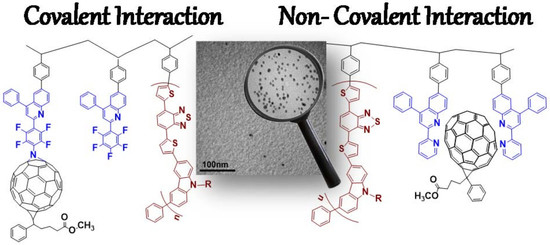
Copolymers and Hybrids Based on Carbazole Derivatives and Their Nanomorphology Investigation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Characterization
3. Results
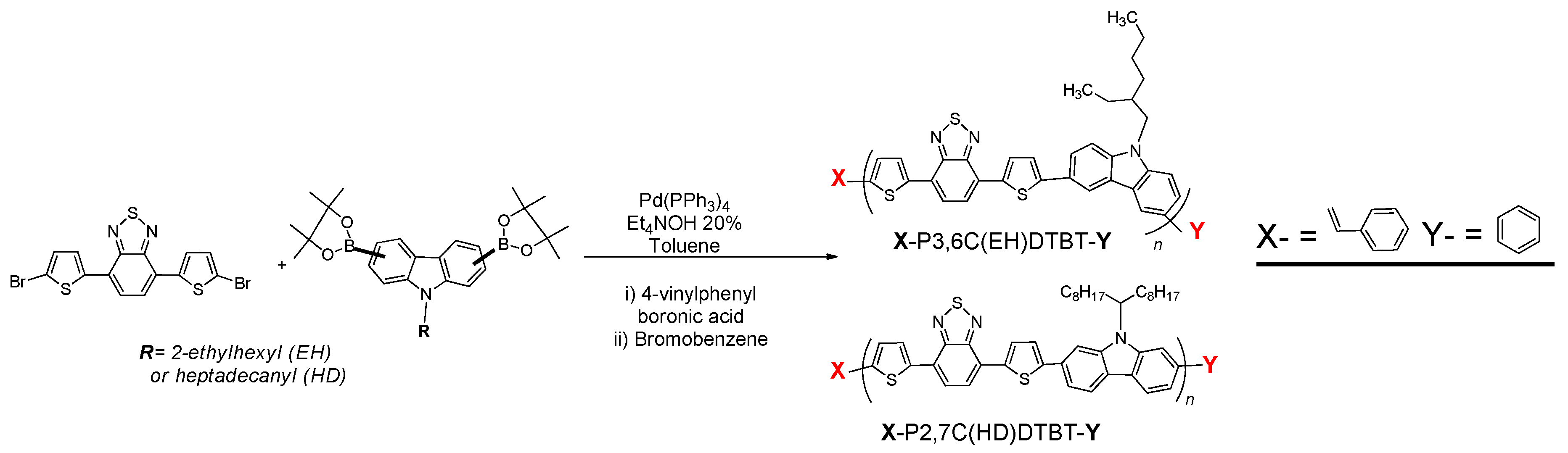
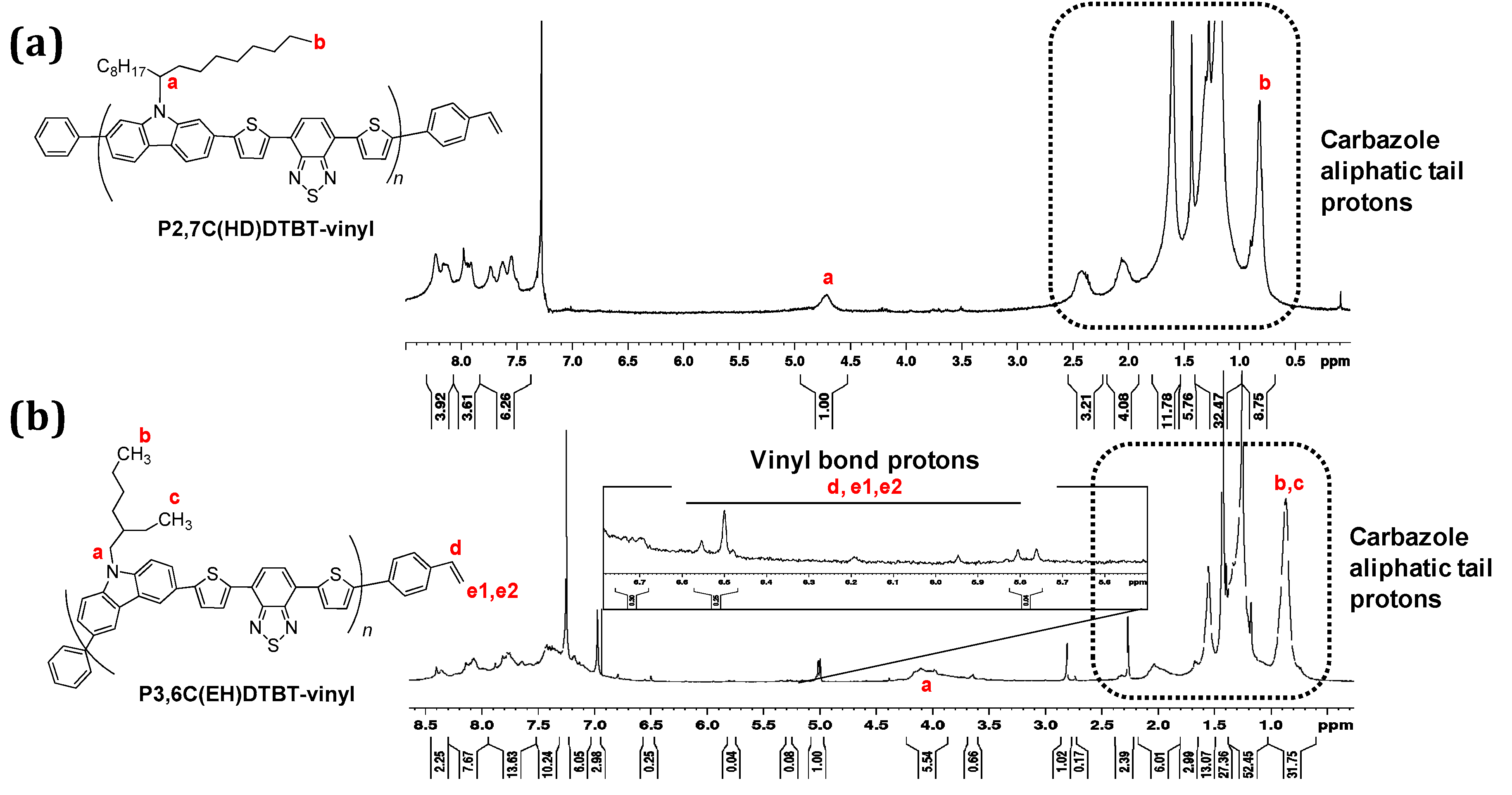
3.1. Synthesis of Functionalized Vinyl PCDTBT-Based Macromonomers
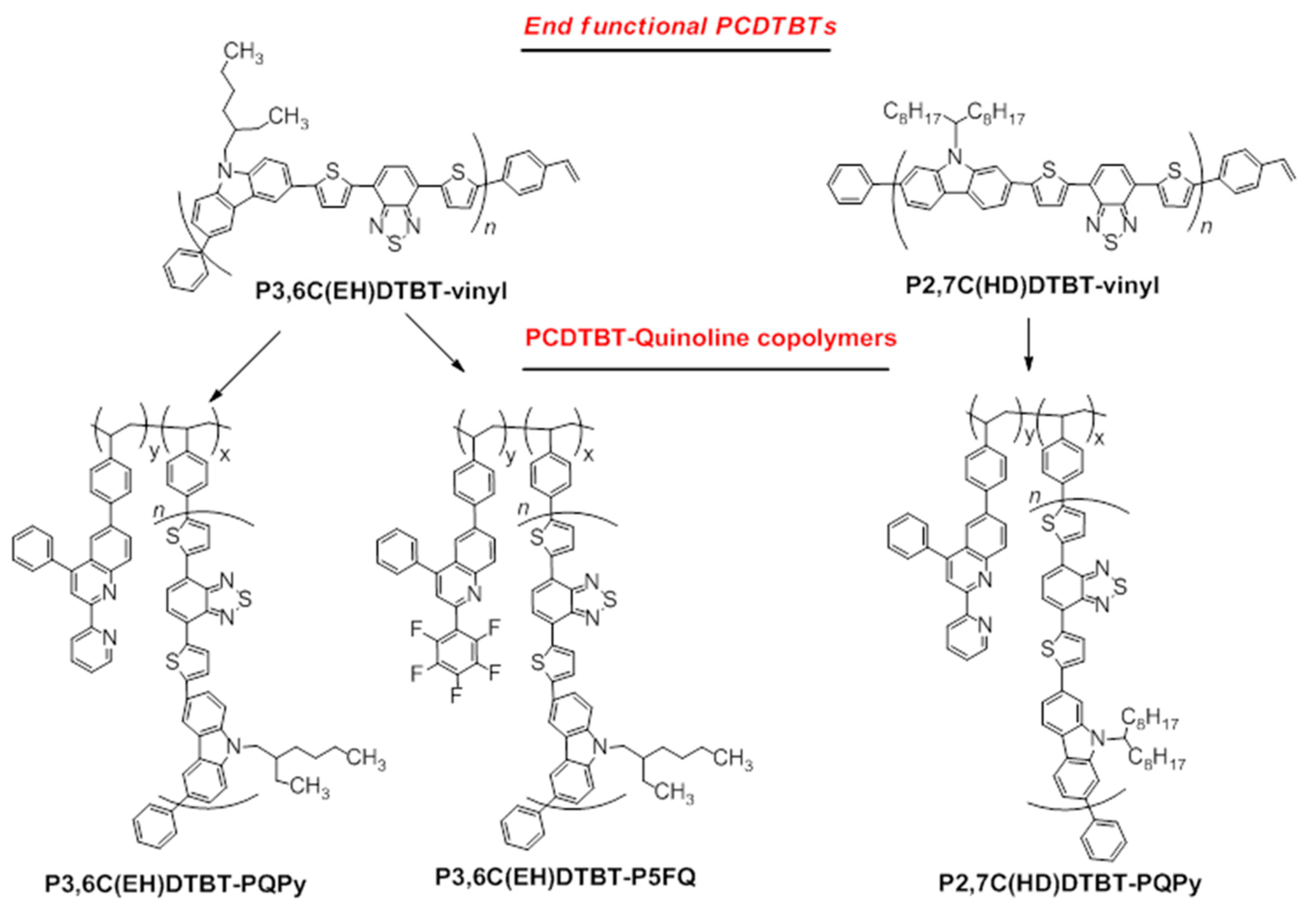
3.2. Synthesis of PCDTBT-Quinoline Copolymers
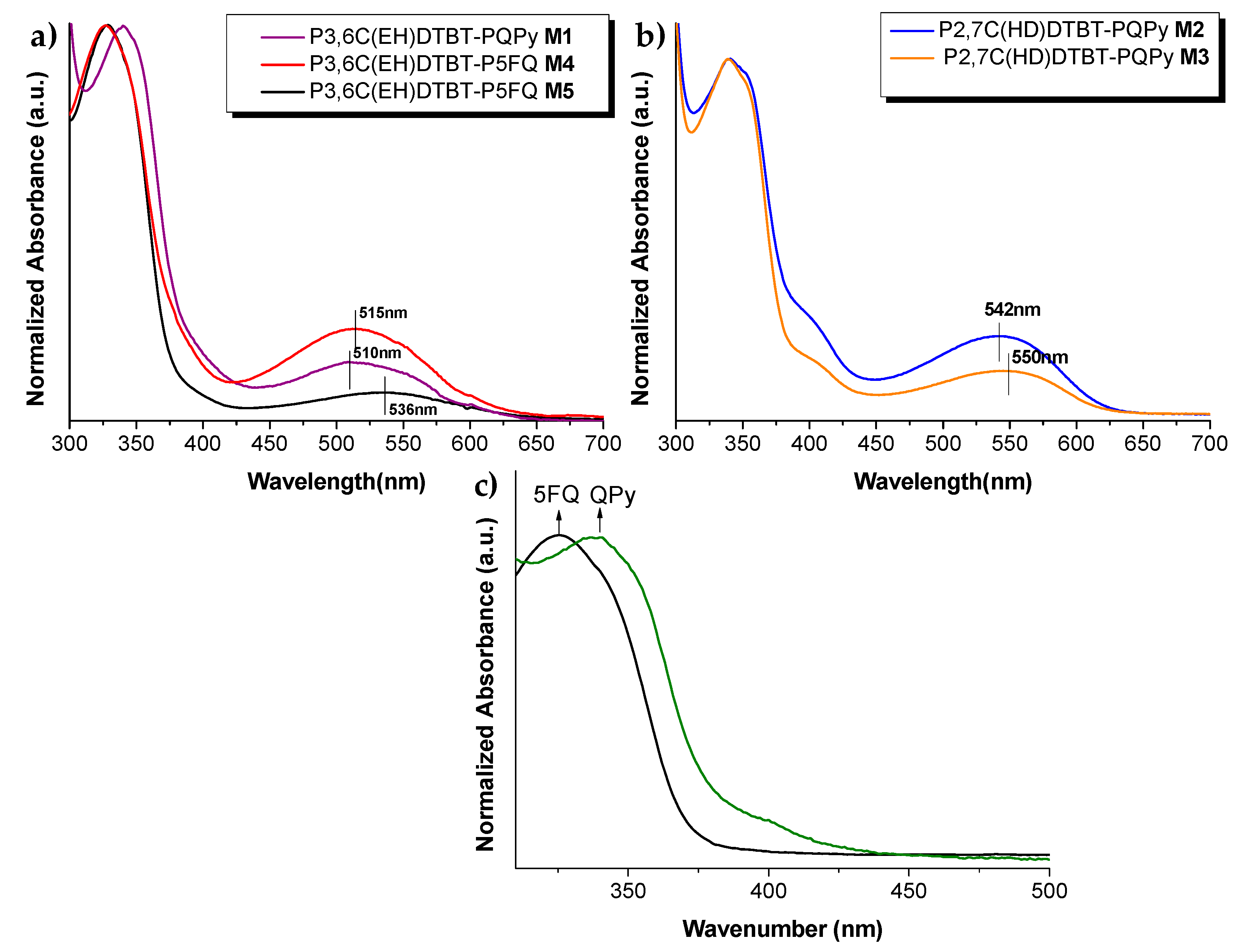
3.3. Optical Characterization of Copolymers
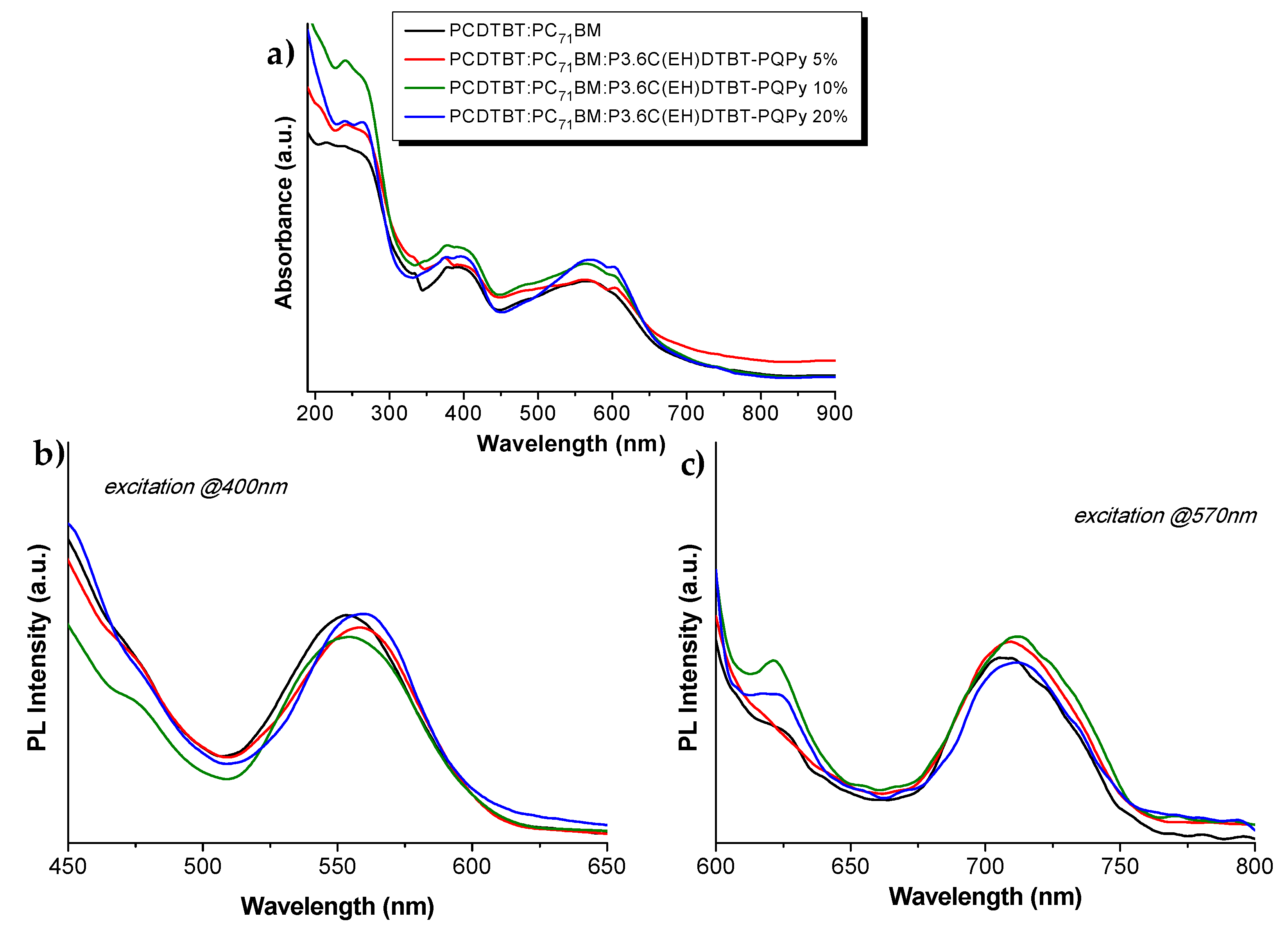
3.4. Blends of PCDTBT:PC71BM with PCDTBT-PQPy Copolymers
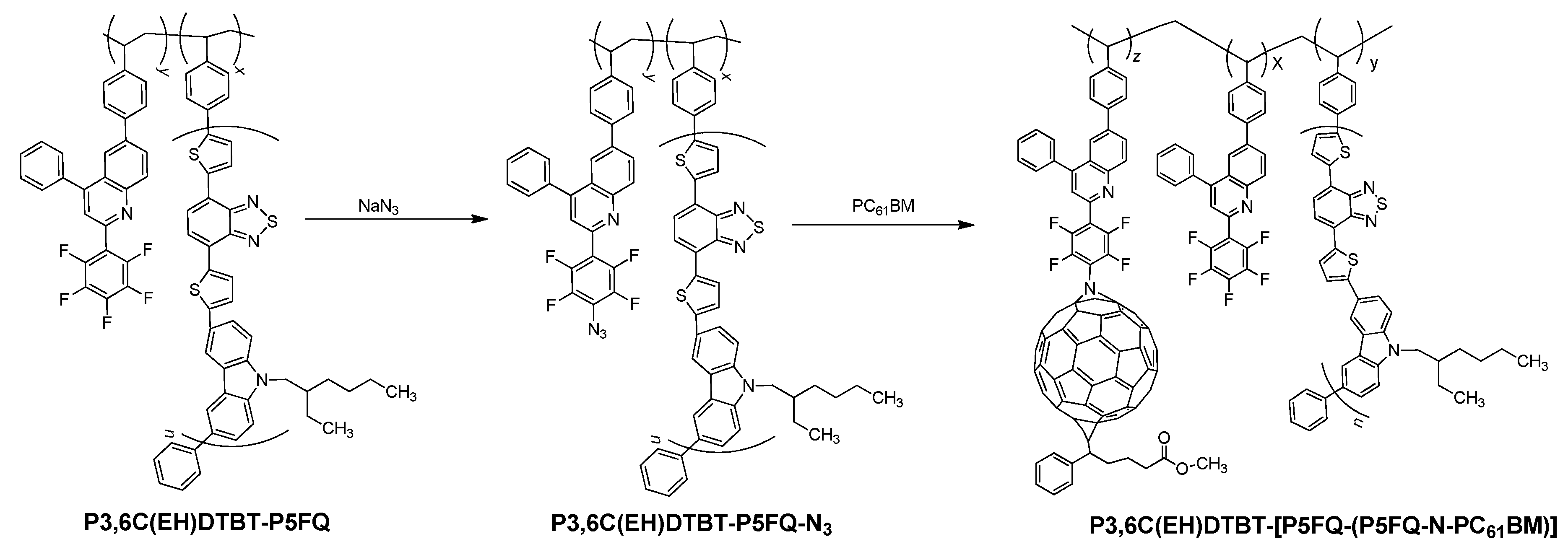
3.5. Hybrid Materials from the PCDTBT-P5FQ Copolymer
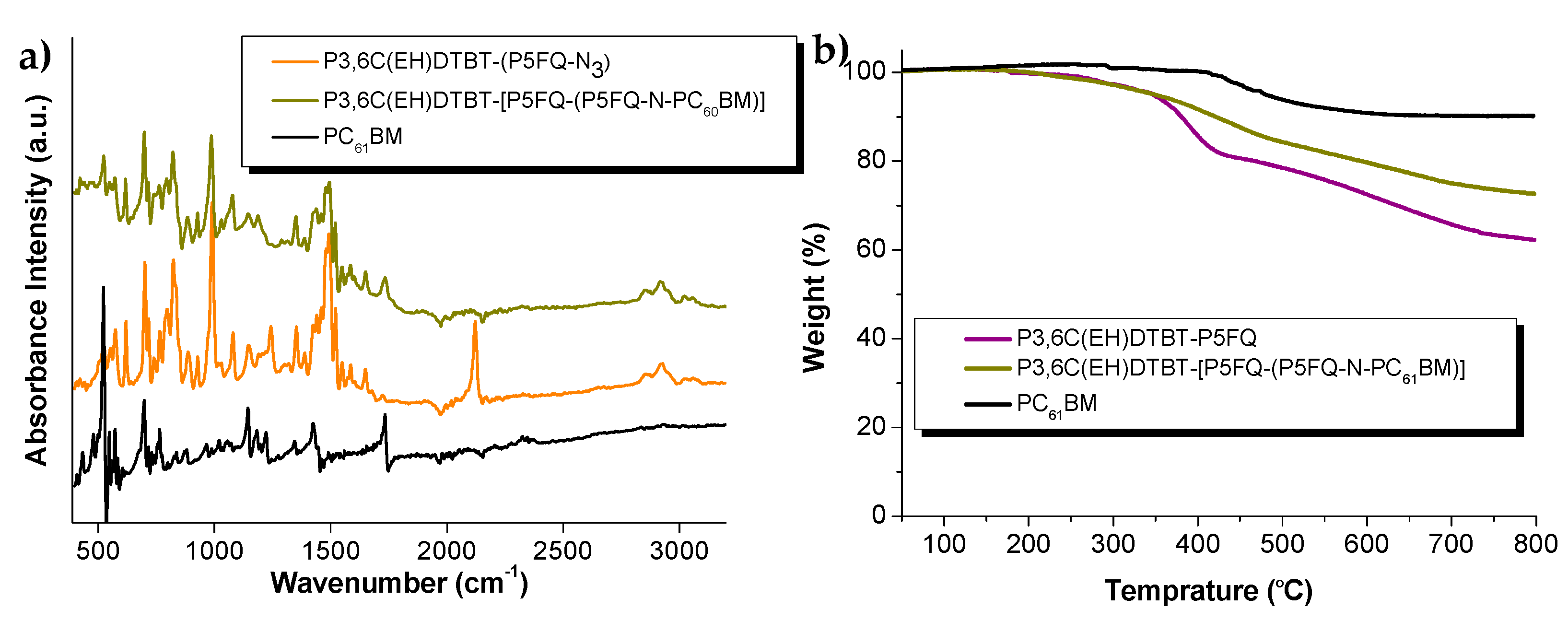
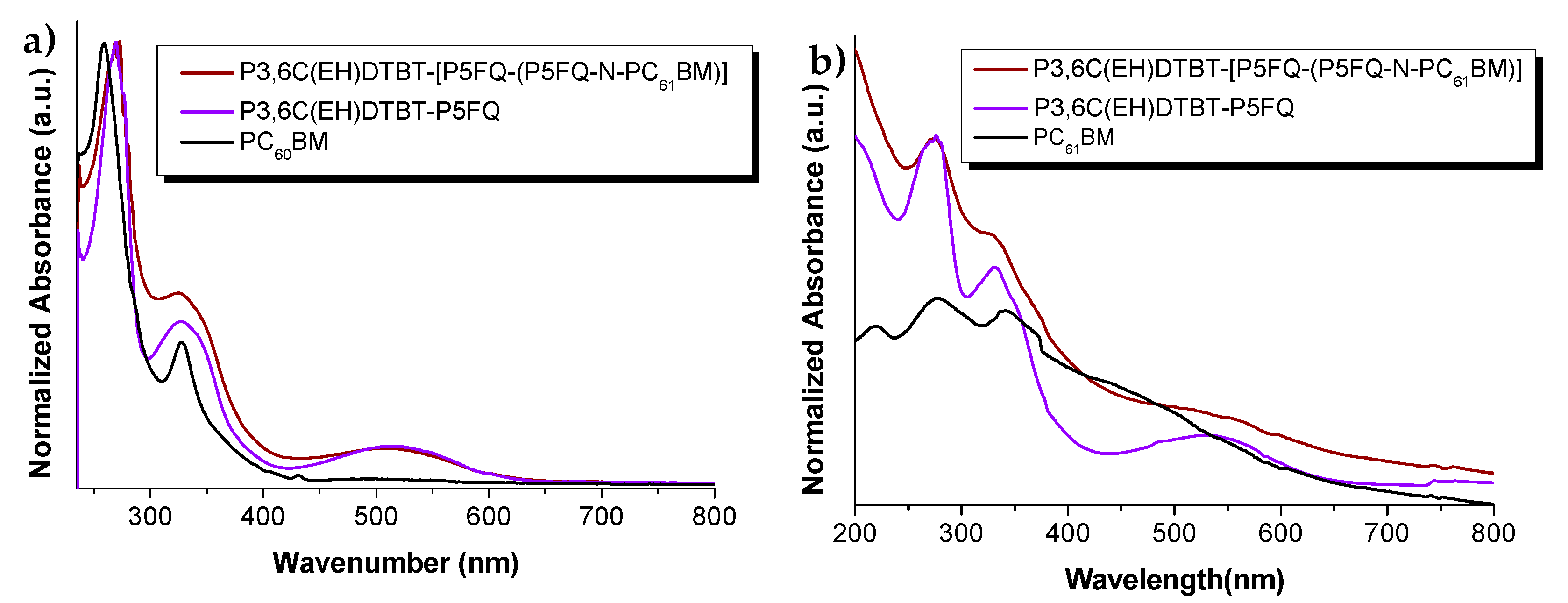
3.6. Optical Properties and Morphology Characteriaztion of Hybrid-Material
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Thompson, B.C.; Fréchet, J.M. Polymer-fullerene composite solar cells. Angew. Chem. Int. Ed. 2008, 47, 58–77. [Google Scholar] [CrossRef] [PubMed]
- Gunes, S.; Neugebauer, H.; Sariciftci, N.S. Conjugated polymer-based organic solar cells. Chem. Rev. 2007, 107, 1324–1338. [Google Scholar] [CrossRef] [PubMed]
- Logothetidis, S.; Laskarakis, A. Towards the optimization of materials and processes for flexible organic electronics devices. EPJ Appl. Phys. 2009, 46, 12502. [Google Scholar] [CrossRef]
- Liu, X.; Wu, Y.; Li, X.; Zhang, W.; Zhao, L.; Wang, H.; Fang, J. CdS–phenanthroline derivative hybrid cathode interlayers for high performance inverted organic solar cells. J. Mater. Chem. A 2016, 4, 297–302. [Google Scholar] [CrossRef]
- He, Z.; Xiao, B.; Liu, F.; Wu, H.; Yang, Y.; Xiao, S.; Wang, C.; Russell, P.T.; Cao, Y. Single-junction polymer solar cells with high efficiency and photovoltage. Nat. Photonics 2015, 9, 174–179. [Google Scholar] [CrossRef]
