Salmonella Detection in Food Using a HEK-hTLR5 Reporter Cell-Based Sensor
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Cultures and Motility Testing
2.2. Cell Culture
2.3. Cell-Based Biosensor for Detection of Bacterial Cultures
2.4. Specificity/Selectivity of Cell-Based Sensor
2.5. Limit of Detection of Cell-Based Sensor
2.6. Salmonella Enteritidis Analysis in Spiked Food Samples
2.7. Detection of Stressed Cells
2.8. Cytotoxicity Assays
2.9. Polymerase Chain Reaction (PCR) Confirmation
2.10. Statistical Analysis
3. Results
3.1. Optimization of HEK Dual hTLR5 Sensor for Detection of Salmonella
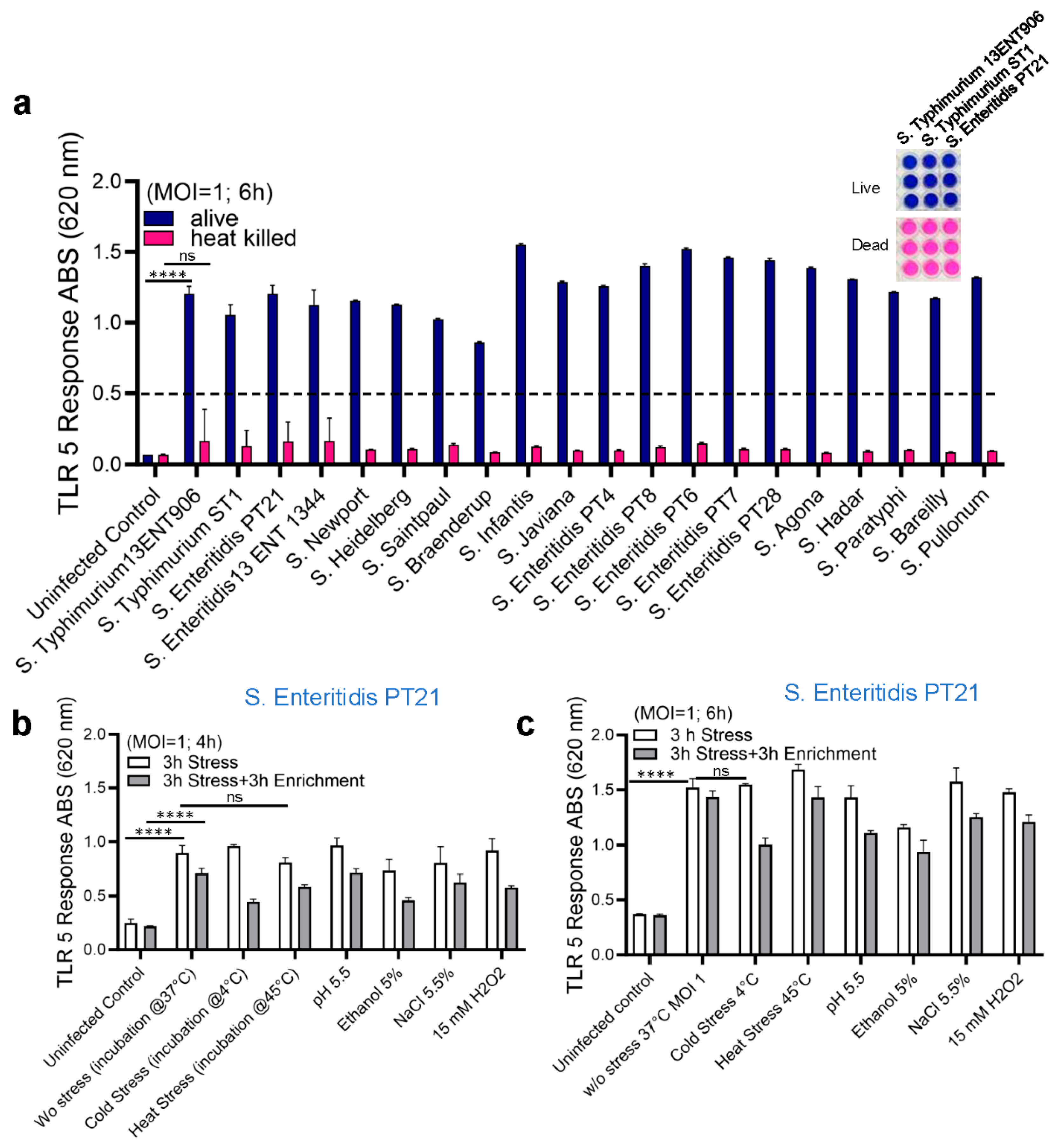
3.2. Specificity of HEK Dual TLR5 Sensor for Detection of Salmonella Serovars
3.3. Detection of Live and Stress-Exposed Salmonella Using HEK Dual TLR5 Sensor
