Perspective about Cellulose-Based Pressure and Strain Sensors for Human Motion Detection
Abstract
:1. Introduction
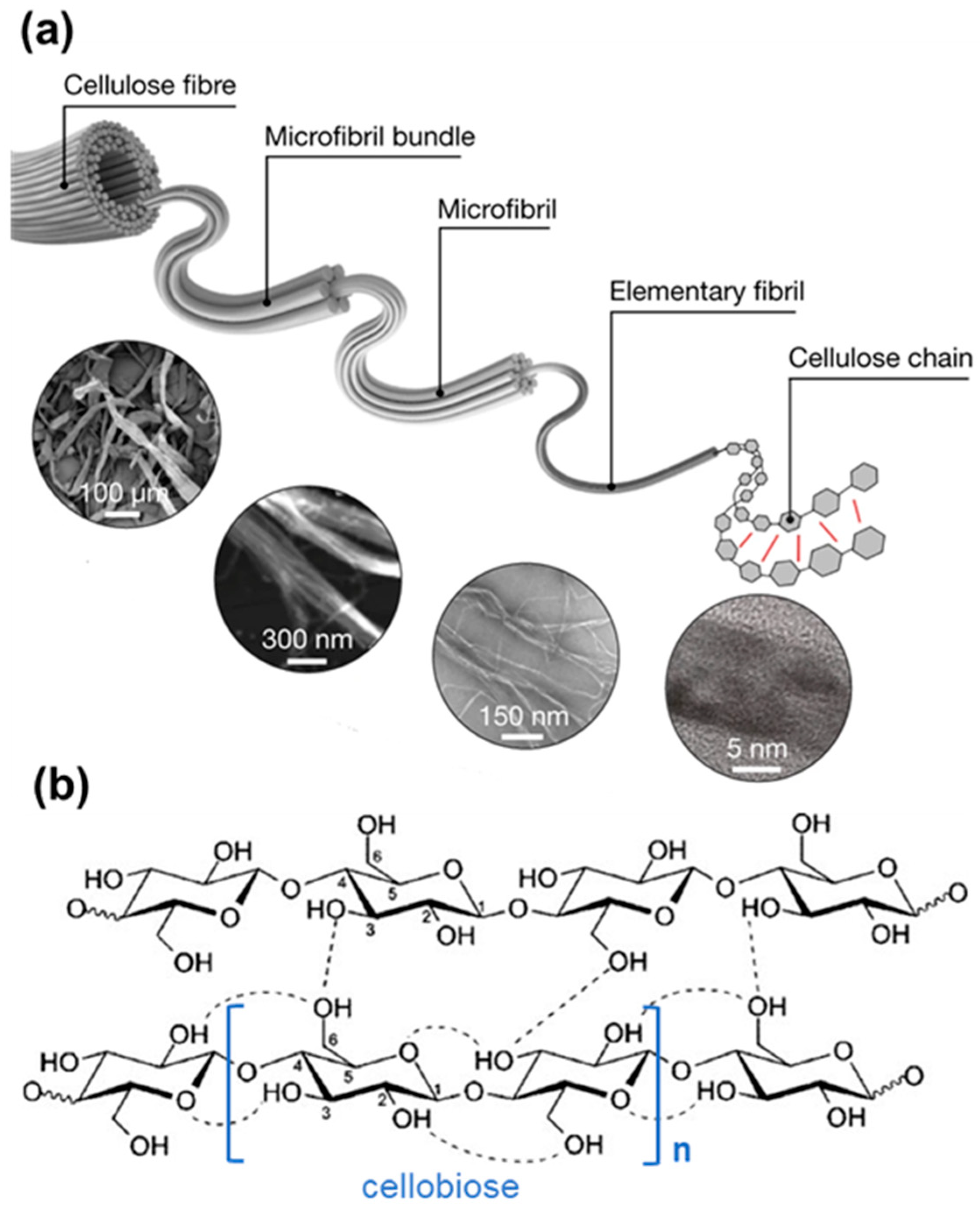
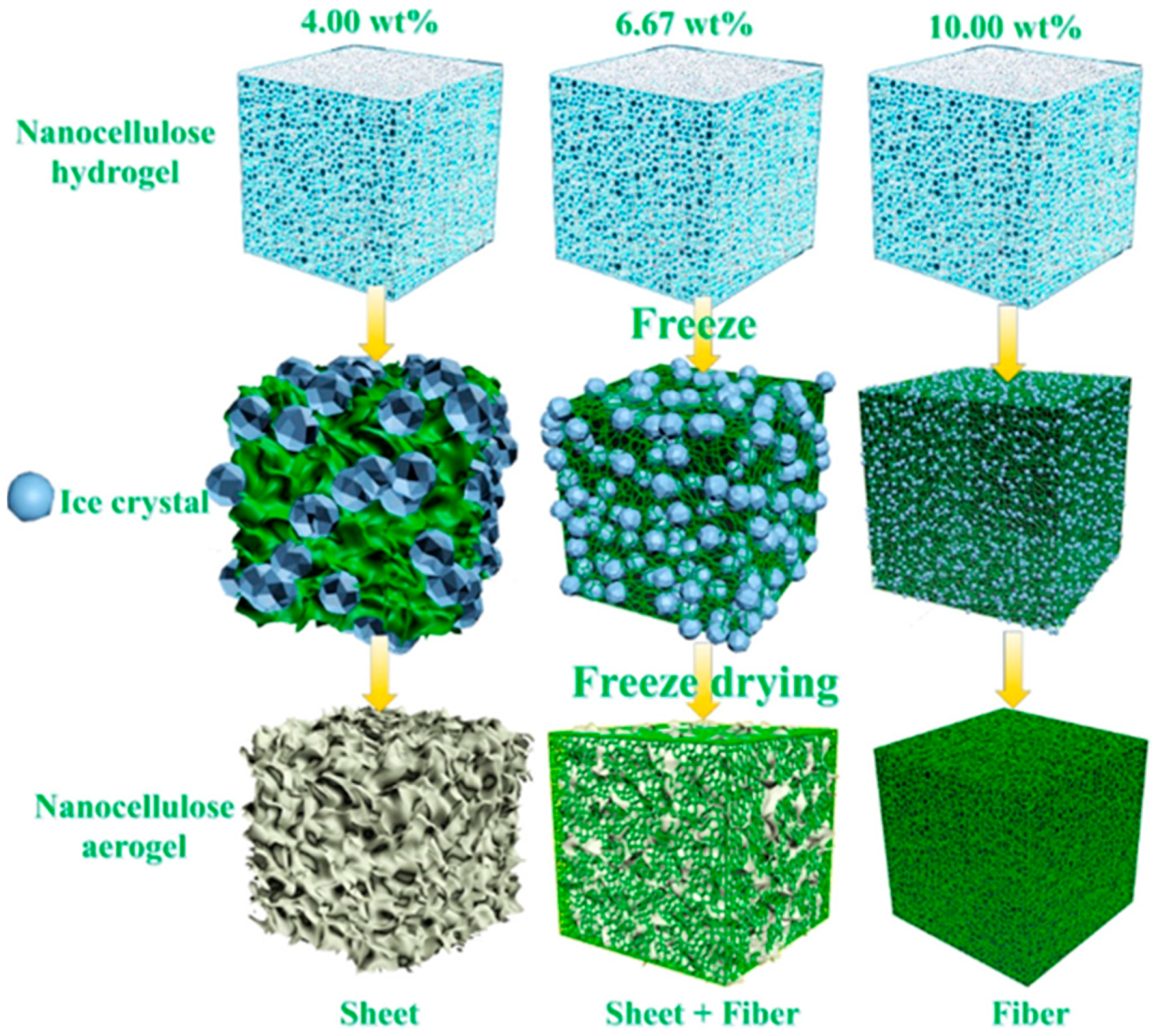
2. Cellulosic Porous Materials
3. Working Principle of Piezoresistive Sensors
3.1. Pressure Sensor
3.2. Strain Sensor
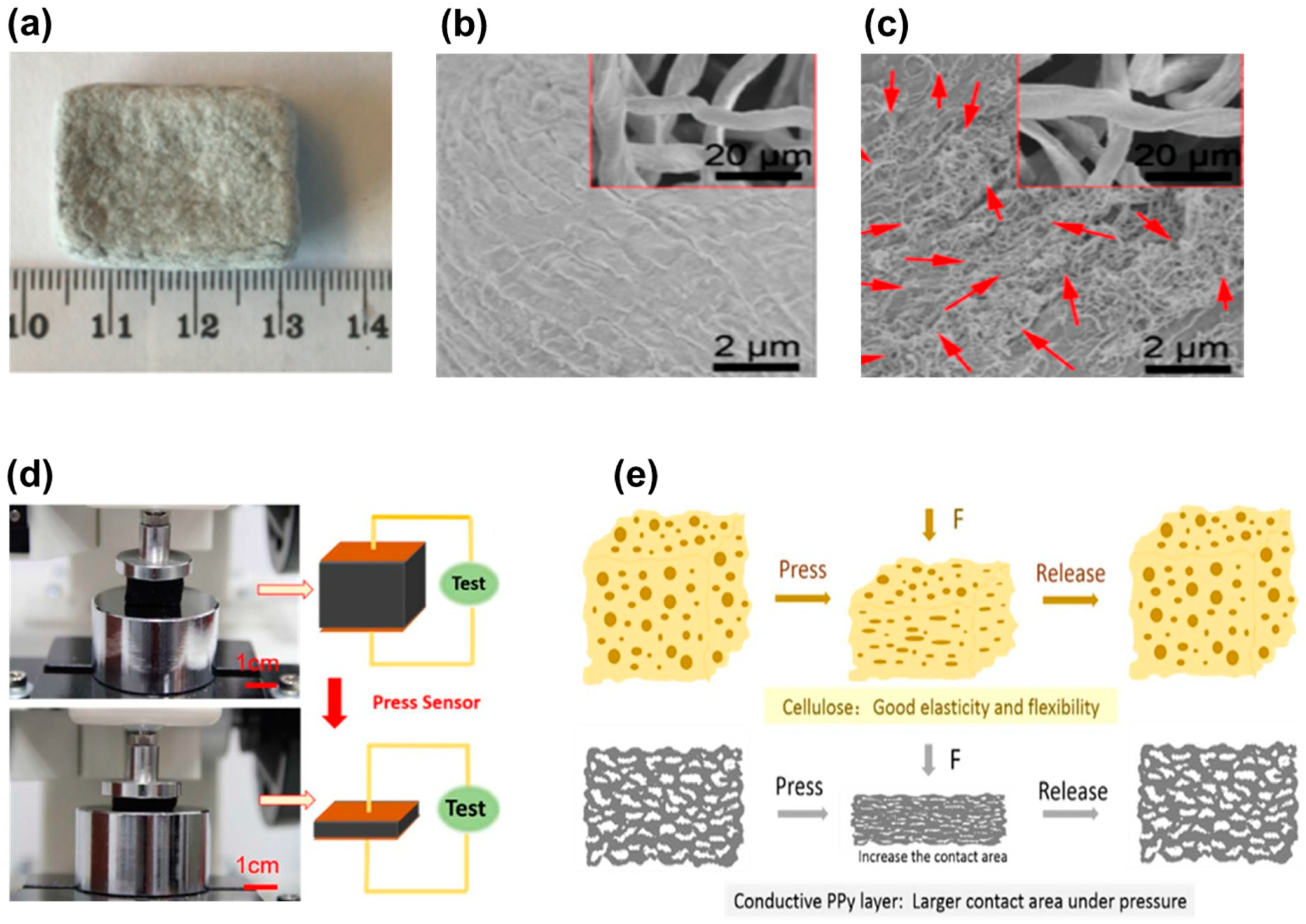
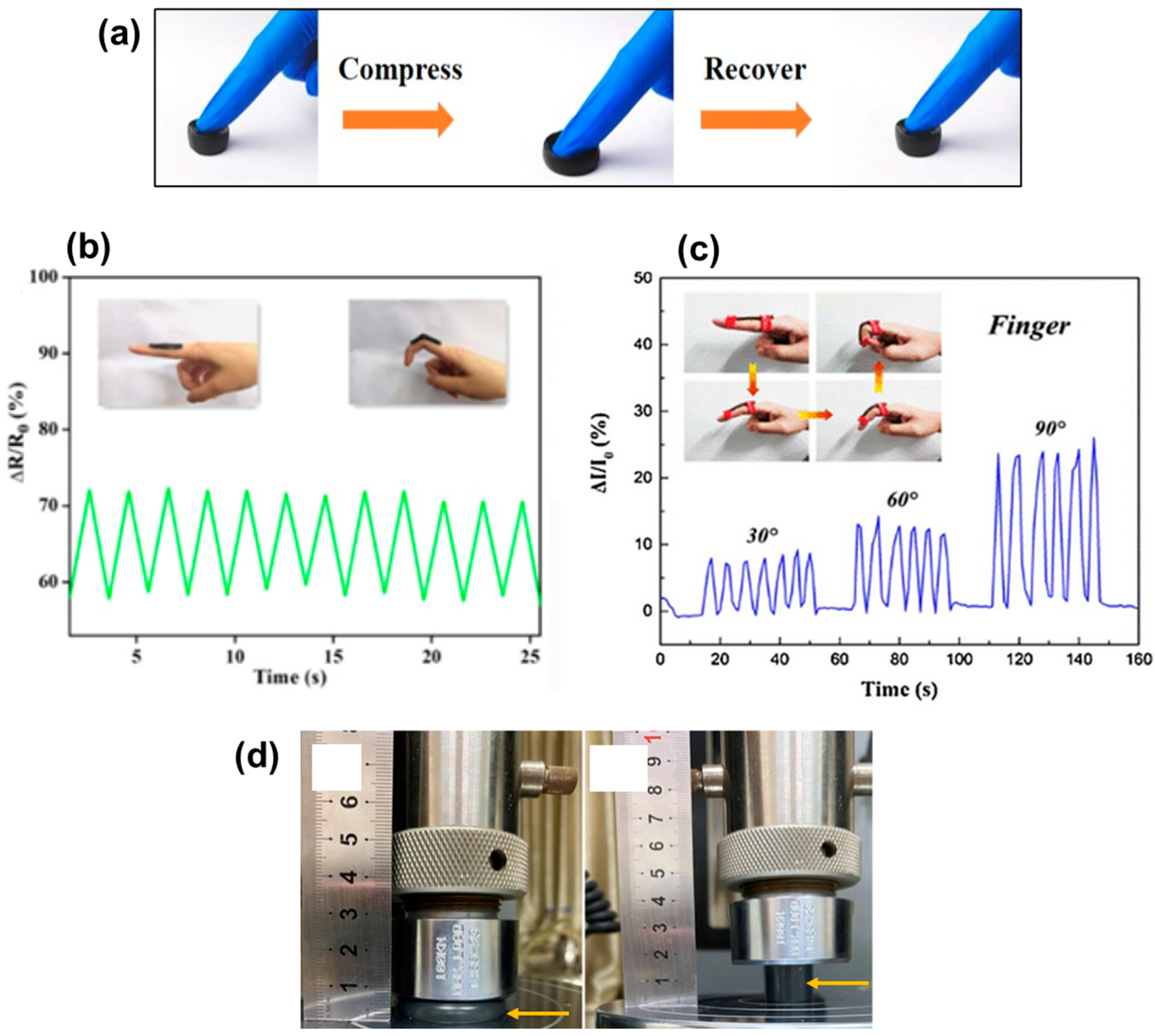
4. Cellulosic Air-Porous Materials for Pressure Sensors
5. Cellulosic Hydrogels for Pressure Sensors
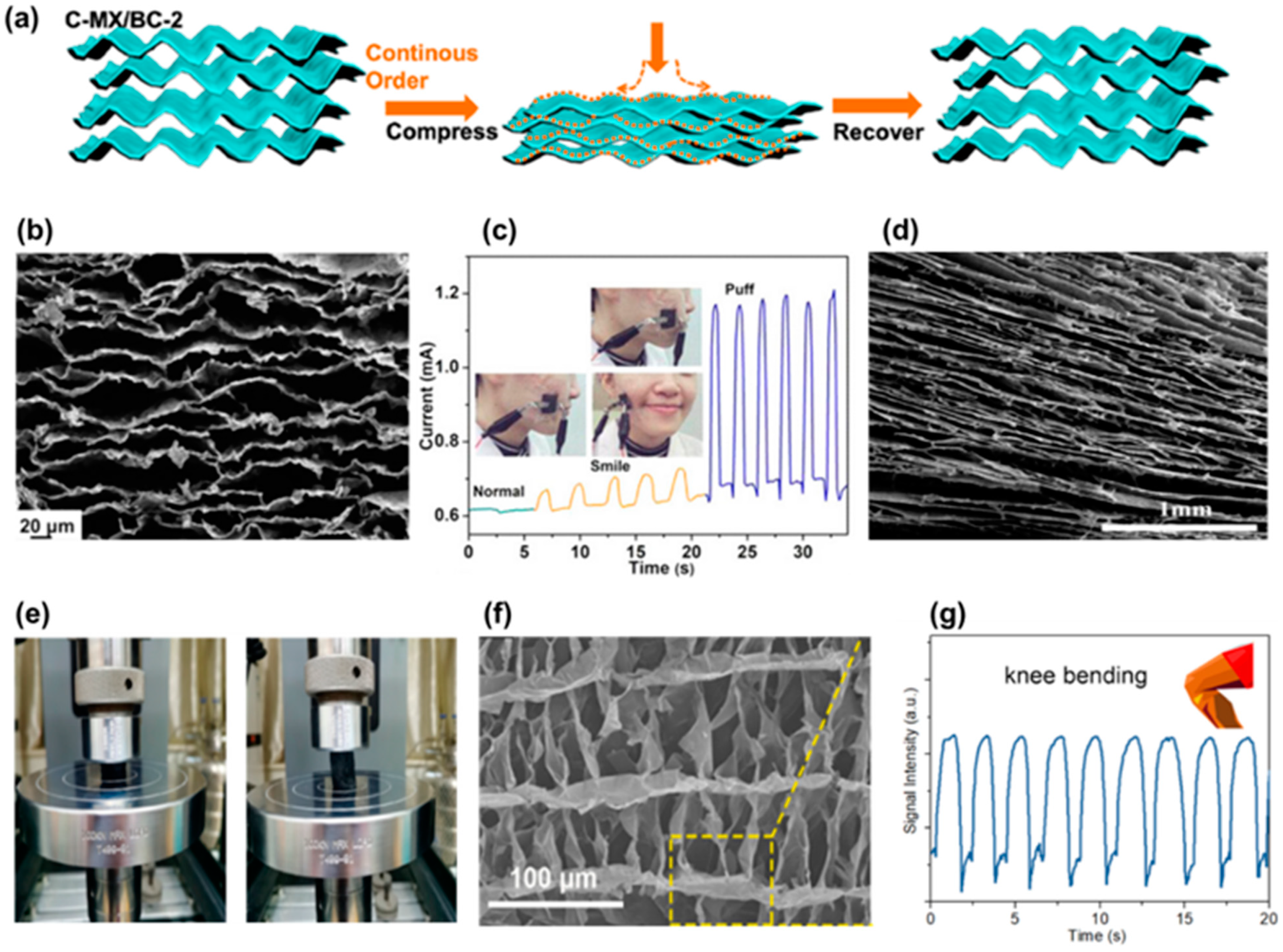
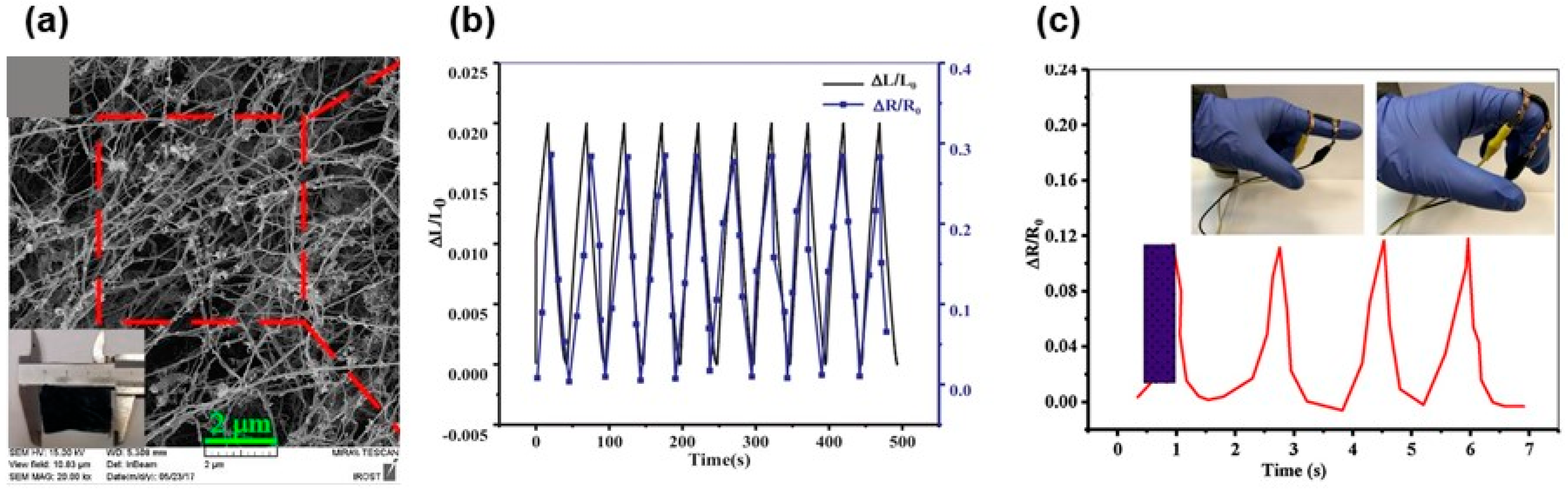
6. Cellulosic Air-Porous Materials for Strain Sensors
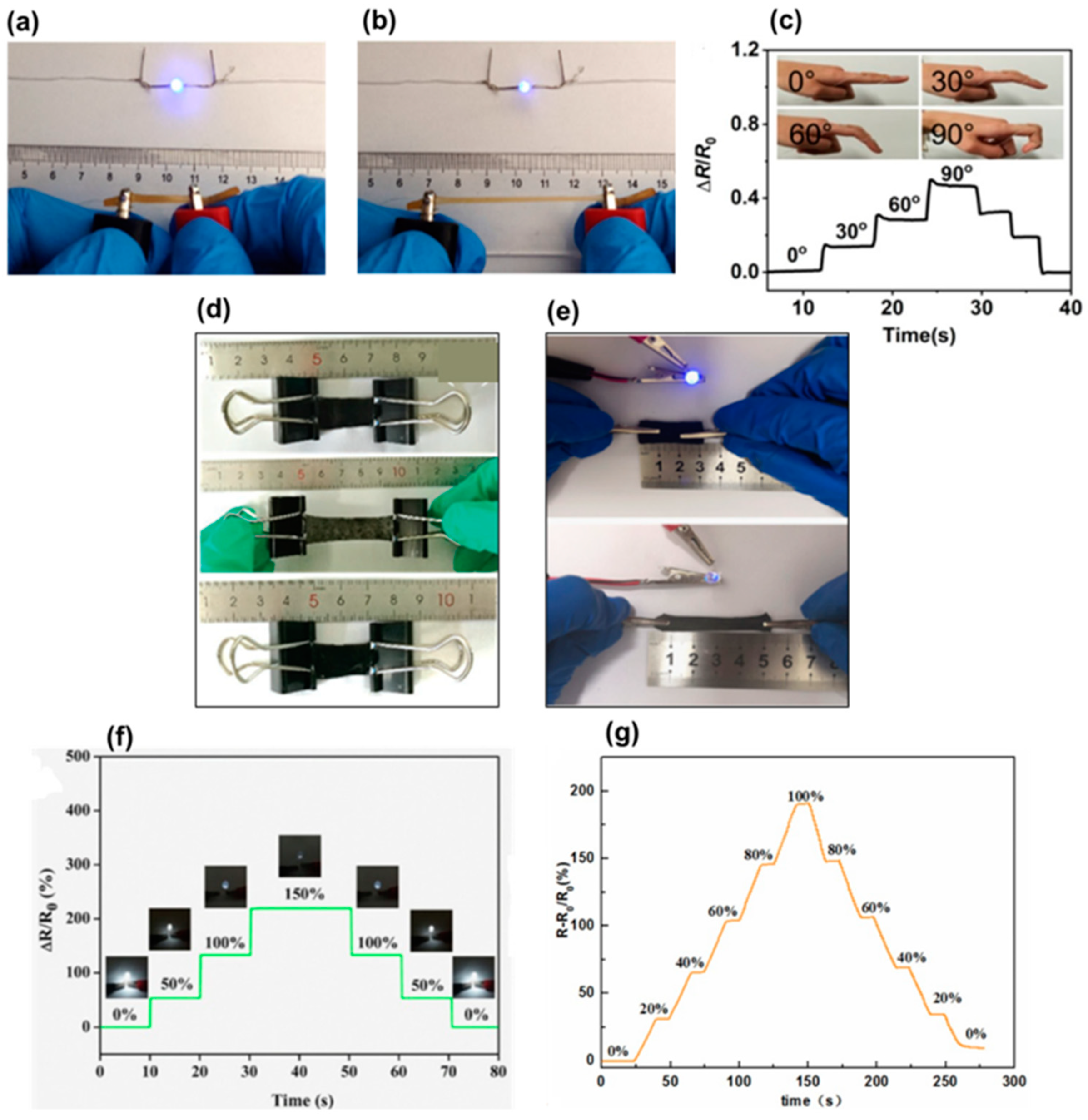
7. Cellulosic Hydrogels for Strain Sensors
8. Outlook and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Sreenilayam, S.P.; Ahad, I.U.; Nicolosi, V.; Acinas Garzon, V.; Brabazon, D. Advanced materials of printed wearables for physiological parameter monitoring. Mater. Today 2020, 32, 147–177. [Google Scholar] [CrossRef]
- Nasiri, S.; Khosravani, M.R. Progress and challenges in fabrication of wearable sensors for health monitoring. Sens. Actuators A Phys. 2020, 312, 112105. [Google Scholar] [CrossRef]
- Mordor Intelligence. WEARABLE SENSORS MARKET-GROWTH, TRENDS, COVID-19 IMPACT, AND FORECASTS (2022–2027); Mortor Intelligence: Hyderabad, India, 2020. [Google Scholar]
- IDTechEx. No Wearable Sensors 2018–2028: Technologies, Markets & PlayersTitle; IDTechEx: Cambridge, UK, 2020. [Google Scholar]
- Duan, L.; D’hooge, D.R.; Cardon, L. Recent progress on flexible and stretchable piezoresistive strain sensors: From design to application. Prog. Mater. Sci. 2020, 114, 100617. [Google Scholar] [CrossRef]
- Wang, X.; Yu, J.; Cui, Y.; Li, W. Research progress of flexible wearable pressure sensors. Sens. Actuators A Phys. 2021, 330, 112838. [Google Scholar] [CrossRef]
