An Assessment of the Effect of Green Synthesized Silver Nanoparticles Using Sage Leaves (Salvia officinalis L.) on Germinated Plants of Maize (Zea mays L.)
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Material
2.2. Deionised Water, pH, and Ion Analysis
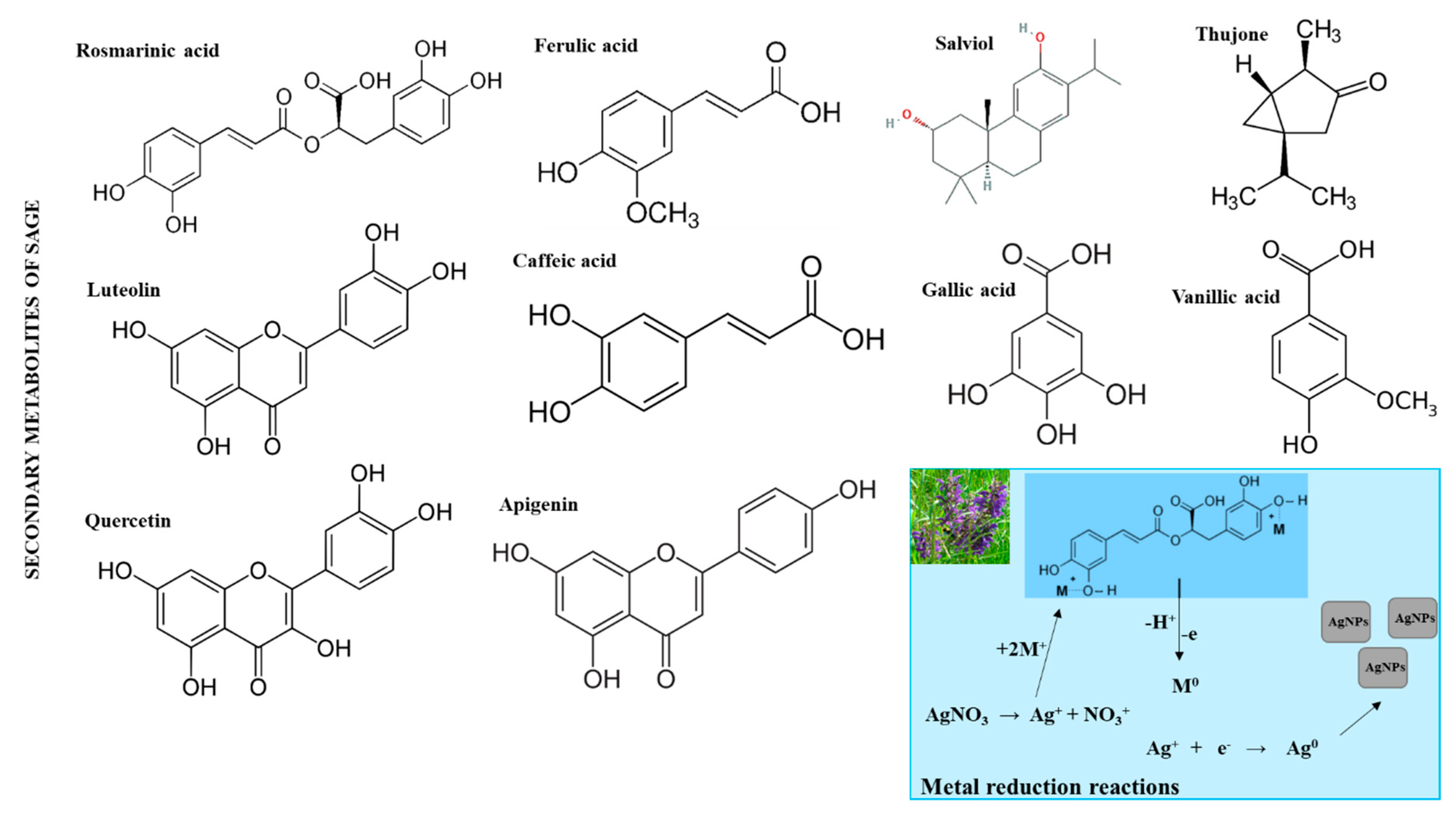
2.3. Preparation of the Sage Plant Extracts
2.4. Synthesis of AgNPs
2.5. Germination of Maize Seeds
2.6. Planting
2.7. Greenhouse Conditions
2.8. Harvesting Description
2.9. Photosynthetic Dyes Analysis
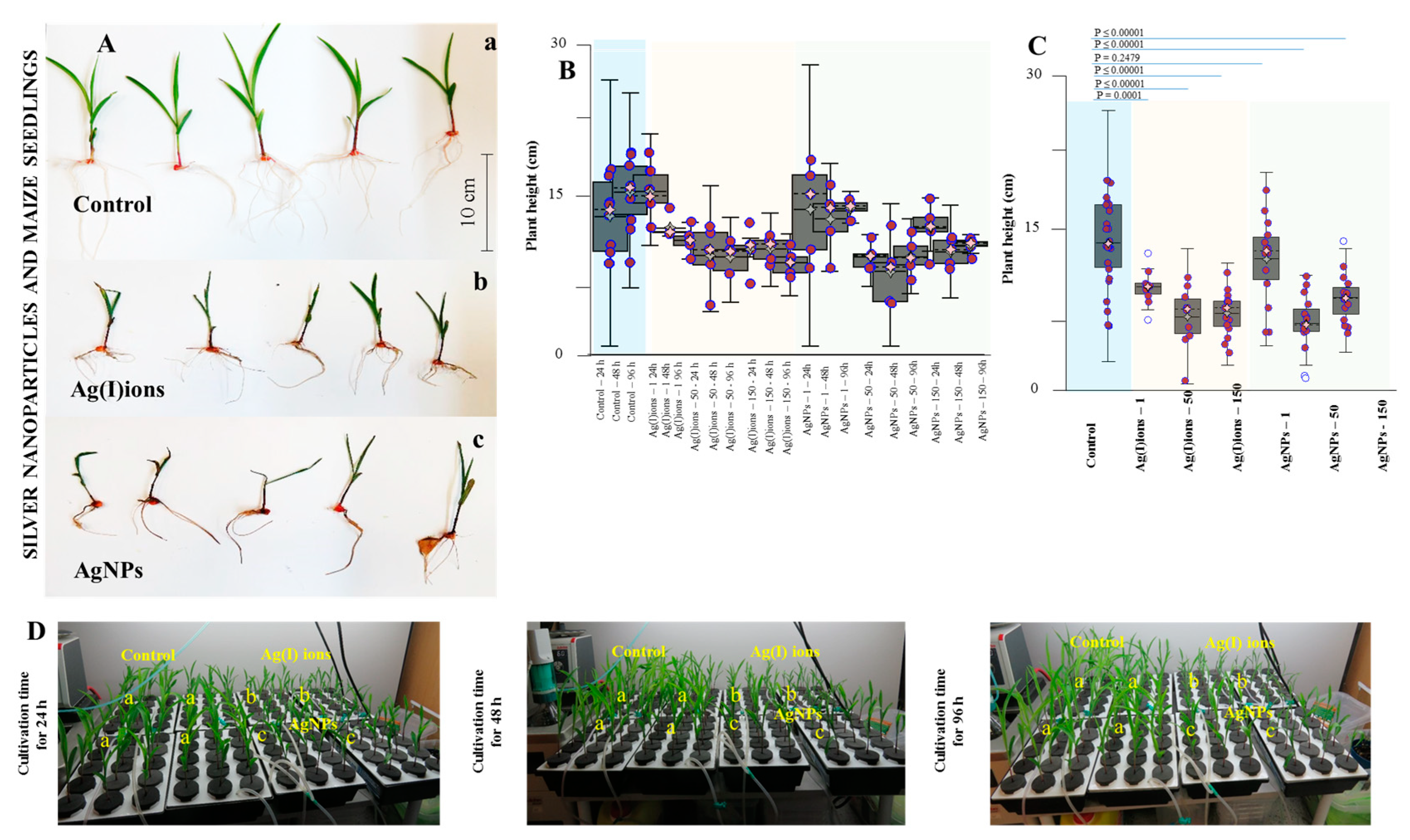
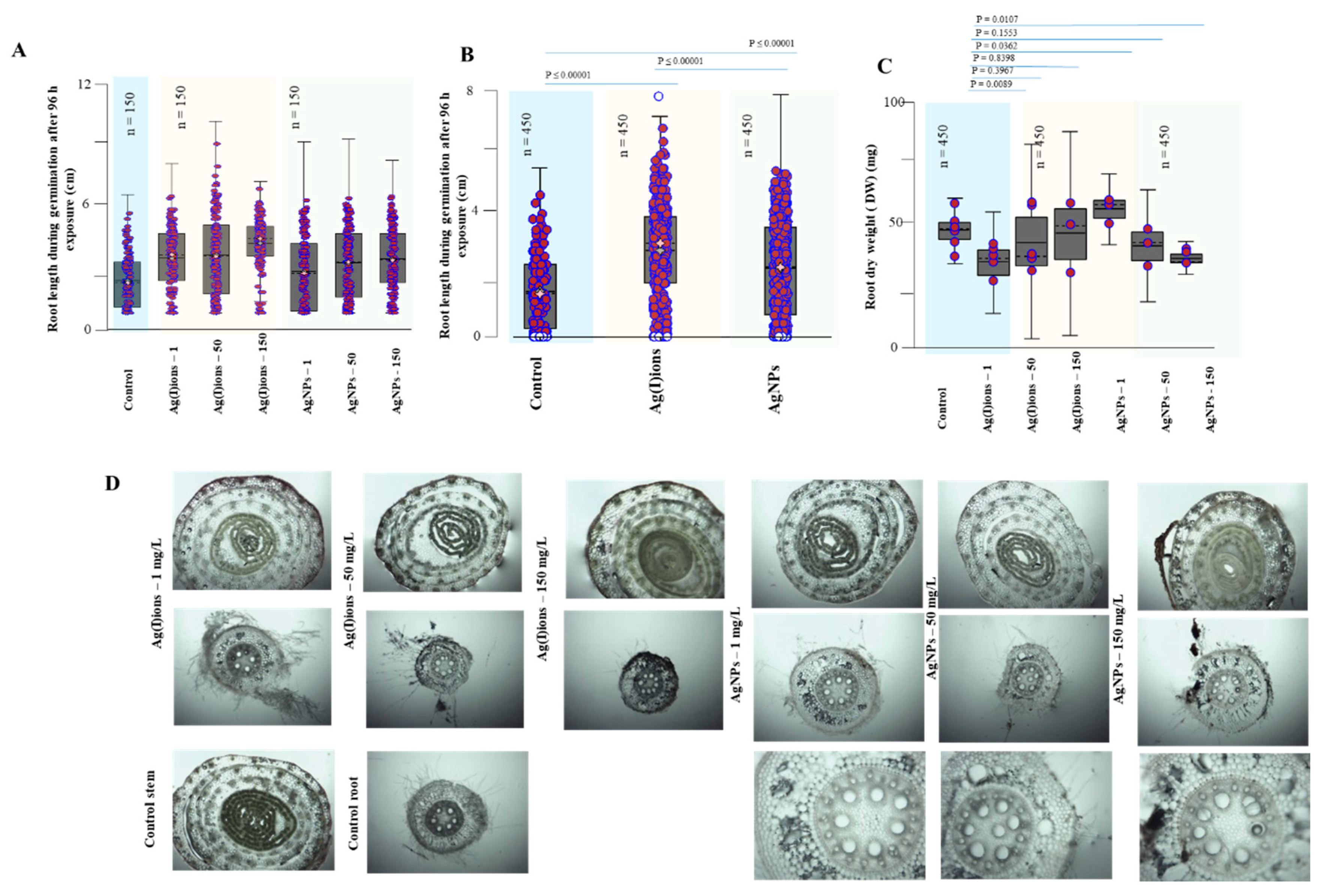
2.10. Plant Growth Metrics
2.11. Morphological and Anatomical Studies
2.12. Quenching of Radicals
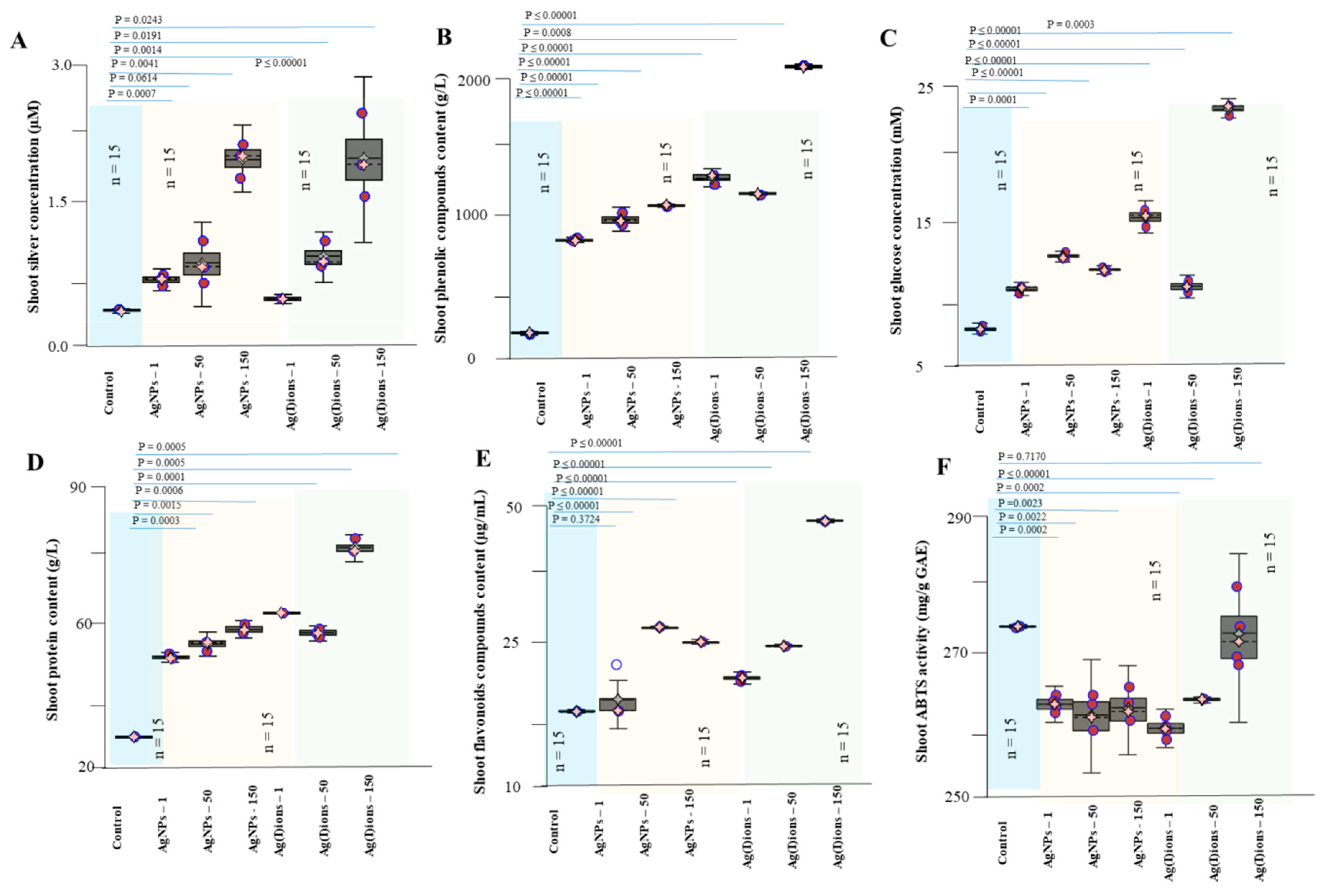
2.13. Total Phenolic Content (TPC) Determination
2.14. Total Flavonoid Content Determination
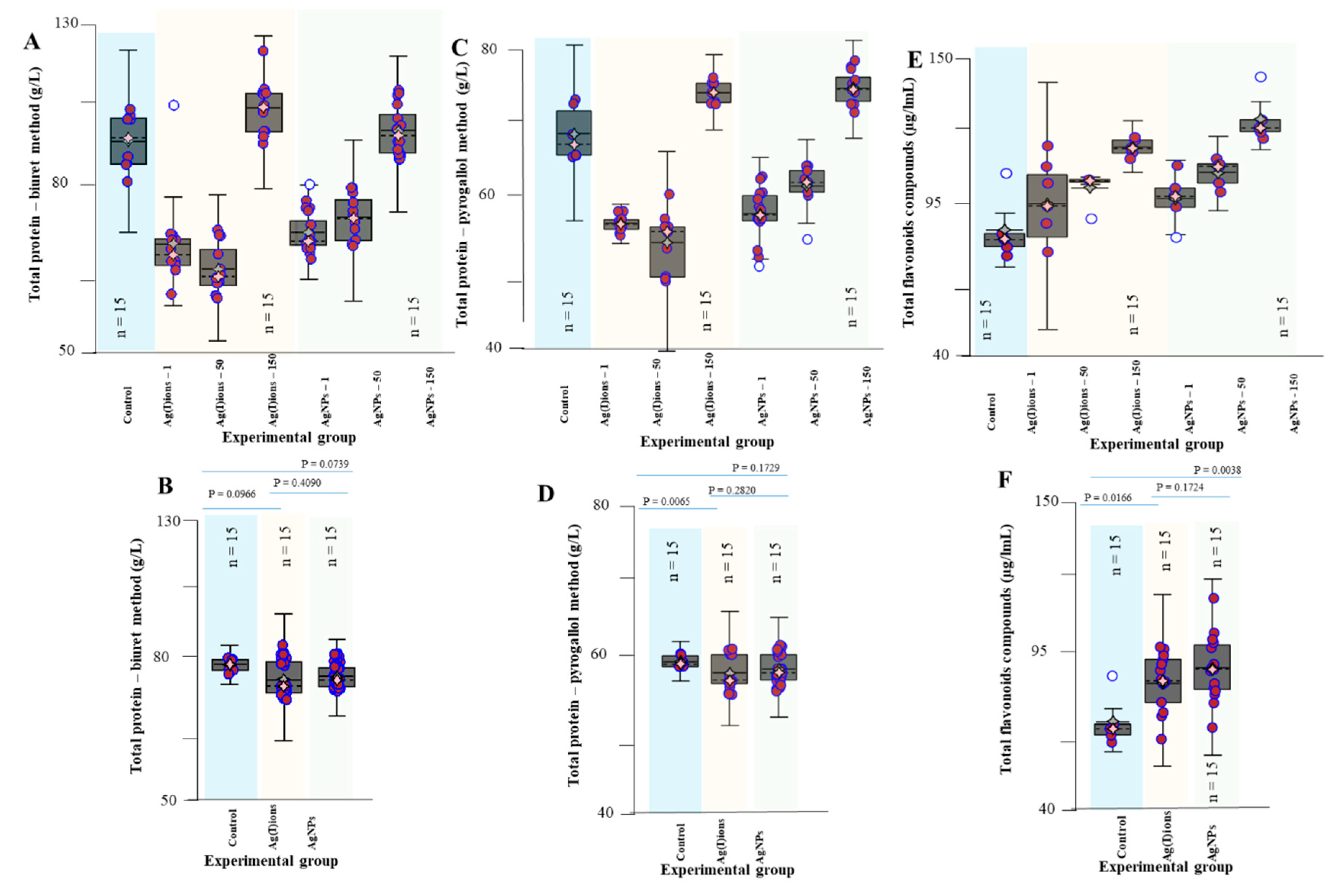
2.15. Total Protein (Biuret Method and Pyrogallol Red-Molybdate Method)
2.16. Pseudo-Peroxidase Assay
2.17. Silver Sample Collection-Electrochemical Measurement
2.18. Electron Microscopy of the AgNPs
2.19. Absorbance Measurements
2.20. Zetasizer Analysis of Nanoparticles
2.21. Data Treatment and Descriptive Statistics
3. Results and Discussion
3.1. Preparation of Sage Extract
3.2. Synthesis and Characterization of AgNPs
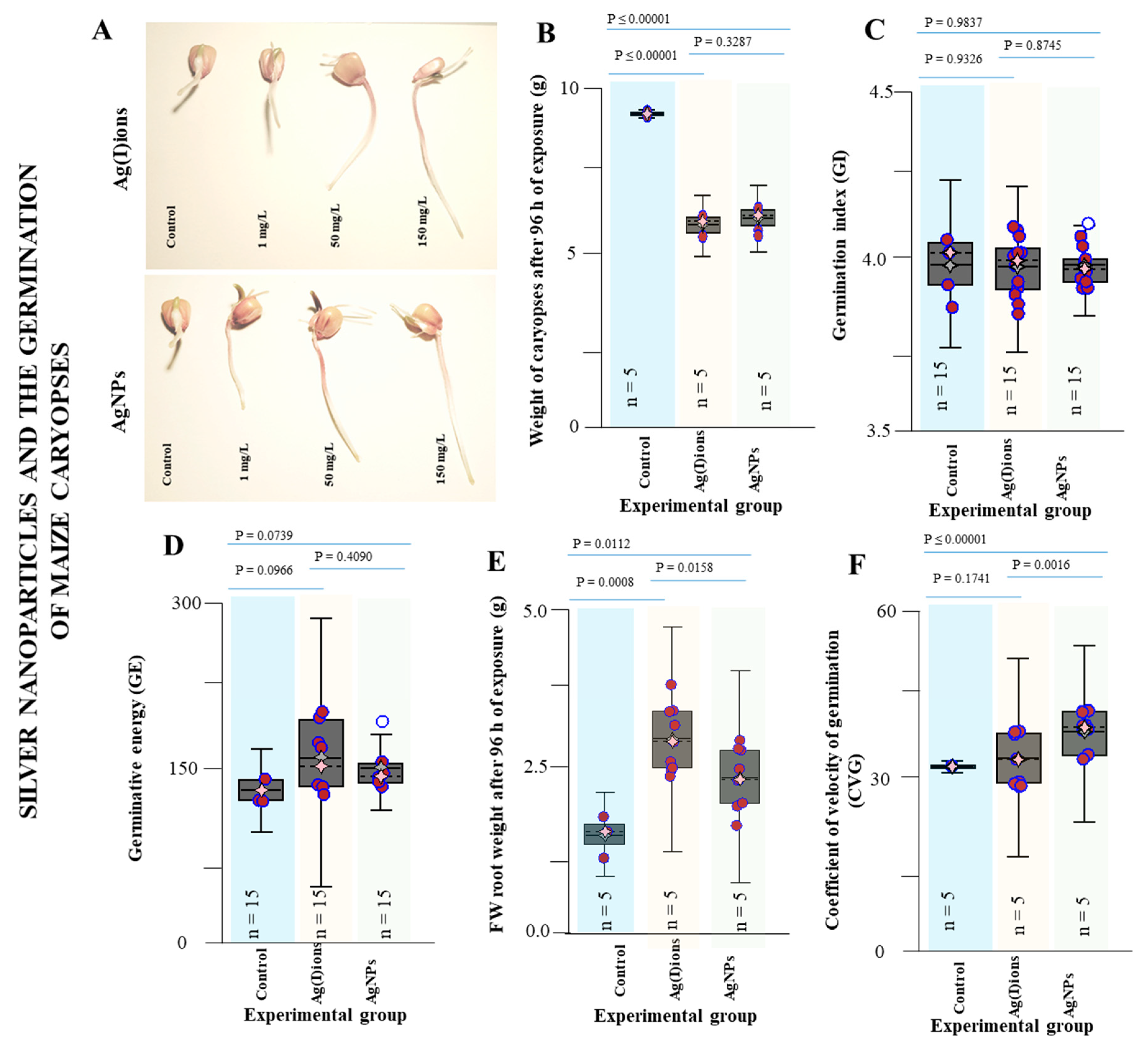
3.3. Effect of AgNPs and Ag(I) Ions on Germination
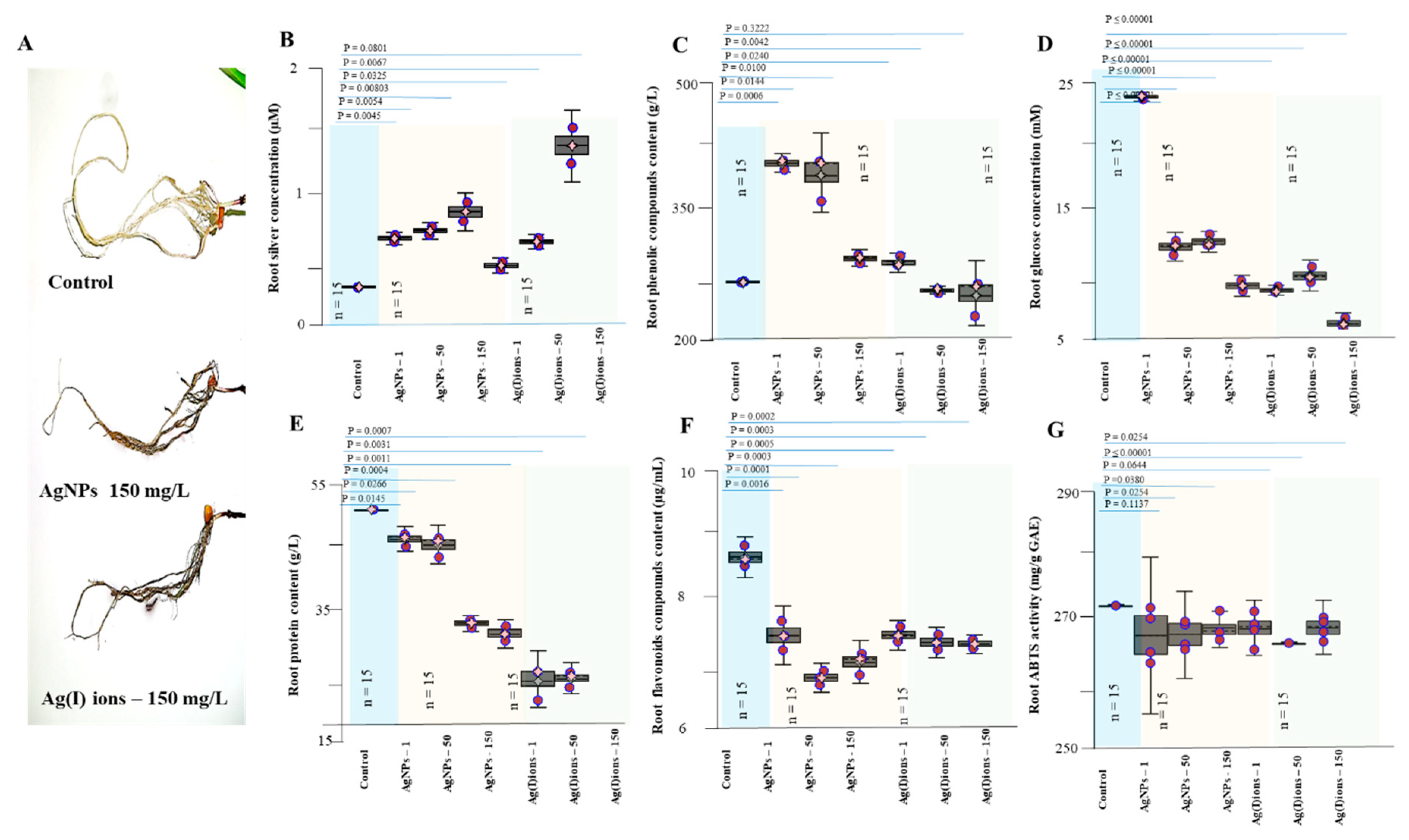
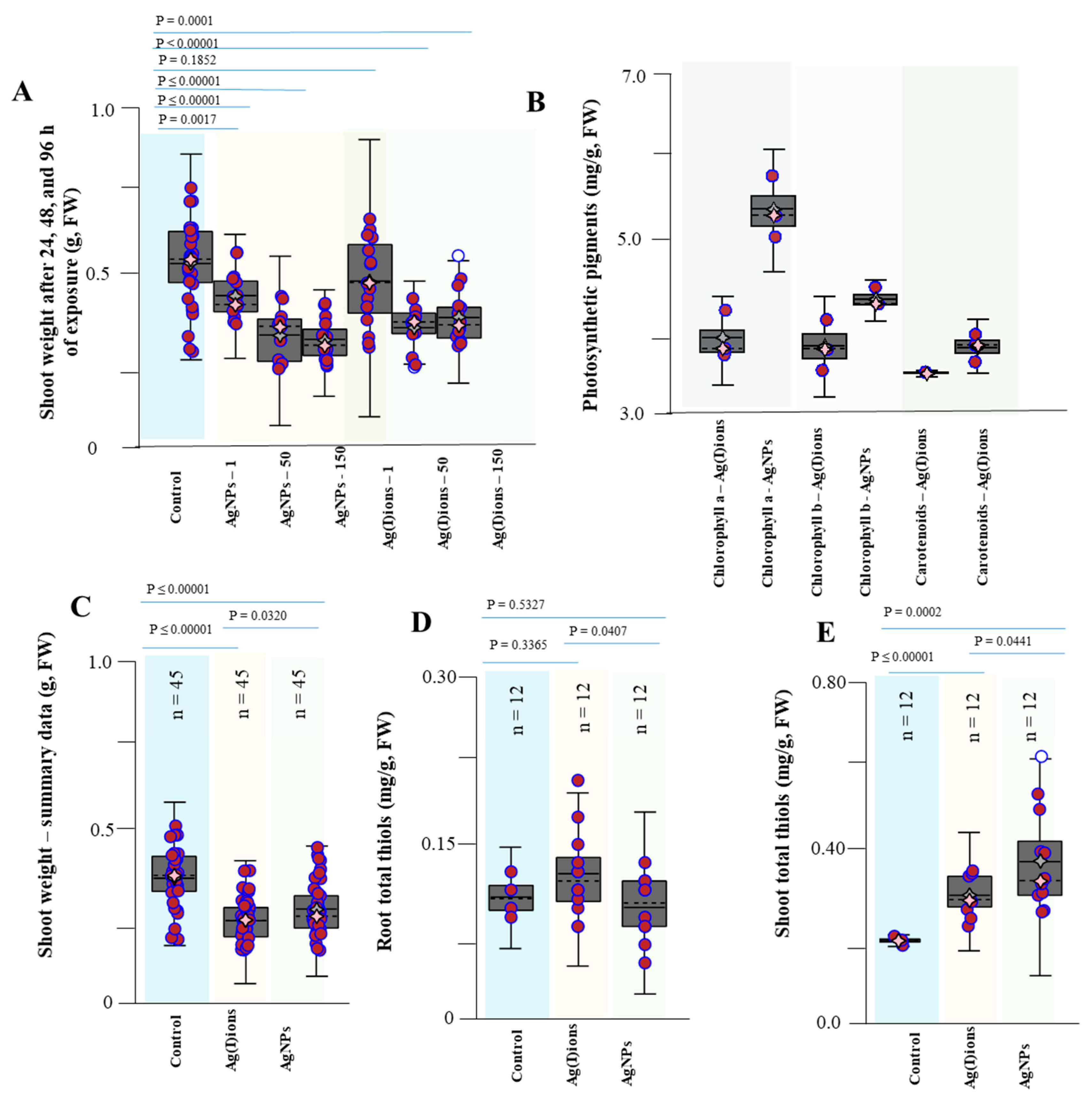
3.4. Effect of AgNPs and Ag(I) Ions on Planting Maize Seedlings
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ABTS | 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) |
| AgNP | silver nanoparticles |
| APX | ascorbate peroxidase |
| CAT | catalase |
| DNA | deoxyribonucleic acid |
| DPPH | 2,2-diphenyl-1-picrylhydrazyl |
| DW | dry weight |
| FRAP | Ferric Reducing Antioxidant Power |
| FW | fresh weight |
| HRP | horseradish peroxidase |
| IAA | indole-3-acetic acid |
| IBA | indole-3-butyric acid |
| LOD | limit of detection |
| LOQ | limit of quantification |
| NPs | nanoparticles |
| P | p-value |
| POX | plant peroxidase |
| R | correlation coefficient |
| RSD | relative standard deviation |
| SOD | superoxide dismutase |
| TMB | 3,3′,5,5′-tetramethylbenzidine |
References
- Darroudi, M.; Sabouri, Z.; Oskuee, R.K.; Zak, A.K.; Kargar, H.; Hamid, M.H.N.A. Green chemistry approach for the synthesis of zno nanopowders and their cytotoxic effects. Ceram. Int. 2014, 40, 4827–4831. [Google Scholar] [CrossRef]
- Zak, A.K.; Hashim, A.M.; Darroudi, M. Optical properties of zno/baco 3 nanocomposites in uv and visible regions. Nanoscale Res. Lett. 2014, 9, 399. [Google Scholar] [CrossRef] [PubMed]
- Yadav, K.; Giri, M.; Jaggi, N. Synthesis, characterization and photocatalytic studies of znse and ag: Znse nanoparticles. Res. Chem. Intermed. 2015, 41, 9967–9978. [Google Scholar] [CrossRef]
- Zamiri, R.; Zakaria, A.; Ahmad, M.B.; Sadrolhosseini, A.R.; Shameli, K.; Darroudi, M.; Mahdi, M.A. Investigation of spatial self-phase modulation of silver nanoparticles in clay suspension. Optik 2011, 122, 836–838. [Google Scholar] [CrossRef]
- Zamiri, R.; Azmi, B.; Darroudi, M.; Sadrolhosseini, A.R.; Husin, M.; Zaidan, A.; Mahdi, M. Preparation of starch stabilized silver nanoparticles with spatial self-phase modulation properties by laser ablation technique. Appl. Phys. A 2011, 102, 189–194. [Google Scholar] [CrossRef]
