Core-Shell Heterostructured and Visible-Light-Driven Titanoniobate/TiO2 Composite for Boosting Photodegradation Performance
Abstract
:1. Introduction
2. Materials and Methods
3. Results
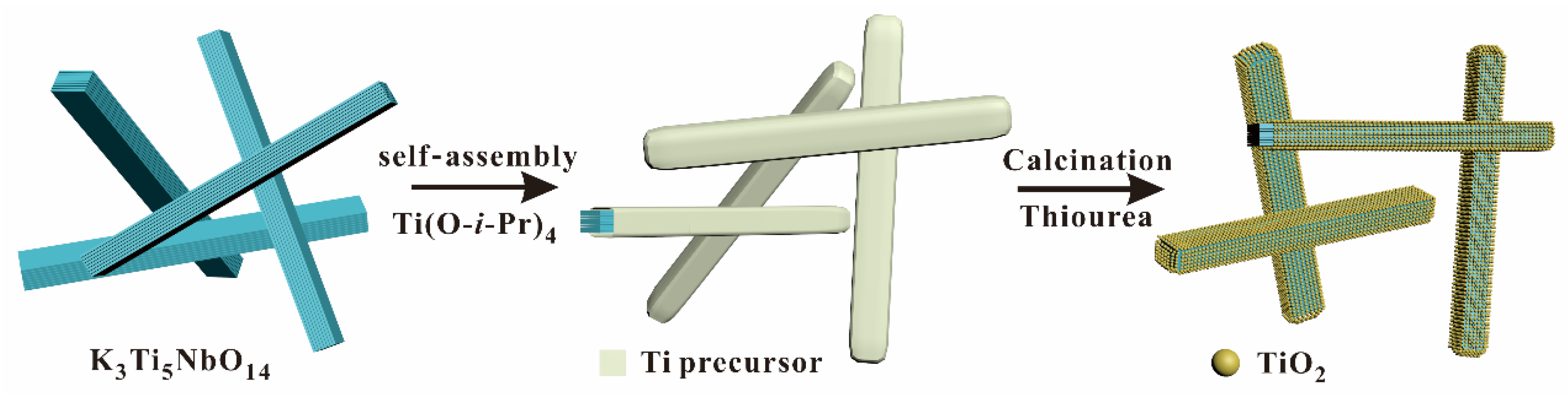
3.1. Structure and Morphology
