Multi-Functional Silver Nanoparticles for High-Throughput Endospore Sensing
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
2.2. Extraction of DPA from Endospore
2.3. Synthesis of Functional Silver Nanoparticles
2.4. Characterization of Nanoparticles
3. Results and Discussion
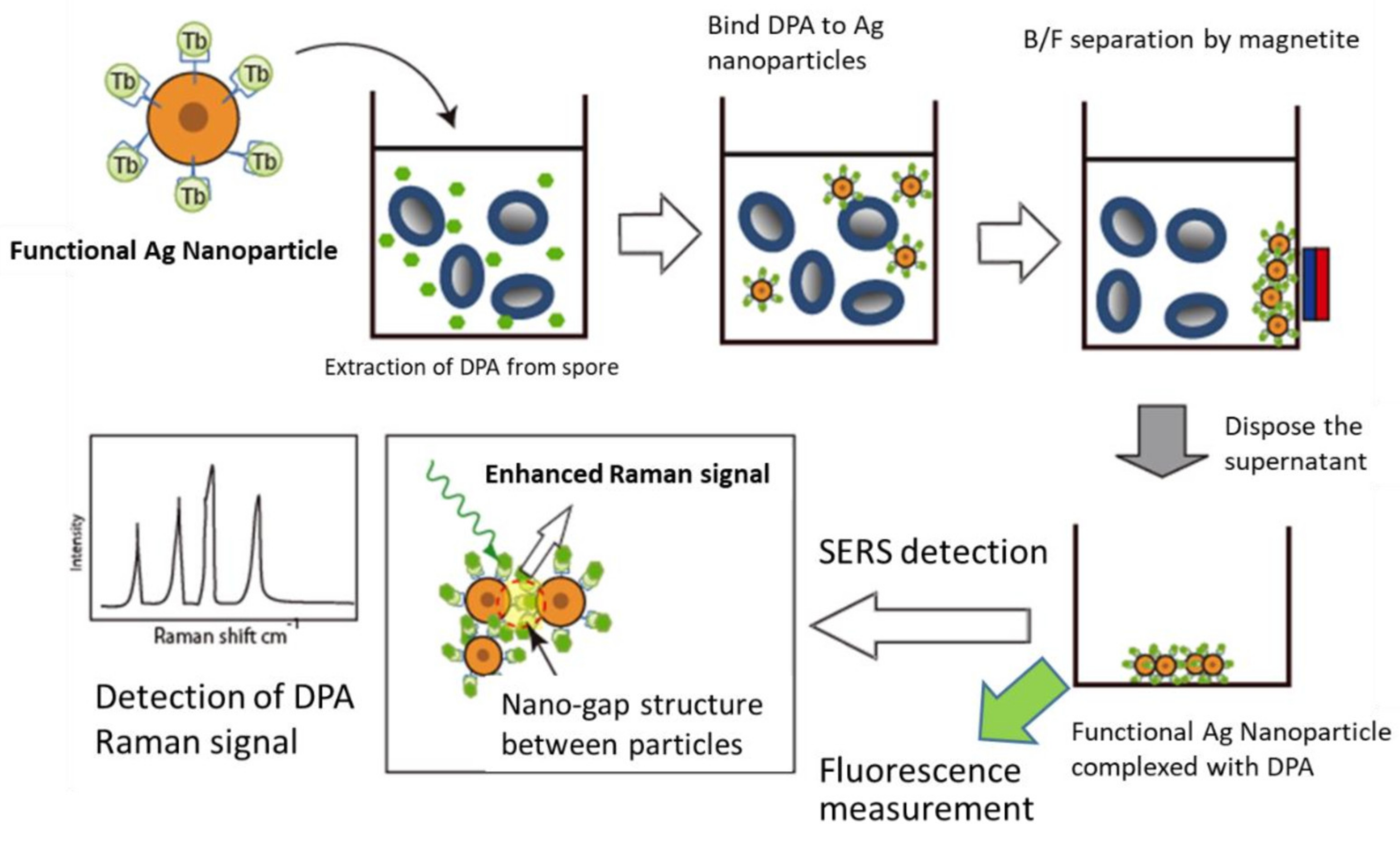
3.1. The Sensing System in This Study
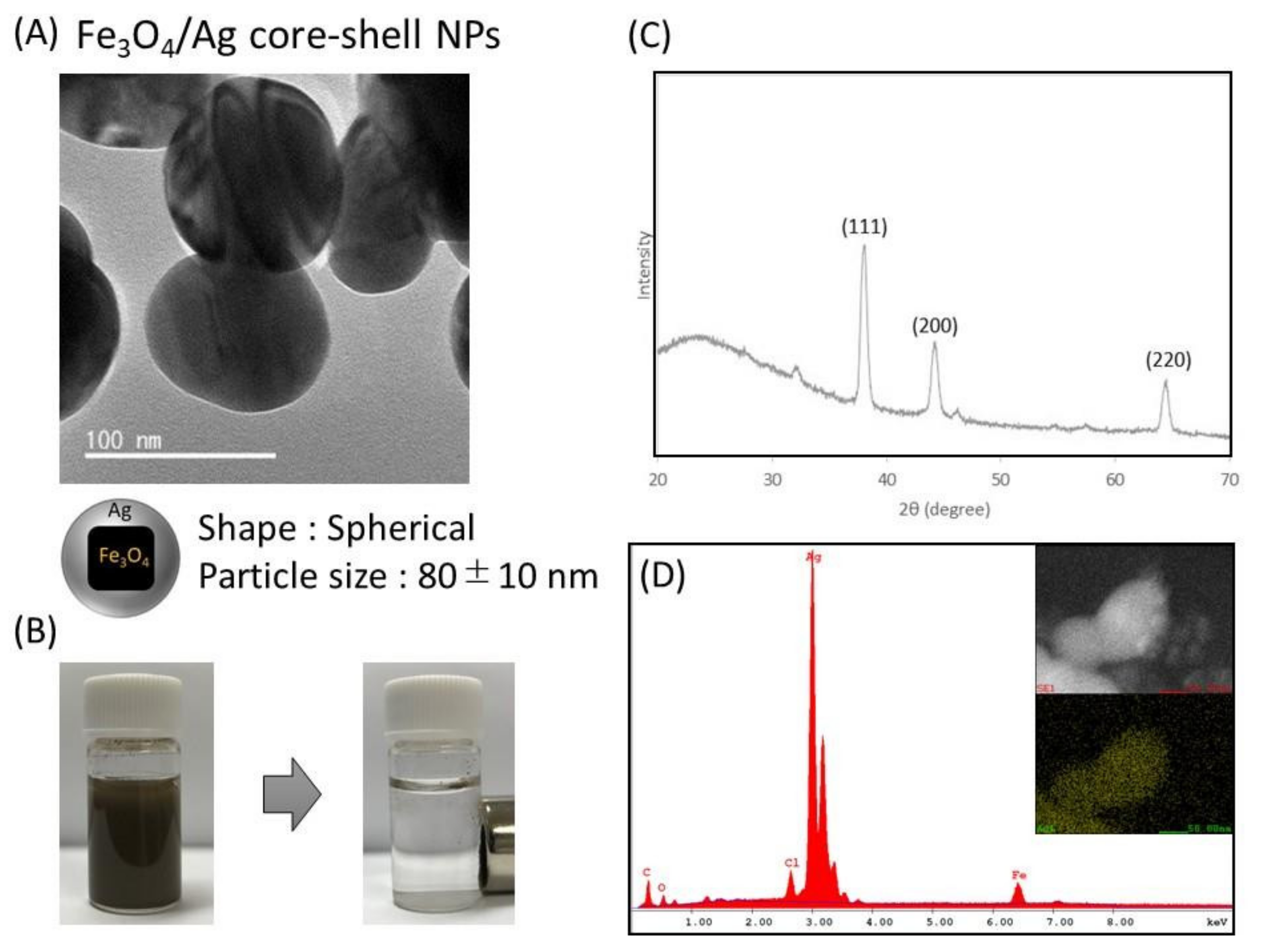
3.2. Preparation and Characterization of Functionalized Nanoparticles
3.3. Molecular Modification and Fluorescence Detection Characterization
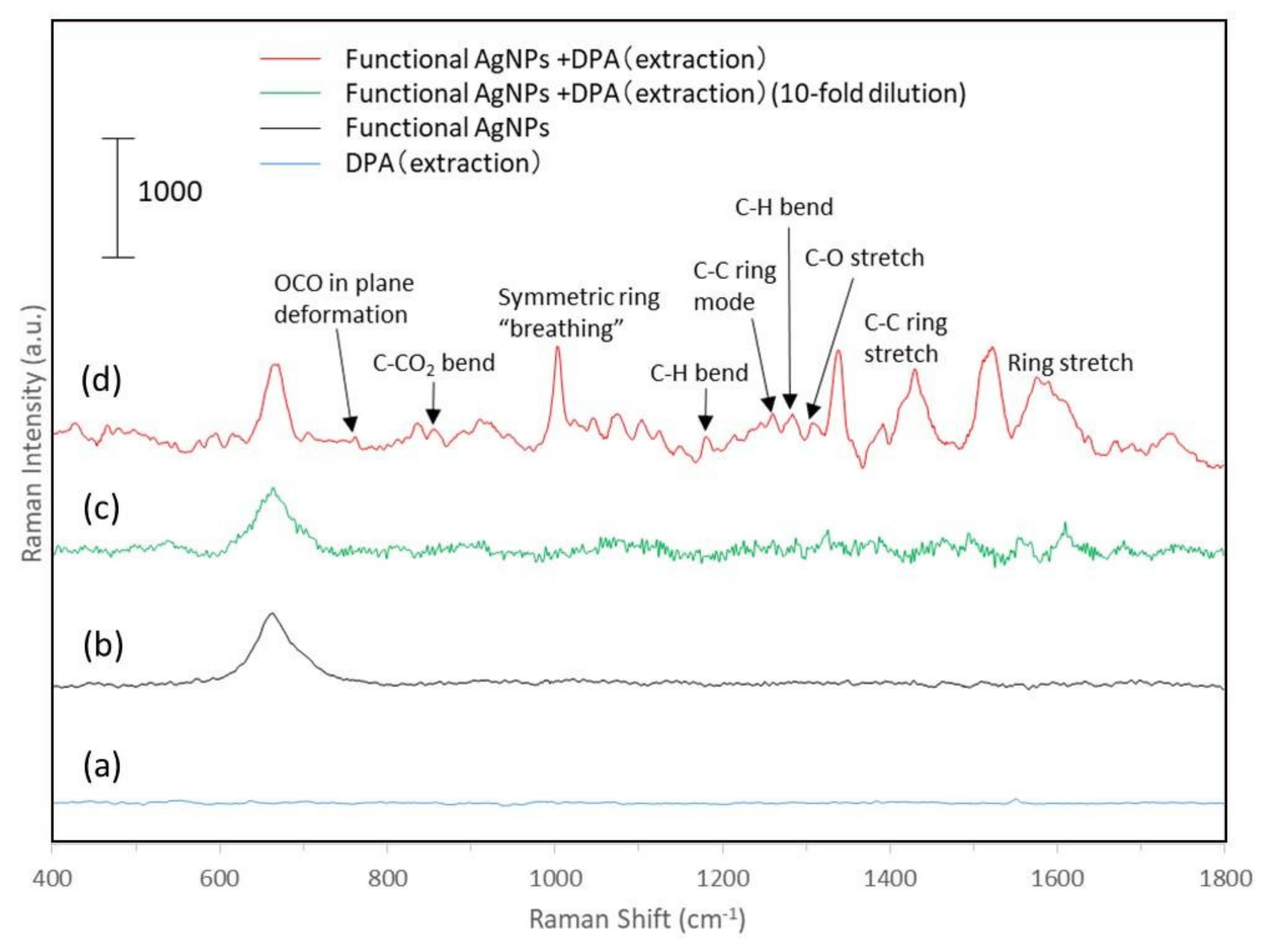
3.4. Qualitative Analysis of DPA by Functionalized Silver Nanoparticles and Raman Spectroscopy
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Crawford, L.M.; Ruff, E.H. A review of the safety of cold pasteurization through irradiation. Food Control 1996, 7, 87–97. [Google Scholar] [CrossRef]
- Aaliya, B.; Sunooj, K.V.; Navaf, M.; Akhila, P.P.; Sudheesh, C.; Mir, S.A.; Sabu, S.; Sasidharan, A.; Hlaing, M.T.; George, J. Recent trends in bacterial decontamination of food products by hurdle technology: A synergistic approach using thermal and non-thermal processing techniques. Food Res. Int. 2021, 147, 110514. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, W.L.; Munakata, N.; Horneck, G.; Melosh, H.J.; Setlow, P. Resistance of Bacillus Endospores to Extreme Terrestrial and Extraterrestrial Environments. Microbiol. Mol. Biol. Rev. 2000, 64, 548–572. