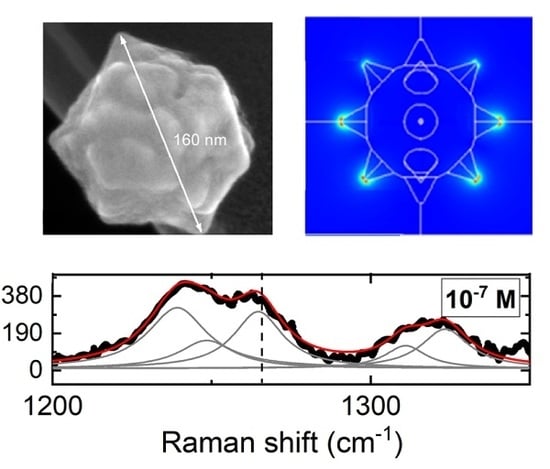
Detection of Histamine Dihydrochloride at Low Concentrations Using Raman Spectroscopy Enhanced by Gold Nanostars Colloids
Abstract
:1. Introduction
2. Methods
2.1. Synthesis of Nanoparticles
2.2. Numerical Simulation
2.3. Spectroscopic Measurements
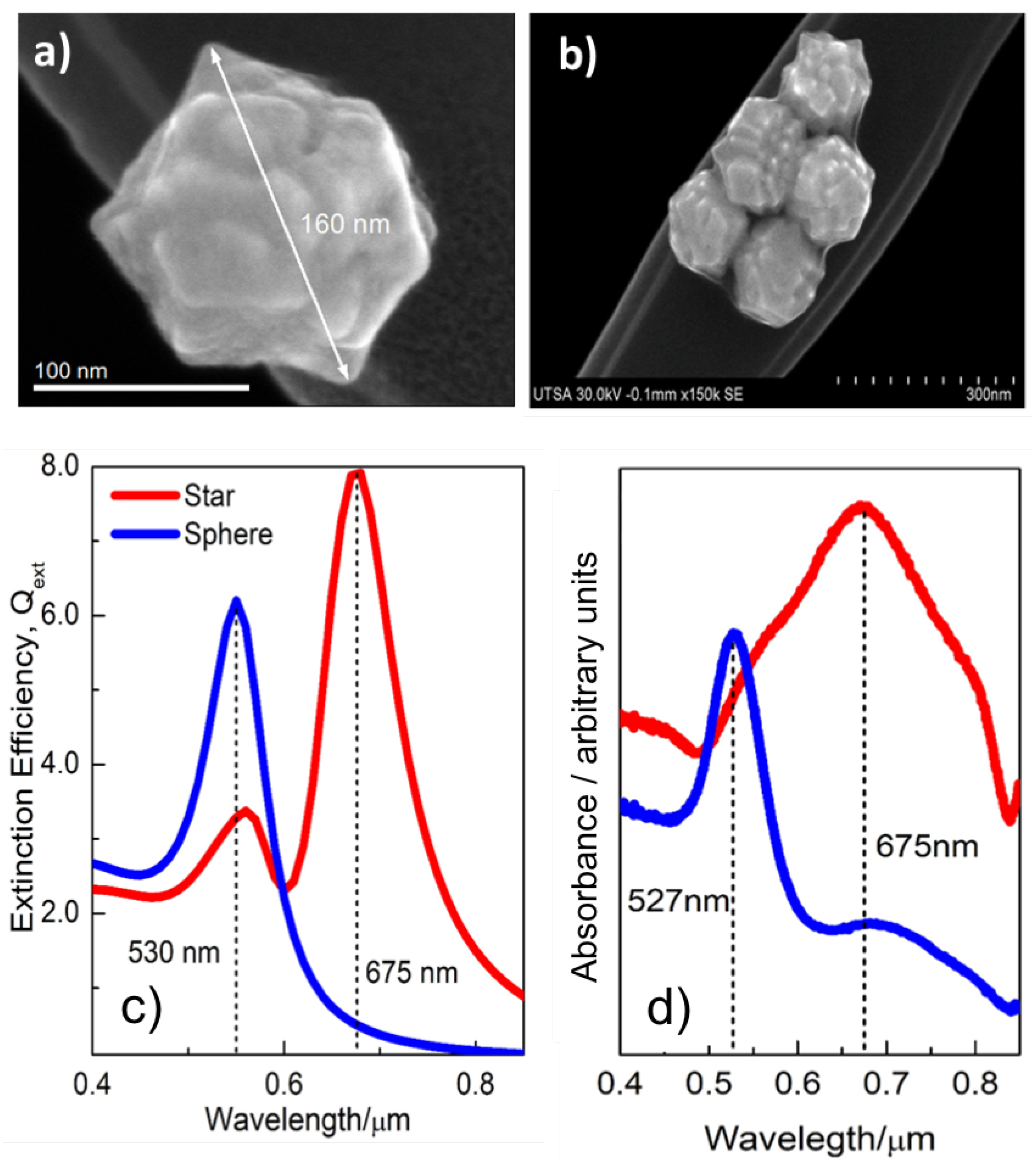
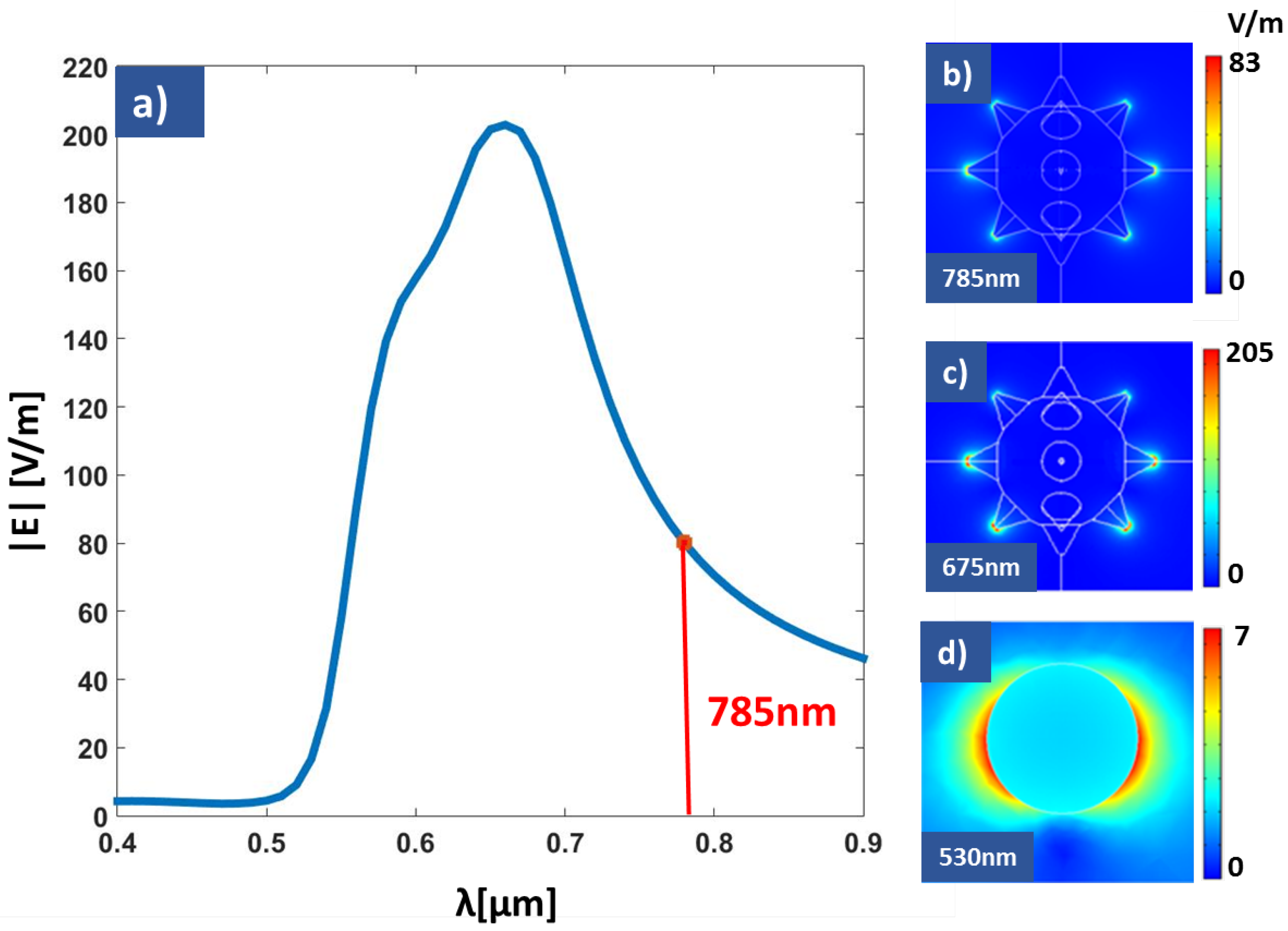
3. Results
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sharma, B.; Frontiera, R.R.; Henry, A.I.; Ringe, E.; Duyne, R.P.V. SERS: Materials, applications, and the future. Mater. Today 2012, 15, 16–25. [Google Scholar] [CrossRef]
- Kneipp, K.; Wang, Y.; Kneipp, H.; Perelman, L.T.; Itzkan, I.; Dasari, R.R.; Feld, M.S. Single Molecule Detection Using Surface-Enhanced Raman Scattering (SERS). Phys. Rev. Lett. 1997, 78, 1667–1670. [Google Scholar] [CrossRef]
- Willets, K.A.; Van Duyne, R.P. Localized Surface Plasmon Resonance Spectroscopy and Sensing. Annu. Rev. Phys. Chem. 2007, 58, 267–297. [Google Scholar] [CrossRef] [PubMed]
- Kneipp, K.; Kneipp, H.; Itzkan, I.; Dasari, R.R.; Feld, M.S. Ultrasensitive Chemical Analysis by Raman Spectroscopy. Chem. Rev. 1999, 99, 2957–2976. [Google Scholar] [CrossRef] [PubMed]
- Stiles, P.L.; Dieringer, J.A.; Shah, N.C.; Van Duyne, R.P. Surface-Enhanced Raman Spectroscopy. Ann. Rev. Anal. Chem. 2008, 1, 601–626. [Google Scholar] [CrossRef] [PubMed]
