Development of Tin Oxide-Based Nanosensors for Electronic Nose Environmental Applications
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Precursor Solution
2.3. Synthesis of Tin Oxide Nanofibres
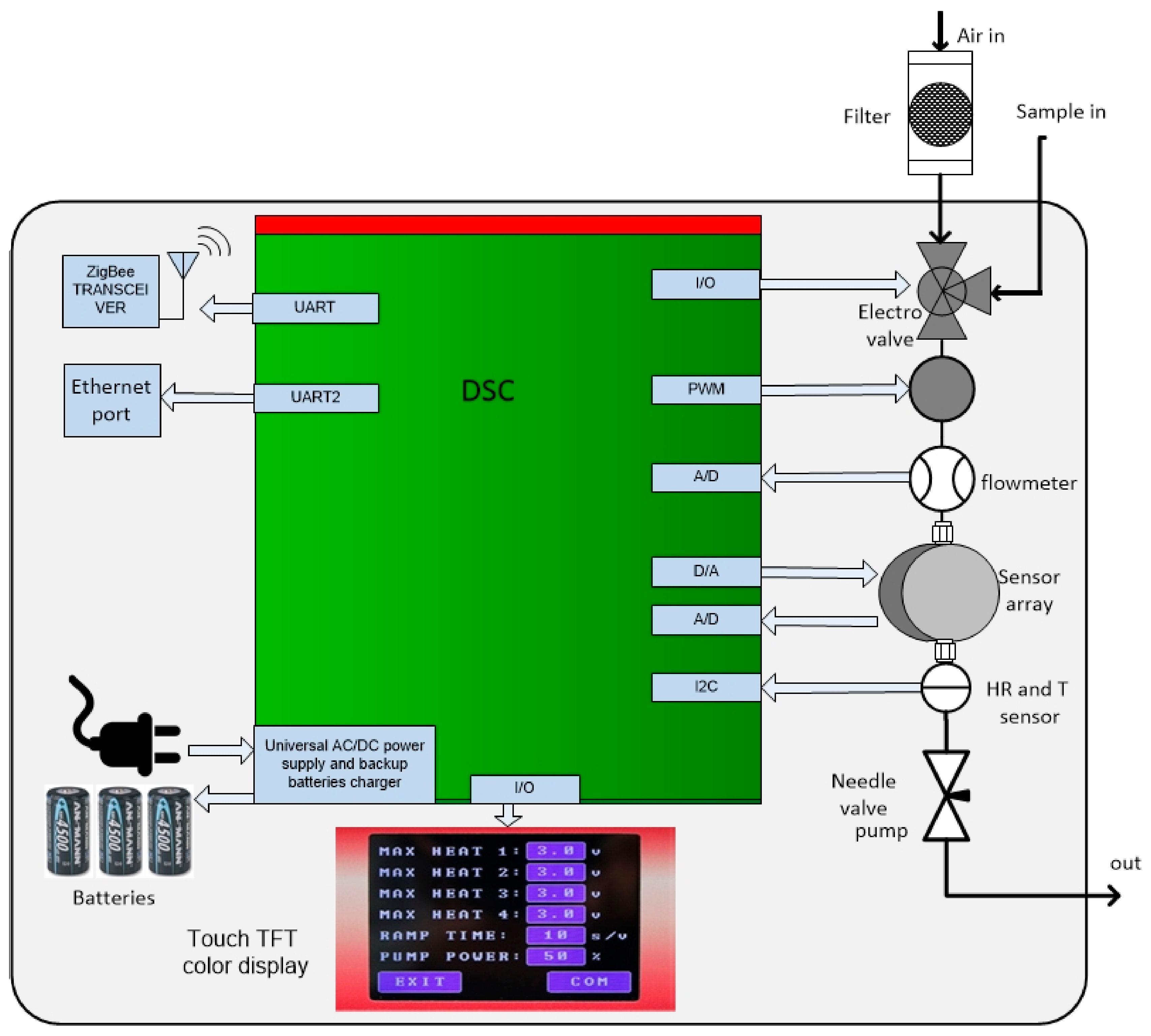
2.4. Experimental Setup of the E-Nose System
Electronic Noses
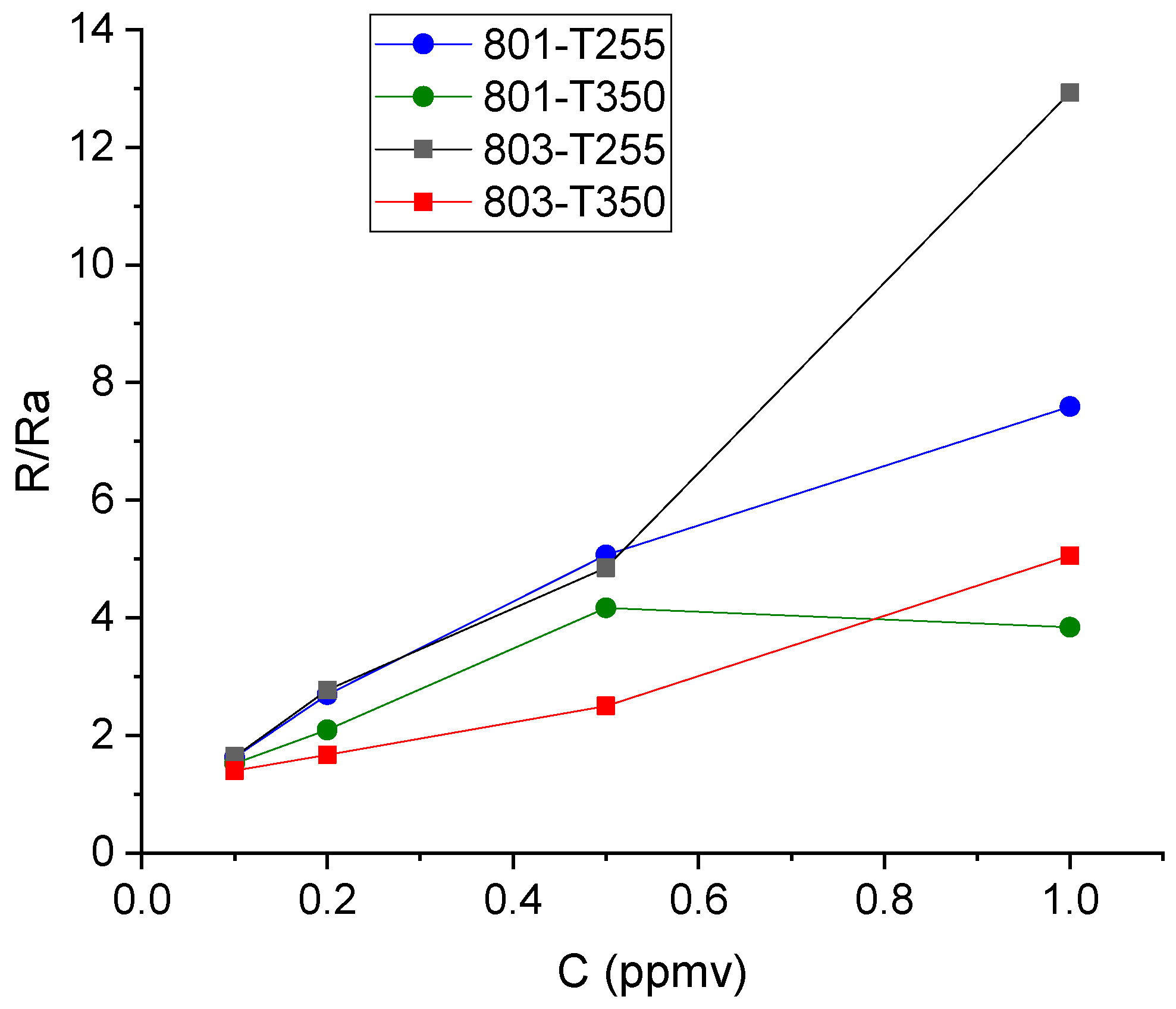
2.5. Sensor Tested
3. Results
3.1. Morphological Characterization of Tin Oxide Nanofibres