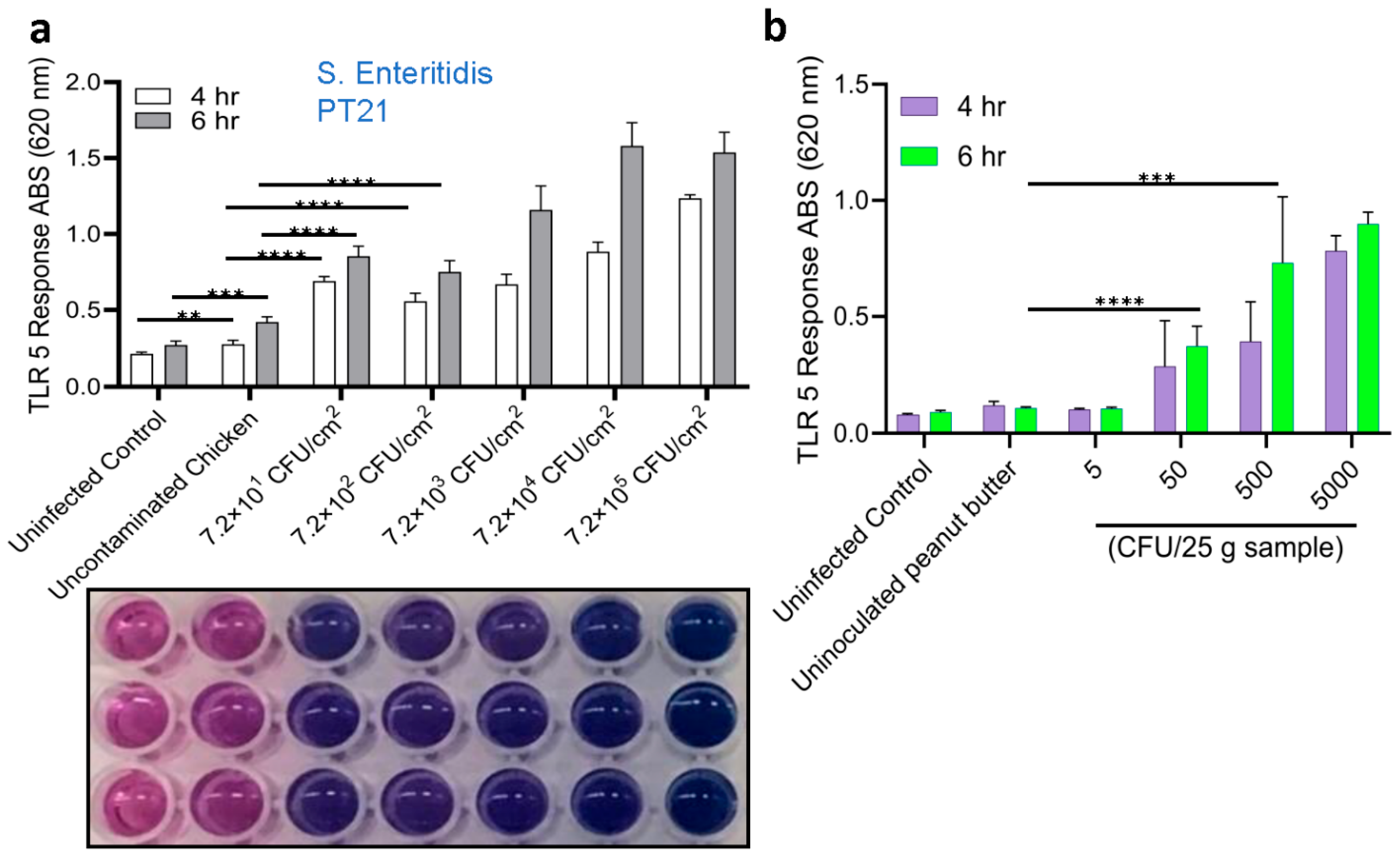
3.4. Detection of Salmonella from Spiked Food Samples Using HEK Dual TLR5 Sensor
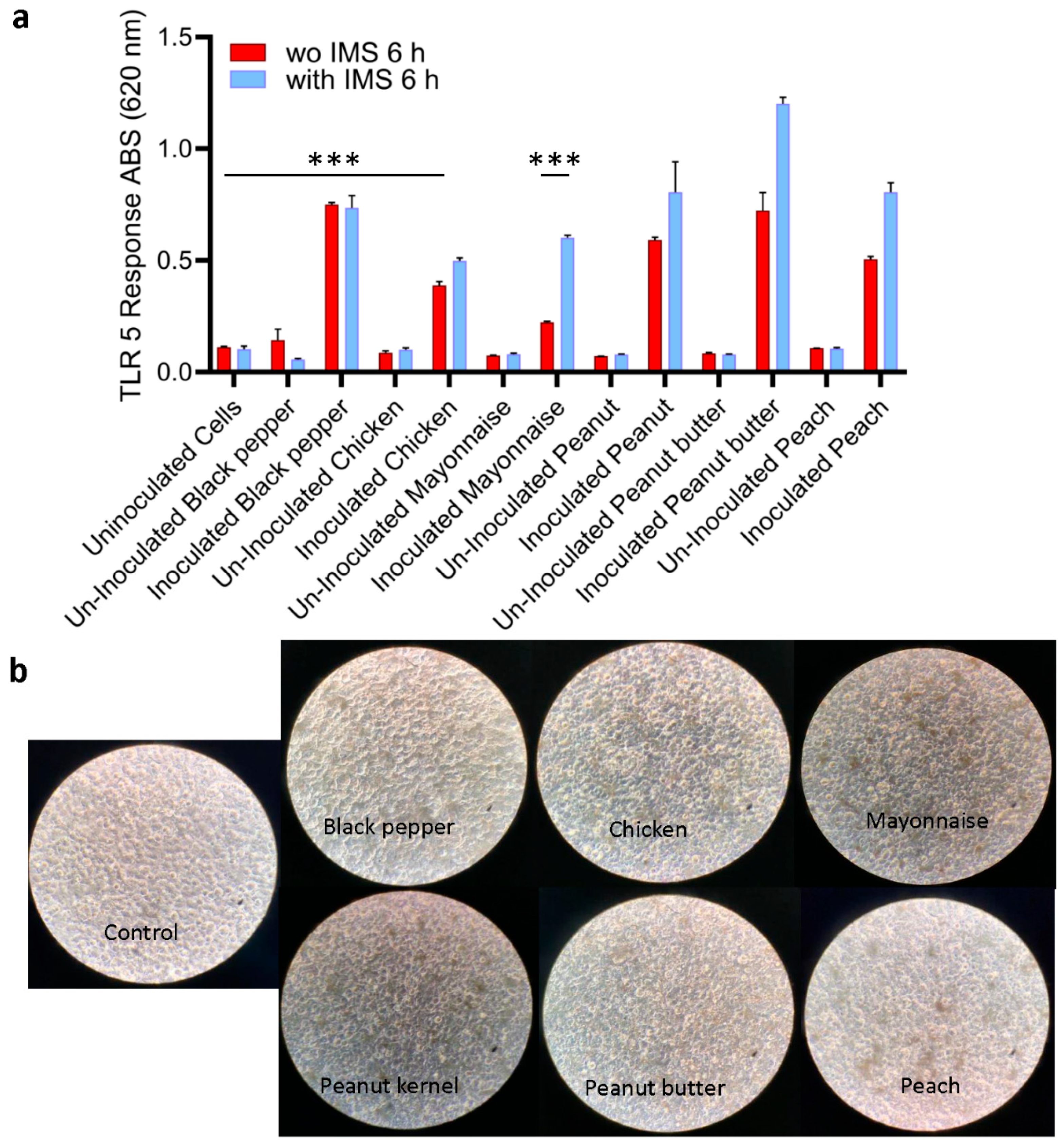
3.5. Validation of Sensor with Spiked Food Samples
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bhunia, A.K. (Ed.) Salmonella enterica. In Foodborne Microbial Pathogens: Mechanisms and Pathogenesis; Springer: New York, NY, USA, 2018; pp. 271–287. [Google Scholar] [CrossRef]
- Teklemariam, A.D.; Al-Hindi, R.R.; Albiheyri, R.S.; Alharbi, M.G.; Alghamdi, M.A.; Filimban, A.A.; Al Mutiri, A.S.; Al-Alyani, A.M.; Alseghayer, M.S.; Almaneea, A.M.; et al. Human Salmonellosis: A Continuous Global Threat in the Farm-to-Fork Food Safety Continuum. Foods 2023, 12, 1756. [Google Scholar] [CrossRef]
- Popa, G.L.; Papa, M.I. Salmonella spp. infection-A continuous threat worldwide. Germs 2021, 11, 88. [Google Scholar] [CrossRef] [PubMed]
- Guard, J. Through the Looking Glass: Genome, Phenome, and Interactome of Salmonella enterica. Pathogens 2022, 11, 581. [Google Scholar] [CrossRef] [PubMed]
- O’Bryan, C.A.; Ricke, S.C.; Marcy, J.A. Public health impact of Salmonella spp. on raw poultry: Current concepts and future prospects in the United States. Food Control 2022, 132, 108539. [Google Scholar] [CrossRef]
- Kowalska, B. Fresh vegetables and fruit as a source of Salmonella bacteria. Ann. Agric. Environ. Med. 2023, 30, 9–14. [Google Scholar] [CrossRef] [PubMed]
- CDC. Salmonella; CDC: Atlanta, GA, USA, 2020. Available online: https://www.cdc.gov/salmonella/index.html (accessed on 1 February 2020).
