Biochemical Changes in Irradiated Oral Mucosa: A FTIR Spectroscopic Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Clinical Samples
2.2. Light Microscopy (LM)
2.3. Fourier-Transform Infrared (FTIR) Scanning
2.4. Data Analysis
3. Results
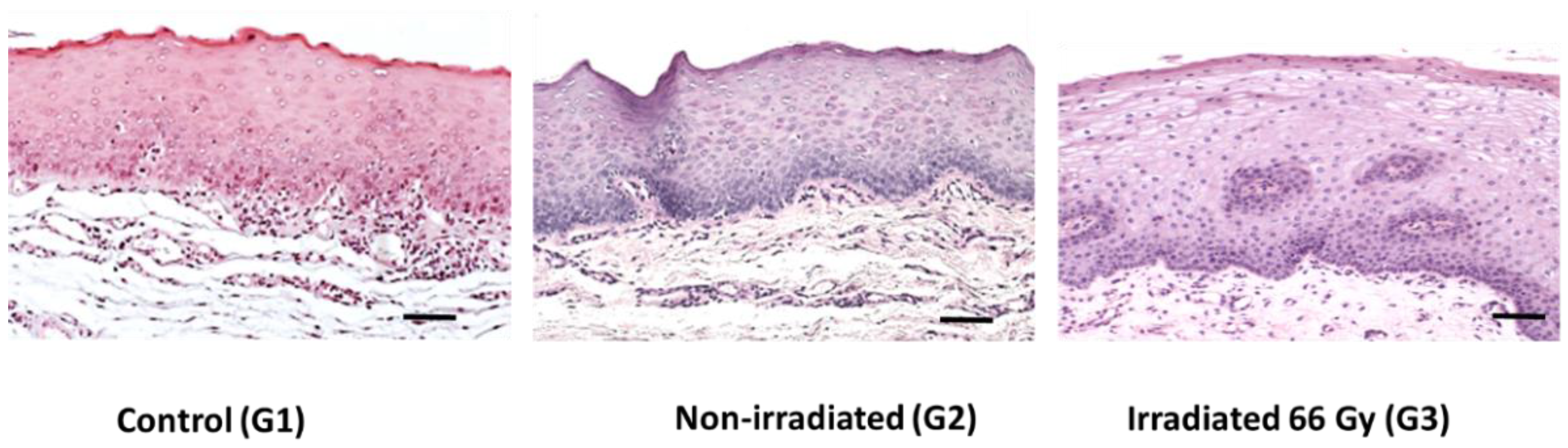
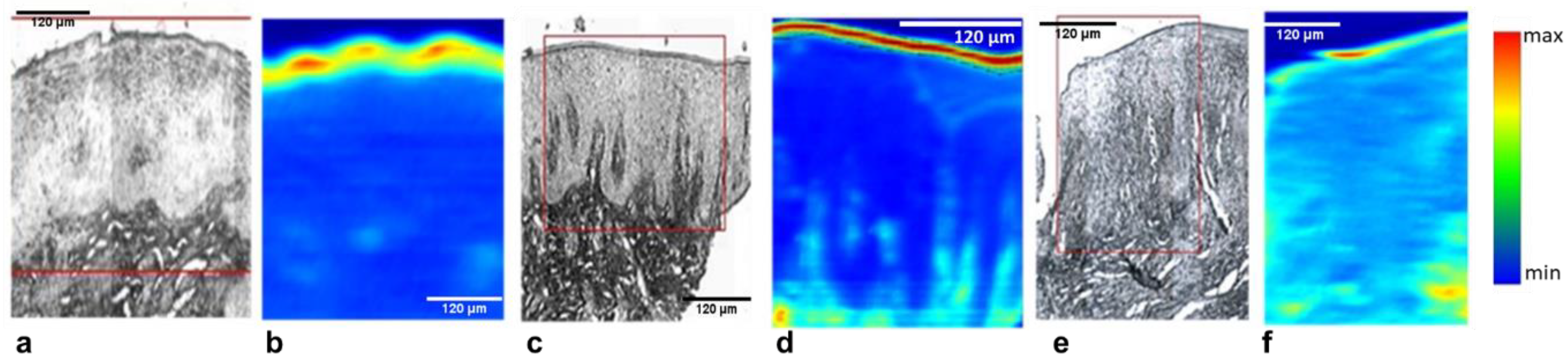
3.1. Histological Analysis using Light Microscopy
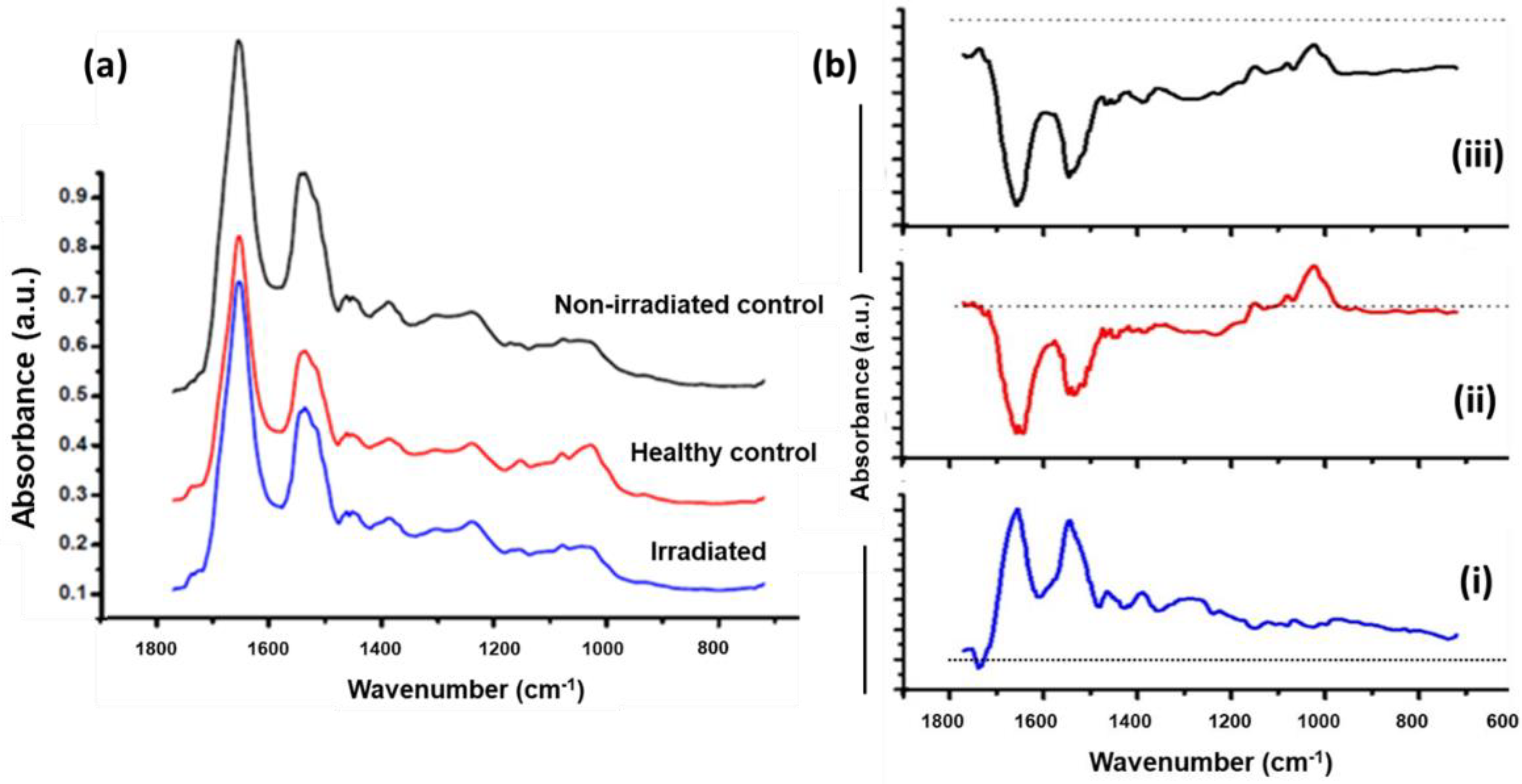
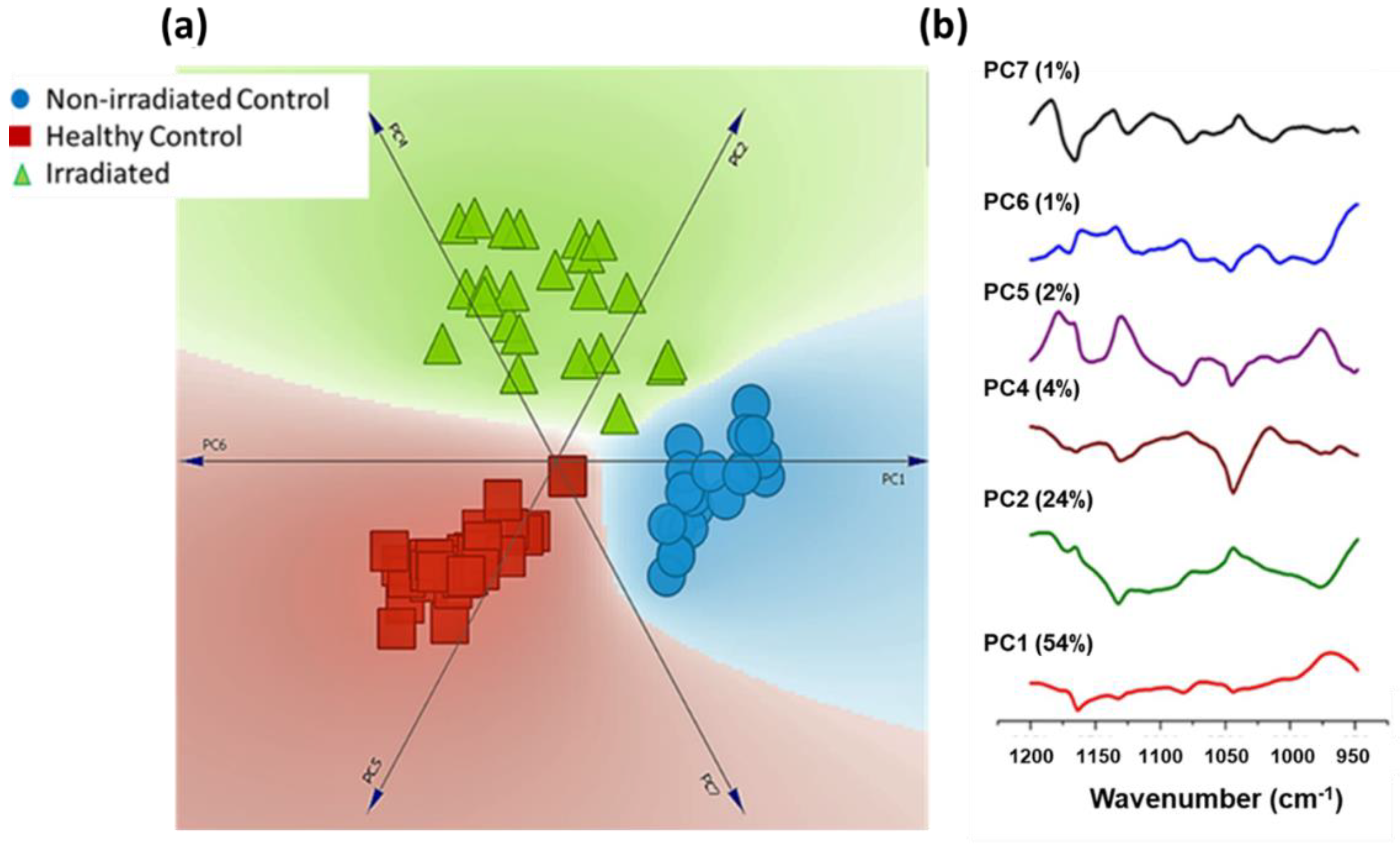
3.2. FTIR analysis
4. Discussion
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Caudell, J.; Torres-Roca, J.F.; Gillies, R.J.; Enderling, H.; Kim, S.; Rishi, A.; Moros, E.G.; Harrison, L.B. The future of personalized radiotherapy for head and neck cancer. Lancet Oncol. 2017, 18, e266–e273. [Google Scholar] [CrossRef]
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef] [PubMed]
- Sonis, S.T. Oral mucositis. Anti-Cancer Drugs 2011, 22, 607–612. [Google Scholar] [CrossRef] [PubMed]
- Delanian, S.; Lefaix, J.L. Current management for late normal tissue injury: Radiation-induced fibrosis and necrosis. Semin. Radiat. Oncol. 2007, 17, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Hashibe, M.; Ritz, B.; Le, A.D.; Li, G.; Sankaranarayanan, R.; Zhang, Z.F. Radiotherapy for oral cancer as a risk factor for second primary cancers. Cancer Lett. 2005, 220, 185–195. [Google Scholar] [CrossRef] [PubMed]
- Hannig, C.; Hannig, M.; Kensche, A.; Carpenter, G. The mucosal pellicle—An underestimated factor in oral physiology. Arch. Oral Biol. 2017, 80, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Ukkonen, H.; Pirhonen, P.; Herrala, M.; Mikkonen, J.J.; Singh, S.P.; Sormunen, R.; Kullaa, A.M. Oral mucosal epithelial cells express the membrane anchored mucin MUC1. Arch. Oral Biol. 2017, 73, 269–273. [Google Scholar] [CrossRef]
