Bare Iron Oxide Nanoparticles: Surface Tunability for Biomedical, Sensing and Environmental Applications
Abstract
1. Introduction
2. BIONs for Food Industry
3. BIONs for Biomedicine
4. BIONs for the Environment
5. Overall Conclusions and Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Chavali, M.S.; Nikolova, M.P. Metal oxide nanoparticles and their applications in nanotechnology. SN Appl. Sci. 2019, 1, 607–637. [Google Scholar] [CrossRef]
- Franke, M.E.; Koplin, T.J.; Simon, U. Metal and metal oxide nanoparticles in chemiresistors: Does the nanoscale matter? Small 2006, 2, 36–50. [Google Scholar] [CrossRef] [PubMed]
- Wilkes, J.S.; Zaworotko, M.J. Air and water stable 1-ethyl-3-methylimidazolium based ionic liquids. Chem. Commun. 1992, 13, 965–967. [Google Scholar] [CrossRef]
- Nuraje, N.; Su, K.; Haboosheh, A.; Samson, J.; Manning, E.P.; Yang, N.-I.; Matsui, H. Room temperature synthesis of ferroelectric barium titanate nanoparticles using peptide nanorings as templates. Adv. Mater. 2006, 18, 807–811. [Google Scholar] [CrossRef] [PubMed]
- Yahiro, J.; Oaki, Y.; Imai, H. Biomimetic synthesis of wurtzite ZnO nanowires possessing a mosaic structure. Small 2006, 2, 1183–1187. [Google Scholar] [CrossRef] [PubMed]
- Margaryan, A.A.; Liu, W. Prospects of using germanium dioxide-based glasses for optics. Opt. Eng. 1993, 32, 1995–1996. [Google Scholar] [CrossRef]
- Lee, S.-Y.; Gao, X.; Matsui, H. Biomimetic and aggregation driven crystallization route for the room-temperature material synthesis: The growth of β-Ga2O3 nanoparticles on peptide assemblies as nanoreactors. J. Am. Chem. Soc. 2007, 129, 2954–2958. [Google Scholar] [CrossRef] [PubMed]
- Klem, M.T.; Mosolf, J.; Young, M.; Douglas, T. Photochemical mineralization of europium, titanium, and iron oxyhydroxide nanoparticles in the ferritin protein cage. Inorg. Chem. 2008, 47, 2237–2239. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Zhang, D.; Fan, T.; Ding, J.; Guo, Q.; Ogawa, H. Fabrication of ZnO microtubes with adjustable nanopores on the walls by the templating of butterfly wing scales. Nanotechnology 2006, 17, 840–844. [Google Scholar] [CrossRef]
- Aizenberg, J.; Hanson, J.; Koetzle, T.F.; Weiner, S.; Addadi, L. Control of macromolecule distribution within synthetic and biogenic single calcite crystals. J. Am. Chem. Soc. 1997, 119, 881–886. [Google Scholar] [CrossRef]
- Biro, L.P.; Balint, Z.; Kertesz, K.; Vertesy, Z.; Mark, G.I.; Tapaszto, L.; Vigneron, J.P.; Lousse, V. Photonic crystal structures of biologic origin: Butterfly wing scales. Mater. Res. Soc. Symp. Proc. 2007, 1014. [Google Scholar] [CrossRef]
- Zou, D.; Xu, C.; Luo, H.; Wang, L.; Ying, T. Synthesis of Co3O4 nanoparticles via an ionic liquid-assisted methodology at room temperature. Mater. Lett. 2008, 62, 1976–1978. [Google Scholar] [CrossRef]
- Vinay, M.M.; Nayaka, Y.A. Iron oxide (Fe2O3) nanoparticles modified carbon paste electrode as an advanced material for electrochemical investigation of paracetamol and dopamine. J. Sci. Adv. Mater. Dev. 2019, 4, 442–450. [Google Scholar] [CrossRef]
- Jakubec, P.; Urbanová, V.; Medříková, Z.; Zbořil, R. Advanced sensing of antibiotics with magnetic gold nanocomposite: Electrochemical detection of chloramphenicol. Chem. Eur. J. 2016, 22, 14279–14284. [Google Scholar] [CrossRef] [PubMed]
- Manal, A.; Azab, S.M.; Hendawy, H.A. A facile nano-iron oxide sensor for the electrochemical detection of the anti-diabetic drug linagliptin in the presence of glucose and metformin. Bull. Nat. Res. Cent. 2019, 43, 95. [Google Scholar]
- Xie, J.; Jon, S. Magnetic nanoparticle-based theranostics. Theranostics 2012, 2, 122–124. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.H.; Zhang, S.Q.; Chen, X.L.; Zhuang, Z.X.; Xu, J.G.; Wang, X.R. Magnetite-containing spherical silica nanoparticles for biocatalysis and bioseparations. Anal. Chem. 2004, 76, 1316–1321. [Google Scholar] [CrossRef] [PubMed]
