Gold@Silica Nanoparticles Functionalized with Oligonucleotides: A Prominent Tool for the Detection of the Methylated Reprimo Gene in Gastric Cancer by Dynamic Light Scattering
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Lines
2.2. Reagents
2.3. Oligonucleotides
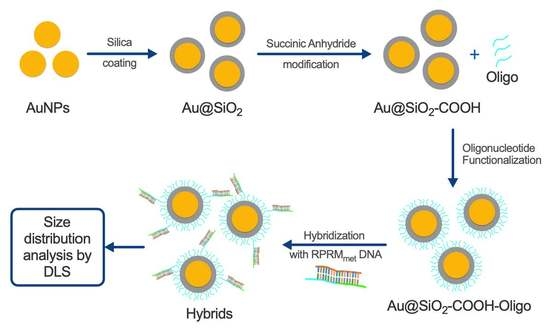
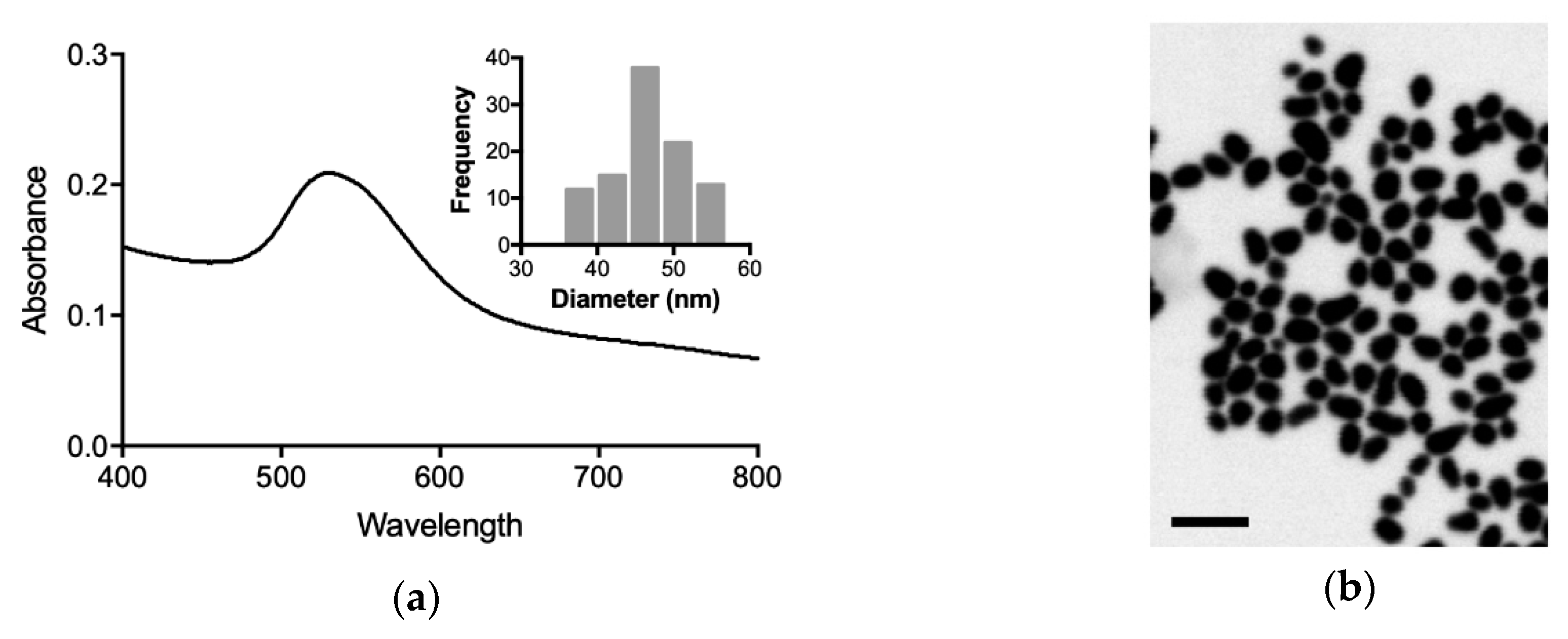
2.4. Synthesis of AuNPs
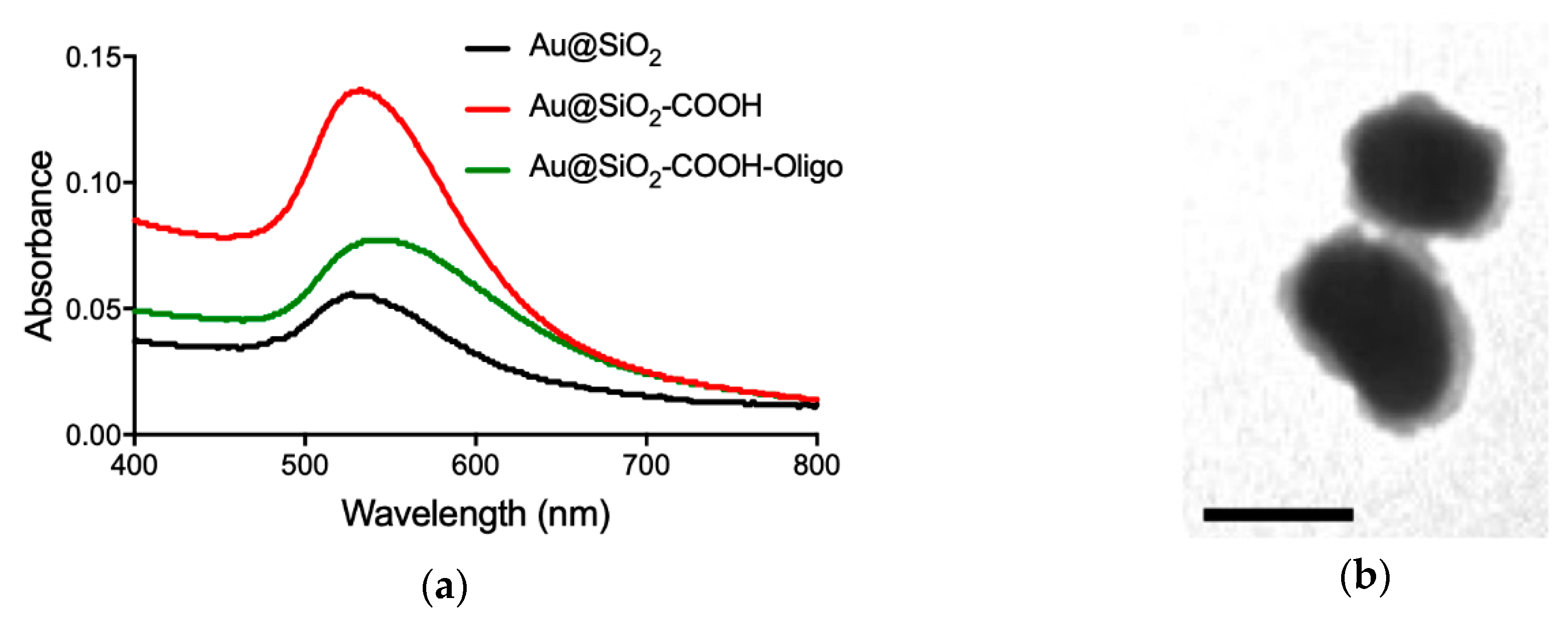
2.5. Synthesis of Core–Shell Au@SiO2–COOH
2.6. Functionalization of Au@SiO2–COOH with Oligonucleotides
2.7. DNA Samples
2.8. DNAmet-RPRM Captured by Hybridization and PCR Assay
2.9. Hybridization Assay
2.10. Statistical Analysis
3. Results and Discussion
3.1. Characterization of Gold Nanoparticles
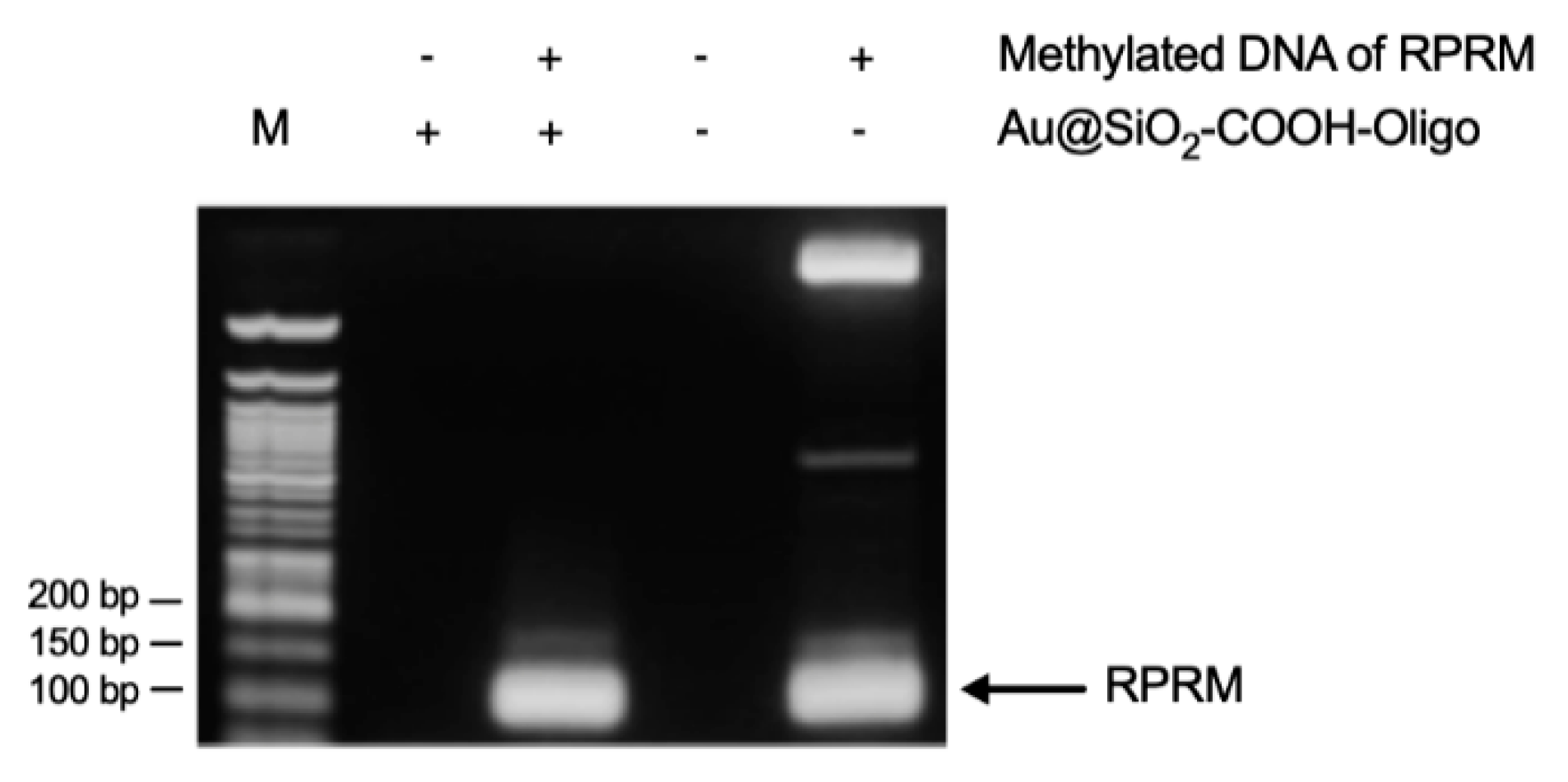
3.2. Capture Assay and Functionality of Au@SiO2–COOH-Oligo
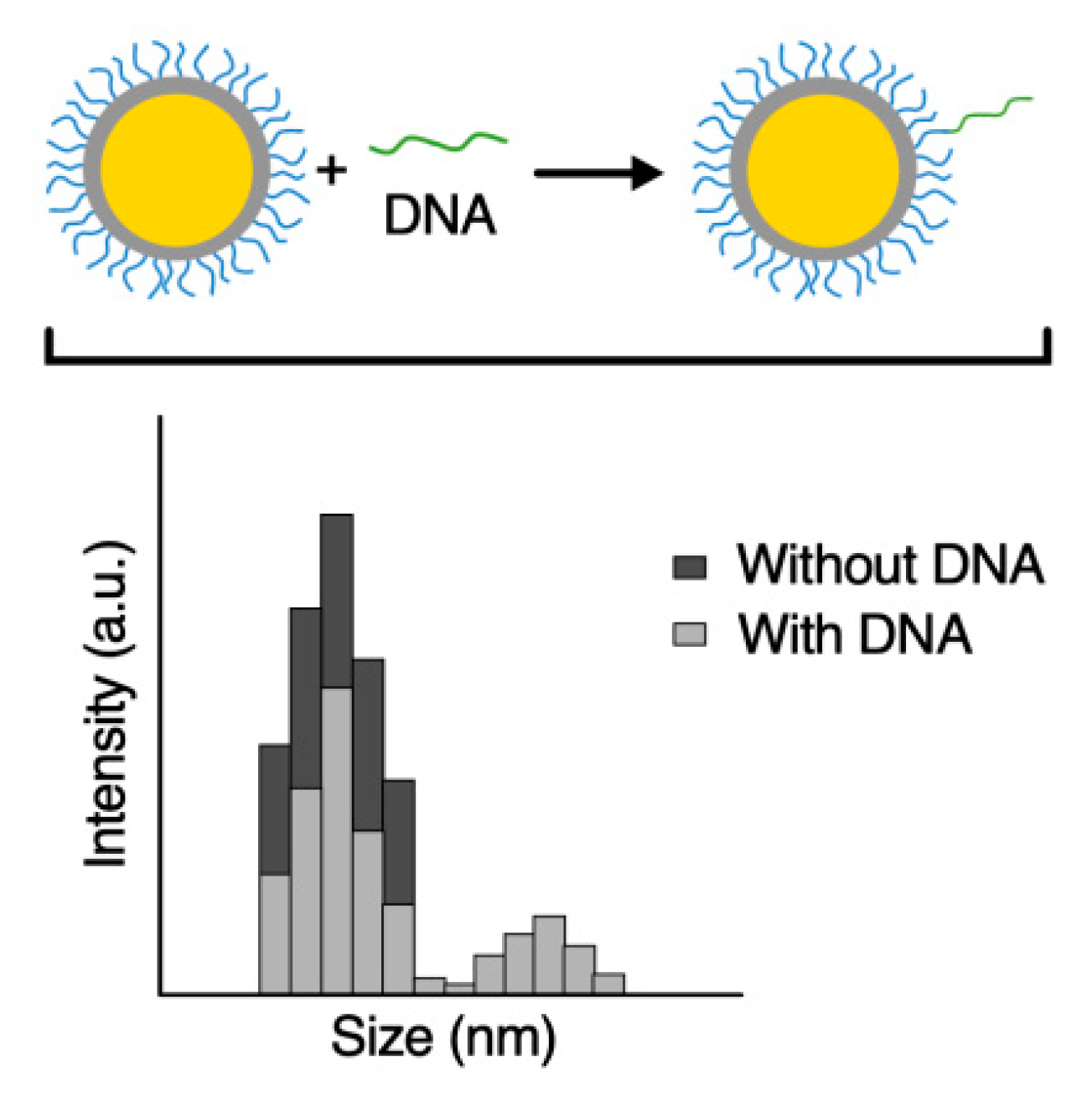
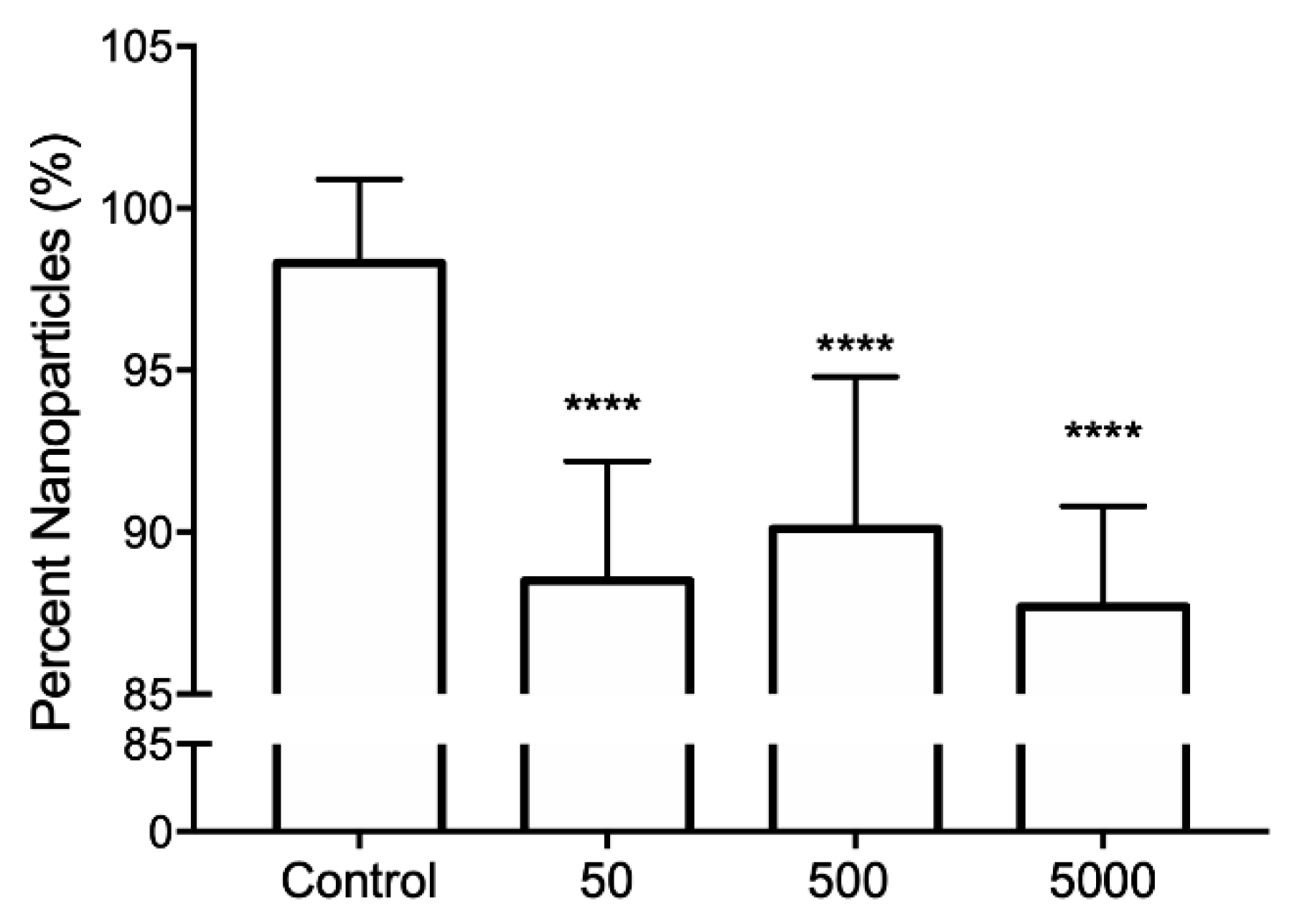
3.3. Hybridization Assay of Au@SiO2–COOH-Oligo with a Synthetic Fragment of RPRM-DNA Analyzed by DLS
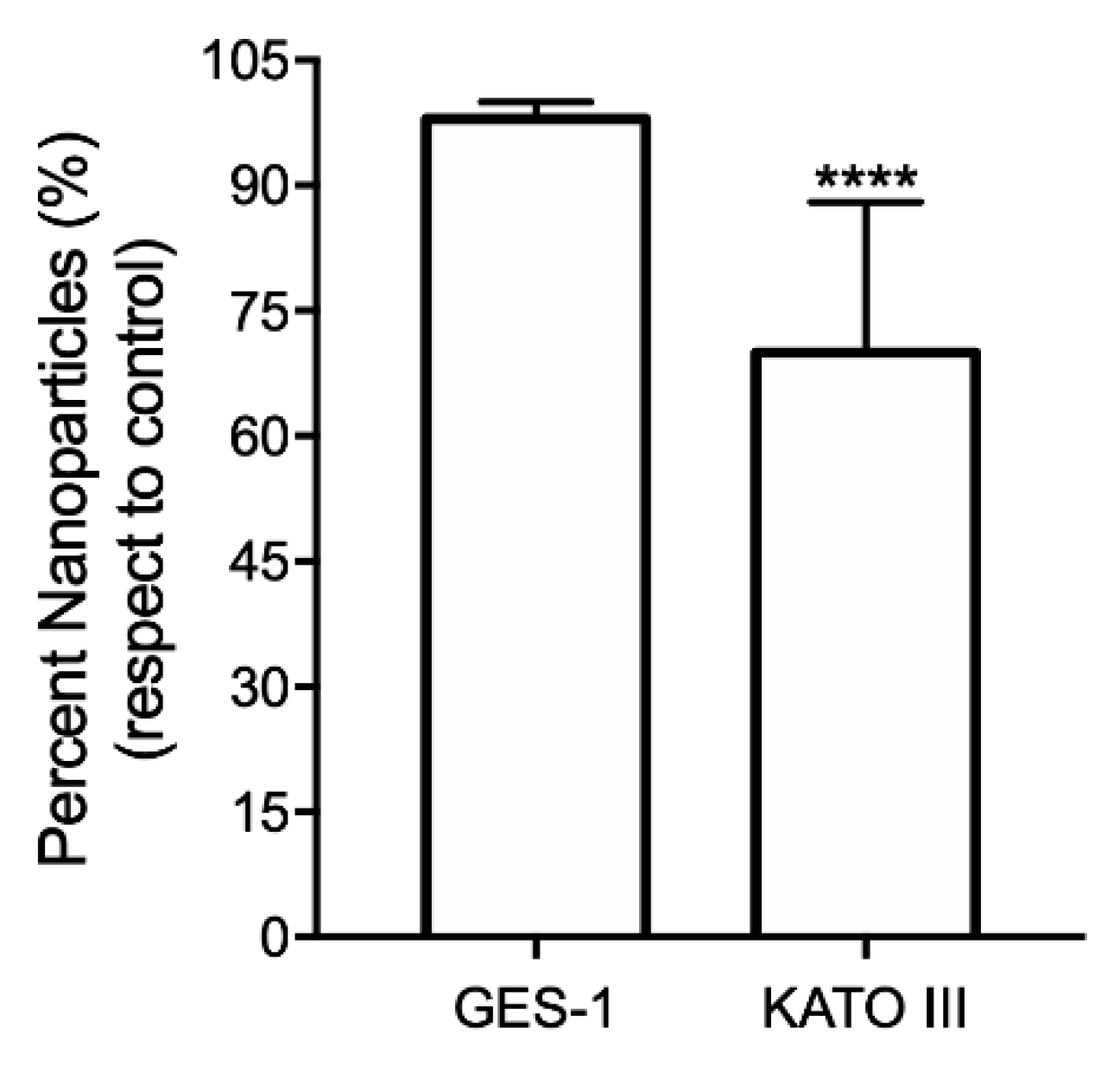