- Kullaa, A.M.; Asikainen, P.; Herrala, M.; Ukkonen, H.; Mikkonen, J.J. Microstructure of oral epithelial cells as an underlying basis for salivary mucosal pellicle. Ultrastruct. Pathol. 2014, 38, 382–386. [Google Scholar] [CrossRef]
- Asikainen, P.; Mikkonen, J.J.; Ruotsalainen, T.J.; Koistinen, A.P.; Kullaa, A.M. Microstructure of the superficial epithelial cells of the human oral mucosa. Ultrastruct. Pathol. 2014, 38, 6–12. [Google Scholar] [CrossRef]
- Asikainen, P.; Mikkonen, J.J.; Kullaa, A.M. Surface morphology of superficial cells in irradiated oral mucosa: An experimental study in beagle dog. Ultrastruct. Pathol. 2014, 38, 268–272. [Google Scholar] [CrossRef]
- Pillai, K.R.; Kannan, S.; Koshy, P.; Mathew, B.; Thampi, B.S.; Nalinakumari, K.R.; Nair, M.K.; Menon, V.P. Scanning electron microscopy of different types of oral leukoplakia: Comparison with normal and malignant oral mucosa. Eur. J. Cancer Part B Oral Oncol. 1994, 30, 400–404. [Google Scholar] [CrossRef]
- Asikainen, P.J.; Dekker, H.; Sirvio, E.; Mikkonen, J.; Schulten, E.; Bloemena, E.; Koistinen, A.; Ten Bruggenkate, C.M.; Kullaa, A.M. Radiation-induced changes in the microstructure of epithelial cells of the oral mucosa: A comparative light and electron microscopic study. J. Oral Pathol. Med. 2017, 46, 1004–1010. [Google Scholar] [CrossRef] [PubMed]
- Kendall, C.; Isabelle, M.; Bazant-Hegemark, F.; Hutchings, J.; Orr, L.; Babrah, J.; Baker, R.; Stone, N. Vibrational spectroscopy: A clinical tool for cancer diagnostics. Analyst 2009, 134, 1029–1045. [Google Scholar] [CrossRef] [PubMed]