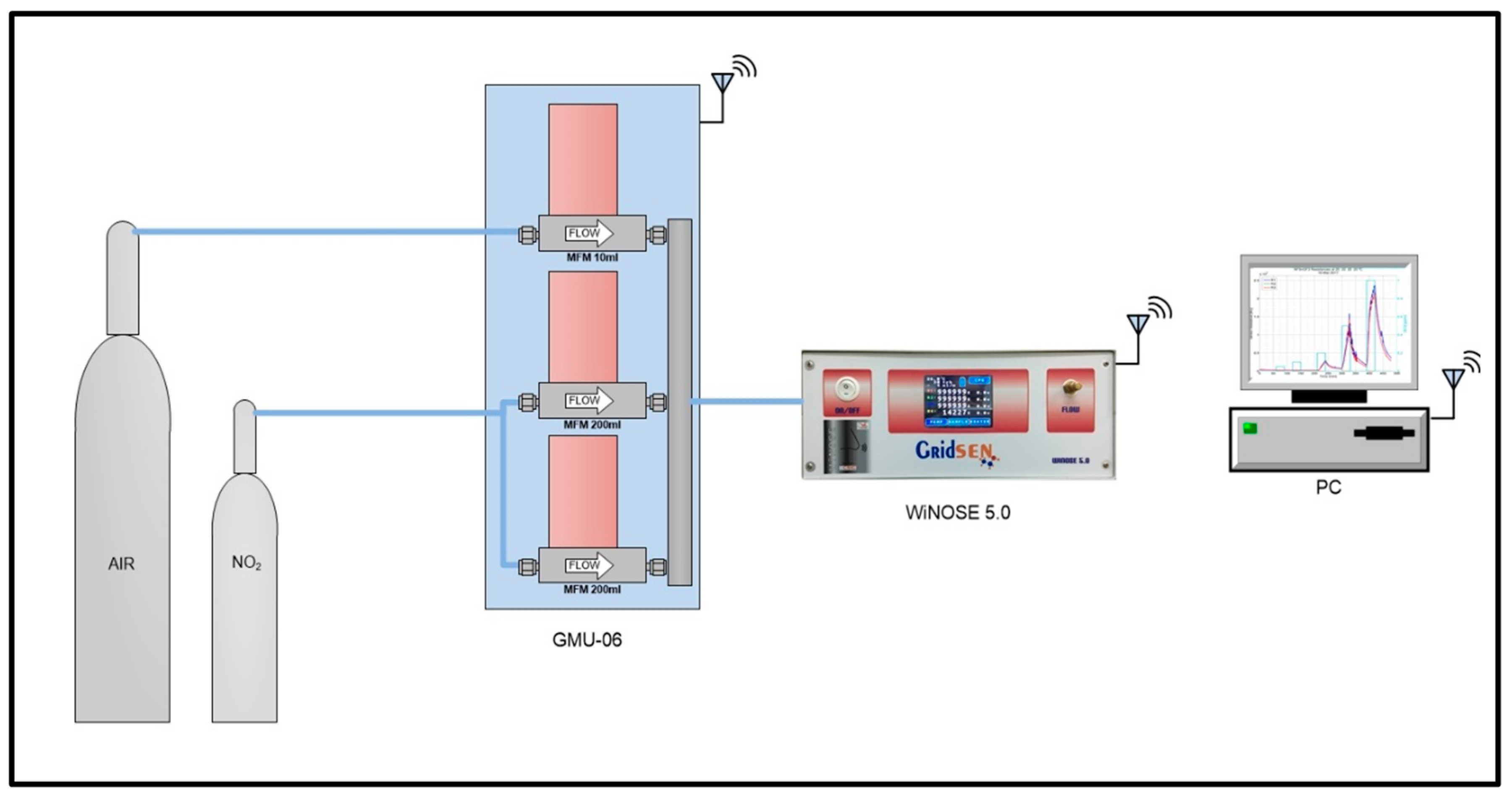
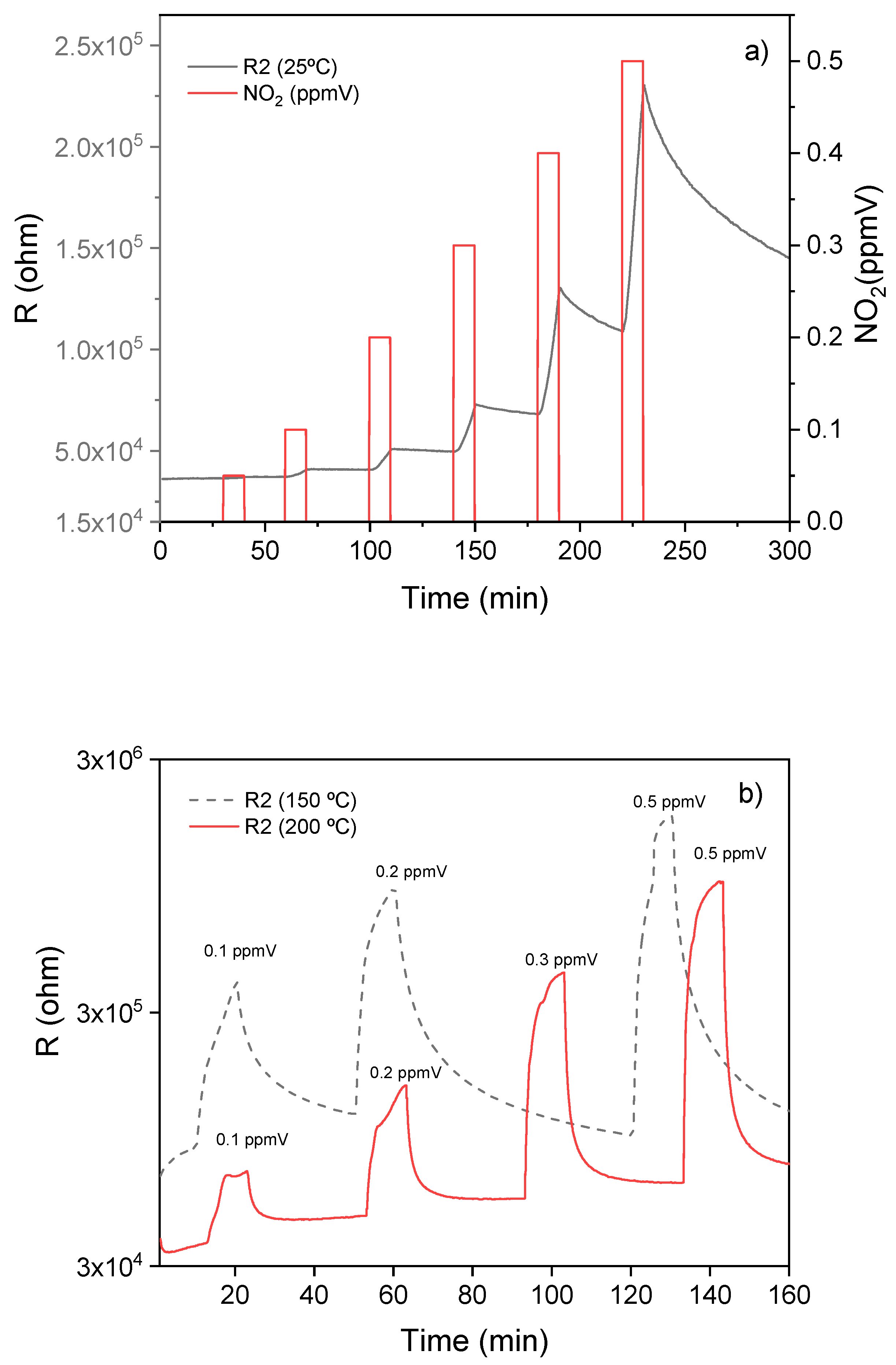
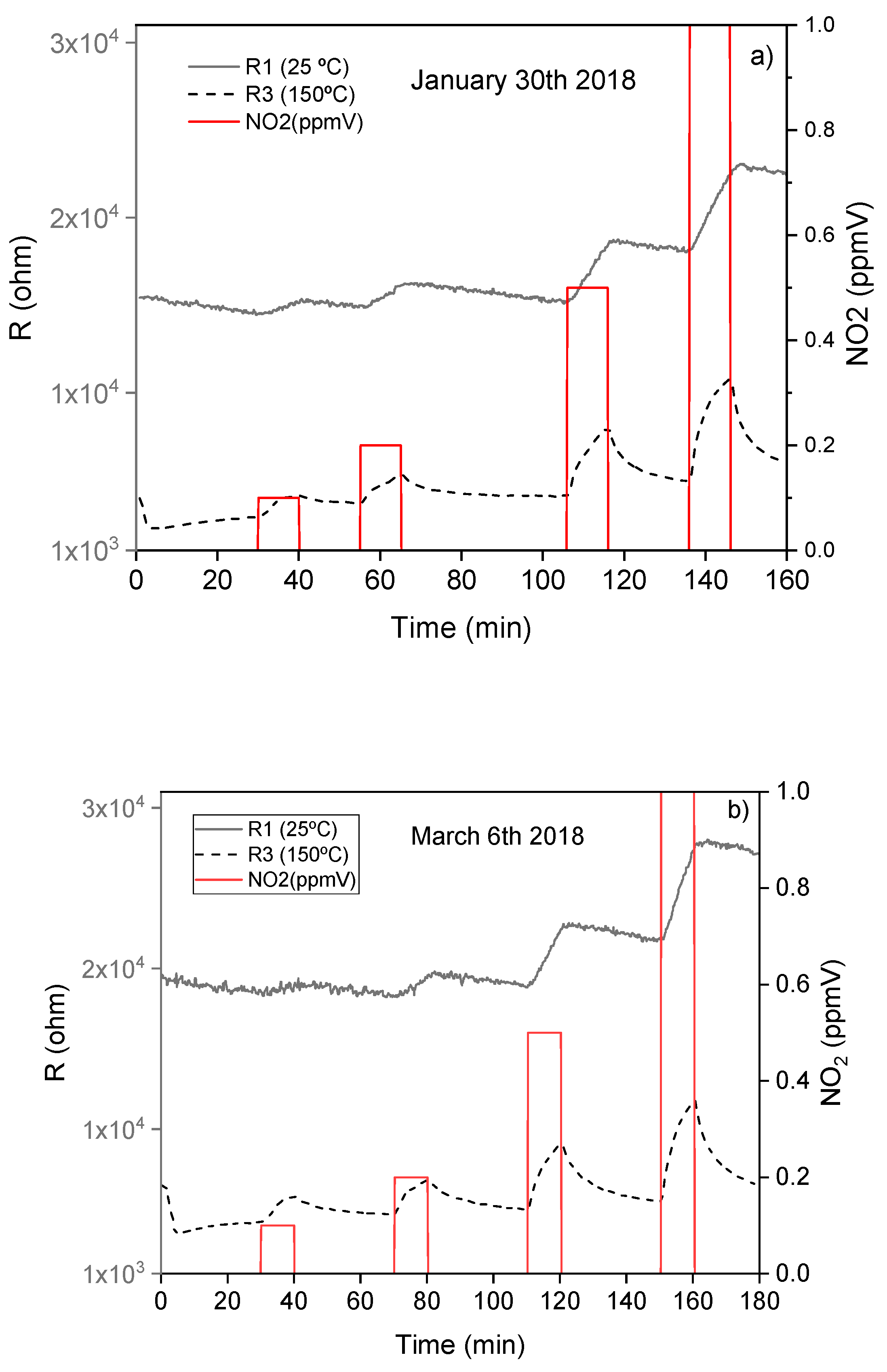
3.2. WiNOSE 5.0
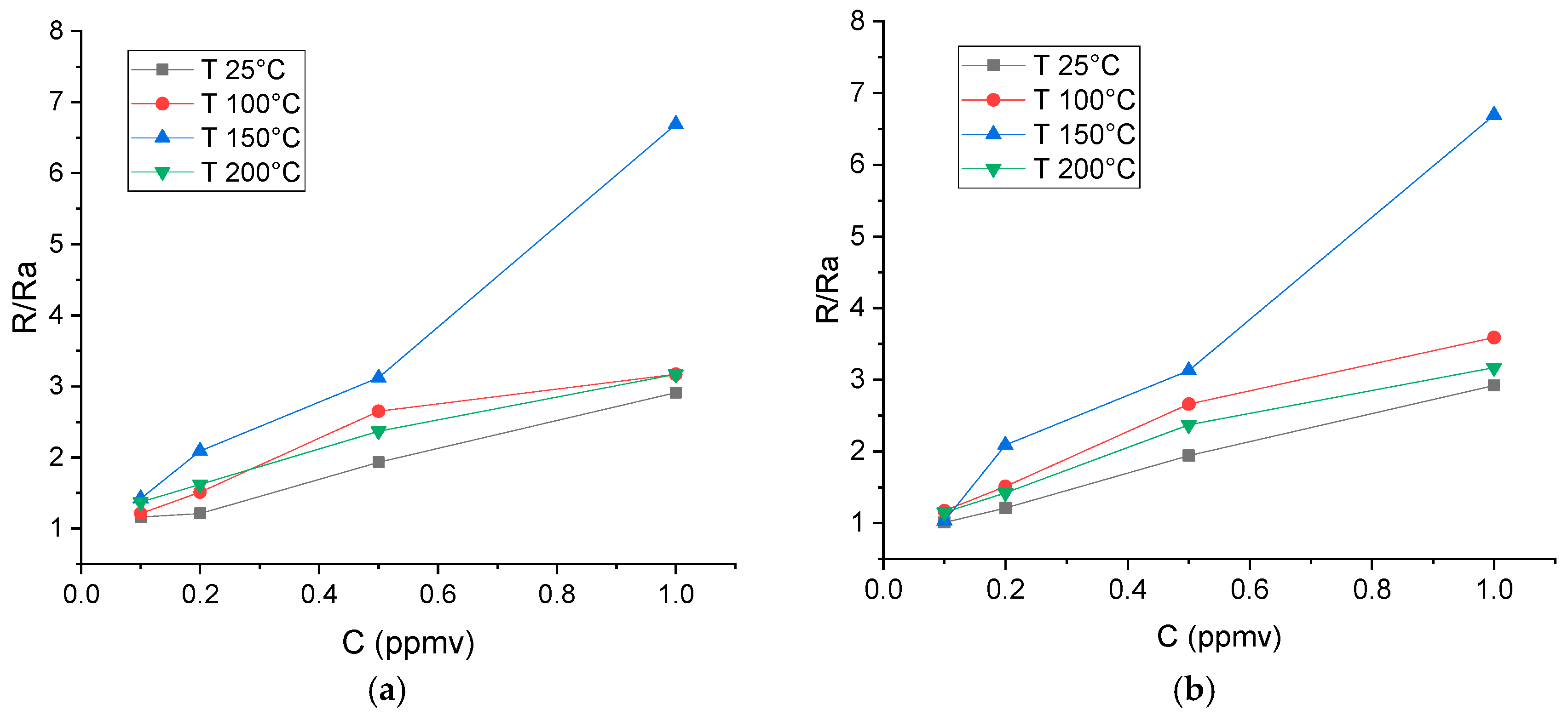
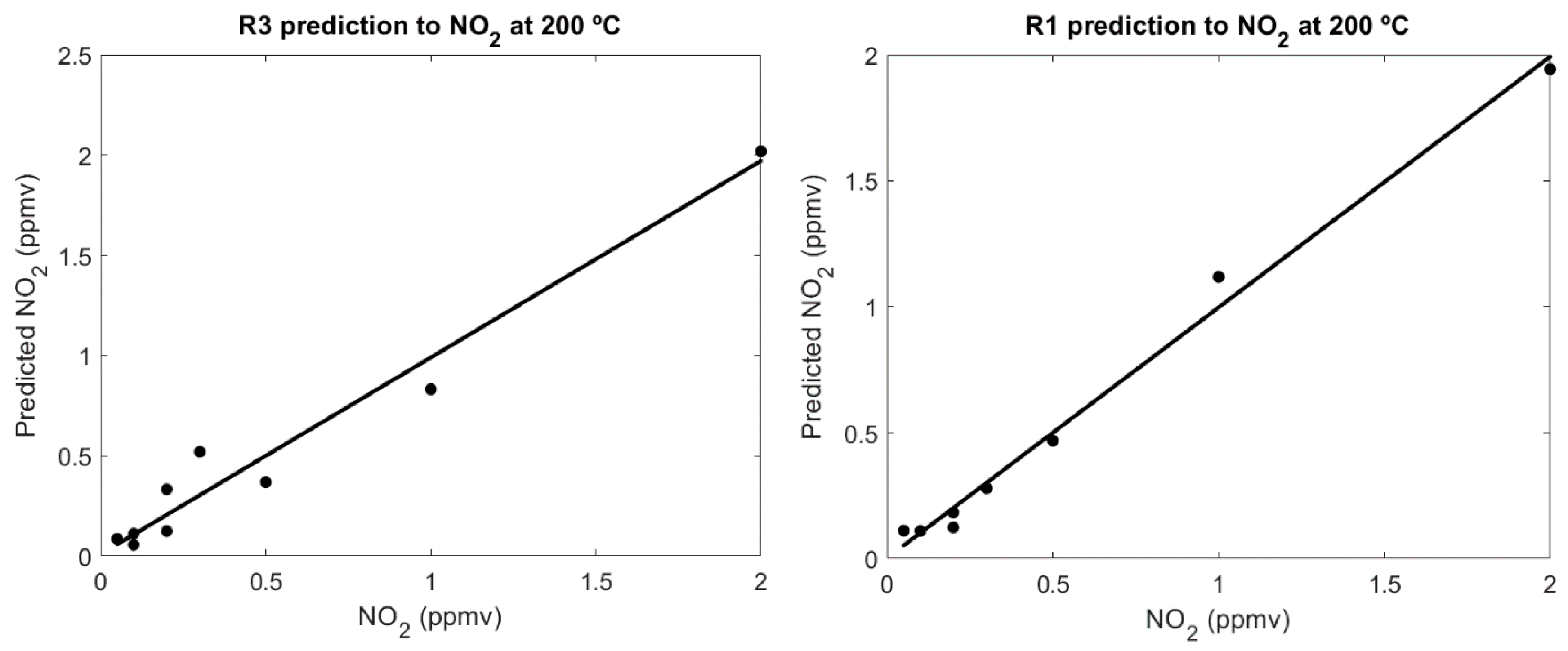
Calibrations of Nanosensors
3.3. WiNOSE 6.0
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Low-Cost Sensors for the Measurement of Atmospheric Composition: Overview of Topic and Future Applications|Climate & Clean Air Coalition. Available online: http://www.ccacoalition.org/en/resources/low-cost-sensors-measurement-atmospheric-composition-overview-topic-and-future (accessed on 25 October 2018).
