Sniffing Out Urinary Tract Infection—Diagnosis Based on Volatile Organic Compounds and Smell Profile
Abstract
1. Introduction
2. Urinary Tract Infection
3. Current Diagnostic Methods for Urinary Tract Infection
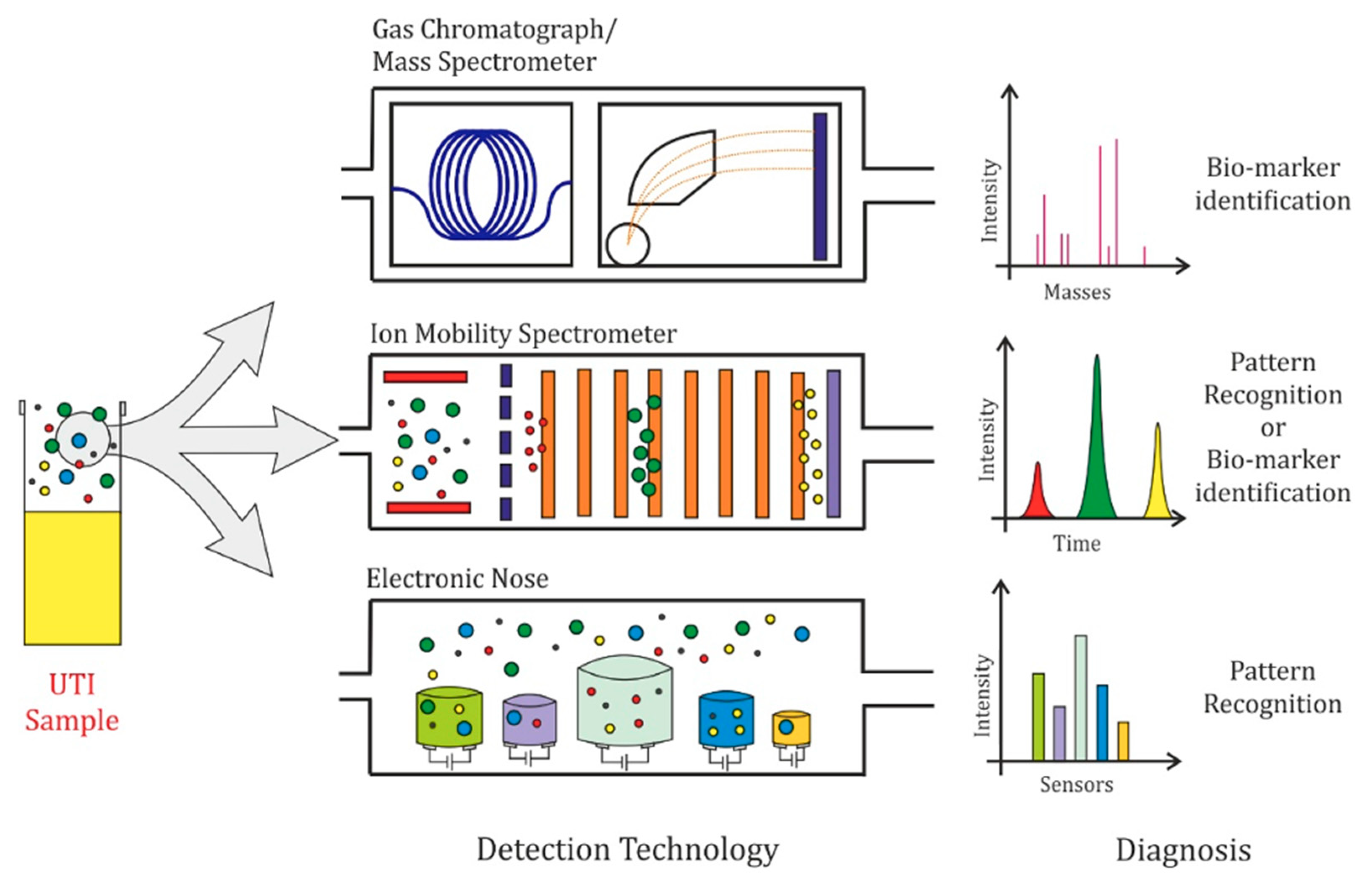
4. Volatile Organic Compounds Detection Technology and Data Analysis
5. Detection of Urinary Tract Infection Using Volatile Organic Compounds
6. Electronic Nose and Ion Mobility Spectrometry Use
7. Discussion and Conclusions
8. Methods
Author Contributions
Funding
Conflicts of Interest
References
- Flores-Mireles, A.L.; Walker, J.N.; Caparon, M.; Hultgren, S.J. Urinary tract infections: Epidemiology, mechanisms of infection and treatment options. Nat. Rev. Microbiol. 2015, 13, 269–284. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.W.; Chlebicki, M.P. Urinary tract infections in adults. Singap. Med. J. 2016, 57, 485–490. [Google Scholar] [CrossRef] [PubMed]
- Ellis, A.K.; Verma, S. Quality of Life in Women with Urinary Tract Infections: Is Benign Disease a Misnomer? J. Am. Board Fam. Med. 2000, 13, 392–397. [Google Scholar] [CrossRef] [PubMed]
- Beahm, N.P.; Nicolle, L.E.; Bursey, A.; Smyth, D.J.; Tsuyuki, R.T. The assessment and management of urinary tract infections in adults: Guidelines for pharmacists. Can. Pharm. J. Rev. Pharm. Can. 2017, 150, 298–305. [Google Scholar] [CrossRef] [PubMed]
- Griebling, T.L. Urologic diseases in America project: Trends in resource use for urinary tract infections in women. J. Urol. 2005, 173, 1288–1294. [Google Scholar] [CrossRef] [PubMed]
- Renard, J.; Ballarini, S.; Mascarenhas, T.; Zahran, M.; Quimper, E.; Choucair, J.; Iselin, C.E. Recurrent Lower Urinary Tract Infections Have a Detrimental Effect on Patient Quality of Life: A Prospective, Observational Study. Infect. Dis. Ther. 2015, 4, 125–135. [Google Scholar] [CrossRef] [PubMed]
- Najeeb, S.; Munir, T.; Rehman, S.; Hafiz, A.; Gilani, M.; Latif, M. Comparison of Urine Dipstick Test with Conventional Urine Culture in Diagnosis of Urinary Tract Infection. J. Coll. Physicians Surg. 2015, 25, 108–110. [Google Scholar]
- Simon, M.G.; Davis, C.E. Instrumentation and sensors for human breath analysis. In Advances in Biomedical Sensing, Measurements, Instrumentation and Systems; Mukhopadhyay, S.C., Lay-Ekuakille, A., Eds.; Lecture Notes in Electrical Engineering; Springer: Berlin/Heidelberg, Germany, 2010; Volume 55, pp. 144–165. ISBN 978-3-642-05166-1. [Google Scholar]
- Porter, R. The early years. In The Greatest Benefit to Mankind: A Medical History of Humanity from Antiquity to the Present; Harper Collins: London, UK, 1999; pp. 147–162. ISBN 978-0-00-637454-1. [Google Scholar]
- Wilson, A.D.; Baietto, M. Advances in Electronic-Nose Technologies Developed for Biomedical Applications. Sensors 2011, 11, 1105–1176. [Google Scholar] [CrossRef]
- Williams, H.; Pembroke, A. Sniffer Dogs in the Melanoma Clinic? Lancet 1989, 333, 734. [Google Scholar] [CrossRef]
- Willis, C.M.; Church, S.M.; Guest, C.M.; Cook, W.A.; McCarthy, N.; Bransbury, A.J.; Church, M.R.T.; Church, J.C.T. Olfactory detection of human bladder cancer by dogs: Proof of principle study. BMJ 2004, 329, 712. [Google Scholar] [CrossRef]
- McCulloch, M.; Turner, K.; Broffman, M. Lung cancer detection by canine scent: Will there be a lab in the lab? Eur. Respir. J. 2012, 39, 511–512. [Google Scholar] [CrossRef] [PubMed]
- McCulloch, M.; Jezierski, T.; Broffman, M.; Hubbard, A.; Turner, K.; Janecki, T. Diagnostic Accuracy of Canine Scent Detection in Early- and Late-Stage Lung and Breast Cancers. Integr. Cancer Ther. 2006, 5, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Cornu, J.-N.; Cancel-Tassin, G.; Ondet, V.; Girardet, C.; Cussenot, O. Olfactory Detection of Prostate Cancer by Dogs Sniffing Urine: A Step Forward in Early Diagnosis. Eur. Urol. 2011, 59, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Horvath, G.; Järverud, G.A.K.; Järverud, S.; Horváth, I. Human Ovarian Carcinomas Detected by Specific Odor. Integr. Cancer Ther. 2008, 7, 76–80. [Google Scholar] [CrossRef]
- Van Oort, P.M.; Povoa, P.; Schnabel, R.; Dark, P.; Artigas, A.; Bergmans, D.C.J.J.; Felton, T.; Coelho, L.; Schultz, M.J.; Fowler, S.J.; et al. The potential role of exhaled breath analysis in the diagnostic process of pneumonia—a systematic review. J. Breath Res. 2018, 12, 024001. [Google Scholar] [CrossRef]
- Gao, Q.; Lee, W.-Y. Urinary Metabolites for Urological Cancer Detection: A Review on the Application of Volatile Organic Compounds for Cancers. Am. J. Clin. Exp. Urol. 2019, 7, 232. [Google Scholar]
- Silva, C.L.; Passos, M.; Câmara, J.S. Solid phase microextraction, mass spectrometry and metabolomic approaches for detection of potential urinary cancer biomarkers—A powerful strategy for breast cancer diagnosis. Talanta 2012, 89, 360–368. [Google Scholar] [CrossRef]
- Sethi, S.; Nanda, R.; Chakraborty, T. Clinical Application of Volatile Organic Compound Analysis for Detecting Infectious Diseases. Clin. Microbiol. Rev. 2013, 26, 462–475. [Google Scholar] [CrossRef]
- Banday, K.M.; Pasikanti, K.K.; Chan, E.C.Y.; Singla, R.; Rao, K.V.S.; Chauhan, V.S.; Nanda, R.K. Use of Urine Volatile Organic Compounds to Discriminate Tuberculosis Patients from Healthy Subjects. Anal. Chem. 2011, 83, 5526–5534. [Google Scholar] [CrossRef]
- Monteiro, M.; Carvalho, M.; Henrique, R.; Jerónimo, C.; Moreira, N.; De Lourdes Bastos, M.; De Pinho, P.G. Analysis of volatile human urinary metabolome by solid-phase microextraction in combination with gas chromatography–mass spectrometry for biomarker discovery: Application in a pilot study to discriminate patients with renal cell carcinoma. Eur. J. Cancer 2014, 50, 1993–2002. [Google Scholar] [CrossRef]
- Weber, C.M.; Cauchi, M.; Patel, M.; Bessant, C.; Turner, C.; Britton, L.E.; Willis, C.M. Evaluation of a gas sensor array and pattern recognition for the identification of bladder cancer from urine headspace. Analyst 2011, 136, 359–364. [Google Scholar] [CrossRef]
- Khalid, T.; Aggio, R.; White, P.; De Lacy Costello, B.; Persad, R.; Al-Kateb, H.; Jones, P.; Probert, C.S.; Ratcliffe, N. Urinary Volatile Organic Compounds for the Detection of Prostate Cancer. PLoS ONE 2015, 10, e0143283. [Google Scholar] [CrossRef] [PubMed]
- Mach, K.E.; Wong, P.K.; Liao, J.C. Biosensor diagnosis of urinary tract infections: A path to better treatment? Trends Pharmacol. Sci. 2011, 32, 330–336. [Google Scholar] [CrossRef] [PubMed]
- Foxman, B. The epidemiology of urinary tract infection. Nat. Rev. Urol. 2010, 7, 653–660. [Google Scholar] [CrossRef] [PubMed]
- Hooton, T.M. Uncomplicated Urinary Tract Infection. N. Engl. J. Med. 2012, 366, 1028–1037. [Google Scholar] [CrossRef] [PubMed]
- Nicolle, L. Complicated Urinary Tract Infection in Adults. Can. J. Infect. Dis. Med. Microbiol. 2005, 16, 349–360. [Google Scholar] [CrossRef] [PubMed]
- Seymour, C.W.; Gesten, F.; Prescott, H.C.; Friedrich, M.E.; Iwashyna, T.J.; Phillips, G.S.; Lemeshow, S.; Osborn, T.; Terry, K.M.; Levy, M.M. Time to Treatment and Mortality during Mandated Emergency Care for Sepsis. N. Engl. J. Med. 2017, 376, 2235–2244. [Google Scholar] [CrossRef]
- Hatfield, K.M.; Dantes, R.B.; Baggs, J.; Sapiano, M.R.P.; Fiore, A.E.; Jernigan, J.A.; Epstein, L. Assessing Variability in Hospital-Level Mortality Among, U.S. Medicare Beneficiaries with Hospitalizations for Severe Sepsis and Septic Shock. Crit. Care Med. 2018, 46, 1753–1760. [Google Scholar] [CrossRef]
- François, M.; Hanslik, T.; Dervaux, B.; Le Strat, Y.; Souty, C.; Vaux, S.; Maugat, S.; Rondet, C.; Sarazin, M.; Heym, B.; et al. The economic burden of urinary tract infections in women visiting general practices in France: A cross-sectional survey. BMC Health Serv. Res. 2016, 16, 365. [Google Scholar] [CrossRef]
