Carbon Dots and Graphene Quantum Dots in Electrochemical Biosensing
Abstract
1. Introduction
2. CDs
3. GQDs
4. GQDs and CDs in Electrochemical Biosensing
4.1. Electrochemical Enzymatic Biosensing Involving GQDs
4.2. Electrochemical Affinity Biosensing Involving GQDs or CDs
4.2.1. GQDs in the Preparation of Electrochemical Immunosensors
4.2.2. Electrochemical Nucleic Acid Biosensors and Aptasensors Involving GQDs or CDs
5. Conclusions, Main Challenges to Solve, and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- García-Mendiola, T.; Bravo, I.; Lopez-Moreno, M.; Pariente, F.; Wannemacher, R.; Weber, K.; Popp, J.; Lorenzo, E. Carbon nanodots based biosensors for gene mutation detection. Sens. Actuator B Chem. 2018, 256, 226–233. [Google Scholar] [CrossRef]
- Yáñez-Sedeño, P.; Campuzano, S.; Pingarrón, J.M. Carbon nanostructures for tagging in electrochemical biosensing: A Review. Carbon 2017, 3, 3. [Google Scholar] [CrossRef]
- Teradal, N.L.; Jelinek, R. Carbon nanomaterials in biological studies and biomedicine. Adv. Healthc. Mater. 2017, 6, 1700574. [Google Scholar] [CrossRef]
- Suvarnaphaet, P.; Pechprasarn, S. Graphene-Based Materials for Biosensors: A Review. Sensors 2017, 17, 2161. [Google Scholar] [CrossRef]
- Borenstein, A.; Hanna, O.; Attias, R.; Luski, S.; Brousse, T.; Aurbach, D. Carbon-based composite materials for supercapacitor electrodes: A review. J. Mater. Chem. A 2017, 5, 12653–12672. [Google Scholar] [CrossRef]
- Sun, H.; Wu, L.; Wei, W.; Qu, X. Recent advances in graphene quantum dots for sensing. Mater. Today 2013, 16, 433–442. [Google Scholar] [CrossRef]
- Pedrero, M.; Campuzano, S.; Pingarrón, J.M. Electrochemical (bio)sensing of clinical markers using quantum dots. Electroanalysis 2017, 29, 24–37. [Google Scholar] [CrossRef]
- Devi, N.R.; Vignesh Kumar, T.H.; Sundramoorthy, A.K. Electrochemically exfoliated carbon quantum dots modified electrodes for detection of dopamine neurotransmitter. J. Electrochem. Soc. 2018, 165, G3112–G3119. [Google Scholar] [CrossRef]
- Mehta, J.; Bhardwaj, N.; Bhardwaj, S.K.; Tuteja, S.K.; Vinayak, P.; Paul, A.K.; Kim, K.-H.; Deep, A. Graphene quantum dot modified screen printed immunosensor for the determination of parathion. Anal. Biochem. 2017, 523, 1–9. [Google Scholar] [CrossRef]
- Bhatnagar, D.; Kaur, I.; Kumar, A. Ultrasensitive cardiac troponin I antibody based nanohybrid sensor for rapid detection of human heart attack. Int. J. Biol. Macromol. 2017, 95, 505–510. [Google Scholar] [CrossRef]
- Chen, F.; Gao, W.; Qiu, X.; Zhang, H.; Liu, L.; Liao, P.; Fu, W.; Luo, Y. Graphene quantum dots in biomedical applications: Recent advances and future challenges. Front. Lab. Med. 2017, 1, 192–199. [Google Scholar] [CrossRef]
- Baluta, S.; Lesiak, A.; Cabaj, J. Graphene quantum dots-based electrochemical biosensor for catecholamine neurotransmitters detection. Electroanalysis 2018, 30, 1781–1790. [Google Scholar] [CrossRef]
- Faridbod, F.; Sanati, A.L. Graphene quantum dots in electrochemical sensors/biosensors. Curr. Anal. Chem. 2019, 15, 103–123. [Google Scholar] [CrossRef]
- Hashemzadeh, N.; Hasanzadeh, M.; Shadjou, N.; Eivazi-Ziaei, J.; Khoubnasabjafari, M.; Jouyban, A. Graphene quantum dot modified glassy carbon electrode for the determination of doxorubicin hydrochloride in human plasma. J. Pharm. Anal. 2016, 6, 235–241. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.V.; Richter, L.; Moulick, A.; Xhaxhiu, K.; Kudr, J.; Cernei, N.; Polansk, H.; Heger, Z.; Masarik, M.; Kopel, P.; Stiborov, M.; Eckschlager, T.; Adam, V.; Kizek, R. Electrochemical sensing of etoposide using carbon quantum dot modified glassy carbon electrode. Analyst 2016, 141, 2665–2675. [Google Scholar] [CrossRef]
- Wang, L.; Tricard, S.; Yue, P.; Zhao, J.; Fang, J.; Shen, W. Polypyrrole and graphene quantum dots @ Prussian Blue hybrid film on graphite felt electrodes: Application for amperometric determination of L-cysteine. Biosens. Bioelectron. 2016, 77, 1112–1118. [Google Scholar] [CrossRef] [PubMed]
- Jian, X.; Liu, X.; Yang, H.; Guo, M.; Song, X.; Dai, H.; Liang, Z. Graphene quantum dots modified glassy carbon electrode via electrostatic self-assembly strategy and its application. Electrochim. Acta 2016, 190, 455–462. [Google Scholar] [CrossRef]
