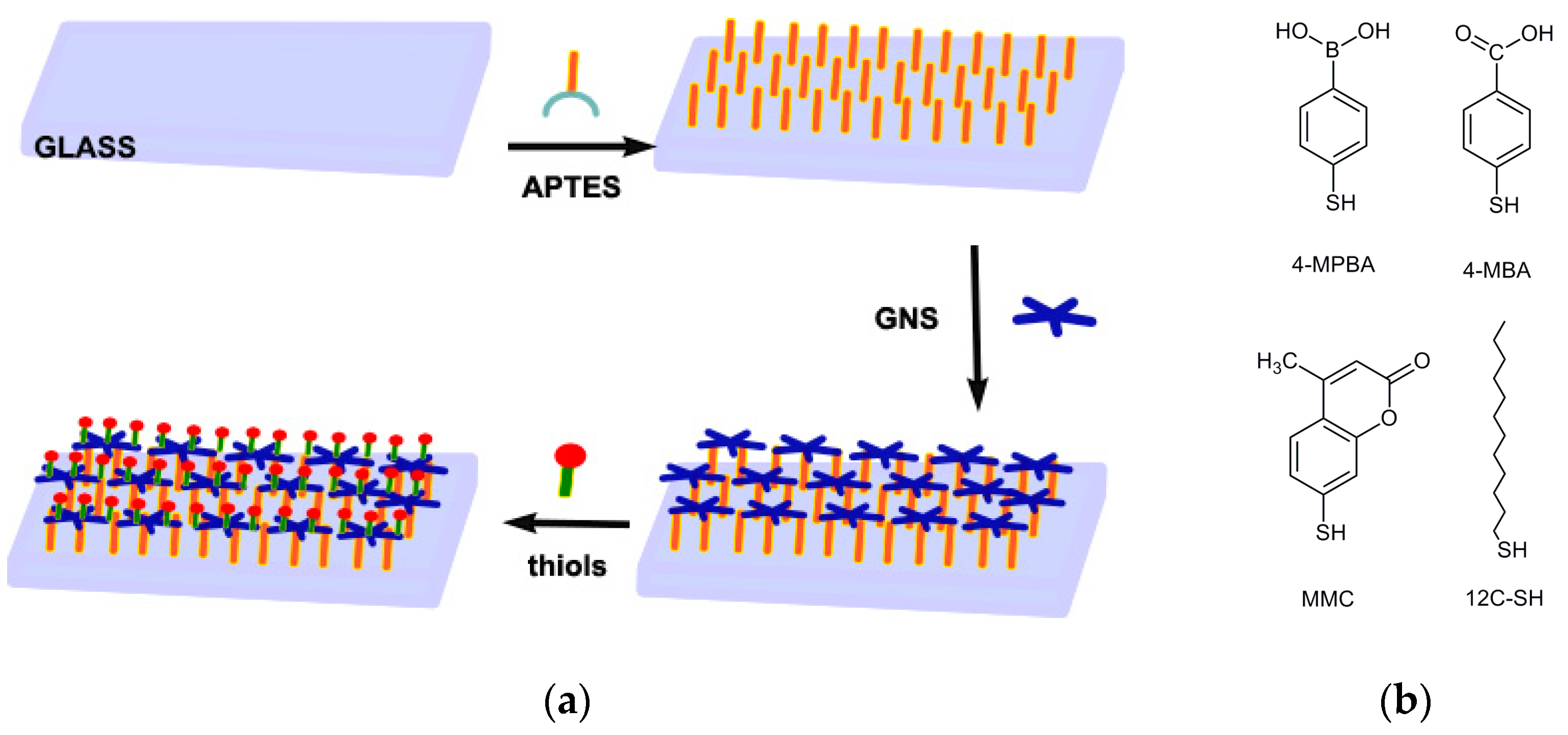
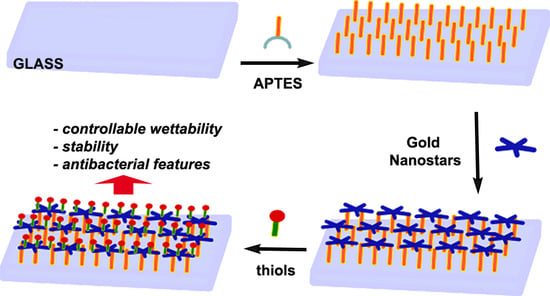
3.1. Preparation of GNS Monolayer on Glass
APTES SAM are prepared on microscopy cover glass slides (21 mm × 26 mm) by means of a reported procedure, with a simple immersion of the slides in a Hellendhal type glass staining jar containing 10% APTES solution in methanol [
4]. Success of silanization is evidenced by the contact angle (c.a.) measurements, showing a change from a value < 10° for the starting glass samples (piranha cleaned) to a value of 56 (5)°.