- Bhalodi, A.A.; Van Engelen, T.S.R.; Virk, H.S.; Wiersinga, W.J. Impact of antimicrobial therapy on the gut microbiome. J. Antimicrob. Chemother. 2019, 74, i6–i15. [Google Scholar] [CrossRef]
- O’Neill, J. Antimicrobial Resistance: Tackling a Crisis for the Health and Wealth of Nations; Wellcome Trust: London, UK, 2014; pp. 1–20. [Google Scholar]
- Zowawi, H.M.; Harris, P.N.A.; Roberts, M.J.; Tambyah, P.A.; Schembri, M.A.; Pezzani, M.D.; Williamson, D.A.; Paterson, D.L. The emerging threat of multidrug-resistant Gram-negative bacteria in urology. Nat. Rev. Urol. 2015, 12, 570–584. [Google Scholar] [CrossRef] [PubMed]
- Spees, A.M.; Wangdi, T.; Lopez, C.A.; Kingsbury, D.D.; Xavier, M.N.; Winter, S.E.; Tsolis, R.M.; Bäumler, A.J. Streptomycin-Induced Inflammation Enhances Escherichia coli Gut Colonization Through Nitrate Respiration. mBio 2013, 4, e00430-13. [Google Scholar] [CrossRef] [PubMed]
- Hay, A.D.; Birnie, K.; Busby, J.; Delaney, B.; Downing, H.; Dudley, J.; Durbaba, S.; Fletcher, M.; Harman, K.; Hollingworth, W.; et al. The Diagnosis of Urinary Tract infection in Young children (DUTY): A diagnostic prospective observational study to derive and validate a clinical algorithm for the diagnosis of urinary tract infection in children presenting to primary care with an acute illness. Health Technol. Assess. 2016, 20, 1–294. [Google Scholar] [CrossRef] [PubMed]
- Devillé, W.L.; Yzermans, J.C.; van Duijn, N.P.; Bezemer, P.D.; van der Windt, D.A.; Bouter, L.M. The urine dipstick test useful to rule out infections. A meta-analysis of the accuracy. BMC Urol. 2004, 4, 4. [Google Scholar] [CrossRef]
- Bent, S. Does This Woman Have an Acute Uncomplicated Urinary Tract Infection? JAMA 2002, 287, 2701. [Google Scholar] [CrossRef]
- Beale, D.; Jones, O.; Karpe, A.; Dayalan, S.; Oh, D.; Kouremenos, K.; Ahmed, W.; Palombo, E. A Review of Analytical Techniques and Their Application in Disease Diagnosis in Breathomics and Salivaomics Research. Int. J. Mol. Sci. 2016, 18, 24. [Google Scholar] [CrossRef]
- Xu, M.; Tang, Z.; Duan, Y.; Liu, Y. GC-Based Techniques for Breath Analysis: Current Status, Challenges, and Prospects. Crit. Rev. Anal. Chem. 2016, 46, 291–304. [Google Scholar] [CrossRef]
- Covington, J.A.; Van der Schee, M.P.; Edge, A.S.L.; Boyle, B.; Savage, R.S.; Arasaradnam, R.P. The application of FAIMS gas analysis in medical diagnostics. Analyst 2015, 140, 6775–6781. [Google Scholar] [CrossRef]
- Arasaradnam, R.P.; McFarlane, M.J.; Ryan-Fisher, C.; Westenbrink, E.; Hodges, P.; Thomas, M.G.; Chambers, S.; O’Connell, N.; Bailey, C.; Harmston, C.; et al. Detection of Colorectal Cancer (CRC) by Urinary Volatile Organic Compound Analysis. PLoS ONE 2014, 9, e108750. [Google Scholar] [CrossRef]
- Westhoff, M.; Litterst, P.; Maddula, S.; Bödeker, B.; Rahmann, S.; Davies, A.N.; Baumbach, J.I. Differentiation of chronic obstructive pulmonary disease (COPD) including lung cancer from healthy control group by breath analysis using ion mobility spectrometry. Int. J. Ion. Mobil. Spectrom. 2010, 13, 131–139. [Google Scholar] [CrossRef]
- Wilson, A.D. Applications of Electronic-Nose Technologies for Noninvasive Early Detection of Plant, Animal and Human Diseases. Chemosensors 2018, 6, 45. [Google Scholar] [CrossRef]
- Madrolle, S.; Grangeat, P.; Jutten, C. A Linear-Quadratic Model for the Quantification of a Mixture of Two Diluted Gases with a Single Metal Oxide Sensor. Sensors 2018, 18, 1785. [Google Scholar] [CrossRef] [PubMed]
- Szulczyński, B.; Gębicki, J. Currently Commercially Available Chemical Sensors Employed for Detection of Volatile Organic Compounds in Outdoor and Indoor Air. Environments 2017, 4, 21. [Google Scholar] [CrossRef]
- Persaud, K.C. Polymers for chemical sensing. Mater. Today 2005, 8, 38–44. [Google Scholar] [CrossRef]
- Kim, Y.S.; Ha, S.-C.; Yang, Y.; Kim, Y.J.; Cho, S.M.; Yang, H.; Kim, Y.T. Portable electronic nose system based on the carbon black–polymer composite sensor array. Sens. Actuators B Chem. 2005, 108, 285–291. [Google Scholar] [CrossRef]
- Nakhleh, M.K.; Broza, Y.Y.; Haick, H. Monolayer-capped gold nanoparticles for disease detection from breath. Nanomedicine 2014, 9, 1991–2002. [Google Scholar] [CrossRef]
- Tisch, U.; Haick, H. Arrays of chemisensitive monolayer-capped metallic nanoparticles for diagnostic breath testing. Rev. Chem. Eng. 2010, 26. [Google Scholar] [CrossRef]
- Fernandes, M.P.; Venkatesh, S.; Sudarshan, B.G. Early Detection of Lung Cancer Using Nano-Nose—A Review. Open Biomed. Eng. J. 2015, 9, 228–233. [Google Scholar] [CrossRef]
- Wang, S.; Inci, F.; De Libero, G.; Singhal, A.; Demirci, U. Point-of-care assays for tuberculosis: Role of nanotechnology/microfluidics. Biotechnol. Adv. 2013, 31, 438–449. [Google Scholar] [CrossRef]
- Liu, X.; Cheng, S.; Liu, H.; Hu, S.; Zhang, D.; Ning, H. A Survey on Gas Sensing Technology. Sensors 2012, 12, 9635–9665. [Google Scholar] [CrossRef]
- Wilson, A.; Baietto, M. Applications and Advances in Electronic-Nose Technologies. Sensors 2009, 9, 5099–5148. [Google Scholar] [CrossRef] [PubMed]
- Wilson, A. Advances in Electronic-Nose Technologies for the Detection of Volatile Biomarker Metabolites in the Human Breath. Metabolites 2015, 5, 140–163. [Google Scholar] [CrossRef] [PubMed]
- Wilson, A.D. Review of Electronic-nose Technologies and Algorithms to Detect Hazardous Chemicals in the Environment. Procedia Technol. 2012, 1, 453–463. [Google Scholar] [CrossRef]
- Jian, Y.; Hu, W.; Zhao, Z.; Cheng, P.; Haick, H.; Yao, M.; Wu, W. Gas Sensors Based on Chemi-Resistive Hybrid Functional Nanomaterials. Nano-Micro Lett. 2020, 12, 71. [Google Scholar] [CrossRef]
- Korotcenkov, G.; Brinzari, V.; Cho, B.K. Conductometric gas sensors based on metal oxides modified with gold nanoparticles: A review. Microchim. Acta 2016, 183, 1033–1054. [Google Scholar] [CrossRef]
- Westenbrink, E.; Arasaradnam, R.P.; O’Connell, N.; Bailey, C.; Nwokolo, C.; Bardhan, K.D.; Covington, J.A. Development and application of a new electronic nose instrument for the detection of colorectal cancer. Biosens. Bioelectron. 2015, 67, 733–738. [Google Scholar] [CrossRef]
- Wilson, A. Application of Electronic-Nose Technologies and VOC-Biomarkers for the Noninvasive Early Diagnosis of Gastrointestinal Diseases. Sensors 2018, 18, 2613. [Google Scholar] [CrossRef]
- Sohn, J.H.; Atzeni, M.; Zeller, L.; Pioggia, G. Characterisation of humidity dependence of a metal oxide semiconductor sensor array using partial least squares. Sens. Actuators B Chem. 2008, 131, 230–235. [Google Scholar] [CrossRef]
- Fryder, M.; Holmberg, M.; Winquist, F.; Lundstrom, I. A calibration technique for an electronic nose. In Proceedings of the International Solid-State Sensors and Actuators Conference—TRANSDUCERS’95, Stockholm, Sweden, 25–29 June 1995; IEEE: Piscataway, NJ, USA, 1995; Volume 1, pp. 683–686. [Google Scholar]
- Lechner, M.; Karlseder, A.; Niederseer, D.; Lirk, P.; Neher, A.; Rieder, J.; Tilg, H.H. pylori Infection Increases Levels of Exhaled Nitrate. Helicobacter 2005, 10, 385–390. [Google Scholar] [CrossRef]
- Zhan, X.; Duan, J.; Duan, Y. Recent developments of proton-transfer reaction mass spectrometry (PTR-MS) and its applications in medical research. Mass Spectrom. Rev. 2013, 32, 143–165. [Google Scholar] [CrossRef]
- Blake, G.J.; Otvos, J.D.; Rifai, N.; Ridker, P.M. Low-Density Lipoprotein Particle Concentration and Size as Determined by Nuclear Magnetic Resonance Spectroscopy as Predictors of Cardiovascular Disease in Women. Circulation 2002, 106, 1930–1937. [Google Scholar] [CrossRef] [PubMed]