- Fadeel, B.; Farcal, L.; Hardy, B.; Vazquez-Campos, S.; Hristozov, D.; Marcomini, A.; Lynch, I.; Valsami-Jones, E.; Alenius, H.; Savolainen, K. Advanced tools for the safety assessment of nanomaterials. Nat. Nanotechnol. 2018, 13, 537–543. [Google Scholar] [CrossRef] [PubMed]
- Manna, I.; Bandyopadhyay, M. A review on the biotechnological aspects of utilizing engineered nanoparticles as delivery systems in plants. Plant Gene 2019, 17, 100167. [Google Scholar] [CrossRef]
- Joseph, T.; Morrison, M. Nanotechnology in agriculture and food. Nanoforum Rep. 2006, 2, 2–3. [Google Scholar]
- Zhang, L.; Gu, F.; Chan, J.; Wang, A.; Langer, R.; Farokhzad, O. Nanoparticles in medicine: Therapeutic applications and developments. Clin. Pharmacol. Ther. 2008, 83, 761–769. [Google Scholar] [CrossRef]
- Priester, J.H.; Ge, Y.; Mielke, R.E.; Horst, A.M.; Moritz, S.C.; Espinosa, K.; Gelb, J.; Walker, S.L.; Nisbet, R.M.; An, Y.-J.; et al. Soybean susceptibility to manufactured nanomaterials with evidence for food quality and soil fertility interruption. Proc. Natl. Acad. Sci. USA 2012, 109, E2451–E2456. [Google Scholar] [CrossRef]
- Stavrinidou, E.; Gabrielsson, R.; Gomez, E.; Crispin, X.; Nilsson, O.; Simon, D.T.; Berggren, M. Electronic plants. Sci. Adv. 2015, 1, 8. [Google Scholar] [CrossRef] [PubMed]
- Anjum, N.A.; Rodrigo, M.A.M.; Moulick, A.; Heger, Z.; Kopel, P.; Zitka, O.; Adam, V.; Lukatkin, A.S.; Duarte, A.C.; Pereira, E.; et al. Transport phenomena of nanoparticles in plants and animals/humans. Environ. Res. 2016, 151, 233–243. [Google Scholar] [CrossRef] [PubMed]
- Gardea-Torresdey, J.L.; Rico, C.M.; White, J.C. Trophic transfer, transformation, and impact of engineered nanomaterials in terrestrial environments. Environ. Sci. Technol. 2014, 48, 2526–2540. [Google Scholar] [CrossRef] [PubMed]
- Siddiqi, K.S.; Husen, A.; Rao, R.A.K. A review on biosynthesis of silver nanoparticles and their biocidal properties. J. Nanobiotechnol. 2018, 16, 28. [Google Scholar] [CrossRef] [PubMed]
- Iravani, S.; Korbekandi, H.; Mirmohammadi, S.V.; Zolfaghari, B. Synthesis of silver nanoparticles: Chemical, physical and biological methods. Res. Pharm. Sci. 2014, 9, 385. [Google Scholar] [PubMed]
- Darroudi, M.; KhandaKhandan Nasab, N.; Salimizand, H.; Dehnad, A. Green synthesis and antibacterial activity of zinc selenide (znse) nanoparticles. Nanomed. J. 2019. [Google Scholar]
- Majeed, S.; Bakhtiar, N.F.B.; Danish, M.; Ibrahim, M.M.; Hashim, R. Green approach for the biosynthesis of silver nanoparticles and its antibacterial and antitumor effect against osteoblast mg-63 and breast mcf-7 cancer cell lines. Sustain. Chem. Pharm. 2019, 12, 100138. [Google Scholar] [CrossRef]
- Naik, R.R.; Stringer, S.J.; Agarwal, G.; Jones, S.E.; Stone, M.O. Biomimetic synthesis and patterning of silver nanoparticles. Nat. Mater. 2002, 1, 169–172. [Google Scholar] [CrossRef]
- Singh, P.; Kim, Y.-J.; Zhang, D.; Yang, D.-C. Biological synthesis of nanoparticles from plants and microorganisms. Trends Biotech. 2016, 34, 588–599. [Google Scholar] [CrossRef]
- Bahlol, H.S.; Foda, M.F.; Ma, J.; Han, H. Robust synthesis of size-dispersal triangular silver nanoprisms via chemical reduction route and their cytotoxicity. Nanomaterials 2019, 9, 674. [Google Scholar] [CrossRef]
- Ovais, M.; Khalil, A.T.; Raza, A.; Khan, M.A.; Ahmad, I.; Ul Islam, N.; Saravanan, M.; Ubaid, M.F.; Ali, M.; Shinwari, Z.K. Green synthesis of silver nanoparticles via plant extracts: Beginning a new era in cancer theranostics. Nanomedicine 2016, 11, 21. [Google Scholar] [CrossRef] [PubMed]
