1. Introduction
Graphite is the most widely used anode material in lithium-ion batteries (LIBs) owing to its rich reserves, good electrical conductivity, low charge/discharge potential, and excellent cycling stability. Unfortunately, the electrochemical performance of graphite as an anode material is poor in sodium-ion batteries (SIBs) using a conventional carbonate electrolyte. This is due to the passivation layer formed on the graphite surface being less effective than the protective layer formed in a LIB, and due to the continuous decomposition of electrolytes [
1]. In addition, due to the thermodynamic instability of sodium, only a small amount of sodium can be stored in graphite. The sodium storage capacity of graphite in a SIB is approximately 1/10th of the lithium storage capacity in a LIB. The reason for this low reversible capacity is not simply the larger ionic radius of Na than that of Li (K is stored more in spite of its larger ionic radius than that of Na). It has been reported that sodium-graphite intercalation compounds (Na-GICs) are more thermodynamically unstable than other alkali-GICs.
In particular, it is estimated that there are three reasons for this instability [
2,
3,
4]. First, the binding energy of GICs are composed of, not only the ion-binding component, but also the covalent bond between the metal and the carbon atom [
2]. That is, in the case of alkali metals, as the atomic number increases, the electronegativity decreases, and the alkali-graphite bond stabilizes. In the case of lithium, the covalent contribution of lithium-graphite bonds further stabilizes the bonding state, even if the atom is small. However, sodium has no covalent contribution, and its atomic diameter is smaller than that of other alkali elements, making it more unstable. Next, among the energies required for the M-GIC formation process (energy
Ed required for the alkaline element to detach from the bulk state, energy
Eg necessary to strain the graphite, and energy
Eb reduced by inserting the M atom into the expanded graphite,
Ef =
Ed +
Eg +
Eb), the instability is seen as a result of the weakening of
Eb. In other words, sodium is the most weakly bonded to the substrate, which is caused by competition between the ionization energy trend below the column of the periodic table and the ion-substrate coupling energy [
3]. Finally, the local repulsive interaction between the Na ion and the graphene layer predominantly destabilizes Na-GICs [
4]. Therefore, co-intercalation reaction, which promotes the intercalation of Na in graphite by preventing direct interaction between Na and graphene by screening Na with ether solvent molecules, is proposed as an effective strategy [
5]. In recent years, carbonate-based electrolytes have been replaced by ether-based electrolytes, and the use of ether-based co-intercalation reactions have been reported to dramatically increase the reversible capacity of graphite [
5,
6,
7]. However, it is still unsatisfactory for practical needs, and the coulombic efficiency (C.E.) varies regardless of the specific surface area.
Therefore, a new carbon material for SIBs was designed to increase the reversible capacity considerably and bring the C.E. to the practical level. The surface area in this study (at least for C.E.) has not been considered because previous studies on storing sodium ions via the co-intercalation reaction have revealed the following results. First, the formation of the solid-electrolyte interphase (SEI) has no relation to the magnitude of specific surface area, and in some cases, no SEI is formed at all. In addition, a very thin, strong SEI has been reported to form [
5,
7,
8,
9,
10]. Therefore, it was concluded that the larger the specific surface area (that is, the surface pore structure is well developed), the more effective co-intercalation reaction can be generated after the electrolyte forms a complex with sodium without interfering with the pore. It was further concluded that SEI formation could be suppressed and the first cycle C.E. could be greatly improved. An attempt was made to increase the reversible capacity by creating many voids in which sodium could be stored.
It was found that a carbonaceous material with a large storage space is the most suitable because it has a large specific surface area, a wide interlayer distance due to the turbostratic structure, a smooth capacitive reaction, and many internal voids. It was considered that the optimum structure which can have this feature is the carbon black structure. That is, as the primary particles are small, the diffusion distance is short and the specific surface area is large, a capacitive reaction may occur due to the disorder in the interior because of the many internal voids.
In this study, a carbon material featuring these characteristics was synthesized from carbon black for SIBs and very good results were obtained. The carbon material was synthesized using the solution plasma process and the details of synthesis and material analysis are discussed below.