- Wang, X.; Liu, Z.; Zhang, T. Flexible Sensing Electronics for Wearable/Attachable Health Monitoring. Small 2017, 13, 1602790. [Google Scholar] [CrossRef]
- Trung, T.Q.; Lee, N.-E. Flexible and Stretchable Physical Sensor Integrated Platforms for Wearable Human-Activity Monitoringand Personal Healthcare. Adv. Mater. 2016, 28, 4338–4372. [Google Scholar] [CrossRef]
- Nesser, H.; Lubineau, G. Strain Sensing by Electrical Capacitive Variation: From Stretchable Materials to Electronic Interfaces. Adv. Electron. Mater. 2021, 7, 2100190. [Google Scholar] [CrossRef]
- Chen, J.; Zheng, J.; Gao, Q.; Zhang, J.; Zhang, J.; Omisore, O.M.; Wang, L.; Li, H. Polydimethylsiloxane (PDMS)-Based Flexible Resistive Strain Sensors for Wearable Applications. Appl. Sci. 2018, 8, 345. [Google Scholar] [CrossRef] [Green Version]
- Gao, Y.; Liu, D.; Xie, Y.; Song, Y.; Zhu, E.; Shi, Z.; Yang, Q.; Xiong, C. Flexible and sensitive piezoresistive electronic skin based on TOCN/PPy hydrogel films. J. Appl. Polym. Sci. 2021, 138, 51367. [Google Scholar] [CrossRef]
- Fu, Q.; Cui, C.; Meng, L.; Hao, S.; Dai, R.; Yang, J. Emerging cellulose-derived materials: A promising platform for the design of flexible wearable sensors toward health and environment monitoring. Mater. Chem. Front. 2021, 5, 2051–2091. [Google Scholar] [CrossRef]
- Zhai, J.; Zhang, Y.; Cui, C.; Li, A.; Wang, W.; Guo, R.; Qin, W.; Ren, E.; Xiao, H.; Zhou, M. Flexible Waterborne Polyurethane/Cellulose Nanocrystal Composite Aerogels by Integrating Graphene and Carbon Nanotubes for a Highly Sensitive Pressure Sensor. ACS Sustain. Chem. Eng. 2021, 9, 14029–14039. [Google Scholar] [CrossRef]
- Zhang, H.; Sun, X.; Hubbe, M.; Pal, L. Flexible and Pressure-Responsive Sensors from Cellulose Fibers Coated with Multiwalled Carbon Nanotubes. ACS Appl. Electron. Mater. 2019, 1, 1179–1188. [Google Scholar] [CrossRef]