As already reported, GNS are prepared using an seed-growth method in water, [
18,
19] the synthesis exploiting a zwitterionic surfactant, laurylsulfobetaine (LSB), which acts as the shape-directing agent, and the presence of ~10% (mol) Ag
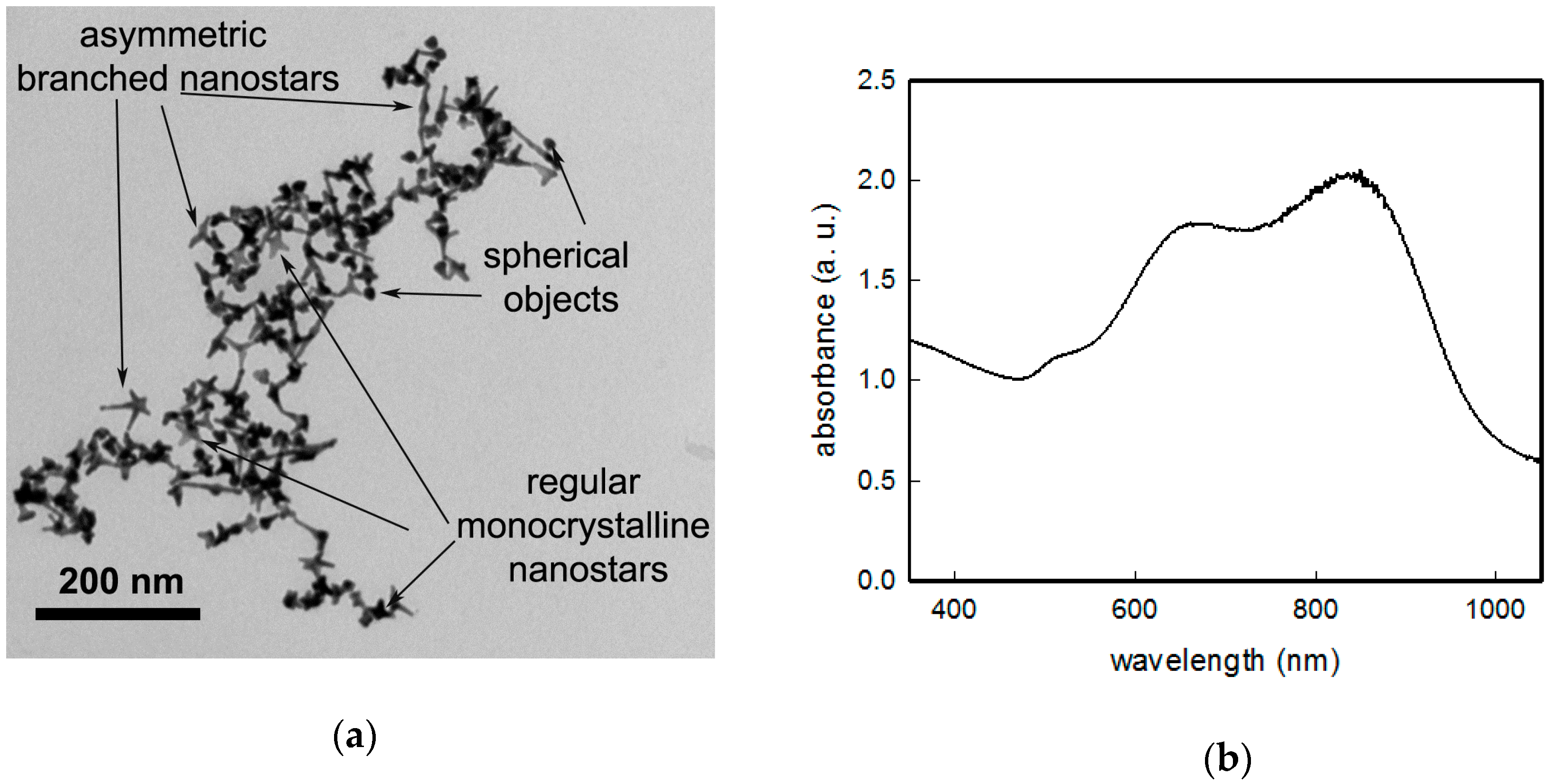
+. The product obtained using this approach consists of colloidal suspensions containing mainly two kinds of anisotropic objects: (i) Asymmetric branched gold nanostars and (ii) monocrystalline symmetric nanostars in a lower percentage. A representative TEM image is reported in
Figure 1a. Composition of these mixtures, as well the dependence of shape features of the two kind of objects is strictly depending on the synthetic conditions, and this has been already described and fully characterized in our previous works [
8,
18] and will not be discussed here. Their extinction spectrum in colloidal suspension (see
Figure 1 for a standard preparation) is dominated by the LSPR of the main (about 45% of objects) component, asymmetric branched gold nanostars, whose maximum can range in the 700–1150 nm interval as a function of synthetic parameters. A second LSPR band can be found in the 690–720 nm interval, owing to the more regular, monocrystalline symmetric nanostars (which represent about 35% of objects), with a central core diameter of 18 ± 4 nm and branches with a length of 12 ± 4 nm. A third, weak LSPR band close to 520 nm is due to the resonance of the spherical cores of the anisotropic objects and to spherical objects coming from seeds, which has undergone to a negligible developing of branches. It is important to notice that also slight and uncontrollable variations in synthetic conditions, which have been prepared as identical can yield slightly different shapes and populations of objects, resulting in colloidal suspensions, which may present small differences in position and intensities of LSPRs bands. Anyway, for this work we managed to obtain products having LSPR optical maxima for the highest wavelength band with a value always close to 850 (20) nm as reported in