- Jeanmaire, D.L.; Duyne, R.P.V. Surface raman spectroelectrochemistry: Part I. Heterocyclic, aromatic, and aliphatic amines adsorbed on the anodized silver electrode. J. Electroanal. Chem. Interfacial Electrochem. 1977, 84, 1–20. [Google Scholar] [CrossRef]
- Lazcka, O.; Campo, F.J.D.; Munoz, F.X. Pathogen detection: A perspective of traditional methods and biosensors. Biosens. Bioelectron. 2007, 22, 1205–1217. [Google Scholar] [CrossRef] [PubMed]
- Velusamy, V.; Arshak, K.; Korostynska, O.; Oliwa, K.; Adley, C. An overview of foodborne pathogen detection: In the perspective of biosensors. Biotechnol. Adv. 2010, 28, 232–254. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.; Cheon, D.; Liu, F.; Lee, K.; Seo, T. A Graphene Oxide Based Immuno-biosensor for Pathogen Detection. Angew. Chem. 2010, 122, 5844–5847. [Google Scholar] [CrossRef]
- Sekhon, S.S.; Kim, S.G.; Lee, S.H.; Jang, A.; Min, J.; Ahn, J.Y.; Kim, Y.-H. Advances in pathogen-associated molecules detection using Aptamer based biosensors. Mol. Cell. Toxicol. 2013, 9, 311–317. [Google Scholar] [CrossRef]
- Izquierdo-Lorenzo, I.; Alda, I.; Sanchez-Cortes, S.; Garcia-Ramos, J.V. Adsorption and Detection of Sport Doping Drugs on Metallic Plasmonic Nanoparticles of Different Morphology. Langmuir 2012, 28, 8891–8901. [Google Scholar] [CrossRef]
- Otsuka, H.; Nagasaki, Y.; Kataoka, K. PEGylated nanoparticles for biological and pharmaceutical applications. Adv. Drug Deliv. Rev. 2012, 64, 246–255. [Google Scholar] [CrossRef]
- Grabar, K.C.; Freeman, R.G.; Hommer, M.B.; Natan, M.J. Preparation and Characterization of Au Colloid Monolayers. Anal. Chem. 1995, 67, 735–743. [Google Scholar] [CrossRef]
