Critical Admission Temperature of H2 and CH4 in Nanopores of Exchanged ERI Zeolites
Abstract
1. Introduction
2. Materials and Methods
2.1. Characterization
2.2. Adsorption of H2 and CH4
3. Results
3.1. Characterization
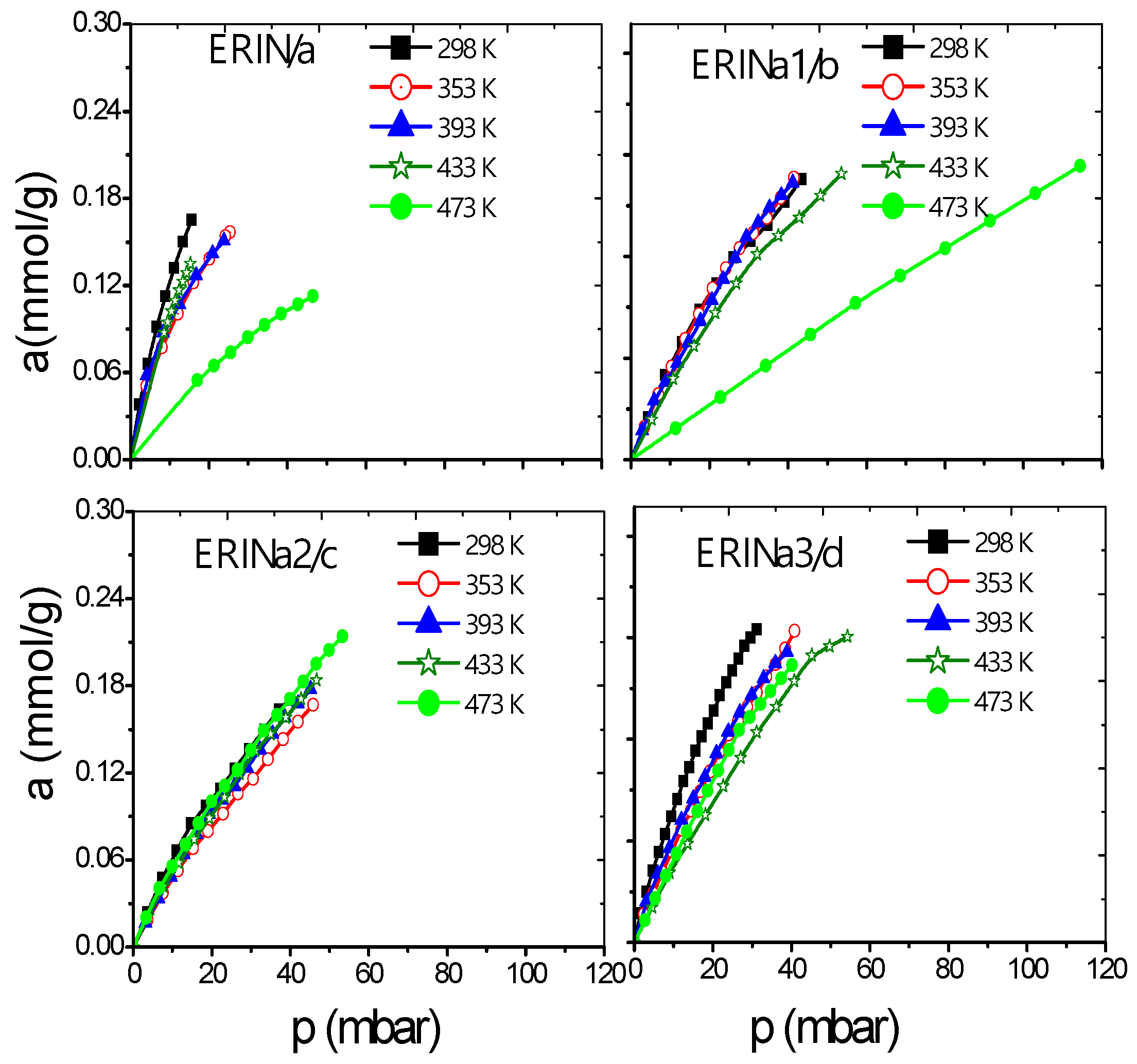
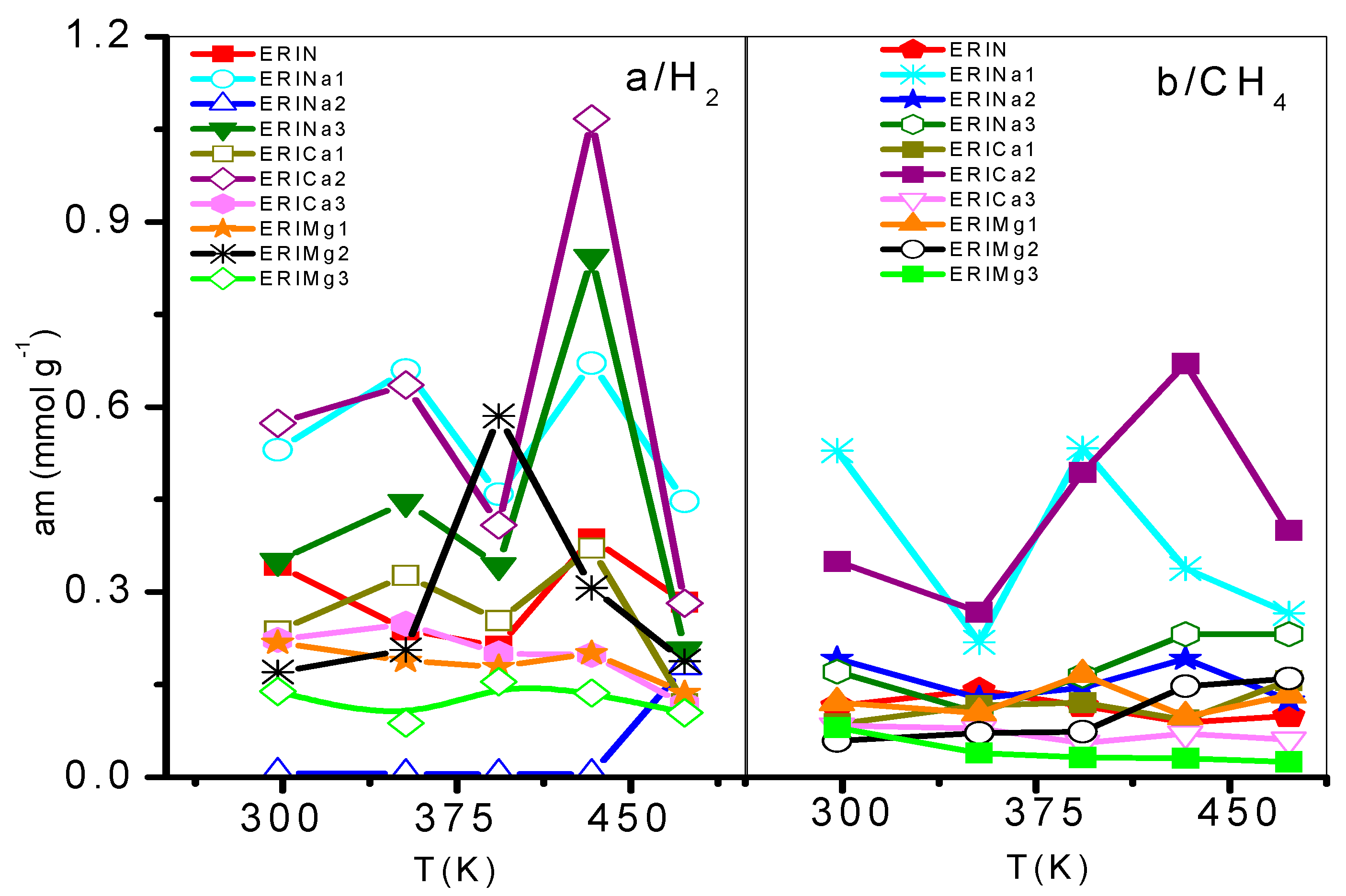
3.2. Adsorption Dependence with the Admission Temperature
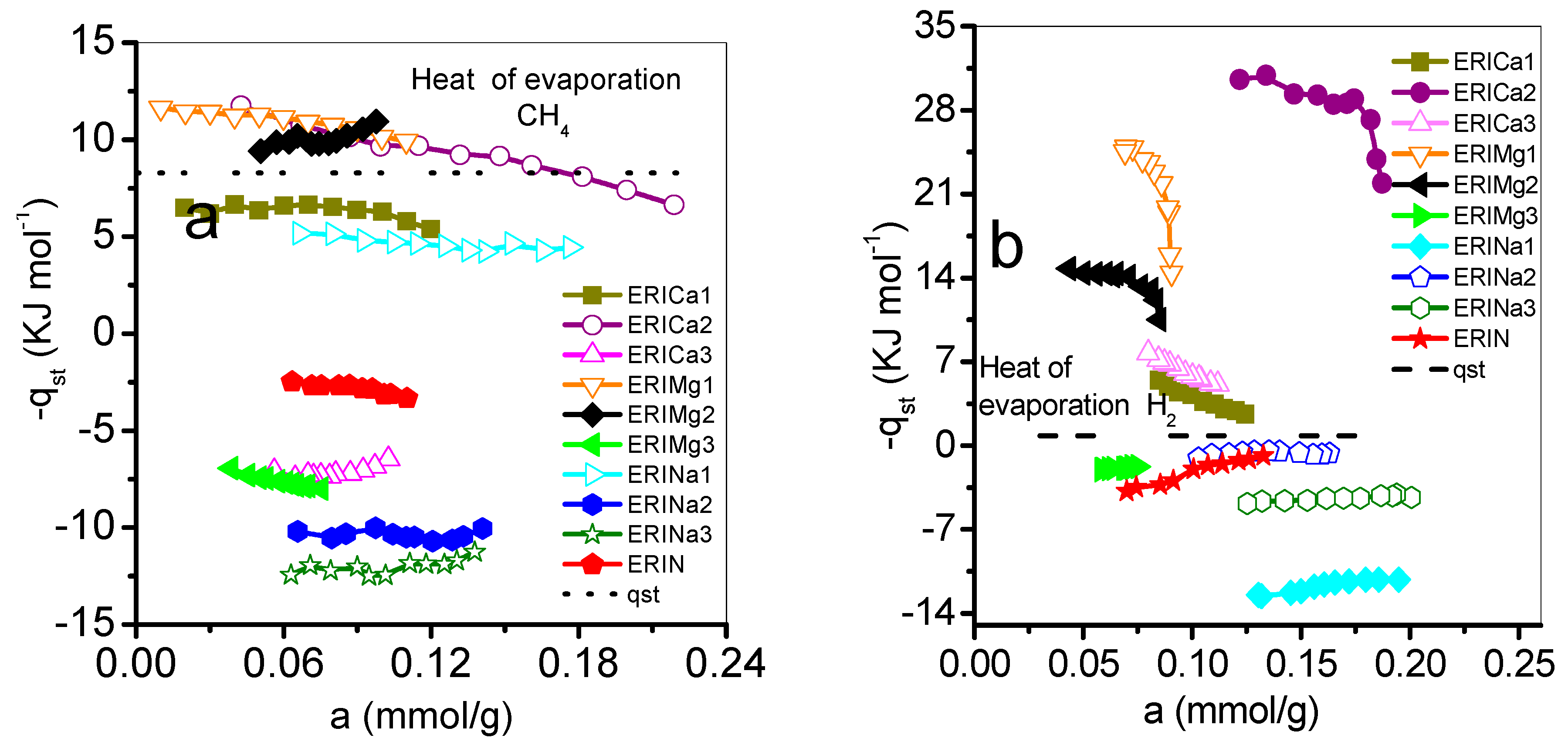
3.3. Isosteric Heat of Adsorption
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hernandez, M.A.; Pestryakov, A.; Portillo, R.; Salgado, A.; Rojas, F.; Rubio, E.; Ruiz, S.; Petranovskii, V. CO2 sequestration by natural zeolite for greenhouse effect control. Procedia Chem. 2015, 15, 33–41. [Google Scholar] [CrossRef]
- Bucior, B.J.; Chen, D.L.; Liu, J.; Jhonson, K. Porous carbon nanotube membranes for separation of H2/CH4 and CO2/CH4 mixtures. J. Phys. Chem. C 2012, 116, 25904–25910. [Google Scholar] [CrossRef]
- Atci, E.; Erucar, I.; Keskin, S. Adsorption and Transport of CH4, CO2, H2 Mixtures in a Bio-MOF Material from Molecular Simulations. J. Phys. Chem. C 2011, 115, 6833–6840. [Google Scholar] [CrossRef]
- Delgado, J.A.; Agueda, V.I.; Uguina, M.A.; Sotelo, J.L.; Brea, P.; Grande, C.A. Adsorption and diffusion of H2, CO, CH4 and CO2 in BPL activated carbon and zeolite 13X: Evaluation of performance in PSA hydrogen purification by simulation. Ind. Eng. Chem. Res. 2014, 53, 15414–15426. [Google Scholar] [CrossRef]
- Ackley, M.W.; Rege, S.U.; Saxena, H. Application of natural zeolites in the purification and separation of gases. Microporous Mesoporous Mater. 2003, 61, 25–42. [Google Scholar] [CrossRef]
