Exploration of the Structure–Function Relationships of a Novel Frog Skin Secretion-Derived Bioactive Peptide, t-DPH1, through Use of Rational Design, Cationicity Enhancement and In Vitro Studies
Abstract
1. Introduction
2. Results
2.1. Molecular Cloning of cDNA Encoding t-DPH1 Precursor
2.2. Purification and Identification of Synthesised t-DPH1 and Its Analogues
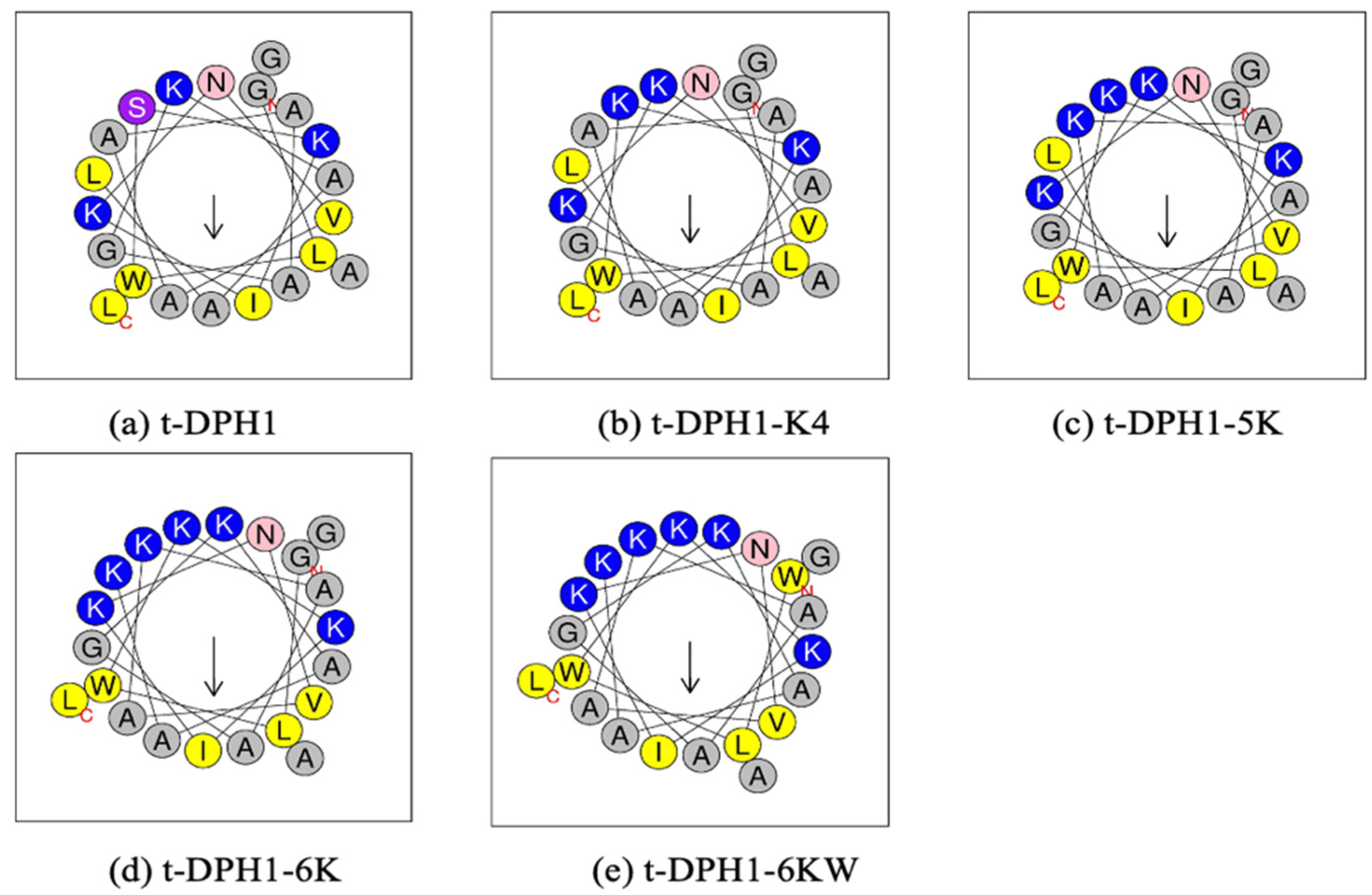
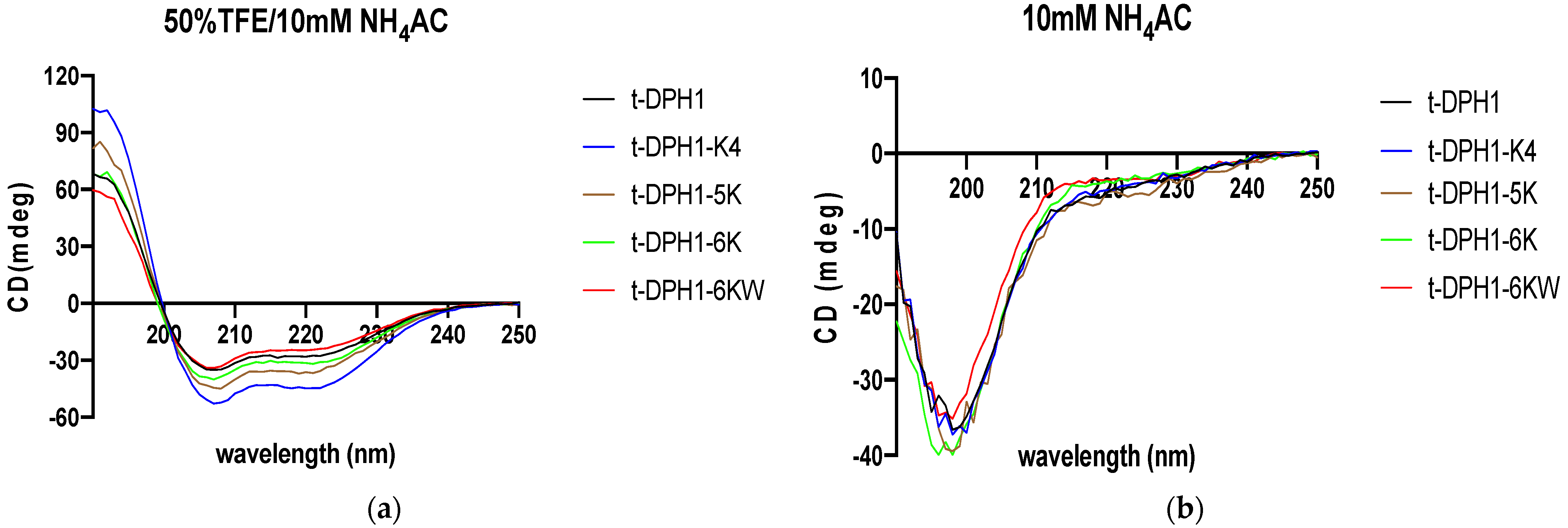
2.3. Peptide Design and Secondary Structure Prediction Analysis of t-DPH1 and Its Analogues
2.4. Antimicrobial Activities of t-DPH1 and Its Analogues
2.5. Anti-Biofilm Activities of t-DPH1 and Its Analogues
2.6. Time–Killing Kinetics of t-DPH1, t-DPH1-K4, t-DPH1-5K and t-DPH1-6K
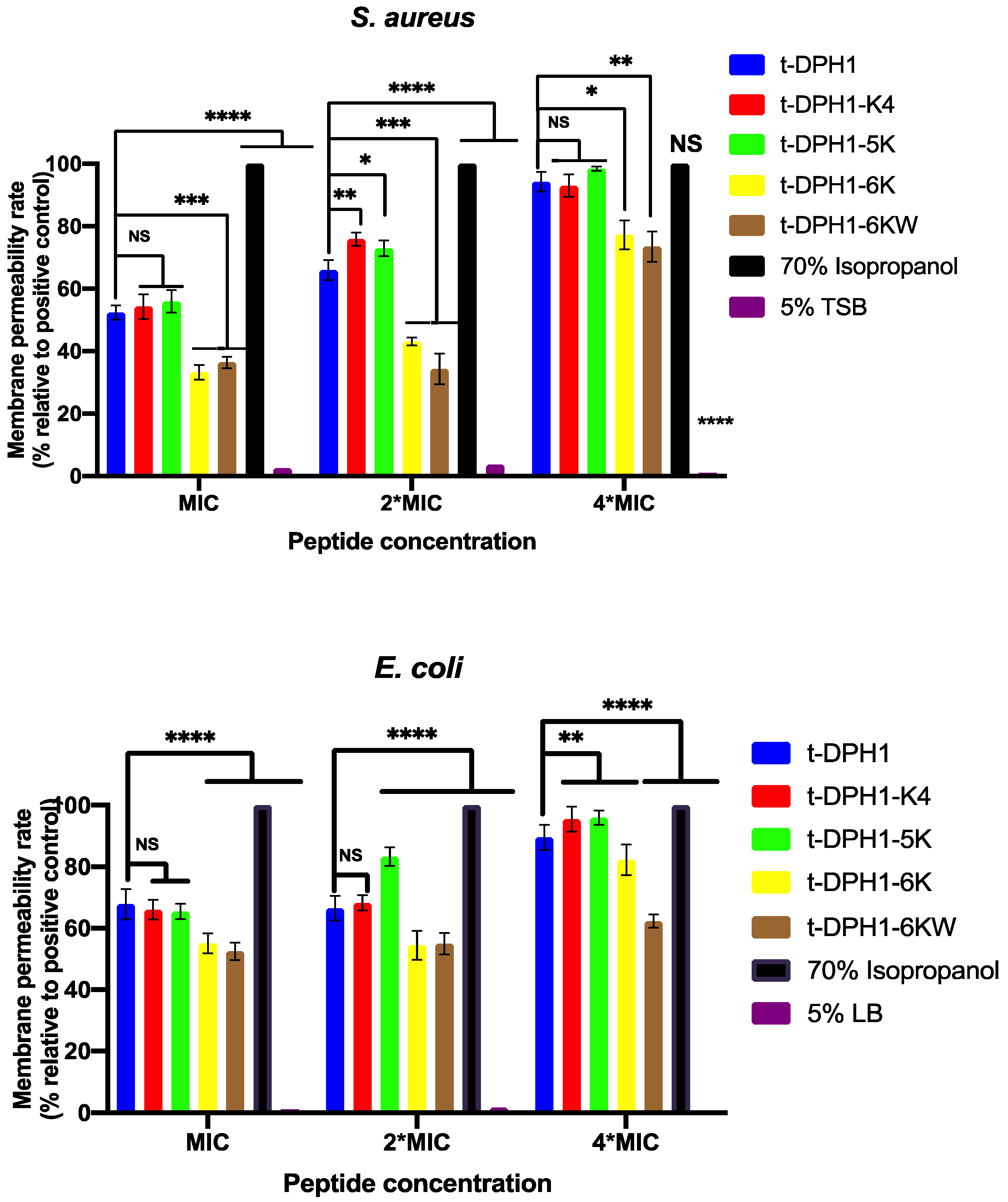
2.7. Permeabilisation Effects of t-DPH1 and Its Analogues on the Bacterial Cell Membrane
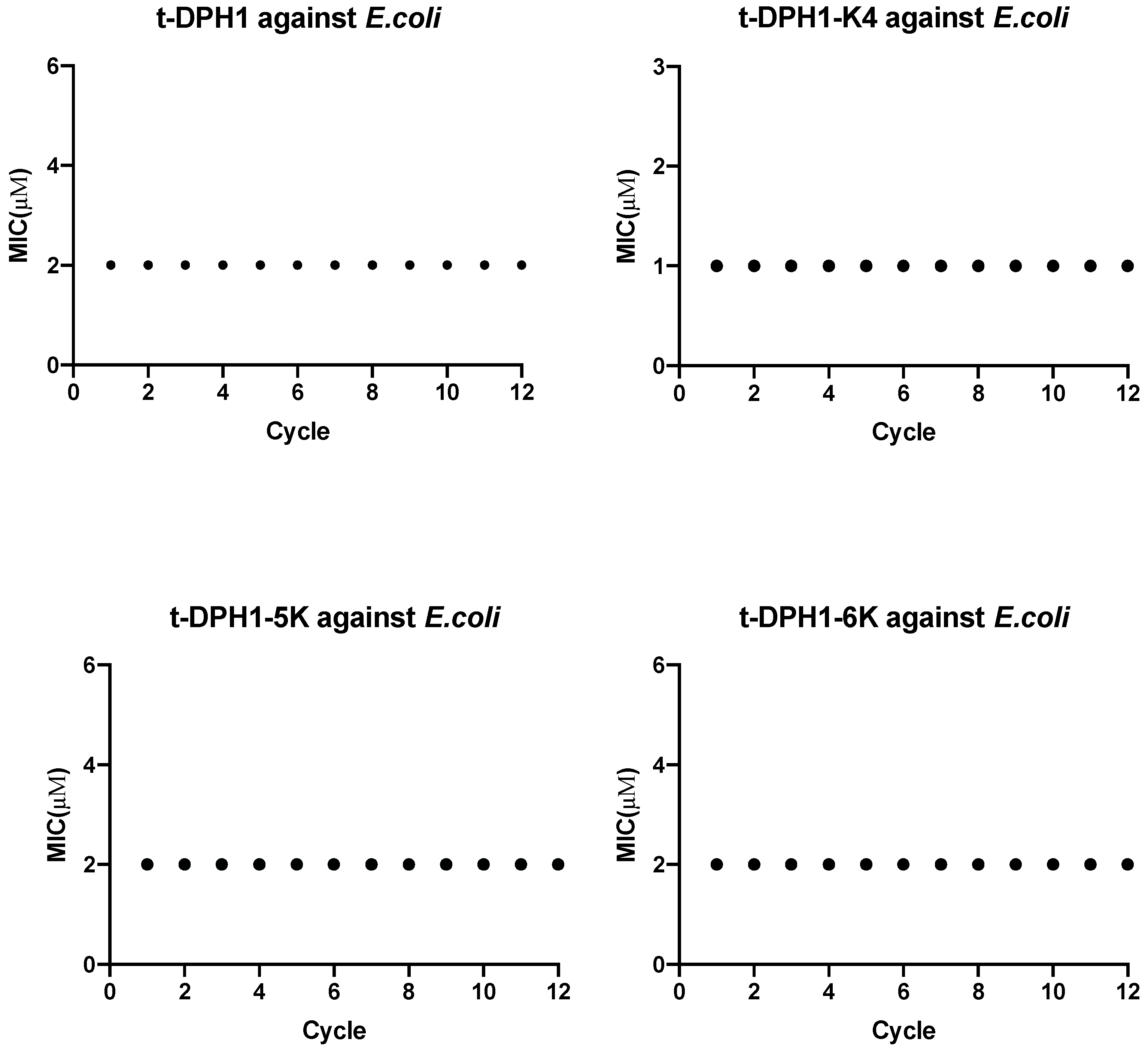
2.8. Assessments of Resistance Induction of t-DPH1 and Its Analogues
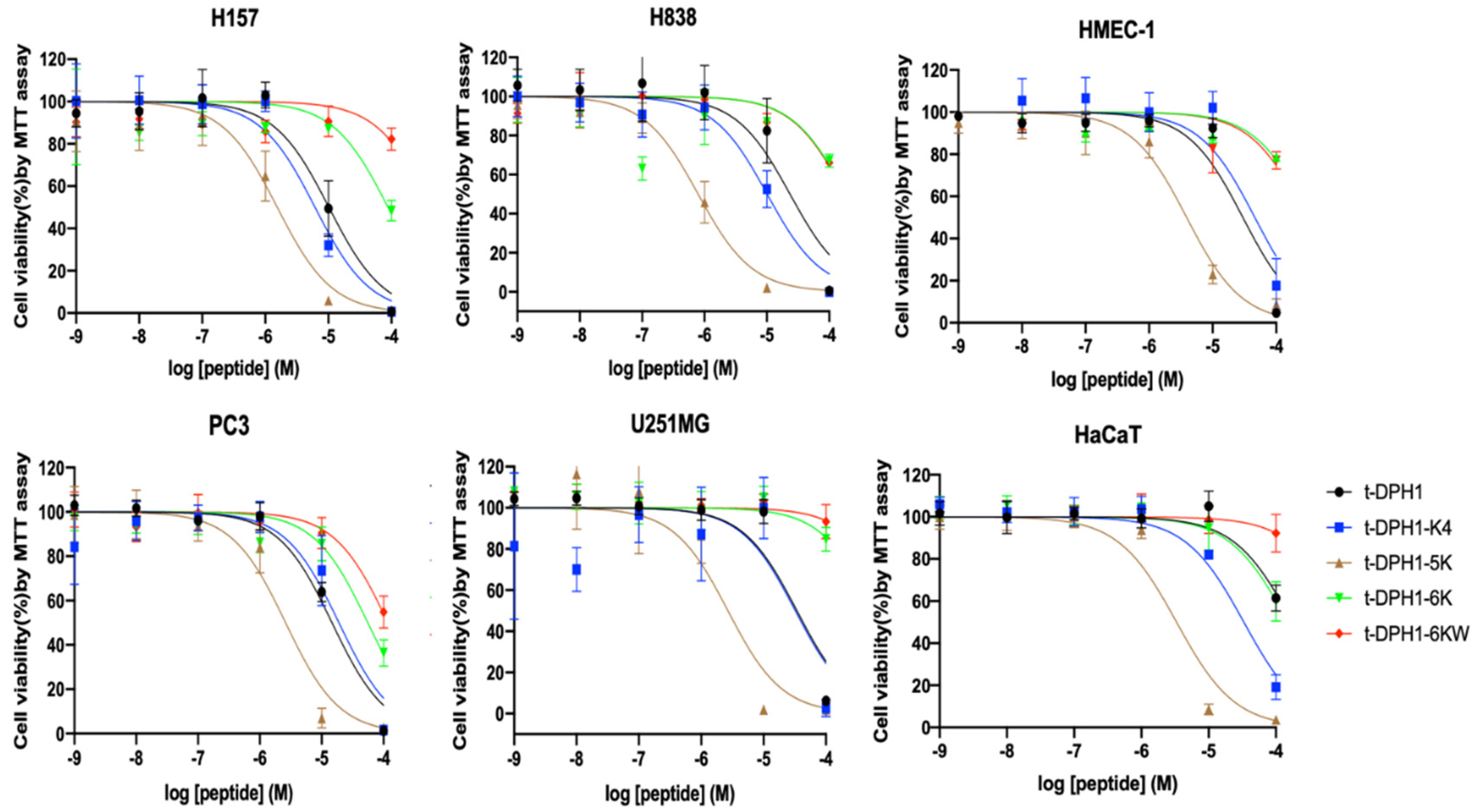
2.9. Anti-Proliferation Activities of t-DPH1 and Its Analogues
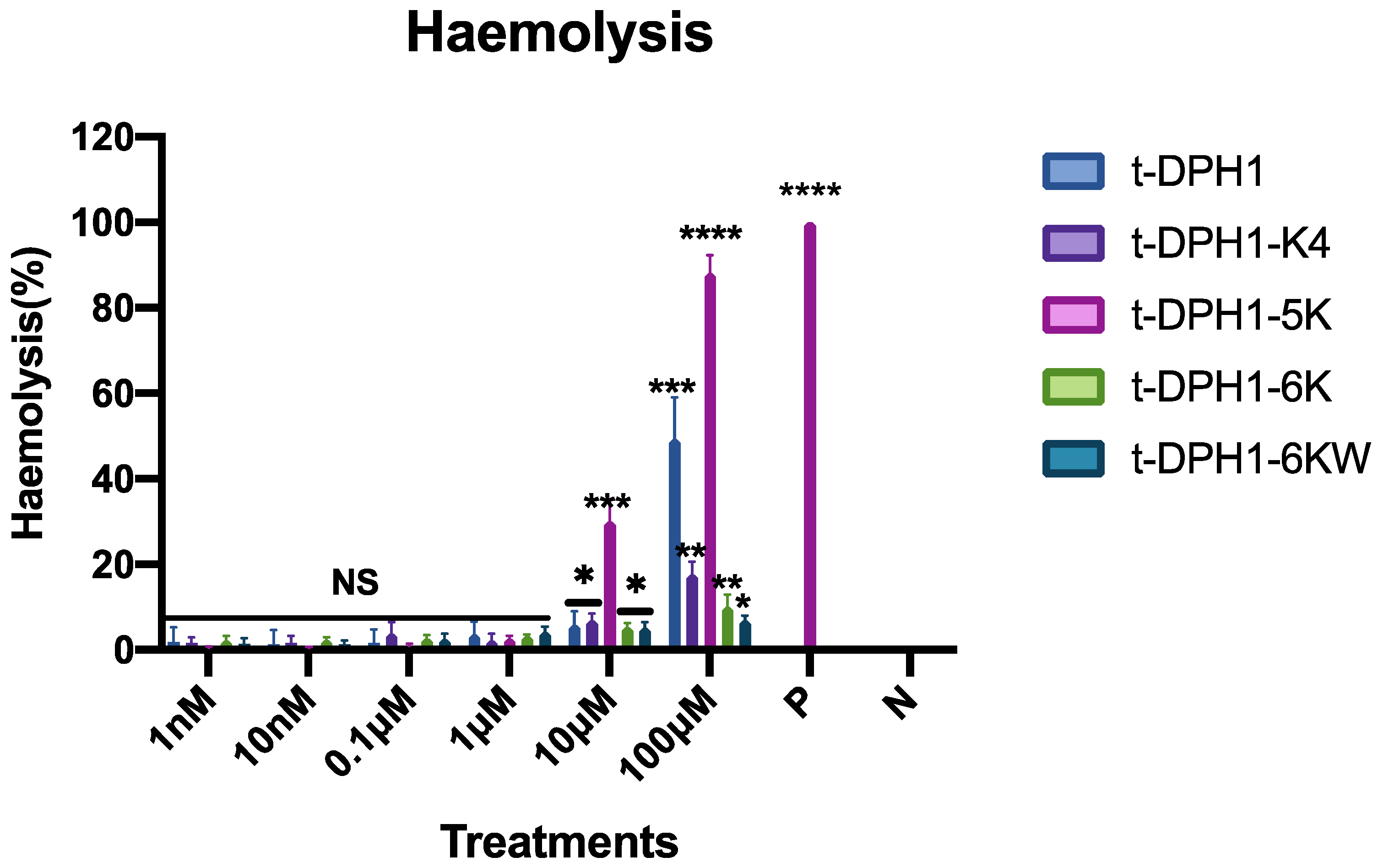
2.10. Haemolysis Activities of t-DPH1 and Its Analogues
3. Discussion
4. Materials and Methods
4.1. Skin Secretion Harvesting from P. hypochondrialis
4.2. Identification of Precursor-Encoding cDNAs from the Skin Secretion
4.3. Peptide Synthesis
4.4. Physicochemical Properties Analyses, Secondary Structure Predictions and Determinations
4.5. MIC and MBC Determinations
4.6. MBIC and MBEC Determinations
4.7. Sytox-Green Bacteria Cell Membrane Permeabilisation
4.8. Time–Killing Kinetics Determination
4.9. Resistance Induction by Serial Passages
4.10. Assessment of Mammalian Cell Proliferation Inhibitory Effect
4.11. Haemolysis Assays
4.12. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Bera, S.; Ghosh, A.; Sharma, S.; Debnath, T.; Giri, B.; Bhunia, A. Probing the role of Proline in the antimicrobial activity and lipopolysaccharide binding of indolicidin. J. Colloid Interface Sci. 2015, 452, 148–159. [Google Scholar] [CrossRef] [PubMed]
- La Manna, S.; Di Natale, C.; Florio, D.; Marasco, D. Peptides as Therapeutic Agents for Inflammatory-Related Diseases. Int. J. Mol. Sci. 2018, 19, 2714. [Google Scholar] [CrossRef] [PubMed]