- Guillén, S.; Nadal, L.; Álvarez, I.; Mañas, P.; Cebrián, G. Impact of the Resistance Responses to Stress Conditions Encountered in Food and Food Processing Environments on the Virulence and Growth Fitness of Non-Typhoidal Salmonellae. Foods 2021, 10, 617. [Google Scholar] [CrossRef] [PubMed]
- Gruzdev, N.; Pinto, R.; Sela, S. Effect of desiccation on tolerance of Salmonella enterica to multiple stresses. Appl. Environ. Microbiol. 2011, 77, 1667–1673. [Google Scholar] [CrossRef] [PubMed]
- Gustavsen, S.; Breen, O. Investigation of an outbreak of Salmonella Oranienburg infections in Norway, caused by contaminated black pepper. Am. J. Epidemiol. 1984, 119, 806–812. [Google Scholar] [CrossRef]
- McCormic, Z.D.; Patel, K.; Higa, J.; Bancroft, J.; Donovan, D.; Edwards, L.; Cheng, J.; Adcock, B.; Bond, C.; Pereira, E. Bi-national outbreak of Salmonella Newport infections linked to onions: The United States experience. Epidemiol. Infect. 2022, 150, e199. [Google Scholar] [CrossRef]
- Mitchell, M.R., Jr.; Kirchner, M.; Schneider, B.; McClure, M.; Neil, K.P.; Madad, A.; Jemaneh, T.; Tijerina, M.; Nolte, K.; Wellman, A. Multistate outbreak of Salmonella Oranienburg infections linked to bulb onions imported from Mexico–United States, 2021. Food Control 2024, 160, 110325. [Google Scholar] [CrossRef]
- CDC. Multistate Outbreak of Human Salmonella Montevideo Infections. Available online: https://www.cdc.gov/salmonella/2010/montevideo-5-4-2010.html (accessed on 3 April 2020).
- Kirk, M.D.; Little, C.L.; Lem, M.; Fyfe, M.; Genobile, D.; Tan, A.; Threlfall, J.; Paccagnella, A.; Lightfoot, D.; Lyi, H.; et al. An outbreak due to peanuts in their shell caused by Salmonella enterica serotypes Stanley and Newport--sharing molecular information to solve international outbreaks. Epidemiol. Infect. 2004, 132, 571–577. [Google Scholar] [CrossRef] [PubMed]
- Scheil, W.; Cameron, S.; Dalton, C.; Murray, C.; Wilson, D. A South Australian Salmonella Mbandaka outbreak investigation using a database to select controls. Aust. N. Z. J. Public Health 1998, 22, 536–539. [Google Scholar] [CrossRef] [PubMed]
- Calhoun, S.; Post, L.; Warren, B.; Thompson, S.; Bontempo, A.R. Prevalence and concentration of Salmonella on raw shelled peanuts in the United States. J. Food Prot. 2013, 76, 575–579. [Google Scholar] [CrossRef] [PubMed]
- CDC. Outbreak of Salmonella Enteritidis Infections Linked to Peaches. Available online: https://www.cdc.gov/salmonella/enteritidis-08-20/index.html (accessed on 21 March 2021).
- CDC. Multistate Outbreak of Multidrug-Resistant Salmonella Heidelberg Infections Linked to Foster Farms Brand Chicken. Available online: https://www.cdc.gov/salmonella/heidelberg-10-13/index.html (accessed on 3 April 2020).
- CDC. Outbreak of Salmonella Heidelberg Infections Linked to Tyson Brand Mechanically Separated Chicken at a Correctional Facility. Available online: https://www.cdc.gov/salmonella/heidelberg-01-14/index.html (accessed on 3 April 2020).
- CDC. Outbreak of Salmonella Infections Linked to Chicken. Available online: https://www.cdc.gov/salmonella/chicken-08-18/index.html (accessed on 1 February 2020).
- Mitchell, E.; O’Mahony, M.; Lynch, D.; Ward, L.R.; Rowe, B.; Uttley, A.; Rogers, T.; Cunningham, D.G.; Watson, R. Large outbreak of food poisoning caused by Salmonella typhimurium definitive type 49 in mayonnaise. Br. Med. J. 1989, 298, 99–101. [Google Scholar] [CrossRef]
- Mason, B.W.; Williams, N.; Salmon, R.L.; Lewis, A.; Price, J.; Johnston, K.M.; Trott, R.M. Outbreak of Salmonella Indiana associated with egg mayonnaise sandwiches at an acute NHS hospital. Commun. Dis. Public Health 2001, 4, 300–304. [Google Scholar]
- Carneiro, M.R.; Cabello, P.H.; Albuquerque-Junior, R.L.; Jain, S.; Candido, A.L. Characterization of a foodborne outbreak caused by Salmonella Enteritidis in Aracaju, State of Sergipe, Brazil. Rev. Soc. Bras. Med. Trop. 2015, 48, 334–337. [Google Scholar] [CrossRef]