- Tan, F.; Cong, L.; Li, X.; Zhao, Q.; Zhao, H.; Quan, X.; Chen, J. An electrochemical sensor based on molecularly imprinted polypyrrole/graphene quantum dots composite for detection of bisphenol A in water samples. Sens. Actuators B Chem. 2016, 233, 599–606. [Google Scholar] [CrossRef]
- Zhu, X.; Wu, G.; Lu, N.; Yuan, X.; Li, B. A miniaturized electrochemical toxicity biosensor based on graphene oxide quantum dots/carboxylated carbon nanotubes for assessment of priority pollutants. J. Hazard. Mater. 2017, 324, 272–280. [Google Scholar] [CrossRef]
- Lu, L.; Zhou, L.; Chen, J.; Yan, F.; Liu, J.; Dong, X.; Xi, F.; Chen, P. Nanochannel-confined graphene quantum dots for ultrasensitive electrochemical analysis of complex samples. ACS Nano 2018, 12, 12673–12681. [Google Scholar] [CrossRef] [PubMed]
- Punrat, E.; Maksuk, C.; Chuanuwatanakul, S.; Wonsawat, W.; Chailapakul, O. Polyaniline/graphene quantum dot-modified screen-printed carbon electrode for the rapid determination of Cr (VI) using stopped-flow analysis coupled with voltammetric technique. Talanta 2016, 150, 198–205. [Google Scholar] [CrossRef]
- Li, Y.; Jiang, Y.; Mo, T.; Zhou, H.; Li, Y.; Li, S. Highly selective dopamine sensor based on graphene quantum dots self-assembled monolayers modified electrode. J. Electroanal. Chem. 2016, 767, 84–90. [Google Scholar] [CrossRef]
- Aoun, S.B. Nanostructured carbon electrode modified with N-doped graphene quantum dots–chitosan nanocomposite: A sensitive electrochemical dopamine sensor. R. Soc. Open Sci. 2017, 4, 171199. [Google Scholar] [CrossRef]
- Zhang, Y.; Wu, C.; Zhou, X.; Wu, X.; Yang, Y.; Wu, H.; Guo, S.; Zhang, J. Graphene quantum dots/gold electrode and its application in living cell H2O2 detection. Nanoscale 2013, 5, 1816–1819. [Google Scholar] [CrossRef]
- Ju, J.; Chen, W. In situ growth of surfactant-free gold nanoparticles on nitrogen-doped graphene quantum dots for electrochemical detection of hydrogen peroxide in biological environments. Anal. Chem. 2015, 87, 1903–1910. [Google Scholar] [CrossRef]
- Xi, J.; Xie, C.; Zhang, Y.; Wang, L.; Xiao, J.; Duan, X.; Ren, J.; Xiao, F.; Wang, S. Pd nanoparticles decorated N-doped graphene quantum dots@N-doped carbon hollow nanospheres with high electrochemical sensing performance in cancer detection. ACS Appl. Mater. Interfaces 2016, 8, 22563–22573. [Google Scholar] [CrossRef] [PubMed]
- Mollarasouli, F.; Asadpour-Zeynali, K.; Campuzano, S.; Yáñez-Sedeño, P.; Pingarrón, J.M. Non-enzymatic hydrogen peroxide sensor based on graphene quantum dots-chitosan/methylene blue hybrid nanostructures. Electrochim. Acta 2017, 246, 303–314. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, W.; Zhang, L.; Li, J.; Su, Z.; Wei, G. Sequence-designed peptide nanofibers bridged conjugation of graphene quantum dots with graphene oxide for high performance electrochemical hydrogen peroxide biosensor. Adv. Mater. Interfaces 2017, 4, 1600895. [Google Scholar] [CrossRef]
- Hu, S.; Trinchi, A.; Atkin, P.; Cole, I. Tunable photoluminescence across the entire visible spectrum from carbon dots excited by white light. Angew. Chem. Int. Ed. 2015, 54, 2970–2974. [Google Scholar] [CrossRef] [PubMed]
- Atkin, P.; Daeneke, T.; Wang, Y.; Careya, B.J.; Berean, K.J.; Clark, R.M.; Ou, J.Z.; Trinchi, A.; Cole, I.S.; Kalantar-zadeh, K. 2D WS2/Carbon Dot Hybrids with Enhanced Photocatalytic Activity. J. Mater. Chem. A 2016, 4, 13563–13571. [Google Scholar] [CrossRef]
- Martínez-Periñán, E.; Bravo, I.; Rowley-Neale, S.J.; Lorenzo, E.; Banks, C.E. Carbon nanodots as electrocatalysts towards the oxygen reduction reaction. Electroanalysis 2018, 30, 436–444. [Google Scholar] [CrossRef]
- Li, H.; Chen, L.; Wu, H.; He, H.; Jin, Y. Ionic liquid-functionalized fluorescent carbon nanodots and their applications in electrocatalysis, biosensing, and cell imaging. Langmuir 2014, 30, 15016–15021. [Google Scholar] [CrossRef] [PubMed]
- Ji, H.; Zhou, F.; Gu, J.; Shu, C.; Xi, K.; Jia, X. Nitrogen-doped carbon dots as a new substrate for sensitive glucose determination. Sensors 2016, 16, 630. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, Z.; Rui, Y.; Li, M. Horseradish peroxidase immobilization on carbon nanodots/CoFe layered double hydroxides: Direct electrochemistryand hydrogen peroxide sensing. Biosens. Bioelectron. 2015, 64, 57–62. [Google Scholar] [CrossRef]