- Raja, Z.; André, S.; Abbassi, F.; Humblot, V.; Lequin, O.; Bouceba, T.; Correia, I.; Casale, S.; Foulon, T.; Sereno, D.; et al. Insight into the mechanism of action of temporin-SHa, a new broad-spectrum antiparasitic and antibacterial agent. PLoS ONE 2017, 12, e0174024. [Google Scholar] [CrossRef] [PubMed]
- Starr, C.G.; Maderdrut, J.L.; He, J.; Coy, D.H.; Wimley, W.C. Pituitary adenylate cyclase-activating polypeptide is a potent broad-spectrum antimicrobial peptide: Structure-activity relationships. Peptides 2018, 104, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Kumari, T.; Verma, D.P.; Afshan, T.; Verma, N.K.; Pant, G.; Ali, M.; Shukla, P.K.; Mitra, K.; Ghosh, J.K. A Noncytotoxic Temporin L Analogue with In Vivo Antibacterial and Antiendotoxin Activities and a Nonmembrane-Lytic Mode of Action. ACS Infect. Dis. 2020, 6, 2369–2385. [Google Scholar] [CrossRef]
- Thompson, A.H.; Bjourson, A.J.; Orr, D.F.; Shaw, C.; McClean, S. A combined mass spectrometric and cDNA sequencing approach to the isolation and characterization of novel antimicrobial peptides from the skin secretions of Phyllomedusa hypochondrialis azurea. Peptides 2007, 28, 1331–1343. [Google Scholar] [CrossRef] [PubMed]
- Mookherjee, N.; Anderson, M.A.; Haagsman, H.P.; Davidson, D.J. Antimicrobial host defence peptides: Functions and clinical potential. Nat. Rev. Drug Discov. 2020, 19, 311–332. [Google Scholar] [CrossRef]
- Lei, J.; Sun, L.; Huang, S.; Zhu, C.; Li, P.; He, J.; Mackey, V.; Coy, D.H.; He, Q. The antimicrobial peptides and their potential clinical applications. Am. J. Trans. Res. 2019, 11, 3919–3931. [Google Scholar]
- Huang, Y.; Huang, J.; Chen, Y. Alpha-helical cationic antimicrobial peptides: Relationships of structure and function. Protein Cell 2010, 1, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Kumar, T.V.; Sanil, G.; Sanil, T.V.K.A.G. A Review of the Mechanism of Action of Amphibian Antimicrobial Peptides Focusing on Peptide-Membrane Interaction and Membrane Curvature. Curr. Protein Pept. Sci. 2017, 18, 1263–1272. [Google Scholar] [CrossRef]
- Mwangi, J.; Hao, X.; Lai, R.; Zhang, Z.-Y. Antimicrobial peptides: New hope in the war against multidrug resistance. Zool. Res. 2019, 40, 488. [Google Scholar] [CrossRef]