- Jagadamma, L.K.; Al-Senani, M.; El-Labban, A.; Gereige, I.; Ngongang Ndjawa, G.O.; Faria, J.C.D.; Kim, T.; Zhao, K.; Cruciani, F.; Anjum, D.H.; et al. Polymer Solar Cells: Polymer Solar Cells with Efficiency >10% Enabled via a Facile Solution-Processed Al-Doped ZnO Electron Transporting Layer. Adv. Energy Mater. 2015, 5, 1500204. [Google Scholar] [CrossRef]
- Kong, J.; Hwang, I.W.; Lee, K. Top-down approach for nanophase reconstruction in bulk heterojunction solar cells. Adv. Mater. 2014, 26, 6275–6283. [Google Scholar] [CrossRef]
- Rogers, J.T.; Schmidt, K.; Toney, M.; Kramer, E.J.; Bazan, G.C. Structural Order in Bulk Heterojunction Films Prepared with Solvent Additives. Adv. Mater. 2011, 23, 2284–2288. [Google Scholar] [CrossRef]
- He, Y.; Chen, H.Y.; Hou, J.; Li, Y. Indene−C60 Bisadduct: A New Acceptor for High-Performance Polymer Solar Cells. J. Am. Chem. Soc. 2010, 132, 1377–1382. [Google Scholar] [CrossRef]
- Guo, X.; Zhang, M.; Cui, C.; Hou, J.; Li, Y. Efficient Polymer Solar Cells Based on Poly(3-hexylthiophene) and Indene–C60 Bisadduct Fabricated with Non-halogenated Solvents. ACS Appl. Mater. Interfaces 2014, 6, 8190–8198. [Google Scholar] [CrossRef]
- Zhang, Y.; Bovill, E.; Kingsley, J.; Buckley, A.R.; Yi, H.; Iraqi, A.; Wang, T.; Lidzey, D.G. PCDTBT based solar cells: One year of operation under real-world conditions. Sci. Rep. 2016, 6, 21632. [Google Scholar] [CrossRef] [PubMed]
- Shim, C.; Kim, M.; Ihn, S.G.; Choi, Y.S.; Kim, Y.; Cho, K. Controlled nanomorphology of PCDTBT–fullerene blends via polymer end-group functionalization for high efficiency organic solar cells. Chem. Commun. 2012, 48, 7206–7208. [Google Scholar] [CrossRef] [PubMed]
- Ye, L.; Zhang, S.; Zhao, W.; Yao, H.; Hou, J. Highly Efficient 2D-Conjugated Benzodithiophene-Based Photovoltaic Polymer with Linear Alkylthio Side Chain. Chem. Mater. 2014, 26, 3603–3605. [Google Scholar] [CrossRef]