- Huang, Q.; Hu, S.; Zhang, H.; Chen, J.; He, Y.; Li, F.; Weng, W.; Ni, J.; Bao, X.; Lin, Y. Carbon dots and chitosan composite film based biosensor for the sensitive and selective determination of dopamine. Analyst 2013, 138, 5417–5423. [Google Scholar] [CrossRef]
- Zhang, L.; Han, Y.; Zhu, J.; Zhai, Y.; Dong, S. Simple and sensitive fluorescent andelectrochemical trinitrotoluene sensors based on aqueous carbon dots. Anal. Chem. 2015, 87, 2033–2036. [Google Scholar] [CrossRef]
- Guo, W.; Pia, F.; Zhang, H.; Sun, J.; Zhang, Y.; Sun, X. A novel molecularly imprinted electrochemical sensor modified with carbon dots, chitosan, gold nanoparticles for the determination of patulin. Biosens. Bioelectron. 2017, 98, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Wu, H.; Jiang, Y.; Xu, J.; Li, X.; Zhang, W.; Qiu, F. A molecularly-imprinted-electrochemical-sensor modified with nanocarbon-dots with high sensitivity and selectivity for rapid determination of glucose. Anal. Biochem. 2018, 555, 42–49. [Google Scholar] [CrossRef]
- Xiang, Q.; Huang, J.; Huang, H.; Mao, W.; Ye, Z. A label-free electrochemical platform for the highly sensitive detection of hepatitis B virus DNA using graphene quantum dots. RSC Adv. 2018, 8, 1820–1825. [Google Scholar] [CrossRef]
- Tuteja, S.K.; Chen, R.; Kukkar, M.; Song, C.K.; Mutreja, R.; Singh, S.; Paul, A.K.; Lee, H.; Kim, K.-H.; Deep, A.; Suri, C.R. A label-free electrochemical immunosensor for the detection of cardiac marker using graphene quantum dots (GQDs). Biosens. Bioelectron. 2016, 86, 548–556. [Google Scholar] [CrossRef] [PubMed]
- Hu, T.; Zhang, L.; Wen, W.; Zhang, X.; Wang, S. Enzyme catalytic amplification of miRNA-155 detection with graphene quantum dot-based electrochemical biosensor. Biosens. Bioelectron. 2016, 77, 451–456. [Google Scholar] [CrossRef]
- Pedrero, M.; Campuzano, S.; Pingarrón, J.M. Quantum dots as components of electrochemical sensing platforms for the detection of environmental and food pollutants: A review. J. AOAC Int. 2017, 100, 950–961. [Google Scholar] [CrossRef]
- Mollarasouli, F.; Serafín, V.; Campuzano, S.; Yáñez-Sedeño, P.; Pingarrón, J.M.; Asadpour-Zeynali, K. Ultrasensitive determination of receptor tyrosine kinase with a label-free electrochemical immunosensor using graphene quantum dotsmodified screen-printed electrodes. Anal. Chim. Acta 2018, 1011, 28–34. [Google Scholar] [CrossRef]
- Wei, H.; Wang, E.K. Nanomaterials with enzyme-like characteristics (nanozymes): Next-generation artificial enzymes. Chem. Soc. Rev. 2013, 42, 6060–6093. [Google Scholar] [CrossRef]
- Liu, W.; Yang, H.; Ma, C.; Ding, Y.-N.; Ge, S.; Yu, J.; Yan, M. Graphene–palladium nanowires based electrochemical sensor using ZnFe2O4–graphene quantum dots as an effective peroxidase mimic. Anal. Chim. Acta 2014, 852, 181–188. [Google Scholar] [CrossRef]
- Lin, L.P.; Song, X.H.; Chen, Y.Y.; Rong, M.C.; Zhao, T.T.; Wang, Y.R.; Jiang, Y.Q.; Chen, X. Intrinsic peroxidase-like catalytic activity of nitrogen-doped graphene quantum dots and their application in the colorimetric detection of H2O2 and glucose. Anal. Chim. Acta 2015, 869, 89–95. [Google Scholar] [CrossRef]
- Serafín, V.; Valverde, A.; Martinez-Garcia, G.; Martinez-Perinan, E.; Comba, F.; Garranzo-Asensio, M.; Barderas, R.; Yañez-Sedeño, P.; Campuzano, S.; Pingarrón, J.M. Graphene quantum dots-functionalized multi-walled carbon nanotubes as nanocarriers in electrochemical immunosensing. Determination of IL-13 receptor α2 in colorectal cells and tumor tissues with different metastatic potential. Sens. Actuators B Chem. 2019, 284, 711–722. [Google Scholar] [CrossRef]
- Serafín, V.; Valverde, A.; Garranzo-Asensio, M.; Barderas, R.; Campuzano, S.; Yáñez-Sedeño, P.; Pingarrón, J.M. Simultaneous determination of the emerging metastatic biomarkers IL-13Rα2 and CDH-17 using amperometric immunosensors involving grafted screen-printed electrodes and quantum dots/carbon nanotubes as carrier tags for signal amplification. Microchim. Acta. in revision.
