Point Prevalence Survey of Antimicrobial Utilization in Ghana’s Premier Hospital: Implications for Antimicrobial Stewardship
Abstract
:1. Introduction
2. Results
2.1. Antimicrobial Prevalence
2.2. Patient Demographics
2.3. Treatment Indications
2.3.1. Community-Acquired versus Healthcare Associated Infections
2.3.2. Prophylactic Antimicrobials
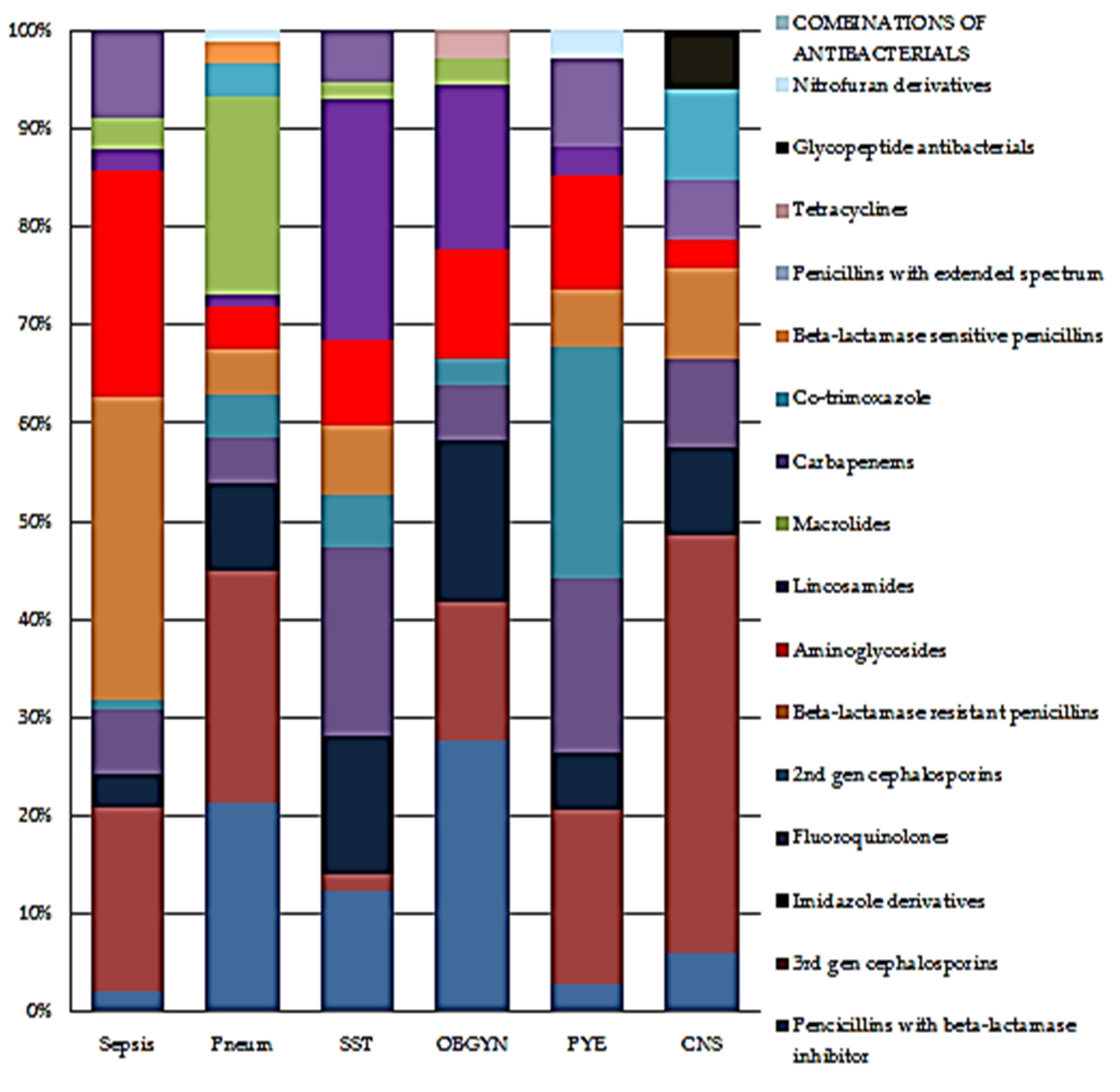
2.3.3. Most Common Diagnoses Treated with Therapeutic Antimicrobials
2.4. Antibiotic Quality Indicators by Activity
2.5. Antibacterial Resistance
2.6. Antimicrobial Class
2.6.1. Antibacterials for Systemic Use (Therapeutic and Prophylactic)
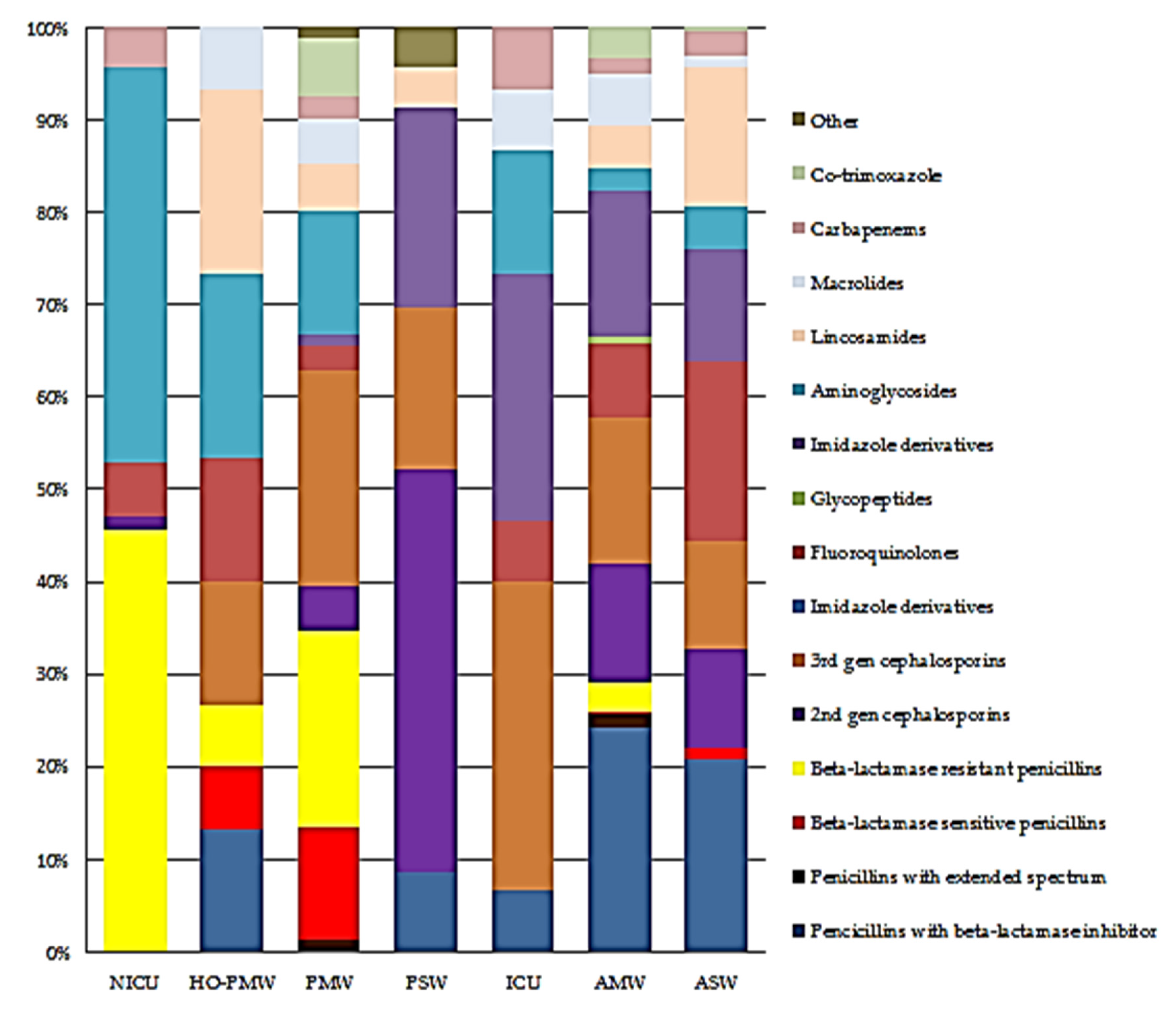
2.6.2. Proportional Antibiotic Use by Class on Ward
3. Discussion
3.1. Antimicrobial Prevalence
3.2. Most Common Antimicrobials Prescribed
3.3. Most Common Diagnosis for Antimicrobial Use
3.4. Targeted versus Empiric Treatment
3.5. Quality Indicators for Prescribing
3.6. Limitations
4. Materials and Methods
4.1. Study Design
4.1.1. Study Site
4.1.2. Pre-Survey
4.1.3. Study Population
4.1.4. Sampling Method
4.1.5. Inclusion Criteria
4.1.6. Exclusion Criteria
4.2. Data Collection
4.3. Data Entry
4.4. Data Analysis
4.5. Ethical Issues
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gelband, H.; Molly Miller, P.; Pant, S.; Gandra, S.; Levinson, J.; Barter, D.; White, A.; Laxminarayan, R. The State of the World’s Antibiotics 2015; Center for Disease Dynamics, Economics and Policy: Washington, DC, USA, 2015. [Google Scholar]