- Ito, A.; Shinkai, M.; Honda, H.; Kobayashi, T. Medical application of functionalized magnetic nanoparticles. J. Biosci. Bioeng. 2005, 100, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Matsunaga, T. Fully automated chemiluminescence immunoassay of insulin using antibody-protein A-bacterial magnetic particle complexes. Anal. Chem. 2000, 72, 3518–3522. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Liu, Y.; Yang, H.; Yang, Y.; Shen, G.; Yu, R. A phenol biosensor based on immobilizing tyrosinase to modified core-shell magnetic nanoparticles supported at a carbon paste electrode. Anal. Chim. Acta 2005, 533, 3–9. [Google Scholar] [CrossRef]
- Neuberger, T.; Schopf, B.; Hofmann, H.; Hofmann, M.; Rechenberg, B.V. Superparamagnetic nanoparticles for biomedical applications: Possibilities and limitations of a new drug delivery system. J. Magn. Magn. Mater. 2005, 293, 483–496. [Google Scholar] [CrossRef]
- Saito, S.; Tsugeno, M.; Koto, D.; Mori, Y.; Yoshioka, Y.; Nohara, S.; Murase, K. Impact of surface coating and particle size on the uptake of small and ultrasmall superparamagnetic iron oxide nanoparticles by macrophages. Int. J. Nanomed. 2012, 7, 5415–5421. [Google Scholar]
- Tassa, C.; Shaw, S.Y.; Weissleder, R. Dextran-coated iron oxide nanoparticles: A versatile platform for targeted molecular imaging, molecular diagnostics, and therapy. Acc. Chem. Res. 2011, 44, 842–852. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Huang, Y.; David, A.E.; Chertok, B.; Zhang, L.; Yu, F.; Yang, V.C. Magnetic nanoparticles for MRI of brain tumors. Curr. Pharm. Biotechnol. 2012, 13, 2403–2416. [Google Scholar] [CrossRef] [PubMed]
- Tran, P.H.-L.; Tran, T.T.-D.; Vo, T.V.; Lee, B.-J. Promising iron oxide-based magnetic nanoparticles in biomedical engineering. Arch. Pharm. Res. 2012, 35, 2045–2061. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, Y.; Usuki, N.; Matsuo, K.; Kishimoto, M. Development of NanoCAP technology for high-density recording. IEEE Trans. Magn. 2005, 41, 3241–3243. [Google Scholar] [CrossRef]
- Fouda, M.F.R.; El-Kholy, M.B.; Moustafa, S.A.; Hussien, A.I.; Wahba, M.A.; El-Shahat, M.F. Synthesis and characterization of nanosized Fe2O3 pigments. Int. J. Inorg. Chem. 2012, 2012, 989281. [Google Scholar] [CrossRef]
- Magro, M.; Baratella, D.; Pianca, N.; Toninello, A.; Grancara, S.; Zboril, R.; Vianello, F. Electrochemical determination of hydrogen peroxide production by isolated mitochondria: A novel nanocomposite carbon–maghemite nanoparticle electrode. Sens. Actuator B Chem. 2013, 176, 315–322. [Google Scholar] [CrossRef]
- Laurent, S.; Forge, D.; Port, M.; Roch, A.; Robic, C.; Vander Elst, L.; Muller, R.N. Magnetic iron oxide nanoparticles: Synthesis, stabilization, vectorization, physicochemical characterizations, and biological applications. Chem. Rev. 2008, 108, 2064–2110. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, N.; Fessi, H.; Elaissari, A. Theranostic applications of nanoparticles in cancer. Drug Discov. Today 2012, 17, 928–934. [Google Scholar] [CrossRef] [PubMed]
- Soenen, S.J.; Hodenius, M.; De Cuyper, M. Magnetoliposomes: Versatile innovative nanocolloids for use in biotechnology and biomedicine. Nanomedicine 2009, 4, 177–191. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Insin, N.; Lee, J.; Han, H.S.; Cordero, J.M.; Liu, W.; Bawendi, M.G. Compact zwitterion-coated iron oxide nanoparticles for biological applications. Nano Lett. 2012, 12, 22–25. [Google Scholar] [CrossRef] [PubMed]
- Wahajuddin, S.S.P.; Arora, S. Superparamagnetic iron oxide nanoparticles: Magnetic nanoplatforms as drug carriers. Int. J. Nanomed. 2012, 7, 3445–3471. [Google Scholar] [CrossRef] [PubMed]
- Meyers, S.R.; Grinstaff, M.W. Biocompatible and bioactive surface modifications for prolonged in vivo efficacy. Chem. Rev. 2012, 112, 1615–1632. [Google Scholar] [CrossRef] [PubMed]
- Hudson, R.; Feng, Y.; Varma, R.S.; Moores, A. Bare magnetic nanoparticles: Sustainable synthesis and applications in catalytic organic transformations. Green Chem. 2014, 16, 4493–4505. [Google Scholar] [CrossRef]