- Bozzini, B.; D’Urzo, L.; Lacitignola, D.; Mele, C.; Sgura, I.; Tondo, E. Investigation into dynamics of Au electrodeposition based on analysis of SERS spectral time series. Trans. IMF 2009, 87, 193–200. [Google Scholar] [CrossRef]
- Bozzini, B.; Gaudenzi, G.P.D.; Mele, C. A SERS investigation of the electrodeposition of Ag-Au alloys from free-cyanide solutions—Part II. J. Electroanal. Chem. 2004, 570, 29–34. [Google Scholar] [CrossRef]
- El-Zahry, M.R.; Mahmoud, A.; Refaat, I.H.; Mohamed, H.A.; Bohlmann, H.; Lendl, B. Antibacterial effect of various shapes of silver nanoparticles monitored by SERS. Talanta 2015, 138, 183–189. [Google Scholar] [CrossRef] [PubMed]
- García-Cámara, B.; Algorri, J.F.; Cuadrado, A.; Urruchi, V.; Sánchez-Pena, J.M.; Serna, R.; Vergaz, R. All-Optical Nanometric Switch Based on the Directional Scattering of Semiconductor Nanoparticles. J. Phys. Chem. C 2015, 119, 19558–19564. [Google Scholar] [CrossRef]
- Niu, W.; Chua, Y.A.A.; Zhang, W.; Huang, H.; Lu, X. Highly Symmetric Gold Nanostars: Crystallographic Control and Surface-Enhanced Raman Scattering Property. J. Am. Chem. Soc. 2015, 137, 10460–10463. [Google Scholar] [CrossRef]
- Dacarro, G.; Pallavicini, P.; Bertani, S.M.; Chirico, G.; D’Alfonso, L.; Falqui, A.; Marchesi, N.; Pascale, A.; Sironi, L.; Taglietti, A.; et al. Synthesis of reduced-size gold nanostars and internalization in SH-SY5Y cells. J. Colloid Interface Sci. 2017, 505, 1055–1064. [Google Scholar] [CrossRef]