Figure 1b.
Resulting GNS were coated by a double layer of surfactant (LSB), and this situation imparted a Z potential of ~−15 mV, as obtained by dynamic light scattering DLS measurements, while the pH of the colloidal suspension was close to 5.5. At this pH, the amino groups of APTES-terminated bulk glass samples were protonated, thus their immersion in the GNS suspension yielded the self-assembly of a GNS monolayer on glass samples, due to the electrostatic interactions between positively charged ammonium and negatively charged nano-objects. After 15 h of immersion of APTES silanized glass samples, samples were taken off and immersed in ethanol and then in water for washing, and finally treated with a flux of nitrogen.
Formation of the GNS SAM turned slides to an evident blue color. As already reported, SEM images show a homogeneous coating (
vide infra) [
11,
17]. Typically, a LSPR blue shift of about 50–70 nm was observed moving from the colloidal suspension in water to the SAM on glass. The shift was due to the change of the local refractive index (n) on passing from water (n = 1.3339) to glass and air at the interface (n = 1.0003).
The c.a. of the samples bringing the GNS monolayer was found to be 51(3)°.
As a result of the described blue-shift, the GNS monolayers samples were found to have LSPR maxima in the range 770–790 nm, a range due to the already mentioned variability of original colloidal suspensions of GNS used. Extinction of the long LSPR band was found to range between 0.20 and 0.25. Samples with spectral features outside of these ranges were discarded (less than 10% of prepared samples).