- Schwaminger, S.P.; Fraga-García, P.; Blank-Shim, S.A.; Straub, T.; Haslbeck, M.; Muraca, F.; Dawson, K.A.; Berensmeier, S. Magnetic one-Step purification of his-tagged protein by bare iron oxide nanoparticles. ACS Omega 2019, 4, 3790–3799. [Google Scholar] [CrossRef] [PubMed]
- Schwaminger, S.P.; Blank-Shim, S.A.; Scheifele, I.; Pipich, V.; Fraga-García, P.; Berensmeier, S. Design of interactions between nanomaterials and proteins: A highly affine peptide tag to bare iron oxide nanoparticles for magnetic protein separation. Biotechnol. J. 2019, 14, 1800055. [Google Scholar] [CrossRef] [PubMed]
- Fraga-García, P.; Kubbutat, P.; Brammen, M.; Schwaminger, S.; Berensmeier, S. Bare iron oxide nanoparticles for magnetic harvesting of microalgae: From interaction behavior to process realization. Nanomaterials 2018, 8, 292. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.K.; Stiles, A.R.; Guo, C.; Liu, C.Z. Harvesting microalgae by magnetic separation: A review. Algal Res. 2015, 9, 178–185. [Google Scholar] [CrossRef]
- Xu, L.; Guo, C.; Wang, F.; Zheng, S.; Liu, C.Z. A simple and rapid harvesting method for microalgae by in situ magnetic separation. Bioresour. Technol. 2011, 102, 10047–10051. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.C.; Lee, K.; Oh, Y.K. Recent nanoparticle engineering advances in microalgal cultivation and harvesting processes of biodiesel production: A review. Bioresour. Technol. 2015, 184, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Safarik, I.; Pospiskova, K.; Baldikova, E. Magnetic particles for microalgae separation and biotechnology. In Food Bioactives; Puri, M., Ed.; Springer: Cham, Switzerland, 2017; pp. 153–169. [Google Scholar]
- Prochazkova, G.; Safarik, I.; Branyik, T. Harvesting microalgae with microwave synthesized magnetic microparticles. Bioresour. Technol. 2013, 130, 472–477. [Google Scholar] [CrossRef] [PubMed]
- Toh, P.Y.; Ng, B.W.; Ahmad, A.L.; Chieh, D.C.J.; Lim, J. The role of particle-to-cell interactions in dictating nanoparticle aided magnetophoretic separation of microalgal cells. Nanoscale 2014, 6, 12838–12848. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.; Lu, D.; Liu, Z. Removal of Arsenic Contaminants with Magnetic γ-Fe2O3 Nanoparticles. Chem. Eng. J. 2012, 211, 46–52. [Google Scholar] [CrossRef]
- Liu, R.; Liu, J.F.; Zhang, L.Q.; Sun, J.F.; Jiang, G.B. Low temperature synthesized ultrathin γ-Fe2O3 nanosheets show similar adsorption behavior for As(III) and As(V). J. Mater. Chem. A 2016, 4, 7606–7614. [Google Scholar] [CrossRef]
- Rana, S.; Yeh, Y.C.; Rotello, V.M. Engineering the nanoparticle protein interface: Applications and possibilities. Curr. Opin. Chem. Biol. 2010, 14, 828–834. [Google Scholar] [CrossRef] [PubMed]
- Velusamy, V.; Arshak, K.; Korostynska, O.; Oliwa, K.; Adley, C. An overview of foodborne pathogen detection: In the perspective of biosensors. Biotechnol. Adv. 2010, 28, 232–254. [Google Scholar] [CrossRef] [PubMed]
- Hierlemann, A.; Gutierrez-Osuna, R. Higher-order chemical sensing. Chem. Rev. 2008, 108, 563–613. [Google Scholar] [CrossRef] [PubMed]
- Urbanova, V.; Magro, M.; Gedanken, A.; Baratella, D.; Vianello, F.; Zboril, R. Nanocrystalline Iron Oxides, Composites, and Related Materials as a Platform for Electrochemical, Magnetic, and Chemical Biosensors. Chem. Mater. 2014, 26, 6653–6673. [Google Scholar] [CrossRef]
- Baratella, D.; Magro, M.; Jakubec, P.; Bonaiuto, E.; de Almeida Roger, J.; Gerotto, E.; Zoppellaro, G.; Tucek, J.; Safarova, K.C.; Zboril, R.; et al. Electrostatically stabilized hybrids of carbon and maghemite nanoparticles: Electrochemical study and application. Phys. Chem. Chem. Phys. 2017, 19, 11668–11677. [Google Scholar] [CrossRef] [PubMed]
- Magro, M.; Baratella, D.; Miotto, G.; Frömmel, J.; Šebela, M.; Kopečná, M.; Agostinelli, E.; Vianello, F. Enzyme self-assembly on naked iron oxide nanoparticles for aminoaldehyde biosensing. Amino Acids 2019, 51, 679–690. [Google Scholar] [CrossRef] [PubMed]