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Setlow, P. Spores of Bacillus subtilis: Their resistance to and killing by radiation, heat and chemicals. J. Appl. Microbiol. 2006, 101, 514–525. [Google Scholar] [CrossRef]
- Slieman, T.A.; Nicholson, W.L. Role of dipicolinic acid in survival of Bacillus subtilis spores exposed to artificial and solar UV radiation. Appl. Environ.Microbiol. 2001, 67, 1274–1279. [Google Scholar] [CrossRef] [Green Version]
- Goepfert, J.M.; Spira, W.M.; Kim, H.U. Bacillus Cereus: Food poisoning organism. A review. J. Milk Food Technol. 1972, 35, 213–227. [Google Scholar] [CrossRef]
- Tewari, A.; Abdullah, S. Bacillus cereus food poisoning: International and Indian perspective. J. Food Sci. Technol. 2015, 52, 2500–2511. [Google Scholar] [CrossRef] [Green Version]
- Guinebretiere, M.H.; Girardin, H.; Dargaignaratz, C.; Carlin, F.; Nguyen, C. Contamination flows of Bacillus cereus and spore-forming aerobic bacteria in a cooked, pasteurised and chilled zucchini purée processing line. Int. J. Food Microbiol. 2003, 82, 223–232. [Google Scholar] [CrossRef]
- Peck, M.W. Clostridium botulinum and the safety of minimally heated, chilled foods: An emerging issue? J. Appl. Microbiol. 2006, 101, 556–570. [Google Scholar] [CrossRef]
- Lewis, J.C. Determination of dipicolinic acid in bacterial spores by ultraviolet spectrometry of the calcium chelate. Anal. Biochem. 1967, 19, 327–337. [Google Scholar] [CrossRef]