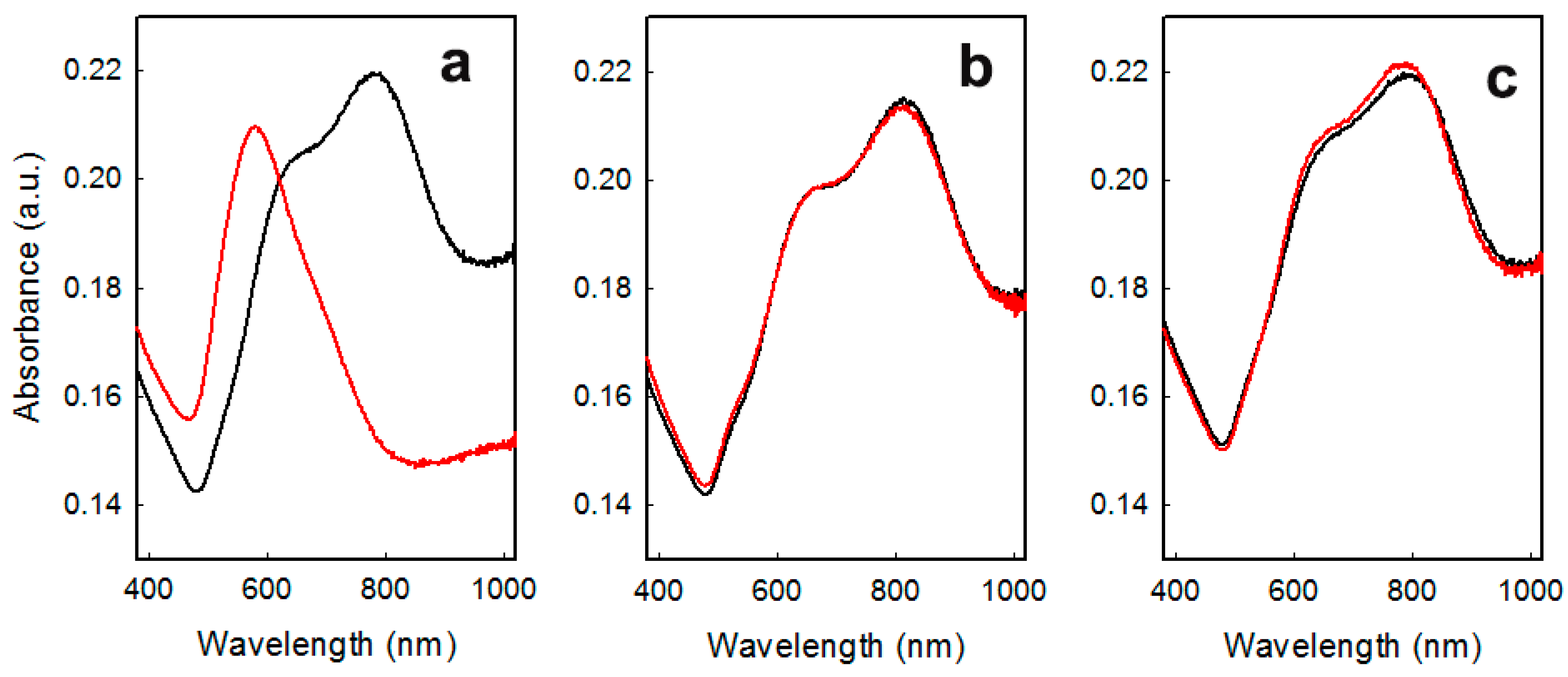
Samples were stable in air only for a few weeks, as evidenced by changes of the LSPR spectra (see
Figure 2a), which suggests the almost total loss of anisotropic features after a couple of months. This is also evidenced by color changes from dark blue to wine red, suggesting that the gold nano-objects had become almost spherical. A similar behavior was observed after prolonged (> 5 h) immersion in water. This represents a striking difference in stability compared to the case of GNS grafted on glass by means of a (3-mercaptopropyl)trimethoxysilane (MPTS) layer we have described in the past [
8]; this is not easily explainable, but could be attributed to the differences in the binding interactions, which are covalent for sulphur (S–Au bond) and electrostatic in the case of amines [
17]. Recently, we have proposed silica coating as a tool to overcome this limited stability of APTES grafted GNS layers [
11,
17].