- Wang, S.; Xiang, J.; Sun, Y.; Wang, H.; Du, X.; Cheng, X.; Du, Z.; Wang, H. Skin-inspired nanofibrillated cellulose-reinforced hydrogels with high mechanical strength, long-term antibacterial, and self-recovery ability for wearable strain/pressure sensors. Carbohydr. Polym. 2021, 261, 117894. [Google Scholar] [CrossRef]
- Chen, C.; Wang, Y.; Wu, Q.; Wan, Z.; Li, D.; Jin, Y. Highly strong and flexible composite hydrogel reinforced by aligned wood cellulose skeleton via alkali treatment for muscle-like sensors. Chem. Eng. J. 2020, 400, 125876. [Google Scholar] [CrossRef]
- Hosseini, H.; Kokabi, M.; Mousavi, S.M. Conductive bacterial cellulose/multiwall carbon nanotubes nanocomposite aerogel as a potentially flexible lightweight strain sensor. Carbohydr. Polym. 2018, 201, 228–235. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Wu, X.; Qin, Z.; Sun, X.; Zhang, H.; Yu, Q.; Yao, M.; He, S.; Dong, X.; Yao, F.; et al. Dual physically cross-linked carboxymethyl cellulose-based hydrogel with high stretchability and toughness as sensitive strain sensors. Cellulose 2020, 27, 9975–9989. [Google Scholar] [CrossRef]
- Brenes, R.G.R.; Grieco, M.A.B.; Bojorge, N.; Pereira, N. Nanocellulose: Production and Processing for Biomedical Applications. ChemNanoMat 2021. [Google Scholar] [CrossRef]
- Mali, P.; Sherje, A.P. Cellulose nanocrystals: Fundamentals and biomedical applications. Carbohydr. Polym. 2022, 275, 118668. [Google Scholar] [CrossRef]
- Divya; Mahapatra, S.; Srivastava, V.R.; Chandra, P. Nanobioengineered Sensing Technologies Based on Cellulose Matrices for Detection of Small Molecules, Macromolecules, and Cells. Biosensors 2021, 11, 168. [Google Scholar] [CrossRef]