- Farukawa, H.; Yaghi, O. Storage of Hydrogen, Methane and Carbon Dioxide in highly porous covalent organic frameworks for clean energy applications. J. Am. Chem. Soc. 2009, 131, 8875–8883. [Google Scholar] [CrossRef] [PubMed]
- Ma, S.; Zhou, H. A metal-organic framework with with entatic metal centers exhibiting high gas adsorption affinity. J. Am. Chem. Soc. 2006, 128, 11734–11735. [Google Scholar] [CrossRef]
- Shang, J.; Li, G.; Singh, R.; Xiao, P.; Liu, J.; Webley, P. Determination of composition range for “molecular trapdoor” effect in chabazite zeolite. J. Phys. Chem. 2013, 117, 12841–12847. [Google Scholar] [CrossRef]
- Moliner, M.; Martínez, C.; Corma, A. Synthesis strategies for preparing useful small pore zeolites and zeotypes for gas separations and catalysis. Chem. Mater. 2014, 26, 246–258. [Google Scholar] [CrossRef]
- Zito, P.F.; Caravella, A.; Brunetti, A.; Drioli, E.; Barbieri, G. Estimation of Langmuir and Sips models adsorption parameters for NaX and NaY FAU zeolites. J. Chem. Eng. Data 2015, 60, 2858–2868. [Google Scholar] [CrossRef]
- Fang, H.; Awati, R.; Boulfelfel, S.; Ravikovitch, P.; Sholl, D. First principles-derived force fields for CH4 adsorption and diffusion in siliceous zeolites. J. Phys. Chem. C 2018, 122, 12880–12891. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, D.; Yang, Q.; Zhong, C.; Mi, J. Comparative study of sepration performance of COFs and MOFs for CH4/CO2/H2 mixtures. J. Eng. Chem. Res. 2010, 49, 2902–2906. [Google Scholar] [CrossRef]
- Keskin, S. Adsorption, diffusion, and separation of CH4/H2 mixtures in covalent organic frameworks: Molecular somulations and theoretical predictions. J. Phys. Chem. C 2012, 116, 1772–1779. [Google Scholar] [CrossRef]