- Khara, J.S.; Lim, F.K.; Wang, Y.; Ke, X.-Y.; Voo, Z.X.; Yang, Y.Y.; Lakshminarayanan, R.; Ee, P.L.R. Designing α-helical peptides with enhanced synergism and selectivity against Mycobacterium smegmatis: Discerning the role of hydrophobicity and helicity. Acta Biomater. 2015, 28, 99–108. [Google Scholar] [CrossRef]
- Nguyen, L.T.; Haney, E.F.; Vogel, H.J. The expanding scope of antimicrobial peptide structures and their modes of action. Trends Biotechnol. 2011, 29, 464–472. [Google Scholar] [CrossRef]
- Tossi, A.; Sandri, L.; Giangaspero, A. Amphipathic, alpha-helical antimicrobial peptides. Biopolymers 2000, 55, 4–30. [Google Scholar] [CrossRef]
- Du, Q.; Hou, X.; Ge, L.; Li, R.; Zhou, M.; Wang, H.; Wang, L.; Wei, M.; Chen, T.; Shaw, C. Cationicity-Enhanced Analogues of the Antimicrobial Peptides, AcrAP1 and AcrAP2, from the Venom of the Scorpion, Androctonus crassicauda, Display Potent Growth Modulation Effects on Human Cancer Cell Lines. Int. J. Biol. Sci. 2014, 10, 1097–1107. [Google Scholar] [CrossRef]
- Chen, L.; Shen, T.; Liu, Y.; Zhou, J.; Shi, S.; Wang, Y.; Zhao, Z.; Yan, Z.; Liao, C.; Wang, C. Enhancing the antibacterial activity of antimicrobial peptide PMAP-37(F34-R) by cholesterol modification. BMC Vet. Res. 2020, 16, 419. [Google Scholar] [CrossRef] [PubMed]
- Mohanram, H.; Bhattacharjya, S. Resurrecting Inactive Antimicrobial Peptides from the Lipopolysaccharide Trap. Antimicrob. Agents Chemother. 2014, 58, 1987–1996. [Google Scholar] [CrossRef] [PubMed]
- Mohanram, H.; Bhattacharjya, S. ‘Lollipop’-shaped helical structure of a hybrid antimicrobial peptide of temporin B-lipopolysaccharide binding motif and mapping cationic residues in antibacterial activity. Biochim. Biophys. Acta Gen. Subj. 2016, 1860, 1362–1372. [Google Scholar] [CrossRef]
- Sinha, S.; Zheng, L.; Mu, Y.; Ng, W.J.; Bhattacharjya, S. Structure and Interactions of A Host Defense Antimicrobial Peptide Thanatin in Lipopolysaccharide Micelles Reveal Mechanism of Bacterial Cell Agglutination. Sci. Rep. 2017, 7, 17795. [Google Scholar] [CrossRef]
- Huang, L.; Chen, D.; Wang, L.; Lin, C.; Ma, C.; Xi, X.; Chen, T.; Shaw, C.; Zhou, M. Dermaseptin-PH: A Novel Peptide with Antimicrobial and Anticancer Activities from the Skin Secretion of the South American Orange-Legged Leaf Frog, Pithecopus (Phyllomedusa) hypochondrialis. Molecules 2017, 22, 1805. [Google Scholar] [CrossRef] [PubMed]