- O’Neill, J. Review on Antimicrobial Resistance: Tackling Drug-Resistant Infections Globally: Final Report and Recommendations; Wellcome Trust: London, UK, 2016. [Google Scholar]
- European Centre for Disease Prevention and Control, European Medicines Agency. The Bacterial Challenge: Time to React. A Call to Narrow the Gap between Multidrug-Resistant Bacteria in the EU and the Development of New Antibacterial Agents. Available online: https://www.ema.europa.eu/en/news/bacterial-challenge-time-react-call-narrow-gap-between-multidrug-resistant-bacteria-eu-development (accessed on 1 December 2021).
- da Costa, M.E.; Machado, H.S. Evolution of antimicrobial resistance in Europe: A factual review. J. Allergy Ther. 2017, 8, 2. [Google Scholar]
- World Health Organization. Antimicrobial Resistance Global Report on Surveillance: 2014 Summary. Available online: https://apps.who.int/iris/bitstream/handle/10665/112647/WHO_HSE_PED_AIP_?sequence=1 (accessed on 1 December 2021).
- Le Doare, K.; Bielicki, J.; Heath, P.T.; Sharland, M. Systematic review of antibiotic resistance rates among gram-negative bacteria in children with sepsis in resource-limited countries. J. Pediatric Infect. Dis. Soc. 2015, 4, 11–20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leopold, S.J.; van Leth, F.; Tarekegn, H.; Schultsz, C. Antimicrobial drug resistance among clinically relevant bacterial isolates in sub-Saharan Africa: A systematic review. J. Antimicrob. Chemother. 2014, 69, 2337–2353. [Google Scholar] [CrossRef] [Green Version]