- Li, T.; Chen, C.; Brozena, A.H.; Zhu, J.Y.; Xu, L.; Driemeier, C.; Dai, J.; Rojas, O.J.; Isogai, A.; Wågberg, L.; et al. Developing fibrillated cellulose as a sustainable technological material. Nature 2021, 590, 47–56. [Google Scholar] [CrossRef]
- Wang, L.; Li, K.; Copenhaver, K.; Mackay, S.; Lamm, M.E.; Zhao, X.; Dixon, B.; Wang, J.; Han, Y.; Neivandt, D.; et al. Review on Nonconventional Fibrillation Methods of Producing Cellulose Nanofibrils and Their Applications. Biomacromolecules 2021, 22, 4037–4059. [Google Scholar] [CrossRef] [PubMed]
- Larsson, P.A.; Riazanova, A.V.; Cinar Ciftci, G.; Rojas, R.; Øvrebø, H.H.; Wågberg, L.; Berglund, L.A. Towards optimised size distribution in commercial microfibrillated cellulose: A fractionation approach. Cellulose 2019, 26, 1565–1575. [Google Scholar] [CrossRef] [Green Version]
- Yi, T.; Zhao, H.; Mo, Q.; Pan, D.; Liu, Y.; Huang, L.; Xu, H.; Hu, B.; Song, H. From Cellulose to Cellulose Nanofibrils—A Comprehensive Review of the Preparation and Modification of Cellulose Nanofibrils. Materials (Basel) 2020, 13, 5062. [Google Scholar] [CrossRef] [PubMed]
- Serra-Parareda, F.; Tarrés, Q.; Sanchez-Salvador, J.L.; Campano, C.; Pèlach, M.À.; Mutjé, P.; Negro, C.; Delgado-Aguilar, M. Tuning morphology and structure of non-woody nanocellulose: Ranging between nanofibers and nanocrystals. Ind. Crops Prod. 2021, 171, 113877. [Google Scholar] [CrossRef]
- Wahid, F.; Huang, L.-H.; Zhao, X.-Q.; Li, W.-C.; Wang, Y.-Y.; Jia, S.-R.; Zhong, C. Bacterial cellulose and its potential for biomedical applications. Biotechnol. Adv. 2021, 53, 107856. [Google Scholar] [CrossRef]
- Mokhena, T.C.; John, M.J. Cellulose nanomaterials: New generation materials for solving global issues. Cellulose 2020, 27, 1149–1194. [Google Scholar] [CrossRef]