3.2. Coating the GNS Monolayers with a Monolayer of Thiol
Coating of the described GNS monolayers grafted on glass slides was performed by simple immersion of a set of slides (usually eight) in a Hellendhal type glass staining jar filled (40 cm3) with a 10−5 M thiol solution in EtOH for 1 h. The use of higher thiol concentrations or higher immersion times did not yield different values in the features of the coated monolayers (vide infra). After immersion and subsequent washing, samples could be stored and characterized.
The chosen thiols are reported in
Table 1.
In every case, a moderate red-shift in LSPR spectra was observed, especially for the long band, as a result of the change of the local refractive index experienced by GNS when thiols displace the labile double layer given by LSB. The extent of the red-shift is a function of the thiol features, and mean values observed are reported in
Table 1, and in all cases brought the value of the LSPR maxima into the range of 790–830 nm. The LSPR spectra features were conserved upon coating, indicating that no changes in nano-object morphologies, shape and dimensions occurred during thiol coating.
Measurement of static contact angles gave further evidence of success of thiol coating on the GNS grafted on glass. The value of c.a. moved from the original value of 51(3)° to values, which are peculiar of the functions brought on the used thiols, which are summarized in
Table 1 (for some examples of contact angle measurements images see the
supplementary material). From these data, a first, expected result can be clearly indicated: Modulation of wettability of gold nano-objects layers was obtained with this simple coating method, allowing to reach marked hydrophilicity imparted from 4-Mercaptophenylboronic acid (4-MPBA) and 4-Mercaptobenzoic acid (4-MBA), to intermediate values as in the case MMC, to the high hydrophobicity (120°) imparted by 1-dodecanthiol (12C-SH).
Another interesting feature of these thiol-coated samples was their improved stability in time. As is evidenced by the spectra repeated after 3 months of storing in air without any particular care, no changes in LSPR spectra were observed for coated samples, as reported in
Figure 2 for the coating with 4-MBA (
Figure 2b) and 4-MPBA (
Figure 2c). This behavior, observed for all thiols investigated, was strikingly different compared to what happened for samples aged in the absence of thiol coating for which prolonged storing in air produced a dramatic change in LSPR features, denouncing the loss of anisotropic features of grafted objects. This fact was immediately perceivable also by naked eye, by the color change described above [
17].
Figure 3 represents the SEM images obtained (a) for a freshly prepared and uncoated GNS layer; (b) for the same layer after 2 months of storage in air; (c) for a freshly prepared layer coated with 4-MBA and (d) and for the same layer after 3 months of aging.
As can be observed, the loss of the anisotropic features for the uncoated samples was confirmed, by the fact that only rounded objects could be observed on the surface after 2 months storing, as already suggested by the LSPR spectra. On the other side, the coated samples (whose SEM images were identical to the freshly prepared uncoated ones) showed no changes after a three month storing period. In this case the behavior was also anticipated by the UV-Vis spectra, as described above. SEM data for 4-MPBA coated GNS monolayers, freshly prepared and after 3 months of storing, are reported in the
supplementary material (Figure S1).
The reason of this stabilization was not investigated further, even if it is not completely unexpected. The “native” GNS possesses a layer of LSB surfactant that is quite labile, as demonstrated by the fact that it is readily replaced by added thiols. Coating with thiols apparently freezes the anisotropic shape, avoiding object reorganization towards the more thermodynamically stable spheroidal shape, a fact that cannot be avoided in the presence of the labile surfactant layer, and which is observed in air and, much more faster, when immersed in water. As already cited, this instability is not observed when the same GNS are grafted on glass by means of a MPTS layer. This could be due to the difference in the nature of the bond features: The covalent S–Au bond versus the electrostatic nature of interaction between GNS and protonated amines in the APTES layer. A certain degree of mobility of gold nano-objects was already observed on APTES grafted gold nano-objects [
19], which could contribute to instability. Nevertheless, as already stressed for objects grafted on glass using MPTS instead of APTES, the presence of thiols and of S–Au bonds seems to increase the stability of the nano-objects.