- von Wissmann, B.; Klinc, C.; Schulze, R.; Wolf, A.; Schreiner, H.; Rabsch, W.; Prager, R.; Hautmann, W. Outbreak of salmonellosis after a wedding party, Bavaria, Germany, summer 2010: The importance of implementing food safety concepts. Eurosurveillance 2012, 17, 20076. [Google Scholar] [CrossRef]
- Bhunia, A.K. One day to one hour: How quickly can foodborne pathogens be detected? Future Microbiol. 2014, 9, 935–946. [Google Scholar] [CrossRef]
- Al-Hindi, R.R.; Teklemariam, A.D.; Alharbi, M.G.; Alotibi, I.; Azhari, S.A.; Qadri, I.; Alamri, T.; Harakeh, S.; Applegate, B.M.; Bhunia, A.K. Bacteriophage-Based Biosensors: A Platform for Detection of Foodborne Bacterial Pathogens from Food and Environment. Biosensors 2022, 12, 905. [Google Scholar] [CrossRef]
- Foddai, A.C.G.; Grant, I.R. Methods for detection of viable foodborne pathogens: Current state-of-art and future prospects. Appl. Microbiol. Biotechnol. 2020, 104, 4281–4288. [Google Scholar] [CrossRef]
- Daquigan, N.; Grim, C.J.; White, J.R.; Hanes, D.E.; Jarvis, K.G. Early Recovery of Salmonella from Food Using a 6-Hour Non-selective Pre-enrichment and Reformulation of Tetrathionate Broth. Front. Microbiol. 2016, 7, 2103. [Google Scholar] [CrossRef] [PubMed]
- Koyuncu, S.; Andersson, M.G.; Häggblom, P. Accuracy and Sensitivity of Commercial PCR-Based Methods for Detection of Salmonella enterica in Feed. Appl. Environ. Microbiol. 2010, 76, 2815. [Google Scholar] [CrossRef] [PubMed]
- Moreirinha, C.; Trindade, J.; Saraiva, J.A.; Almeida, A.; Delgadillo, I. MIR spectroscopy as alternative method for further confirmation of foodborne pathogens Salmonella spp. and Listeria monocytogenes. J. Food Sci. Technol. 2018, 55, 3971–3978. [Google Scholar] [CrossRef] [PubMed]
- Villamizar-Rodríguez, G.; Fernández, J.; Marín, L.; Muñiz, J.; González, I.; Lombó, F. Multiplex detection of nine food-borne pathogens by mPCR and capillary electrophoresis after using a universal pre-enrichment medium. Front. Microbiol. 2015, 6, 1194. [Google Scholar] [CrossRef]
- Xu, L.; Bai, X.; Bhunia, A.K. Current State of Biosensors Development and their Application in Foodborne Pathogen Detection. J. Food Prot. 2021, 84, 1213–1227. [Google Scholar] [CrossRef]
- Eijkelkamp, J.M.; Aarts, H.J.M.; van der Fels-Klerx, H.J. Suitability of Rapid Detection Methods for Salmonella in Poultry Slaughterhouses. Food Anal. Methods 2009, 2, 1–13. [Google Scholar] [CrossRef]
- Wang, Q.; Chang, K.; Yang, Q.; Wu, W. Semiconductor-based surface-enhanced Raman scattering sensing platforms: State of the art, applications and prospects in food safety. Trends Food Sci. Technol. 2024, 147, 104460. [Google Scholar] [CrossRef]
- Chang, K.; Zhao, Y.; Wang, M.; Xu, Z.; Zhu, L.; Xu, L.; Wang, Q. Advances in metal-organic framework-plasmonic metal composites based SERS platforms: Engineering strategies in chemical sensing, practical applications and future perspectives in food safety. Chem. Eng. J. 2023, 459, 141539. [Google Scholar] [CrossRef]
- Xu, L.; Bai, X.; Tenguria, S.; Liu, Y.; Drolia, R.; Bhunia, A.K. Mammalian cell-based immunoassay for detection of viable bacterial pathogens. Front. Microbiol. 2020, 11, 575615. [Google Scholar] [CrossRef]
- Bhunia, A.K.; Singh, A.K.; Parker, K.; Applegate, B.M. Petri-plate, bacteria, and laser optical scattering sensor. Front. Cell. Infect. Microbiol. 2022, 12, 1087074. [Google Scholar] [CrossRef]
- To, C.; Banerjee, P.; Bhunia, A.K. Cell-Based Biosensor for Rapid Screening of Pathogens and Toxins. In Handbook of Cell Biosensors; Thouand, G., Ed.; Springer International Publishing: Cham, Switzerland, 2020; pp. 1–16. [Google Scholar]
- Lu, X.; Ye, Y.; Zhang, Y.; Sun, X. Current research progress of mammalian cell-based biosensors on the detection of foodborne pathogens and toxins. Crit. Rev. Food Sci. Nutr. 2021, 61, 3819–3835. [Google Scholar] [CrossRef] [PubMed]