- Long, Q.; Li, L.; Wang, H.; Li, M.; Wang, L.; Zhou, M.; Su, Q.; Chen, T.; Wu, Y. Novel peptide dermaseptin-PS1 exhibits anticancer activity via induction of intrinsic apoptosis signalling. J. Cell. Mol. Med. 2018, 23, 1300–1312. [Google Scholar] [CrossRef] [PubMed]
- Zairi, A.; Tangy, F.; Bouassida, K.; Hani, K. Dermaseptins and Magainins: Antimicrobial Peptides from Frogs’ Skin—New Sources for a Promising Spermicides Microbicides—A Mini Review. J. Biomed. Biotechnol. 2009, 2009, 452567. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Zhou, X.; Chen, X.; Huang, L.; Xi, X.; Ma, C.; Zhou, M.; Wang, L.; Chen, T. Evaluating the Bioactivity of a Novel Antimicrobial and Anticancer Peptide, Dermaseptin-PS4(Der-PS4), from the Skin Secretion of Phyllomedusa sauvagii. Molecules 2019, 24, 2974. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Feng, Q.; Yan, Q.; Hao, X.; Chen, Y. Alpha-Helical Cationic Anticancer Peptides: A Promising Candidate for Novel Anticancer Drugs. Mini-Rev. Med. Chem. 2015, 15, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Gong, Z.; Pei, X.; Ren, S.; Chen, X.; Wang, L.; Ma, C.; Xi, X.; Chen, T.; Shaw, C.; Zhou, M. Identification and Rational Design of a Novel Antibacterial Peptide Dermaseptin-AC from the Skin Secretion of the Red-Eyed Tree Frog Agalychnis callidryas. Antibiotics 2020, 9, 243. [Google Scholar] [CrossRef] [PubMed]
- Ge, L.; Lyu, P.; Zhou, M.; Zhang, H.; Wan, Y.; Li, B.; Li, R.; Wang, L.; Chen, T.; Shaw, C. AcT-2: A Novel Myotropic and Antimicrobial Type 2 Tryptophyllin from the Skin Secretion of the Central American Red-Eyed Leaf Frog, Agalychnis callidryas. Sci. World J. 2014, 2014, 158546. [Google Scholar] [CrossRef]
- Molchanova, N.; Hansen, P.R.; Franzyk, H. Advances in Development of Antimicrobial Peptidomimetics as Potential Drugs. Molecules 2017, 22, 1430. [Google Scholar] [CrossRef]
- Epand, R.M.; Epand, R.F. Bacterial membrane lipids in the action of antimicrobial agents. J. Pept. Sci. 2010, 17, 298–305. [Google Scholar] [CrossRef]
- Wiradharma, N.; Sng, M.Y.S.; Khan, M.; Ong, Z.-Y.; Yang, Y.-Y. Rationally Designed α-Helical Broad-Spectrum Antimicrobial Peptides with Idealized Facial Amphiphilicity. Macromol. Rapid Commun. 2013, 34, 74–80. [Google Scholar] [CrossRef] [PubMed]