- Moka, D.; Vorreuther, R.; Schicha, H.; Spraul, M.; Humpfer, E.; Lipinski, M.; Foxall, P.J.D.; Nicholson, J.K.; Lindon, J.C. Biochemical classification of kidney carcinoma biopsy samples using magic-angle-spinning 1H nuclear magnetic resonance spectroscopy. J. Pharm. Biomed. Anal. 1998, 17, 125–132. [Google Scholar] [CrossRef]
- Shulman, G.I.; Alger, J.R.; Prichard, J.W.; Shulman, R.G. Nuclear magnetic resonance spectroscopy in diagnostic and investigative medicine. J. Clin. Investig. 1984, 74, 1127–1131. [Google Scholar] [CrossRef] [PubMed]
- Bajtarevic, A.; Ager, C.; Pienz, M.; Klieber, M.; Schwarz, K.; Ligor, M.; Ligor, T.; Filipiak, W.; Denz, H.; Fiegl, M.; et al. Noninvasive detection of lung cancer by analysis of exhaled breath. BMC Cancer 2009, 9, 348. [Google Scholar] [CrossRef] [PubMed]
- Wlodzimirow, K.A.; Abu-Hanna, A.; Schultz, M.J.; Maas, M.A.W.; Bos, L.D.J.; Sterk, P.J.; Knobel, H.H.; Soers, R.J.T.; Chamuleau, R.A.F.M. Exhaled breath analysis with electronic nose technology for detection of acute liver failure in rats. Biosens. Bioelectron. 2014, 53, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Smolinska, A.; Hauschild, A.-C.; Fijten, R.R.R.; Dallinga, J.W.; Baumbach, J.; Van Schooten, F.J. Current breathomics—A review on data pre-processing techniques and machine learning in metabolomics breath analysis. J. Breath Res. 2014, 8, 027105. [Google Scholar] [CrossRef]
- Pereira, J.; Porto-Figueira, P.; Cavaco, C.; Taunk, K.; Rapole, S.; Dhakne, R.; Nagarajaram, H.; Câmara, J. Breath Analysis as a Potential and Non-Invasive Frontier in Disease Diagnosis: An Overview. Metabolites 2015, 5, 3–55. [Google Scholar] [CrossRef]
- Guyon, I.; Elisseff, A. An introduction to variable and feature selection. J. Mach. Learn. Res. 2003, 3, 1157–1182. [Google Scholar]
- Woollam, M.; Teli, M.; Liu, S.; Daneshkhah, A.; Siegel, A.P.; Yokota, H.; Agarwal, M. Urinary Volatile Terpenes Analyzed by Gas Chromatography–Mass Spectrometry to Monitor Breast Cancer Treatment Efficacy in Mice. J. Proteome Res. 2020, 19, 1913–1922. [Google Scholar] [CrossRef]
- Mozdiak, E.; Wicaksono, A.N.; Covington, J.A.; Arasaradnam, R.P. Colorectal cancer and adenoma screening using urinary volatile organic compound (VOC) detection: Early results from a single-centre bowel screening population (UK BCSP). Tech. Coloproctol. 2019, 23, 343–351. [Google Scholar] [CrossRef]
- Mazzone, P.J.; Wang, X.-F.; Lim, S.; Choi, H.; Jett, J.; Vachani, A.; Zhang, Q.; Beukemann, M.; Seeley, M.; Martino, R.; et al. Accuracy of volatile urine biomarkers for the detection and characterization of lung cancer. BMC Cancer 2015, 15, 1001. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, K.; Opiekun, M.; Oka, H.; Vachani, A.; Albelda, S.M.; Yamazaki, K.; Beauchamp, G.K. Urinary Volatile Compounds as Biomarkers for Lung Cancer: A Proof of Principle Study Using Odor Signatures in Mouse Models of Lung Cancer. PLoS ONE 2010, 5, e8819. [Google Scholar] [CrossRef] [PubMed]
- Hayward, N.J.; Jeavons, T.H.; Nicholson, A.J.; Thornton, A.G. Methyl mercaptan and dimethyl disulfide production from methionine by Proteus species detected by head-space gas-liquid chromatography. J. Clin. Microbiol. 1977, 6, 187–194. [Google Scholar] [PubMed]
- Hayward, N.J.; Jeavons, T.H.; Nicholson, A.J.; Thornton, A.G. Development of specific tests for rapid detection of Escherichia coli and all species of Proteus in urine. J. Clin. Microbiol. 1977, 6, 195–201. [Google Scholar]
- Hayward, N.J.; Jeavons, T.H. Assessment of Technique for Rapid Detection of Escherichia Coli and Proteus Species in Urine by Head-Space Gas-Liquid Chromatography. J. Clin. Microbiol. 1977, 6, 202–208. [Google Scholar]
- Coloe, P.J. Ethanol formed from arabinose: A rapid method for detecting Escherichia coli. J. Clin. Pathol. 1978, 31, 361–364. [Google Scholar] [CrossRef]
- Coloe, P.J. Head-space gas liquid chromatography for rapid detection of Escherichia coli and Proteus mirabilis in urine. J. Clin. Pathol. 1978, 31, 365–369. [Google Scholar] [CrossRef]
- Manja, K.S.; Rao, K.M. Gas-chromatographic detection of urinary tract infections caused by Escherichia coli and Klebsiella sp. J. Clin. Microbiol. 1983, 17, 264–266. [Google Scholar] [CrossRef]
- Davies, T.J.; Hayward, N.J. Volatile products from acetylcholine as markers in the rapid urine test using head-space gas—liquid chromatography. J. Chromatogr. B Biomed. Sci. Appl. 1984, 307, 11–21. [Google Scholar] [CrossRef]
- Storer, M.K.; Hibbard-Melles, K.; Davis, B.; Scotter, J. Detection of volatile compounds produced by microbial growth in urine by selected ion flow tube mass spectrometry (SIFT-MS). J. Microbiol. Methods 2011, 87, 111–113. [Google Scholar] [CrossRef]
- Thorn, R.M.S.; Reynolds, D.M.; Greenman, J. Multivariate analysis of bacterial volatile compound profiles for discrimination between selected species and strains in vitro. J. Microbiol. Methods 2011, 84, 258–264. [Google Scholar] [CrossRef] [PubMed]
- Bean, H.D.; Dimandja, J.-M.D.; Hill, J.E. Bacterial volatile discovery using solid phase microextraction and comprehensive two-dimensional gas chromatography–time-of-flight mass spectrometry. J. Chromatogr. B 2012, 901, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Karami, N.; Mirzajani, F.; Rezadoost, H.; Karimi, A.; Fallah, F.; Ghassempour, A.; Aliahmadi, A. Initial study of three different pathogenic microorganisms by gas chromatography-mass spectrometry. F1000Research 2018, 6, 1415. [Google Scholar] [CrossRef] [PubMed]
- Altaee, N.; Kadhim, M.J.; Hameed, I.H. Detection of Volatile Compounds Produced by Pseudomonas aeruginosa Isolated from UTI Patients by Gas Chromatography-Mass Spectrometry. Int. J. Curr. Pharm. Rev. Res. 2017, 7, 8–24. [Google Scholar]
- Smart, A.; De Lacy Costello, B.; White, P.; Avison, M.; Batty, C.; Turner, C.; Persad, R.; Ratcliffe, N. Sniffing out resistance—Rapid identification of urinary tract infection-causing bacteria and their antibiotic susceptibility using volatile metabolite profiles. J. Pharm. Biomed. Anal. 2019, 167, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Adebiyi, A.; Swaminathan, S.; Than, N.; Abdi, M.; Bowers, A.N.; Fasoli, A.; Mannari, A.; Bozano, L. Rapid Strain Differentiation of E. coli-inoculated Urine Using Olfactory-based Smart Sensors. Sens. Transducers 2019, 238, 94–98. [Google Scholar]
- Ratiu, I.A.; Bocos-Bintintan, V.; Patrut, A.; Moll, V.H.; Turner, M.; Thomas, C.L.P. Discrimination of bacteria by rapid sensing their metabolic volatiles using an aspiration-type ion mobility spectrometer (a-IMS) and gas chromatography-mass spectrometry GC-MS. Anal. Chim. Acta 2017, 982, 209–217. [Google Scholar] [CrossRef]
- DeJong, C.S.; Wang, D.I.; Polyakov, A.; Rogacs, A.; Simske, S.J.; Shkolnikov, V. Bacterial Detection and Differentiation via Direct Volatile Organic Compound Sensing with Surface Enhanced Raman Spectroscopy. ChemistrySelect 2017, 2, 8431–8435. [Google Scholar] [CrossRef]
- Rees, C.A.; Burklund, A.; Stefanuto, P.-H.; Schwartzman, J.D.; Hill, J.E. Comprehensive volatile metabolic fingerprinting of bacterial and fungal pathogen groups. J. Breath Res. 2018, 12, 026001. [Google Scholar] [CrossRef]
- Wiesner, K.; Jaremek, M.; Pohle, R.; Von Sicard, O.; Stuetz, E. Monitoring of Bacterial Growth and Rapid Evaluation of Antibiotic Susceptibility by Headspace Gas Analysis. Procedia Eng. 2014, 87, 332–335. [Google Scholar] [CrossRef]
- Sovová, K.; Čepl, J.; Markoš, A.; Španěl, P. Real time monitoring of population dynamics in concurrent bacterial growth using SIFT-MS quantification of volatile metabolites. Analyst 2013, 138, 4795. [Google Scholar] [CrossRef] [PubMed]