- Yang, Y.; Liu, Q.; Liu, Y.; Cui, J.; Liu, H.; Wang, P.; Li, Y.; Chen, L.; Zhao, Z.; Dong, Y. A novel label-free electrochemical immunosensor based on functionalized nitrogen-doped graphene quantum dots for carcinoembryonic antigen detection. Biosens. Bioelectron. 2017, 90, 31–38. [Google Scholar] [CrossRef]
- Dong, Y.; Shao, J.; Chen, C.; Li, H.; Wang, R.; Chi, Y.; Lin, X.; Chen, G. Blue luminescent graphene quantum dots and graphene oxide prepared by tuning the carbonization degree of citric acid. Carbon 2012, 50, 4738–4743. [Google Scholar] [CrossRef]
- Gupta, S.; Smith, T.; Banaszak, A.; Boeck, J. Graphene quantum dots electrochemistry and sensitive electrocatalytic glucose sensor development. Nanomaterials 2017, 7, 301. [Google Scholar] [CrossRef] [PubMed]
- Muthurasu, A.; Ganesh, V. Horseradish peroxidase enzyme immobilized graphene quantum dots as electrochemical biosensors. Appl. Biochem. Biotechnol. 2014, 174, 945–959. [Google Scholar] [CrossRef] [PubMed]
- Mohammad-Rezaei, R.; Razmi, H. Preparation and characterization of hemoglobin immobilized on graphene quantum dots-chitosan nanocomposite as a sensitive and stable hydrogen peroxide biosensor. Sens. Lett. 2016, 14, 685–691. [Google Scholar] [CrossRef]
- Vasilescu, I.; Eremia, S.A.V.; Kusko, M.; Radoi, A.; Vasile, E.; Radu, G.L. Molybdenum disulphide and graphene quantum dots as electrode modifiers for laccase biosensor. Biosens. Bioelectron. 2016, 75, 232–237. [Google Scholar] [CrossRef] [PubMed]