- Baratella, D.; Magro, M.; Sinigaglia, G.; Zboril, R.; Salviulo, G.; Vianello, F. A glucose biosensor based on surface active maghemite nanoparticles. Biosens. Bioelectron. 2013, 45, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Magro, M.; Baratella, D.; Colò, V.; Vallese, F.; Nicoletto, C.; Santagata, S.; Sambo, P.; Molinari, S.; Salviulo, G.; Venerando, A.; et al. Electrocatalytic Nanostructured Ferric Tannates as platform for enzyme conjugation: Electrochemical determination of phenolic compounds. Bioelectrochemistry 2019. [Google Scholar] [CrossRef]
- Shinde, S.B.; Fernandes, C.B.; Patravale, V.B. Recent trends in in-vitro nanodiagnostics for detection of pathogens. J. Control. Release 2012, 159, 164–180. [Google Scholar] [CrossRef] [PubMed]
- Sanvicens, N.; Pastells, C.; Pascual, N.; Marco, M.P. Nanoparticle based biosensors for detection of pathogenic bacteria. TrAC Trends Anal. Chem. 2009, 28, 1243–1252. [Google Scholar] [CrossRef]
- Bonaiuto, E.; Magro, M.; Fasolato, L.; Novelli, E.; Shams, S.; Piccirillo, A.; Bakhshi, B.; Moghadam, T.T.; Baratella, D.; Vianello, F. Versatile nano-platform for tailored immuno-magnetic carriers. Anal. Bioanal. Chem. 2018, 410, 7575–7589. [Google Scholar] [CrossRef] [PubMed]
- Martin, N.H.; Murphy, S.C.; Ralyea, R.D.; Wiedmann, M.; Boor, K.J. When cheese gets the blues: Pseudomonas fluorescens as the causative agent of cheese spoilage. J. Dairy Sci. 2011, 94, 3176–3183. [Google Scholar] [CrossRef] [PubMed]
- Andreani, N.A.; Martino, M.E.; Fasolato, L.; Carraro, L.; Montemurro, F.; Mioni, R.; Bordin, P.; Cardazzo, B. Tracking the blue: A MLST approach to characterize the Pseudomonas fluorescens group. Food Microbiol. 2014, 39, 116–126. [Google Scholar] [CrossRef] [PubMed]
- Chierici, M.; Picozzi, C.; La Spina, M.G.; Orsi, C.; Vigentini, I.; Zambrini, V.; Foschino, R. Strain diversity of Pseudomonas fluorescens group with potential blue pigment phenotype isolated from dairy products. J. Food Prot. 2016, 79, 1430–1435. [Google Scholar] [CrossRef] [PubMed]
- Andreani, N.A.; Carraro, L.; Martino, M.E.; Fondi, M.; Fasolato, L.; Miotto, G.; Magro, M.; Vianello, F.; Cardazzo, B. A genomic and transcriptomic approach to investigate the blue pigment phenotype in Pseudomonas fluorescens. Int. J. Food Microbiol. 2015, 213, 88–98. [Google Scholar] [CrossRef] [PubMed]
- Andreani, N.A.; Carraro, L.; Zhang, L.; Vos, M.; Cardazzo, B. Transposon mutagenesis in Pseudomonas fluorescens reveals genes involved in blue pigment production and antioxidant protection. Food Microbiol. 2019, 82, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Magro, M.; Baratella, D.; Jakubec, P.; Corraducci, V.; Fasolato, L.; Cardazzo, B.; Novelli, E.; Zoppellaro, G.; Zboril, R.; Vianello, F. H2O2 Tolerance in Pseudomonas fluorescens: Synergy between Pyoverdine-Iron(III) Complex and a Blue Extracellular Product by a Nanotechnology based Electrochemical Approach. ChemElectroChem. 2019. [Google Scholar] [CrossRef]
- Kiran-Kumar, P.; Badarinath, V.; Halami, P. Isolation of anti-listerial bacteriocin producing Lactococcus lactis CFR-B3 from Beans (Phaseolus vulgaris). Internet J. Microbiol. 2008, 6, 1–6. [Google Scholar]
- Magro, M.; Bonaiuto, E.; Baratella, D.; de Almeida Roger, J.; Jakubec, P.; Corraducci, V.; Tucek, J.; Malina, O.; Zboril, R.; Vianello, F. Electrocatalytic nanostructured ferric tannates: Characterization and application of a polyphenol nanosensor. ChemPhysChem 2016, 17, 3196–3203. [Google Scholar] [CrossRef] [PubMed]
- De Almeida Roger, J.; Magro, M.; Spagnolo, S.; Bonaiuto, E.; Baratella, D.; Fasolato, L.; Vianello, F. Antimicrobial and magnetically removable tannic acid nanocarrier: A processing aid for Listeria monocytogenes treatment for food industry applications. Food Chem. 2018, 267, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Hadjidemetriou, M.; Al-Ahmady, Z.; Buggio, M.; Swift, J.; Kostarelos, K. A novel scavenging tool for cancer biomarker discovery based on the blood-circulating nanoparticle protein corona. Biomaterials 2019, 188, 118–129. [Google Scholar] [CrossRef] [PubMed]