- Warth, A.D. Determination of dipicolinic acid in bacterial spores by derivative spectroscopy. Anal. Biochem. 1983, 130, 502–505. [Google Scholar] [CrossRef]
- Narula, A.; Rao, C.P. Fluorophoric Conjugate of N-Alkyl Naphthalimide in Sodium Dodecyl Sulfate As a Tunable and Sustainable Sensing System: Differential Sensing of Zn2+ and Al3+ and the Application of Its Zn2+ Complex in Detecting Dipicolinic Acid, a Component of Anthrax Bacterial Endospores. J. Phys. Chem. C 2019, 123, 21271–21280. [Google Scholar]
- Hindle, A.A.; Hall, E.A. Dipicolinic acid (DPA) assay revisited and appraised for spore detection. Analyst 1999, 124, 1599–1604. [Google Scholar] [CrossRef] [PubMed]
- Shafaat, H.S.; Ponce, A. Applications of a rapid endospore viability assay for monitoring UV inactivation and characterizing arctic ice cores. Appl. Environ. Microbiol. 2006, 72, 6808–6814. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Navarro, A.K.; Pena, A.; Perez-Guevara, F. Endospore dipicolinic acid detection during Bacillus thuringiensis culture. Lett. Appl. Microbiol. 2008, 46, 166–170. [Google Scholar] [CrossRef]
- Rosen, D.L.; Sharpless, C.; McGown, L.B. Bacterial spore detection and determination by use of terbium dipicolinate photoluminescence. Anal. Chem. 1997, 69, 1082–1085. [Google Scholar] [CrossRef]