- Guillot, J.-M. E-Noses: Actual Limitations and Perspectives for Environmental Odour Analysis. Chem. Eng. Trans. 2016, 54, 223–228. [Google Scholar]
- Szulczyński, B.; Wasilewski, T.; Wojnowski, W.; Majchrzak, T.; Dymerski, T.; Namieśnik, J.; Gębicki, J. Different Ways to Apply a Measurement Instrument of E-Nose Type to Evaluate Ambient Air Quality with Respect to Odour Nuisance in a Vicinity of Municipal Processing Plants. Sensors 2017, 17, 2671. [Google Scholar] [CrossRef] [PubMed]
- Jasinski, G.; Wozniak, L.; Kalinowski, P.; Jasinski, P. Evaluation of the Electronic Nose Used for Monitoring Environmental Pollution. In Proceedings of the 2018 IEEE XV International Scientific Conference on Optoelectronic and Electronic Sensors (COE), Orlando, FL, USA, 10–13 November 2018; pp. 1–4. [Google Scholar]
- Panneerselvam, G.; Thirumal, V.; Pandya, H.M. Review of Surface Acoustic Wave Sensors for the Detection and Identification of Toxic Environmental Gases/vapours. Arch. Acoust. 2018, 43, 357–367. [Google Scholar]
- Dung, T.; Oh, Y.; Choi, S.-J.; Kim, I.-D.; Oh, M.-K.; Kim, M.; Dung, T.T.; Oh, Y.; Choi, S.-J.; Kim, I.-D.; et al. Applications and Advances in Bioelectronic Noses for Odour Sensing. Sensors 2018, 18, 103. [Google Scholar] [CrossRef] [PubMed]
- Capezzuto, L.; Abbamonte, L.; De Vito, S.; Massera, E.; Formisano, F.; Fattoruso, G.; Di Francia, G.; Buonanno, A. A Maker Friendly Mobile and Social Sensing Approach to Urban Air Quality Monitoring. In Proceedings of the 2014 IEEE Sensors, Valencia, Spain, 2–5 November 2014. [Google Scholar]
- Hannon, A.; Lu, Y.; Li, J.; Meyyappan, M. A Sensor Array for the Detection and Discrimination of Methane and Other Environmental Pollutant Gases. Sensors 2016, 16, 1163. [Google Scholar] [CrossRef] [PubMed]
- Herrero, J.L.; Lozano, J.; Santos, J.P.; Fernandez, J.A.; Marcelo, J.I.S. A Web-Based Approach for Classifying Environmental Pollutants Using Portable E-Nose Devices. IEEE Intell. Syst. 2016, 31, 108–112. [Google Scholar] [CrossRef]
- Sun, Y.-F.; Liu, S.-B.; Meng, F.-L.; Liu, J.-Y.; Jin, Z.; Kong, L.-T.; Liu, J.-H.; Sun, Y.-F.; Liu, S.-B.; Meng, F.-L.; et al. Metal Oxide Nanostructures and their Gas Sensing Properties: A Review. Sensors 2012, 12, 2610–2631. [Google Scholar] [CrossRef] [PubMed]
- Comini, E.; Baratto, C.; Faglia, G.; Ferroni, M.; Vomiero, A.; Sberveglieri, G. Quasi-One Dimensional Metal Oxide Semiconductors: Preparation, Characterization and Application as Chemical Sensors. Prog. Mater. Sci. 2009, 54, 1–67. [Google Scholar] [CrossRef]
- Pan, J.; Shen, H.; Mathur, S. One-Dimensional SnO2 Nanostructures: Synthesis and Applications. J. Nanotechnol. 2012, 2012, 1–12. [Google Scholar] [CrossRef]
- Yu, F.; Tang, D.; Hai, K.; Luo, Z.; Chen, Y.; He, X.; Peng, Y.; Yuan, H.; Zhao, D.; Yang, Y. Fabrication of SnO2 One-Dimensional Nanosturctures with Graded Diameters by Chemical Vapor Deposition Method. J. Cryst. Growth 2010, 312, 220–225. [Google Scholar] [CrossRef]
- Luan, C.; Zhu, Z.; Mi, W.; Ma, J. Structural and Electrical Properties of SnO2 Films Grown on R-Cut Sapphire at Different Substrate Temperature by MOCVD. Vacuum 2014, 99, 110–114. [Google Scholar] [CrossRef]