- Fan, H.; Mundstock, A.; Feldhoff, A.; Knebel, A.; Gu, J.; Meng, H.; Caro, J. Covalent organic framework-covalent organic framework bilayer membranes for highly selective gas separation. J. Am. Chem. Soc. 2018, 140, 10094–10098. [Google Scholar] [CrossRef]
- Fischer, M.; Bell, R.G. Influence of Zeolite Topology on CO2/N2 Separation Behavior: Force-Field Simulations Using a DFT-Derived Charge Model. J. Phys. Chem. C 2012, 116, 26449–26463. [Google Scholar] [CrossRef]
- Curtis, R.A.; Deem, M.W. A statistical mechanics study of ring size, ring shape, and the relation to pores found in zeolites. J. Phys. Chem. B 2003, 107, 8612–8620. [Google Scholar] [CrossRef]
- Breck, D.W. Zeolite Molecular Sieves: Structure, Chemistry and Use; John Wiley & Sons Inc.: New York, NY, USA, 1984; pp. 77–78. ISBN 0-471-09985-6. [Google Scholar]
- Ortega, K.; Hernandez, M.A.; Portillo, R.; Ayala, E.; Romero, O.; Rojas, F.; Rubio, E.; Pestryakov, A.; Petranovskii, V. Adsorption of Ar and N2 on dealuminated mordenite tuffs. Procedia Chem. 2015, 15, 65–71. [Google Scholar] [CrossRef]
- Yang, J.; Zhao, Q.; Xu, H.; Li, L.; Dong, J.; Li, J. Adsorption of CO2, CH4, and N2 on gas diameter grade ion-exchange small pore zeolites. J. Chem. Eng. Data 2012, 57, 3701–3709. [Google Scholar] [CrossRef]
- Liu, Z.; Ottaviani, M.F.; Abrams, L.; Lei, X.; Turro, N. Characterization of the External Surface of Silicalites Employing Electron Paramagnetic Resonance. J. Phys. Chem. A 2004, 108, 8040–8047. [Google Scholar] [CrossRef]
- Weber, G.; Simonot-Grange, M.H. Characterization of the dealumination effect into H faujasites by adsorption: Part 2. The hexane molecule as a pore volume probe. Zeolites 1994, 14, 433–438. [Google Scholar] [CrossRef]