- GARP–Tanzania National Working Group. Situation Analysis and Recommendations: Antibiotic Resistance in Tanzania; Dar es Salaam, Tanzania, 2015. Available online: Https://cddep.org/publications/garp_tanzania_situation_analysis/ (accessed on 3 March 2021).
- Vandepitte, J.; Hughes, P.; Matovu, G.; Bukenya, J.; Grosskurth, H.; Lewis, D.A. High prevalence of ciprofloxacin-resistant gonorrhea among female sex workers in Kampala, Uganda (2008–2009). Sex. Transm. Dis. 2014, 41, 233–237. [Google Scholar] [CrossRef]
- Kariuki, S.; Gordon, M.A.; Feasey, N.; Parry, C.M. Antimicrobial resistance and management of invasive Salmonella disease. Vaccine 2015, 33, C21–C29. [Google Scholar] [CrossRef] [Green Version]
- Kariuki, S.; Gichia, M.; Kakai, R. Situation Analysis: Antibiotic Use and Resistance in Kenya; The GARP-Kenya National Working Group; Kenya Medical Research Institute: Nairobi, Kenya, 2011; Available online: https://cddep.org/wp-content/uploads/2017/08/garp-kenya_es.pdf (accessed on 1 December 2021).
- HPSC Annual Epidemiological Reports 2014. European Antimicrobial Resistance Surveillance Network (EARS-Net). EARS-Net Report; Ireland. Available online: Https://www.hpsc.ie/a-z/microbiologyantimicrobialresistance/europeanantimicrobialresistancesurveillancesystemearss/ears-netdataandreports/annualreports/File,15531,en.pdf (accessed on 6 June 2021).
- O’Neill, J. Antimicrobial Resistance: Tackling a Crisis for the Health and Wealth of Nations. 2014. Available online: Https://amr-review.org/sites/default/files/AMR%20Review%20Paper%20-%20Tackling%20a%20crisis%20for%20the%20health%20and%20wealth%20of%20nations_1.pdf. (accessed on 3 March 2021).