- Stenta, C.; Molina, D.; Viterisi, A.; Montero-Rama, M.P.; Pla, S.; Cambarau, W.; Fernández-Lázaro, F.; Palomares, E.; Marsal, L.F.; Sastre-Santos, A. Diphenylphenoxy-Thiophene-PDI Dimers as Acceptors for OPV Applications with Open Circuit Voltage Approaching 1 Volt %. Nanomaterials 2018, 8, 211. [Google Scholar] [CrossRef]
- Li, Y.N.; Vamvounis, G.; Holdcroft, S. Facile functionalization of poly(3-alkylthiophene)s via electrophilic substitution. Macromolecules 2001, 34, 141–143. [Google Scholar] [CrossRef]
- Kim, I.K.; Jo, J.H.; Yun, J.-H. Morphology-Controlled High-Efficiency Small Molecule Organic Solar Cells without Additive Solvent Treatment. Nanomaterials 2016, 6, 64. [Google Scholar] [CrossRef] [PubMed]
- Lu, Z.; Liu, W.; Li, J.; Fang, T.; Li, W.; Zhang, J.; Feng, F.; Li, W. The Influence of Fluorination on Nano-Scale Phase Separation and Photovoltaic Performance of Small Molecular/PC71BM Blends. Nanomaterials 2016, 6, 80. [Google Scholar] [CrossRef] [PubMed]
- Vahdani, P.; Li, X.; Zhang, C.; Holdcroft, S.; Frisken, B.J. Morphological characterization of a new low-bandgap thermocleavable polymer showing stable photovoltaic properties. J. Mater. Chem. A 2016, 4, 10650–10658. [Google Scholar] [CrossRef]
- Seibers, Z.D.; Le, T.P.; Lee, Y.; Gomez, E.D.; Kilbey, S.M. Impact of Low Molecular Weight Poly(3-hexylthiophene)s as Additives in Organic Photovoltaic Devices. ACS Appl. Mater. Interfaces 2018, 10, 2752–2761. [Google Scholar] [CrossRef]
- Karagiannidis, P.G.; Georgiou, D.; Pitsalidis, C.; Laskarakis, A.; Logothetidis, S. Evolution of vertical phase separation in P3HT:PCBM thin films induced by thermal annealing. Mater. Chem. Phys. 2011, 129, 1207–1213. [Google Scholar] [CrossRef]
- Kim, Y.; Cook, S.; Kirkpatrick, J.; Nelson, J.; Durrant, J.R.; Bradley, D.D.C.; Giles, M.; Heeney, M.; Hamilton, R.; McCulloch, I. Effect of the End Group of Regioregular Poly(3-hexylthiophene) Polymers on the Performance of Polymer/Fullerene Solar Cells. J. Phys. Chem. 2007, 111, 8137–8141. [Google Scholar] [CrossRef]