- Armatas, G.S.; Petrakis, D.E.; Pomonis, P.J. A method of distinction between microporosity and mesoporosity using BET–Scatchard plots. Microporous Mesoporous Mater. 2005, 83, 251–261. [Google Scholar] [CrossRef]
- Zhu, H.Y.; Lu, G.Q. Estimating pore size distribution from the differential curves of comparison plots. Stud. Surf. Sci. Catal. 2000, 128, 248–250. [Google Scholar] [CrossRef]
- Hernandez, M.A.; Rojas, F. Adsorption Characteristics of Natural Erionite, Clinoptilolite and Mordenite Zeolites from Mexico. Adsorption 2000, 6, 33–45. [Google Scholar] [CrossRef]
- Ballirano, P.; Cametti, G. Crystal chemical and structural modificatons of erionite fibers leached with simulated lung fuids. Am. Mineral. 2015, 100, 1003–1012. [Google Scholar] [CrossRef]
- Hernández, M.A.; Rojas, F.; Portillo, R.; Salgado, M.; Rubio, E.; Ruiz, S. Creating nanoporosity in Na, Ca and Mg exchanged erionite zeolite. Int. J. Nanotechnol. 2016, 13, 28–43. [Google Scholar] [CrossRef]
- Reid, R.C.; Prausnitz, J.M.; Sherwood, T.K. The Properties of Gases and Liquids, 3rd ed.; McGraw-Hill: New York, NY, USA, 1977; pp. 629–635. [Google Scholar]
- Alberti, A.; Martucci, A.; Galli, E.; Vezzalini, G. A reexamination of the crystal structure of erionite. Zeolites 1997, 19, 349–352. [Google Scholar] [CrossRef]
- Tsitsihvili, G.V.; Andronikashvili, T.G.; Kirov, G.N.; Filizova, L.D. Natural Zeolites (Ellis Horwood Series in Inorganic Chemestry); Ellis Horwood: Chichester, UK, 1992; pp. 82–87. ISBN 13 978-0136120377. [Google Scholar]
- Choundary, V.R.; Mantry, K. Adsorption of aromatic hydrocarbons on highly siliceous MCM-41. Langmuir 2000, 16, 7031–7037. [Google Scholar] [CrossRef]