3.4. Hybridization Assay of Au@SiO2–COOH-Oligo with DNA from Cell Lines Analyzed by DLS
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Luo, G.; Zhang, Y.; Guo, P.; Wang, L.; Huang, Y.; Li, K. Global patterns and trends in stomach cancer incidence: Age, period and birth cohort analysis. Int. J. Cancer 2017, 141, 1333–1344. [Google Scholar] [CrossRef] [PubMed]
- Abbas, M.; Habib, M.; Naveed, M.; Karthik, K.; Dhama, K.; Shi, M.; Dingding, C. The relevance of gastric cancer biomarkers in prognosis and pre-and post-chemotherapy in clinical practice. Biomed. Pharmacother. 2017, 95, 1082–1090. [Google Scholar] [CrossRef] [PubMed]
- Ohki, R.; Nemoto, J.; Murasawa, H.; Oda, E.; Inazawa, J.; Tanaka, N.; Taniguchi, T. Reprimo, a new candidate mediator of the p53-mediated cell cycle arrest at the G2 phase. J. Biol. Chem. 2000, 275, 22627–22630. [Google Scholar] [CrossRef] [PubMed]
- Bernal, C.; Aguayo, F.; Villarroel, C.; Vargas, M.; Díaz, I.; Ossandon, F.J.; Santibañez, E.; Palma, M.; Aravena, E.; Barrientos, C.; et al. Reprimo as a potential biomarker for early detection in gastric cancer. Clin. Cancer Res. 2008, 14, 6264–6269. [Google Scholar] [CrossRef] [PubMed]
- Lai, J.; Wang, H.; Luo, Q.; Huang, S.; Lin, S.; Zheng, Y.; Chen, Q. The relationship between DNA methylation and Reprimo gene expression in gastric cancer cells. Oncotarget 2017, 8, 108610–108623. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Yang, X. Implication of Reprimo and hMLH1 gene methylation in early diagnosis of gastric carcinoma. Int. J. Clin. Exp. Pathol. 2015, 8, 14977. [Google Scholar] [PubMed]
- Zhang, X.Q.; Xu, X.; Bertrand, N.; Pridgen, E.; Swami, A.; Farokhzad, O.C. Interactions of nanomaterials and biological systems: Implications to personalized nanomedicine. Adv. Drug Deliv. Rev. 2012, 64, 1363–1384. [Google Scholar] [CrossRef]
- Majeed, M.I.; Bhatti, H.N.; Nawaz, H.; Kashif, M. Nanobiotechnology: Applications of Nanomaterials in Biological Research. In Integrating Green Chemistry and Sustainable Engineering; John Wiley & Sons: Hoboken, NJ, USA, 2019; pp. 581–615. [Google Scholar]