- Kim, J.-H.; Abideen, Z.U.; Zheng, Y.; Kim, S.S. Improvement of Toluene-Sensing Performance of SnO2 Nanofibers by Pt Functionalization. Sensors 2016, 16, 1857. [Google Scholar] [CrossRef] [PubMed]
- Park, J.Y.; Asokan, K.; Choi, S.-W.; Kim, S.S. Growth Kinetics of Nanograins in SnO2 Fibers and Size Dependent Sensing Properties. Sens. Actuators B Chem. 2011, 152, 254–260. [Google Scholar] [CrossRef]
- Kenry; Lim, C.T. Nanofiber Technology: Current Status and Emerging Developments. Prog. Polym. Sci. 2017, 70, 1–17. [Google Scholar] [CrossRef]
- Li, D.; McCann, J.T.; Xia, Y.; Marquez, M. Electrospinning: A Simple and Versatile Technique for Producing Ceramic Nanofibers and Nanotubes. J. Am. Ceram. Soc. 2006, 89, 1861–1869. [Google Scholar] [CrossRef]
- Huang, Z.-M.; Zhang, Y.-Z.; Kotaki, M.; Ramakrishna, S. A Review on Polymer Nanofibers by Electrospinning and Their Applications in Nanocomposites. Compos. Sci. Technol. 2003, 63, 2223–2253. [Google Scholar] [CrossRef]
- Lim, S.K.; Hwang, S.-H.; Chang, D.; Kim, S. Preparation of Mesoporous In2O3 Nanofibers by Electrospinning and Their Application as a CO Gas Sensor. Sens. Actuators B Chem. 2010, 149, 28–33. [Google Scholar] [CrossRef]
- Kim, I.-D.; Rothschild, A. Nanostructured Metal Oxide Gas Sensors Prepared by Electrospinning. Polym. Adv. Technol. 2011, 22, 318–325. [Google Scholar] [CrossRef]
- Ding, B.; Wang, M.; Yu, J.; Sun, G.; Ding, B.; Wang, M.; Yu, J.; Sun, G. Gas Sensors Based on Electrospun Nanofibers. Sensors 2009, 9, 1609–1624. [Google Scholar] [CrossRef] [PubMed]
- Latza, U.; Gerdes, S.; Baur, X. Effects of nitrogen dioxide on human health: Systematic review of experimental and epidemiological studies conducted between 2002 and 2006. Int. J. Hyg. Environ. Health 2009, 212, 271–287. [Google Scholar] [CrossRef] [PubMed]
- Air Quality in Europe—2018 Report. Available online: https://www.eea.europa.eu/publications/air-quality-in-europe-2018 (accessed on 15 november 2018).
- Ambient (Outdoor) Air Quality and Health. Available online: http://www.who.int/news-room/fact-sheets/detail/ambient-(outdoor)-air-quality-and-health (accessed on 25 October 2018).
- European Environment Agency. Communication from the Commission to the Council, the European Parliament, the European Economic and Social Committee and the Committee of the Regions—“A Clean Air Programme for Europe”, COM (2013) 918 final. Available online: http://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:52013DC0918&from=EN (accessed on 1 February 2019).
- European Environment Agency. Directive 2008/50/EC of the European Parliament and of the Council of 21 May 2008 on ambient air quality and cleaner air for Europe. Available online: http://eur-lex.europa.eu/legal-content/en/ALL/?uri=CELEX:32008L0050 (accessed on 1 February 2019).
- TD1105—European Network on New Sensing Technologies for Air-Pollution Control and Environmental Sustainability—EuNetAir. Available online: https://www.cost.eu/actions/TD1105/#tabs|Name:overview (accessed on 1 February 2019).