- Tamba, Y.; Yamazaki, M. Single Giant Unilamellar Vesicle Method Reveals Effect of Antimicrobial Peptide Magainin 2 on Membrane Permeability. Biochemistry 2005, 44, 15823–15833. [Google Scholar] [CrossRef]
- Ladram, A.; Nicolas, P. Antimicrobial peptides from frog skin biodiversity and therapeutic promises. Front. Biosci. 2016, 21, 1341–1371. [Google Scholar] [CrossRef]
- Powers, J.P.; Hancock, R.E. The relationship between peptide structure and antibacterial activity. Peptides 2003, 24, 1681–1691. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.-K.; Shih, L.-Y.; Chang, K.Y. Large-Scale Analysis of Antimicrobial Activities in Relation to Amphipathicity and Charge Reveals Novel Characterization of Antimicrobial Peptides. Molecules 2017, 22, 2037. [Google Scholar] [CrossRef] [PubMed]
- Yasir, M.; Willcox, M.D.P.; Dutta, D. Action of Antimicrobial Peptides against Bacterial Biofilms. Materials 2018, 11, 2468. [Google Scholar] [CrossRef]
- Kim, J.; Mosior, M.; Chung, L.; Wu, H.; McLaughlin, S. Binding of peptides with basic residues to membranes containing acidic phospholipids. Biophys. J. 1991, 60, 135–148. [Google Scholar] [CrossRef]
- Bechinger, B.; Gorr, S.-U. Antimicrobial Peptides: Mechanisms of Action and Resistance. J. Dent. Res. 2016, 96, 254–260. [Google Scholar] [CrossRef] [PubMed]
- Gaspar, D.; Veiga, A.S.; Castanho, M.A. From antimicrobial to anticancer peptides. A review. Front. Microbiol. 2013, 4, 294. [Google Scholar] [CrossRef]
- Torres, M.D.T.; Sothiselvam, S.; Lu, T.K.; De La Fuente-Nunez, C. Peptide Design Principles for Antimicrobial Applications. J. Mol. Biol. 2019, 431, 3547–3567. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Du, Q.; Ma, C.; Xi, X.; Wang, L.; Zhou, M.; Burrows, J.F.; Chen, T.; Wang, H. Structure–activity relationship of an antimicrobial peptide, Phylloseptin-PHa: Balance of hydrophobicity and charge determines the selectivity of bioactivities. Drug Des. Dev. Ther. 2019, 13, 447–458. [Google Scholar] [CrossRef] [PubMed]
- Mishra, B.; Lakshmaiah Narayana, J.; Lushnikova, T.; Wang, X.; Wang, G. Low cationicity is important for systemic in vivo efficacy of database-derived peptides against drug-resistant Gram-positive pathogens. Proc. Natl. Acad. Sci. USA 2019, 116, 13517–13522. [Google Scholar] [CrossRef]