- Quiroz, K.; Hernández, M.A.; Rojas, F.; Portillo, R.; Rubio, E.; López, L. N2 and CO2 adsorption by soils with high kaolinite content from San Juan Amecac, Puebla, México. Minerals 2016, 6, 73. [Google Scholar] [CrossRef]
- Pacella, A.; Cremisini, C.; Nardi, E.; Montereali, M.R.; Pettiti, I.; Giordani, M.; Mattioli, M.; Ballirano, P. Different erionite species bind iron into the structure: A potential explanation for fibrous erionita toxicity. Minerals 2018, 8, 36. [Google Scholar] [CrossRef]
- Cametti, G.; Pacella, A.; Mura, F.; Rossi, M.; Ballirano, P. New morphological, chemical, and structural data of woolly erionite-Na from Durkee, Oregon, U.S.A. Am. Mineral. 2013, 98, 2155–2163. [Google Scholar] [CrossRef]
- Ballirano, P.; Andreozzi, G.; Dogan, M.; Dogan, U. Crystal structure and iron topochemistry of erionite-K from Rome, Oregon, U.S.A. Am. Mineral. 2009, 94, 1262–1270. [Google Scholar] [CrossRef]
- Gualtieri, A.; Gandolfi, N.; Pollastri, S.; Pollok, K.; Langenhorst, F. Where is iron in erionite? A multidisciplinary study on fibrous erionite-Na from Jersey (Nevada, USA). Sci. Rep. 2016, 6, 37981. [Google Scholar] [CrossRef] [PubMed]
- Pacella, A.; Fantauzzi, M.; Atzei, D.; Cremisine, C.; Nardi, E.; Montereali, M.; Rossi, A.; Ballirano, P. Iron within the erionite cavity and its potential role in inducing its toxicity: Evidence of Fe (III) segregation as extra-framework cation. Microporous Mesoporous Mater. 2017, 237, 168–179. [Google Scholar] [CrossRef]
- Galarneau, A.; Villemot, F.; Rodríguez, J.; Fajula, F.; Coasne, B. Validity of the t-plot method to assess microporosity in Hierarchical micro/mesoporous materials. Langmuir 2014, 30, 13266–13274. [Google Scholar] [CrossRef]