- Zhong, C. Industrial-Scale Production and Applications of Bacterial Cellulose. Front. Bioeng. Biotechnol. 2020, 8, 1425. [Google Scholar] [CrossRef]
- Wei, Z.; Wu, C.; Li, R.; Yu, D.; Ding, Q. Nanocellulose based hydrogel or aerogel scaffolds for tissue engineering. Cellulose 2021, 28, 7497–7520. [Google Scholar] [CrossRef]
- De France, K.J.; Hoare, T.; Cranston, E.D. Review of Hydrogels and Aerogels Containing Nanocellulose. Chem. Mater. 2017, 29, 4609–4631. [Google Scholar] [CrossRef]
- Sun, Y.; Chu, Y.; Wu, W.; Xiao, H. Nanocellulose-based lightweight porous materials: A review. Carbohydr. Polym. 2021, 255, 117489. [Google Scholar] [CrossRef]
- Huang, D.; Wu, M.; Wang, C.; Kuga, S.; Huang, Y. Effect of Partial Dehydration on Freeze-Drying of Aqueous Nanocellulose Suspension. ACS Sustain. Chem. Eng. 2020, 8, 11389–11395. [Google Scholar] [CrossRef]
- Ganesan, K.; Barowski, A.; Ratke, L.; Milow, B. Influence of hierarchical porous structures on the mechanical properties of cellulose aerogels. J. Sol-Gel Sci. Technol. 2019, 89, 156–165. [Google Scholar] [CrossRef]
- Isobe, N.; Komamiya, T.; Kimura, S.; Kim, U.-J.; Wada, M. Cellulose hydrogel with tunable shape and mechanical properties: From rigid cylinder to soft scaffold. Int. J. Biol. Macromol. 2018, 117, 625–631. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Zhang, Y.; Wang, X.; Liu, S.; Yao, Y. Characterization of the nano-cellulose aerogel from mixing CNF and CNC with different ratio. Mater. Lett. 2018, 229, 103–106. [Google Scholar] [CrossRef]
- Teodoro, K.B.R.; Sanfelice, R.C.; Migliorini, F.L.; Pavinatto, A.; Facure, M.H.M.; Correa, D.S. A Review on the Role and Performance of Cellulose Nanomaterials in Sensors. ACS Sens. 2021, 6, 2473–2496. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Pan, Z.-Z.; Lv, W.; Liu, B.; Wei, J.; Lv, X.; Luo, Y.; Nishihara, H.; Yang, Q.