- Miotto, G.; Magro, M.; Terzo, M.; Zaccarin, M.; Da Dalt, L.; Bonaiuto, E.; Baratella, D.; Gabai, G.; Vianello, F. Protein corona as a proteome fingerprint: The example of hidden biomarkers for cow mastitis. Colloids Surf. B Biointerfaces 2016, 140, 40–49. [Google Scholar] [CrossRef] [PubMed]
- Magro, M.; Zaccarin, M.; Miotto, G.; Da Dalt, L.; Baratella, D.; Fariselli, P.; Gabai, G.; Vianello, F. Analysis of hard protein corona composition on selective iron oxide nanoparticles by MALDI-TOF mass spectrometry: Identification and amplification of a hidden mastitis biomarker in milk proteome. Anal. Bioanal. Chem. 2018, 410, 2949–2959. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, M.I.; Magro, M.; Ming, L.C.; da Silva, M.B.; Rodrigues, L.F.O.S.; do Prado, D.Z.; Bonaiuto, E.; Baratella, D.; Lima, G.P.P.; de Almeida Roger, J.; et al. Sustainable production of high purity curcuminoids from Curcuma longa by magnetic nanoparticles: A case study in Brazil. J. Clean. Prod. 2017, 154, 233–241. [Google Scholar] [CrossRef]
- Magro, M.; Esteves Moritz, D.; Bonaiuto, E.; Baratella, D.; Terzo, M.; Jakubec, P.; Malina, O.; Cépe, K.; de Falcao Aragao, G.M.; Zboril, R.; et al. Citrinin mycotoxin recognition and removal by naked magnetic nanoparticles. Food Chem. 2016, 203, 505–512. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Li, W.; Han, P.; Zhou, X.; Cheng, J.; Wen, H.; Xue, W. Carbon quantum dots: Synthesis, properties, and sensing applications as a potential clinical analytical method. Anal. Methods 2019, 11, 2240–2258. [Google Scholar] [CrossRef]
- Dykman, L.A.; Khlebtsov, N.G. Gold Nanoparticles in Biology and Medicine: Recent Advances and Prospects. Acta Naturae 2011, 3, 34–55. [Google Scholar] [CrossRef] [PubMed]
- Baetke, S.C.; Lammers, T.; Kiessling, F. Applications of nanoparticles for diagnosis and therapy of cancer. Br. J. Radiol. 2015, 88, 20150207. [Google Scholar] [CrossRef] [PubMed]
- Elsabahy, M.; Wooley, K.L. Design of polymeric nanoparticles for biomedical delivery applications. Chem. Soc. Rev. 2012, 41, 2545–2561. [Google Scholar] [CrossRef] [PubMed]
- Capeletti, L.B.; Loiola, L.M.D.; Picco, A.S.; da Silva Liberato, M.; Cardoso, M.B. Silica Nanoparticle Applications in the Biomedical Field. In Smart Nanoparticles for Biomedicine; Elsevier: Amsterdam, The Netherlands, 2018; pp. 115–129. [Google Scholar]
- Shirshahi, V.; Soltani, M. Solid silica nanoparticles: Applications in molecular imaging. Contrast Media Mol. Imaging 2015, 10, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhao, Q.; Han, N.; Bai, L.; Li, J.; Liu, J.; Che, E.; Hu, L.; Zhang, Q.; Jiang, T.; et al. Mesoporous silica nanoparticles in drug delivery and biomedical applications. Nanomed. Nanotechnol. Biol. Med. 2015, 11, 313–327. [Google Scholar] [CrossRef] [PubMed]
- Braun, K.; Stürzel, C.M.; Biskupek, J.; Kaiser, U.; Kirchhoff, F.; Lindén, M. Comparison of different cytotoxicity assays for in vitro evaluation of mesoporous silica nanoparticles. Toxicol. In Vitro 2018, 52, 214–221. [Google Scholar] [CrossRef] [PubMed]
- Tamba, B.I.; Dondas, A.; Leon, M.; Neagu, A.N.; Dodi, G.; Stefanescu, C.; Tijani, A. Silica nanoparticles: Preparation, characterization and in vitro/in vivo biodistribution studies. Eur. J. Pharm. Sci. 2015, 71, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Colombo, M.; Carregal-Romero, S.; Casula, M.F.; Gutierrez, L.; Morales, M.P.; Böhm, I.B.; Heverhagen, J.T.; Prosperi, D.; Parak, W.J. Biological applications of magnetic nanoparticles. Chem. Soc. Rev. 2012, 41, 4306–4334. [Google Scholar] [CrossRef] [PubMed]
- Basso, C.R.; Crulhas, B.P.; Magro, M.; Vianello, F.; Pedrosa, V.A. A new immunoassay of hybrid nanomater conjugated to aptamers for the detection of dengue virus. Talanta 2019, 197, 482–490. [Google Scholar] [CrossRef] [PubMed]