- Donmez, M.; Yilmaz, M.D.; Kilbas, B. Fluorescent detection of dipicolinic acid as a biomarker of bacterial spores using lanthanide-chelated gold nanoparticles. J. Hazard. Mater. 2017, 324, 593–598. [Google Scholar] [CrossRef] [PubMed]
- Tan, H.; Li, Q.; Ma, C.; Song, Y.; Xu, F.; Chen, S.; Wang, L. Lanthanide-functionalized silver nanoparticles for detection of an anthrax biomarker and test paper fabrication. J. Nanopart. Res. 2014, 16, 2151. [Google Scholar] [CrossRef]
- Cable, M.L.; Kirby, J.P.; Sorasaenee, K.; Gray, H.B.; Ponce, A. Bacterial spore detection by [Tb3+(macrocycle)(dipicolinate)] luminescence. J. Am. Chem. Soc. 2007, 129, 1474–1475. [Google Scholar] [CrossRef] [Green Version]
- Cable, M.L.; Kirby, J.P.; Levine, D.J.; Manary, M.J.; Gray, H.B.; Ponce, A. Detection of bacterial spores with lanthanide-macrocycle binary complexes. J. Am. Chem. Soc. 2009, 131, 9562–9570. [Google Scholar] [CrossRef] [Green Version]
- Fleischmann, M.; Hendra, P.J.; McQuillan, A.J. Raman spectra of pyridine adsorbed at a silver electrode. Chem. Phys. Lett. 1974, 26, 163–166. [Google Scholar] [CrossRef]
- Jeanmaire, D.L.; Van Duyne, R.P. Surface Raman spectroelectrochemistry part I. heterocyclic, aromatic, and aliphatic amines adsorbed on the anodized silver electrode. J. Electroanal. Chem. 1977, 84, 1–20. [Google Scholar] [CrossRef]
- Kneipp, K.; Wang, Y.; Kneipp, H.; Perelman, L.T.; Itzkan, I.; Dasari, R.R.; Feld, M.S. Single molecule detection using surface-enhanced Raman scattering (SERS). Phys. Rev. Lett. 1997, 78, 1667–1670. [Google Scholar] [CrossRef] [Green Version]