-H. A Directional Strain Sensor Based on Anisotropic Microhoneycomb Cellulose Nanofiber-Carbon Nanotube Hybrid Aerogels Prepared by Unidirectional Freeze Drying. Small 2019, 15, 1805363. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Hu, Y.; Zhuo, H.; Liu, L.; Jing, S.; Zhong, L.; Peng, X.; Sun, R. Compressible, Elastic, and Pressure-Sensitive Carbon Aerogels Derived from 2D Titanium Carbide Nanosheets and Bacterial Cellulose for Wearable Sensors. Chem. Mater. 2019, 31, 3301–3312. [Google Scholar] [CrossRef]
- Chen, S.; Chen, Y.; Li, D.; Xu, Y.; Xu, F. Flexible and Sensitivity-Adjustable Pressure Sensors Based on Carbonized Bacterial Nanocellulose/Wood-Derived Cellulose Nanofibril Composite Aerogels. ACS Appl. Mater. Interfaces 2021, 13, 8754–8763. [Google Scholar] [CrossRef]
- Luo, M.; Li, M.; Li, Y.; Chang, K.; Liu, K.; Liu, Q.; Wang, Y.; Lu, Z.; Liu, X.; Wang, D. In-situ polymerization of PPy/cellulose composite sponge with high elasticity and conductivity for the application of pressure sensor. Compos. Commun. 2017, 6, 68–72. [Google Scholar] [CrossRef]
- Li, M.; Tu, Q.; Long, X.; Zhang, Q.; Jiang, H.; Chen, C.; Wang, S.; Min, D. Flexible conductive hydrogel fabricated with polyvinyl alcohol, carboxymethyl chitosan, cellulose nanofibrils, and lignin-based carbon applied as strain and pressure sensor. Int. J. Biol. Macromol. 2021, 166, 1526–1534. [Google Scholar] [CrossRef]
- Pi, M.; Jiang, L.; Wang, Z.; Cui, W.; Shi, L.; Ran, R. Robust and ultrasensitive hydrogel sensors enhanced by MXene/cellulose nanocrystals. J. Mater. Sci. 2021, 56, 8871–8886. [Google Scholar] [CrossRef]
- Huang, J.; Zhao, M.; Cai, Y.; Zimniewska, M.; Li, D.; Wei, Q. A Dual-Mode Wearable Sensor Based on Bacterial Cellulose Reinforced Hydrogels for Highly Sensitive Strain/Pressure Sensing. Adv. Electron. Mater. 2020, 6, 1900934. [Google Scholar] [CrossRef]