- Liang, D.; Li, H.; Xu, X.; Liang, J.; Dai, X.; Zhao, W. Design, screening and antimicrobial activity of novel peptides against Streptococcus mutans. J. South. Med. Univ. 2019, 39, 823–829. [Google Scholar] [CrossRef]
- Milano, A.; Sulejmani, A.; Intra, J.; Sala, M.R.; Leoni, V.; Carcione, D. Antimicrobial Resistance Trends of Escherichia coli Isolates from Outpatient and Inpatient Urinary Infections over a 20-Year Period. Microb. Drug Resist. 2021. Online ahead of print. [Google Scholar] [CrossRef]
- Sanders, C.C.; Sanders, W.E. beta-Lactam Resistance in Gram-Negative Bacteria: Global Trends and Clinical Impact. Clin. Infect. Dis. 1992, 15, 824–839. [Google Scholar] [CrossRef]
- Schoutens, E.; Yourassowsky, E. Speed of Bactericidal Action of Penicillin G, Ampicillin, and Carbenicillin on Bacteroides fragilis. Antimicrob. Agents Chemother. 1974, 6, 227–231. [Google Scholar] [CrossRef][Green Version]
- Kondejewski, L.H.; Jelokhani-Niaraki, M.; Farmer, S.W.; Lix, B.; Kay, C.M.; Sykes, B.D.; Hancock, R.; Hodges, R.S. Dissociation of Antimicrobial and Hemolytic Activities in Cyclic Peptide Diastereomers by Systematic Alterations in Amphipathicity. J. Biol. Chem. 1999, 274, 13181–13192. [Google Scholar] [CrossRef]
- Liscano, Y.; Oñate-Garzón, J.; Delgado, J.P. Peptides with Dual Antimicrobial–Anticancer Activity: Strategies to Overcome Peptide Limitations and Rational Design of Anticancer Peptides. Molecules 2020, 25, 4245. [Google Scholar] [CrossRef]
- Cutrona, K.; Kaufman, B.A.; Figueroa, D.; Elmore, D.E. Role of arginine and lysine in the antimicrobial mechanism of histone-derived antimicrobial peptides. FEBS Lett. 2015, 589, 3915–3920. [Google Scholar] [CrossRef]
- Luong, H.X.; Kim, D.-H.; Lee, B.-J.; Kim, Y.-W. Effects of lysine-to-arginine substitution on antimicrobial activity of cationic stapled heptapeptides. Arch. Pharmacal Res. 2018, 41, 1092–1097. [Google Scholar] [CrossRef]
- Rice, A.; Wereszczynski, J. Probing the disparate effects of arginine and lysine residues on antimicrobial peptide/bilayer association. Biochim. Biophys. Acta Biomembr. 2017, 1859, 1941–1950. [Google Scholar] [CrossRef]