- Van Boeckel, T.P.; Gandra, S.; Ashok, A.; Caudron, Q.; Grenfell, B.T.; Levin, S.A.; Laxminarayan, R. Global antibiotic consumption 2000 to 2010: An analysis of national pharmaceutical sales data. Lancet Infect. Dis. 2014, 14, 742–750. [Google Scholar] [CrossRef]
- Onwueme, K.; Fadairo, Y.; Idoko, L.; Onuh, J.; Alao, O.; Agaba, P.; Lawson, L.; Ukomadu, C.; Idoko, J. High prevalence of toxinogenic Clostridium difficile in Nigerian adult HIV patients. Trans. R. Soc. Trop. Med. Hyg. 2011, 105, 667–669. [Google Scholar] [CrossRef]
- Versporten, A.; Zarb, P.; Caniaux, I.; Gros, M.F.; Drapier, N.; Miller, M.; Jarlier, V.; Nathwani, D.; Goossens, H.; Koraqi, A.; et al. Antimicrobial consumption and resistance in adult hospital inpatients in 53 countries: Results of an internet-based global point prevalence survey. Lancet Glob. Health 2018, 6, e619–e629. [Google Scholar] [CrossRef] [Green Version]
- Newman, M.J.; Frimpong, E.; Donkor, E.S.; Opintan, J.A.; Asamoah-Adu, A. Resistance to antimicrobial drugs in Ghana. Infect. Drug Resist. 2011, 4, 215. [Google Scholar]
- Nweneka, C.V.; Tapha-Sosseh, N.; Sosa, A. Curbing the menace of antimicrobial resistance in developing countries. Harm Reduct. J. 2009, 6, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Harbarth, S.; Balkhy, H.H.; Goossens, H.; Jarlier, V.; Kluytmans, J.; Laxminarayan, R.; Saam, M.; Van Belkum, A.; Pittet, D. Antimicrobial Resistance: One World, One Fight! Antimicrob. Resist. Infect. Control. 2015, 4. [Google Scholar] [CrossRef] [Green Version]
- World Health Organization. Global Antimicrobial Resistance Surveillance System (GLASS) Report: Early Implementation 2016–2017. Available online: https://apps.who.int/iris/bitstream/handle/10665/259744/9789241513449-eng.pdf (accessed on 1 December 2021).
- Barlam, T.F.; Cosgrove, S.E.; Abbo, L.M.; MacDougall, C.; Schuetz, A.N.; Septimus, E.J.; Srinivasan, A.; Dellit, T.H.; Falck-Ytter, Y.T.; Fishman, N.O.; et al. Implementing an antibiotic stewardship program: Guidelines by the Infectious Diseases Society of America and the Society for Healthcare Epidemiology of America. Clin. Infect. Dis. 2016, 62, e51–e77. [Google Scholar] [CrossRef]
- Davey, P.; Brown, E.; Charani, E.; Fenelon, L.; Gould, I.M.; Holmes, A.; Ramsay, C.R.; Wiffen, P.J.; Wilcox, M. Interventions to improve antibiotic prescribing practices for hospital inpatients. Cochrane Database Syst. Rev. 2013, 4, eCD003543. [Google Scholar] [CrossRef]
- Schuts, E.C.; Hulscher, M.E.; Mouton, J.W.; Verduin, C.M.; Stuart, J.W.; Overdiek, H.W.; van der Linden, P.D.; Natsch, S.; Hertogh, C.M.; Wolfs, T.F.; et al. Current evidence on hospital antimicrobial stewardship objectives: A systematic review and meta-analysis. Lancet Infect. Dis. 2016, 16, 847–856. [Google Scholar] [CrossRef]
- ECDC. Point Prevalence Survey of Healthcare-Associated Infections and Antimicrobial Use in European Acute Care Hospitals; European Centre for Disease Prevention and Control: Stockholm, Sweden, 2013; Available online: https://ecdc.europa.eu/en/ healthcare-associated-infections-acute-care-hospitals (accessed on 12 November 2018).