- Blout, E.R.; Mellors, R.C. Infrared Spectra of Tissues. Science 1949, 110, 137–138. [Google Scholar] [CrossRef] [PubMed]
- Nasse, M.J.; Walsh, M.J.; Mattson, E.C.; Reininger, R.; Kajdacsy-Balla, A.; Macias, V.; Bhargava, R.; Hirschmugl, C.J. High-resolution Fourier-transform infrared chemical imaging with multiple synchrotron beams. Nat. Methods 2011, 8, 413–416. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Menzies, G.E.; Fox, H.R.; Marnane, C.; Pope, L.; Prabhu, V.; Winter, S.; Derrick, A.V.; Lewis, P.D. Fourier transform infrared for noninvasive optical diagnosis of oral, oropharyngeal, and laryngeal cancer. Transl. Res. 2014, 163, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.; Pal, M.; Chakrabarty, J.; Petibois, C.; Paul, R.R.; Giri, A.; Chatterjee, J. Fourier-transform-infrared-spectroscopy based spectral-biomarker selection towards optimum diagnostic differentiation of oral leukoplakia and cancer. Anal. Bioanal. Chem. 2015, 407, 7935–7943. [Google Scholar] [CrossRef]
- Ukkonen, H.; Kumar, S.; Mikkonen, J.; Salo, T.; Singh, S.P.; Koistinen, A.; Goormaghtigh, E.; Kullaa, A.M. Changes in the microenvironment of invading melanoma and carcinoma cells identified by FTIR imaging. Vib. Spectrosc. 2015, 79, 24–30. [Google Scholar] [CrossRef]
- Benard, A.; Desmedt, C.; Smolina, M.; Szternfeld, P.; Verdonck, M.; Rouas, G.; Kheddoumi, N.; Rothe, F.; Larsimont, D.; Sotiriou, C.; et al. Infrared imaging in breast cancer: Automated tissue component recognition and spectral characterization of breast cancer cells as well as the tumor microenvironment. Analyst 2014, 139, 1044–1056. [Google Scholar] [CrossRef]