It is important to notice that the addition of thiols also offers the way to remove the surfactant molecules used for the seed-growth synthesis and stabilization of GNS: Surfactants may have an harmful effect when used in applications related to human health, as indeed it has been demonstrated that in some cases they are toxic for our cells [
20]. Therefore their post-synthetic substitution with molecules, which are not toxic and/or more stably bound to nano-objects, should be considered a necessary improvement.
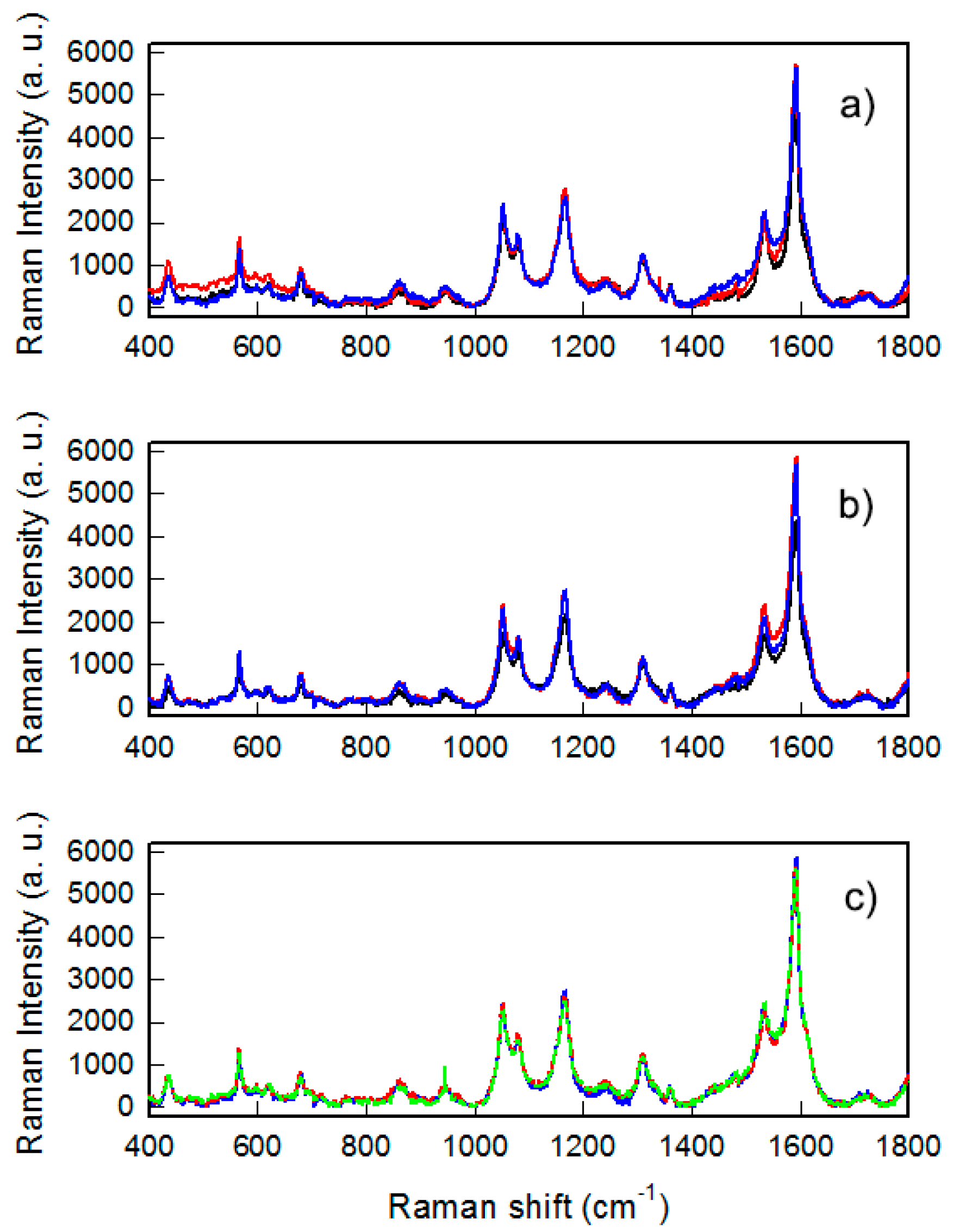
The coating by thiols was also characterized by SERS experiments. SERS enhancement of the usually extremely weak Raman signal allowed us to reveal very low amounts of molecules bound or adsorbed to a nano-structured metallic surface. The enhancement factor (EF) of a specific Raman signal and the spectral features of SERS spectra could give information about specific groups involved in bonds and/or specific geometries with which the molecules are bound or adsorbed [
21]. We measured the spectra of the samples coated with three Raman active thiols: MMC, 4-MBA and 4-MPBA. The SERS spectra taken on different points of a single sample and on different samples for MMC are reported in
Figure 4. As can be clearly noticed, spectra recorder from different points of the sample and from different samples were almost overlapped, with differences in peak intensities of about 10%, a value considered as a marker of highly reproducible SERS substrates [
22]. In particular this is true for the signals in the range 1000–1800 cm
−1 where fall the main Raman modes due to the in-plane C=C stretching of the lactone ring and to the modes inside the benzene ring [
23]. These results indicate the success of the thiol coating because the MMC molecule at this amount can be seen only exploiting the SERS effect. In turn the data reveal both a high uniformity of the coating on the samples coupled to a high reproducibility of the preparations (
Figure 4a,b).
Preparations were also repeated increasing the quantity of MMC in the coating solutions used from 10
−5M to 10
−3M (
Figure 4c): No changes in the whole SERS yield were measured, indicating the fact that the conditions used for a standard coating procedure (40 mL of 10
−5 solutions of thiols for eight slides) ensure the formation of a monolayer of thiols fully covering the available surface of GNS, and that increasing the thiol concentration did not produce any increase of thiol binding, as already suggested by LSPR shift and c.a. values determinations.
SERS experiments were repeated with 4-MBA and 4-MPBA. The spectra are reported in the
supplementary material (