- Labi, A.-K.; Obeng-Nkrumah, N.; Nartey, E.T.; Bjerrum, S.; Adu-Aryee, N.A.; Ofori-Adjei, Y.A.; Yawson, A.E.; Newman, M.J. Antibiotic use in a tertiary healthcare facility in Ghana: A point prevalence survey. Antimicrob. Resist. Infect. Control 2018, 7, 15. [Google Scholar] [CrossRef]
- Newman, M.J. Nosocomial and community acquired infections in Korle Bu teaching hospital, Accra. West Afr. J. Med. 2009, 28, 300–303. [Google Scholar] [CrossRef] [Green Version]
- ECTMIH 2017 (Poster N 5P95): Global Point Prevalence Survey on Antimicrobial Use and Resistance (Global-PPS): Implications for Antibiotic Stewardship Programme for Komfo Anokye Teaching Hospital in Ghana. Available online: https://www.globalpps.com/wp-content/uploads/ECTMIH-2017-5P95-Ghana.pdf (accessed on 11 August 2021).
- Dodoo, C.C.; Orman, E.; Alalbila, T.; Mensah, A.; Jato, J.; Mfoafo, K.A.; Folitse, I.; Hutton-Nyameaye, A.; Okon Ben, I.; Mensah-Kane, P.; et al. Antimicrobial Prescription Pattern in Ho Teaching Hospital, Ghana: Seasonal Determination Using a Point Prevalence Survey. Antibiotics 2021, 10, 199. [Google Scholar] [CrossRef]
- Afriyie, D.K.; Sefah, I.A.; Sneddon, J.; Malcolm, W.; McKinney, R.; Cooper, L.; Kurdi, A.; Godman, B.; Seaton, R.A. Antimicrobial point prevalence surveys in two Ghanaian hospitals: Opportunities for antimicrobial stewardship. Jac Antimicrob. Resist. 2020, 2, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Iosifidis, E.; Antachopoulos, C.; Tsivitanidou, M.; Katragkou, A.; Farmaki, E.; Tsiakou, M.; Kyriazi, T.; Sofianou, D.; Roilides, E. Differential correlation between rates of antimicrobial drug consumption and prevalence of antimicrobial resistance in a tertiary Care Hospital in Greece. Infect. Control Hosp. Epidemiol. 2008, 29, 615–622. [Google Scholar] [CrossRef]
- Vandael, E.; Latour, K.; Goossens, H.; Magerman, K.; Drapier, N.; Catry, B.; Versporten, A.; The Belgian Point Prevalence Survey Study Group. Point prevalence survey of antimicrobial use and healthcare associated infections in Belgian acute care hospitals: Results of the Global–PPS and ECDC-PPS, 2017. Antimicrob. Resist. Infect. Control 2020, 9, 13. [Google Scholar] [CrossRef] [Green Version]
- Momanyi, L.; Opanga, S.; Nyamu, D.; Oluka, M.; Kurdi, A.; Godman, B. Antibiotic prescribing pattern at a leading referral hospital in Kenya: A Point Prevalence Survey. J. Res. Pharm Pr. 2019, 8, 149–154. [Google Scholar] [CrossRef]
- Laxminarayan, R.; Heymann, D.L. Challenges of drug resistance in the developing world. BMJ 2012, 344, e1567. [Google Scholar] [CrossRef] [Green Version]
- Goossens, H.; Ferech, M.; Vander Stichele, R.; Elseviers, M.; The ESAC Project Group. Outpatient antibiotic use in Europe and association with resistance: A cross-national database study. Lancet 2005, 365, 579–582. [Google Scholar] [CrossRef]
- Megraud, F.; Coenen, S.; Versporten, A.; Kist, M.; Lopez-Brea, M.; Hirschl, A.M.; Andersen, L.P.; Grossens, H.; Glupczynski, Y. Helicobacter pylori resistance to antibiotics in Europe and it’s relationship to antibiotic consumption. Gut 2013, 62, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Shorr, A.F. Epidermiology of Staphylococcal resistance. Clin. Infect. Dis. 2007, 45, S171–S176. [Google Scholar] [CrossRef] [Green Version]
- Moran, G.J.; Krishnadasan, A.; Gorwitz, R.J.; Forscheim, G.E.; McDougal, L.K.; Carey, R.B.; Talan, D.A. Methicillin resistant S. Aureus infections among patients in the emergency department. N. Eng. J. Med. 2006, 355, 66674. [Google Scholar] [CrossRef]