- Venerando, R.; Miotto, G.; Magro, M.; Dallan, M.; Baratella, D.; Bonaiuto, E.; Zboril, R.; Vianello, F. Magnetic Nanoparticles with Covalently Bound Self-Assembled Protein Corona for Advanced Biomedical Applications. J. Phys. Chem. C 2013, 117, 20320–20331. [Google Scholar] [CrossRef]
- Skopalik, J.; Polakova, K.; Havrdova, M.; Justan, I.; Magro, M.; Milde, D.; Knopfova, L.; Smarda, J.; Polakova, H.; Gabrielova, E.; et al. Mesenchymal stromal cell labeling by new uncoated superparamagnetic maghemite nanoparticles in comparison with commercial Resovist—An initial in vitro study. Int. J. Nanomed. 2014, 9, 5355–5372. [Google Scholar] [CrossRef] [PubMed]
- Cmiel, V.; Skopalik, J.; Polakova, K.; Solar, J.; Havrdova, M.; Milde, D.; Justan, I.; Magro, M.; Starcuk, Z.; Provaznik, I. Rhodamine bound maghemite as a long-term dual imaging nanoprobe of adipose tissue-derived mesenchymal stromal cells. Eur. Biophys. 2017, 46, 433–444. [Google Scholar] [CrossRef] [PubMed]
- Magro, M.; Campos, R.; Baratella, D.; Lima, G.P.P.; Hola, K.; Divoky, C.; Stollberger, R.; Malina, O.; Aparicio, C.; Zoppellaro, G.; et al. A Magnetically Drivable Nanovehicle for Curcumin with Antioxidant Capacity and MRI Relaxation Properties. Chem. Eur. J. 2014, 20, 11913–11920. [Google Scholar] [CrossRef] [PubMed]
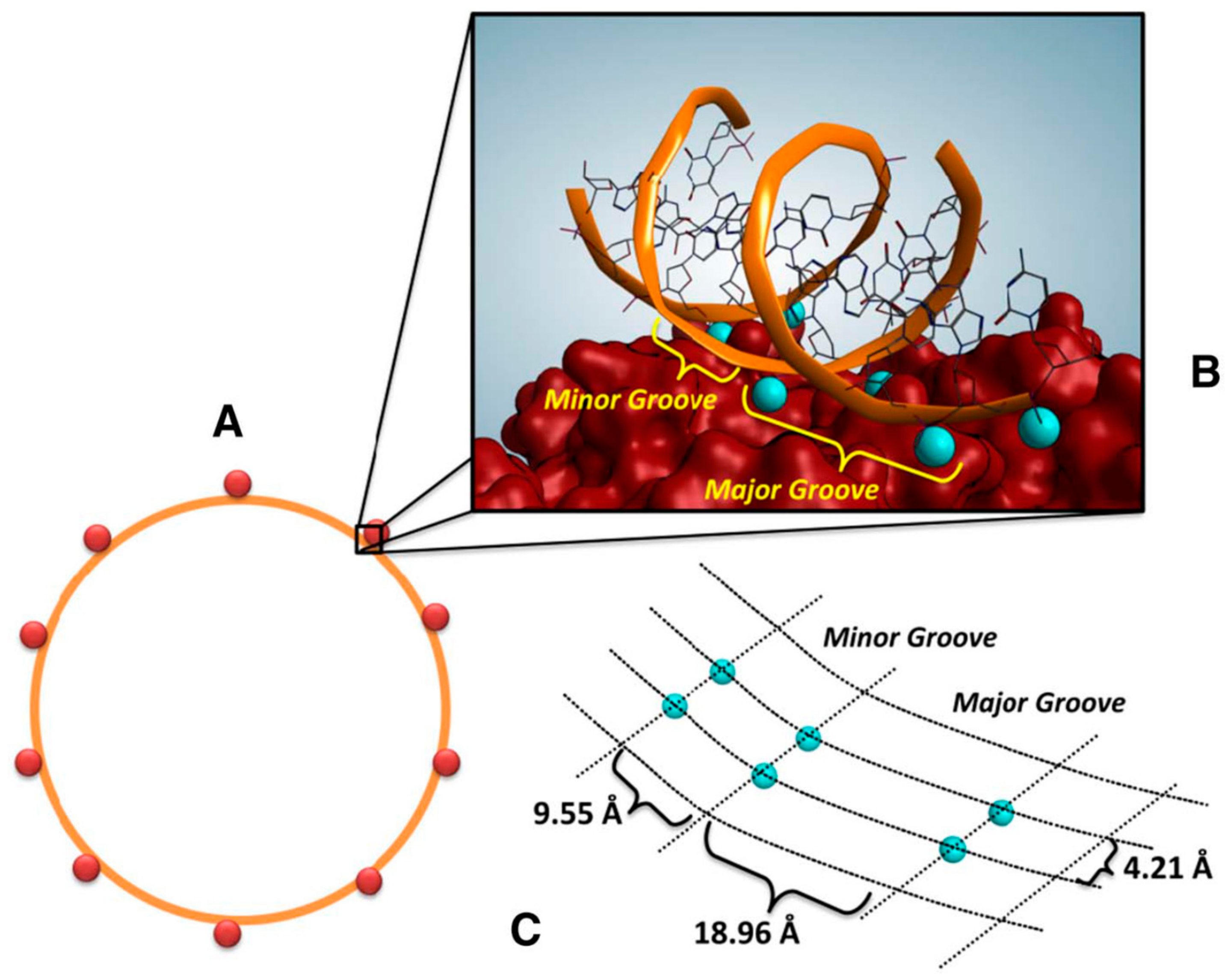
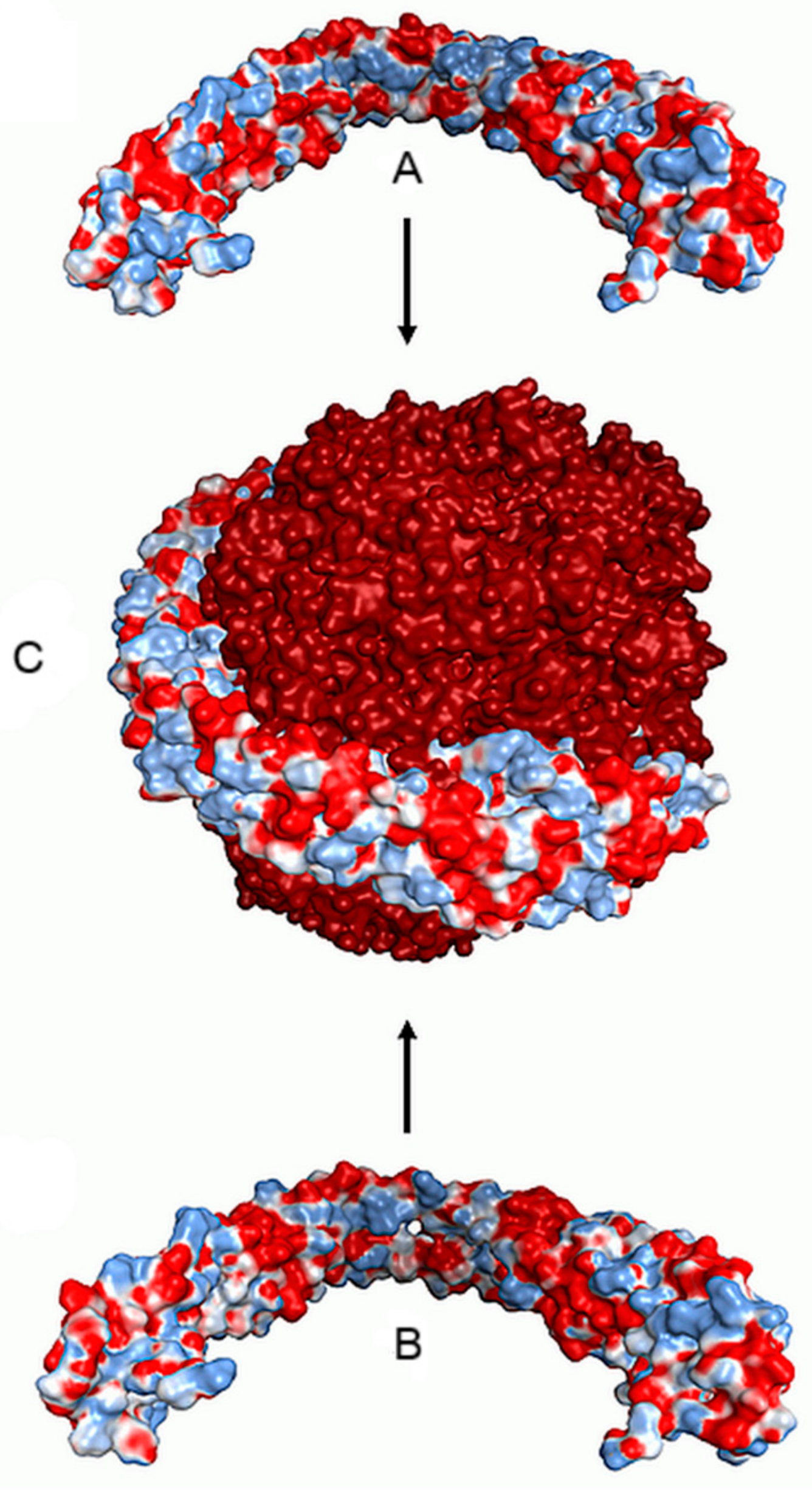
- Magro, M.; Martinello, T.; Bonaiuto, E.; Gomiero, C.; Baratella, D.; Zoppellaro, G.; Cozza, G.; Patruno, M.; Zboril, R.; Vianello, F. Covalently bound DNA on naked iron oxide nanoparticles: Intelligent colloidal nano-vector for cell transfection. Biochim. Biophys. Acta Gen. Sub. 2017, 1861, 2802–2810. [Google Scholar] [CrossRef] [PubMed]
- Venerando, A.; Magro, M.; Baratella, D.; Ugolotti, J.; Zanin, S.; Malina, O.; Zboril, R.; Lin, H.; Vianello, F. Biotechnological applications of nanostructured hybrids of polyamine carbon quantum dots and iron oxide nanoparticles. Amino Acids 2019, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Anderson, C.J.; Bulte, J.W.; Chen, K.; Chen, X.; Khaw, B.A.; Shokeen, M.; Wooley, K.L.; VanBrocklin, H.F. Design of targeted cardiovascular molecular imaging probes. J. Nucl. Med. 2010, 51, 3S–17S. [Google Scholar] [CrossRef] [PubMed]
- Kanwar, J.R.; Roy, K.; Kanwar, R.K. Chimeric aptamers in cancer cell-targeted drug delivery. Crit. Rev. Biochem. Mol. Biol. 2011, 46, 459–477. [Google Scholar] [CrossRef] [PubMed]
- Kubinova, S.; Sykova, E. Nanotechnologies in regenerative medicine. Minim. Invasive Ther. Allied Technol. 2010, 19, 144–156. [Google Scholar] [CrossRef] [PubMed]
- Kiessling, F.; Huppert, J.; Zhang, C.; Jayapaul, J.; Zwick, S.; Woenne, E.C.; Mueller, M.M.; Zentgraf, H.; Eisenhut, M.; Addadi, Y.; et al. RGD-labeled USPIO inhibits adhesion and endocytotic activity of alpha v beta3-integrin-expressing glioma cells and only accumulates in the vascular tumor compartment. Radiology 2009, 253, 462–469. [Google Scholar] [CrossRef] [PubMed]
- Dias, A.M.; Hussain, A.; Marcos, A.S.; Roque, A.C. A biotechnological perspective on the application of iron oxide magnetic colloids modified with polysaccharides. Biotechnol. Adv. 2011, 29, 142–155. [Google Scholar] [CrossRef] [PubMed]
- Chemello, G.; Piccinetti, C.; Randazzo, B.; Carnevali, O.; Maradonna, F.; Magro, M.; Bonaiuto, E.; Vianello, F.; Pasquaroli, S.; Radaelli, G.; et al. Oxytetracycline delivery in adult female zebrafish by iron oxide nanoparticles. Zebrafish 2016, 13, 495–503. [Google Scholar] [CrossRef] [PubMed]
- Magro, M.; Baratella, D.; Bonaiuto, E.; de Almeida Roger, J.; Chemello, G.; Pasquaroli, S.; Mancini, I.; Olivotto, I.; Zoppellaro, G.; Ugolotti, J.; et al. Stealth iron oxide nanoparticles for organotropic drug targeting. Biomacromolecules 2019, 20, 1375–1384. [Google Scholar] [CrossRef] [PubMed]
- Chemello, G.; Randazzo, B.; Zarantoniello, M.; Fifi, A.P.; Aversa, S.; Ballarin, C.; Radaelli, G.; Magro, M.; Olivotto, I. Safety assessment of antibiotic administration by magnetic nanoparticles in in vitro zebrafish liver and intestine cultures. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2019, 108559. [Google Scholar] [CrossRef] [PubMed]
