Biosensors for the Detection of Antibiotics in Poultry Industry—A Review
Abstract
:1. Introduction
Antibiotics and Food Safety
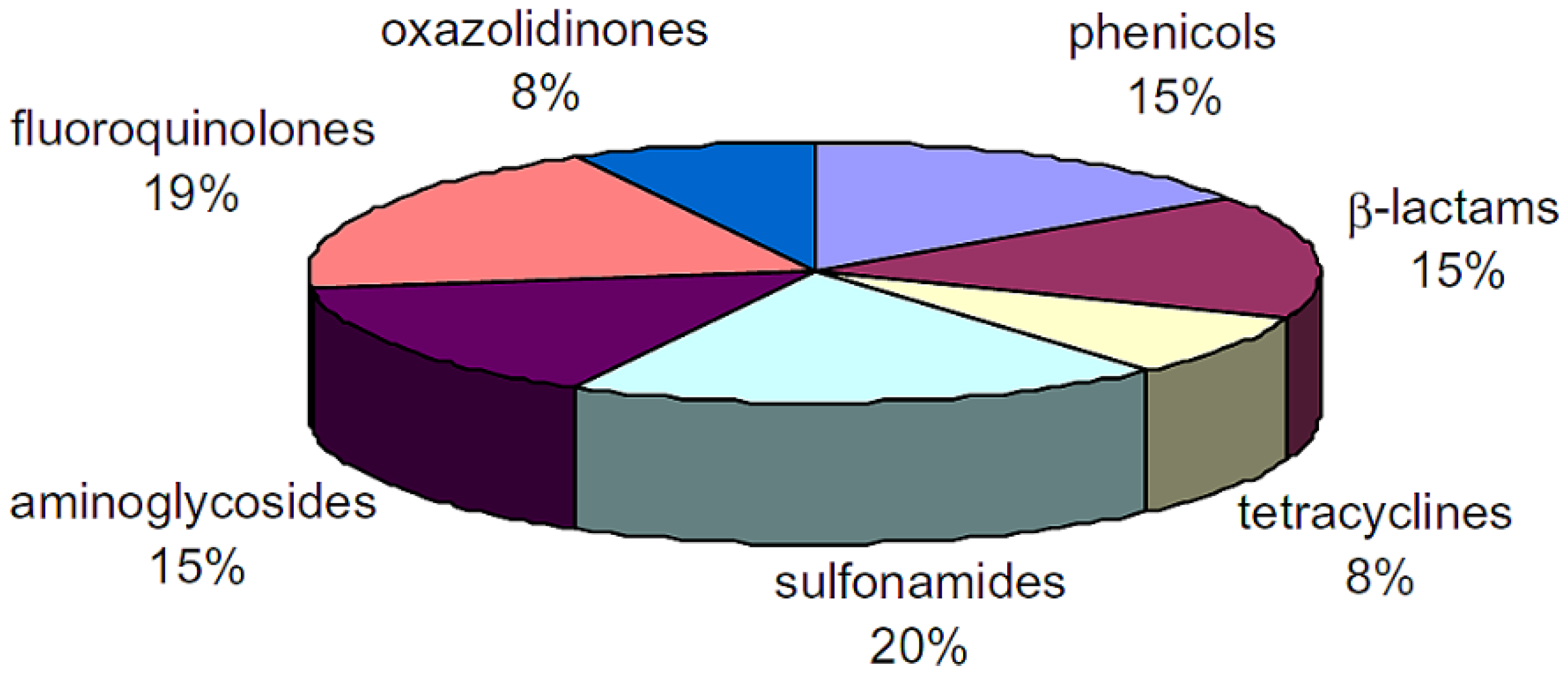
2. Antibiotics Detection Techniques
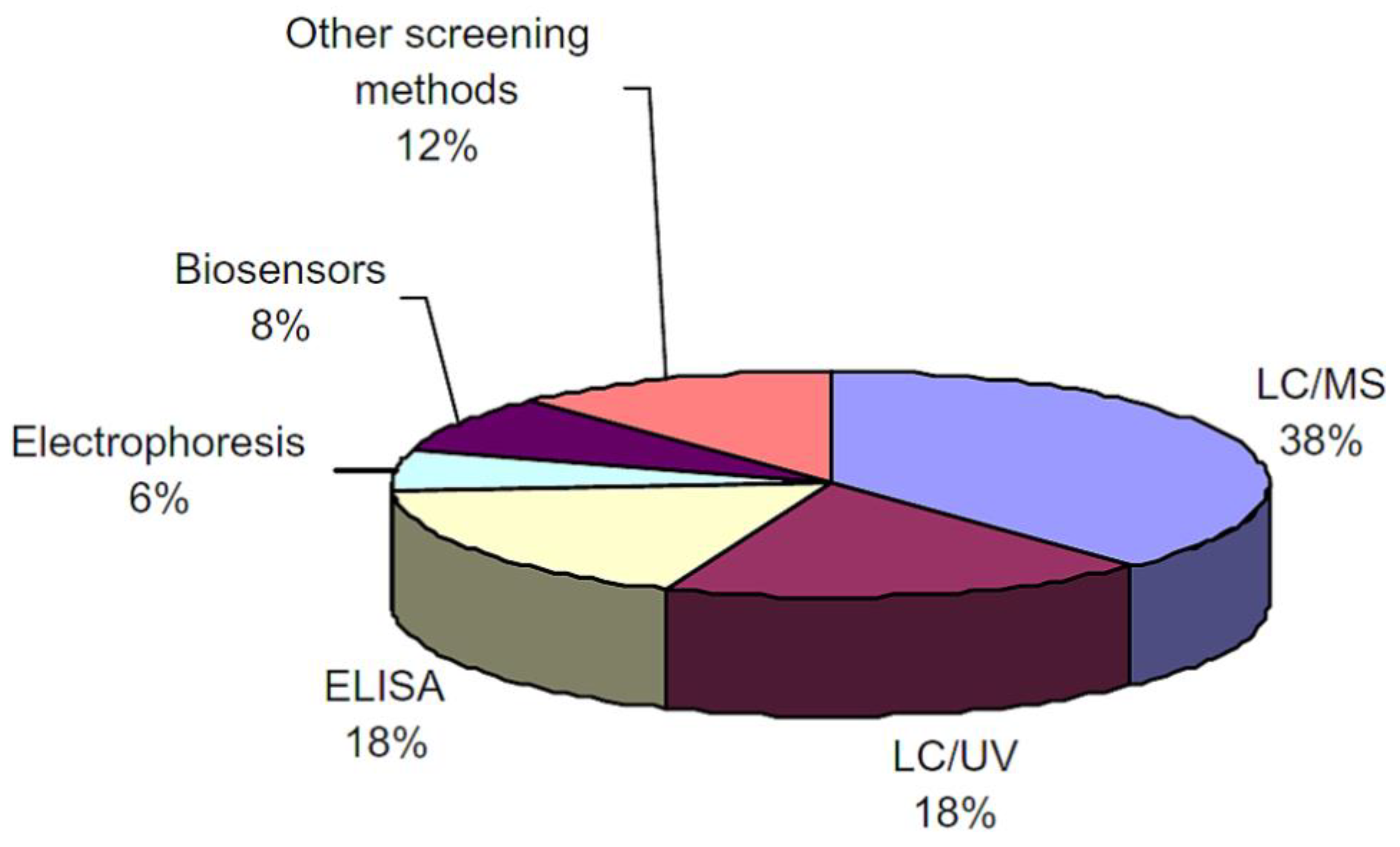
| Detection Method | Principle | Advantages | Drawback |
|---|---|---|---|
| Enzyme-Linked Immunosorbent Assay (ELISA) |
| ||
| Biosensors |
|
| |
| Liquid Chromatography-Mass Spectrometry (LC-MS) | The LC-MS coupling is an effective