3. Results and Discussion
Figure 1a shows the transmission electron microscopy (TEM) image of the synthesized SCB. As can be seen, spherical primary particles of about 20 to 30 nm size are entangled in different directions, and the aggregate size is at least 1 µm.
Figure 1b is a magnified TEM image of the primary particles, which shows that the microcrystalline domains form a turbostratic structure and are oriented in different directions. These domains are composed of only a few layers and have a width of approximately less than 5 nm. Numerous microcrystalline domains of this type aggregated to form spherical primary particle agglomerates, and many voids formed between these domains. Therefore, the aggregate has the structure of carbon black, while the internal structure of the primary particles is almost the same as that of hard carbon. The XRD pattern also shows a peak that is typical of amorphous carbon (
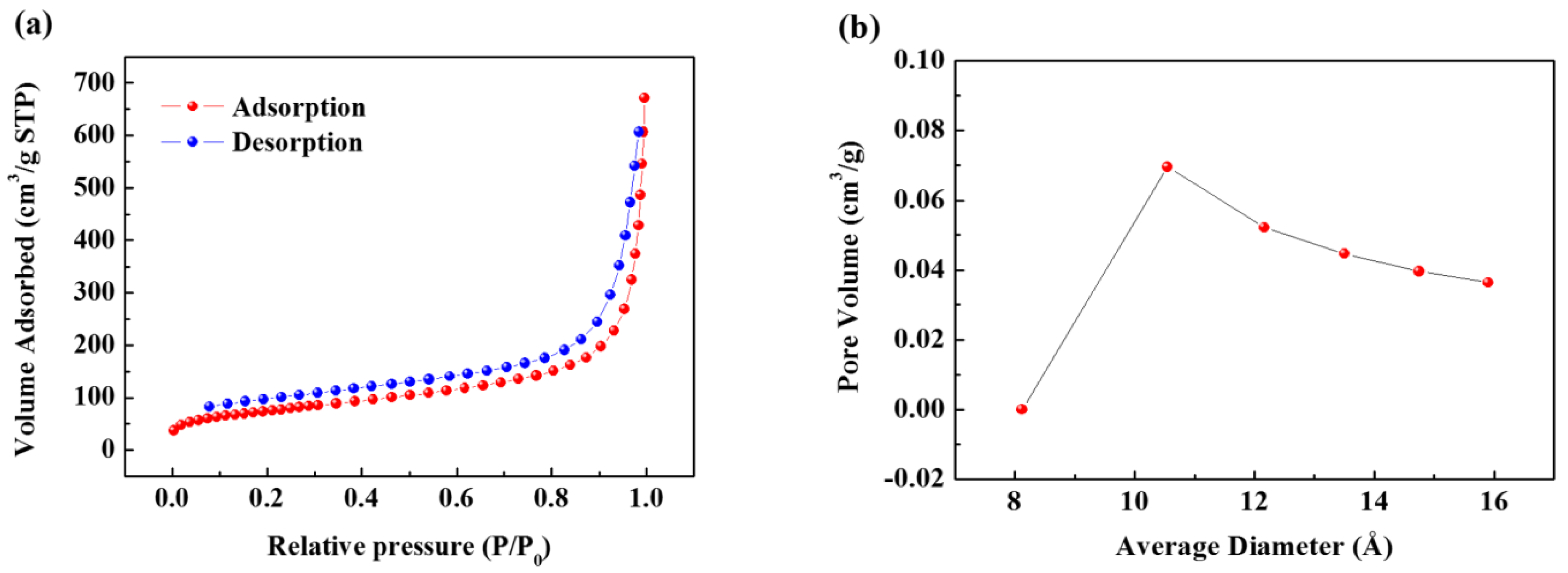
Figure S1). Next, N
2 gas adsorption-desorption curves were obtained by the BET method to evaluate the specific surface area of the SCB (
Figure 2a). The specific surface area calculated from the isotherm curve was approximately 268 m
2 g
−1. This value is similar to commercial carbon black, and the large specific surface area results from the pore structure that formed on the surface. The pore size distribution (
Figure 2b) is calculated from the desorption curve, and it is confirmed that many pores of approximately 10 Å exist on the surface. Therefore, based on the results of TEM and BET, it is confirmed that the SCB is a spherical particle with 10 Å pores on average on its surface and a void structure inside. On the other hand, the calculated planar area of [Na-DEGDME]
+ was reported to be 7.27 Å × 9.37 Å. Therefore, it is small enough to diffuse into the SCB. In addition, triple complex intercalation of [Na-DEGDME]
+ is also possible if the distance between graphite layers is wide enough.