- Sankar, R.; Rahman, P.K.S.M.; Varunkumar, K.; Anusha, C.; Kalaiarasi, A.; Shivashangari, K.S.; Ravikumar, V. Facile synthesis of curcuma longa tuber powder engineered metal nanoparticles for bioimaging applications. J. Mol. Struct. 2017, 1129, 8–16. [Google Scholar] [CrossRef]
- Kumar, A.; Vemula, P.K.; Ajayan, P.M.; John, G. Silver-nanoparticle-embedded antimicrobial paints based on vegetable oil. Nat. Mater. 2008, 7, 236–241. [Google Scholar] [CrossRef] [PubMed]
- Richter, A.P.; Brown, J.S.; Bharti, B.; Wang, A.; Gangwal, S.; Houck, K.; Hubal, E.A.C.; Paunov, V.N.; Stoyanov, S.D.; Velev, O.D. An environmentally benign antimicrobial nanoparticle based on a silver-infused lignin core. Nat. Nanotech. 2015, 10, 817–831. [Google Scholar] [CrossRef]
- Panacek, A.; Kvitek, L.; Smekalova, M.; Vecerova, R.; Kolar, M.; Roderova, M.; Dycka, F.; Sebela, M.; Prucek, R.; Tomanec, O.; et al. Bacterial resistance to silver nanoparticles and how to overcome it. Nat. Nanotechnol. 2018, 13, 65–71. [Google Scholar] [CrossRef]
- Salehi, S.; Shandiz, S.A.S.; Ghanbar, F.; Darvish, M.R.; Ardestani, M.S.; Mirzaie, A.; Jafari, M. Phytosynthesis of silver nanoparticles using artemisia marschalliana sprengel aerial part extract and assessment of their antioxidant, anticancer, and antibacterial properties. Int. J. Nanomed. 2016, 11, 1835–1846. [Google Scholar]
- Dakshayani, S.S.; Marulasiddeshwara, M.B.; Sharath Kumar, M.N.; Ramesh, G.; Raghavendra Kumar, P.; Devaraja, S.; Rashmi, H. Antimicrobial, anticoagulant and antiplatelet activities of green synthesized silver nanoparticles using selaginella (sanjeevini) plant extract. Int. J. Biol. Macromol. 2019, 131, 787–797. [Google Scholar]
- Almeida, L.M.; Magno, L.N.; Pereira, A.C.; Guidelli, E.J.; Baffa, O.; Kinoshita, A.; Goncalves, P.J. Toxicity of silver nanoparticles released by hancornia speciosa (mangabeira) biomembrane. Spectrochim. Acta Pt. A Mol. Biomol. Spectrosc. 2019, 210, 329–334. [Google Scholar] [CrossRef]
- Prakash, A.; Sharma, S.; Ahmad, N.; Ghosh, A.; Sinha, P. Bacteria mediated extracellular synthesis of metallic nanoparticles. Int. Res. J. Biotechnol. 2010, 1, 071–079. [Google Scholar]
- Wiley, B.; Sun, Y.; Mayers, B.; Xia, Y. Shape-controlled synthesis of metal nanostructures: The case of silver. Chem. Eur. J. 2005, 11, 454–463. [Google Scholar] [CrossRef]
- Supalkova, V.; Huska, D.; Diopan, V.; Hanustiak, P.; Zitka, O.; Stejskal, K.; Baloun, J.; Pikula, J.; Havel, L.; Zehnalek, J.; et al. Electroanalysis of plant thiols. Sensors 2007, 7, 932–959. [Google Scholar] [CrossRef]
- Ghosh, M.; Ghosh, I.; Godderis, L.; Hoet, P.; Mukherjee, A. Genotoxicity of engineered nanoparticles in higher plants. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2019, 842, 132–145. [Google Scholar] [CrossRef] [PubMed]
- Tolaymat, T.; Genaidy, A.; Abdelraheem, W.; Dionysiou, D.; Andersen, C. The effects of metallic engineered nanoparticles upon plant systems: An analytic examination of scientific evidence. Sci. Total Environ. 2017, 579, 93–106. [Google Scholar] [CrossRef] [PubMed]
- Zheng, S.M.; Zhou, Q.X.; Chen, C.H.; Yang, F.X.; Cai, Z.; Li, D.; Geng, Q.J.; Feng, Y.M.; Wang, H.Q. Role of extracellular polymeric substances on the behavior and toxicity of silver nanoparticles and ions to green algae chlorella vulgaris. Sci. Total Environ. 2019, 660, 1182–1190. [Google Scholar] [CrossRef] [PubMed]
- Nair, R.; Varghese, S.H.; Nair, B.G.; Maekawa, T.; Yoshida, Y.; Kumar, D.S. Nanoparticulate material delivery to plants. Plant Sci. 2010, 179, 154–163. [Google Scholar] [CrossRef]