- Navarro, J.R.G.; Manchon, D.; Lerouge, F.; Blanchard, N.P.; Marotte, S.; Leverrier, Y.; Marvel, J.; Chaput, F.; Micouin, G.; Gabudean, A.M.; et al. Synthesis of PEGylated gold nanostars and bipyramids for intracellular uptake. Nanotechnology 2012, 23, 465602. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.M.; Yu, J.; Chang, C.A.; Chiou, A.; Chiang, H.K.; Chuang, Y.C.; Wu, C.H.; Hsu, C.H.; Chen, P.A.; Huang, C.C. One-step shell polymerization of inorganic nanoparticles and their applications in SERS/nonlinear optical imaging, drug delivery, and catalysis. Sci. Rep. 2014, 4, 5593. [Google Scholar] [CrossRef]
- Huang, W.S.; Sun, I.W.; Huang, C.C. Promotion of SERS and catalytic activities with bimetallic and ternary concave nanolayers. J. Mater. Chem. A 2018, 6, 13041–13049. [Google Scholar] [CrossRef]
- Black, J.W.; Duncan, W.A.M.; Durant, C.J.; Ganellin, C.R.; Parsons, E.M. Definition and Antagonism of Histamine H2-receptors. Nature 1972, 236, 385–390. [Google Scholar] [CrossRef]
- Bodmer, S.; Imark, C.; Kneubühl, M. Biogenic amines in foods: Histamine and food processing. Inflamm. Res. 1999, 48, 296–300. [Google Scholar] [CrossRef]
- Maintz, L.; Novak, N. Histamine and histamine intolerance. Am. J. Clin. Nutr. 2007, 85, 1185–1196. [Google Scholar] [CrossRef]
- Rahmani, H.; Ingram, C. Histamine controls food intake in sheep via H1 receptors. Small Rumin. Res. 2007, 70, 110–115. [Google Scholar] [CrossRef]