- Hu, J.; Wu, Y.; Yang, Q.; Zhou, Q.; Hui, L.; Liu, Z.; Xu, F.; Ding, D. One-pot freezing-thawing preparation of cellulose nanofibrils reinforced polyvinyl alcohol based ionic hydrogel strain sensor for human motion monitoring. Carbohydr. Polym. 2022, 275, 118697. [Google Scholar] [CrossRef] [PubMed]
- Qin, H.; Chen, Y.; Huang, J.; Wei, Q. Bacterial Cellulose Reinforced Polyaniline Electroconductive Hydrogel with Multiple Weak H-Bonds as Flexible and Sensitive Strain Sensor. Macromol. Mater. Eng. 2021, 306, 2100159. [Google Scholar] [CrossRef]


| Material Type | Conductive Material | Cellulose Type | Sensitivity (kPa−1) | Detection Limit (kPa) | Response (ms) | Recovery (ms) | Cyclic Stability | Human Body Monitoring | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| Air-porous | MXene | Bacterial cellulose | 12.5 | - | 167 | 121 | 100,000 (50% strain) | Finger bending Wrist bending Elbow bending Face expression Arm pulse Jugular venous pulse | [39] |
| CNT/Graphene | CNC | 0.25 | 0.112 | - | - | - | Finger bending Wrist bending Elbow bending Squatting Walking Running | [13] | |
| Carbonized bacterial cellulose | TEMPO CNF | 0.003–0.358 | 0.0025 | 50 | 110 | 10,000 | Wrist bending Knee bending Finger touch Breath blow | [40] | |
| Multiwalled carbon nanotube (MWCNT) | Cotton balls | 0.0159–0.0197 | - | 20 | 20 | - | Finger compression | [14] | |
| Polypyrrole | Cellulose microcrystalline | 58.9 | - | - | - | 10 | - | [41] | |
| Hydrogel | Lignin carbon | CNF | - | - | - | - | - | Finger bending Elbow bending Palm gripping | [42] |
| Silver nanoparticle | TEMPO CNF | 9.5 | - | - | - | 1000 | Face expression Elbow bending Neck forward Walking Jumping | [15] | |
| MXene | CNC | - | - | - | - | - | - | [43] |