- Skjolding, L.M.; Sørensen, S.N.; Hartmann, N.B.; Hjorth, R.; Hansen, S.F.; Baun, A. A critical review of aquatic ecotoxicity testing of nanoparticles—The quest for disclosing nanoparticle effects. Angew. Chem. Int. Ed. 2016, 55, 15224–15239. [Google Scholar] [CrossRef] [PubMed]
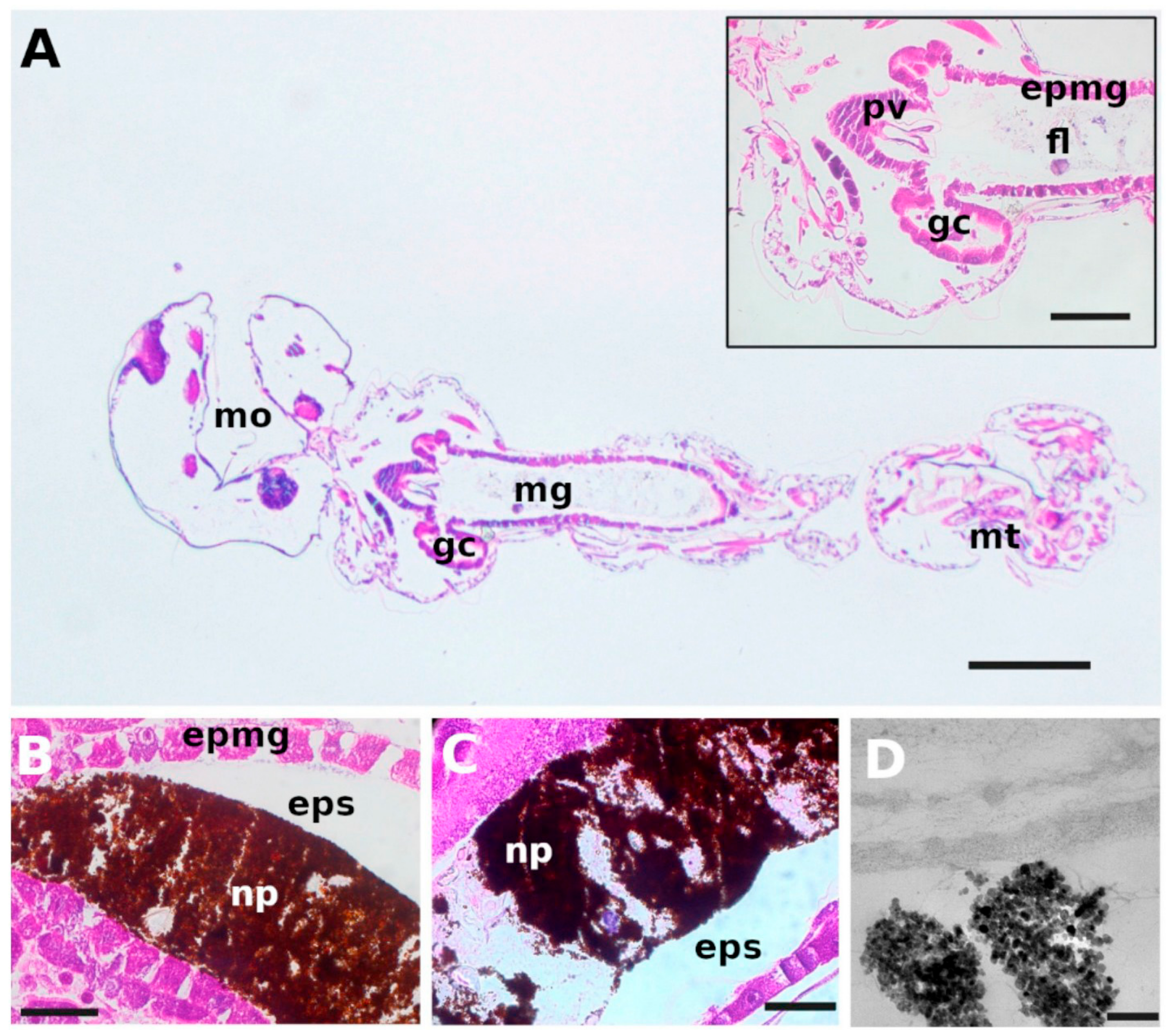
- Magro, M.; De Liguoro, M.; Franzago, E.; Baratella, D.; Vianello, F. The surface reactivity of iron oxide nanoparticles as a potential hazard for aquatic environments: A study on Daphnia magna adults and embryos. Sci. Rep. 2018, 8, 13017. [Google Scholar] [CrossRef] [PubMed]
- Magro, M.; Bramuzzo, S.; Baratella, D.; Ugolotti, J.; Zoppellaro, G.; Chemello, G.; Olivotto, I.; Ballarin, C.; Radaelli, G.; Arcaro, B.; et al. Self-assembly of chlorin-e6 on γ-Fe2O3 nanoparticles: Application for larvicidal activity against Aedes aegypti. J. Photochem. Photobiol. B Biol. 2019, 194, 21–31. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.C.N.; Lo, I.M.C. Magnetic nanoparticles: Essential factors for sustainable environmental applications. Water Res. 2013, 47, 2613–2632. [Google Scholar] [CrossRef] [PubMed]
- Plachtová, P.; Medříková, Z.; Zbořil, R.; Tuček, J.; Varma, R.S.; Maršálek, B. Iron and iron oxide nanoparticles synthesized with green tea extract: Differences in ecotoxicological profile and ability to degrade malachite green. ACS Sustain. Chem. Eng. 2018, 6, 8679–8687. [Google Scholar] [CrossRef] [PubMed]
- Markova, Z.; Novak, P.; Kaslik, J.; Plachtova, P.; Brazdova, M.; Jancula, D.; Siskova, K.M.; Machala, L.; Marsalek, B.; Zboril, R.; et al. Iron(II,III)-polyphenol complex nanoparticles derived from green tea with remarkable ecotoxicological impact. ACS Sustain. Chem. Eng. 2014, 2, 1674–1680. [Google Scholar] [CrossRef]
- Mwilu, S.K.; Siska, E.; Baig, R.B.N.; Varma, R.S.; Heithmar, E.; Rogers, K.R. Separation and measurement of silver nanoparticles and silver ions using magnetic particles. Sci. Total Environ. 2014, 472, 316–323. [Google Scholar] [CrossRef] [PubMed]
- Coyne, R.; Smith, P.; Moriarty, C. The fate of oxytetracycline in the marine environment of a salmon cage farm, Marine Environment and Health Series No. 3, Marine Institute 2001. Mar. Environ. Health Ser. 2001, 3, 1–24. [Google Scholar]
- Wise, R. Antimicrobial resistance: Priorities for action. J. Antimicrob. Chemother. 2002, 49, 585–586. [Google Scholar] [CrossRef] [PubMed]
- De La Torre, A.; Iglesias, I.; Carballo, M.; Ramírez, P.; Muñoz, M.J. An approach for mapping the vulnerability of European Union soils to antibiotic contamination. Sci. Total Environ. 2012, 414, 672–679. [Google Scholar] [CrossRef] [PubMed]
- Rakshit, S.; Sarkar, D.; Elzinga, E.; Punamiya, P.; Datta, R. Surface complexation of oxytetracycline by magnetite: Effect of solution properties. Vadose Zone J. 2014, 13. [Google Scholar] [CrossRef]
- Figueroa, R.A.; Mackay, A.A. Sorption of oxytetracycline to iron oxides and iron oxide-rich soils. Environ. Sci. Technol. 2005, 39, 6664–6671. [Google Scholar] [CrossRef] [PubMed]
- Ihara, I.; Toyoda, K.; Beneragama, N.; Umetsu, K. Magnetic separation of antibiotics by electrochemical magnetic seeding. J. Phys. Conf. Ser. 2009, 156, 012034. [Google Scholar] [CrossRef]
- Xiao, L.; Li, J.; Brougham, D.F.; Fox, E.K.; Feliu, N.; Bushmelev, A.; Schmidt, A.; Mertens, N.; Kiessling, F.; Valldor, M.; et al. Water-soluble superparamagnetic magnetite nanoparticles with biocompatible coating for enhanced magnetic resonance imaging. ACS Nano 2011, 5, 6315–6324. [Google Scholar] [CrossRef] [PubMed]
- Magro, M.; Baratella, D.; Molinari, S.; Venerando, A.; Salviulo, G.; Chemello, G.; Olivotto, I.; Zoppellaro, G.; Ugolotti, J.; Aparicio, C.; et al. Biologically safe colloidal suspensions of naked iron oxide nanoparticles for in situ antibiotic suppression. Colloids Surf. B Biointerfaces 2019, 181, 102–111. [Google Scholar] [CrossRef] [PubMed]