- Zechman, J.M.; Aldinger, S.; Labows, J.N. Characterization of pathogenic bacteria by automated headspace concentration—gas chromatography. J. Chromatogr. B Biomed. Sci. Appl. 1986, 377, 49–57. [Google Scholar] [CrossRef]
- Luchner, M.; Gutmann, R.; Bayer, K.; Dunkl, J.; Hansel, A.; Herbig, J.; Singer, W.; Strobl, F.; Winkler, K.; Striedner, G. Implementation of proton transfer reaction-mass spectrometry (PTR-MS) for advanced bioprocess monitoring. Biotechnol. Bioeng. 2012, 109, 3059–3069. [Google Scholar] [CrossRef] [PubMed]
- Edman, D.C.; Brooks, J.B. Gas—liquid chromatography—frequency pulse-modulated electron-capture detection in the diagnosis of infectious diseases. J. Chromatogr. B Biomed. Sci. Appl. 1983, 274, 1–25. [Google Scholar] [CrossRef]
- Zhu, J.; Bean, H.D.; Kuo, Y.-M.; Hill, J.E. Fast Detection of Volatile Organic Compounds from Bacterial Cultures by Secondary Electrospray Ionization-Mass Spectrometry. J. Clin. Microbiol. 2010, 48, 4426–4431. [Google Scholar] [CrossRef]
- Jünger, M.; Vautz, W.; Kuhns, M.; Hofmann, L.; Ulbricht, S.; Baumbach, J.I.; Quintel, M.; Perl, T. Ion mobility spectrometry for microbial volatile organic compounds: A new identification tool for human pathogenic bacteria. Appl. Microbiol. Biotechnol. 2012, 93, 2603–2614. [Google Scholar] [CrossRef] [PubMed]
- Tavakkol, A.; Drucker, D.B. Gas chromatographic analysis of bacterial amines as their free bases. J. Chromatogr. B Biomed. Sci. Appl. 1983, 274, 37–44. [Google Scholar] [CrossRef]
- Labows, J.N.; McGinley, K.J.; Webster, G.F.; Leyden, J.J. Headspace Analysis of Volatile Metabolites of Pseudomonas Aeruginosa and Related Species by Gas Chromatography-Mass Spectrometry. J. Clin. Microbiol. 1980, 12, 521–526. [Google Scholar] [CrossRef]
- Preti, G.; Thaler, E.; Hanson, C.W.; Troy, M.; Eades, J.; Gelperin, A. Volatile compounds characteristic of sinus-related bacteria and infected sinus mucus: Analysis by solid-phase microextraction and gas chromatography–mass spectrometry. J. Chromatogr. B 2009, 877, 2011–2018. [Google Scholar] [CrossRef]
- Boots, A.W.; Smolinska, A.; Van Berkel, J.J.B.N.; Fijten, R.R.R.; Stobberingh, E.E.; Boumans, M.L.L.; Moonen, E.J.; Wouters, E.F.M.; Dallinga, J.W.; Van Schooten, F.J. Identification of microorganisms based on headspace analysis of volatile organic compounds by gas chromatography–mass spectrometry. J. Breath Res. 2014, 8, 027106. [Google Scholar] [CrossRef]
- Kuzma, J.; Nemecek-Marshall, M.; Pollock, W.H.; Fall, R. Bacteria produce the volatile hydrocarbon isoprene. Curr. Microbiol. 1995, 30, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Filipiak, W.; Sponring, A.; Baur, M.; Filipiak, A.; Ager, C.; Wiesenhofer, H.; Nagl, M.; Troppmair, J.; Amann, A. Molecular analysis of volatile metabolites released specifically by staphylococcus aureus and pseudomonas aeruginosa. BMC Microbiol. 2012, 12, 113. [Google Scholar] [CrossRef] [PubMed]
- Schöller, C.; Molin, S.; Wilkins, K. Volatile metabolites from some gram-negative bacteria. Chemosphere 1997, 35, 1487–1495. [Google Scholar] [CrossRef]
- Maddula, S.; Blank, L.M.; Schmid, A.; Baumbach, J.I. Detection of volatile metabolites of Escherichia coli by multi capillary column coupled ion mobility spectrometry. Anal. Bioanal. Chem. 2009, 394, 791–800. [Google Scholar] [CrossRef]
- Bunge, M.; Araghipour, N.; Mikoviny, T.; Dunkl, J.; Schnitzhofer, R.; Hansel, A.; Schinner, F.; Wisthaler, A.; Margesin, R.; Märk, T.D. On-Line Monitoring of Microbial Volatile Metabolites by Proton Transfer Reaction-Mass Spectrometry. Appl. Environ. Microbiol. 2008, 74, 2179–2186. [Google Scholar] [CrossRef]
- Hamilton-Kemp, T.; Newman, M.; Collins, R.; Elgaali, H.; Yu, K.; Archbold, D. Production of the Long-Chain Alcohols Octanol, Decanol, and Dodecanol by Escherichia coli. Curr. Microbiol. 2005, 51, 82–86. [Google Scholar] [CrossRef]
- Cox, C.D.; Parker, J. Use of 2-aminoacetophenone Production in Identification of Pseudomonas Aeruginosa. J. Clin. Microbiol. 1979, 9, 479–484. [Google Scholar]
- Carroll, W.; Lenney, W.; Wang, T.; Španěl, P.; Alcock, A.; Smith, D. Detection of volatile compounds emitted by Pseudomonas aeruginosa using selected ion flow tube mass spectrometry. Pediatr. Pulmonol. 2005, 39, 452–456. [Google Scholar] [CrossRef]
- Allardyce, R.A.; Hill, A.L.; Murdoch, D.R. The rapid evaluation of bacterial growth and antibiotic susceptibility in blood cultures by selected ion flow tube mass spectrometry. Diagn. Microbiol. Infect. Dis. 2006, 55, 255–261. [Google Scholar] [CrossRef]
- Allardyce, R.A.; Langford, V.S.; Hill, A.L.; Murdoch, D.R. Detection of volatile metabolites produced by bacterial growth in blood culture media by selected ion flow tube mass spectrometry (SIFT-MS). J. Microbiol. Methods 2006, 65, 361–365. [Google Scholar] [CrossRef]
- Scotter, J.M.; Allardyce, R.A.; Langford, V.S.; Hill, A.; Murdoch, D.R. The rapid evaluation of bacterial growth in blood cultures by selected ion flow tube–mass spectrometry (SIFT-MS) and comparison with the BacT/ALERT automated blood culture system. J. Microbiol. Methods 2006, 65, 628–631. [Google Scholar] [CrossRef]
- Scott-Thomas, A.J.; Syhre, M.; Pattemore, P.K.; Epton, M.; Laing, R.; Pearson, J.; Chambers, S.T. 2-Aminoacetophenone as a potential breath biomarker for Pseudomonas aeruginosa in the cystic fibrosis lung. BMC Pulm. Med. 2010, 10, 56. [Google Scholar] [CrossRef] [PubMed]
- Lawal, O.; Muhamadali, H.; Ahmed, W.M.; White, I.R.; Nijsen, T.M.E.; Goodacre, R.; Fowler, S.J. On behalf of the BreathDx Consortium. Headspace volatile organic compounds from bacteria implicated in ventilator-associated pneumonia analysed by TD-GC/MS. J. Breath Res. 2018, 12, 026002. [Google Scholar] [CrossRef]
- Drees, C.; Vautz, W.; Liedtke, S.; Rosin, C.; Althoff, K.; Lippmann, M.; Zimmermann, S.; Legler, T.J.; Yildiz, D.; Perl, T.; et al. GC-IMS headspace analyses allow early recognition of bacterial growth and rapid pathogen differentiation in standard blood cultures. Appl. Microbiol. Biotechnol. 2019, 103, 9091–9101. [Google Scholar] [CrossRef] [PubMed]
- Julák, J.; Stránská, E.; Rosová, V.; Geppert, H.; Španěl, P.; Smith, D. Bronchoalveolar lavage examined by solid phase microextraction, gas chromatography–mass spectrometry and selected ion flow tube mass spectrometry. J. Microbiol. Methods 2006, 65, 76–86. [Google Scholar] [CrossRef] [PubMed]
- Julák, J.; Stránská, E.; Procházková-Francisci, E.; Rosová, V. Blood Cultures Evaluation by Gas Chromatography of Volatile Fatty Acids. Med. Sci. Monit. 2000, 6, 605–610. [Google Scholar] [PubMed]
- Dolch, M.E.; Janitza, S.; Boulesteix, A.-L.; Graßmann-Lichtenauer, C.; Praun, S.; Denzer, W.; Schelling, G.; Schubert, S. Gram-negative and -positive bacteria differentiation in blood culture samples by headspace volatile compound analysis. J. Biol. Res.-Thessalon. 2016, 23, 3. [Google Scholar] [CrossRef][Green Version]
- Aathithan, S.; Plant, J.C.; Chaudry, A.N.; French, G.L. Diagnosis of Bacteriuria by Detection of Volatile Organic Compounds in Urine Using an Automated Headspace Analyzer with Multiple Conducting Polymer Sensors. J. Clin. Microbiol. 2001, 39, 2590–2593. [Google Scholar] [CrossRef]
- Pavlou, A.K.; Magan, N.; McNulty, C.; Jones, J.M.; Sharp, D.; Brown, J.; Turner, A.P.F. Use of an electronic nose system for diagnoses of urinary tract infections. Biosens. Bioelectron. 2002, 17, 893–899. [Google Scholar] [CrossRef]
- Kodogiannis, V.S.; Lygouras, J.N.; Tarczynski, A.; Chowdrey, H.S. Artificial Odor Discrimination System Using Electronic Nose and Neural Networks for the Identification of Urinary Tract Infection. IEEE Trans. Inf. Technol. Biomed. 2008, 12, 707–713. [Google Scholar] [CrossRef]