system whereby the mass spectrometry component functions by transforming the ionised (charged) state of molecules using the mass-to-charge ratio. There are several methods of LC-MS, including the electrospray ionisation source, direct injection methods and mobile phase [14]. | Highly sensitive and able to handle complex mixtures [14] |
3. Overview of Biosensors
3.1. Definition of Biosensors
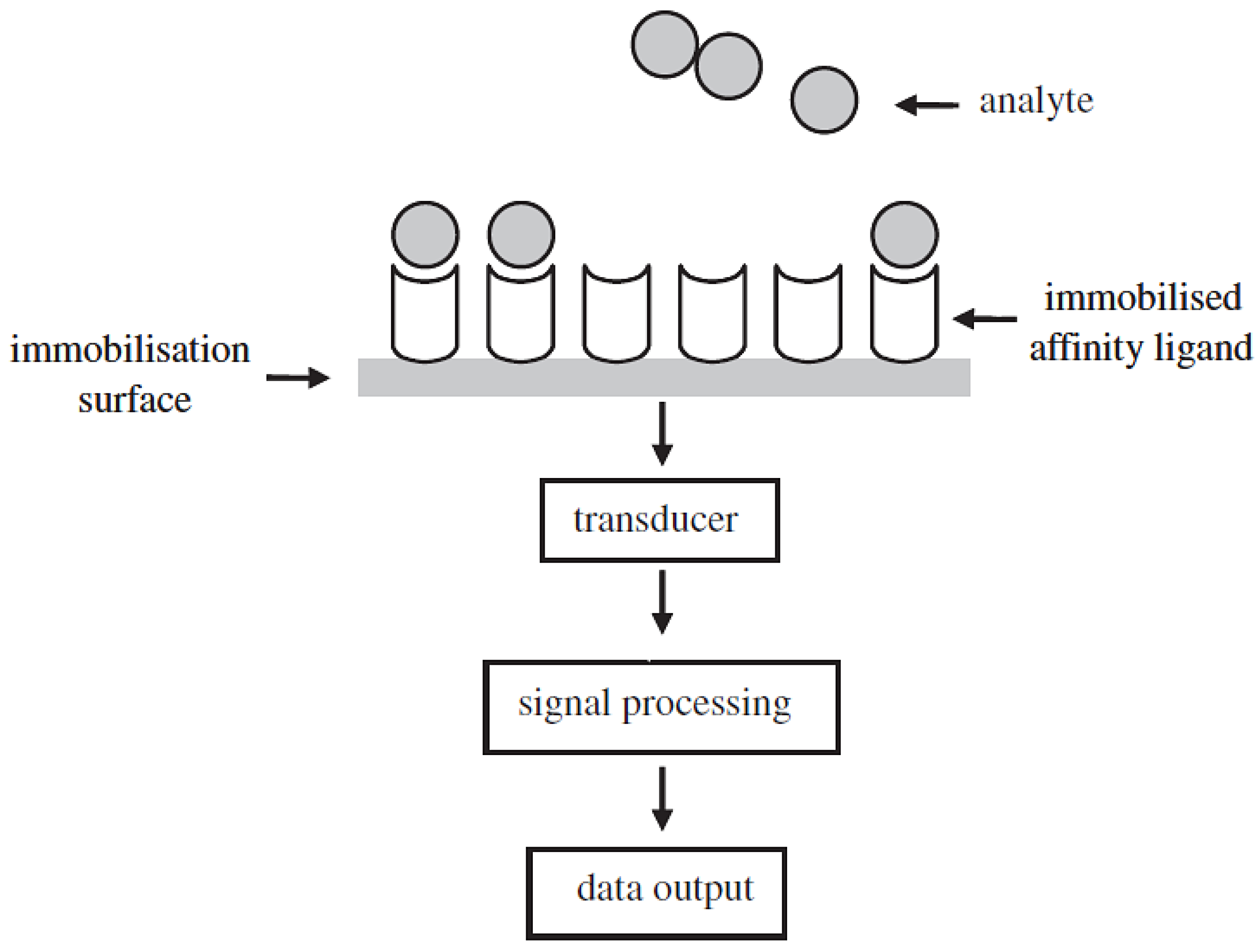
3.2. Classification of Biosensors

