Label-Free Electrochemical Immunoassay for C-Reactive Protein
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents and Chemicals
2.2. CRP Measurements in Blood Serum
2.3. Instrumentation
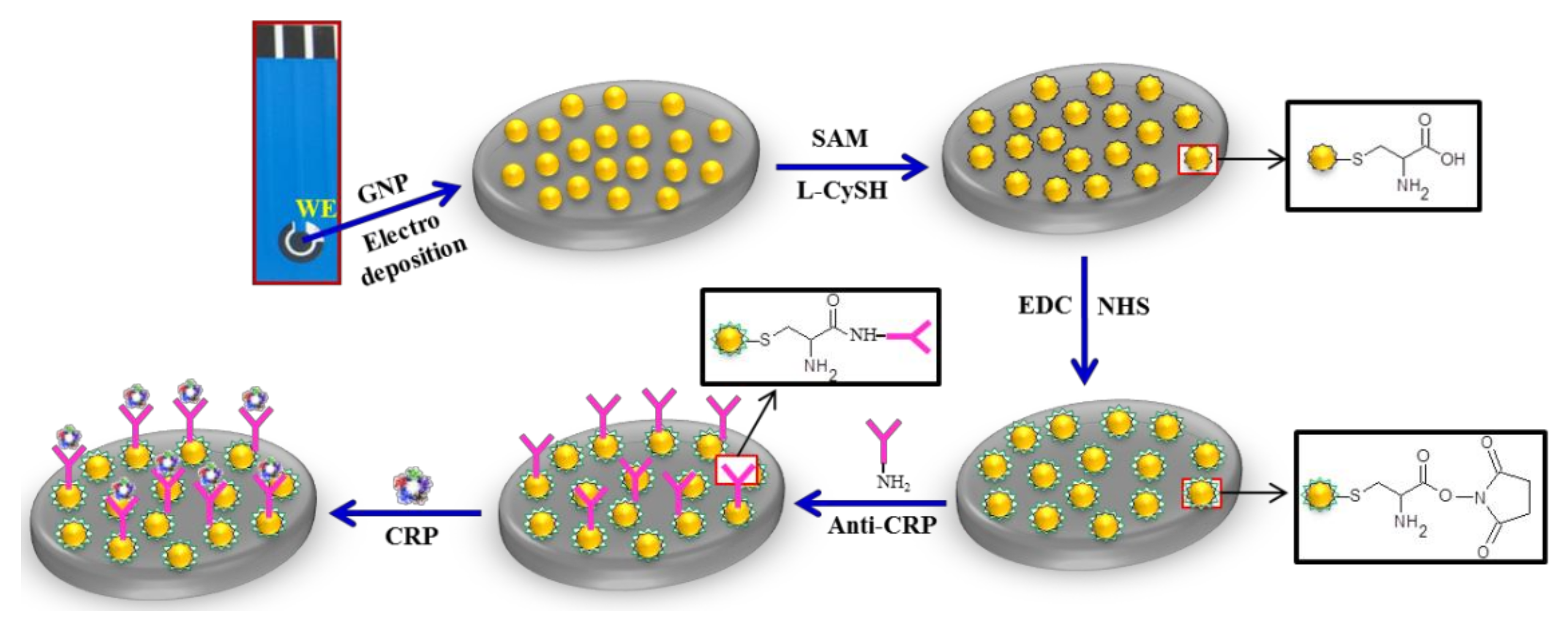
2.4. Immunosensor Fabrication
2.5. Immunoreaction of CRP with Anti-CRP
3. Results and Discussion
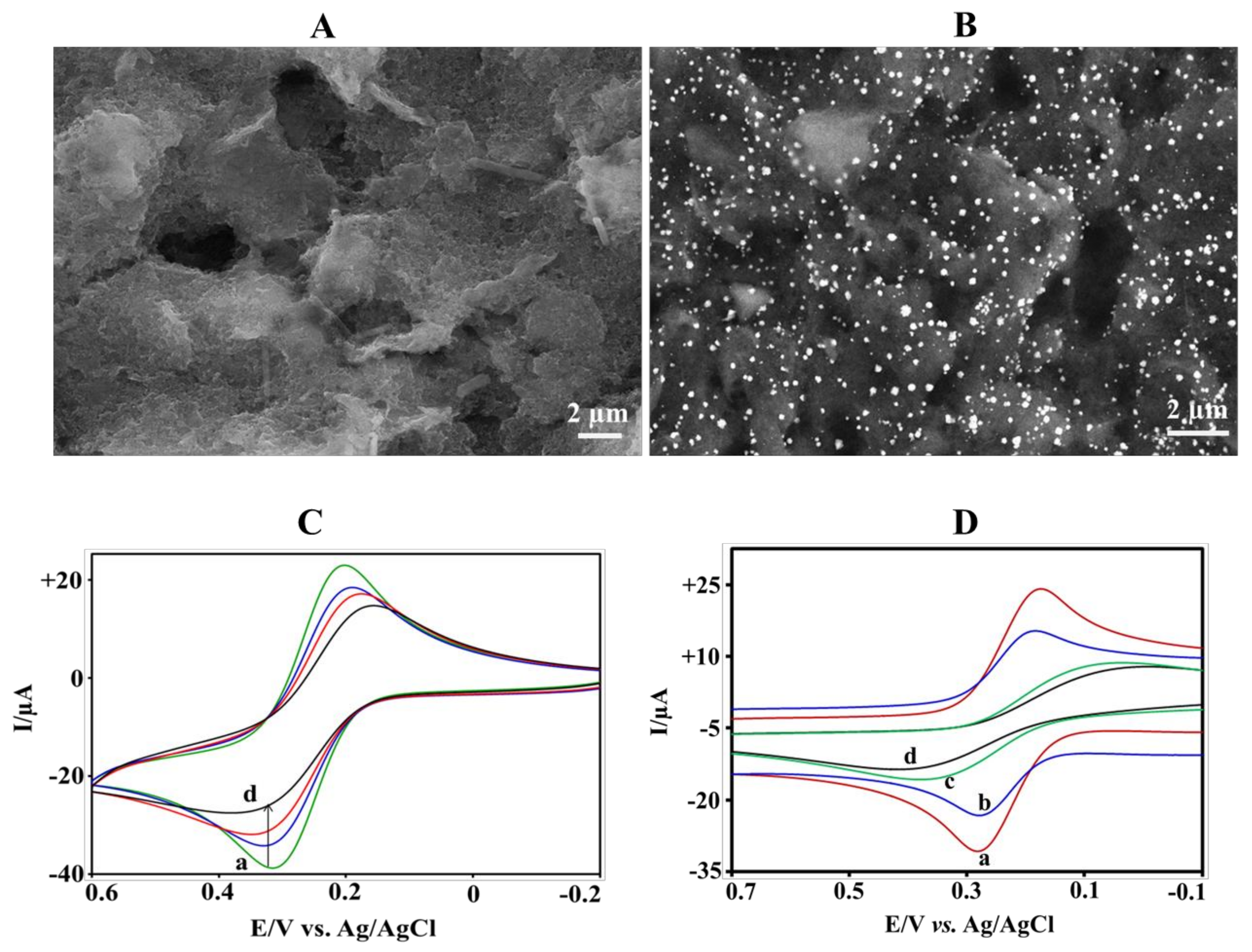
3.1. Morphological Characterization
3.2. Electrochemical Characterization
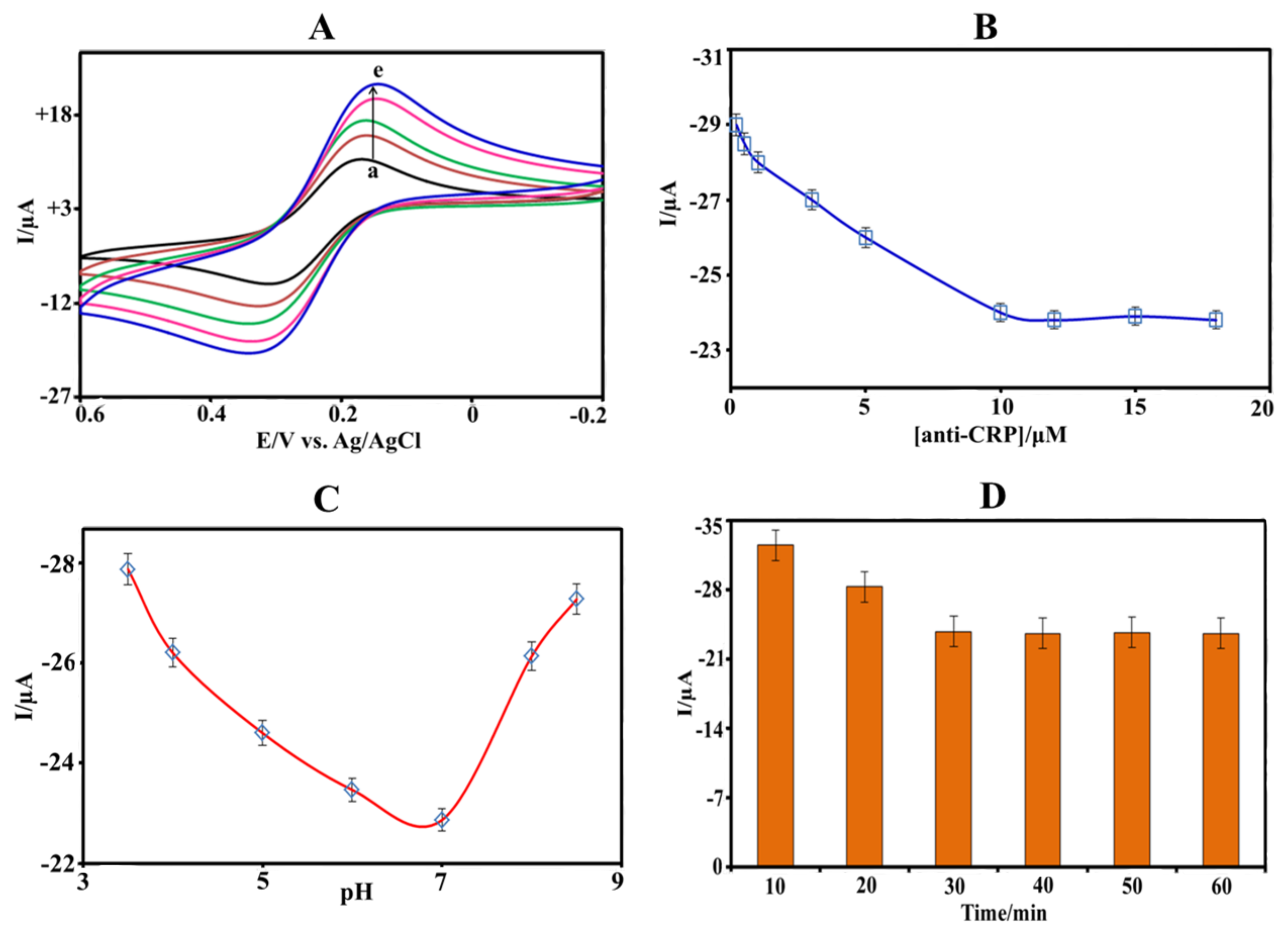
3.3. Optimization
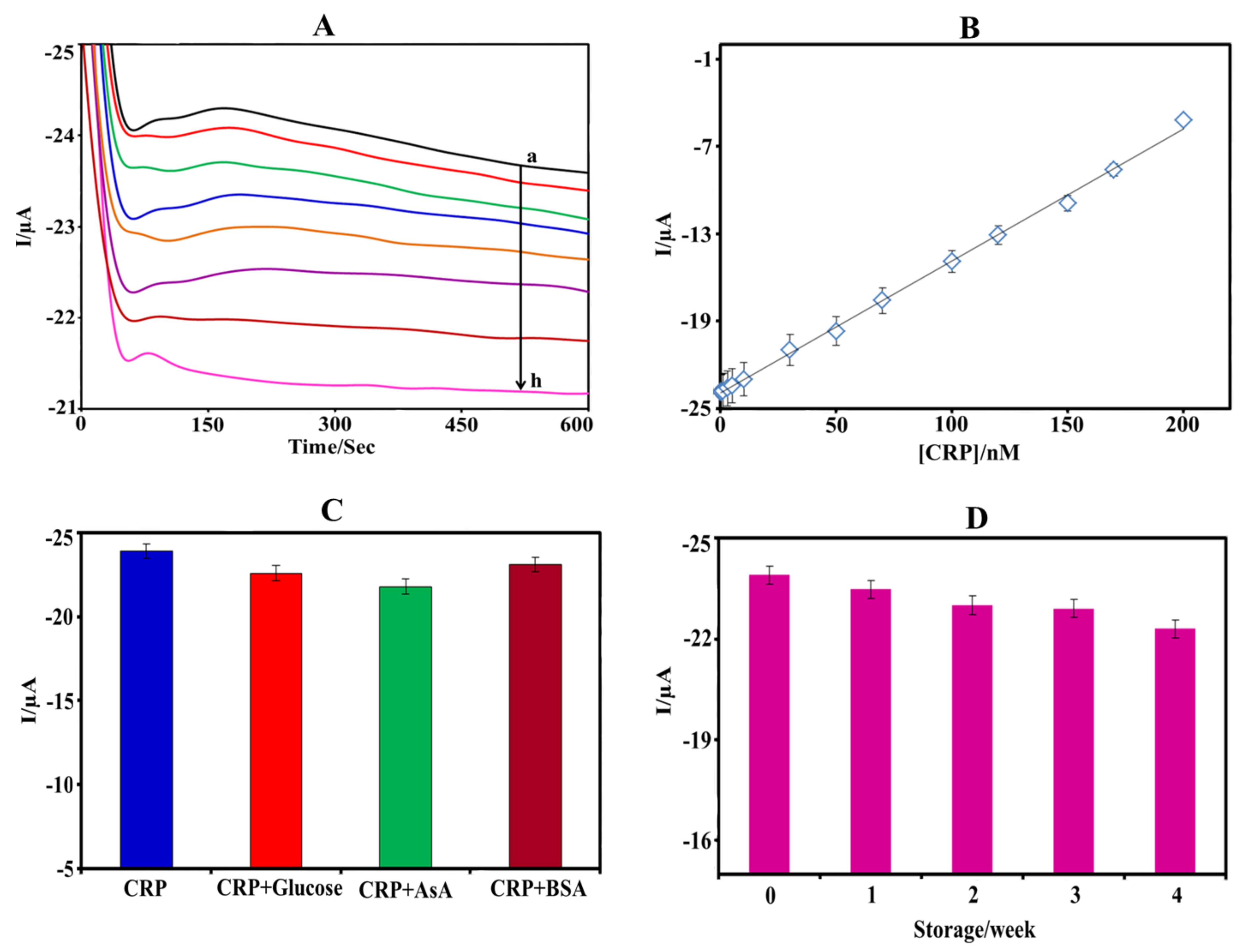
3.4. CRP Measurement
3.5. Selectivity, Stability, and Reproducibility
3.6. Real Sample Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Heinecke, J.W. Chemical knockout of C-reactive protein in cardiovascular disease. Nat. Chem. Biol. 2006, 2, 300–301. [Google Scholar] [CrossRef] [PubMed]
- Pepys, M.B.; Hirschfield, G.M.; Tennent, G.A.; Ruth Gallimore, J.; Kahan, M.C.; Bellotti, V.; Hawkins, P.N.; Myers, R.M.; Smith, M.D.; Polara, A.; et al. Targeting C-reactive protein for the treatment of cardiovascular disease. Nature 2006, 440, 1217–1221. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M. Clinical application of C-reactive protein for cardiovascular disease detection and prevention. Circulation 2003, 107, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M. C-reactive protein: A simple test to help predict risk of heart attack and stroke. Circulation 2003, 108, 81–85. [Google Scholar] [CrossRef] [PubMed]