- Petryayeva, E.; Krull, U.J. Localized surface plasmon resonance: Nanostructures, bioassays and biosensing; A Review. Anal. Chim. Acta 2011, 706, 8–24. [Google Scholar] [CrossRef]
- Mayer, K.M.; Hafner, J.H. Localized surface plasmon resonance sensors. Chem. Rev. 2011, 111, 3828–3857. [Google Scholar] [CrossRef]
- Guo, L.; Jackman, J.A.; Yang, H.H.; Chen, P.; Cho, N.J.; Kim, D.H. Strategies for enhancing the sensitivity of plasmonic nanosensors. Nano Today 2015, 10, 213–239. [Google Scholar] [CrossRef]
- Stetefeld, J.; McKenna, S.A.; Patel, T.R. Dynamic light scattering: a practical guide and applications in biomedical sciences. Biophys. Rev. 2016, 8, 409–427. [Google Scholar] [CrossRef] [PubMed]
- Qadami, F.; Molaeirad, A.; Alijanianzadeh, M.; Azizi, A.; Kamali, N. Localized Surface Plasmon Resonance (LSPR)-Based Nanobiosensor for Methamphetamin Measurement. Plasmonics 2018, 13, 1–8. [Google Scholar] [CrossRef]
- Seow, N.; Tan, Y.N.; Yung, L.Y.L.; Su, X. DNA-directed assembly of nanogold dimers: a unique dynamic light scattering sensing probe for transcription factor detection. Sci. Rep. 2015, 5, 18293. [Google Scholar] [CrossRef] [PubMed]
- Turkevich, J.; Stevenson, P.C.; Hillier, J. A study of the nucleation and growth processes in the synthesis of colloidal gold. Discuss. Faraday Soc. 1951, 11, 55–75. [Google Scholar] [CrossRef]
- Liz-Marzán, L.M.; Giersig, M.; Mulvaney, P. Synthesis of nanosized gold-silica core-shell particles. Langmuir 1996, 12, 4329–4335. [Google Scholar] [CrossRef]
- Yuan, L.; He, Y. Effect of surface charge of PDDA-protected gold nanoparticles on the specificity and efficiency of DNA polymerase chain reaction. Analyst 2013, 138, 539–545. [Google Scholar] [CrossRef] [PubMed]
- Vanzha, E.; Pylaev, T.; Khanadeev, V.; Konnova, S.; Fedorova, V.; Khlebtsov, N. Gold nanoparticle-assisted polymerase chain reaction: effects of surface ligands, nanoparticle shape and material. RSC Adv. 2016, 6, 10146–110154. [Google Scholar] [CrossRef]