4. Types of Biosensors
4.1. Electrochemical Biosensors
- ▪
- Potentiometric-based biosensors sense changes in pH and ion concentrations upon the antigen/antibody interaction.
- ▪
- Amperometric-based biosensors sense the difference in current potentials during redox reactions when antigen/antibody pairing occurs.
- ▪
- Conductometric/capacitive biosensors sense the change of electrical charge in a solution under constant voltage. This method is not recommended because of poor signal-to-noise ratio.
- ▪
- Impedimetric-based biosensors sense changes in impedances upon antigen/antibody interaction.
4.2. Mass-Based Biosensors
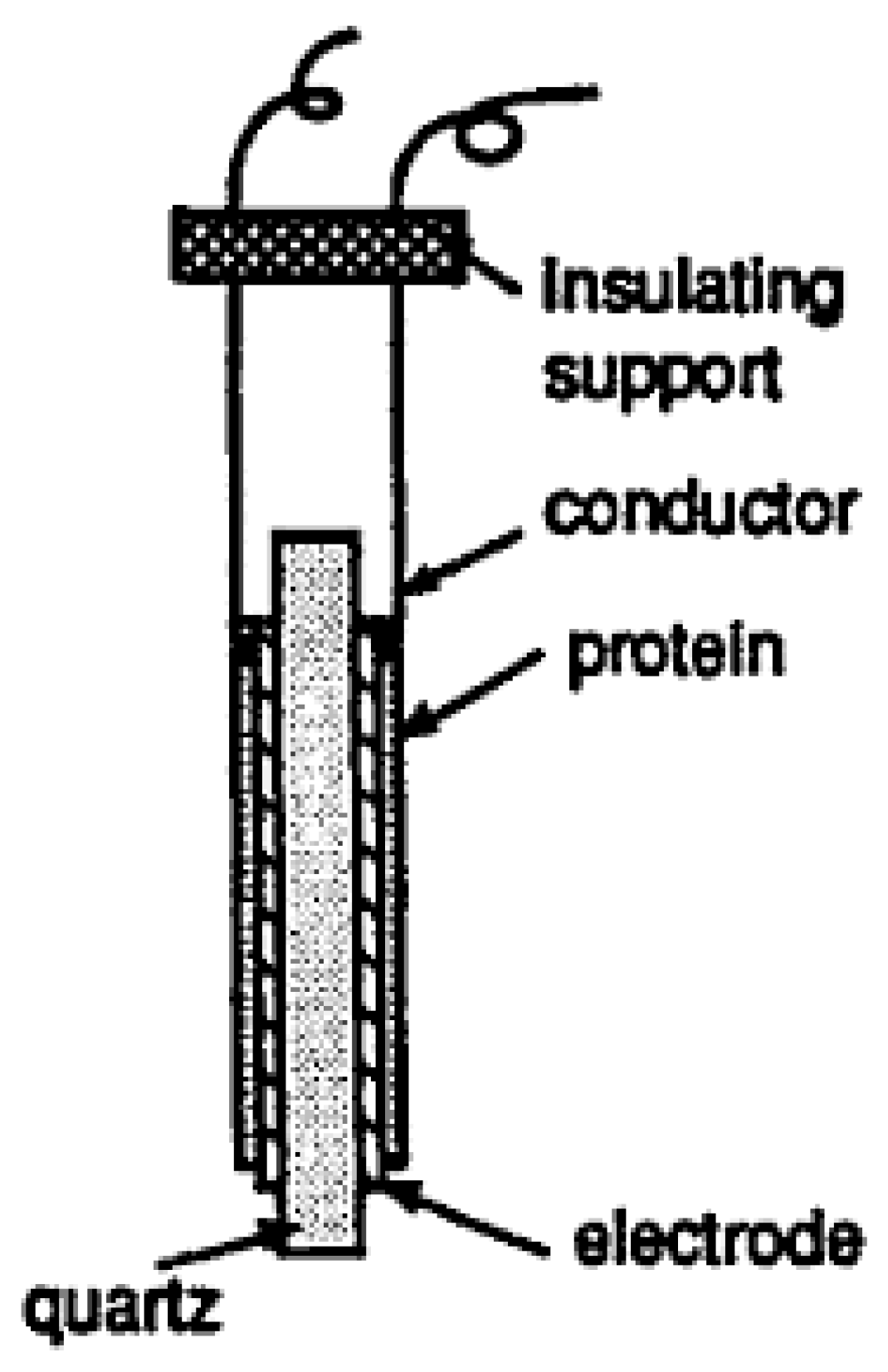
4.3. Surface-Enhanced Raman Scattering (SERS)

4.4. Optics Biosensors
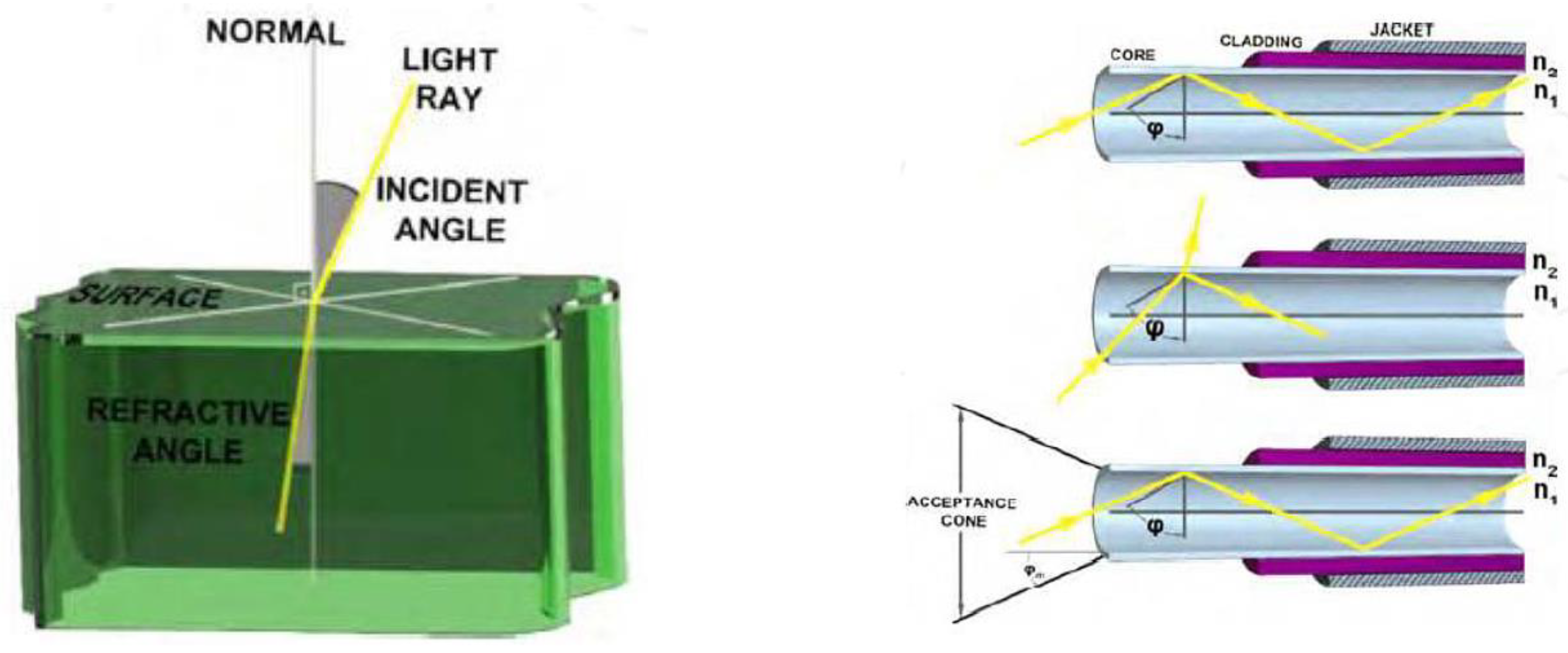
4.4.1. Absorbance Fibre-Optic Biosensors
- A = Optical Absorbance.
- Io = Incident light intensity.
- I = Transmitted light intensity.
- є = Molar absorption coefficient.