- Espe, K.; Galler, A.; Raila, J.; Kiess, W.; Schweigert, F. High-normal C-reactive protein levels do not affect the vitamin A transport complex in serum of children and adolescents with type 1 diabetes. Pediatr. Res. 2007, 62, 741–745. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Luo, Y.; Zhang, B.; Chen, M.; Jiang, T.; Zhou, D.; Huang, J.; Fu, W. Sensitive and rapid quantification of C-reactive protein using quantum dot-labeled microplate immunoassay. J. Transl. Med. 2012, 10, 24. [Google Scholar] [CrossRef] [PubMed]
- Ducloux, D.; Bresson-Vautrin, C.; Kribs, M.; Abdelfatah, A.; Chalopin, J.-M. C-reactive protein and cardiovascular disease in peritoneal dialysis patients. Kidney Int. 2002, 62, 1417–1422. [Google Scholar] [CrossRef] [PubMed]
- Kitayama, Y.; Takeuchi, T. Localized Surface Plasmon Resonance Nanosensing of C-Reactive Protein with Poly(2-methacryloyloxyethyl phosphorylcholine)-Grafted Gold Nanoparticles Prepared by Surface-Initiated Atom Transfer Radical Polymerization. Anal. Chem. 2014, 86, 5587–5594. [Google Scholar] [CrossRef]
- Vance, S.A.; Sandros, M.G. Zeptomole Detection of C-Reactive Protein in Serum by a Nanoparticle Amplified Surface Plasmon Resonance Imaging Aptasensor. Sci. Rep. 2014, 4, 5129. [Google Scholar] [CrossRef] [PubMed]
- Yen, Y.K.; Lai, Y.C.; Hong, W.T.; Pheanpanitporn, Y.; Chen, C.S.; Huang, L.S. Electrical detection of C-reactive protein using a single free-standing, thermally controlled piezoresistive microcantilever for highly reproducible and accurate measurements. Sensors 2013, 13, 9653–9668. [Google Scholar] [CrossRef] [PubMed]
- Kim, N.; Kim, D.-K.; Cho, Y.-J. Development of indirect-competitive quartz crystal microbalance immunosensor for C-reactive protein. Sens. Actuators B Chem. 2009, 143, 444–448. [Google Scholar] [CrossRef]