- Yates, J.W.T.; Chappell, M.J.; Gardner, J.W.; Dow, C.S.; Dowson, C.; Hamood, A.; Bolt, F.; Beeby, L. Data reduction in headspace analysis of blood and urine samples for robust bacterial identification. Comput. Methods Progr. Biomed. 2005, 79, 259–271. [Google Scholar] [CrossRef]
- Sabeel, T.M.A.; CheHarun, F.K.; Eluwa, S.E.; Sabeel, S.M.A. Detection of volatile compounds in urine using an electronic nose instrument. In Proceedings of the 2013 International Conference on Computing, Electrical and Electronics Engineering (ICCEEE), Khartoum, Sudan, 26–28 August 2013; IEEE: Piscataway, NJ, USA, 2013; pp. 1–4. [Google Scholar]
- Roine, A.; Saviauk, T.; Kumpulainen, P.; Karjalainen, M.; Tuokko, A.; Aittoniemi, J.; Vuento, R.; Lekkala, J.; Lehtimäki, T.; Tammela, T.L.; et al. Rapid and Accurate Detection of Urinary Pathogens by Mobile IMS-Based Electronic Nose: A Proof-of-Principle Study. PLoS ONE 2014, 9, e114279. [Google Scholar] [CrossRef] [PubMed]
- Hayward, N.J. Head-space gas—Liquid chromatography for the rapid laboratory diagnosis of urinary tract infections caused by enterobacteria. J. Chromatogr. B Biomed. Sci. Appl. 1983, 274, 27–35. [Google Scholar] [CrossRef]
- Gardner, J.W.; Yates, J.W.T.; Dow, C.S.; Chappell, M.J. Identification of bacterial pathogens using quadrupole mass spectrometer data and radial basis function neural networks. IEE Proc. Sci. Meas. Technol. 2005, 152, 97–102. [Google Scholar] [CrossRef]
- Bruins, M.; Bos, A.; Petit, P.L.C.; Eadie, K.; Rog, A.; Bos, R.; van Ramshorst, G.H.; Van Belkum, A. Device-independent, real-time identification of bacterial pathogens with a metal oxide-based olfactory sensor. Eur. J. Clin. Microbiol. Infect. Dis. 2009, 28, 775–780. [Google Scholar] [CrossRef] [PubMed]
- Neale, R.; Berman, D. The Long Read: We Must do Better for UTI Patients. Available online: https://longitudeprize.org/blog-post/long-read-we-must-do-better-uti-patients (accessed on 8 June 2020).
- Drabińska, N.; Jarocka-Cyrta, E.; Ratcliffe, N.; Krupa-Kozak, U. The Profile of Urinary Headspace Volatile Organic Compounds After 12-Week Intake of Oligofructose-Enriched Inulin by Children and Adolescents with Celiac Disease on a Gluten-Free Diet: Results of a Pilot, Randomized, Placebo-Controlled Clinical Trial. Molecules 2019, 24, 1341. [Google Scholar] [CrossRef] [PubMed]
- Queralto, N.; Berliner, A.N.; Goldsmith, B.; Martino, R.; Rhodes, P.; Lim, S.H. Detecting cancer by breath volatile organic compound analysis: A review of array-based sensors. J. Breath Res. 2014, 8, 027112. [Google Scholar] [CrossRef]
- Aggio, R.B.M.; Mayor, A.; Coyle, S.; Reade, S.; Khalid, T.; Ratcliffe, N.M.; Probert, C.S.J. Freeze-drying: An alternative method for the analysis of volatile organic compounds in the headspace of urine samples using solid phase micro-extraction coupled to gas chromatography - mass spectrometry. Chem. Cent. J. 2016, 10, 9. [Google Scholar] [CrossRef]
- Kouri, T.; Fogazzi, G.; Gant, V.; Hallander, H.; Hofmann, W.; Guder, W.G. European Urinalysis Guidelines. Scand. J. Clin. Lab. Investig. 2000, 60, 1–96. [Google Scholar] [CrossRef]
- Pernille, H.; Lars, B.; Marjukka, M.; Volkert, S.; Anne, H. Sampling of urine for diagnosing urinary tract infection in general practice—First-void or mid-stream urine? Scand. J. Prim. Health Care 2019, 37, 113–119. [Google Scholar] [CrossRef]
- Delanghe, J.; Speeckaert, M. Preanalytical requirements of urinalysis. Biochem. Medica 2014, 89–104. [Google Scholar] [CrossRef] [PubMed]
- Jobu, K.; Sun, C.; Yoshioka, S.; Yokota, J.; Onogawa, M.; Kawada, C.; Inoue, K.; Shuin, T.; Sendo, T.; Miyamura, M. Metabolomics Study on the Biochemical Profiles of Odor Elements in Urine of Human with Bladder Cancer. Biol. Pharm. Bull. 2012, 35, 639–642. [Google Scholar] [CrossRef]
- Rist, M.; Muhle-Goll, C.; Görling, B.; Bub, A.; Heissler, S.; Watzl, B.; Luy, B. Influence of Freezing and Storage Procedure on Human Urine Samples in NMR-Based Metabolomics. Metabolites 2013, 3, 243–258. [Google Scholar] [CrossRef] [PubMed]
- Esfahani, S.; Sagar, N.; Kyrou, I.; Mozdiak, E.; O’Connell, N.; Nwokolo, C.; Bardhan, K.; Arasaradnam, R.; Covington, J. Variation in Gas and Volatile Compound Emissions from Human Urine as It Ages, Measured by an Electronic Nose. Biosensors 2016, 6, 4. [Google Scholar] [CrossRef] [PubMed]
- Silva, C.L.; Passos, M.; Câmara, J.S. Investigation of urinary volatile organic metabolites as potential cancer biomarkers by solid-phase microextraction in combination with gas chromatography-mass spectrometry. Br. J. Cancer 2011, 105, 1894–1904. [Google Scholar] [CrossRef] [PubMed]
- Netto, D.C.; Reis, R.M.; Mendes, C.B.; Gomes, P.C.F.L.; Martins, I.; Siqueira, M.E.P.B. Headspace solid-phase microextraction procedure for gas-chromatography analysis of toluene in urine. J. Braz. Chem. Soc. 2008, 19, 1201–1206. [Google Scholar] [CrossRef]
- Robards, K.; Haddad, P.R.; Jackson, P.E. Headspace gas chromatography. In Principles and Practice of Modern Chromatographic Methods; Elsevier: Amsterdam, The Netherlands, 2004; pp. 75–177. ISBN 978-0-08-057178-2. [Google Scholar]
- Heers, H.; Gut, J.M.; Hegele, A.; Hofman, R.; Boeselt, T.; Hattesohl, A.; Koczulla, A.R. Non-invasive Detection of Bladder Tumors Through Volatile Organic Compounds: A Pilot Study with an Electronic Nose. Anticancer Res. 2018, 38, 833–837. [Google Scholar]
- Sakura, N.; Nishimura, S.; Fujita, N.; Namera, A.; Yashiki, M.; Kojima, T. Determination of acrolein in human urine by headspace gas chromatography and mass spectrometry. J. Chromatogr. B Biomed. Sci. Appl. 1998, 719, 209–212. [Google Scholar] [CrossRef]
- Oliveira, D.P.D.; Siqueira, M.E.P.B.D. A simple and rapid method for urinary acetone analysis by headspace/gas chromatography. Quím. Nova 2007, 30, 1362–1364. [Google Scholar] [CrossRef]
- Bosch, S.; El Manouni el Hassani, S.; Covington, J.A.; Wicaksono, A.N.; Bomers, M.K.; Benninga, M.A.; Mulder, C.J.J.; De Boer, N.K.H.; De Meij, T.G.J. Optimized Sampling Conditions for Fecal Volatile Organic Compound Analysis by Means of Field Asymmetric Ion Mobility Spectrometry. Anal. Chem. 2018, 90, 7972–7981. [Google Scholar] [CrossRef]
- Tiele, A.; Wicaksono, A.; Ayyala, S.K.; Covington, J.A. Development of a Compact, IoT-Enabled Electronic Nose for Breath Analysis. Electronics 2020, 9, 84. [Google Scholar] [CrossRef]
- Hu, W.; Wan, L.; Jian, Y.; Ren, C.; Jin, K.; Su, X.; Bai, X.; Haick, H.; Yao, M.; Wu, W. Electronic Noses: From Advanced Materials to Sensors Aided with Data Processing. Adv. Mater. Technol. 2018, 1800488. [Google Scholar] [CrossRef]
- Mochalski, P.; Ruzsanyi, V.; Wiesenhofer, H.; Mayhew, C.A. Instrumental sensing of trace volatiles—A new promising tool for detecting the presence of entrapped or hidden people. J. Breath Res. 2018, 12, 027107. [Google Scholar] [CrossRef]
- Schieweck, A.; Gunschera, J.; Varol, D.; Salthammer, T. Analytical procedure for the determination of very volatile organic compounds (C3–C6) in indoor air. Anal. Bioanal. Chem. 2018, 410, 3171–3183. [Google Scholar] [CrossRef] [PubMed]
- Palma, S.I.C.J.; Traguedo, A.P.; Porteira, A.R.; Frias, M.J.; Gamboa, H.; Roque, A.C.A. Machine learning for the meta-analyses of microbial pathogens’ volatile signatures. Sci. Rep. 2018, 8, 3360. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.-Y.; Cogswell, M.E.; Loria, C.M.; Chen, T.-C.; Pfeiffer, C.M.; Swanson, C.A.; Caldwell, K.L.; Perrine, C.G.; Carriquiry, A.L.; Liu, K.; et al. Urinary Excretion of Sodium, Potassium, and Chloride, but Not Iodine, Varies by Timing of Collection in a 24-Hour Calibration Study. J. Nutr. 2013, 143, 1276–1282. [Google Scholar] [CrossRef]