4.4.2. Fluorescence Fibre-Optic Biosensors
4.4.3. Luminescence Fibre-Optic Biosensors
ATP Bioluminescence
Bacterial Bioluminescence

5. Design Strategies of Fluorescent Biosensors
Auto-Fluorescent Protein (AFP)-Based Biosensors
Analyte-Sensitive Sensors
Conformation-Sensitive Sensors
6. Biosensor Recognition

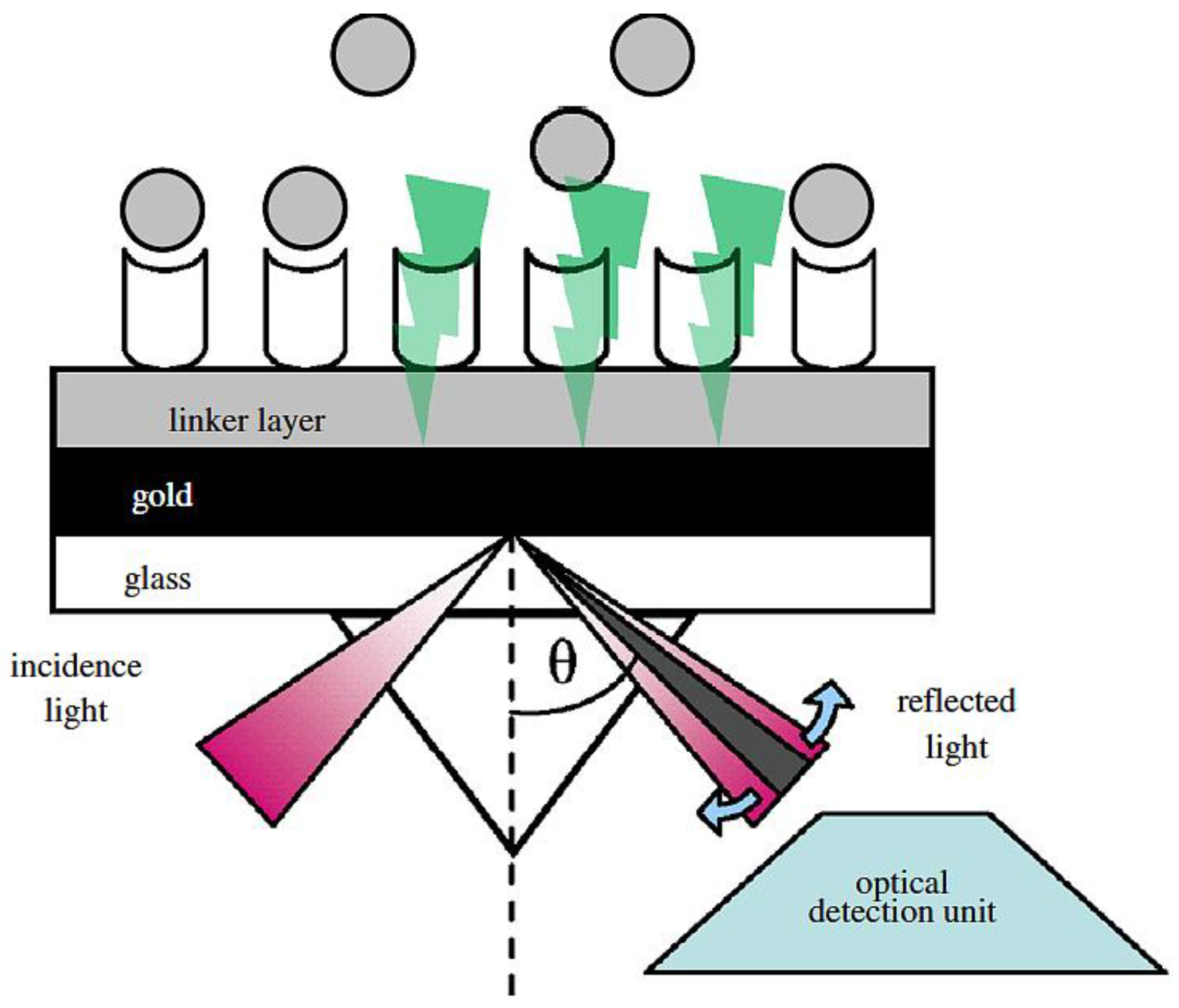
7. Biosensor Transducing Element: Surface Plasmon Resonance (SPR)
7.1. SPR Transducing Recognition
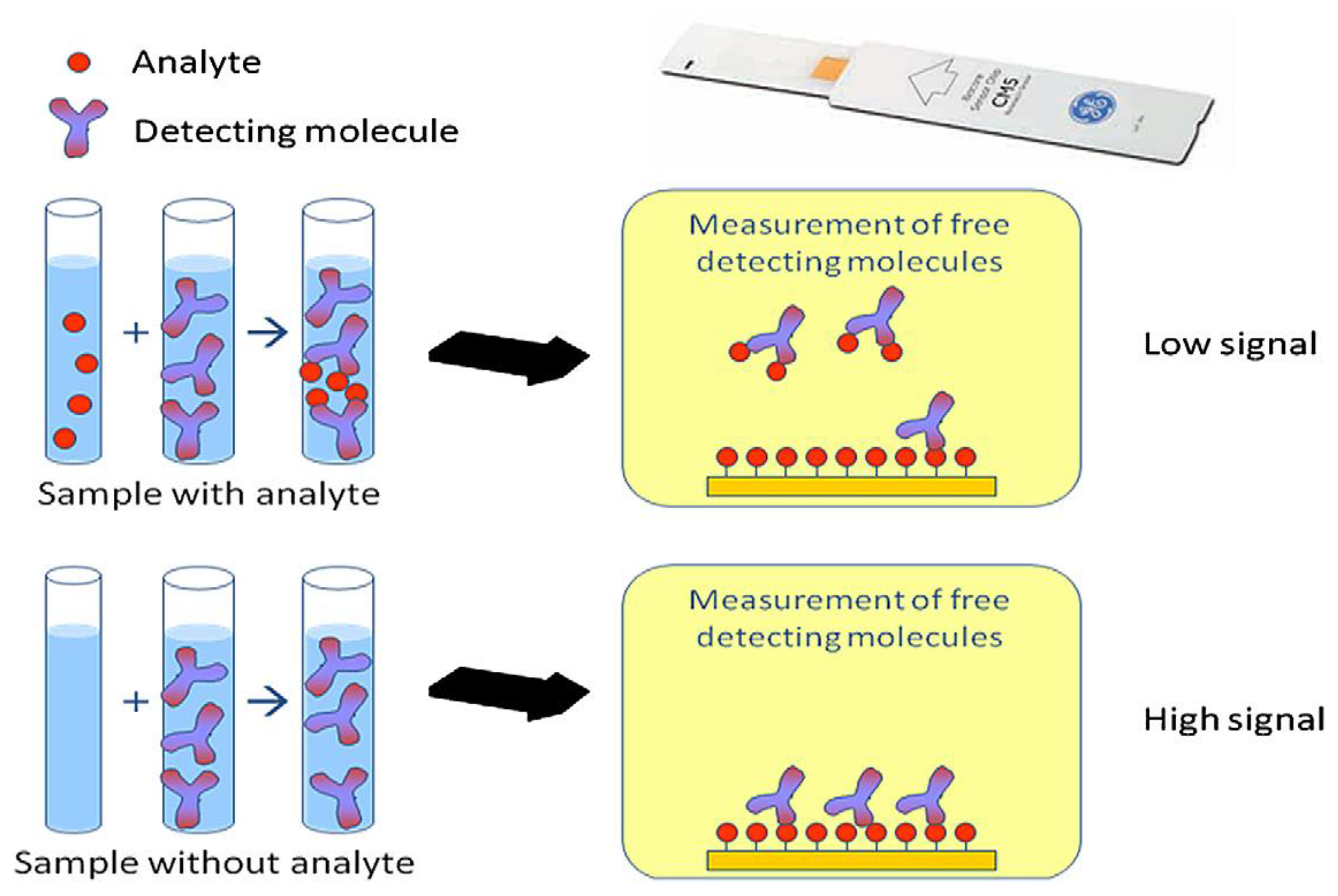
7.2. Biorecognition Element of SPR
| Antibiotics | Matrix | Biosensor Type | LOD (Limit Of detection) | Sample Pre-Treatment & Assay time | References |
|---|---|---|---|---|---|
| Aminoglycosides: Streptomycin, dihydrostreptomycin | Pig muscle | Optical biosensor—SPR | 70 ng/g muscle | Buffer + adjustment pH, kidney and muscle: 3% trichloroacetic acid or 3.6% HClO4 or 0.2 M Na2HPO4 at pH 9.2 4 h at 45 °C in tube (Accusphere test) | [25] |
| Quinolones: Norfloxacin, enrofloxacin, sarafloxacin, flumequine, ciprofloxacin, difloxacin | Chicken muscle | Optical biosensor—dual SPR + LC electrospray time-of-flight MS | LC50: between 2.7 and 5.9 ng/g for multi-FQs and 3.8 ng/g for flumequine | Homogenize with water, filtration of supernatant, ultrafiltration 3 h at 67 °C in tube (Farm test) | [26] |
| Macrolides/Fenicol: Fenicols: CAP, CAP glucuronide | Poultry muscle | Optical biosensor—SPR | 0.005 ng/g for poultry muscle | (PBS) + ethyl acetate then evaporation supernatant 4 h at 45 °C in tube (Accusphere test) | [27] |
| Tetracyclines: Chlortetracycline, oxytetracycline, tetracycline, doxycycline and their 4-epimer metabolites | Poultry muscle | Bioluminescent biosensor bacterial | 5 ng/g for doxycycline, 7.5 ng/g for chlortetracycline, and 25 ng/g for (oxy)tetracycline | Heating, centrifugationto obtain meat fluid sample. 3 h at 67 °C in tube (Farm test) | [28] |