- McBride, J.D.; Cooper, M.A. A high sensitivity assay for the inflammatory marker C-Reactive protein employing acoustic biosensing. J. Nanobiotechnol. 2008, 6, 5. [Google Scholar] [CrossRef] [PubMed]
- Hennessey, H.; Afara, N.; Omanovic, S.; Padjen, A.L. Electrochemical investigations of the interaction of C-reactive protein (CRP) with a CRP antibody chemically immobilized on a gold surface. Anal. Chim. Acta 2009, 643, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Buch, M.; Rishpon, J. An Electrochemical Immunosensor for C-Reactive Protein Based on Multi-Walled Carbon Nanotube-Modified Electrodes. Electroanalysis 2008, 20, 2592–2594. [Google Scholar] [CrossRef]
- Hays, H.C.W.; Millner, P.A.; Prodromidis, M.I. Development of capacitance based immunosensors on mixed self-assembled monolayers. Sens. Actuators B Chem. 2006, 114, 1064–1070. [Google Scholar] [CrossRef]
- Dawan, S.; Kanatharana, P.; Wongkittisuksa, B.; Limbut, W.; Numnuam, A.; Limsakul, C.; Thavarungkul, P. Label-free capacitive immunosensors for ultra-trace detection based on the increase of immobilized antibodies on silver nanoparticles. Anal. Chim. Acta 2011, 699, 232–241. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Jiang, J.; Shen, G.; Yu, R. A label-free electrochemical impedance immunosensor for the sensitive detection of aflatoxin B1. Anal. Methods 2015, 7, 2354–2359. [Google Scholar] [CrossRef]