- Gao, F.; Grant, E.; Lu, X. Determination of histamine in canned tuna by molecularly imprinted polymers-surface enhanced Raman spectroscopy. Anal. Chim. Acta 2015, 901, 68–75. [Google Scholar] [CrossRef]
- Stratton, J.E.; Hutkins, R.W.; Taylor, S.L. Biogenic Amines in Cheese and other Fermented Foods: A Review. J. Food Prot. 1991, 54, 460–470. [Google Scholar] [CrossRef]
- Kovacova-Hanuskova, E.; Buday, T.; Gavliakova, S.; Plevkova, J. Histamine, histamine intoxication and intolerance. Allergol. Immunopathol. 2015, 43, 498–506. [Google Scholar] [CrossRef]
- De Cerio, O.G.D.; Barrutia-Borque, A.; Gardeazabal-García, J. Scombroid Poisoning: A Practical Approach. Actas Dermo-Sifiliográficas (Engl. Ed.) 2016, 107, 567–571. [Google Scholar] [CrossRef]
- Harsing, L.G.; Nagashima, H.; Duncalf, D.; Vizi, E.S.; Goldiner, P.L. Determination of Histamine Concentrations in Plasma by Liquid Chromatography/electrochemistry. Clin. Chem. 1986, 32, 1823–1827. [Google Scholar]
- Cinquina, A.; Longo, F.; Cali, A.; Santis, L.D.; Baccelliere, R.; Cozzani, R. Validation and comparison of analytical methods for the determination of histamine in tuna fish samples. J. Chromatogr. A 2004, 1032, 79–85. [Google Scholar] [CrossRef]
- Janci, T.; Mikac, L.; Ivanda, M.; Marusic Radovcic, N.; Medic, H.; Vidacek, S. Optimization of parameters for histamine detection in fish muscle extracts by surface-enhanced Raman spectroscopy using silver colloid SERS substrates. J. Raman Spectrosc. 2016, 48, 64–72. [Google Scholar] [CrossRef]