- Tyler, M.J.; Stone, D.J.; Bowie, J.H. A novel method for the release and collection of dermal, glandular secretions from the skin of frogs. J. Pharmacol. Toxicol. Methods 1992, 28, 199–200. [Google Scholar] [CrossRef]
- Gautier, R.; Douguet, D.; Antonny, B.; Drin, G. HELIQUEST: A web server to screen sequences with specific-helical properties. Bioinformatics 2008, 24, 2101–2102. [Google Scholar] [CrossRef]
- Andrews, J.M. Determination of minimum inhibitory concentrations. J. Antimicrob. Chemother. 2001, 48, 5–16. [Google Scholar] [CrossRef]
- Dolzani, L.; Milan, A.; Scocchi, M.; Lagatolla, C.; Bressan, R.; Benincasa, M. Sub-MIC effects of a proline-rich antibacterial peptide on clinical isolates of Acinetobacter baumannii. J. Med. Microbiol. 2019, 68, 1253–1265. [Google Scholar] [CrossRef]

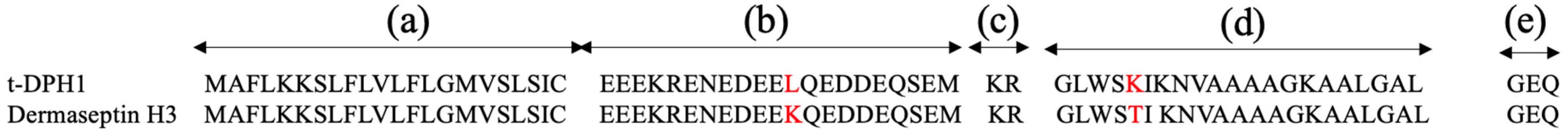

| Peptide Name | Peptide Sequence | Net Charge (z) | Hydrophobicity (H) | Hydrophobic Moment (μH) |
|---|---|---|---|---|
| t-DPH1 | GLWSKIKNVAAAAGKAALGAL-NH2 | 4 | 0.425 | 0.337 |
| t-DPH1-K4 | GLWKKIKNVAAAAGKAALGAL-NH2 | 5 | 0.380 | 0.414 |
| t-DPH1-5K | GLWKKIKNVAKAAGKAALGAL-NH2 | 6 | 0.318 | 0.452 |
| t-DPH1-6K | GLWKKIKNVAKAAGKAAKGAL-NH2 | 7 | 0.190 | 0.513 |
| t-DPH1-6KW | WLWKKIKNVAKAAGKAAKGAL-NH2 | 7 | 0.297 | 0.443 |
| Peptide Name | % of α-Helix in 50% TFE |
|---|---|
| t-DPH1 | 53.64 |
| t-DPH1-K4 | 69.33 |
| t-DPH1-5K | 63.02 |
| t-DPH1-6K | 60.38 |
| t-DPH1-6KW | 54.42 |
| t-DPH1 | t-DPH1-K4 | t-DPH1-5K | t-DPH1-6K | t-DPH1-6KW | |
|---|---|---|---|---|---|
| S. aureus (ATCC 6538) | 8/16 | 2/4 | 2/2 | 4/8 | 128/128 |
| Methicillin-resistant Staphylococcus aureus (MRSA) (NCTC 12493) | 16/32 | 2/4 | 2/4 | 32/64 | 128/256 |
| Enterococcus faecalis (E. faecalis) (NCTC 12697) | 128/256 | 32/32 | 8/16 | 256/512 | 512/>512 |
| E. coli (ATCC 8739) | 2/4 | 1/2 | 2/4 | 2/4 | 2/4 |
| K. pneumoniae (ATCC 43816) | 8/16 | 2/4 | 2/4 | 4/8 | 64/64 |
| P. aeruginosa (ATCC 9027) | 16/32 | 8/16 | 4/8 | 8/16 | 64/128 |
| C. albicans (ATCC 10231) | 64/128 | 32/64 | 256/512 | 64/128 | 512/>512 |
| t-DPH1 | t-DPH1-K4 | t-DPH1-5K | t-DPH1-6K | t-DPH1-6KW | |
|---|---|---|---|---|---|
| S. aureus (ATCC 6538) | 16/>256 | 8/256 | 4/>256 | 16/>256 | 128/>256 |
| MRSA (NCTC 12493) | 32/>256 | 8/>256 | 4/>256 | 64/>256 | >256/>256 |
| E. faecalis (NCTC 12697) | 256/>256 | 64/>256 | 16/>256 | >256/>256 | >256/>256 |