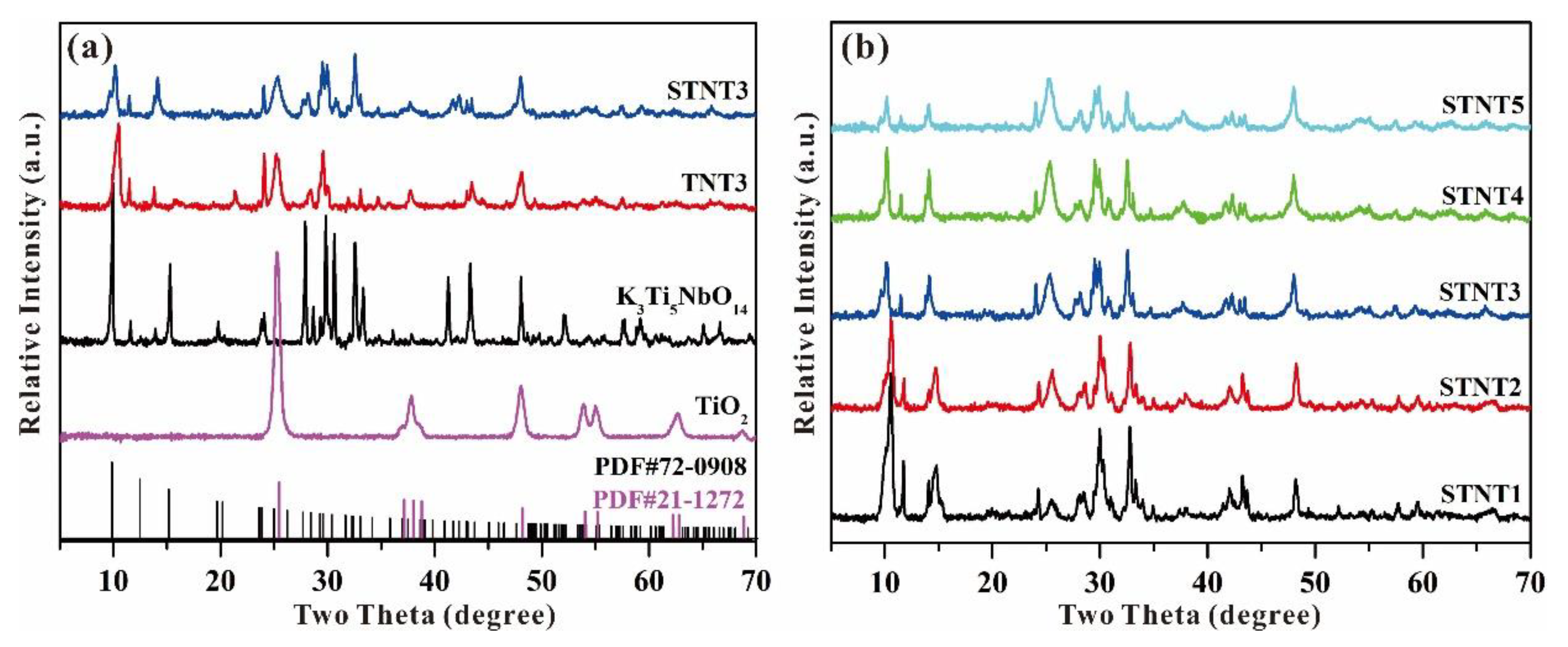
3.2. Powder X-Ray Diffraction Analysis
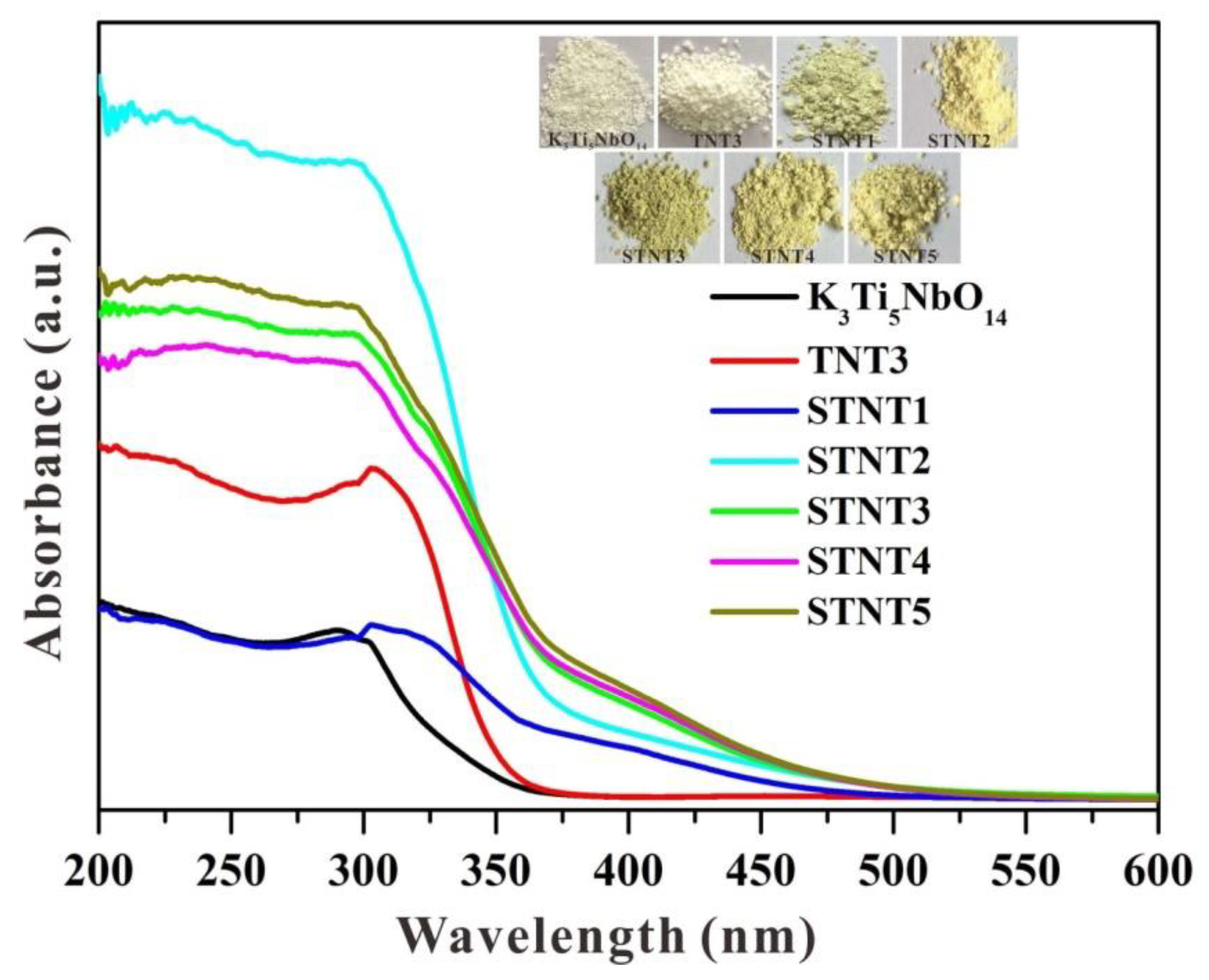
3.3. UV-Vis Analysis
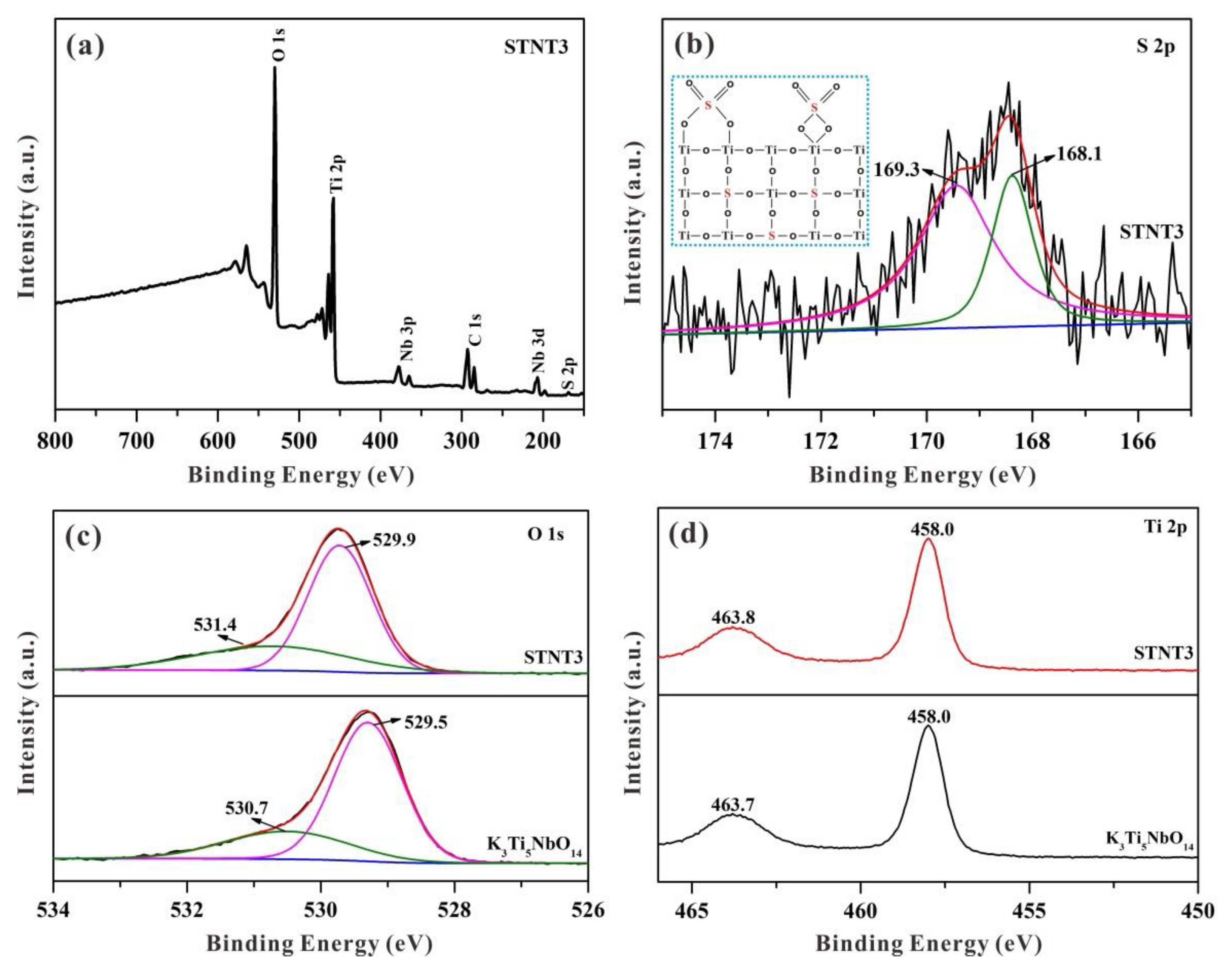
3.4. XPS Analysis
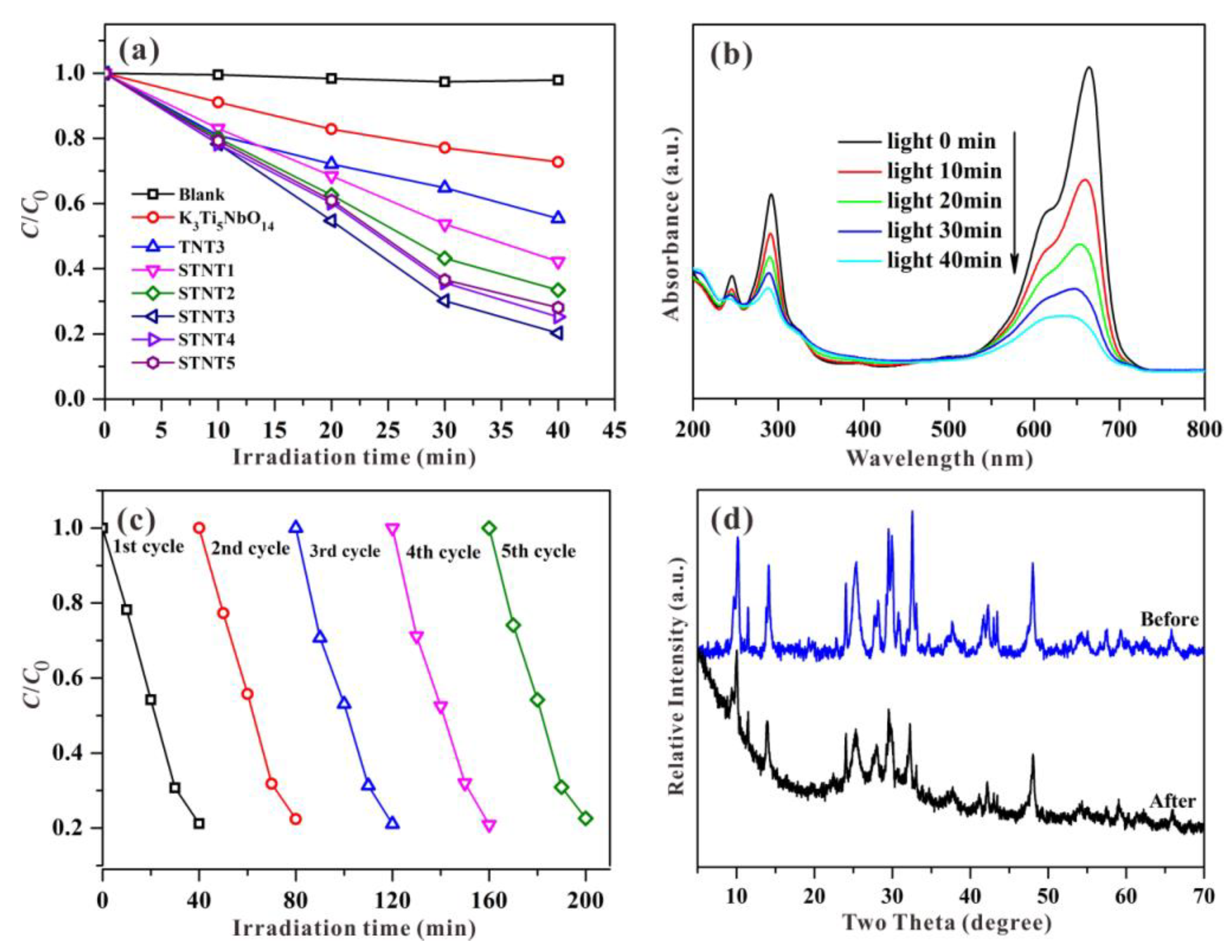