- Abdelsalam, N.R.; Kandil, E.E.; Al-Msari, M.A.F.; Al-Jaddadi, M.A.M.; Ali, H.M.; Salem, M.Z.M.; Elshikh, M.S. Effect of foliar application of npk nanoparticle fertilization on yield and genotoxicity in wheat (triticum aestivum L.). Sci. Total. Environ. 2019, 653, 1128–1139. [Google Scholar] [CrossRef]
- Zheng, Y.L.; Hou, L.J.; Liu, M.; Newell, S.E.; Yin, G.Y.; Yu, C.D.; Zhang, H.L.; Li, X.F.; Gao, D.Z.; Gao, J.; et al. Effects of silver nanoparticles on nitrification and associated nitrous oxide production in aquatic environments. Sci. Adv. 2017, 3, 11. [Google Scholar] [CrossRef]
- Flores-Lopez, L.Z.; Espinoza-Gomez, H.; Somanathan, R. Silver nanoparticles: Electron transfer, reactive oxygen species, oxidative stress, beneficial and toxicological effects. Mini review. J. Appl. Toxicol. 2019, 39, 16–26. [Google Scholar] [CrossRef]
- Hong, F.; Zhou, J.; Liu, C.; Yang, F.; Wu, C.; Zheng, L.; Yang, P. Effect of nano-tio 2 on photochemical reaction of chloroplasts of spinach. Biol. Trace Elem. Res. 2005, 105, 269–279. [Google Scholar] [CrossRef]
- Krishnaraj, C.; Jagan, E.G.; Ramachandran, R.; Abirami, S.M.; Mohan, N.; Kalaichelvan, P.T. Effect of biologically synthesized silver nanoparticles on bacopa monnieri (linn.) wettst. Plant growth metabolism. Process Biochem. 2012, 47, 651–658. [Google Scholar] [CrossRef]
- Dayem, A.A.; Hossain, M.K.; Lee, S.B.; Kim, K.; Saha, S.K.; Yang, G.M.; Choi, H.Y.; Cho, S.G. The role of reactive oxygen species (ros) in the biological activities of metallic nanoparticles. Int. J. Mol. Sci. 2017, 18, 21. [Google Scholar]
- Khodakovskaya, M.; Dervishi, E.; Mahmood, M.; Xu, Y.; Li, Z.; Watanabe, F.; Biris, A.S. Carbon nanotubes are able to penetrate plant seed coat and dramatically affect seed germination and plant growth. ACS Nano 2009, 3, 3221–3227. [Google Scholar] [CrossRef] [PubMed]
- Kumari, M.; Mukherjee, A.; Chandrasekaran, N. Genotoxicity of silver nanoparticles in allium cepa. Sci. Total Environ. 2009, 407, 5243–5246. [Google Scholar] [CrossRef]
- Akter, M.; Sikder, M.T.; Rahman, M.M.; Ullah, A.K.M.A.; Hossain, K.F.B.; Banik, S.; Hosokawa, T.; Saito, T.; Kurasaki, M. A systematic review on silver nanoparticles-induced cytotoxicity: Physicochemical properties and perspectives. J. Adv. Res. 2018, 9, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Scherer, M.D.; Sposito, J.C.; Falco, W.F.; Grisolia, A.B.; Andrade, L.H.; Lima, S.M.; Machado, G.; Nascimento, V.A.; Gonçalves, D.A.; Wender, H. Cytotoxic and genotoxic effects of silver nanoparticles on meristematic cells of allium cepa roots: A close analysis of particle size dependence. Sci. Total Environ. 2019, 660, 459–467. [Google Scholar] [CrossRef] [PubMed]
- Abdelsalam, N.R.; Fouda, M.M.G.; Abdel-Megeed, A.; Ajarem, J.; Allam, A.A.; El-Naggar, M.E. Assessment of silver nanoparticles decorated starch and commercial zinc nanoparticles with respect to their genotoxicity on onion. Int. J. Biol. Macromol. 2019, 133, 1008–1018. [Google Scholar] [CrossRef] [PubMed]
- Karami Mehrian, S.; De Lima, R. Nanoparticles cyto and genotoxicity in plants: Mechanisms and abnormalities. Environ. Nanotech. Monit. Manag. 2016, 6, 184–193. [Google Scholar] [CrossRef]
- Sharifi-Rad, M.; Ozcelik, B.; Altin, G.; Daskaya-Dikmen, C.; Martorell, M.; Ramirez-Alarcon, K.; Alarcon-Zapata, P.; Morais-Braga, M.F.B.; Carneiro, J.N.P.; Leal, A.; et al. Salvia spp. Plants-from farm to food applications and phytopharmacotherapy. Trends Food Sci. Technol. 2018, 80, 242–263. [Google Scholar] [CrossRef]