- Kyriakidou, M.; Mavrogenis, A.F.; Kyriazis, S.; Markouizou, A.; Theophanides, T.; Anastassopoulou, J. An FT-IR Spectral Analysis of the Effects of gamma-Radiation on Normal and Cancerous Cartilage. In Vivo 2016, 30, 599–604. [Google Scholar]
- Pallua, J.D.; Pezzei, C.; Zelger, B.; Schaefer, G.; Bittner, L.K.; Huck-Pezzei, V.A.; Schoenbichler, S.A.; Hahn, H.; Kloss-Brandstaetter, A.; Kloss, F.; et al. Fourier transform infrared imaging analysis in discrimination studies of squamous cell carcinoma. Analyst 2012, 137, 3965–3974. [Google Scholar] [CrossRef] [PubMed]
- Slaughter, D.P.; Southwick, H.W.; Smejkal, W. Field cancerization in oral stratified squamous epithelium; clinical implications of multicentric origin. Cancer 1953, 6, 963–968. [Google Scholar] [CrossRef]
- Dotto, G.P. Multifocal epithelial tumors and field cancerization: Stroma as a primary determinant. J. Clin. Investig. 2014, 124, 1446–1453. [Google Scholar] [CrossRef] [PubMed]
- Mohan, M.; Jagannathan, N. Oral field cancerization: An update on current concepts. Oncol. Rev. 2014, 8, 244. [Google Scholar] [CrossRef] [PubMed]
- Sundaram, J.; Park, B.; Hinton, A., Jr.; Yoon, S.C.; Windham, W.R.; Lawrence, K.C. Classification and structural analysis of live and dead Salmonella cells using Fourier transform infrared spectroscopy and principal component analysis. J. Agric. Food Chem. 2012, 60, 991–1004. [Google Scholar] [CrossRef] [PubMed]
- Paidi, S.K.; Rizwan, A.; Zheng, C.; Cheng, M.; Glunde, K.; Barman, I. Label-Free Raman Spectroscopy Detects Stromal Adaptations in Premetastatic Lungs Primed by Breast Cancer. Cancer Res. 2017, 77, 247–256. [Google Scholar] [CrossRef] [PubMed]