- Razmi, H.; Mohammad-Rezaei, R. Graphene quantum dots as a new substrate for immobilization and direct electrochemistry of glucose oxidase: Application to sensitive glucose determination. Biosens. Bioelectron. 2013, 41, 498–504. [Google Scholar] [CrossRef]
- Valipour, A.; Roushani, M. Using silver nanoparticle and thiol graphene quantum dots nanocomposite as a substratum to load antibody for detection of hepatitis C virus core antigen: Electrochemical oxidation of riboflavin was used as redox probe. Biosens. Bioelectron. 2017, 89, 946–951. [Google Scholar] [CrossRef] [PubMed]
- Tufa, L.T.; Oh, S.; Tran, V.T.; Kim, J.; Jeong, K.-J.; Park, T.J.; Kim, H.-J.; Lee, J. Electrochemical immunosensor using nanotriplex of graphene quantum dots, Fe3O4, and Ag nanoparticles for tuberculosis. Electrochim. Acta 2018, 290, 369–377. [Google Scholar] [CrossRef]
- Mars, A.; Hamami, M.; Bechnak, L.; Patra, D.; Raouafi, N. Curcumin-graphene quantum dots for dual mode sensing platform: Electrochemical and fluorescence detection of APOe4, responsible of Alzheimer’s disease. Anal. Chim. Acta 2018, 1036, 141–146. [Google Scholar] [CrossRef]
- Shahdost-fard, F.; Roushani, M. Designing an ultra-sensitive aptasensor based on an AgNPs/thiol-GQD nanocomposite for TNT detection at femtomolar levels using the electrochemical oxidation of Rutin as a redox probe. Biosens. Bioelectron. 2017, 87, 724–731. [Google Scholar] [CrossRef]


| Electrode | Bioassay format | Nanomaterial(Role) | Target | Technique | Linear range | LOD | Assay time | Sample | Reference |
|---|---|---|---|---|---|---|---|---|---|