- Justino, C.I.L.; Freitas, A.C.; Amaral, J.P.; Rocha-Santos, T.A.P.; Cardoso, S.; Duarte, A.C. Disposable immunosensors for C-reactive protein based on carbon nanotubes field effect transistors. Talanta 2013, 108, 165–170. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Javadi, A.; Gong, S. Sensitive electrochemical immunosensor for the detection of cancer biomarker using quantum dot functionalized graphene sheets as labels. Sens. Actuators B Chem. 2011, 155, 357–360. [Google Scholar] [CrossRef]
- Mao, K.; Wu, D.; Li, Y.; Ma, H.; Ni, Z.; Yu, H.; Luo, C.; Wei, Q.; Du, B. Label-free electrochemical immunosensor based on graphene/methylene blue nanocomposite. Anal. Biochem. 2012, 422, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Madasamy, T.; Pandiaraj, M.; Balamurugan, M.; Karnewar, S.; Benjamin, A.R.; Venkatesh, K.A.; Vairamani, K.; Kotamraju, S.; Karunakaran, C. Virtual electrochemical nitric oxide analyzer using copper, zinc superoxide dismutase immobilized on carbon nanotubes in polypyrrole matrix. Talanta 2012, 100, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Madasamy, T.; Pandiaraj, M.; Kanugula, A.K.; Rajesh, S.; Bhargava, K.; Sethy, N.K.; Kotamraju, S.; Karunakaran, C. Gold Nanoparticles with Self-Assembled Cysteine Monolayer Coupled to Nitrate Reductase in Polypyrrole Matrix Enhanced Nitrate Biosensor. Adv. Chem. Lett. 2013, 1, 2–9. [Google Scholar] [CrossRef]
- Pandiaraj, M.; Madasamy, T.; Gollavilli, P.N.; Balamurugan, M.; Kotamraju, S.; Rao, V.K.; Bhargava, K.; Karunakaran, C. Nanomaterial-based electrochemical biosensors for cytochrome c using cytochrome c reductase. Bioelectrochemistry 2013, 91, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Madasamy, T.; Pandiaraj, M.; Balamurugan, M.; Bhargava, K.; Sethy, N.K.; Karunakaran, C. Copper, zinc superoxide dismutase and nitrate reductase coimmobilized bienzymatic biosensor for the simultaneous determination of nitrite and nitrate. Biosens. Bioelectron. 