- Kim, J.S.; Lee, Y.; Lee, J.H.; Park, J.H.; Kim, J.K.; Cho, K. High-Efficiency Organic Solar Cells Based on End-Functional-Group-Modified Poly(3-hexylthiophene). Adv. Mater. 2010, 22, 1355–1360. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Han, A.-R.; Cho, C.-H.; Kang, H.; Cho, H.-H.; Lee, M.Y.; Fréchet, J.M.J.; Oh, J.H.; Kim, B.J. Solvent-Resistant Organic Transistors and Thermally Stable Organic Photovoltaics Based on Cross-linkable Conjugated Polymers. Chem. Mater. 2012, 24, 215–221. [Google Scholar] [CrossRef]
- Yassara, A.; Miozzoa, L.; Girondaa, R.; Horowitz, G. Rod–coil and all conjugated block copolymers for photovoltaic applications. Prog. Polym. Sci. 2013, 38, 791–844. [Google Scholar] [CrossRef]
- Kallitsis, J.K.; Anastasopoulos, C.; Andreopoulou, A.K. Functional semiconductors targeting copolymer architectures and hybrid nanostructures. MRS Commun. 2015, 5, 365–382. [Google Scholar] [CrossRef]
- Kakogianni, S.; Andreopoulou, A.K.; Kallitsis, J.K. Synthesis of Polythiophene–Fullerene Hybrid Additives as Potential Compatibilizers of BHJ Active Layers. Polymers 2016, 8, 440. [Google Scholar] [CrossRef]
- Kakogianni, S.; Lebedeva, M.; Paloumbis, G.; Andreopoulou, A.K.; Porfyrakis, K.; Kallitsis, J.K. Semiconducting End-Perfluorinated P3HT-Fullerenic Hybrids as Potential Additives for P3HT/IC70BA blends. RSC Adv. 2016, 6, 100. [Google Scholar] [CrossRef]
- Sygellou, L.; Kakogianni, S.; Andreopoulou, A.K.; Theodosiou, K.; Leftheriotis, G.; Kallitsis, J.K.; Siokou, A.E. Evaluation of the electronic properties of perfluorophenyl functionalized quinolines and their hybrids with carbon nanostructures. Phys. Chem. Chem. Phys. 2016, 18, 5. [Google Scholar] [CrossRef]
- Yuan, K.; Chen, L.; Chen, Y. Nanostructuring compatibilizers of block copolymers for organic Photovoltaics. Polym. Int. 2014, 63, 593–606. [Google Scholar] [CrossRef]