| GCE | Direct immunosensor | AgNPs/thiol-GQDs (electrode modifiers) | HCV | DPV (riboflavin) | 0.05 pg mL−1–60 ng mL−1 | 3 fg mL−1 | 30 min + AgNPs (2 h) + Thiol-GQDs (54 h) + CAb/AgNPs/thiol-GQD/GCE (>3 h) | Spiked human serum | [56] |
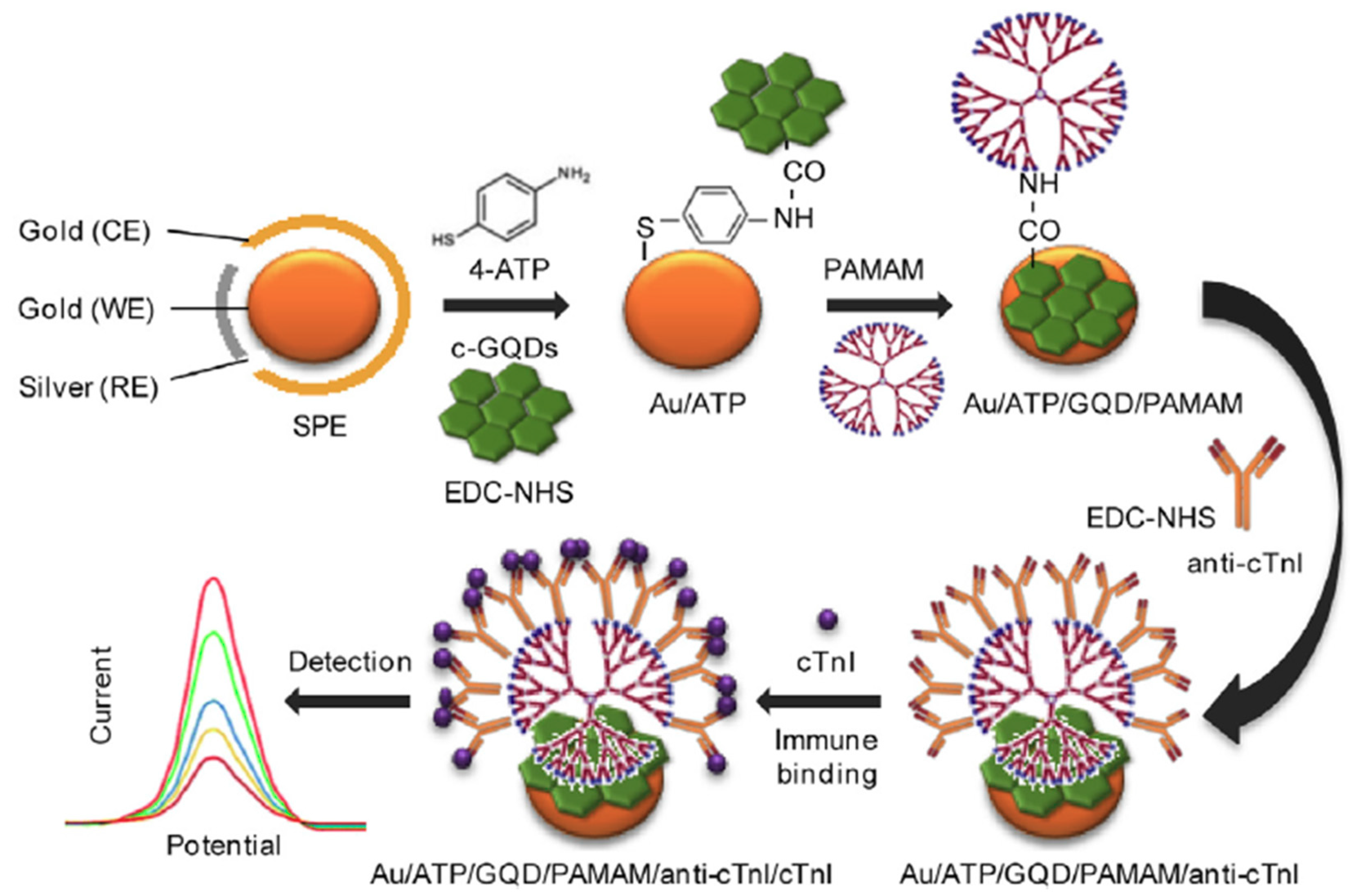
| Au-SPE | Direct immunosensor | PAMAM/GQDs (electrode modifiers) | cTnI | CV, DPV (Fe(CN)63-) | 1.0 10−6–1.0 × 10−3 | 20 fg mL−1 (CV) 25 fg mL−1 (DPV) | 10 min once prepared the Au/GQD/PAMAM/CAb (30 h) | Human blood serum | [10] |
| SPCE | Direct immunosensor | GQDs (electrode modifiers) | cMyo | DPV (Fe3+ group of the cMyo) | 0.01–100 ng mL−1 | 0.01 ng mL−1 | 10 min once prepared the CAb/GQDs/SPCE (1 h 10 min) | Spiked human serum | [40] |
| SPCE | Direct immunosensor | GQDs grafted with 2-ABA (electrode modifiers) | Parathion | EIS ([Fe(CN)6]3-/4-) | 0.01–106 ng L−1 | 46 pg L−1 | 5–30 min once prepared the CAb/GQD/SPCE (~3.5 h) | — | [9] |
| SPCE | Direct immunosensor | GQDs grafted with 2-ABA (electrode modifiers) | AXL | DPV ([Fe(CN)6]3-/4-) | 1.7−1000 pg mL−1 | 0.5 pg mL−1 | 90 min once prepared the CAb/GQD/SPCE (~4 h) | Human serum | [43] |
| GCE | Direct immunosensor | PtPd/N-GQDs@Au (electrode modifiers) | CEA | Amperometry (H2O2) | 5 fg mL−1–50 ng mL−1 | 2 fg mL−1 | 1 h + CAb/PtPd/N-GQDs@Au/GCE (2 h) + PtPd/N-GQDs@Au (~47 h) | Spiked human serum | [49] |
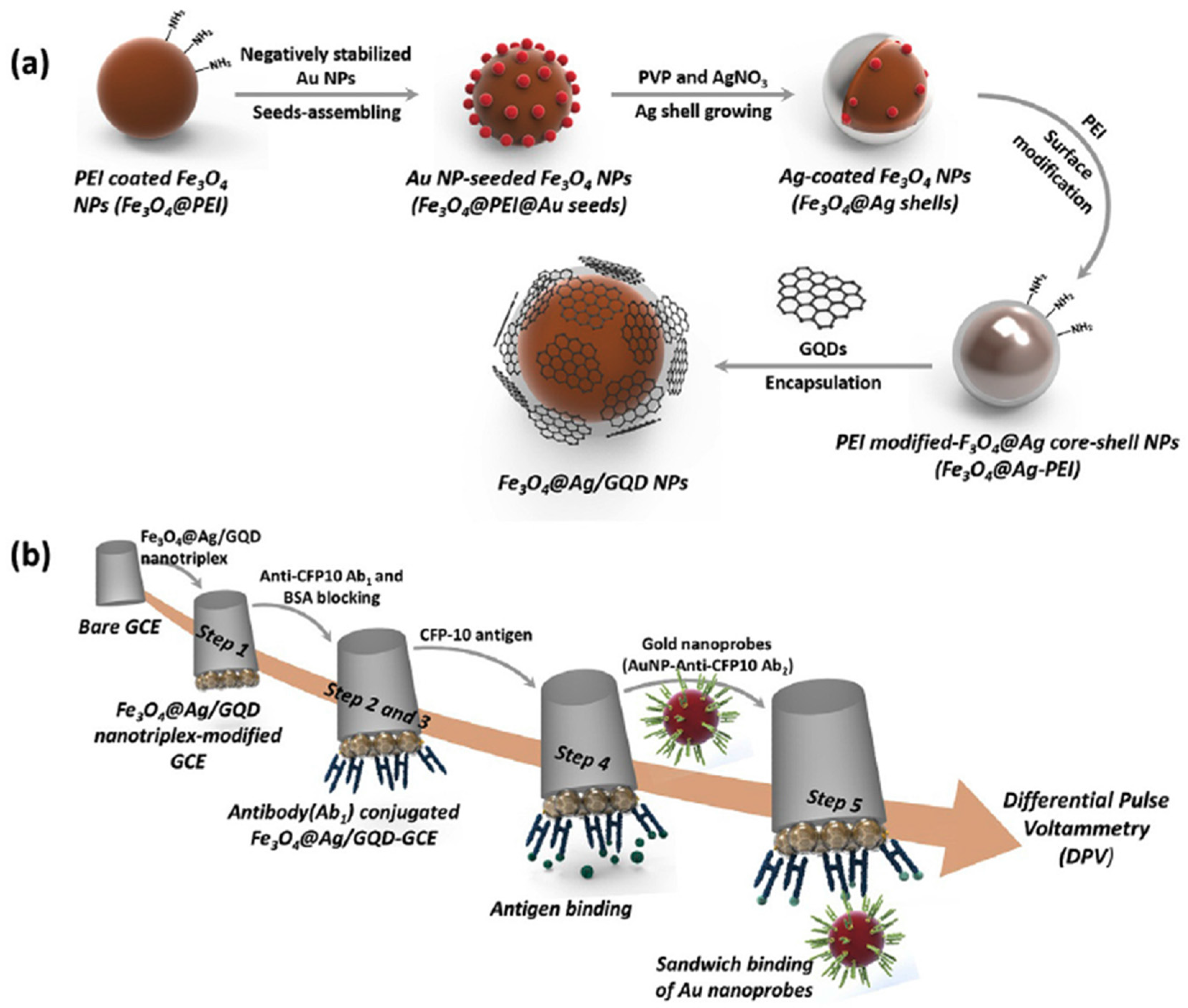
| GCE | Sandwich immunosensor | Fe3O4@Ag/GQDs (electrode modifiers) and DAb-AuNPs as labels | CFP-10 | DPV (Au3+) | 0.005–500 mg mL−1 | 0.33 ng mL−1 | 2 h once prepared the CAb/Fe3O4@Ag/GQD/GCE (~13 h) | Spiked human urine | [57] |