- Novembre, D.; Sabatino, B.; Gimeno, D.; García, M.; Martínez, S. Synthesis of Na-X zeolites from tripolaceous deposits (Cronote, Italy) and volcanic zeolitised rocks (Vico, Volcano, Italy). Microporous Mesoporous Mater. 2004, 75, 1–11. [Google Scholar] [CrossRef]
- Fan, M.; Sun, J.; Bai, S.; Penezai, H. Size effects of extraframework monovalent cations on the termal stability and nitrogen adsorption of LSX zeolite. Microporous Mesoporous Mater. 2015, 202, 44–49. [Google Scholar] [CrossRef]
- Musyoka, N.; Petrik, L.; Hums, E.; Kuhnt, A.; Schwieger, W. Thermal stability studies of zeolites A and X synthesized from South African coal fly ash. Res. Chem. Intermed. 2015, 41, 575–582. [Google Scholar] [CrossRef]
- Van Reeuwijk, L.P. The Thermal Dehydration of Natural Zeolites; H. Veenman & Zonen, B.V.: Wageningen, The Netherlands, 1974; pp. 1–88. [Google Scholar]
- Sharma, P.; Song, J.; Hee Han, M.; Cho, C. GIS-NAP1 zeolite microspheres as potencial water adsorption material: Influence of initial silica concentration on adsorptive and physical/topological properties. Sci. Rep. 2016, 6, 22734. [Google Scholar] [CrossRef]
- Ballirano, P.; Cametti, G. Dehydration dynamics and thermal stability of erionite-K: Experimental evidence of the ‘‘internal ionic exchange’’ mechanism. Microporous Mesoporous 2012, 163, 160–168. [Google Scholar] [CrossRef]
- Kennedy, D.; Mujcin, M.; Trudeau, E.; Tezel, H. Pure and binary adsorption equilibria of methane and nitrogen on activated carbons, desiccants, and zeolites at different pressures. J. Chem. Eng. Data 2016, 61, 3163–3176. [Google Scholar] [CrossRef]