- Slupsky, C.M.; Rankin, K.N.; Wagner, J.; Fu, H.; Chang, D.; Weljie, A.M.; Saude, E.J.; Lix, B.; Adamko, D.J.; Shah, S.; et al. Investigations of the Effects of Gender, Diurnal Variation, and Age in Human Urinary Metabolomic Profiles. Anal. Chem. 2007, 79, 6995–7004. [Google Scholar] [CrossRef]
- Lenz, E.M.; Bright, J.; Wilson, I.D.; Hughes, A.; Morrisson, J.; Lindberg, H.; Lockton, A. Metabonomics, dietary influences and cultural differences: A 1H NMR-based study of urine samples obtained from healthy British and Swedish subjects. J. Pharm. Biomed. Anal. 2004, 36, 841–849. [Google Scholar] [CrossRef]
- Perrier, E.; Rondeau, P.; Poupin, M.; Le Bellego, L.; Armstrong, L.E.; Lang, F.; Stookey, J.; Tack, I.; Vergne, S.; Klein, A. Relation between urinary hydration biomarkers and total fluid intake in healthy adults. Eur. J. Clin. Nutr. 2013, 67, 939–943. [Google Scholar] [CrossRef]
- Gradišek, A.; Van Midden, M.; Koterle, M.; Prezelj, V.; Strle, D.; Štefane, B.; Brodnik, H.; Trifkovič, M.; Kvasić, I.; Zupanič, E.; et al. Improving the Chemical Selectivity of an Electronic Nose to TNT, DNT and RDX Using Machine Learning. Sensors 2019, 19, 5207. [Google Scholar] [CrossRef]
- Farraia, M.V.; Cavaleiro Rufo, J.; Paciência, I.; Mendes, F.; Delgado, L.; Moreira, A. The electronic nose technology in clinical diagnosis: A systematic review. Porto Biomed. J. 2019, 4, e42. [Google Scholar] [CrossRef] [PubMed]
- Tambyah, P.A. Catheter-associated urinary tract infections: Diagnosis and prophylaxis. Int. J. Antimicrob. Agents 2004, 24, 44–48. [Google Scholar] [CrossRef] [PubMed]
- Loh, K.Y.; Sivalingam, N. Urinary Tract Infections in Pregnancy. Malays. Fam. Physician 2007, 2, 54–57. [Google Scholar] [PubMed]
| Technology | eNoses | IMS | GC-MS | |
|---|---|---|---|---|
| CP | MOS | |||
| Portability | Good | Good | Good | Poor |
| Cost | Low | Low | Low | High |
| Trained personnel | No | No | No | Yes |
| Sample throughput | High | High | Medium | Low |
| Speed | Real-time | Real-time | Real-time | Off-line |
| Metabolite detection | No | No | No 1 | Yes |
| Pattern recognition | Yes | Yes | Yes | Yes |
| Chemical insight | No | No | Yes | Yes |
| Sensor drift | Yes | Yes | Minor | Minor |
| Compound | EC | PM | EF | PA | SA | KP | Compound | EC | PM | EF | PA | SA | KP |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Alcohol | Ester | ||||||||||||
| Ethanol | √ | √ | √ | √ | √ | ethyl acetate | √ | √ | √ | √ | √ | ||
| Methanol | √ | N-propylacetate | √ | √ | |||||||||
| Propanol | √ | n-butyl acetate | √ | ||||||||||
| 1-propanol | √ | Ethyl phenylacetate | √ | ||||||||||
| N-propanol | √ | isopentyl acetate | √ | √ | √ | √ | |||||||
| 2-(Methylthio)-ethanol | √ | √ | √ | √ | 3-Methylbutyl 2-methylpropanoate | √ | |||||||
| phenol | √ | √ | 2-methylbutyl isobutyrate | √ | |||||||||
| Butanol | √ | √ | 2-Phenyl ethyl acetate | √ | |||||||||
| 1-butanol | √ | √ | Phenylethyl butyrate | √ | |||||||||
| 2-butanol | √ | √ | √ | √ | methyl 2-methylbutyrate | √ | |||||||
| Isobutanol | √ | √ | √ | √ | 2-methylbutyl 2-methylbutyrate | √ | |||||||
| 2-methyl-1-butanol | √ | methyl methacrylate | √ | √ | |||||||||
| 3-Methyl-1-butanol | √ | √ | √ | √ | ethyl 2-methylbutyrate | √ | |||||||
| octanol | √ | isoamyl butyrate | √ | √ | |||||||||
| Decanol | √ | amyl isovalerate | √ | ||||||||||
| 1-Decanol | √ | ethyl isovalerate | √ | ||||||||||
| dodecanol | √ | ethyl formate | √ | ||||||||||
| Benzyl Alcohol | √ | ethyl butanoate | √ | √ | |||||||||
| Isopentanol (isoamyl alcohol) | √ | √ | √ | √ | Propanoic acid 2-hydroxy-2-methyl-methyl ester | √ | |||||||
| phenethyl alcohol | √ | √ | ethyl formate | √ | |||||||||
| ethylene glycol | √ | √ | Phenylacetic acid propylester | √ | |||||||||
| 4-methylphenol | √ | √ | Ketone | ||||||||||
| 2-methyl-1-propanol | √ | acetone | √ | √ | √ | √ | |||||||
| acetol (hydroxyacetone) | √ | Acetoin (3-hydroxybutanone) | √ | √ | |||||||||
| 2-phenylethyl alcohol | √ | butanedione (diacetyl) | √ | ||||||||||
| methylbutenol | √ | √ | √ | Butanone | √ | √ | √ | ||||||
| 2-butanone | √ | √ | √ | ||||||||||
| Aldehyde | 2,3-butanedione | √ | √ | √ | √ | √ | |||||||
| Acetaldehyde | √ | √ | 2-heptanone | √ | √ | √ | √ | √ | |||||
| Benzaldehyde | √ | √ | 4-heptanone | √ | |||||||||
| 2-Heptenal | √ | √ | 2-nonanone | √ | √ | √ | √ | √ | |||||
| (3-methylbutanal) Isovaleraldehyde | √ | √ | √ | √ | √ | 2-decanone | √ | ||||||
| 2-methylbutanal | √ | √ | √ | 2-hexanone | √ | ||||||||
| methylbutanal | √ | √ | √ | 2-Tridecanone | √ | ||||||||
| isobutyraldehyde | √ | 2-undecanone | √ | √ | √ | √ | √ | ||||||
| 3-(ethylthio)-propanal | √ | 2,3-Heptanedione | √ | ||||||||||
| Propanal | √ | 2-pentanone | √ | √ | √ | ||||||||
| 3-methyl-2-butenal | √ | 2-aminoacetophenone | √ | √ | |||||||||
| 2-ethylacrolein | √ | 3-octanone | √ | ||||||||||
| (Z)-2-methyl-2-butenal | √ | methyl isobutylketone | √ | ||||||||||
| (E)-2-methyl-2-butenal | √ | mercaptoacetone | √ | ||||||||||
| Methacrolein | √ | 1-hydroxy-2-propanone | √ | ||||||||||
| 2-methylpropanal | √ | ||||||||||||
| Nonanal | √ | 1-methyl-4-(1-methylethenyl)cyclohexane | √ | √ | √ | √ | |||||||
| formaldehyde | √ | √ | √ | √ | √ | ||||||||
| Acids | |||||||||||||
| Hydrocarbons | isobutyric acid | √ | |||||||||||
| Toluene | √ | √ | √ | √ | √ | Butyric acid | √ | ||||||
| 1-Methyl-naphthalene | √ | 4-methylhexanoic acid | √ | ||||||||||
| 2-Methyl-naphthalene | √ | Picolinic acid N-oxide | √ | ||||||||||
| Isoprene | √ | √ | Isoamyl benzoate | √ | |||||||||
| 2-Butene | √ | Sulphur | |||||||||||
| (Z)-2-butene | √ | methyl mercaptan (Methanethiol) | √ | √ | √ | √ | √ | √ | |||||
| (E)-2-butene | √ | dimethyl disulfide | √ | √ | √ | √ | √ | √ | |||||
| Undecane | √ | √ | dimethyl sulfide | √ | √ | √ | √ | ||||||
| 2,4-dimethylheptane | √ | √ | √ | √ | dimethyl trisulfide | √ | √ | √ | √ | ||||
| 3-methylheptane | √ | Benzyl methyl sulfide | √ | ||||||||||
| undecene | √ | 2-methoxy-5-methylthiophene | √ | ||||||||||
| 1-undecene | √ | S-Methyl thiobenzoate | √ | ||||||||||
| 2-methyl-2-butene | √ | 2,4-dithiapentane | √ | ||||||||||
| 1,10-undecadiene | √ | ||||||||||||
| 1-nonene | √ | Chlorine | |||||||||||
| 2-Nonene | √ | Trichloromethane | √ | √ | |||||||||
| 1-decene | √ | 4-Chloro-1H-indole | √ | ||||||||||
| 1-dodecene | √ | ||||||||||||
| Butane | √ | Inorganic | |||||||||||
| n-butane | √ | ammonia | √ | √ | √ | √ | |||||||
| 10-methyl-1-undecene | √ | Hydrogen Sulfide | √ | √ | √ | ||||||||
| 1,3-butadiene | √ | ||||||||||||
| 2-methylpropene | √ | Other | |||||||||||
| Propane | √ | pyrazine derivative | √ | √ | |||||||||
| 2-Nonene, 3-methyl | √ | benzene derivative | √ | √ | √ | √ | √ | ||||||
| 2,3,4-Trimethylhexane | √ | phenol derivative | √ | √ | √ | ||||||||
| hexane | √ | pentanone/toluene | √ | √ | √ | ||||||||
| 2,3,3-trimethylpentane | √ | ||||||||||||
| Acetic acid | √ | √ | √ | Nitrogen | |||||||||
| Isovaleric acid | √ | indole dimer | √ | ||||||||||
| 2-methylbutyric acid | √ | ethylamine | √ | ||||||||||
| Indole | √ | √ | |||||||||||
| Nitrogen | 3-Methyl-1H-indole | √ | |||||||||||
| methylpyrazine | √ | √ | Cadaverine | √ | √ | ||||||||
| benzonitrile | √ | √ | √ | √ | putrescine | √ | |||||||
| 2,3,5-trimethylpyrazine | √ | Acetonitrile | √ | √ | √ | ||||||||
| N-(Phenylmethylene)-methanamine | √ | beta-phenylethylamine | √ | ||||||||||
| N,N′-Dibenzylideneethylenediamine | √ | ||||||||||||
| N-(Phenylmethylene)-1-propanamine | √ | References | [95] | [96] | [84] | [95] | [96] | [83] | |||||
| N-(Phenylmethylene)-1-butanamine | √ | √ | [97] | [84] | [93] | [84] | [84] | [84] | |||||
| n-nitrosodimethylamine | √ | [83] | [83] | [93] | [93] | [96] | |||||||
| pyrrolidine | √ | [80] | [98] | [99] | [99] | [93] | |||||||