- Nie, S.; Emory, S.R. Probing single molecules and single nanoparticles by surface-enhanced Raman scattering. Science 1997, 275, 1102–1106. [Google Scholar] [CrossRef]
- Cheng, H.W.; Huan, S.Y.; Wu, H.L.; Shen, G.L.; Yu, R.Q. Surface-enhanced Raman spectroscopic detection of a bacteria biomarker using gold nanoparticle immobilized substrates. Anal. Chem. 2009, 81, 9902–9912. [Google Scholar] [CrossRef]
- Bell, S.E.J.; Mackle, J.N.; Sirimuthu, N.M.S. Quantitative surface-enhanced Raman spectroscopy of dipicolinic acid—Towards rapid anthrax endospore detection. Analyst 2005, 130, 545–549. [Google Scholar] [CrossRef]
- Ghiamati, E.; Manoharan, R.; Nelson, W.H.; Sperry, J.F. UV Resonance Raman spectra of Bacillus spores. Appl. Spectrosc. 1992, 46, 357–364. [Google Scholar] [CrossRef]
- Ikeno, S.; Maekawa, T. Instantaneous extraction of DPA from endospore for high-throughput endospore sensing. Nanobio Bionano 2016, 3, 12–14. [Google Scholar]
- Goon, I.Y.; Lai, L.M.H.; Lim, M.; Munroe, P.; Gooding, J.J.; Amal, R. Fabrication and dispersion of gold-shell-protected magnetite nanoparticles: Systematic control using polyethyleneimine. Chem. Mater. 2009, 21, 673–681. [Google Scholar] [CrossRef]
- Pillai, Z.S.; Prashant, V.K. What factors control the size and shape of silver nanoparticles in the citrate ion reduction method? J. Phys. Chem. B 2004, 108, 945–951. [Google Scholar] [CrossRef]