Next, the electrochemical performance of the SCB was evaluated.
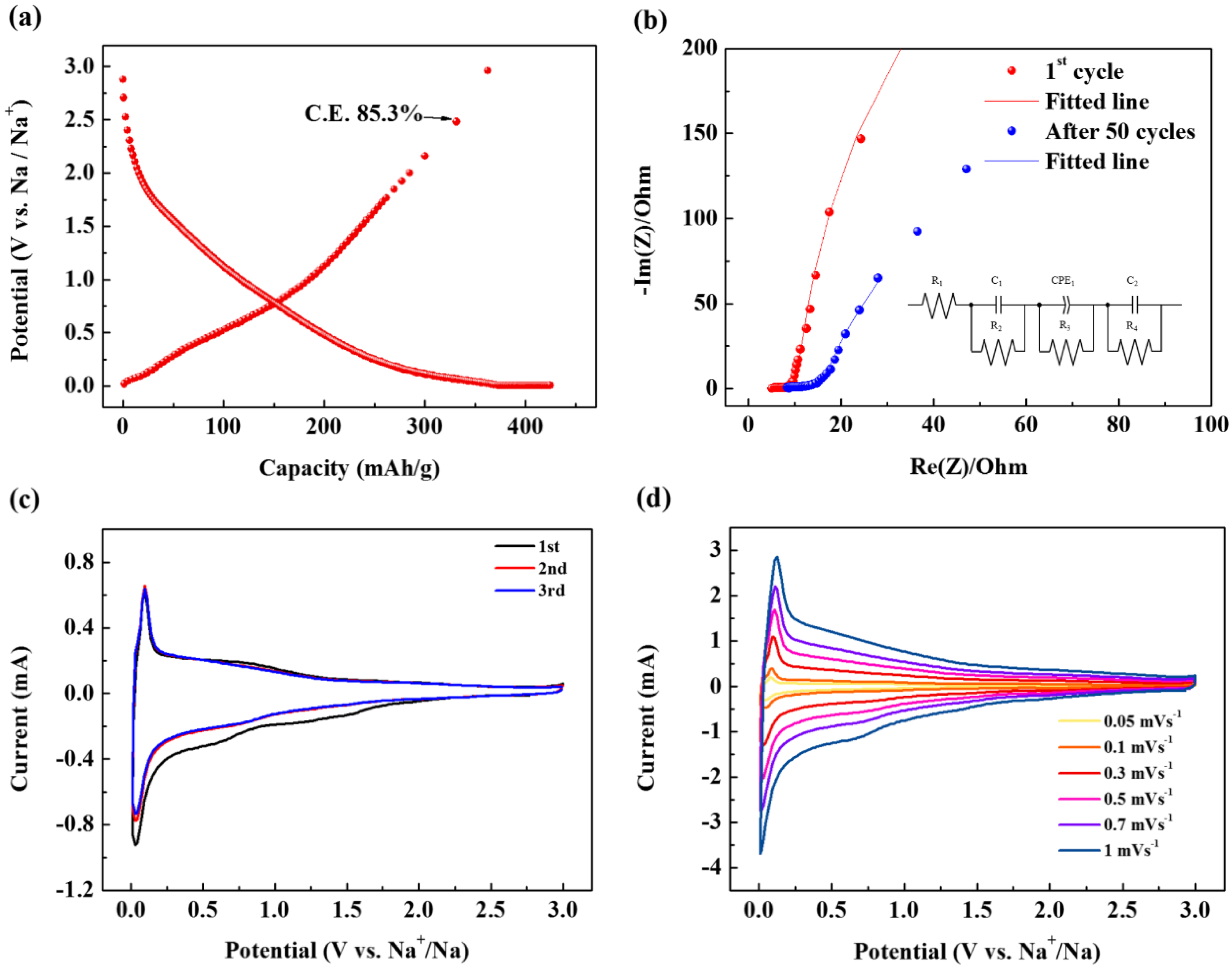
Figure 3a represents the first cycle charge/discharge profile of SCB. Here, it is noteworthy that the first cycle C.E. exceeds 85% despite the large surface area of the material. This shows that there is no correlation between the specific surface area and C.E. In fact, graphite with a specific surface area of 1 m
2 g
−1, which is 1/330 of the material synthesized in this study, showed 50% C.E. in the ether-based electrolyte via the co-intercalation reaction [
5]. In the case of materials with a specific surface area of 100 m
2 g
−1, which is one-third of the materials synthesized in this study, the C.E. is extremely low at 55%, even though the same ether-based electrolyte is used [
15]. In addition, N330 carbon black with the same carbon black morphology has a specific surface area of only 75 m
2 g
−1, but a C.E. of 64%, which is much lower than the SCB [
7]. These results indicate that there is no direct relationship between specific surface area and C.E. In other words, co-intercalation cannot occur because a SEI cannot be formed if a wide pathway that can cause a co-intercalation reaction, rather than the specific surface area being well developed. Thus, the surprisingly high efficiency demonstrated in this study can be a strong indicator of the absence of a SEI layer. The absence of a SEI was also evident from the comparison of electrochemical impedance spectroscopy (EIS) measurements as seen in
Figure 3b. The depressed nature of the semicircle can be attributed to the merging of the two different semicircles. One is due to the SEI and the other is from the charge-transfer process. Therefore,
R1 means the internal resistance (including the resistance of the electrolyte and electrode) and