- Opinta, J.A.; Newman, M.J.; Arhin, R.E.; Donkor, E.S.; Gyansa-Lutterodt, M.; Mills-Pappoe, W. Laboratory-based nationwide surveillance of antimicrobial resistance in Ghana. Infect. Drug Resist. 2015, 8, 379. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anand Paramadhas, B.D.; Tiroyakgosi, C.; Mpinda-Joseph, P.; Morokotso, M.; Matome, M.; Sinkala, F.; Gaolebe, M.; Malone, B.; Molosiwa, E.; Shanmugam, M.G.; et al. Point Prevalence study of antimicrobial use among hospitals across Botswana; findings and implications. Expert Rev. Anti-Infect. 2019, 17, 535–546. [Google Scholar] [CrossRef]
- Talaat, M.; Saied, T.; Kandeel, A.; El-Ata, G.A.A.; El-Kholy, A.; Hafez, S.; Osman, A.; Razik, M.A.; Ismail, G.; El-Masry, S.; et al. A point prevalence survey of antibiotic use in 18 hospitals in Egypt. Antibiotics 2014, 3, 450–460. [Google Scholar] [CrossRef]
- Lefebvre, M.A.; Versporten, A.; Carrier, M.; Chang, S.; Comeau, J.L.; Emond, Y.; Frenette, C.; Khan, S.; Landry, D.L.; MacLaggab, T.D.; et al. The 2018 Global Point Prevalence Survey of Antimicrobial Consumption and Resistance: Pediatric Results from 26 Canadian Hospitals. Open Forum Infect. 2019, 9, 1–9. [Google Scholar]
- Porto, A.P.; Goossens, H.; Versporten, A.; Costa, S.F. Brazilian Global-PPS Working Group. Global point prevalence of antimicrobial consumption in Brazilian hospitals. J. Hosp. Infect. 2020, 1014, 165–171. [Google Scholar] [CrossRef]
- Zivanovic, V.; Gojkovic-Bukarica, L.; Scepanovic, R.; Vitorovic, T.; Novakovic, R.; Milanov, N.; Bukumiric, Z.; Carevic, B.; Trajkovic, J.; Rajkovic, J.; et al. Differences in antimicrobial consumption, prescribing and isolation rate of multidrug resistant Klebsiella pneumonia, Pseudomonas aeruginosa and Acinetobacter baumannii on surgical and medical wards. PLoS ONE 2017, 12, e0175689. [Google Scholar] [CrossRef]
- Doherty, A.F.; Ikuerowo, S.O.; Jeje, E.A.; Ibrahim, N.A.; Ojongbede, O.L.; Mutiu, W.B.; Omisanjo, O.A.; Abolarinwa, A.A. A Prospective Randomized Comparative Study of Targeted versusEmpirical Prophylactic Antibiotics in the Prevention of Infective Complications following Transrectal Ultrasound-Guided ProstateBiopsy. Ann. Afr. Med. 2019, 18, 132–137. [Google Scholar] [PubMed]
- Roberts, M.J.; Williamson, D.A.; Hadway, P.; Suhail ARDoi, S.A.R.; Gardiner, R.A.; Paterson, D.L. Baseline prevalence of antimicrobial resistance and subsequent infection following prostate biopsy using empirical or altered prophylaxis: A bias-adjusted meta-analysis. Int. J. Antimicrob. Agents 2014, 43, 301–309. [Google Scholar] [CrossRef] [PubMed]
- Gould, I.M. A review of the role of antibiotic policies in the control of antibiotic resistance. J. Antimicrob. Chemother. 1999, 43, 459–465. [Google Scholar] [CrossRef] [PubMed]
- Fijn, R.; Chow, M.C.; Schuur, P.M.; De Jong-Van den Berg, L.T.; Brouwers, J.R. Multicentre evaluation of prescribing concurrence with anti-infective guidelines: Epidemiological assessment of indicators. Pharm. Drug Saf. 2002, 11, 361–372. [Google Scholar] [CrossRef] [PubMed]
- Halm, E.A.; Atlas, S.J.; Borowsky, L.H.; Benzer, T.I.; Metlay, J.P.; Chang, Y.C.; Singer, D.E. Understanding physician adherence with a pneumonia practice guideline: Effects of patient, system, and physician factors. Arch. Intern. Med. 2000, 160, 98–104. [Google Scholar] [CrossRef] [Green Version]
- Van de Beek, D.; de Gans, J.; Spanjard, L.; Vermeulen, M.; Dankert, J. Antibiotic guidelines and antibiotic use in adult bacterial meningitis in the Netherlands. J. Antimicrob. Chemother. 2002, 49, 661–666. [Google Scholar] [CrossRef] [Green Version]
- Global Point Prevalence Survey on Antimicrobial Consumption and Resistance. Global-PPS Website. Available online: http://www.global-pps.com/ (accessed on 30 November 2021).