- Ye, Y.; Guo, H.; Sun, X. Recent progress on cell-based biosensors for analysis of food safety and quality control. Biosens. Bioelectron. 2019, 126, 389–404. [Google Scholar] [CrossRef] [PubMed]
- Burkholder, K.; Bhunia, A. Salmonella enterica serovar Typhimurium adhesion and cytotoxicity during epithelial cell stress is reduced by Lactobacillus rhamnosus GG. Gut Pathog. 2009, 1, 14. [Google Scholar] [CrossRef]
- Birhanu, B.T.; Park, N.-H.; Lee, S.-J.; Hossain, M.A.; Park, S.-C. Inhibition of Salmonella Typhimurium adhesion, invasion, and intracellular survival via treatment with methyl gallate alone and in combination with marbofloxacin. Vet. Res. 2018, 49, 101. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.M.; Kaniga, K.; Galan, J.E. Salmonella spp. are cytotoxic for cultured macrophages. Mol. Microbiol. 1996, 21, 1101–1115. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Yan, H. TLR5: Beyond the recognition of flagellin. Cell. Mol. Immunol. 2017, 14, 1017–1019. [Google Scholar] [CrossRef]
- Song, W.S.; Jeon, Y.J.; Namgung, B.; Hong, M.; Yoon, S.I. A conserved TLR5 binding and activation hot spot on flagellin. Sci. Rep. 2017, 7, 40878. [Google Scholar] [CrossRef]
- Andersen-Nissen, E.; Smith, K.D.; Strobe, K.L.; Barrett, S.L.R.; Cookson, B.T.; Logan, S.M.; Aderem, A. Evasion of Toll-like receptor 5 by flagellated bacteria. Proc. Natl. Acad. Sci. USA 2005, 102, 9247–9252. [Google Scholar] [CrossRef]
- Mendonca, M.; Conrad, N.; Conceicao, F.; Moreira, A.; da Silva, W.; Aleixo, J.; Bhunia, A. Highly specific fiber optic immunosensor coupled with immunomagnetic separation for detection of low levels of Listeria monocytogenes and L. ivanovii. BMC Microbiol. 2012, 12, 275. [Google Scholar] [CrossRef]
- de Cássia Dos Santos da Conceição, R.; Moreira, A.N.; Ramos, R.J.; Goularte, F.L.; Carvalhal, J.B.; Aleixo, J.A.G. Detection of Salmonella sp in chicken cuts using immunomagnetic separation. Braz. J. Microbiol. 2008, 39, 173–177. [Google Scholar] [CrossRef]
- Mansfield, L.P.; Forsythe, S.J. The detection of Salmonella using a combined immunomagnetic separation and ELISA end-detection procedure. Lett. Appl. Microbiol. 2000, 31, 279–283. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.K.; Bettasso, A.M.; Bae, E.; Rajwa, B.; Dundar, M.M.; Forster, M.D.; Liu, L.; Barrett, B.; Lovchik, J.; Robinson, J.P.; et al. Laser optical sensor, a label-free on-plate Salmonella enterica colony detection tool. mBio 2014, 5, e01019-13. [Google Scholar] [CrossRef] [PubMed]
- Kearns, D.B.; Losick, R. Swarming motility in undomesticated Bacillus subtilis. Mol. Microbiol. 2003, 49, 581–590. [Google Scholar] [CrossRef] [PubMed]
- Hahm, B.K.; Bhunia, A.K. Effect of environmental stresses on antibody-based detection of Escherichia coli O157:H7, Salmonella enterica serotype Enteritidis and Listeria monocytogenes. J. Appl. Microbiol. 2006, 100, 1017–1027. [Google Scholar] [CrossRef] [PubMed]
- Ha, D.-G.; Kuchma, S.L.; O’Toole, G.A. Plate-Based Assay for Swimming Motility in Pseudomonas aeruginosa. In Pseudomonas Methods and Protocols; Filloux, A., Ramos, J.-L., Eds.; Springer: New York, NY, USA, 2014; pp. 59–65. [Google Scholar]
- USDA-FSIS. Isolation and Identification of Salmonella from Meat, Poultry, Pasteurized Egg and Catfish Products. In Microbiology Laboratory Guidebook; Method number MLG 4.06; USDA-FSIS: Washington, DC, USA, 2013. Available online: http://www.fsis.usda.gov/wps/wcm/connect/700c05fe-06a2-492a-a6e1-3357f7701f52/MLG-4.pdf?MOD=AJPERES (accessed on 1 February 2020).
- Bell, R.L.; Jarvis, K.G.; Ottesen, A.R.; McFarland, M.A.; Brown, E.W. Recent and emerging innovations in Salmonella detection: A food and environmental perspective. Microb. Biotechnol. 2016, 9, 279–292. [Google Scholar] [CrossRef] [PubMed]
- FDA. Bacteriological Analytical Manual Online, 8th ed.; AOAC International: Arlington, VA, USA, 2001. Available online: http://www.fda.gov/Food/ScienceResearch/LaboratoryMethods/BacteriologicalAnalyticalManualBAM/default.htm (accessed on 1 February 2020).