- Winnard, P.T., Jr.; Zhang, C.; Vesuna, F.; Kang, J.W.; Garry, J.; Dasari, R.R.; Barman, I.; Raman, V. Organ-specific isogenic metastatic breast cancer cell lines exhibit distinct Raman spectral signatures and metabolomes. Oncotarget 2017, 8, 20266–20287. [Google Scholar] [CrossRef]
| Patients | Number (M/F) | Age (Mean; SD) |
|---|---|---|
| Healthy control | 11 (7/4) | 33–74 (59.9; 11.2) years |
| Non-Irradiated control | 5 (4/1) | 57–80 (65.8; 8.6) years |
| Irradiated | 9 (5/4) | 58–82 (70.4; 8.2) years |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ukkonen, H.; Vuokila, S.; Mikkonen, J.J.W.; Dekker, H.; Schulten, E.A.J.M.; Bloemena, E.; Koistinen, A.; Valdez, T.A.; Kullaa, A.M.; Singh, S.P. Biochemical Changes in Irradiated Oral Mucosa: A FTIR Spectroscopic Study. Biosensors 2019, 9, 12. https://doi.org/10.3390/bios9010012
Ukkonen H, Vuokila S, Mikkonen JJW, Dekker H, Schulten EAJM, Bloemena E, Koistinen A, Valdez TA, Kullaa AM, Singh SP. Biochemical Changes in Irradiated Oral Mucosa: A FTIR Spectroscopic Study. Biosensors. 2019; 9(1):12. https://doi.org/10.3390/bios9010012
Chicago/Turabian StyleUkkonen, Helena, Simo Vuokila, Jopi J. W. Mikkonen, Hannah Dekker, Engelbert A. J. M. Schulten, Elisabeth Bloemena, Arto Koistinen, Tulio A. Valdez, Arja M. Kullaa, and Surya Pratap Singh. 2019. "Biochemical Changes in Irradiated Oral Mucosa: A FTIR Spectroscopic Study" Biosensors 9, no. 1: 12. https://doi.org/10.3390/bios9010012
APA StyleUkkonen, H., Vuokila, S., Mikkonen, J. J. W., Dekker, H., Schulten, E. A. J. M., Bloemena, E., Koistinen, A., Valdez, T. A., Kullaa, A. M., & Singh, S. P. (2019). Biochemical Changes in Irradiated Oral Mucosa: A FTIR Spectroscopic Study. Biosensors, 9(1), 12. https://doi.org/10.3390/bios9010012