| Material Type | Conductive Material | Cellulose Type | Gauge Factor | Response (ms) | Cyclic Stability | Human Body Monitoring | Ref. |
|---|---|---|---|---|---|---|---|
| Air-porous | Multiwalled carbon nanotube (MWCNT) | Bacterial cellulose | 21 | 390 | 1000 | Finger bending | [17] |
| Hydrogel | Na+ Fe3+ | Carboxymethyl cellulose | 1.99 (0–50%) 4.02 (50–600%) | 260 | - | Finger bending Wrist bending Elbow bending Swallowing Speaking | [18] |
| MWCNT Carbon black | Bacterial cellulose | 0.725 (0–60%) 2.216 (60–145.2%) 5.010 (145.2–200%) | - | - | Wrist bending Elbow bending | [44] | |
| Carbonized lignin | CNF | - | - | - | Finger bending Wrist bending Elbow bending | [42] | |
| Zn2+ | TEMPO CNF | 1.70 | - | 500 | Finger bending Wrist bending Elbow bending Knee bending Neck movement Speaking | [45] | |
| Polyaniline | Bacterial cellulose | 0.85 | 560 | 200 | Finger bending Wrist bending Elbow bending Knee bending | [46] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Basarir, F.; Kaschuk, J.J.; Vapaavuori, J. Perspective about Cellulose-Based Pressure and Strain Sensors for Human Motion Detection. Biosensors 2022, 12, 187. https://doi.org/10.3390/bios12040187
Basarir F, Kaschuk JJ, Vapaavuori J. Perspective about Cellulose-Based Pressure and Strain Sensors for Human Motion Detection. Biosensors. 2022; 12(4):187. https://doi.org/10.3390/bios12040187
Chicago/Turabian StyleBasarir, Fevzihan, Joice Jaqueline Kaschuk, and Jaana Vapaavuori. 2022. "Perspective about Cellulose-Based Pressure and Strain Sensors for Human Motion Detection" Biosensors 12, no. 4: 187. https://doi.org/10.3390/bios12040187
APA StyleBasarir, F., Kaschuk, J. J., & Vapaavuori, J. (2022). Perspective about Cellulose-Based Pressure and Strain Sensors for Human Motion Detection. Biosensors, 12(4), 187. https://doi.org/10.3390/bios12040187