| Isoamylamine | √ | [81] | [81] | [100] | [100] | [100] | |||||||
| Isobutylamine | √ | [93] | [101] | [102] | [103] | [104] | |||||||
| 3-Methyl-N-(3-methylbutylidene)-1-butanamine | √ | [99] | [77] | [105] | [106] | ||||||||
| Benzyl nitrile | √ | [100] | [91] | [107] | [91] | ||||||||
| N-Butyl-benzenamine | √ | [108] | [100] | [103] | [104] | ||||||||
| 2-(3-Methylbutyl)-3,5-dimethylpyrazine | √ | √ | [105] | [106] | |||||||||
| 3-Methyl-N-(2-phenylethylidene)-1-butanamine | √ | [109] | [104] | ||||||||||
| p-Pentylaniline | √ | [110] | |||||||||||
| N-(1,1-Dimethylethyl)-benzamide | √ | [91,104] | |||||||||||
| N-n-Butylphthalimide | √ | ||||||||||||
| 4-methyl-quinazoline | √ | ||||||||||||
| Dimethylpyrazine | √ | ||||||||||||
| pyrrole | √ | ||||||||||||
| 3-methylpyrrole | √ | ||||||||||||
| 1-vinyl aziridine | √ | ||||||||||||
| Pyrimidine | √ | ||||||||||||
| 2-Acetylthiazole | √ | ||||||||||||
| Trimethylamine | √ | √ | √ |
| Reference and Year | Technology | Number | Culture for Headspace Analysis | Experiment/Findings |
|---|---|---|---|---|
| Hayward et al. [77], 1977 | Gas-liquid chromatograph | 68 strains | Basal defined media with potassium lactate, amino acids and salts. Nutrient Broth (Oxoid) and MacConkey agar (Oxoid). Incubation still/shaken (160 rpm) at 37 °C | Samples were mechanically shaken for 5 min at 37 °C followed by 3 min incubation at 60 °C prior to analysis. It was found that Proteus species will produce dimethyl disulfide and methyl mercaptan from L-methionine. |
| Hayward et al. [78], 1977 | Gas-liquid chromatograph | 14 bacterial species | Basal defined media with lactate, amino acids, salts and vitamins cultured still at 37 °C or yeast extract broth still/shaken at 160 rpm. | Proteus mirabilis produces dimethyl disulfide and methyl mercaptan when cultured with L-methionine. E. coli produces ethanol from lactose. Detection of E. coli and P. mirabilis in urine based on ethanol production was achieved in 5 h (ethanol for E. coli methyl mercaptan for P. mirabilis) or 4 h (dimethyl disulfide for P. mirabilis). Other bacteria tested for dimethyl disulfide, methyl mercaptan or ethanol production were: C. freundii, E. cloacae, S. marcescens, K. aerogenes, K, oxytoca, K. ozaenae, P. aeruginosa, S. aureus, S. epidermidis, S. marcescens and S. faecalis. Samples were mechanically shaken for 5 min at 37 °C followed by 3 min incubation at 60 °C prior to analysis. |
| Coloe et al. [80], 1978 | Gas-liquid chromatograph | 49 strains | 24 h, unshaken cultures. Media: arabinose, amino acid mixture, salt mixture, pH 7.4, nicotinic acid (0.5 mg) and calcium pantothenate | 39/49 clinical samples: urine—29, faeces—8, pus—1, sputum—1. E. coli H19 incubation resulted in ethanol production from arabinose (maximum concentration after 8 h). For headspace analysis the mixture (culture fluid with 6 g of anhydrous K2CO3) was shaken at 160 rpm at 37 °C for 5 min and then heated at 60 °C in a water bath for 5 min. |
| Coloe et al. [81], 1978 | Gas-liquid chromatograph | 122 urine samples | 37 °C, 4 h shaking in 6 mL yeast-extract peptone water enriched with arabinose and methionine | 94 samples from UTI suspected patients, 28 uninfected controls. Results available in 4 h. E. coli produces ethanol from arabinose and P. mirabilis dimethyl disulfide from methionine. |
| Hayward et al. [128], 1983 | Gas-liquid chromatograph | 382 urine samples | Methionine yeast-extract peptone medium supplemented with arabinose incubated for 3.5 h. | E. coli, Klebsiella, Citrobacter and Proteus species. Ethanol production from lactose/arabinose of E. coli and methyl mercaptan from methionine of Proteus spp (methyl mercaptan oxidizes to dimethyl disulfide). No false positives and false negatives. Liquid was heated to 60 °C for headspace sampling. |
| Manja et al. [82], 1983 | Gas-chromatography | 96 urine samples | E. coli detection M-9 salt mixture, M-9L medium at 44 °C, 5 h. Klebsiella detection M-9A medium (M-9L with lactose replaced by adonitol and carbenicillin), 37 °C. | 16 cases caused by E coli, 4 by Klebsiella and 2 were missed by GC. Ethanol presence indicates the bacteria present (in M-9L of E. coli and in M-9A of Klebsiella). All samples cultured in both media. |
| Davies et al. [83], 1984 | Gas-liquid chromatograph | 125 strains | Urine culture, 37 °C, 3.5 h, unshaken, media made of yeast-extract peptone medium concentrate. | For headspace samples—liquid temperature 60 °C for 5 min. Production of trimethylamine from acetylcholine biomarker for P. mirabilis, P. vulgaris, P. retgeri, P. inconstants A (not in P. morganii and P. inconstans B). Same species also produced ethanol and ethyl acetate. Ethanol production from arabinose was found in E. coli, K. aerogenes, all strains of S. faecalis. Ethyl acetate produced by K. aerogenes, S. faecalis, E. coli (with n-propanol) and P. inconstans B. No VOCs found P. morganii, S. epidermidis, S. aureus, P. aeruginosa. |
| Aathithan et al. [122], 2001 | Osmetech Microbial Analyzer (OMA) Polymer sensor array | 534 clinical samples | No | Sensitivity of 83.48%, specificity of 87.59% (infection defined as >1 × 105 CFU/mL). 72.3% sensitivity and 89.38% Specificity at 104 CFU/mL cut-off. Threshold PCA values were set using control experiments with reconstituted urine specimens inoculated with bacteria (4 cultures for each organism—result average 4 replicates)—4 blanks for each organism. Organisms: E. coli, E. faecalis, S. aureus, Klebsiella spp., S. saprophyticus and P. mirabilis |
| Pavlou et al. [123], 2002 | Bloodhound BH114 | 25 and 45 urine samples | 4.5 h incubation in enhanced media at 37 °C. Media included: 60% brain heart infusion broth, 40% cooked meat broth. | 37 °C water bath for VOC sampling. First experiment: 20 out of 25 samples UTI confirmed—9 E. coli, 5 P. mirabilis, 6 mixed infection of gram positive cocci and Proteus species, 5 normal urine samples. Genetic Algorithms, Back-Propagation Neural Networks (GA-NN) used—100% prediction on the training set, the 9 samples not used for training were successfully identified. Second experiment: 30 out of 45 samples UTI—13 E.coli, 9 Proteus. spp, 8 coagulase negative Staphylococcus species. First NN 98% prediction rate, 13/14 unknown samples identified correctly. Second NN 95% prediction rate—18/19 unknown identified. |
| Yates et al. [125], 2005 | Cyranose 320 electronic nose and Agilent 4440 Chemosensor | 189 Sensor Responses for Urine (Cyranose 320) | No | Data reduction and optimisation using non-linear model with a kernel width parameter achieved 80% accuracy. Different statistical methods were used accuracy attained: ARX model 65%, 67% (using data from 19/32 sensors), 71% using the most negatively correlated sensor; RBF—50%, using correlation results 65%, Hybrid NARX model—80%. (73% when sensor number was reduced). |
| Yates et al. [129], 2005 | Agilent 4440 | 28 and 40 samples for S. aureus. 32 data points for E. coli | Yes | Agilent 4440 was used along with a pattern recognition algorithm (radial basis function network) to distinguish between methicillin resistant and susceptible S. aureus (100% accuracy obtained) and identify the growth phase of E. coli (68.75% and 81.25% accuracy)—however the ones missed were misclassified by one growth stage. |
| Kodogiannis et al. [124], 2008 | Bloodhound BH114 | 45 urine samples | Culture with Volatile generating kit, 5 h incubation at 37 °C | Headspace sample—37 °C water bath. 45 samples, 30 UTI (confirmed by microscopy + culture)—13 E. coli, 9 Proteus spp, 8 coagulase—Staphylococcus spp. 31 samples used for training, 14 for validation. Averaging the output of 4 feature-based networks (Divergence, Absorption, Desorption, Area) the overall accuracy was 100%. |