- Jakovljevic, M.; Jokic, S.; Molnar, M.; Jasic, M.; Babic, J.; Jukic, H.; Banjari, I. Bioactive profile of various salvia officinalis L. Preparations. Plants-Basel 2019, 8, 30. [Google Scholar] [CrossRef]
- Lu, Y.R.; Foo, L.Y. Antioxidant activities of polyphenols from sage (salvia officinalis). Food Chem. 2001, 75, 197–202. [Google Scholar] [CrossRef]
- Wang, M.F.; Li, J.G.; Rangarajan, M.; Shao, Y.; LaVoie, E.J.; Huang, T.C.; Ho, C.T. Antioxidative phenolic compounds from sage (salvia officinalis). J. Agric. Food Chem. 1998, 46, 4869–4873. [Google Scholar] [CrossRef]
- Er, M.; Tugay, O.; Ozcan, M.M.; Ulukus, D.; Al-Juhaimi, F. Biochemical properties of some salvia L. Species. Environ. Monit. Assess. 2013, 185, 5193–5198. [Google Scholar] [CrossRef] [PubMed]
- Howes, M.J.R.; Perry, N.S.L.; Houghton, P.J. Plants with traditional uses and activities, relevant to the management of alzheimer’s disease and other cognitive disorders. Phytother. Res. 2003, 17, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Scholey, A.B.; Tildesley, N.T.J.; Ballard, C.G.; Wesnes, K.A.; Tasker, A.; Perry, E.K.; Kennedy, D.O. An extract of salvia (sage) with anticholinesterase properties improves memory and attention in healthy older volunteers. Psychopharmacology 2008, 198, 127–139. [Google Scholar] [CrossRef] [PubMed]
- Bozin, B.; Mlmica-Dukic, N.; Samojlik, I.; Jovin, E. Antimicrobial and antioxidant properties of rosemary and sage (rosmarinus officinalis L. And salvia officinalis L., lamiaceae) essential oils. J. Agric. Food Chem. 2007, 55, 7879–7885. [Google Scholar] [CrossRef]
- Moghadam, S.B.; Masoudi, R.; Monsefi, M. Salvia officinalis induces apoptosis in mammary carcinoma cells through alteration of bax to bcl-2 ratio. Iran. J. Sci. Technol. Trans. A Sci. 2018, 42, 297–303. [Google Scholar] [CrossRef]
- Pei, J.W.; Fu, B.F.; Jiang, L.F.; Sun, T.Z. Biosynthesis, characterization, and anticancer effect of plant-mediated silver nanoparticles using coptis chinensis. Int. J. Nanomed. 2019, 14, 1969–1978. [Google Scholar] [CrossRef]
- Zhamanbayeva, G.T.; Aralbayeva, A.N.; Murzakhmetova, M.K.; Tuleukhanov, S.T.; Danilenko, M. Cooperative antiproliferative and differentiation-enhancing activity of medicinal plant extracts in acute myeloid leukemia cells. Biomed. Pharmacother. 2016, 82, 80–89. [Google Scholar] [CrossRef]
- Bekut, M.; Brkic, S.; Kladar, N.; Dragovic, G.; Gavaric, N.; Bozin, B. Potential of selected lamiaceae plants in anti(retro)viral therapy. Pharmacol. Res. 2018, 133, 301–314. [Google Scholar] [CrossRef]
- Roby, M.H.H.; Sarhan, M.A.; Selim, K.A.H.; Khalel, K.I. Evaluation of antioxidant activity, total phenols and phenolic compounds in thyme (thymus vulgaris L.), sage (salvia officinalis L.), and marjoram (origanum majorana L.) extracts. Ind. Crop. Prod. 2013, 43, 827–831. [Google Scholar] [CrossRef]
- Ruttkay-Nedecky, B.; Skalickova, S.; Kepinska, M.; Cihalova, K.; Docekalova, M.; Stankova, M.; Uhlirova, D.; Fernandez, C.; Sochor, J.; Milnerowicz, H.; et al. Development of new silver nanoparticles suitable for materials with antimicrobial properties. J. Nanosci. Nanotechnol. 2019, 19, 2762–2769. [Google Scholar] [CrossRef] [PubMed]
- Almaghrabi, O.A. Impact of drought stress on germination and seedling growth parameters of some wheat cultivars. Life Sci. J. 2012, 9, 590–598. [Google Scholar]