- Rajapaksha, P.; Elbourne, A.; Gangadoo, S.; Brown, R.; Cozzolino, D.; Chapman, J. A review of methods for the detection of pathogenic microorganisms. Analyst 2019, 144, 396–411. [Google Scholar] [CrossRef]
- Ricke, S.C.; Kim, S.A.; Shi, Z.; Park, S.H. Molecular-based identification and detection of Salmonella in food production systems: Current perspectives. J. Appl. Microbiol. 2018, 125, 313–327. [Google Scholar] [CrossRef]
- Lee, K.-M.; Runyon, M.; Herrman, T.J.; Phillips, R.; Hsieh, J. Review of Salmonella detection and identification methods: Aspects of rapid emergency response and food safety. Food Control 2015, 47, 264–276. [Google Scholar] [CrossRef]
- Buzby, J.C.; Farah-Wells, H.; Hyman, J. The Estimated Amount, Value, and Calories of Postharvest Food Losses at the Retail and Consumer Levels in the United States. USDA-ERS Economic Information Bulletin Number 121. Available online: https://ssrn.com/abstract=2501659 (accessed on 1 February 2014). [CrossRef]
- Elkhishin, M.T.; Gooneratne, R.; Hussain, M.A. Microbial safety of foods in the supply chain and food security. Adv. Food Technol. Nutr. Sci. Open J. 2017, 3, 22–32. [Google Scholar] [CrossRef]
- Hayashi, F.; Smith, K.D.; Ozinsky, A.; Hawn, T.R.; Yi, E.C.; Goodlett, D.R.; Eng, J.K.; Akira, S.; Underhill, D.M.; Aderem, A. The innate immune response to bacterial flagellin is mediated by Toll-like receptor 5. Nature 2001, 410, 1099–1103. [Google Scholar] [CrossRef]
- Smith, K.D.; Andersen-Nissen, E.; Hayashi, F.; Strobe, K.; Bergman, M.A.; Barrett, S.L.R.; Cookson, B.T.; Aderem, A. Toll-like receptor 5 recognizes a conserved site on flagellin required for protofilament formation and bacterial motility. Nat. Immunol. 2003, 4, 1247–1253. [Google Scholar] [CrossRef] [PubMed]
- Yoon, S.-I.; Kurnasov, O.; Natarajan, V.; Hong, M.; Gudkov, A.V.; Osterman, A.L.; Wilson, I.A. Structural basis of TLR5-flagellin recognition and signaling. Science 2012, 335, 859–864. [Google Scholar] [CrossRef] [PubMed]
- Thomas, P.; Smart, T.G. HEK293 cell line: A vehicle for the expression of recombinant proteins. J. Pharmacol. Toxicol. Methods 2005, 51, 187–200. [Google Scholar] [CrossRef] [PubMed]
- Miao, E.A.; Alpuche-Aranda, C.M.; Dors, M.; Clark, A.E.; Bader, M.W.; Miller, S.I.; Aderem, A. Cytoplasmic flagellin activates caspase-1 and secretion of interleukin 1β via Ipaf. Nat. Immunol. 2006, 7, 569–575. [Google Scholar] [CrossRef] [PubMed]
- Schrader, C.; Schielke, A.; Ellerbroek, L.; Johne, R. PCR inhibitors–occurrence, properties and removal. J. Appl. Microbiol. 2012, 113, 1014–1026. [Google Scholar] [CrossRef] [PubMed]
- Lin, A.; Singh, A.; Allred, A.; Allard, M.; Waltman, D.; Imanian, B.; Ng, J.H.J.; Sanahmadi, Y.; Khaksar, R. Targeted Next Generation Sequencing Assay for Direct Detection and Serotyping of Salmonella from Enrichment. J. Food Prot. 2024, 87, 100256. [Google Scholar] [CrossRef]
- Vinayaka, A.C.; Ngo, T.A.; Kant, K.; Engelsmann, P.; Dave, V.P.; Shahbazi, M.A.; Wolff, A.; Bang, D.D. Rapid detection of Salmonella enterica in food samples by a novel approach with combination of sample concentration and direct PCR. Biosens. Bioelectron. 2019, 129, 224–230. [Google Scholar] [CrossRef]
- Kuang, H.; Cui, G.; Chen, X.; Yin, H.; Yong, Q.; Xu, L.; Peng, C.; Wang, L.; Xu, C. A one-step homogeneous sandwich immunosensor for Salmonella detection based on magnetic nanoparticles (MNPs) and quantum Dots (QDs). Int. J. Mol. Sci. 2013, 14, 8603–8610. [Google Scholar] [CrossRef]
- Cimaglia, F.; De Lorenzis, E.; Mezzolla, V.; Rossi, F.; Poltronieri, P. Detection of L. monocytogenes in Enrichment Cultures by Immunoseparation and Immunosensors. IEEE Sens. J. 2016, 16, 7045–7052. [Google Scholar] [CrossRef]


| Salmonella enterica Serovars | Non-Salmonella Cultures |
|---|---|
| Enteritidis PT4 | Escherichia coli O157:H¯493-89 (flagella negative control) |