- Sartorio, C.; Campisciano, V.; Chiappara, C.; Cataldo, S.; Scopelliti, M.; Gruttadauria, M.; Giacalone, F.; Pignataro, B. Enhanced Power-Conversion Efficiency in Organic Solar Cells Incorporating Copolymeric Phase-Separation Modulators. J. Mater. Chem. A 2018, 6, 3884–3894. [Google Scholar] [CrossRef]
- Laiho, A.; Ras, R.H.A.; Valkama, S.; Ruokolainen, J.; Österbacka, R.; Ikkala, O. Control of Self-Assembly by Charge-Transfer Complexation between C60 Fullerene and Electron Donating Units of Block Copolymers. Macromolecules 2006, 39, 7648–7653. [Google Scholar] [CrossRef]
- Renaud, C.; Mougnier, S.-J.; Pavlopoulou, E.; Brochon, C.; Fleury, G.; Deribew, D.; Portale, G.; Cloutet, E.; Chambon, S.; Vignau, L.; et al. Block Copolymer as a Nanostructuring Agent for High-Efficiency and Annealing-Free Bulk Heterojunction Organic Solar Cells. Adv. Mater. 2012, 24, 2196–2201. [Google Scholar] [CrossRef] [PubMed]
- Hsu, S.; Chen, C.; Cheng, Y.; Wei, K. New Carbazole-Based Conjugated Polymers Containing Pyridylvinyl Thiophene Units for Polymer Solar Cell Applications: Morphological Stabilization Through Hydrogen Bonding. J. Polym. Sci. Part A Polym. Chem. 2011, 49, 603–611. [Google Scholar] [CrossRef]
- Chen, H.; Chen, J.; Yin, W.; Yu, X.; Shao, M.; Xiao, K.; Hong, K.; Pickel, D.L.; Kochemba, W.M.; Kilbey, S.M., II; Dadmun, M. Correlation of polymeric compatibilizer structure to its impact on the morphology and function of P3HT:PCBM bulk heterojunctions. J. Mater. Chem. A 2013, 1, 5309–5319. [Google Scholar] [CrossRef]
- Kakogianni, S.; Kourkouli, S.N.; Andreopoulou, A.K.; Kallitsis, J.K. A versatile approach for creating hybrid semiconducting polymer–fullerene architectures for organic electronics. J. Mater. Chem. A 2014, 2, 8110–8117. [Google Scholar] [CrossRef]
- Kourkouli, S.N.; Siokou, A.; Stefopoulos, A.A.; Ravani, F.; Plocke, T.; Müller, M.; Maultzsch, J.; Thomsen, C.; Papagelis, K.; Kallitsis, J.K. Electronic Properties of Semiconducting Polymer-Functionalized Single Wall Carbon Nanotubes. Macromolecules 2013, 46, 2590–2598. [Google Scholar] [CrossRef]