- Klejdus, B.; Zehnalek, J.; Adam, V.; Petrek, J.; Kizek, R.; Vacek, J.; Trnkova, L.; Rozik, R.; Havel, L.; Kuban, V. Sub-picomole high-performance liquid chromatographic/mass spectrometric determination of glutathione in the maize (zea mays L.) kernels exposed to cadmium. Anal. Chim. Acta 2004, 520, 117–124. [Google Scholar] [CrossRef]
- Kizek, R.; Vacek, J.; Trnkova, L.; Klejdus, B.; Kuban, V. Electrochemical biosensors in agricultural and environmental analysis. Chem. Listy 2003, 97, 1003–1006. [Google Scholar]
- Mikulaskova, H.; Merlos, M.A.R.; Zitka, O.; Kominkova, M.; Hynek, D.; Adam, V.; Beklova, M.; Kizek, R. Employment of electrochemical methods for assessment of the maize (zea mays L.) and pea (pisum sativum L.) response to treatment with platinum(iv). Int. J. Electrochem. Sci. 2013, 8, 4505–4519. [Google Scholar]
- Potesil, D.; Petrlova, J.; Adam, V.; Vacek, J.; Klejdus, B.; Zehnalek, J.; Trnkova, L.; Havel, L.; Kizek, R. Simultaneous femtomole determination of cysteine, reduced and oxidized glutathione, and phytochelatin in maize (zea mays L.) kernels using high-performance liquid chromatography with electrochemical detection. J. Chromatogr. A 2005, 1084, 134–144. [Google Scholar] [CrossRef] [PubMed]
- Sochor, J.; Ryvolova, M.; Krystofova, O.; Salas, P.; Hubalek, J.; Adam, V.; Trnkova, L.; Havel, L.; Beklova, M.; Zehnalek, J.; et al. Fully automated spectrometric protocols for determination of antioxidant activity: Advantages and disadvantages. Molecules 2010, 15, 8618–8640. [Google Scholar] [CrossRef] [PubMed]
- Bibi, G.; Haq, I.; Ullah, N.; Muazzam, A.G.; Mannan, A.; Mirza, B. Phytochemical evaluation of naturally growing aster tomsonii plant species. IJPIS J. Pharmacogn. Herb. Form. 2012, 2, 33–39. [Google Scholar]
- Cox, A.; Venkatachalam, P.; Sahi, S.; Sharma, N. Silver and titanium dioxide nanoparticle toxicity in plants: A review of current research. Plant Physiol. Biochem. 2016, 107, 147–163. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Yan, J. Plant uptake and accumulation of engineered metallic nanoparticles from lab to field conditions. Curr. Opin. Environ. Sci. Health 2018, 6, 16–20. [Google Scholar] [CrossRef]
- Shrivastava, M.; Srivastav, A.; Gandhi, S.; Rao, S.; Roychoudhury, A.; Kumar, A.; Singhal, R.K.; Jha, S.K.; Singh, S.D. Monitoring of engineered nanoparticles in soil-plant system: A review. Environ. Nanotech. Monit. Manag. 2019, 11, 100218. [Google Scholar] [CrossRef]
- Hochella, M.F.; Lower, S.K.; Maurice, P.A.; Penn, R.L.; Sahai, N.; Sparks, D.L.; Twining, B.S. Nanominerals, mineral nanoparticles, and earth systems. Science 2008, 319, 1631–1635. [Google Scholar] [CrossRef] [PubMed]
- Bhor, G.; Maskare, S.; Hinge, S.; Singh, L.; Nalwade, A. Synthesis of silver nanoparticles using leaflet extract of nephrolepi sexaltata L. And evaluation antibacterial activity against human and plant pathogenic bacteria. Asian J. Pharm. Technol. Innov. 2014, 2, 6. [Google Scholar]
- Gargulak, M.; Strofova, N.; Sehnal, K.; Hosnedlova, B.; Docekalova, M.; Ofomaja, A.E.; Fernandez, C.; Kepinska, M.; Milnerowicz; Kizek, R. Phytotoxicity of silver nanoparticles (agnps) prepared by green synthesis using sage leaves (salvia officinalis). IEEE Malaysia 2019, in press. [Google Scholar]
- Doody, M.A.; Wang, D.J.; Bais, H.P.; Jin, Y. Differential antimicrobial activity of silver nanoparticles to bacteria bacillus subtilis and escherichia coli, and toxicity to crop plant zea mays and beneficial b. Subtilis-inoculated z. Mays. J. Nanopart. Res. 2016, 18, 19. [Google Scholar] [CrossRef]