3.5. Photocatalytic Activity
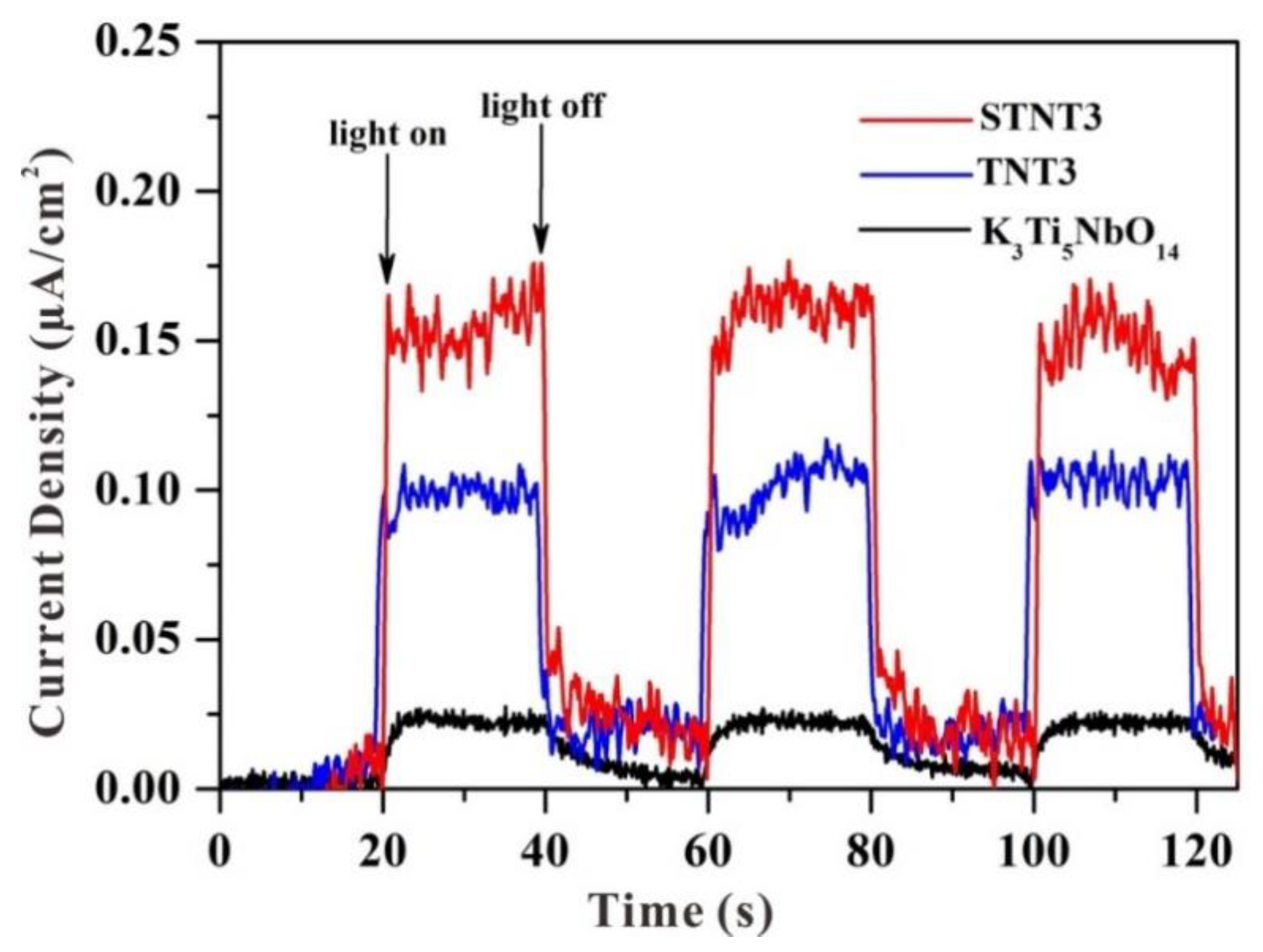
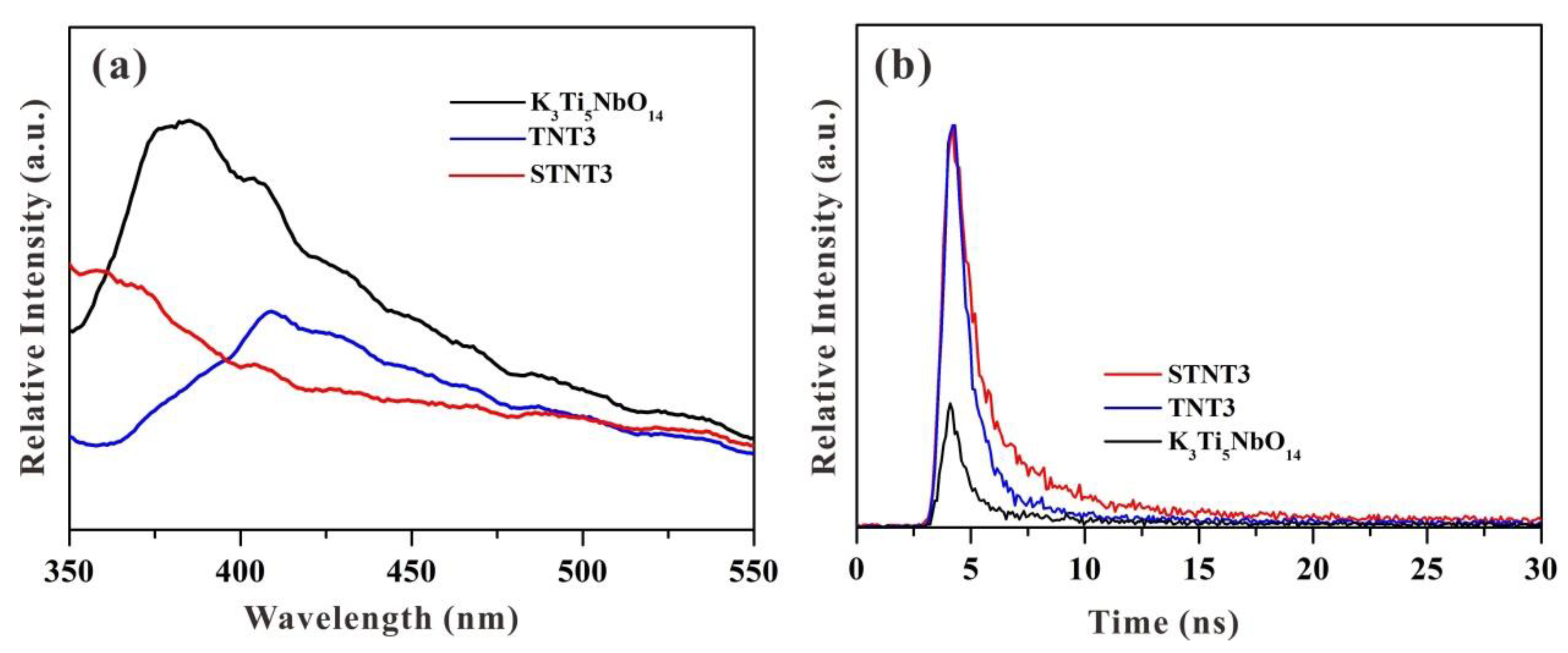
3.6. Electrochemical Analysis and PL Analysis
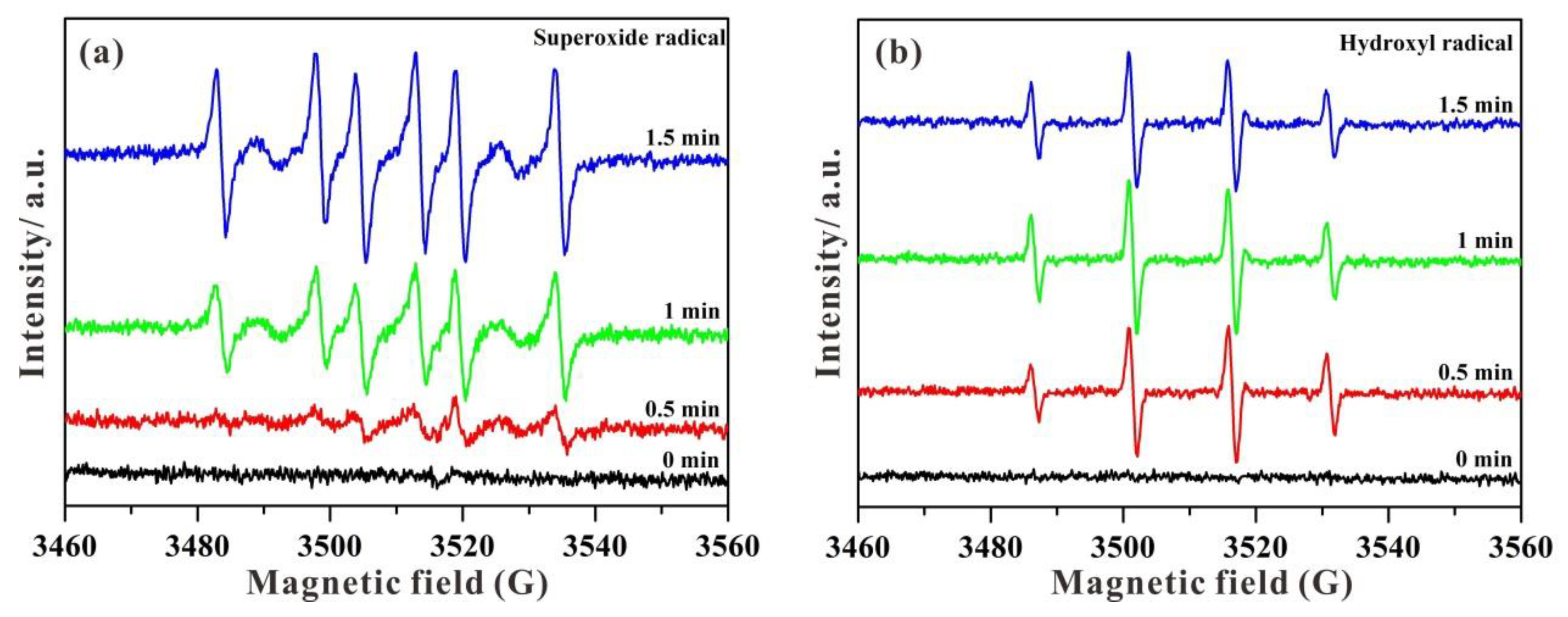
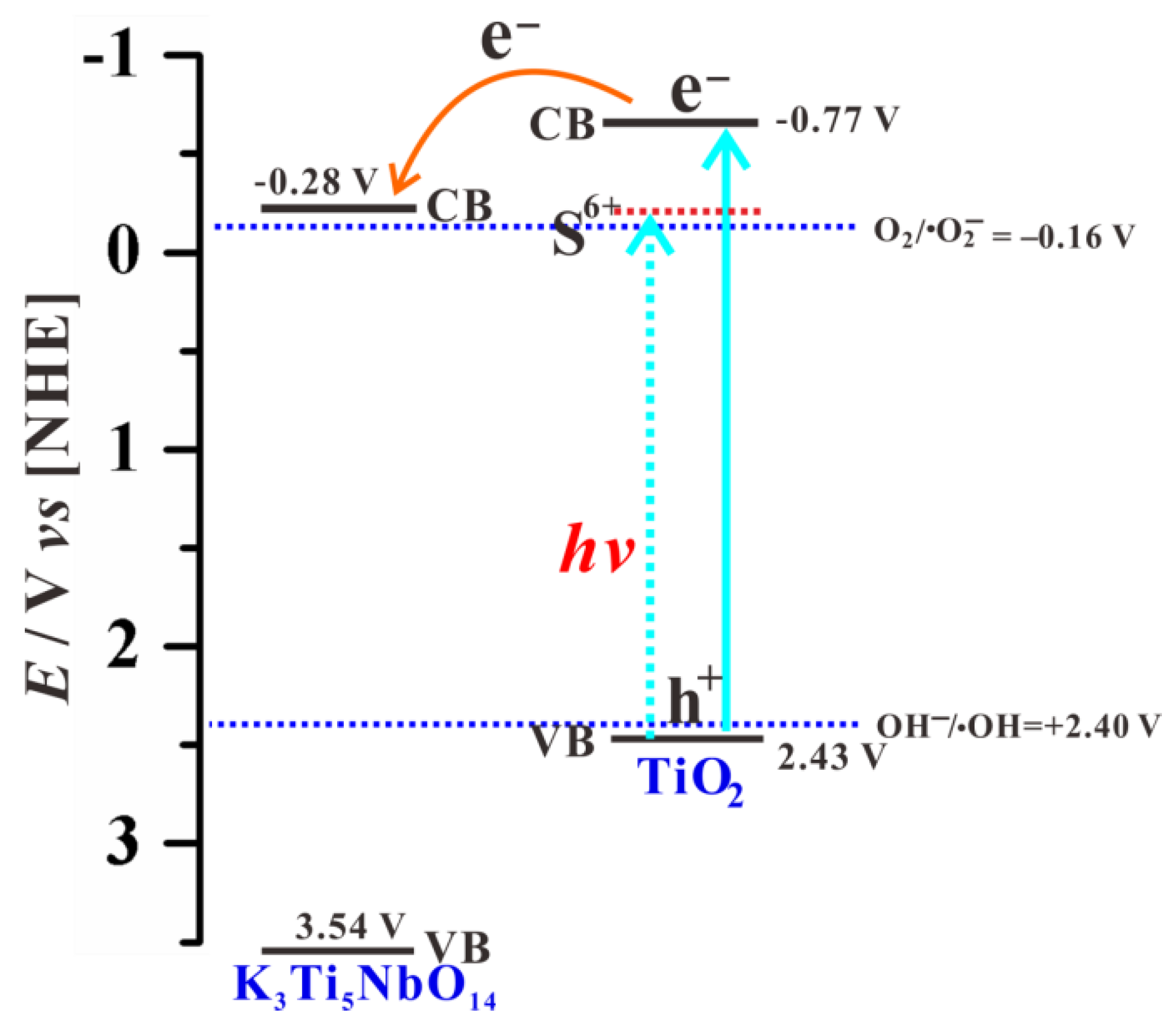