| Enteritidis PT8 | E. coli O26:H11 00971 |
| Enteritidis PT6 | E. coli O103:H2 90-3128 |
| Enteritidis PT7 | E. coli O26:H11 05-6544 |
| Enteritidis PT28 | E. coli O103:H11 SJ12 |
| Enteritidis PT21 | E. coli O157:H7 204P |
| Enteritidis 13ENT1344 | E. coli O157:H7 505B |
| Typhimurium 13ENT906 | E. coli O157:H7 ATCC 43895 |
| Typhimurium ST1 | E. coli O157:H7 ATCC 43890 |
| Agona 12ENT1356 | Citrobacter freundii ATCC 3624 |
| Hadar 13ENT979 | Citrobacter freundii ATCC43864 |
| Paratyphi 11J85 | Citrobacter freundii ATCC8090 |
| Bareilly 12ENT1164 | Hafnia alvei |
| Pullorum DUP-PVUII 1006 | Proteus vulgaris DUP-10086 |
| Newport 13ENT1060 | Proteus mirabilis B-3402 |
| Heidelberg 18ENT1418 | Serratia marcescens ATCC 8100 |
| Saintpaul 13ENT1045 | S. marcescens B-2544 |
| Bradenderup 12ENT1138 | S. marcescens ATCC 43862 |
| Infantis 13ENT866 | Klebsiella pneumoniae B-41958 |
| Javiana 13ENT86F | Listeria monocytogenes 104033S |
| Log 10 (CFU/mL) ± SEM (N = 3) | |||||||
|---|---|---|---|---|---|---|---|
| No Stress | Cold Stress | Heat Stress | Low pH | Ethanol | NaCL | H2O2 | |
| 3-h stress | 9.86 ± 0.19 | 9.38 ± 0.21 | 9.35 ± 0.11 | 9.67 ± 0.14 | 9.60 ± 0.22 | 9.24 ± 0.28 | 9.33 ± 0.21 |
| 3-h stress + 3-h enrichment | 10.49 ± 0.22 | 10.44 ± 0.24 | 10.01 ± 0.21 | 10.21 ± 0.24 | 9.69 ± 0.29 | 10.33 ± 0.19 | 10.65 ± 0.24 |
| Log 10 (CFU/mL) ± SEM (N = 3) | ||||
|---|---|---|---|---|
| Inoculation Level (CFU/25 g Sample) | Pre-Enriched | Enriched | ||
| TSA | XLD | TSA | XLD | |
| 0 | No growth | No growth | No growth | No growth |
| ~5 | No growth | No growth | 2.86 ± 0.21 | 3.09 ± 0.18 |
| ~50 | 2.68 ± 0.17 | 2.51 ± 0.11 | 4.06 ± 0.14 | 4.18 ± 0.19 |
| ~500 | 3.94 ± 0.21 | 3.85 ± 0.19 | 6.32 ± 0.22 | 6.39 ± 0.24 |
| ~500 | 4.35 ± 0.13 | 4.44 ± 0.18 | 6.60 ± 0.23 | 6.42 ± 0.14 |
| Log 10 (CFU/mL) ± SEM (N = 3) | ||||||||
|---|---|---|---|---|---|---|---|---|
| Food Sample | After Selective Enrichment | Without IMS | IMS | Counts (CFU/100 µL) | PCR (invA 796 bp) | |||
| TSA | XLD | TSA | XLD | TSA | XLD | XLD | ||
| Black pepper (U) | 0 | 0 | 0 | 0 | 0 | 0 | 0 | - |
| Black pepper (I) | 3.91 ± 0.18 | 4.04 ± 0.11 | 3.87 ± 0.14 | 4.05 ± 0.21 | 3.98 ± 0.08 | 4.07 ± 0.14 | 3.07 ± 0.11 | - |
| Chicken (U) | 0 | 0 | 0 | 0 | 0 | 0 | 0 | - |
| Mayonnaise (U) | 0 | 0 | 0 | 0 | 0 | 0 | 0 | - |
| Mayonnaise (I) | 3.94 ± 0.22 | 4.02 ± 0.14 | 3.72 ± 0.18 | 3.94 ± 0.12 | 3.72 ± 0.21 | 3.92 ± 0.18 | 2.92 ± 0.13 | - |
| Peanut kernel(U) | 0 | 0 | 0 | 0 | 0 | 0 | 0 | - |
| Peanut kernel (I) | 3.92 ± 0.26 | 4.09 ± 0.19 | 4.26 ± 0.22 | 4.29 ± 0.18 | 4.07 ± 0.14 | 4.13 ± 0.19 | 3.13 ± 0.18 | + |
| Peanut butter (U) | 0 | 0 | 0 | 0 | 0 | 0 | 0 | - |
| Peanut butter (I) | 5.53 ± 0.24 | 5.18 ± 0.18 | 5.36 ± 0.25 | 5.46 ± 0.28 | 5.39 ± 0.22 | 5.47 ± 0.24 | 4.47 ± 0.16 | - |
| Peach (U) | 0 | 0 | 0 | 0 | 0 | 0 | 0 | - |
| Peach (I) | 3.71 ± 0.14 | 3.88 ± 0.16 | 3.72 ± 0.12 | 4.08 ± 0.19 | 3.91 ± 0.16 | 4.33 ± 0.14 | 3.33 ± 0.11 | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eser, E.; Felton, V.A.; Drolia, R.; Bhunia, A.K. Salmonella Detection in Food Using a HEK-hTLR5 Reporter Cell-Based Sensor. Biosensors 2024, 14, 444. https://doi.org/10.3390/bios14090444
Eser E, Felton VA, Drolia R, Bhunia AK. Salmonella Detection in Food Using a HEK-hTLR5 Reporter Cell-Based Sensor. Biosensors. 2024; 14(9):444. https://doi.org/10.3390/bios14090444
Chicago/Turabian StyleEser, Esma, Victoria A. Felton, Rishi Drolia, and Arun K. Bhunia. 2024. "Salmonella Detection in Food Using a HEK-hTLR5 Reporter Cell-Based Sensor" Biosensors 14, no. 9: 444. https://doi.org/10.3390/bios14090444
APA StyleEser, E., Felton, V. A., Drolia, R., & Bhunia, A. K. (2024). Salmonella Detection in Food Using a HEK-hTLR5 Reporter Cell-Based Sensor. Biosensors, 14(9), 444. https://doi.org/10.3390/bios14090444