2014, 52, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.-J.; Xu, J.-Z.; He, J.-T.; Wang, Y.-J.; Miao, Q.; Chen, H.-Y. An Electrochemical Immunosensor for Assays of C-Reactive Protein. Anal. Lett. 2003, 36, 1547–1556. [Google Scholar] [CrossRef]
- Bryan, T.; Luo, X.; Bueno, P.R.; Davis, J.J. An optimised electrochemical biosensor for the label-free detection of C-reactive protein in blood. Biosens. Bioelectron. 2013, 39, 94–98. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.K.; Periyakaruppan, A.; Meyyappan, M.; Koehne, J.E. Label-free detection of C-reactive protein using a carbon nanofiber based biosensor. Biosens. Bioelectron. 2014, 59, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Centi, S.; Bonel Sanmartin, L.; Tombelli, S.; Palchetti, I.; Mascini, M. Detection of C Reactive Protein (CRP) in Serum by an Electrochemical Aptamer-Based Sandwich Assay. Electroanalysis 2009, 21, 1309–1315. [Google Scholar] [CrossRef]
- Fakanya, W.M.; Tothill, I.E. Detection of the Inflammation Biomarker C-Reactive Protein in Serum Samples: Towards an Optimal Biosensor Formula. Biosensors 2014, 4, 340–357. [Google Scholar] [CrossRef] [PubMed]
- Kumar, D.; Prasad, B.B. Multiwalled carbon nanotubes embedded molecularly imprinted polymer-modified screen printed carbon electrode for the quantitative analysis of C-reactive protein. Sens. Actuators B Chem. 2012, 171–172, 1141–1150. [Google Scholar] [CrossRef]