- Janci, T.; Valinger, D.; Kljusuric, J.G.; Mikac, L.; Vidacek, S.; Ivanda, M. Determination of histamine in fish by Surface Enhanced Raman Spectroscopy using silver colloid SERS substrates. Food Chem. 2017, 224, 48–54. [Google Scholar] [CrossRef]
- Lai, C.H.; Wang, G.A.; Ling, T.K.; Wang, T.J.; Chiu, P.-K.; Chau, Y.F.C.; Huang, C.C.; Chiang, H.P. Near infrared surface-enhanced Raman scattering based on star-shaped gold/silver nanoparticles and hyperbolic metamaterial. Sci. Rep. 2017, 7, 5446. [Google Scholar] [CrossRef]
- Kimling, J.; Maier, M.; Okenve, B.; Kotaidis, V.; Ballot, H.; Plech, A. Turkevich Method for Gold Nanoparticle Synthesis Revisited. J. Phys. Chem. B 2006, 110, 15700–15707. [Google Scholar] [CrossRef]
- Childs, A.; Vinogradova, E.; Ruiz-Zepeda, F.; Velazquez-Salazar, J.J.; Jose-Yacaman, M. Biocompatible gold/silver nanostars for surface-enhanced Raman scattering. J. Raman Spectrosc. 2016, 47, 651–655. [Google Scholar] [CrossRef]
- Lin, W.C.; Tsai, T.R.; Huang, H.L.; Shiau, C.Y.; Chiang, H.P. SERS Study of Histamine by Using Silver Film over Nanosphere Structure. Plasmonics 2012, 7, 709–716. [Google Scholar] [CrossRef]
- Rodríguez-Lorenzo, L.; Álvarez Puebla, R.A.; de Abajo, F.J.G.; Liz-Marzán, L.M. Surface Enhanced Raman Scattering Using Star-Shaped Gold Colloidal Nanoparticles. J. Phys. Chem. C 2010, 114, 7336–7340. [Google Scholar] [CrossRef]