| Sulfonamides: Sulfamethazine, sulfisoxazole, sulfachlorpyridazine, sulfachlorpyrazine, sulfamerazine, sulfadiazine, sulfatroxazole, sulfathiazole | Chicken serum | Optical biosensor—SPR | 10 ng/mL for sulfamethazine | None | [29] |
8. Microfluidics and Biosensors
9. Future Outlook of Biosensors
10. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- World Health Organization. Tackling Antibiotic Resistance from a Food Safety Perspective in Europe; World Health Organization Regional Office for Europe: Copenhagen, Denmark, 2011; pp. 1–88. [Google Scholar]
- Levy, S.B.; Marshall, B. Antibacterial resistance worldwide: Causes, challenges and responses. Nat. Med. 2004, 10, S122–S129. [Google Scholar] [CrossRef] [PubMed]
- Crooks, S.R.H.; McCarney, B.; Traynor, I.M.; Thompson, C.S.; Floyd, S.; Elliott, C.T. Detection of levamisole residues in bovine liver and milk by immunobiosensor. Anal. Chim. Acta 2003, 483, 181–186. [Google Scholar] [CrossRef]
- Marshall, B.M.; Levy, S.B. Food Animals and Antimicrobials: Impacts on Human Health. Clin. Microbiol. Rev. 2011, 24, 718–733. [Google Scholar] [CrossRef] [PubMed]
- Kriebel, D.; Tickner, J.; Epstein, P.; Lemons, J.; Levins, R.; Loechler, E.L.; Quinn, M.; Rudel, R.; Schettler, T.; Stoto, M. The precautionary principle in environmental science. Environ. Health Perspect. 2001, 109, 871–876. [Google Scholar] [CrossRef] [PubMed]
- Maximum Residue Limits (MRLs) and the Safety of Food from Animals. Available online: http://www.noah.co.uk/issues/briefingdoc/09-mrls.htm (accessed on 12 September 2014).