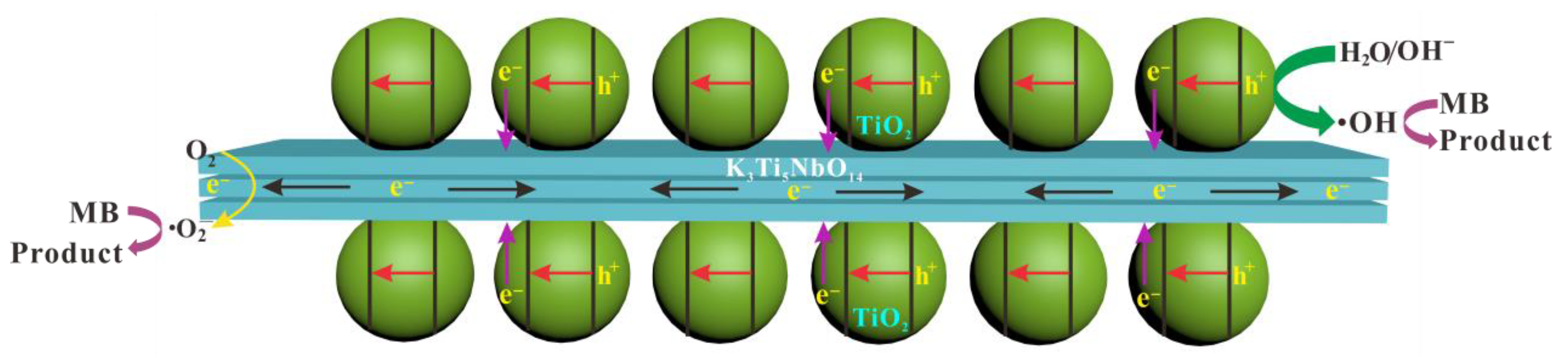
3.7. Photocatalytic Mechanism
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- He, R.; Xu, D.; Cheng, B.; Yu, J.; Ho, W. Review on nanoscale Bi-based photocatalysts. Nanoscale Horiz. 