- Guo, H.; Zhang, Z.; Xing, B.; Mukherjee, A.; Musante, C.; White, J.C.; He, L. Analysis of silver nanoparticles in antimicrobial products using surface-enhanced Raman spectroscopy (SERS). Environ. Sci. Technol. 2015, 49, 4317–4324. [Google Scholar] [CrossRef] [PubMed]
- Stamplecoskie, K.G.; Scaiano, J.C.; Tiwari, V.S.; Anis, H. Optimal size of silver nanoparticles for surface-enhanced Raman spectroscopy. J. Phys. Chem. C 2011, 115, 1403–1409. [Google Scholar] [CrossRef]
- Li, Q.; Yue, D.; Lu, W.; Zhang, X.; Li, C.; Wang, Z. Hybrid luminescence materials assembled by [Ln (DPA)3]3− and mesoporous host through ion-pairing interactions with high quantum efficiencies and long lifetimes. Sci. Rep. 2015, 5, 8385. [Google Scholar] [CrossRef] [PubMed]
- Kolomenskii, A.A.; Schuessler, H.A. Raman spectra of dipicolinic acid in crystalline and liquid environments. Spectrochim. Acta Part A 2005, 61, 647–651. [Google Scholar] [CrossRef]
- Futamata, M.; Yu, Y.; Yajima, T. Elucidation of electrostatic interaction between cationic dyes and Ag nanoparticles generating enormous SERS enhancement in aqueous solution. J. Phys. Chem. C 2011, 115, 5271–5279. [Google Scholar] [CrossRef]
- Otto, A. The ‘chemical’ (electronic) contribution to surface-enhanced Raman scattering. J. Raman Spectrosc. 2005, 36, 497–509. [Google Scholar] [CrossRef]