| E. coli (ATCC 8739) | 8/256 | 2/128 | 4/64 | 2/256 | 4/256 |
| K. pneumoniae (ATCC 43816) | 16/256 | 4/128 | 4/64 | 8/256 | 64/>256 |
| P. aeruginosa (ATCC 9027) | 32/>256 | 16/256 | 8/128 | 16/>256 | 128/>256 |
| PC-3 | H838 | H157 | U251MG | HMEC-1 | HaCaT | |
|---|---|---|---|---|---|---|
| t-DPH1 | 14.67 | 23.51 | 10.20 | 34.25 | 29.85 | 175.6 |
| t-DPH1-K4 | 18.99 | 10.11 | 6.143 | 32.18 | 46.02 | 63.37 |
| t-DPH1-5K | 2.605 | 1.796 | 1.546 | 2.679 | 3.897 | 3.332 |
| t-DPH1-6K | 55.65 | 186.1 | 88.28 | 593.7 | 340.2 | 151.4 |
| t-DPH1-6KW | 118.4 | 180.8 | 427.3 | 1499 | 293.2 | 119.3 |
| Peptides | HC50 (μM) |
|---|---|
| t-DPH1 | 106.4 |
| t-DPH1-K4 | 444.8 |
| t-DPH1-5K | 19.39 |
| t-DPH1-6K | 834.6 |
| t-DPH1-6KW | 2094 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qin, H.; Fang, H.; Chen, X.; Wang, L.; Ma, C.; Xi, X.; Chen, T.; Shaw, C.; Zhou, M. Exploration of the Structure–Function Relationships of a Novel Frog Skin Secretion-Derived Bioactive Peptide, t-DPH1, through Use of Rational Design, Cationicity Enhancement and In Vitro Studies. Antibiotics 2021, 10, 1529. https://doi.org/10.3390/antibiotics10121529
Qin H, Fang H, Chen X, Wang L, Ma C, Xi X, Chen T, Shaw C, Zhou M. Exploration of the Structure–Function Relationships of a Novel Frog Skin Secretion-Derived Bioactive Peptide, t-DPH1, through Use of Rational Design, Cationicity Enhancement and In Vitro Studies. Antibiotics. 2021; 10(12):1529. https://doi.org/10.3390/antibiotics10121529
Chicago/Turabian StyleQin, Haixin, Hantian Fang, Xiaoling Chen, Lei Wang, Chengbang Ma, Xinping Xi, Tianbao Chen, Chris Shaw, and Mei Zhou. 2021. "Exploration of the Structure–Function Relationships of a Novel Frog Skin Secretion-Derived Bioactive Peptide, t-DPH1, through Use of Rational Design, Cationicity Enhancement and In Vitro Studies" Antibiotics 10, no. 12: 1529. https://doi.org/10.3390/antibiotics10121529
APA StyleQin, H., Fang, H., Chen, X., Wang, L., Ma, C., Xi, X., Chen, T., Shaw, C., & Zhou, M. (2021). Exploration of the Structure–Function Relationships of a Novel Frog Skin Secretion-Derived Bioactive Peptide, t-DPH1, through Use of Rational Design, Cationicity Enhancement and In Vitro Studies. Antibiotics, 10(12), 1529. https://doi.org/10.3390/antibiotics10121529