- Cháfer-Pericás, C.; Maquieira, Á.; Puchades, R. Fast screening methods to detect antibiotic residues in food samples. Trends Anal. Chem. 2010, 29, 1038–1049. [Google Scholar] [CrossRef]
- Jiang, W.; Wang, Z.; Beier, R.C.; Jiang, H.; Wu, Y.; Shen, J. Simultaneous determination of 13 fluoroquinolone and 22 sulfonamide residues in milk by a dual-colorimetric enzyme-linked immunosorbent assay. Anal. Chem. 2013, 85, 1995–1999. [Google Scholar] [CrossRef] [PubMed]
- List of Maximum Residue Limits (MRLs) for Veterinary Drugs in Foods. Available online: http://www.hc-sc.gc.ca/dhp-mps/vet/mrl-lmr/mrl-lmr_versus_new-nouveau-eng.php (accessed on 12 September 2014).
- Galarini, R.; Diana, F.; Moretti, S.; Puppini, B.; Saluti, G.; Persic, L. Development and validation of a new qualitative ELISA screening for multiresidue detection of sulfonamides in food and feed. Food Control 2014, 35, 300–310. [Google Scholar] [CrossRef]
- Wang, S.; Xu, B.; Zhang, Y.; He, J.X. Development of enzyme-linked immunosorbent assay (ELISA) for the detection of neomycin residues in pig muscle, chicken muscle, egg, fish, milk and kidney. Meat Sci. 2009, 82, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Petz, M. Recent applications of surface plasmon resonance biosensors for analyzing residues and contaminants in food. Monatsh. Chem. 2009, 140, 953–964. [Google Scholar] [CrossRef]
- Huet, A.; Fodey, T.; Haughey, S.A.; Weigel, S.; Elliott, C.; Delahaut, P. Advances in biosensor-based analysis for antimicrobial residues in foods. Trends Anal. Chem. 2010, 29, 1281–1294. [Google Scholar] [CrossRef]
- Koyun, A.; Ahlatcolu, E.; Koca, Y. Biosensors and their principles. In A Roadmap of Biomedical Engineers and Milestones; Kara, S., Ed.; InTech: Rijeka, Croatia, 2012. [Google Scholar]
- Handrigan, J.P. Rapid Detection of Food-Borne Pathogens. 2010. Available online: http://www.johnpaulhandrigan.net/wp-content/uploads/2012/01/John-Paul-Handrigan-Rapid-Detection-of-Food-borne-Pathogens.pdf (accessed on 12 September 2014).
- Yola, M.L.; Uzun, L.; Özaltın, N.; Denizli, A. Development of molecular imprinted nanosensor for determination of tobramycin in pharmaceuticals and foods. Talanta 2014, 120, 318–324. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; He, L. Surface-enhanced Raman spectroscopy for the chemical analysis of food. Compr. Rev. Food Sci. Food Saf. 2014, 13, 317–328. [Google Scholar] [CrossRef]
- Tainaka, K.; Sakaguchi, R.; Hayashi, H.; Nakano, S.; Liew, F.F.; Morii, T. Design Strategies of Fluorescent Biosensors Based on Biological Macromolecular Receptors. Sensors 2010, 10, 1355–1376. [Google Scholar] [PubMed]
- Reder-Christ, K.; Bendas, G. Biosensor applications in the field of antibiotic research—A review of recent developments. Sensors 2011, 11, 9450–9466. [Google Scholar] [PubMed]
- Chen, D.; Yao, D.S.; Xie, C.F.; Liu, D.L. Development of an aptasensor for electrochemical detection of tetracycline. Food Control 2014, 42, 109–115. [Google Scholar] [CrossRef]
- Hou, H.; Bai, X.; Xing, C.; Gu, N.; Zhang, B.; Tang, J.; Bai, X.; Xing, C.; Gu, N.; Zhang, B.; et al. Aptamer-based cantilever array sensors for oxytetracycline detection. Anal. Chem. 2013, 85, 2010–2014. [Google Scholar] [CrossRef] [PubMed]
- Que, X.; Tang, D.; Xia, B.; Lu, M. Gold nanocatalyst-based immunosensing strategy accompanying catalytic reduction of 4-nitrophenol for sensitive monitoring of chloramphenicol residue. Anal. Chim. Acta 2014, 830, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Shen, L.; Chen, J.; Li, N.; He, P.; Li, Z. Rapid colorimetric sensing of tetracycline antibiotics with in situ growth of gold nanoparticles. Anal. Chim. Acta 2014, 839, 83–90. [Google Scholar] [CrossRef]
- Berling, H.; Jarhede, T.; Larsson, A.; Roos, H. Method of Determining Analyte Concentration. U.S. Patent 8,263,415, 11 September 2012. [Google Scholar]