Figures S2 and S3). For these thiols, SERS measurements also gave evidence that the coating was realized with success and that the coated substrates exhibited high reproducibility and homogeneity. As a completion of SERS characterization we calculated the enhancement factors for the three thiols. In all the cases we estimated the EF values by analyzing the Raman modes appearing in all the spectra around 1600 cm
−1, because in these regions the SERS signal from thiols is not disturbed by other Raman/SERS signals and by unresolved background. As already reported above for MMC the Raman features around 1600 cm
−1 originate from the carbon stretching inside lactone and benzene ring; similar origin, i.e., C–C motion in the benzene ring, can be attributed to the signal at 1590 cm
−1 observed both for 4-MBA and 4-MBPA [
24,
25].
The EF values derived from the three thiols ranged from 5.4 × 10
6 for MMC to 2.3 × 10
7 for 4-MBA with the value for 4-MBPA being intermediate. Considering the non-resonant conditions, these values testified the good SERS performances of the nano-objects [
26].
3.3. Photo-Thermal Dehavior
The efficient photo-thermal properties of the GNS used in this work has been already studied on their colloidal suspensions, and when grafted on a SAM obtained with MPTS or APTES on glass [
8,
11,
18]. Here we tested the photo-thermal behavior for the described samples, coming from the same preparation sets described and characterized so far, for the coatings with 4-MBA, 4-MPBA and C12-SH, which are the thiols giving the most interesting wettability values (i.e., the most hydrophilic and hydrophobic: The sample coated with MMC, having an intermediate c.a. value, were not investigated at this point). The slides were irradiated with continuous laser sources at 808 nm, using increasing power ranging from 39 to 207 mW (laser spot area = 0.785 cm
2, irradiances 0.049–0.264 W/cm
2). The temperature reached on the samples was measured vs. time using a thermocamera. The same steep ascending-plateau profiles were observed for all samples at every irradiance, as reported in
Figure 5a. The maximum equilibrium temperature was reached in about 5 min, with ΔT increasing linearly from 3 to about 20 °C as a function of the applied laser power. Interestingly, no variations were observed in the relation between applied power and ΔT obtained on changing the thiol coating: The slopes of the lines interpolating the experimental ΔT vs. power data were almost the same for all samples, see
Figure 5b. Thus, it was possible to conclude that laser irradiation of the GNS monolayers at 808 nm gave the same efficient photo-thermal conversion independently from the choice of the thiol used for coating, as expected from the observation that thiol coatings produced only negligible variations in the position and intensities of LSPR absorptions, which were only responsible for the photo-thermal efficiency.