- Magro, M.; Domeneghetti, S.; Baratella, D.; Jakubec, P.; Salviulo, G.; Bonaiuto, E.; Venier, P.; Malina, O.; Tuček, J.; Ranc, V.; et al. Colloidal Surface Active Maghemite Nanoparticles for biologically safe CrVI remediation: From core-shell nanostructures to pilot plant development. Chem. Eur. J. 2016, 22, 14219–14226. [Google Scholar] [CrossRef] [PubMed]
- Colino, C.; Millán, C.; Lanao, J. Nanoparticles for signaling in biodiagnosis and treatment of infectious diseases. Int. J. Mol. Sci. 2018, 19, 1627. [Google Scholar] [CrossRef] [PubMed]
- Cho, I.H.; Ku, S. Current technical approaches for the early detection of foodborne pathogens: Challenges and opportunities. Int. J. Mol. Sci. 2017, 18, 2078. [Google Scholar] [CrossRef] [PubMed]
- Magro, M.; Baratella, D.; Bonaiuto, E.; de Almeida Roger, J.; Vianello, F. New Perspectives on Biomedical Applications of Iron Oxide Nanoparticles. Curr. Med. Chem. 2017, 25, 540–555. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi, M.; Lynch, I.; Ejtehadi, M.R.; Monopoli, M.P.; Baldelli Bombelli, F.; Laurent, S. Protein−Nanoparticle Interactions: Opportunities and Challenges. Chem. Rev. 2011, 111, 5610–5637. [Google Scholar] [CrossRef] [PubMed]
- Jersmann, H.P.; Dransfield, I.; Hart, S.P. Fetuin/Alpha2-HS Glycoprotein Enhances Phagocytosis of Apoptotic Cells and Macropinocytosis by Human Macrophages. Clin. Sci. 2003, 105, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Cedervall, T.; Lynch, I.; Lindman, S.; Berggård, T.; Thulin, E.; Nilsson, H.; Dawson, K.A.; Linse, S. Understanding the nanoparticle–protein corona using methods to quantify exchange rates and affinities of proteins for nanoparticles. Proc. Natl. Acad. Sci. USA 2007, 104, 2050–2055. [Google Scholar] [CrossRef] [PubMed]
- Garcia, J.; Zhang, Y.; Taylor, H.; Cespedes, O.; Webb, M.E.; Zhou, D. Multilayer Enzyme-Conjugated Magnetic Nanoparticles as Efficient, Reusable Biocatalysts and Biosensors. Nanoscale 2001, 3, 3721–3730. [Google Scholar] [CrossRef] [PubMed]
- Niemirowicz, K.; Markiewicz, K.H.; Wilczewska, A.Z.; Car, H. Magnetic nanoparticles as new diagnostic tools in medicine. Adv. Med. Sci. 2012, 57, 196–207. [Google Scholar] [CrossRef] [PubMed]
- Mirshafiee, V.; Mahmoudi, M.; Lou, K.; Cheng, J.; Kraft, M.L. Protein corona significantly reduces active targeting yield. Chem. Commun. 2013, 49, 2557–2559. [Google Scholar] [CrossRef] [PubMed]
- Salvati, A.; Pitek, A.S.; Monopoli, M.P.; Prapainop, K.; Bandelli Bombelli, F.; Hristov, D.R.; Kelly, P.M.; Åberg, C.; Mahon, E.; Dawson, K.A. Transferrin-functionalized nanoparticles lose their targeting capabilities when a biomolecule corona adsorbs on the surface. Nat. Nanotechnol. 2013, 8, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Laurent, S.; Burtea, C.; Thirifays, C.; Rezaee, F.; Mahmoudi, M. Significance of cell “observer” and protein source in nanobiosciences. J. Colloid Interface Sci. 2013, 392, 431–445. [Google Scholar] [CrossRef] [PubMed]
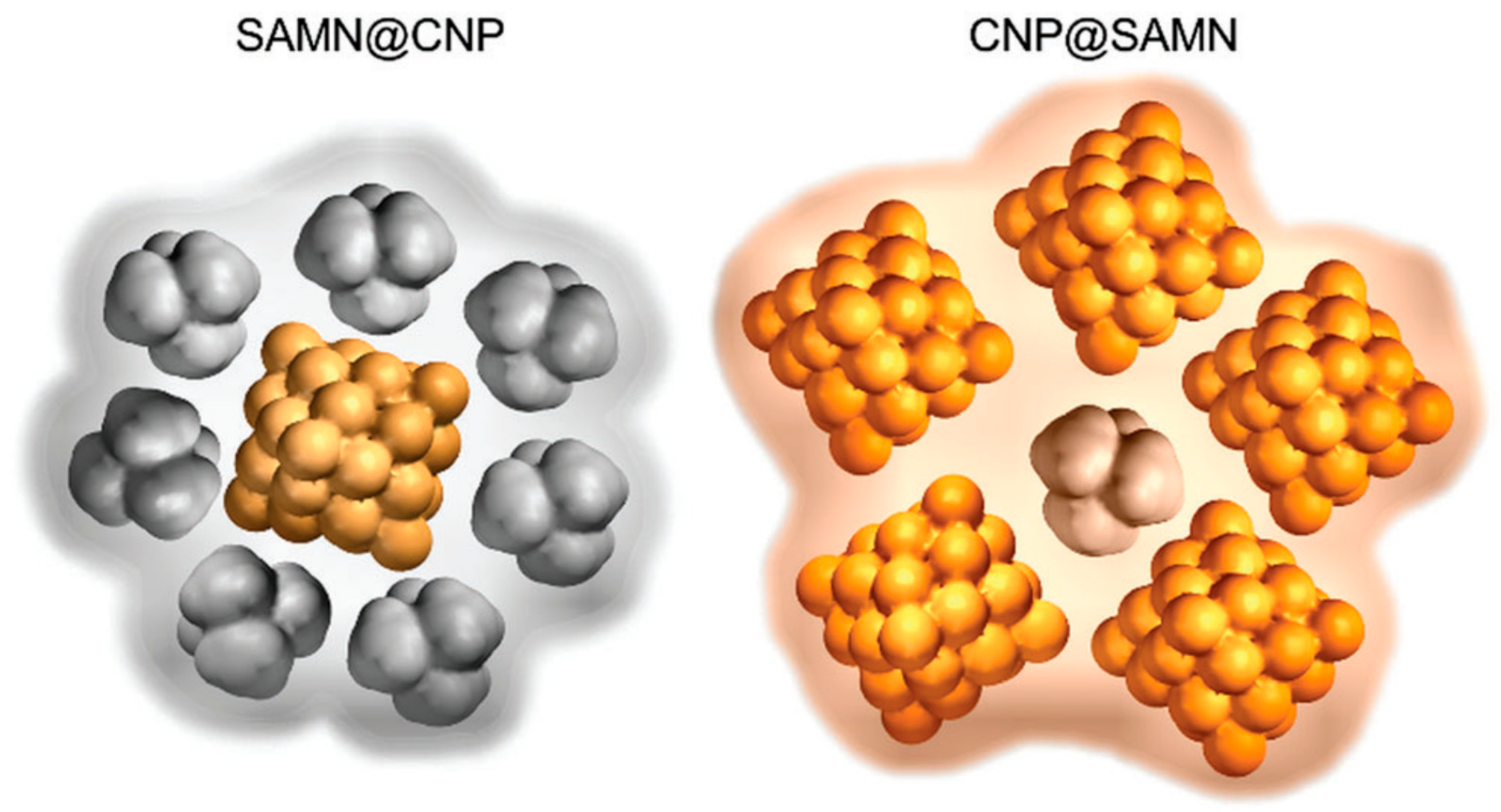
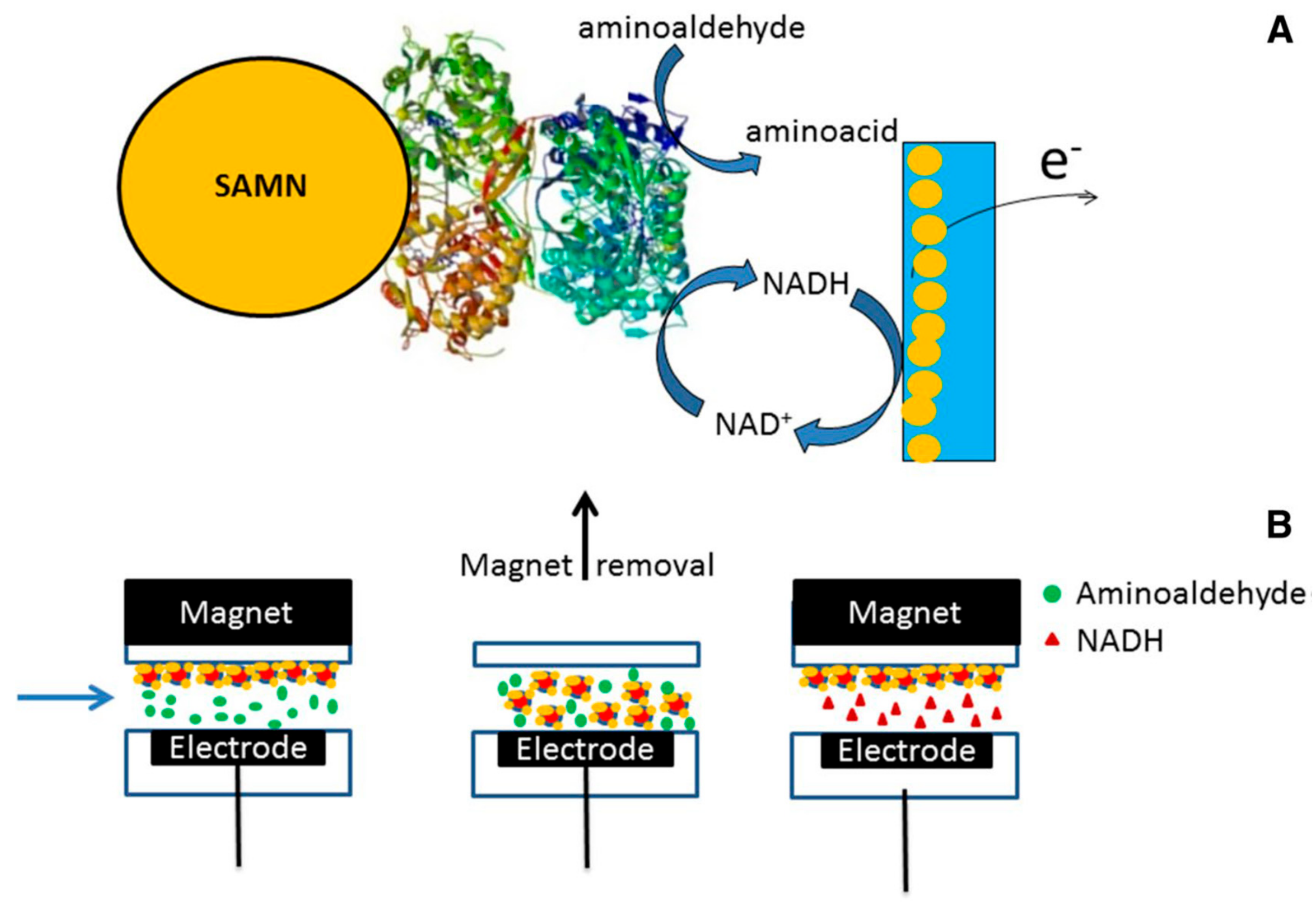
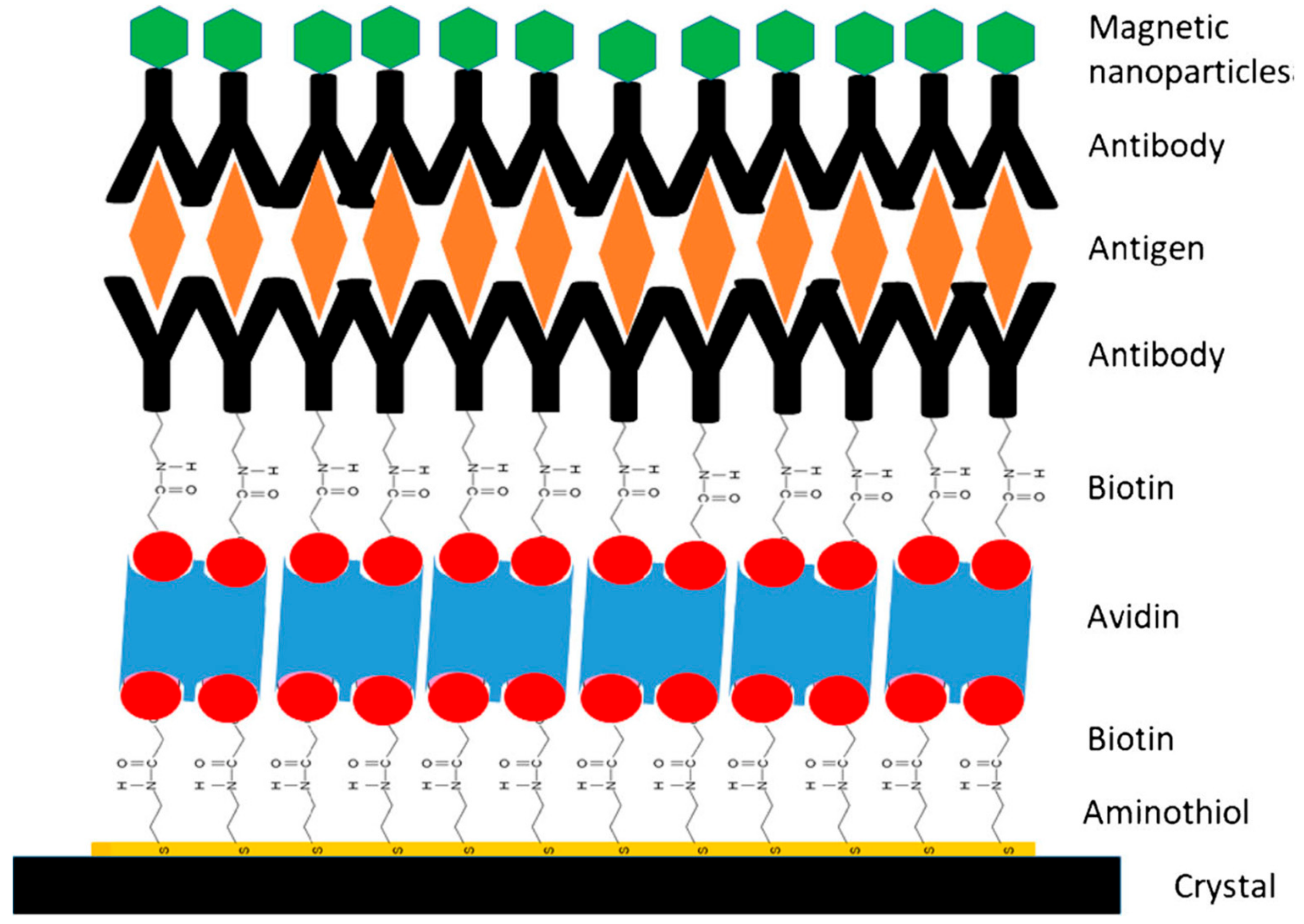
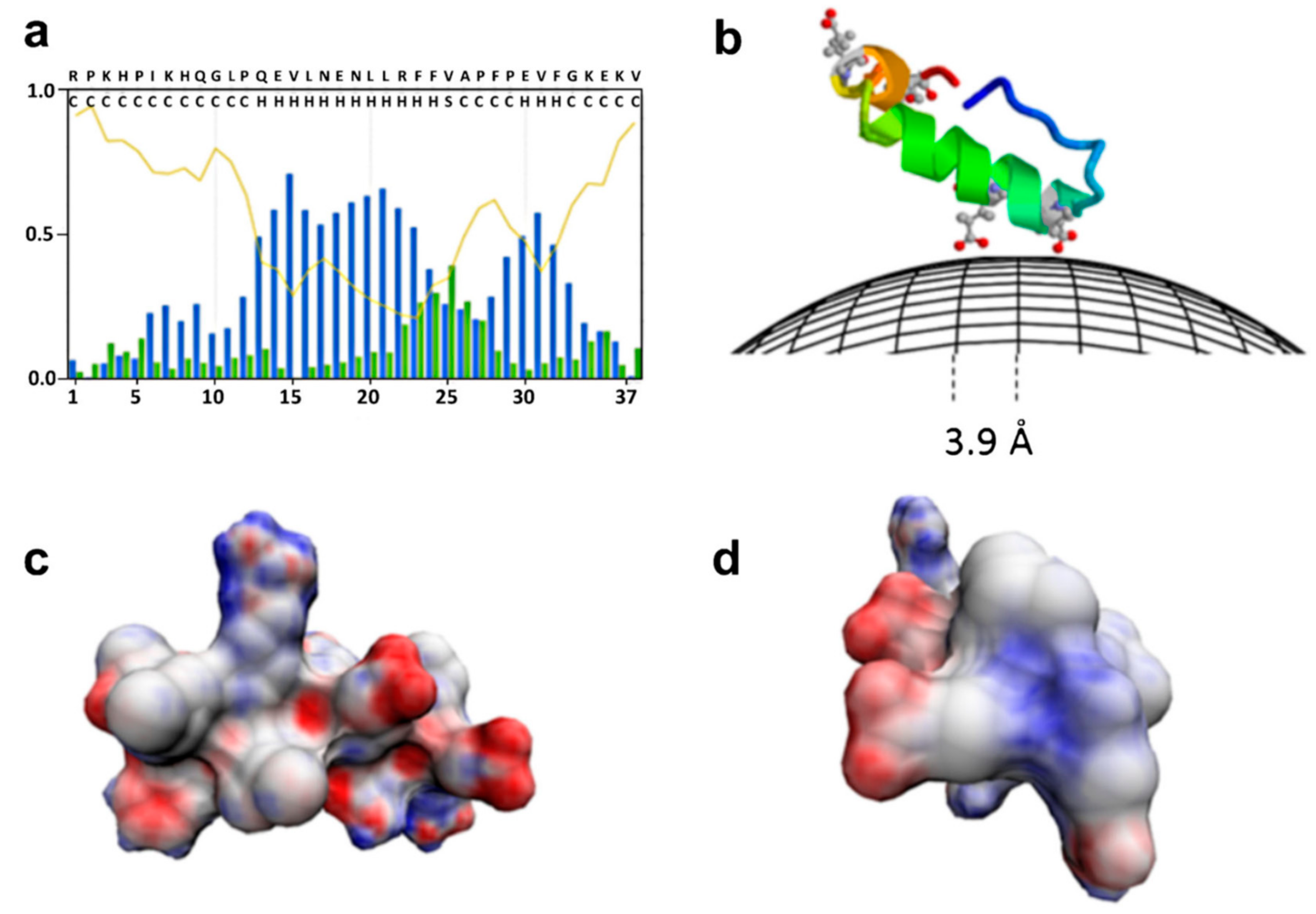

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Magro, M.; Vianello, F. Bare Iron Oxide Nanoparticles: Surface Tunability for Biomedical, Sensing and Environmental Applications. Nanomaterials 2019, 9, 1608. https://doi.org/10.3390/nano9111608
Magro M, Vianello F. Bare Iron Oxide Nanoparticles: Surface Tunability for Biomedical, Sensing and Environmental Applications. Nanomaterials. 2019; 9(11):1608. https://doi.org/10.3390/nano9111608
Chicago/Turabian StyleMagro, Massimiliano, and Fabio Vianello. 2019. "Bare Iron Oxide Nanoparticles: Surface Tunability for Biomedical, Sensing and Environmental Applications" Nanomaterials 9, no. 11: 1608. https://doi.org/10.3390/nano9111608
APA StyleMagro, M., & Vianello, F. (2019). Bare Iron Oxide Nanoparticles: Surface Tunability for Biomedical, Sensing and Environmental Applications. Nanomaterials, 9(11), 1608. https://doi.org/10.3390/nano9111608