| Bruins et al. [130], 2009 | MonoNose | 52 strains | BD-BACTEC™–Plus-Anaerobic/FMedium with the addition of 0.1 mM FeCl3 for measurements. Other commercially available culture broths with different chemicals added were also tested | 104 measurements taken. Bacterial species tested: Escherichia coli, Proteus mirabilis, Enterococcus faecalis, Pseudomonas aeruginosa, Klebsiella pneumoniae, Staphylococcus aureus, Klebsiella oxytoca, Enterobacter cloacae, Clostridium difficile and Salmonella enteriditis, Salmonella typhimurium. Specificity varied with the organism tested—between 67% and 100%. |
| Storer et al. [84], 2011 | SIFT-MS | 90 (10 replicate samples for each microbe) | Inoculation of sterile urine for 6 h. | 10 samples of inoculated urine for each pathogen: E. coli, Proteus vulgaris, P. aeruginosa, S. aureus, S. epidermidis, K. pneumoniae, E. faecalis, or Candida albicans. Presents a table with mean concentration of different VOCs in the urine headspace. |
| Thorn et al. [85], 2011 | SIFT-MS | 11 strains (66—3 repeats for each strain at 5 h and 24 h) | Bacterial plate cultures (20–24 h after inoculation) were emulsified in 5 mL of 1% tryptone-0.5% yeast extract broth, incubated at 37 °C, orbital shaking, 200 rpm, 24 h. | P. aeruginosa, S. aureus, E. coli, P. mirabilis, Burkholderia cepacian, S. pyogenes and E. faecalis were studied. Control media sampling was also used (n = 36). Table with the different VOCs presented and discrimination between species is possible. |
| Wiesner et al. [94], 2011 | IMR-MS | 4 strains | Liquid cultures in Muller-Hinton medium shaken at 200 rpm and 37 °C | Antibiotic sensitive/resistant bacteria strain identification. Two strains of E. Coli (one sensitive and one ampicillin resistant) and two S. aureus strains (oxacillin sensitive/resistant) were used. |
| Bean et al. [86], 2012 | GCxGC-TOF-MS | 2 PA14 cultures and one LB-Lennox blank | 24 h culture at 37 °C in lysogeny broth, Lennox | Discovered 28 new volatiles for P. aeruginosa PA14, 56 compounds in total. Samples heated and stirred for 10 min at 50 °C. Solid-phase microextraction SPME was used for headspace sampling PA14 strains. |
| Jünger et al. [100], 2012 | MCC-IMS | 15 human pathogens | 24 h at 37 °C on Columbia sheep blood agar. | Detection of VOC for E. coli, P. aeruginosa, P. mirabilis, K. pneumoniae, S. aureus, S. epidermidis and other species using MCC-IMS in negative and positive ion mode as well as GC/MS for confirmation. |
| Sabeel et al. [126], 2013 | Cyranose 320 | 13 urine samples, selected 9 | No | Cyranose 320 was introduced in 2 mL of urine 10 times for each of the 13 samples. PCA was used for analysis (first component explained 97.087% of the variation). 9 samples were selected and classified as healthy (2), containing bacteria (1), containing mucus (9)—UTI marker. |
| Roine et al. [127], 2014 | ChemPro 100i, Environics Inc., Mikkeli, Finland | 101 Cultures from clinical samples | Cysteine lactose electrolytedeficient (CLED) medium | Pathogens: E. Coli, S. saprophyticus, Klebsiella spp, E. faecalis. Samples introduced in 36 °C water bath for 15 min prior to headspace analysis. LDA and LR were used. Infected/non-infected discrimination LR—95% sensitivity, 97% specificity; LDA—90% sensitivity, 96% specificity. Bacterial discrimination 95% sensitivity 96% specificity LDA. Validation method leave one out (LOOCV). |
| Boots et al. [104], 2014 | GC-MS | 200 (40 cultures for each bacteria) | Overnight culture, 37 °C, blood agar plates. Bacteria transferred to sterile brain heart infusion broth for 4 h, agitated at 37 °C. | 4 bacteria studied: P. aeruginosa, S. aureus, E. coli, K. pneumoniae. After the overnight incubation the flasks were flushed with high-grade nitrogen (3000 mL over 15 min). Identification of the species is possible as well as methicillin-susceptible/resistant S. aureus strains. |
| Karami et al. [87], 2017 | GC-MS | 18 (2 medium used, 3 time points, 3 organisms) | 24 h in nutrient agar, subcultured at 37 °C in two different broth medium—Muller Hinton Broth and tryptic soy broth. | Identifying VOCs of E. coli, Candida albicans, S. aureus from cultures. For headspace extraction cultures placed on a magnetic stirrer hotplate at 70 °C. Headspace extracted using SPME at 2, 4 and 24 h. |
| Ratiu et al. [91], 2017 | ChemPro-100i (Environics Oy, Finland)and TD-GC-MS (for confirmation) | 90 (3 replicates at 3 time points for each of the 10 cultures) and 30 for confirmation | Agar growth medium, 30 °C for B. subtilis and S. aureus and 37 °C for E. coli | 540 headspace samples were analysed (270 from bacteria and 270 blanks with medium only). Discrimination between blanks and culture, the three species: E. coli, Bacillus subtilis and S. aureus and the different time points (24, 48 and 72 h) was possible. A list of VOC identified with TD-GC-MS is also shown. |
| DeJong et al. [92], 2017 | Surface enhanced Raman spectroscopy | 3 Strains | Liquid Culture in Tryptic Soy Broth Soybean-Casein Digest medium for 16 h at 37 °C transferred to agar-coated plates (Tryptic Soy Agar Soybean-casein Digest). SERS substrate was added. | The technique was applied to both cultures and simulated urine and blood infections with discrimination possible after 16 h. |
| Rees et al. [93], 2018 | GCxGC-TOF-MS | 100 clinical isolates (blood/urine) | 37 °C 200 rpm shaking overnight in Difco Mueller-Hinston Broth (pre-culture) and 1:1000 same conditions for 12 h. | Prior to headspace analysis: 60 min at 37 °C, 200 rpm. Discrimination between the most common infectious pathogens in blood and urine (E. coli, Klebsiella species, P. aeruginosa, P. mirabilis, S. aureus, coagulase-negative Staphylococcus, Acinetobacter species, Candida species, Enterobacter spp, Enterococcus spp,.) was possible with a 95% accuracy using machine learning (random forest) on 203 VOCs or 96% accuracy with 811 VOCs. |
| Smart et al. [89], 2019 | TD-GC-MS | 86 chromatograms, 18 bacterial isolates | Overnight cultures, shaking at 180 rpm, 37 °C. Subcultures grown for 3 h. Antibiotics (cephalexin or ciprofloxacin) added after 2.5 h. | Bacteria: E. coli, K. pneumoniae, P. aeruginosa. Cultures grown in the presence of cephalexin or ciprofloxacin before headspace analysis. Difference in VOC profile was found between resistant and sensitive bacteria |
| Adebiyi et al. [90], 2019 | IBM Electronic Volatile Analyzer | - | 5-day culture in normal human urine at 37 °C | Concentration of bacteria 1 × 109 CFU/mL. Incubated at 37 °C during sampling. Different models (LR, SVM, RF, MLP) had different accuracy (overall, 90%> and some had 100%) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dospinescu, V.-M.; Tiele, A.; Covington, J.A. Sniffing Out Urinary Tract Infection—Diagnosis Based on Volatile Organic Compounds and Smell Profile. Biosensors 2020, 10, 83. https://doi.org/10.3390/bios10080083
Dospinescu V-M, Tiele A, Covington JA. Sniffing Out Urinary Tract Infection—Diagnosis Based on Volatile Organic Compounds and Smell Profile. Biosensors. 2020; 10(8):83. https://doi.org/10.3390/bios10080083
Chicago/Turabian StyleDospinescu, Valentin-Mihai, Akira Tiele, and James A. Covington. 2020. "Sniffing Out Urinary Tract Infection—Diagnosis Based on Volatile Organic Compounds and Smell Profile" Biosensors 10, no. 8: 83. https://doi.org/10.3390/bios10080083
APA StyleDospinescu, V.-M., Tiele, A., & Covington, J. A. (2020). Sniffing Out Urinary Tract Infection—Diagnosis Based on Volatile Organic Compounds and Smell Profile. Biosensors, 10(8), 83. https://doi.org/10.3390/bios10080083