- Shang, J.; Li, G.; Singh, R.; Gu, Q.; Nair, K.; Bastow, T.; Medhekar, N.; Doherty, C.M.; Hill, A.J.; Liu, J.Z.; et al. Discriminative separation of gases by a “Molecular Trapdoor” mechanism in chabazite zeolites. J. Am. Chem. Soc. 2012, 134, 19246–19253. [Google Scholar] [CrossRef] [PubMed]
- Coudert, F.X.; Kohen, D. Molecular insight into CO2 “trapdoor” adsorption in zeolite Na-RHO. Chem. Mater. 2017, 29, 2724–2730. [Google Scholar] [CrossRef]
- Dominguez, G.; Hernandez, R.; Aguilar, G. Isosteric heats of adsoprtion of N2O and NO on natural zeolites. J. Mex. Chem. Soc. 2010, 54, 111–116. [Google Scholar]
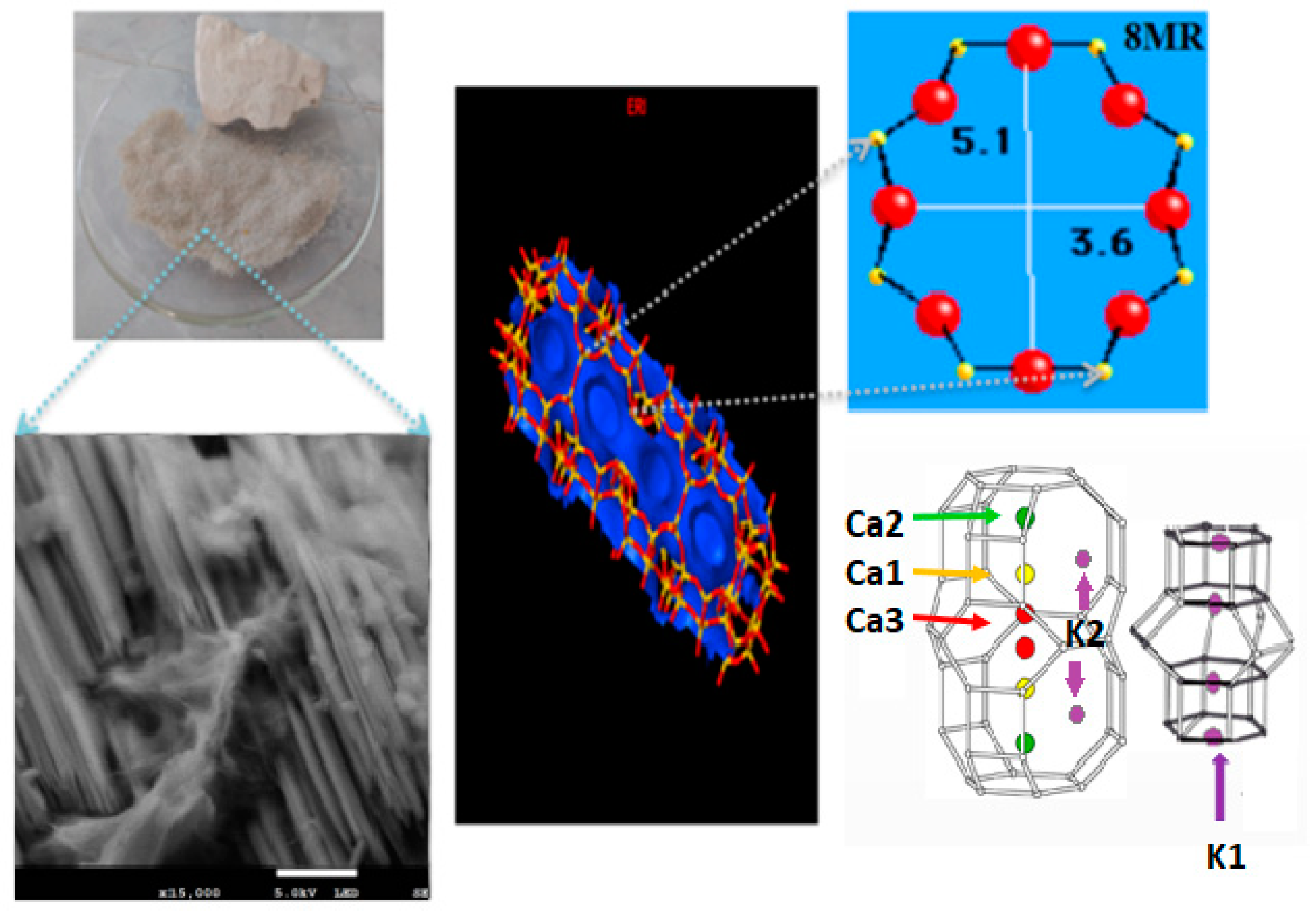
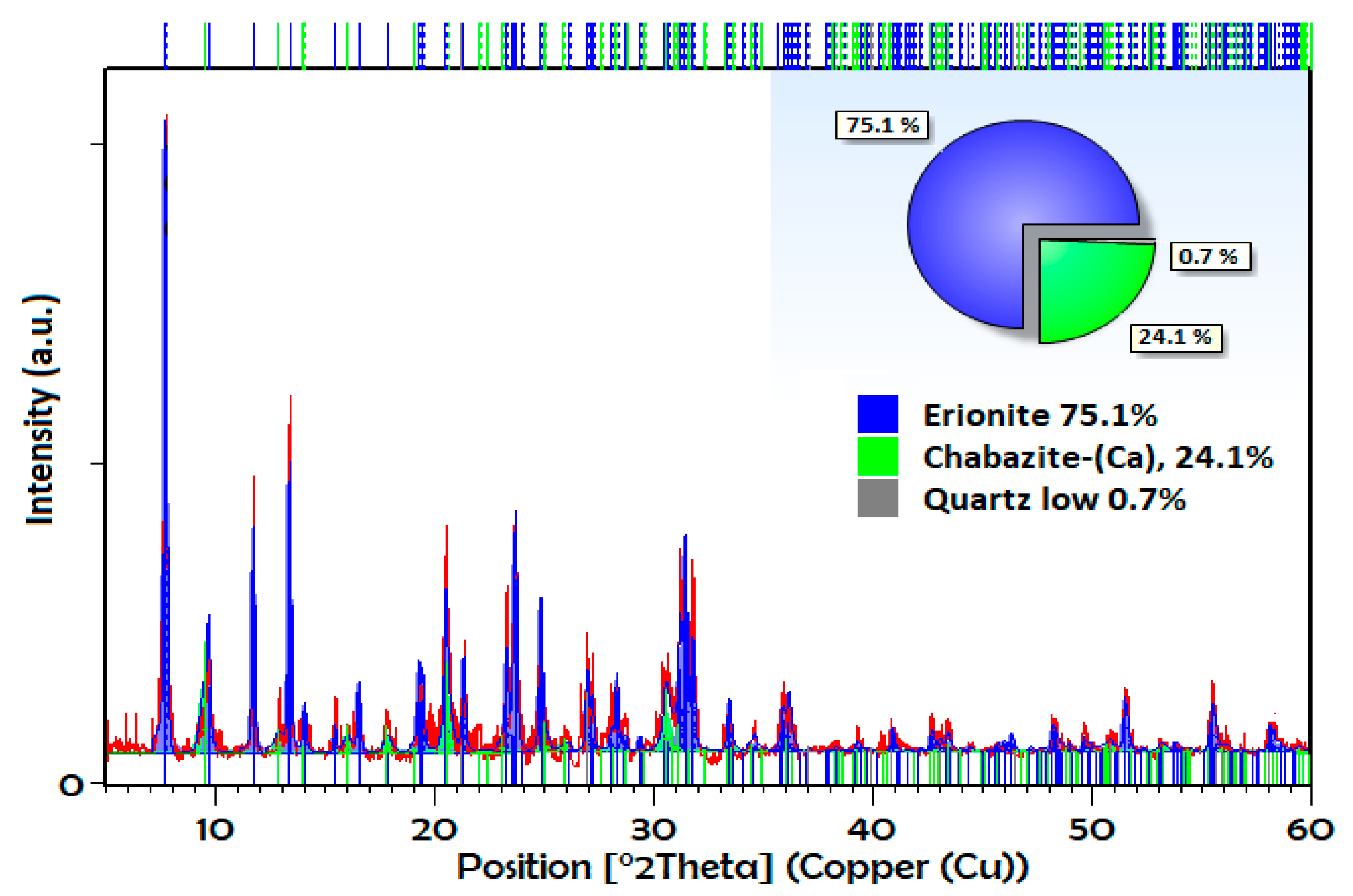

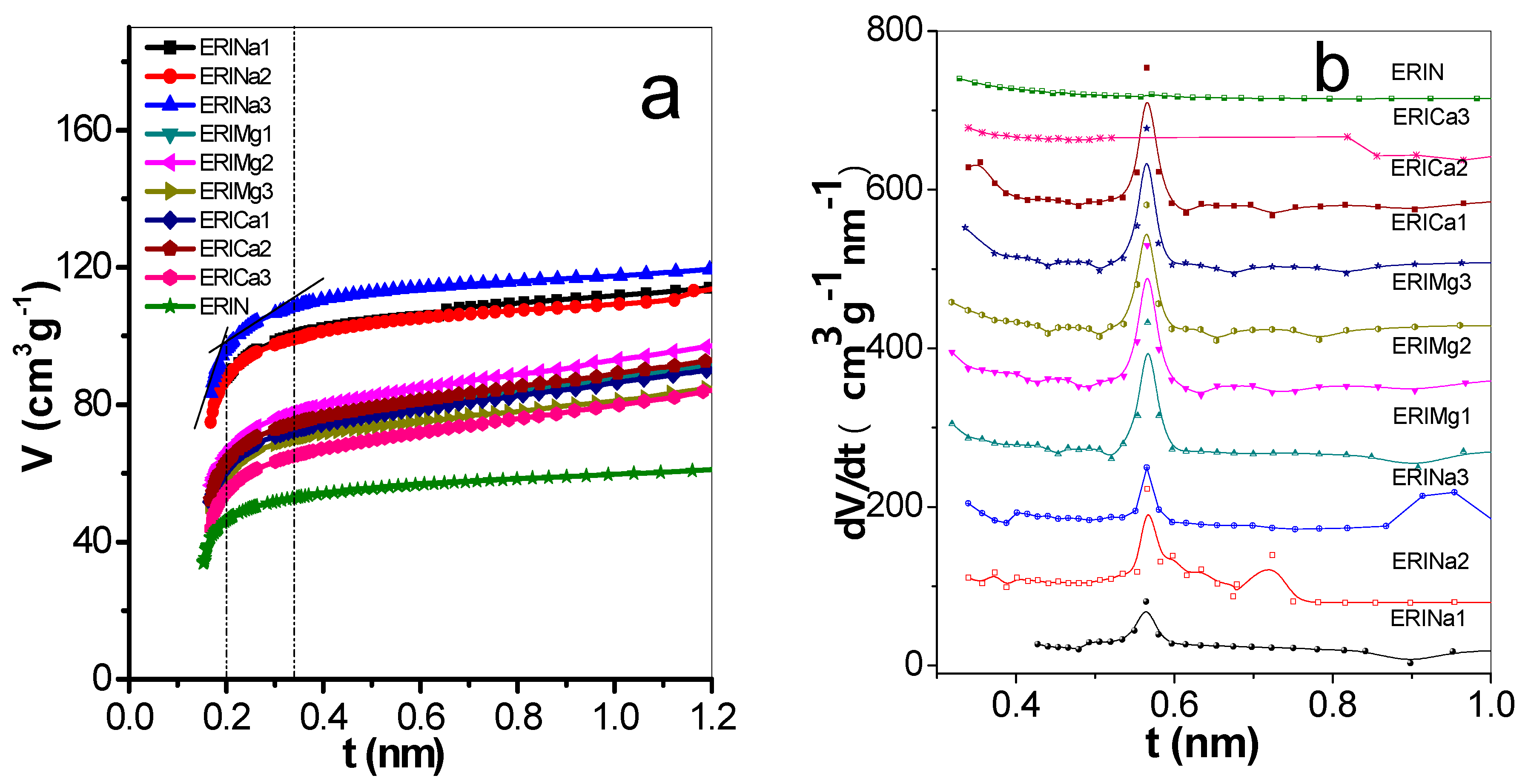
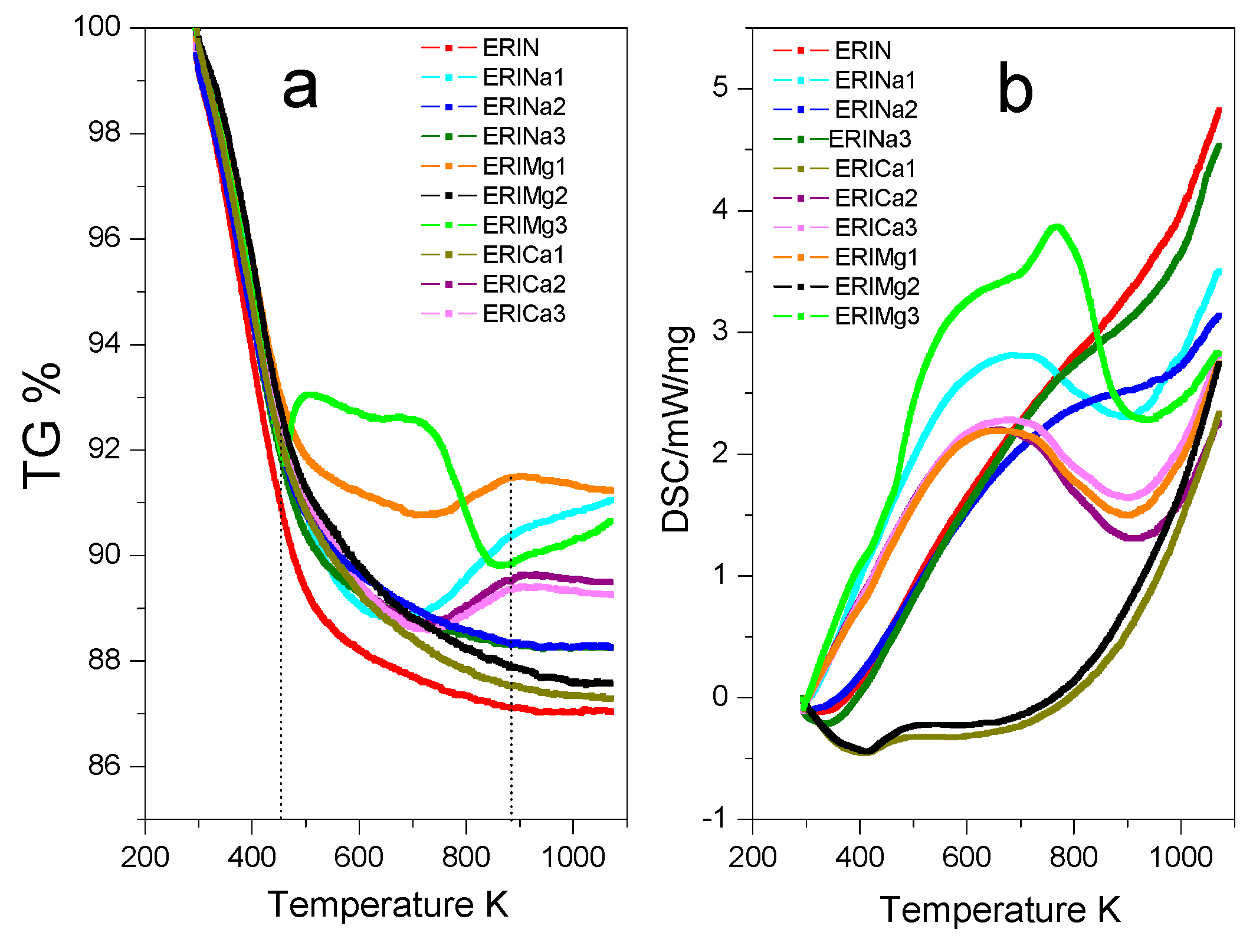



| Gas | Polarizability Å3 | Quadrupole Moment, D Å | Kinetic Diameter, nm |
|---|---|---|---|
| H2 | 0.79 | 0.52 | 0.29 |
| CH4 | 2.45 | 0 | 0.38 |
| ERINat | ERINa1 | ERINa2 | ERINa3 | ERIC1 | ERICa2 | ERICa3 | ERIMg1 | ERIMg2 | ERIMg3 | |
|---|---|---|---|---|---|---|---|---|---|---|
| SiO2 | 59.4 | 59.80 | 60.90 | 60.90 | 64.30 | 62.30 | 60.20 | 61.20 | 60.80 | 59.20 |
| Al2O3 | 12.9 | 13.50 | 13.30 | 14.30 | 13.60 | 13.80 | 13.30 | 13.20 | 12.80 | 13.00 |
| Na2O | 3.52 | 3.76 | 4.69 | 3.85 | 2.80 | 2.40 | 1.21 | 3.07 | 2.55 | 2.94 |
| K2O | 3.56 | 3.20 | 2.66 | 3.35 | 2.66 | 3.07 | 3.13 | 3.21 | 3.17 | 2.76 |
| Fe2O3 | 2.29 | 2.23 | 1.71 | 2.39 | 1.81 | 1.74 | 2.05 | 1.83 | 1.81 | 1.89 |
| MgO | 1.39 | 1.45 | 1.45 | 1.47 | 1.13 | 1.12 | 1.32 | 1.75 | 1.70 | 1.96 |
| CaO | 1.31 | 1.12 | 1.46 | 1.48 | 2.24 | 2.72 | 3.44 | 1.29 | 1.33 | 1.35 |
| TiO2 | 0.4 | 0.19 | 0.20 | 0.23 | 0.20 | 0.20 | 0.32 | - | 0.18 | 0.24 |
| H2O | 15.23 | 14.75 | 13.63 | 12.03 | 11.26 | 12.65 | 15.03 | 14.45 | 15.66 | 16.66 |