- Dent, M.; Dragovic-Uzelac, V.; Penic, M.; Brncic, M.; Bosiljkov, T.; Levaj, B. The effect of extraction solvents, temperature and time on the composition and mass fraction of polyphenols in dalmatian wild sage (salvia officinalis L.) extracts. Food Technol. Biotechnol. 2013, 51, 84–91. [Google Scholar]
- Akkol, E.K.; Göger, F.; Koşar, M.; Başer, K.H.C. Phenolic composition and biological activities of salvia halophila and salvia virgata from turkey. Food Chem. 2008, 108, 942–949. [Google Scholar] [CrossRef]
- Thomas, B.; Vithiya, B.S.M.; Prasad, T.A.A.; Mohamed, S.B.; Magdalane, C.M.; Kaviyarasu, K.; Maaza, M. Antioxidant and photocatalytic activity of aqueous leaf extract mediated green synthesis of silver nanoparticles using passiflora edulis f. Flavicarpa. J. Nanosci. Nanotechnol. 2019, 19, 2640–2648. [Google Scholar] [CrossRef]
- Baharara, J.; Ramezani, T.; Mousavi, M.; Asadi-Samani, M. Antioxidant and anti-inflammatory activity of green synthesized silver nanoparticles using salvia officinalis extract. Ann. Trop. Med. PH 2017, 10, 1265. [Google Scholar]
- Zhang, K.; Liu, X.; Samuel Ravi, S.O.A.; Ramachandran, A.; Aziz Ibrahim, I.A.; Nassir, A.M.; Yao, J. Synthesis of silver nanoparticles (agnps) from leaf extract of salvia miltiorrhiza and its anticancer potential in human prostate cancer lncap cell lines. Artif. Cells Nanomed. Biotechnol. 2019, 47, 2846–2854. [Google Scholar] [CrossRef]
- Tripathi, D.K.; Tripathi, A.; Shweta; Singh, S.; Singh, Y.; Vishwakarma, K.; Yadav, G.; Sharma, S.; Singh, V.K.; Mishra, R.K.; et al. Uptake, accumulation and toxicity of silver nanoparticle in autotrophic plants, and heterotrophic microbes: A concentric review. Front. Microbiol. 2017, 8, 7. [Google Scholar] [CrossRef] [PubMed]
- Orosa-Puente, B.; Leftley, N.; Von Wangenheim, D.; Banda, J.; Srivastava, A.K.; Hill, K.; Truskina, J.; Bhosale, R.; Morris, E.; Srivastava, M.; et al. Root branching toward water involves posttranslational modification of transcription factor arf7. Science 2018, 362, 1407–1410. [Google Scholar] [CrossRef] [PubMed]
- Giehl, R.F.H.; von Wiren, N. Hydropatterning-how roots test the waters. Science 2018, 362, 1358–1359. [Google Scholar] [CrossRef] [PubMed]
- Nair, P.M.G.; Chung, I.M. Physiological and molecular level studies on the toxicity of silver nanoparticles in germinating seedlings of mung bean (vigna radiata L.). Acta Physiol. Plant. 2015, 37, 11. [Google Scholar] [CrossRef]
- Wiechen, M.; Zaharieva, I.; Dau, H.; Kurz, P. Layered manganese oxides for water-oxidation: Alkaline earth cations influence catalytic activity in a photosystem ii-like fashion. Chem. Sci. 2012, 3, 2330–2339. [Google Scholar] [CrossRef]
- Thangavelu, R.M.; Gunasekaran, D.; Jesse, M.I.; Riyaz, S.U.M.; Sundarajan, D.; Krishnan, K. Nanobiotechnology approach using plant rooting hormone synthesized silver nanoparticle as “nanobullets” for the dynamic applications in horticulture—An in vitro and ex vitro study. Arab. J. Chem. 2018, 11, 48–61. [Google Scholar] [CrossRef]
- Almutairi, Z.M.; Alharbi, A. Effect of silver nanoparticles on seed germination of crop plants. J. Adv. Agric. 2015, 4, 283–288. [Google Scholar] [CrossRef]
- Zheng, L.; Hong, F.S.; Lu, S.P.; Liu, C. Effect of nano-TiO2 on strength of naturally and growth aged seeds of spinach. Biol. Trace Elem. Res. 2005, 104, 83–91. [Google Scholar] [CrossRef]
- Mehta, C.; Srivastava, R.; Arora, S.; Sharma, A. Impact assessment of silver nanoparticles on plant growth and soil bacterial diversity. 3 Biotech 2016, 6, 254. [Google Scholar]