| SPCE | Sandwich immunosensor | MWCNTs/GQDs (nanocarriers of DAb+HRP) | IL-13sRα2 | Amperometry (H2O2/HQ) | 2.7–100 ng mL−1 | 0.8 ng mL−1 | 1 h 45 min + BCAb-Strep/p-ABA/SPCEs (3 h 15 min) + MWCNTs/GQDs-HRP-DAb (~30.5 h) | Raw cellular lysates and extracts of paraffin-embedded tissues from CRC patients | [47] |
| SPdCE | Sandwich immunosensor | MWCNTs/GQDs (nanocarriers of DAb+HRP) | 13sRα2 + CDH-17 | Amperometry (H2O2/HQ) | 4.92–100 ng mL−1 (IL-13sRα2) 0.11–10 ng mL−1 (CDH-17) | 1.44 ng mL−1 (IL-13sRα2) and 0.03 ng mL−1 (CDH-17) | 1 h 45 min + BCAb-Strep/p-ABA/SPCEs (3 h 15 min) + MWCNTs/GQDs-HRP-DAb (~30.5 h) | Raw cellular lysates and extracts of paraffin-embedded tissues from CRC patients | [48] |
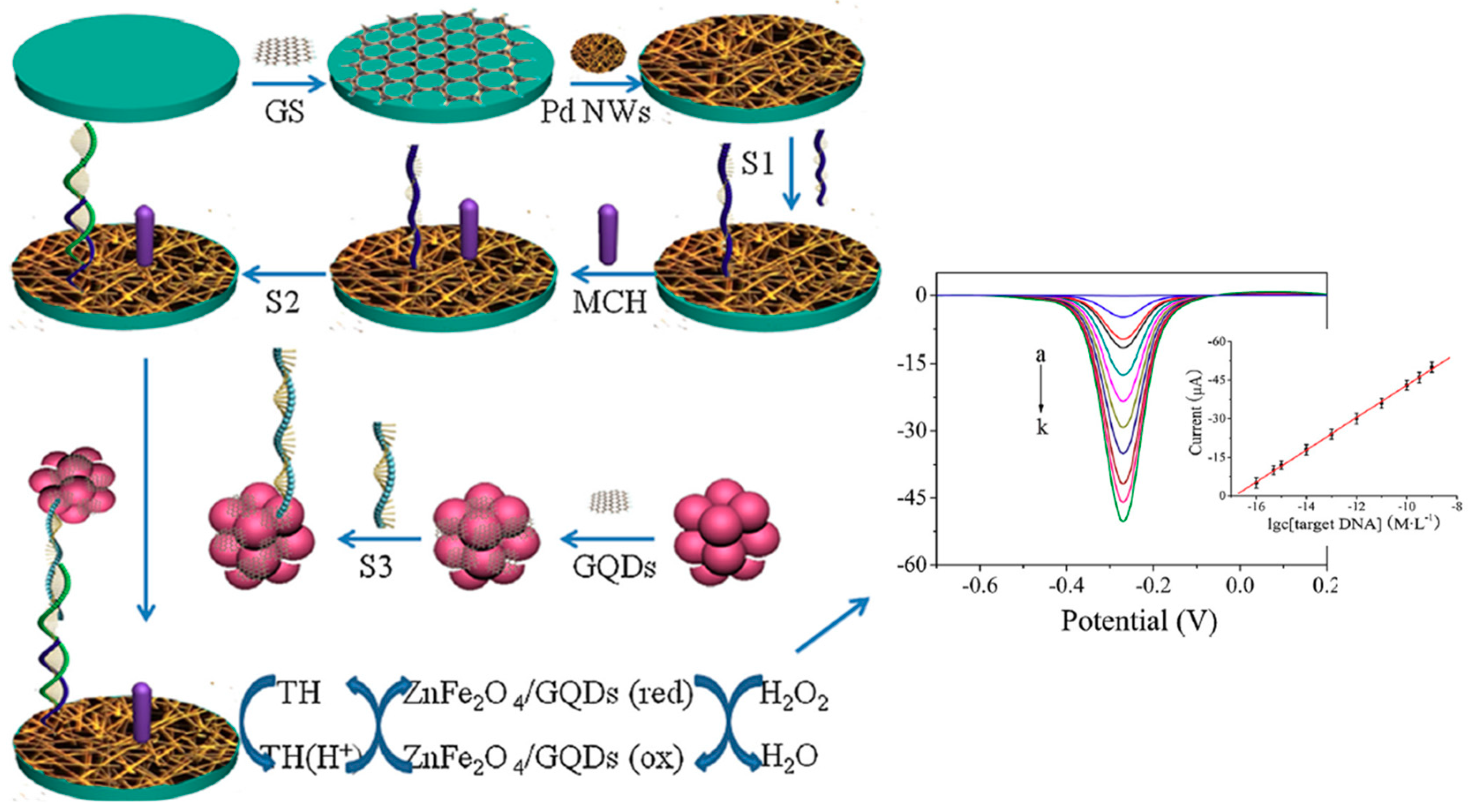
| GCE | Sandwich hybridization sensor | ZnFe2O4/GQDs (Trace label + nanocarrier of Dp) | Target DNA (S2) | DPV (H2O2/TH) | 10−16–5 × 10−9M | 6.2 × 10−17 M | 160 min + MCH/S1/Pd/GS/GCE (110 min) ZnFe2O4/GQDs-S3 (11 h) | Spiked human serum | [45] |
| Au disk electrode (2-mm φ) | Sandwich hybridization sensor | GQDs (platform for HRP immobilization) | miRNA-155 | Chronoamperometry (H2O2/TMB) | 1 fM–100 pM | 0.14 fM | 4 h once prepared the MCH/SHCp/Au (~13 h) | Spiked human serum (1/10 diluted) | [41] |
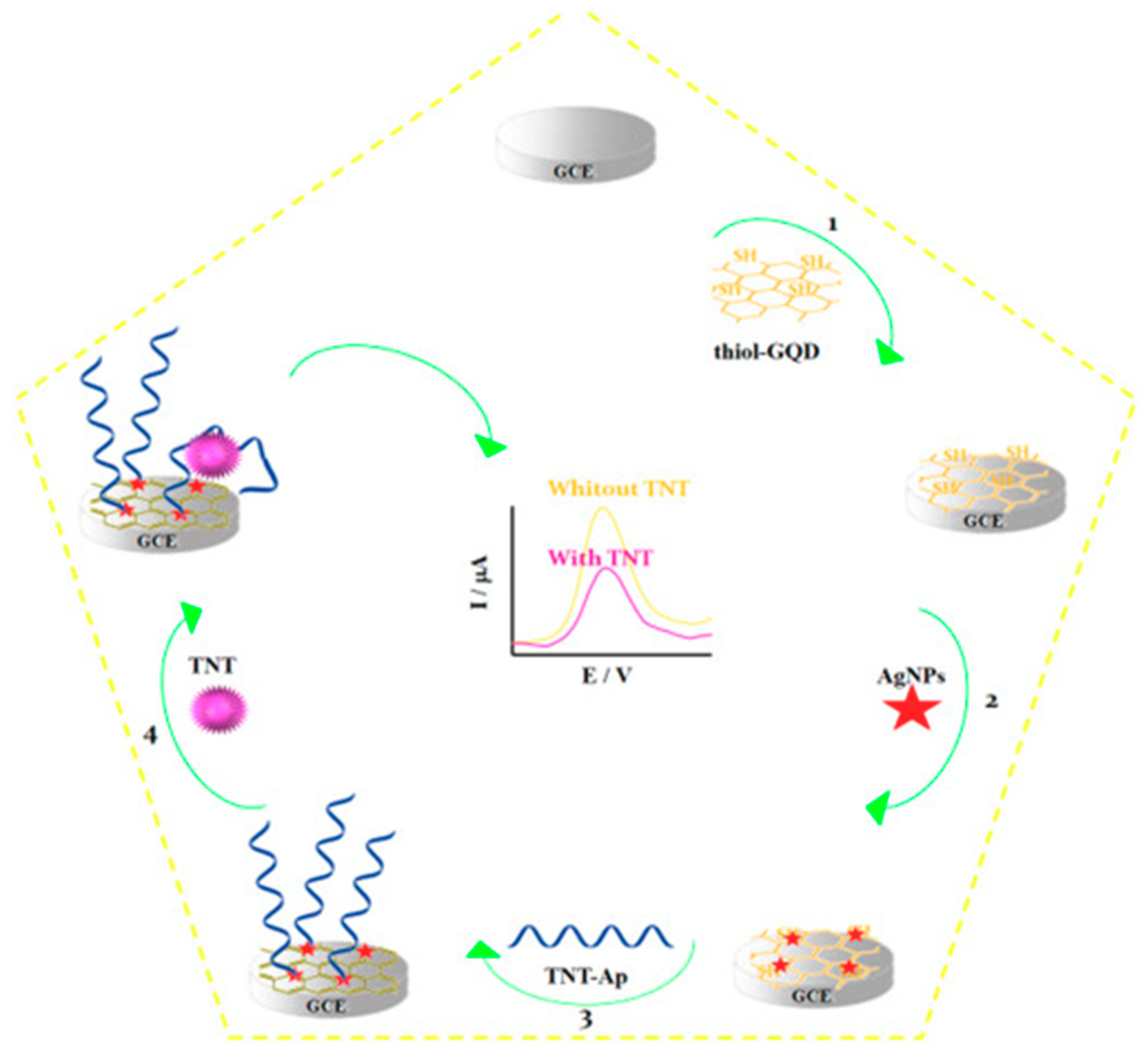
| GCE | Direct aptasensing strategy | AgNPs/thiol-GQDs (electrode modifiers) | TNT | DPV (RU) | 0.001–0.300 pM | 0.33 fM | 35 min + AgNPs (2 h) + Thiol-GQDs (54 h) + Ap/AgNPs/thiol-GQD/GCE (>16 h) | Spiked soil and water samples | [59] |