R2 corresponds to the resistance of SEI layer while
C1 indicates the resultant capacitance from SEI layer.
R3 is the charge transfer resistance in the intermediate-frequency region and constant phase element (CPE) is related to the surface property of the electrode.
R4 is the Warburg resistance and
C2 indicates the double layer capacitance caused by the ion transfer in the electrode material. The EIS graph in
Figure 3b shows that that the resistance of the SEI (
R2) is very small, and even after 50 cycles, it increases very little (
Table 1). In particular, this value is much smaller than a SCB discharged in a carbonate-based electrolyte (
Figure S2 and
Table S1). Therefore, these results are good evidence that no SEI formed on the SCB surface when using the ether-based electrolyte.
Therefore, in this study, the irreversible capacity of approximately 15% in the first cycle is attributed to sodium trapped in some pores in the carbon rather than the SEI formation. When Na is inserted into a soft carbon turbostratic structure, local and macro-structural expansion occurs inside the structure, which is sometimes irreversible. Thus, some Na ions may become trapped in the intercalated site [
16].
Figure 3c shows the cyclic voltammogram (CV) of the SCB. The CV clearly shows peaks arising from reversible oxidation and reduction reactions. These peaks appear when Na is stored in the carbonaceous material together with the electrolyte [
5]. Thus, the appearance of this peak demonstrates the successful co-intercalation of Na and solvent. On the other hand, in the CV, no distinct peak indicating the decomposition of electrolyte appears, and it can be seen that the 2nd and 3rd peaks are superimposed. This implies that no SEI is formed on the electrode surface, and almost all the Na can be reversibly drawn into the carbon while some amount of Na is trapped.