- Stefopoulos, A.A.; Kourkouli, S.N.; Economopoulos, S.; Ravani, F.; Andreopoulou, A.K.; Papagelis, K.; Siokou, A.; Kallitsis, J.K. Polymer and hybrid electron accepting materials based on a semiconducting perfluorophenylquinoline. Macromolecules 2010, 43, 4827–4828. [Google Scholar] [CrossRef]
- Giannopoulos, P.; Raptis, D.; Theodosiou, K.; Andreopoulou, A.K.; Anastasopoulos, C.; Dokouzis, A.; Leftheriotis, G.; Lianos, P.; Kallitsis, J.K. Organic Dyes End-Capped with Perfluorophenyl Anchors: Synthesis, Electrochemical Properties and Assessment of Sensitization Capacity of Titania Photoanodes. Dyes Pigments 2018, 148, 167–179. [Google Scholar] [CrossRef]
- Schimperna, G.; Bianchi, G. Process for the preparation of benzohetero [1,3]-diazole compounds disubstituted with heteoaryl groups. U.S. Patent WO2013021315 A1, 14 February 2013. [Google Scholar]
- Zhang, C. Process for preparing a 4,7-bis(5-halothien-2-yl)-2,1,3-benzothiadiazole and a precursor therefor. U.S. Patent 2004/0229925 A1, 18 November 2004. [Google Scholar]
- Blouin, N.; Michaud, A.; Leclerc, M. A Low-Bandgap Poly(2,7-Carbazole) Derivative for Usein High-Performance Solar Cell. Adv. Mater. 2007, 19, 2295–2300. [Google Scholar] [CrossRef]
- Li, J.; Meng, Q.; Kim, J.; Lee, Y. A Thiophene, Benzothiadiazole, and Carbazole-Based Copolymer Synthesis and Characterization. Bull. Korean Chem. Soc. 2009, 30, 951–954. [Google Scholar] [CrossRef]
- Berton, N.; Ottone, C.; Labet, V.; Bettignies, R.; Bailly, S.; Grand, A.; Morell, C.; Sadki, S.; Chandezon, F. New Alternating Copolymers of 3,6-Carbazoles and Dithienylbenzothiadiazoles: Synthesis, Characterization, and Application in Photovoltaics. Macromol. Chem. Phys. 2011, 212, 2127–2141. [Google Scholar] [CrossRef]
- Coulson, D.R.; Satek, L.C.; Grim, S. Tetrakis(triphenylphosphine)palladium(0). In Inorganic Syntheses; Cotton, F.A., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 1972; Volume 13, ISBN 9780470132449. [Google Scholar]