- Spinelle, L.; Gerboles, M.; Villani, M.G.; Aleixandre, M.; Bonavitacola, F. Field Calibration of a Cluster of Low-Cost Available Sensors for Air Quality Monitoring. Part A: Ozone and Nitrogen Dioxide. Sens. Actuators B Chem. 2015, 215, 249–257. [Google Scholar] [CrossRef]
- Santos, J.; Fernández, M.; Fontecha, J.; Matatagui, D.; Sayago, I.; Horrillo, M.; Gracia, I.; Santos, J.P.; Fernández, M.J.; Fontecha, J.L.; et al. Nanocrystalline Tin Oxide Nanofibers Deposited by a Novel Focused Electrospinning Method. Application to the Detection of TATP Precursors. Sensors 2014, 14, 24231–24243. [Google Scholar] [CrossRef] [PubMed]
- Santos, J.P.; Aleixandre, M.; Arroyo, P.; Suárez, J.I.; Lozano, J. An Advanced Hand Held Electronic Nose for Ambient Air Applications. Chem. Eng. Trans. 2018, 68, 235–240. [Google Scholar]
- Santos, J.P.; Aleixandre, M.; Horrillo, M.C. WiNOSE: Wireless Electronic Nose for Outdoors Applications. Chem. Eng. Trans. 2010, 23, 159–164. [Google Scholar]
- Han, D.; Zhai, L.; Gu, F.; Wang, Z. Highly sensitive NO2 gas sensor of ppb-level detection based on In2O3 nanobricks at low temperature. Sens. Actuators B Chem. 2018, 262, 655–663. [Google Scholar] [CrossRef]
- Xu, X.; Zhang, H.; He, C.; Pu, C.; Leng, Y.; Li, G.; Hou, S.; Zhu, Y.; Fu, L.; Lu, G. Synthesis and NO2 Sensing Properties of Indium Oxide Nanorod Clusters via a Simple Solvothermal Route. RSC Adv. 2016, 6, 47083–47088. [Google Scholar] [CrossRef]
- Ahn, M.-W.; Park, K.-S.; Heo, J.-H.; Kim, D.-W.; Choi, K.J.; Park, J.-G. On-Chip Fabrication of ZnO-Nanowire Gas Sensor with High Gas Sensitivity. Sens. Actuators B Chem. 2009, 138, 168–173. [Google Scholar] [CrossRef]
- Choi, Y.-J.; Hwang, I.-S.; Park, J.-G.; Choi, K.J.; Park, J.-H.; Lee, J.-H. Novel Fabrication of an SnO2 Nanowire Gas Sensor with High Sensitivity. Nanotechnology 2008, 19, 95508. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, D.; Qin, L.; Zhao, P.; Liu, F.; Chuai, X.; Sun, P.; Liang, X.; Gao, Y.; Sun, Y.; et al. Preparation and Gas Sensing Properties of Hierarchical Leaf-like SnO2 Materials. Sens. Actuators B Chem. 2018, 255, 2944–2951. [Google Scholar] [CrossRef]

| Average Annual | Average Hourly |
|---|---|
| 40 µg/m3 (0.02 ppm) | 200 µg/m3 (0.11 ppm) not to exceed more than 18 h per year |
| T (°C) | RMS R1 | RMS R3 | RMS PLS | R2 R1 | R2 R3 | R2 PLS |
|---|---|---|---|---|---|---|
| 25 | 0.186 | 0.0288 | 0.328 | 0.993 | 0.997 | 0.992 |
| 50 | 0.159 | 0.090 | 0.140 | 0.989 | 0.989 | 0.998 |
| 100 | 0.106 | 0.059 | 0.267 | 0.998 | 0.987 | 0.979 |
| 150 | 0.539 | 0.025 | 0.473 | 0.999 | 0.995 | 0.969 |
| 200 | 0.119 | 0.246 | 0.772 | 0.954 | 0.996 | 0.840 |
| 250 | 0.321 | 0.034 | 0.176 | 0.903 | 0.999 | 0.975 |
| 300 | 0.345 | 0.227 | 2.212 | 0.976 | 0.964 | 0.864 |
| T (°C) | RMS S801 | RMS S803 | RMS PLS | R2 S801 | R2 S803 | R2 PLS |
|---|---|---|---|---|---|---|
| 255 | 0.008 | 0.043 | 1.197 | 0.990 | 0.987 | 0.941 |
| 350 | 0.372 | 0.006 | 0.695 | 0.949 | 0.989 | 0.984 |
| Sensitive Layer | Concentration (ppm) | T (°C) | Response (RNO2/Rair) | Ref. |
|---|---|---|---|---|
| In2O3 (nanorod clusters) | 0.5 | 150 | 41 | [34] |
| ZnO (nanowires) | 0.5 | 225 | 18 | [35] |
| SnO2 (nanowires) | 0.5 | 200 | 17 | [36] |
| SnO2 (hierarchical leaf-like) | 0.5 | 65 | 7 | [37] |
| SnO2 (nanofibrefibres) | 0.1/0.5 | 25 | 1.16/1.93 | This work |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sayago, I.; Aleixandre, M.; Santos, J.P. Development of Tin Oxide-Based Nanosensors for Electronic Nose Environmental Applications. Biosensors 2019, 9, 21. https://doi.org/10.3390/bios9010021
Sayago I, Aleixandre M, Santos JP. Development of Tin Oxide-Based Nanosensors for Electronic Nose Environmental Applications. Biosensors. 2019; 9(1):21. https://doi.org/10.3390/bios9010021
Chicago/Turabian StyleSayago, Isabel, Manuel Aleixandre, and José Pedro Santos. 2019. "Development of Tin Oxide-Based Nanosensors for Electronic Nose Environmental Applications" Biosensors 9, no. 1: 21. https://doi.org/10.3390/bios9010021
APA StyleSayago, I., Aleixandre, M., & Santos, J. P. (2019). Development of Tin Oxide-Based Nanosensors for Electronic Nose Environmental Applications. Biosensors, 9(1), 21. https://doi.org/10.3390/bios9010021