- Nalbant Esenturk, E.; Hight Walker, A.R. Surface-enhanced Raman scattering spectroscopy via gold nanostars. J. Raman Spectrosc. 2009, 40, 86–91. [Google Scholar] [CrossRef]
- Zhang, W.; Liu, J.; Niu, W.; Yan, H.; Lu, X.; Liu, B. Tip-Selective Growth of Silver on Gold Nanostars for Surface-Enhanced Raman Scattering. ACS Appl. Mater. Interfaces 2018, 10, 14850–14856. [Google Scholar] [CrossRef]
- Collado, J.A.; Ramírez, F.J. Vibrational spectra and assignments of histamine dication in the solid state and in solution. J. Raman Spectrosc. 2000, 31, 925–931. [Google Scholar] [CrossRef]
- Torreggiani, A.; Tamba, M.; Bonora, S.; Fini, G. Raman and IR study on copper binding of histamine. Biopolymers 2003, 72, 290–298. [Google Scholar] [CrossRef]
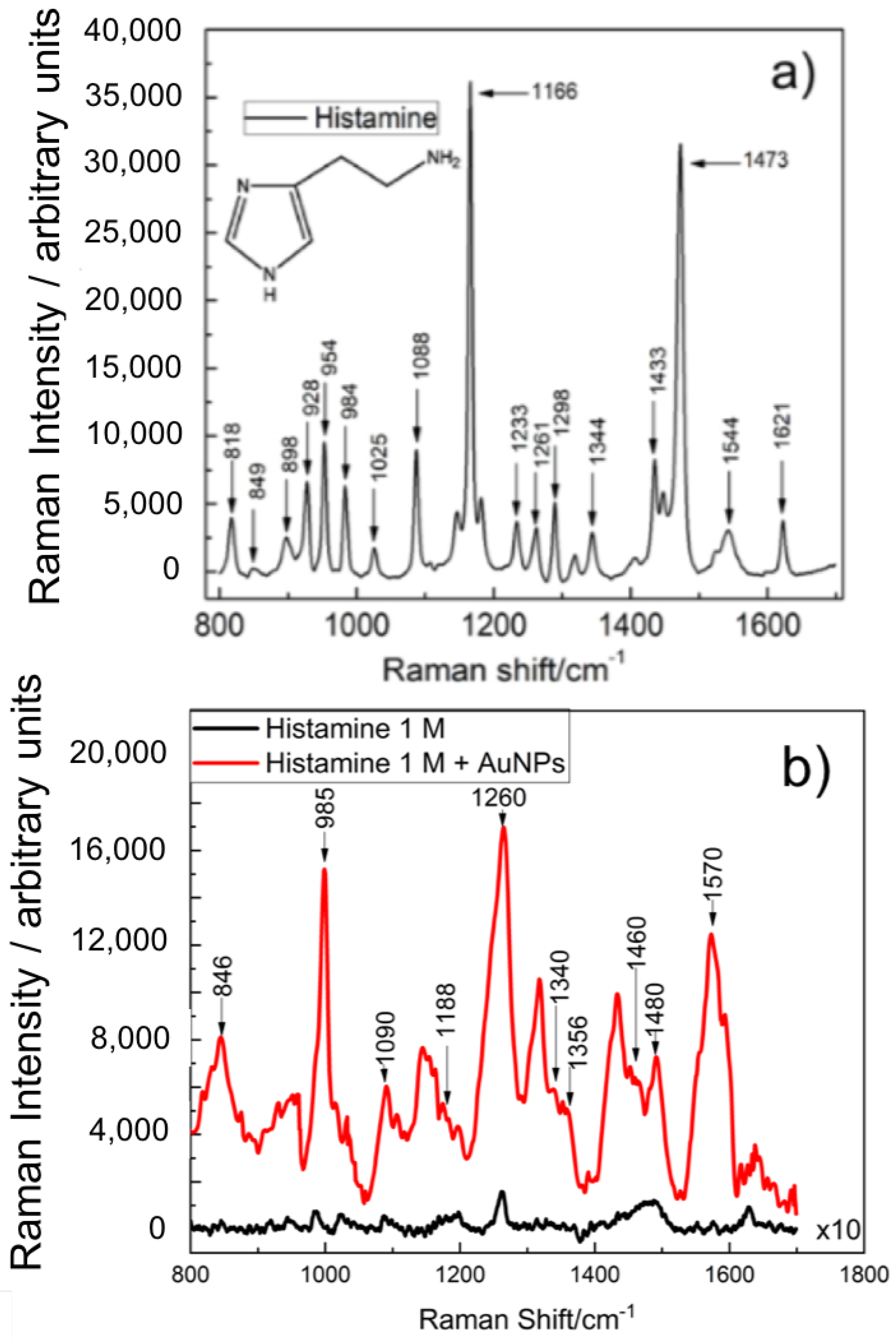
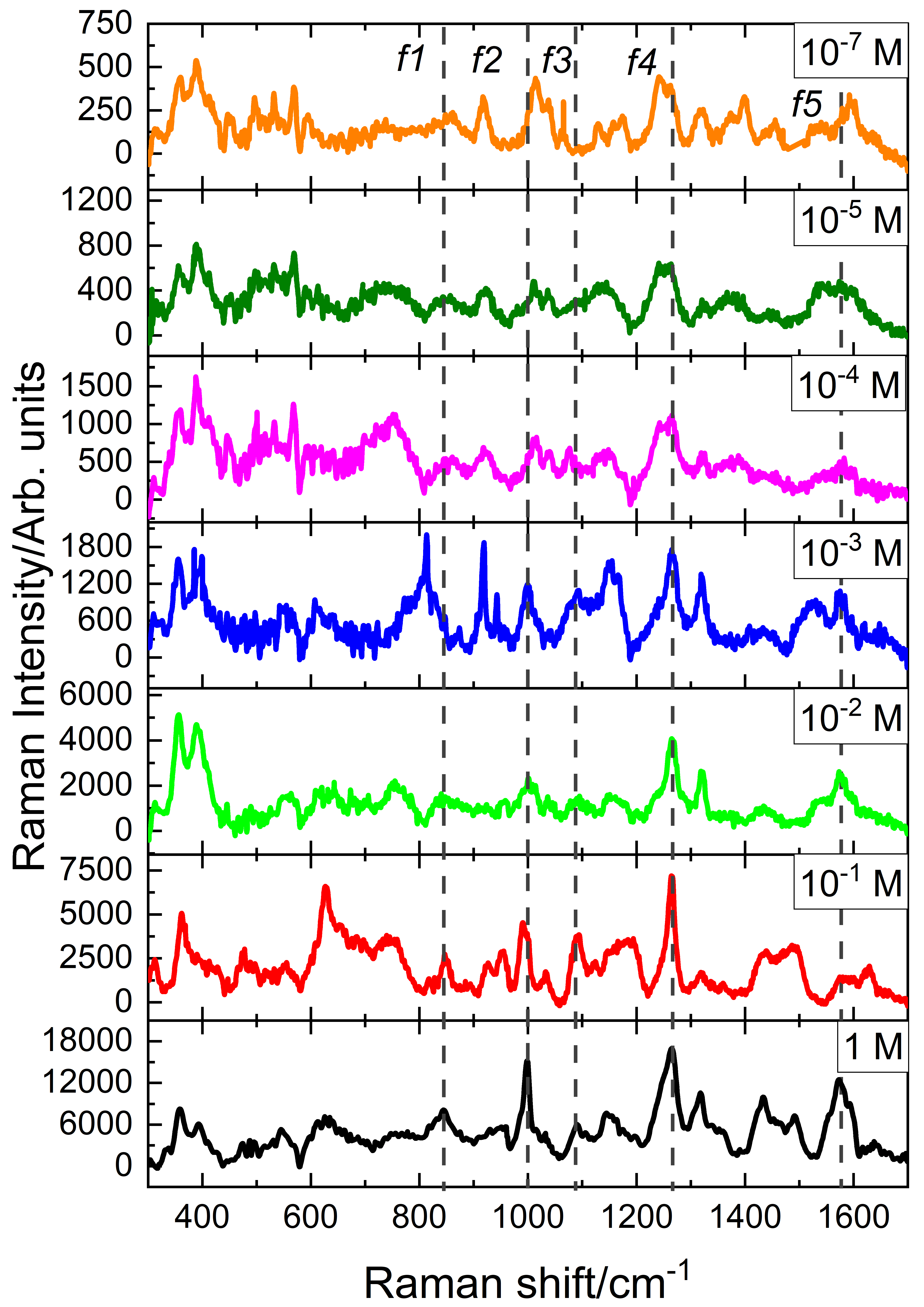
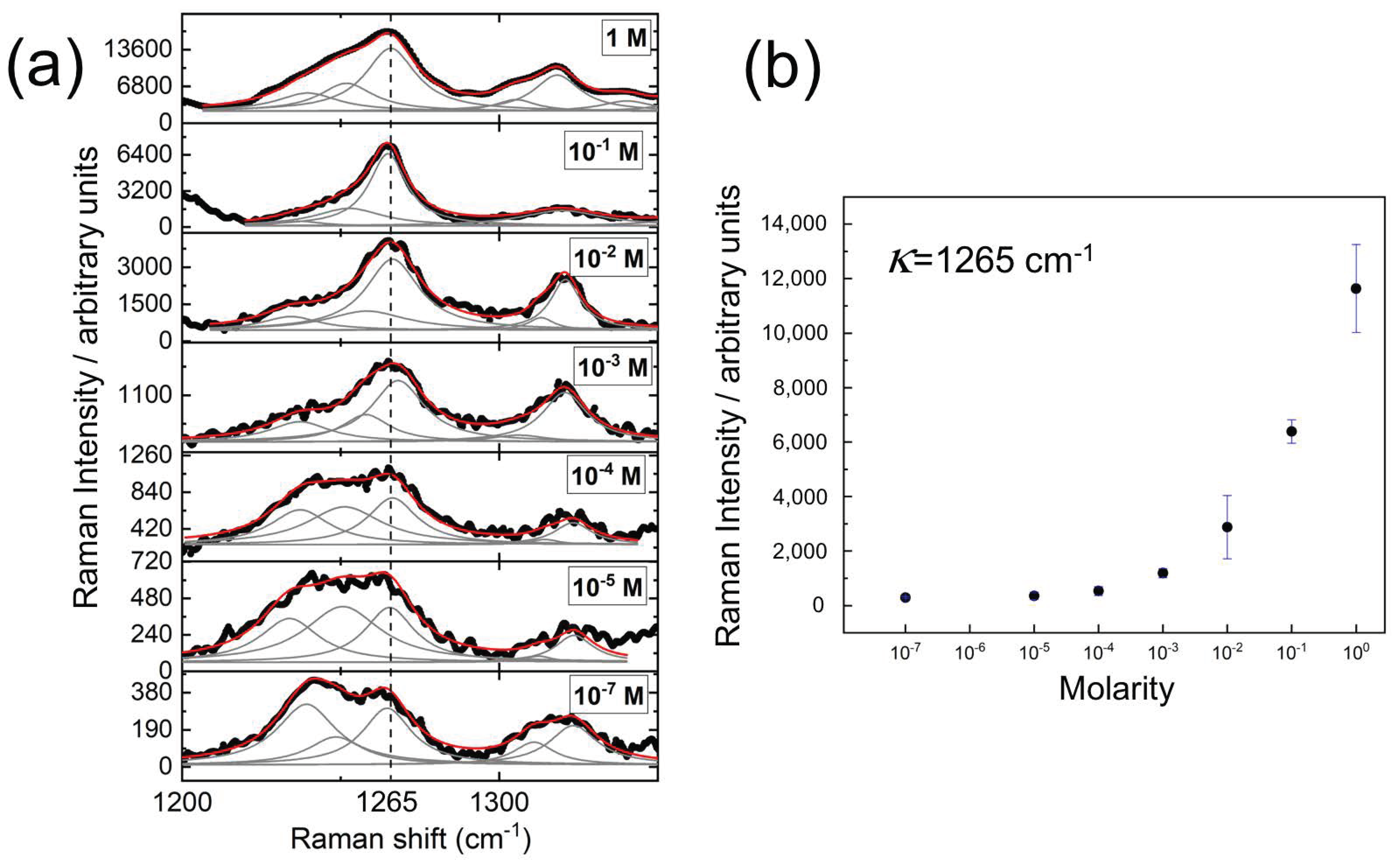
| (cm) | (cm) [38] | (cm) [43] | Assignment |
|---|---|---|---|
| 846 | ring A, wagging C | ||
| 895 | (N3-H) | ||
| 950 | ring A | ||
| 985 | 980 | (N1-H), (c2-H) | |
| 1028 | 1024.73 | 1005 | sy (C5-H), (C2-H), (C1-H) |
| 1090 | 1084.05 | 1088 | sy (N3-H), sy (C5-H) |
| 1188 | wagging C, (C2-H) | ||
| 1260 | 1270 | ring breathing | |
| 1340 | (C1-H), (N1-H) | ||
| 1356 | 1358.07 | translation C, (C1-H), (N1-H) | |
| 1438 | 1435 | ||
| 1460 | 1455.46 | (C4-H), sy (N3-H) | |
| 1480 | sy (C5-H), sy (N3-H) | ||
| 1570 | 1567.61 | 1579 | (N3-H) |