On the other hand, to investigate the surface dynamics of the electrode reaction, an additional CV analysis at a low scan rate (
Figure 3d) was conducted. Generally, in lithium and sodium-ion cells, the CV current is represented by a combination of capacitive and intercalation/deintercalation currents. Therefore, the total charge stored on the electrodes can be distinguished by a combination of capacitive and intercalation mechanisms. According to Power’s law, the voltage-current response of electrode active materials at various sweep rates can be summarized as follows:
where
v is the scan speed (V s
−1), and d and b are adjustable parameters. The value of
b can be determined from the slope of the linear fit of the log
i vs. log
v plot at a fixed potential (V). At
b = 0.5, the peak current is linearly related to the square root of the scan rate (
v1/2), which is considered a typical diffusion-controlled lithium storage process. The peak current at
b = 1 is proportional to the scan speed and indicates surface-controlled charge storage operations [
17,
18,
19]. The plot peak currents (
ip and
is) as a function of the scan rate in the logarithmic scale are shown in
Figure 3 and
Figure S4. Both the curves show a good linear relationship with
R = 0.98 and a slope of 0.9 (close to 1), suggesting a surface-controlled process such as adsorption. Therefore, most of the current at the peak potential is capacitive. This indicates that the reaction rate is surface controlled and therefore very fast [
20]. Based on the dependence between the peak current and scan rate, the contributions of capacitive and intercalation elements can be estimated using the following equations [
21,
22]:
where
k1(
v) and
k2(
v1/2) represent the capacitive current and intercalation/deintercalation current respectively, obtained by modifying the above equation.
where
k1 and
k2 correspond to the slope and slice of the linear fitting of the plot of
i(V)/
v1/2 vs.
v1/2 respectively. If the values of
k1 and
k2 are obtained, the contribution from the intercalation/deintercalation and the capacitive mechanism can be easily distinguished from each potential. The capacitive and diffusion-controlled contributions to the total capacitance are shown in
Figure 3e.
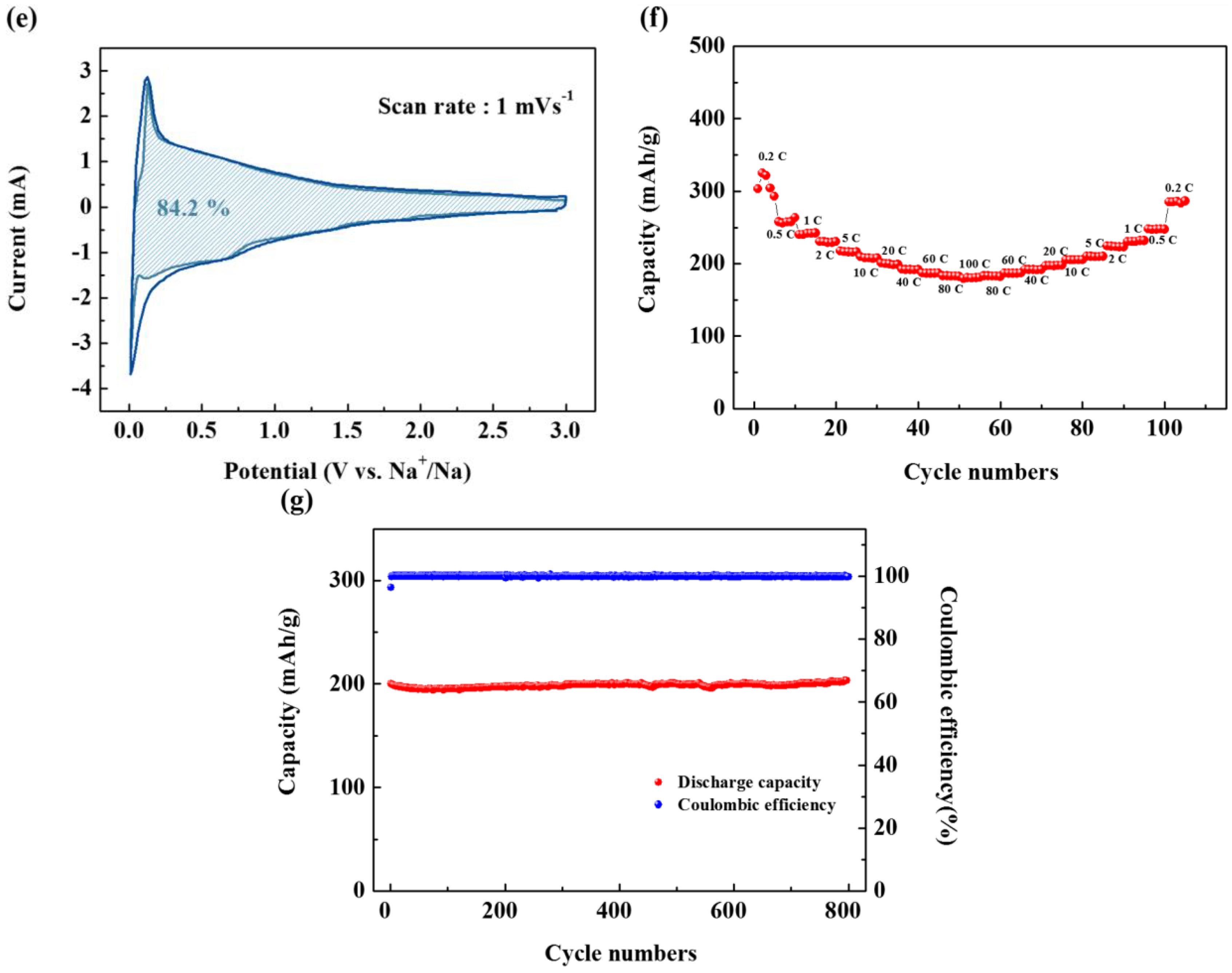
Figure 3e indicates that capacitive charge storage plays an important role in the overall capacity of the electrode, and the ratio accounts for 84.2% of the total charge storage.