| Total | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 |
| Si/Al | 3.91 | 4.07 | 4.04 | 3.76 | 4.18 | 3.99 | 4.00 | 4.09 | 4.20 | 4.02 |
| Sample | Wo cm3/g | Dp nm | AsL [26] m2/g |
|---|---|---|---|
| ERIN | 0.09 | 0.51 | 243.9 |
| ERINa1 | 0.17 | 0.49 | 244.7 |
| ERINa2 | 0.17 | 0.48 | 504.4 |
| ERINa3 | 0.18 | 0.47 | 545.7 |
| ERIMg1 | 0.13 | 0.54 | 322.7 |
| ERIMg2 | 0.14 | 0.54 | 343.8 |
| ERIMg3 | 0.13 | 0.53 | 309.4 |
| ERICa1 | 0.13 | 0.55 | 325.5 |
| ERICa2 | 0.14 | 0.54 | 331.5 |
| ERICa3 | 0.12 | 0.56 | 293.8 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Quiroz-Estrada, K.; Hernández, M.Á.; Felipe-Mendoza, C.; Santamaría-Juárez, J.D.; Petranovskii, V.; Rubio, E. Critical Admission Temperature of H2 and CH4 in Nanopores of Exchanged ERI Zeolites. Nanomaterials 2019, 9, 160. https://doi.org/10.3390/nano9020160
Quiroz-Estrada K, Hernández MÁ, Felipe-Mendoza C, Santamaría-Juárez JD, Petranovskii V, Rubio E. Critical Admission Temperature of H2 and CH4 in Nanopores of Exchanged ERI Zeolites. Nanomaterials. 2019; 9(2):160. https://doi.org/10.3390/nano9020160
Chicago/Turabian StyleQuiroz-Estrada, Karla, Miguel Ángel Hernández, Carlos Felipe-Mendoza, Juana Deisy Santamaría-Juárez, Vitalii Petranovskii, and Efraín Rubio. 2019. "Critical Admission Temperature of H2 and CH4 in Nanopores of Exchanged ERI Zeolites" Nanomaterials 9, no. 2: 160. https://doi.org/10.3390/nano9020160
APA StyleQuiroz-Estrada, K., Hernández, M. Á., Felipe-Mendoza, C., Santamaría-Juárez, J. D., Petranovskii, V., & Rubio, E. (2019). Critical Admission Temperature of H2 and CH4 in Nanopores of Exchanged ERI Zeolites. Nanomaterials, 9(2), 160. https://doi.org/10.3390/nano9020160