| 1629 | 1618 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kolosovas-Machuca, E.S.; Cuadrado, A.; Ojeda-Galván, H.J.; Ortiz-Dosal, L.C.; Hernández-Arteaga, A.C.; Rodríguez-Aranda, M.d.C.; Navarro-Contreras, H.R.; Alda, J.; González, F.J. Detection of Histamine Dihydrochloride at Low Concentrations Using Raman Spectroscopy Enhanced by Gold Nanostars Colloids. Nanomaterials 2019, 9, 211. https://doi.org/10.3390/nano9020211
Kolosovas-Machuca ES, Cuadrado A, Ojeda-Galván HJ, Ortiz-Dosal LC, Hernández-Arteaga AC, Rodríguez-Aranda MdC, Navarro-Contreras HR, Alda J, González FJ. Detection of Histamine Dihydrochloride at Low Concentrations Using Raman Spectroscopy Enhanced by Gold Nanostars Colloids. Nanomaterials. 2019; 9(2):211. https://doi.org/10.3390/nano9020211
Chicago/Turabian StyleKolosovas-Machuca, Eleazar Samuel, Alexander Cuadrado, Hiram Joazet Ojeda-Galván, Luis Carlos Ortiz-Dosal, Aida Catalina Hernández-Arteaga, Maria del Carmen Rodríguez-Aranda, Hugo Ricardo Navarro-Contreras, Javier Alda, and Francisco Javier González. 2019. "Detection of Histamine Dihydrochloride at Low Concentrations Using Raman Spectroscopy Enhanced by Gold Nanostars Colloids" Nanomaterials 9, no. 2: 211. https://doi.org/10.3390/nano9020211
APA StyleKolosovas-Machuca, E. S., Cuadrado, A., Ojeda-Galván, H. J., Ortiz-Dosal, L. C., Hernández-Arteaga, A. C., Rodríguez-Aranda, M. d. C., Navarro-Contreras, H. R., Alda, J., & González, F. J. (2019). Detection of Histamine Dihydrochloride at Low Concentrations Using Raman Spectroscopy Enhanced by Gold Nanostars Colloids. Nanomaterials, 9(2), 211. https://doi.org/10.3390/nano9020211