- Vandeweyer, D.; Lievens, B.; Van Campenhout, L. Identification of bacterial endospores and targeted detection of foodborne viruses in industrially reared insects for food. Nat. Food 2020, 1, 511–516. [Google Scholar] [CrossRef]
- Kędrak-Jabłońska, A.; Budniak, S.; Szczawińska, A.; Reksa, M.; Krupa, M.; Szulowski, K. Evaluation of real-time PCR based on SYBR Green I fluorescent dye for detection of Bacillus anthracis strains in biological samples. J. Vet. Res. 2018, 62, 549–554. [Google Scholar] [CrossRef]

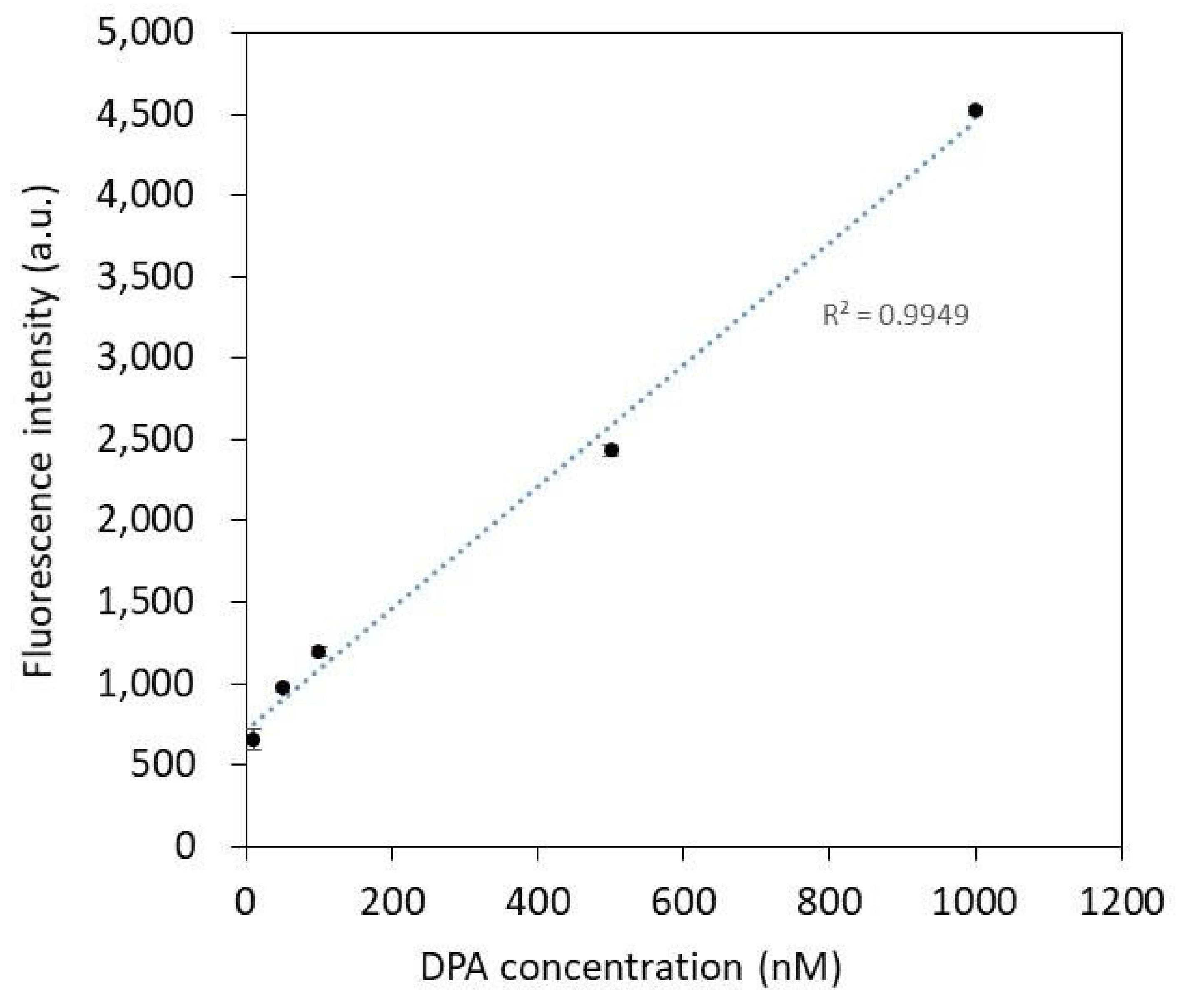


| DPA Concentration (nM) | Fluorescence Intensity (a.u.) |
|---|---|
| 0 | 604 |
| 1 | 603 |
| 5 | 608 |
| 10 | 662 |
| 50 | 974 |
| 100 | 1194 |
| 500 | 2433 |
| 1000 | 4518 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ikeno, S.; Maekawa, T.; Hara, N. Multi-Functional Silver Nanoparticles for High-Throughput Endospore Sensing. Biosensors 2022, 12, 68. https://doi.org/10.3390/bios12020068
Ikeno S, Maekawa T, Hara N. Multi-Functional Silver Nanoparticles for High-Throughput Endospore Sensing. Biosensors. 2022; 12(2):68. https://doi.org/10.3390/bios12020068
Chicago/Turabian StyleIkeno, Shinya, Takahiro Maekawa, and Noriyasu Hara. 2022. "Multi-Functional Silver Nanoparticles for High-Throughput Endospore Sensing" Biosensors 12, no. 2: 68. https://doi.org/10.3390/bios12020068
APA StyleIkeno, S., Maekawa, T., & Hara, N. (2022). Multi-Functional Silver Nanoparticles for High-Throughput Endospore Sensing. Biosensors, 12(2), 68. https://doi.org/10.3390/bios12020068