- Yang, W.; Li, X.; Sun, J.; Shao, Z. Enhanced PCR amplification of GC-rich DNA templates by gold nanoparticles. ACS Appl. Mater. Interfaces 2013, 5, 11520–11524. [Google Scholar] [CrossRef]
- Seow, N.; Tan, Y.N.; Yung, L.Y.L. Gold nanoparticle–dynamic light scattering tandem for the rapid and quantitative detection of the let7 microRNA family. Part. Part. Syst. Charact. 2014, 31, 1260–1268. [Google Scholar] [CrossRef]
- Sutariya, P.G.; Pandya, A.; Lodha, A.; Menon, S.K. A simple and rapid creatinine sensing via DLS selectivity, using calix[4]arene thiol functionalized gold nanoparticles. Talanta 2016, 147, 590–597. [Google Scholar] [CrossRef]
- Saavedra, K.; Valbuena, J.; Olivares, W.; Marchant, M.J.; Rodríguez, A.; Torres-Estay, V.; Carrasco-Avino, G.; Guzman, L.; Aguayo, F.; Roa, J.C.; et al. Loss of expression of reprimo, a p53-induced cell cycle arrest gene, correlates with invasive stage of tumor progression and p73 expression in gastric cancer. PLoS ONE 2015, 10, e0125834. [Google Scholar] [CrossRef] [PubMed]