- Ferguson, J.P.; Baxter, G.A.; McEvoy, J.D.G.; Stead, S.; Rawlings, E.; Sharman, M. Detection of streptomycin and dihydrostreptomycin residues in milk, honey and meat samples using an optical biosensor. Analyst 2002, 127, 951–956. [Google Scholar] [CrossRef] [PubMed]
- Marchesini, G.R.; Hooijerink, H.; Haasnoot, W.; Nielen, M.W.F.; Buijs, J.; Campbell, K.; Elliott, C.T.; Nielen, M.W.F. Towards surface plasmon resonance biosensing combined with bioaffinity-assisted nano HILIC liquid chromatography/time-of-flight mass spectrometry identification of paralytic shellfish poisons. TrAC Trends Anal. Chem. 2009, 28, 792–803. [Google Scholar] [CrossRef]
- Ferguson, J.; Baxter, A.; Young, P.; Kennedy, G.; Elliott, C.; Weigel, S.; Gatermann, R.; Ashwin, H.; Stead, S.; Sharman, M. Detection of chloramphenicol and chloramphenicol glucuronide residues in poultry muscle, honey, prawn and milk using a surface plasmon resonance biosensor and Qflex® kit chloramphenicol. Anal. Chim. Acta 2005, 529, 109–113. [Google Scholar] [CrossRef]
- Pikkemaat, M.G.; Rapallini, M.L.B.A.; Karp, M.T.; Elferink, J.W.A. Application of a luminescent bacterial biosensor for the detection of tetracyclines in routine analysis of poultry muscle samples. Food Addit. Contam. Part A 2010, 27, 1112–1117. [Google Scholar] [CrossRef]
- Haasnoot, W.; Bienenmann-Ploum, M.; Kohen, F. Biosensor immunoassay for the detection of eight sulfonamides in chicken serum. Anal. Chim. Acta 2003, 483, 171–180. [Google Scholar] [CrossRef]
- Rivet, C.; Lee, H.; Hirsch, A.; Hamilton, S.; Lu, H. Microfluidics for medical diagnostics and biosensors. Chem. Eng. Sci. 2011, 66, 1490–1507. [Google Scholar] [CrossRef]
- Situ, C.; Mooney, M.H.; Elliott, C.T.; Buijs, J. Advances in surface plasmon resonance biosensor technology towards high-throughput, food-safety analysis. TrAC Trends Anal. Chem. 2010, 29, 1305–1315. [Google Scholar] [CrossRef]
- Haasnoot, W.; Loomans, E.E.M.G.; Cazemier, G.; Dietrich, R.; Verheijen, R.; Bergwerff, A.A.; Stephany, R.W. Direct versus competitive biosensor immunoassays for the detection of (dihydro)streptomycin residues in milk. Food Agric. Immunol. 2002, 14, 15–27. [Google Scholar]
- Asadollahi-Baboli, M.; Mani-Varnosfaderani, A. Rapid and simultaneous determination of tetracycline and cefixime antibiotics by mean of gold nanoparticles-screen printed gold electrode and chemometrics tools. Measurement 2014, 47, 145–149. [Google Scholar] [CrossRef]
- Jin, W.; Yang, G.; Shao, H.; Qin, A. A label-free impedimetric immunosensor for detection of 1-aminohydantoin residue in food samples based on solegel embedding antibody. Food Control 2014, 39, 185–191. [Google Scholar] [CrossRef]
- Fernández, F.; Hegnerová, K.; Piliarik, M.; Sanchez-Baeza, F.; Homola, J.; Marco, M.-P. A label-free and portable multichannel surface plasmon resonance immunosensor for on site analysis of antibiotics in milk samples. Biosens. Bioelectron. 2010, 26, 1231–1238. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mungroo, N.A.; Neethirajan, S. Biosensors for the Detection of Antibiotics in Poultry Industry—A Review. Biosensors 2014, 4, 472-493. https://doi.org/10.3390/bios4040472
Mungroo NA, Neethirajan S. Biosensors for the Detection of Antibiotics in Poultry Industry—A Review. Biosensors. 2014; 4(4):472-493. https://doi.org/10.3390/bios4040472
Chicago/Turabian StyleMungroo, Nawfal Adam, and Suresh Neethirajan. 2014. "Biosensors for the Detection of Antibiotics in Poultry Industry—A Review" Biosensors 4, no. 4: 472-493. https://doi.org/10.3390/bios4040472
APA StyleMungroo, N. A., & Neethirajan, S. (2014). Biosensors for the Detection of Antibiotics in Poultry Industry—A Review. Biosensors, 4(4), 472-493. https://doi.org/10.3390/bios4040472