- Madasamy, T.; Santschi, C.; Martin, O.J.F. A miniaturized electrochemical assay for homocysteine using screen-printed electrodes with cytochrome c anchored gold nanoparticles. Analyst 2015, 140, 6071–6078. [Google Scholar] [CrossRef] [PubMed]
- Bard, A.J.; Faulkner, L.R. Electrochemical Methods: Fundamentals and Applications; Wiley: Hoboken, NJ, USA, 2000; ISBN 9780471043720. [Google Scholar]
- Thangamuthu, M.; Santschi, C.; Martin, O. Electrochemical Optimization of Gold Nanoparticles for Efficient Electron TransferImplication for Highly Sensitive Biosensing. Sens. Lett. 2017, 15, 126–131. [Google Scholar] [CrossRef]
- Khopkar, S.M. Basic Concepts of Analytical Chemistry; New Academic Science: England, UK, 2008; ISBN 9781906574000. [Google Scholar]
- De Ávila, B.E.-F.; Escamilla-Gómez, V.; Campuzano, S.; Pedrero, M.; Salvador, J.-P.; Marco, M.-P.; Pingarrón, J.M. Ultrasensitive amperometric magnetoimmunosensor for human C-reactive protein quantification in serum. Sens. Actuators B Chem. 2013, 188, 212–220. [Google Scholar] [CrossRef]
- Zeng, S.L.; Zhou, H.K.; Gan, N.; Cao, Y.T. A Renewable C Reactive Protein Amperometric Immunosensor Based on Magnetic Multiwalled Carbon Nanotubes Probles Modified Electrode. Appl. Mech. Mater. 2011, 80, 452–456. [Google Scholar] [CrossRef]
- Zhou, F.; Lu, M.; Wang, W.; Bian, Z.-P.; Zhang, J.-R.; Zhu, J.-J. Electrochemical Immunosensor for Simultaneous Detection of Dual Cardiac Markers Based on a Poly(Dimethylsiloxane)-Gold Nanoparticles Composite Microfluidic Chip: A Proof of Principle. Clin. Chem. 2010, 56, 1701–1707. [Google Scholar] [CrossRef] [PubMed]