- Baur, D.; Gladstone, B.P.; Burkert, F.; Carrara, E.; Foschi, F.; Döbele, S.; Tacconelli, E. Effect of antibiotic stewardship on the incidence of infection and colonisation with antibiotic-resistant bacteria and Clostridium difficile infection: A systematic review and meta-analysis. Lancet Infect. Dis. 2017, 17, 990–1001. [Google Scholar] [CrossRef]
- Standard Treatment Guidelines. Ghana National Drugs Programme. Ministry of Health. Seventh Edition. 2017. Available online: https://www.moh.gov.gh/wp-content/uploads/2020/07/GHANA-STG-2017-1.pdf (accessed on 12 November 2021).
- Cardoso, T.; Almeida, M.; Friedman, N.D.; Aragão, I.; Costa-Pereira, A.; Sarmento, A.E.; Azevedo, L. Classification of healthcare-associated infection: A systematic review 10 years after the first proposal. BMC Med. 2014, 12, 40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Ward | % Treated (Number Treated) |
|---|---|
| General or mixed Adult ICU | 100.0 (7) |
| General or mixed Adult MW | 53.6 (246) |
| General or mixed Adult SW | 50.0 (162) |
| Haematology-Oncology PMW | 47.6 (10) |
| Neonatal Intensive Care Unit | 47.7 (41) |
| Paediatric MW | 76.3 (45) |
| Paediatric SW | 50.0 (16) |
| Antimicrobial prevalence | 53.3 (527) |
| Patients | Total 527 | ||||
|---|---|---|---|---|---|
| Adults ≥ 18 years | 379 | (72%) | |||
| Female | Male | Unknown | |||
| ≥60 years | 210 | 104 | 1 | 315 | (83%) |
| >60 years | 25 | 39 | 64 | (17%) | |
| Median age in yrs (iqr *) | 34 (16) | 45 (29) | |||
| Children < 18 years | 148 | (28%) | |||
| Female | Male | Unknown | |||
| <18 years | 18 | 32 | 50 | (34%) | |
| <5 years | 8 | 7 | 15 | (10%) | |
| <24 months | 41 | 41 | 1 | 83 | (56%) |
| Median age in years (iqr) | 0.3 (7) | 1.5 (8) | |||
| Empirical | Targeted | Total | ||||
|---|---|---|---|---|---|---|
| N | % | N | % | N | % | |
| CAI | 374 | 94.0 | 24 | 6.0 | 398 | 73.4 |
| HAI | 124 | 86.1 | 20 | 13.9 | 144 | 26.6 |
| 498 | 44 | 542 | ||||
| Characteristic | Number | (%) |
|---|---|---|
| Ten commonest diagnoses treated with antimicrobials | ||
| Pneumonia | 58 | 18.4 |
| Skin and soft tissue | 36 | 11.4 |
| Sepsis | 35 | 11.1 |
| Upper respiratory tract infection | 25 | 7.9 |
| Malaria | 24 | 7.6 |
| Infection of central nervous system | 22 | 7.0 |
| Obstetrics/gynaecology infection | 22 | 7.0 |
| Bone and joint infection | 15 | 4.8 |
| Gastro-intestinal infection | 14 | 4.4 |
| Intra-abdominal sepsis | 10 | 3.2 |
| Treatment according to biomarkers (n = 527) | ||
| No | 502 | 95.3 |
| Yes | 25 | 4.7 |
| Antibiotic quality indicators by department | ||
| Medical | ||
| Reason in notes | 235 | 76.1 |
| Guidelines missing | 79 | 25.6 |
| Guideline compliant | 122 | 85.9 |
| Stop/review date | 119 | 38.5 |
| Surgical | ||
| Reason in notes | 183 | 44.3 |
| Guidelines missing | 127 | 30.8 |
| Guideline compliant | 135 | 83.3 |
| Stop/review date | 206 | 49.9 |
| Intensive care unit | ||
| Reason in notes | 35 | 41.2 |
| Guidelines missing | 11 | 12.9 |
| Guideline compliant | 38 | 92.7 |
| Stop/review date | 10 | 11.8 |
| Patients with isolated multi-drug resistant pathogen | ||
| Third generation cephalosporin resistant (TGCR) | 2 | 4.4 |