| Au-SPE | Direct hybridization | CDs (electrode modifiers) | Target DNAs | DPV (safranine) | 0.001–20 μM | 0.16 nM | 1 h + CDs (~3 days) + Cp/CDs/Au-SPEs (~2 days) | DNA isolated from peripheral blood leukocytes from cystic fibrosis patients | [1] |
| GCE | Direct hybridization | GQDs (electrode modifiers) | Target HBV-DNA | DPV (K3[Fe(CN)6]) | 10–500 nM | 1 nM | 30 min + Cp/GQDs/GCE (12.5 h) + GQDs (50 min) | — | [39] |
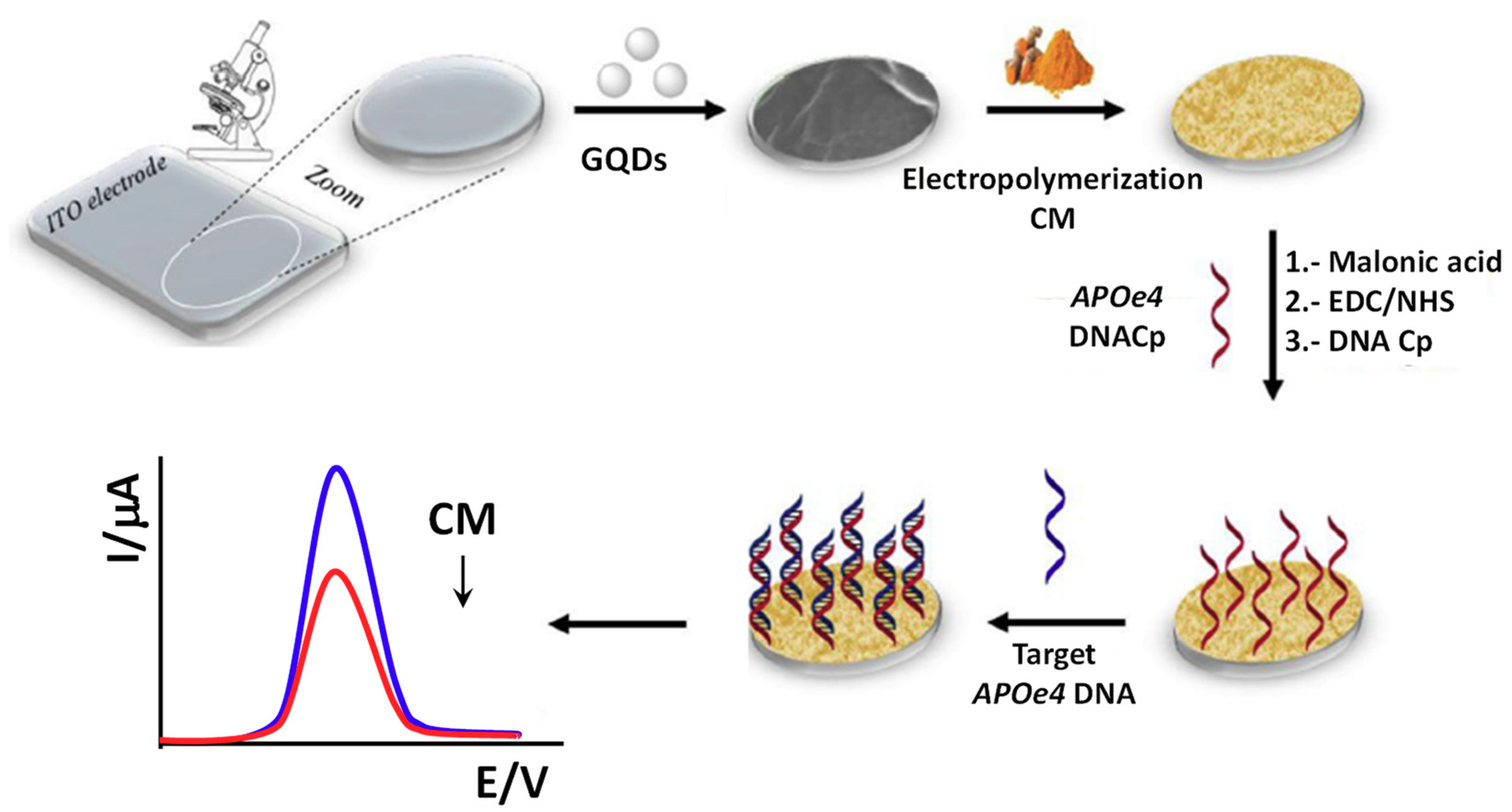
| ITO | Direct hybridization | CM/GQDs (electrode modifiers) | APOe4 DNA | DPV (CM) | 20–400 pg mL−1 | 0.48 pg mL−1 | — + Cp/CM/GQDs/ITO (>30 min) + GQDs (20 min) | 100-fold diluted human blood plasma pre-treated with ammonium sulfate | [58] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Campuzano, S.; Yáñez-Sedeño, P.; Pingarrón, J.M. Carbon Dots and Graphene Quantum Dots in Electrochemical Biosensing. Nanomaterials 2019, 9, 634. https://doi.org/10.3390/nano9040634
Campuzano S, Yáñez-Sedeño P, Pingarrón JM. Carbon Dots and Graphene Quantum Dots in Electrochemical Biosensing. Nanomaterials. 2019; 9(4):634. https://doi.org/10.3390/nano9040634
Chicago/Turabian StyleCampuzano, Susana, Paloma Yáñez-Sedeño, and José M. Pingarrón. 2019. "Carbon Dots and Graphene Quantum Dots in Electrochemical Biosensing" Nanomaterials 9, no. 4: 634. https://doi.org/10.3390/nano9040634
APA StyleCampuzano, S., Yáñez-Sedeño, P., & Pingarrón, J. M. (2019). Carbon Dots and Graphene Quantum Dots in Electrochemical Biosensing. Nanomaterials, 9(4), 634. https://doi.org/10.3390/nano9040634