- Kim, J.; Kwon, Y.S.; Shin, W.S.; Moon, S.J.; Park, T. Carbazole-Based Copolymers: Effects of Conjugation Breaks and Steric Hindrance. Macromolecules 2011, 44, 1909–1919. [Google Scholar] [CrossRef]
- Fu, Y.; Kim, J.; Siva, A.; Shin, W.S.; Moon, S.J.; Park, T. Parameters Influencing the Molecular Weight of 3,6-Carbazole-Based D-p-A-Type Copolymers. J. Polym. Sci. A Polym. Chem. 2011, 49, 4368–4378. [Google Scholar] [CrossRef]
- Svensson, M.; Zhang, F.; Veenstra, S.C.; Verhees, W.J.H.; Hummelen, J.C.; Kroon, J.M.; Inganäs, O.; Andersson, M.R. High-Performance Polymer Solar Cells of an Alternating Polyfluorene Copolymer and a Fullerene Derivative. Adv. Mater. 2003, 15, 988–991. [Google Scholar] [CrossRef] [Green Version]
- Kingsley, J.W.; Marchisio, P.P.; Yi, H.; Iraqi, A.; Kinane, C.J.; Langridge, S.; Thompson, R.L.; Cadby, A.J.; Pearson, A.J.; Lidzey, D.G.; et al. Molecular weight dependent vertical composition profiles of PCDTBT:PC71BM blends for organic photovoltaics. Sci. Rep. 2014, 4, 5286. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, S.H.; Roy, A.; Beaupre, S.; Cho, S.; Coates, N.; Moon, J.S.; Moses, D.; Leclerc, M.; Lee, K.; Heeger, A.J. Bulk heterojunction solar cells with internal quantum efficiency approaching 100%. Nat. Photonics 2009, 3, 297–302. [Google Scholar] [CrossRef]








| Copolymer | PCDTBT-Quinoline Theoretical wt% Ratio | Peak Integrals of 1H NMR | molar % Ratio Calculated from 1H NMR | wt. % Ratio Calculated from 1H NMR | |
|---|---|---|---|---|---|
| 6.95–6.3 ppm 2H-PQuinoline | 4.3–4.1 ppm 2H-carbazole | ||||
| M1 | 11/89 | 3.778 | 0.022 | 5/95 | 7/93 |
| M2 | 11/89 | 0.555 | 0.026 | 5/95 | 9/91 |
| M3 | 11/89 | 5.690 | 1.000 | 15/85 | 24/76 |
| M4 | 11/89 | 0.352 | 0.090 | 20/80 | 23/77 |
| M5 | 11/89 | 0.700 | 0.040 | 5/95 | 6/94 |
| Copolymer | Mn PCDTBT | PCDTBT/Quinoline % molar ratio via 1H NMR | GPC @ 254 nm | GPC @540 nm | ||||