| Carbapenem-resistant enterobacteriaceae | 2 | 4.4 |
| Carbapenem-resistant non fermentor Gram-negative bacilli | 2 | 4.4 |
| ESBL-producing Enterobacteriaceae | 7 | 15.6 |
| ESBL-producing Enterobacteriaceae-TGCR | 1 | 2.2 |
| No MDR recorded | 19 | 42.2 |
| Targeted treatment against other MDR organisms | 10 | 22.2 |
| Vancomycin-resistant enterobacteriaceae (VRE) | 2 | 4.4 |
| ATC Code | Total Systemic Antimicrobials | No (%) | |
|---|---|---|---|
| J01 | Antibacterials for systemic use | 807 | 83.5% |
| P01AB | Nitroimidazole derivatives | 83 | 8.6% |
| P01B | Antimalarials | 29 | 3.0% |
| P01B | 28 | ||
| P01BB | 1 | ||
| J02 | Antimycotics for systemic use | 10 | 1.0% |
| J02AC | |||
| J04 | Antimycobacterials | 10 | 1.0% |
| J04AM | |||
| J05 | Antivirals for systemic use | 20 | 2.1% |
| J05AR | 9 | ||
| J05AF | 6 | ||
| J05AG | 4 | ||
| J05AB | 1 | ||
| A07AA | Intestinal Anti-infectives | 7 | 0.7% |
| D01 | Antifungals for dermatological use | 1 | 0.1% |
| D01BA | |||
| 967 | 100% | ||
| Therapeutic Prescription | No |
|---|---|
| Penicillins with beta-lactamase inhibitor | 141 |
| 3rd gen cephalosporins | 113 |
| 2nd gen cephalosporins | 86 |
| Beta-lactamase resistant penicillins | 67 |
| Beta-lactamase sensitive penicillins | 12 |
| Penicillins with extended spectrum | 8 |
| Imidazole derivatives | 95 |
| Fluoroquinolones | 86 |
| Aminoglycosides | 66 |
| Lincosamides | 62 |
| Macrolides | 28 |
| Carbapenems | 19 |
| Co-trimoxazole | 17 |
| Tetracyclines | 3 |
| Glycopeptide antibacterials | 2 |
| Combinations of antimicrobials | 1 |
| Nitrofuran derivatives | 1 |
| 807 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ankrah, D.; Owusu, H.; Aggor, A.; Osei, A.; Ampomah, A.; Harrison, M.; Nelson, F.; Aboagye, G.O.; Ekpale, P.; Laryea, J.; et al. Point Prevalence Survey of Antimicrobial Utilization in Ghana’s Premier Hospital: Implications for Antimicrobial Stewardship. Antibiotics 2021, 10, 1528. https://doi.org/10.3390/antibiotics10121528
Ankrah D, Owusu H, Aggor A, Osei A, Ampomah A, Harrison M, Nelson F, Aboagye GO, Ekpale P, Laryea J, et al. Point Prevalence Survey of Antimicrobial Utilization in Ghana’s Premier Hospital: Implications for Antimicrobial Stewardship. Antibiotics. 2021; 10(12):1528. https://doi.org/10.3390/antibiotics10121528
Chicago/Turabian StyleAnkrah, Daniel, Helena Owusu, Asiwome Aggor, Anthony Osei, Agneta Ampomah, Mark Harrison, Frempomaa Nelson, Grace Owusu Aboagye, Priscilla Ekpale, Jennifer Laryea, and et al. 2021. "Point Prevalence Survey of Antimicrobial Utilization in Ghana’s Premier Hospital: Implications for Antimicrobial Stewardship" Antibiotics 10, no. 12: 1528. https://doi.org/10.3390/antibiotics10121528
APA StyleAnkrah, D., Owusu, H., Aggor, A., Osei, A., Ampomah, A., Harrison, M., Nelson, F., Aboagye, G. O., Ekpale, P., Laryea, J., Selby, J., Amoah, S., Lartey, L., Addison, O., Bruce, E., Mahungu, J., & Mirfenderesky, M. (2021). Point Prevalence Survey of Antimicrobial Utilization in Ghana’s Premier Hospital: Implications for Antimicrobial Stewardship. Antibiotics, 10(12), 1528. https://doi.org/10.3390/antibiotics10121528