- Tripathi, A.; Liu, S.; Singh, P.K.; Kumar, N.; Pandey, A.C.; Tripathi, D.K.; Chauhan, D.K.; Sahi, S. Differential phytotoxic responses of silver nitrate (agno3) and silver nanoparticle (agnps) in cucumis sativus L. Plant Gene 2017, 11, 255–264. [Google Scholar] [CrossRef]
- Yin, J.-J.; Liu, J.; Ehrenshaft, M.; Roberts, J.E.; Fu, P.P.; Mason, R.P.; Zhao, B. Phototoxicity of nano titanium dioxides in hacat keratinocytes—generation of reactive oxygen species and cell damage. Toxicol. Appl. Pharmacol. 2012, 263, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Yin, L.; Colman, B.P.; McGill, B.M.; Wright, J.P.; Bernhardt, E.S. Effects of silver nanoparticle exposure on germination and early growth of eleven wetland plants. PLoS ONE 2012, 7, e47674. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Jiang, F.; Ma, C.; Rui, Y.; Rui, M.; Adeel, M.; Cao, W.; Xing, B. Alteration of crop yield and quality of wheat upon exposure to silver nanoparticles in a life cycle study. J. Agric. Food Chem. 2018, 66, 2589–2597. [Google Scholar] [CrossRef] [PubMed]
- Dietz, K.-J.; Herth, S. Plant nanotoxicology. Trends Plant Sci. 2011, 16, 582–589. [Google Scholar] [CrossRef]
- Carpita, N.; Sabularse, D.; Montezinos, D.; Delmer, D.P. Determination of the pore size of cell walls of living plant cells. Science 1979, 205, 1144–1147. [Google Scholar] [CrossRef]
- Arnaout, C.L.; Gunsch, C.K. Impacts of silver nanoparticle coating on the nitrification potential of nitrosomonas europaea. Environ. Sci. Technol. 2012, 46, 5387–5395. [Google Scholar] [CrossRef]
- Wang, X.; Yang, X.; Chen, S.; Li, Q.; Wang, W.; Hou, C.; Gao, X.; Wang, L.; Wang, S. Zinc oxide nanoparticles affect biomass accumulation and photosynthesis in arabidopsis. Front. Plant. Sci. 2016, 6, 1243. [Google Scholar] [CrossRef]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sehnal, K.; Hosnedlova, B.; Docekalova, M.; Stankova, M.; Uhlirova, D.; Tothova, Z.; Kepinska, M.; Milnerowicz, H.; Fernandez, C.; Ruttkay-Nedecky, B.; et al. An Assessment of the Effect of Green Synthesized Silver Nanoparticles Using Sage Leaves (Salvia officinalis L.) on Germinated Plants of Maize (Zea mays L.). Nanomaterials 2019, 9, 1550. https://doi.org/10.3390/nano9111550
Sehnal K, Hosnedlova B, Docekalova M, Stankova M, Uhlirova D, Tothova Z, Kepinska M, Milnerowicz H, Fernandez C, Ruttkay-Nedecky B, et al. An Assessment of the Effect of Green Synthesized Silver Nanoparticles Using Sage Leaves (Salvia officinalis L.) on Germinated Plants of Maize (Zea mays L.). Nanomaterials. 2019; 9(11):1550. https://doi.org/10.3390/nano9111550
Chicago/Turabian StyleSehnal, Karel, Bozena Hosnedlova, Michaela Docekalova, Martina Stankova, Dagmar Uhlirova, Zuzana Tothova, Marta Kepinska, Halina Milnerowicz, Carlos Fernandez, Branislav Ruttkay-Nedecky, and et al. 2019. "An Assessment of the Effect of Green Synthesized Silver Nanoparticles Using Sage Leaves (Salvia officinalis L.) on Germinated Plants of Maize (Zea mays L.)" Nanomaterials 9, no. 11: 1550. https://doi.org/10.3390/nano9111550
APA StyleSehnal, K., Hosnedlova, B., Docekalova, M., Stankova, M., Uhlirova, D., Tothova, Z., Kepinska, M., Milnerowicz, H., Fernandez, C., Ruttkay-Nedecky, B., Nguyen, H. V., Ofomaja, A., Sochor, J., & Kizek, R. (2019). An Assessment of the Effect of Green Synthesized Silver Nanoparticles Using Sage Leaves (Salvia officinalis L.) on Germinated Plants of Maize (Zea mays L.). Nanomaterials, 9(11), 1550. https://doi.org/10.3390/nano9111550