|---|---|---|---|---|---|---|---|---|
| Mn | Mw | PDI | Mn | Mw | PDI | |||
| PQPy-based copolymers | ||||||||
| P3,6C(EH)DTBT-PQPy (M1) | 2100 | 5/95 | 3500 | 4100 | 1.17 | 3650 | 4000 | 1.1 |
| P2,7C(HD)DTBT-PQPy (M2) | 3300 | 5/95 | 3450 | 4000 | 1.16 | 3500 | 4000 | 1.14 |
| P2,7C(HD)DTBT-PQPy (M3) | 5700 | 15/85 | 6000 | 7000 | 1.17 | 5700 | 6600 | 1.16 |
| P5FQ-based copolymers | ||||||||
| P3,6C(EH)DTBT-P5FQ (M4) | 2100 | 20/80 | 4900 | 6100 | 1.2 | 5600 | 6700 | 1.2 |
| P3,6C(EH)DTBT-P5FQ (M5) | 2800 | 5/95 | 8200 | 12700 | 1.55 | 7700 | 12000 | 1.56 |
| Blend | PCDTBT:PC71BM wt. % | Copolymer wt. % |
|---|---|---|
| P3,6C(EH)DTBT-PQPy (M1) | ||
| 8 | 100 | 0 |
| 9 | 47,5:47,5 | 5 |
| 10 | 45:45 | 10 |
| 11 | 40:40 | 20 |
| P2,7C(HD)DTBT-PQPy (M2) | ||
| 12 | 47,5:47,5 | 5 |
| 13 | 45:45 | 10 |
| 14 | 40:40 | 20 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aivali, S.; Kakogianni, S.; Anastasopoulos, C.; Andreopoulou, A.K.; Kallitsis, J.K. Copolymers and Hybrids Based on Carbazole Derivatives and Their Nanomorphology Investigation. Nanomaterials 2019, 9, 133. https://doi.org/10.3390/nano9020133
Aivali S, Kakogianni S, Anastasopoulos C, Andreopoulou AK, Kallitsis JK. Copolymers and Hybrids Based on Carbazole Derivatives and Their Nanomorphology Investigation. Nanomaterials. 2019; 9(2):133. https://doi.org/10.3390/nano9020133
Chicago/Turabian StyleAivali, Stefania, Sofia Kakogianni, Charalampos Anastasopoulos, Aikaterini K. Andreopoulou, and Joannis K. Kallitsis. 2019. "Copolymers and Hybrids Based on Carbazole Derivatives and Their Nanomorphology Investigation" Nanomaterials 9, no. 2: 133. https://doi.org/10.3390/nano9020133
APA StyleAivali, S., Kakogianni, S., Anastasopoulos, C., Andreopoulou, A. K., & Kallitsis, J. K. (2019). Copolymers and Hybrids Based on Carbazole Derivatives and Their Nanomorphology Investigation. Nanomaterials, 9(2), 133. https://doi.org/10.3390/nano9020133