- Dai, Q.; Liu, X.; Coutts, J.; Austin, L.; Huo, Q. A one-step highly sensitive method for DNA detection using dynamic light scattering. J. Am. Chem. Soc. 2008, 130, 8138–8139. [Google Scholar] [CrossRef] [PubMed]
| AuNP | λmax (nm) | Size a,b (nm) | Zeta Potential a (mV) |
|---|---|---|---|
| Au | 528 | 46 ± 7 | −62 ± 3 c |
| Au@SiO2 | 530 | 71 ± 2 | −26 ± 1 c |
| Au@SiO2–COOH | 533 | 73 ± 1 | −44 ± 1 c |
| Au@SiO2–COOH-Oligo | 536 | 93 ± 2 | −29 ± 1 d |
| Reaction | Size (nm) a | Percentage of NPs a |
|---|---|---|
| Control | 151 ± 11 | 98 ± 3 |
| 50 | 142 ± 11 | 88 ± 4 b |
| 500 | 149 ± 18 | 90 ± 5 b |
| 5000 | 150 ± 9 | 88 ± 3 b |
| GES-1 | 236 ± 77 | 98 ± 2 b |
| KATO III | 240 ± 98 | 70 ± 18 b |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marchant, M.J.; Guzmán, L.; Corvalán, A.H.; Kogan, M.J. Gold@Silica Nanoparticles Functionalized with Oligonucleotides: A Prominent Tool for the Detection of the Methylated Reprimo Gene in Gastric Cancer by Dynamic Light Scattering. Nanomaterials 2019, 9, 1333. https://doi.org/10.3390/nano9091333
Marchant MJ, Guzmán L, Corvalán AH, Kogan MJ. Gold@Silica Nanoparticles Functionalized with Oligonucleotides: A Prominent Tool for the Detection of the Methylated Reprimo Gene in Gastric Cancer by Dynamic Light Scattering. Nanomaterials. 2019; 9(9):1333. https://doi.org/10.3390/nano9091333
Chicago/Turabian StyleMarchant, María José, Leda Guzmán, Alejandro H. Corvalán, and Marcelo J. Kogan. 2019. "Gold@Silica Nanoparticles Functionalized with Oligonucleotides: A Prominent Tool for the Detection of the Methylated Reprimo Gene in Gastric Cancer by Dynamic Light Scattering" Nanomaterials 9, no. 9: 1333. https://doi.org/10.3390/nano9091333
APA StyleMarchant, M. J., Guzmán, L., Corvalán, A. H., & Kogan, M. J. (2019). Gold@Silica Nanoparticles Functionalized with Oligonucleotides: A Prominent Tool for the Detection of the Methylated Reprimo Gene in Gastric Cancer by Dynamic Light Scattering. Nanomaterials, 9(9), 1333. https://doi.org/10.3390/nano9091333