The high contribution of capacitive charge storage is believed to be due to the turbostratic structure of the surface pores formed on the surface of the primary spherical particles. This provides a large electrolyte contact area, thus facilitating the capacitive reaction. Therefore, the CVs obtained at a low scan rate provide evidence that large pores exist on the surface, as predicted by BET. The large pathway due to the turbostratic structure together with this surface pore structure, plays an important role in the capacitive reaction.
On the other hand, these surface structures also provide conditions for excellent rate capability. The rate capability of the SCB is shown in
Figure 3f. An initial high capacity of 303 mAh g
−1 is observed at a current density of 0.2 C after five cycles. On the other hand, when the C-rate was set continuously as 0.2, 0.5, 1, 2, 5, 10, 20, 40, 60, 80, 100, and 0.2 C, reversible capacities of 300, 260, 240, 230, 216, 209, 200, 191, 187, 183, 180 and 286 mAh g
−1 were obtained. As the current density decreased, the capacity fully recovered. This indicates that the capacitive reaction inside Na-C is stable at various current densities and that the reversible capacity at 0.2 C is 60% of that at 100 C. This implies that even at a high C-rate, the resistance is not large for a charge transfer. Such excellent output characteristics can be achieved only without a SEI. This further reinforces that a SEI was not formed.
Figure 3g shows the cycling performance of the SCB. As can be seen, the SCB shows the same reversible capacity over 1000 cycles, which is about 1.5 times of the previously reported hard carbon [
5].
From the above results, it was confirmed that carbon black-type materials with a turbostratic structure (i.e., a wide pathway) containing many voids are highly efficient structures for the storage of Na ions via capacitive reactions. Therefore, for a SIB fabricated using the capacitive reaction, it is possible to produce an excellent anode material for the SIB by considering this structure and the composition with other materials.
Finally, a SCB sample was taken from the coin cell after 100 cycles and observed it via TEM. The SCB cycled with the ether-based electrolyte using a carbonate-based electrolyte was also compared (
Figure 4). In the case of the SCB taken from the cell using the DEGDME, the SEI layer was not observed, as predicted by the electrochemical analysis. However, in the case of the SCB using the carbonate electrolyte, a thick SEI layer formed on the surface.