| Immunosensor | Linearity (nM) | Sensitivity (nA nM−1 cm−2) | Detection Limit (nM) | Reference |
|---|---|---|---|---|
| antiCRP-SAM-Au | 0.5–50 | - | 0.176 | [26] |
| antiCRP-VACNFs-SiO2 | 0.42–42 | - | 0.090 | [27] |
| antiCRP-SAM-Au | 0.097–9.74 | - | 0.050 | [13] |
| antiCRP-SWCNT-FET | 0.003–0.847 | - | 0.003 | [18] |
| antiCRP-SAM-AuNPs-SPE | 0.4–200 | 90.7 | 0.150 | Present work |
| Analyte | Selectivity (%) |
|---|---|
| CRP | 100 |
| CRP + Glucose | 93 |
| CRP + Ascorbic acid | 91 |
| CRP + Bovine serum albumin | 96 |
| Sample No. | CRP Added (nM) | CRP Measured (nM) | Recovery (%) | RSD (%) |
|---|---|---|---|---|
| 1 | 5 | 5.05 | 101.2 | 1.9 |
| 2 | 10 | 9.36 | 93.60 | 2.3 |
| 3 | 15 | 14.14 | 94.30 | 1.4 |
| 4 | 20 | 20.64 | 103.2 | 2.7 |
| 5 | 25 | 24.45 | 97.80 | 1.8 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thangamuthu, M.; Santschi, C.; J. F. Martin, O. Label-Free Electrochemical Immunoassay for C-Reactive Protein. Biosensors 2018, 8, 34. https://doi.org/10.3390/bios8020034
Thangamuthu M, Santschi C, J. F. Martin O. Label-Free Electrochemical Immunoassay for C-Reactive Protein. Biosensors. 2018; 8(2):34. https://doi.org/10.3390/bios8020034
Chicago/Turabian StyleThangamuthu, Madasamy, Christian Santschi, and Olivier J. F. Martin. 2018. "Label-Free Electrochemical Immunoassay for C-Reactive Protein" Biosensors 8, no. 2: 34. https://doi.org/10.3390/bios8020034
APA StyleThangamuthu, M., Santschi, C., & J. F. Martin, O. (2018). Label-Free Electrochemical Immunoassay for C-Reactive Protein. Biosensors, 8(2), 34. https://doi.org/10.3390/bios8020034