The stability of the layers upon laser irradiation was controlled taking SERS spectra after 30 min of irradiation using the higher irradiance value, and comparing them with the spectra taken before irradiation. This was repeated three times for the samples coated with 4-MBA and 4-MPBA (as 12C-SH had no Raman features to be easily exploited with SERS): No changes in SERS patterns and intensities were perceivable, as can be observed in
Figures S4 and S5. The same was observed when measuring LSPR features of the samples and their c.a. values. All these findings suggest that laser irradiations and the consequent hyperthermia did not produce any change in layers features.
3.4. Hyperthermia Based Antibacterial Effects on Planktonic Bacteria Cells
To test the effects of hyperthermia in eliminating Gram + as Gram – “planktonic” bacterial specimens (i.e., fluctuating, not yet organized in biofilm), samples of glass slides were opportunely cut in order to be completely covered by the laser spot while kept in contact with the desired bacteria suspensions, and were irradiated for 30 min with the described laser source at 808 nm, at an irradiance of 0.264 W/cm2. Strains of E. coli and S. aureus were chosen as representative of Gram – and Gram + bacteria, respectively. The “thermal microbicidal effect”, MET, can be calculated using the formula MET = log NC − log NT, where NC is the number of CFU/mL developed after the contact with a functionalized glass sample in the absence of irradiation, taken as a control, and NT the number of CFU/mL found in a sample kept in identical conditions but after exposure to the laser irradiation.
It is important to observe that in these conditions, the ME
T has to be ascribed only to the hyperthermia given by the photo-thermal effect, as any other influence from the features of the surfaces in the absence of irradiation is considered in the control count. Nevertheless, the so called, “microbicidal effect” (ME) measured in the absence of any irradiation, using the same contact time and calculated using a blank glass as a control, gave very low values (well below 1) in all cases. It is also important to stress that laser irradiation alone, in the absence of plasmonic objects exerting photo-thermal features, does not cause any harm to bacteria, as already observed [
8,
9,
10,
11].
Table 2 reports the ME
T obtained for freshly prepared samples of slides bearing uncoated GNS, and for samples coated with a monolayer of 4-MBA, of 4-MPBA and C12-SH.
As can be noticed, a strong microbicidal effect was obtained in all samples for the two bacteria strains investigated. In all cases the ME
T was higher than 4, a value corresponding to the elimination of at least 99.99% of bacteria and, specifically for
E. coli, no bacteria at all were found after laser treatment. These results show that the laser treatment was able to eliminate almost completely the two investigated strains of planktonic bacteria cells after 30 min of irradiation at 0.264 W/cm
2 (a value which is well below the considered safe for the exposure of skin [
27]) at a wavelength that can be used in-vivo. Indeed, behavior of these surfaces in complex physiological environments, as well as their effect on healthy cells needs further investigation. In any case, the stability in physiological media and safety towards healthy cells has been already assessed in our previous papers for colloidal GNS coated with thiols like 7-Mercapto-4-methylcoumarin and PEG-thiols [
28,
29].
Of course, one can notice that the uncoated GNS layers (i.e., the samples which were not treated with the thiols and which conserve the original surfactant layer on the part of the GNS surface exposed to the environment) were already extremely active in killing planktonic bacteria with their photo-thermal features. Nevertheless their practical use was almost totally hampered by their instability, while, on the other side, the coating with thiols impart stability without altering photo-thermal efficiency in bacteria killing (see
Figure S6 for a schematic illustration of the improved stability).