2018, 3, 464–504. [Google Scholar] [CrossRef]
- Fu, J.; Yu, J.; Jiang, C.; Cheng, B. g-C3N4-Based heterostructured photocatalysts. Adv. Energy Mater. 2018, 8, 1701503. [Google Scholar] [CrossRef]
- Ren, J.; Ouyang, S.; Chen, H.; Umezawa, N.; Lu, D.; Wang, D.; Xu, H.; Ye, J. Effective mineralization of organic dye under visible-light irradiation over electronic-structure-modulated Sn(Nb1−xTax)2O6 solid solutions. Appl. Catal. B 2015, 168, 243–249. [Google Scholar] [CrossRef]
- Li, X.; Yu, J.; Jaroniec, M. Hierarchical photocatalysts. Chem. Soc. Rev. 2016, 45, 2603–2636. [Google Scholar] [CrossRef]
- Zhong, W.; Shen, S.; Feng, S.; Lin, Z.; Wang, Z.; Fang, B. Facile fabrication of alveolate Cu2−xSe microsheets as a new visible-light photocatalyst for discoloration of Rhodamine B. CrystEngComm 2018, 20, 7851–7856. [Google Scholar] [CrossRef]
- Chen, X.; Liu, L.; Huang, F. Black titanium dioxide (TiO2) nanomaterials. Chem. Soc. Rev. 2015, 44, 1861–1885. [Google Scholar] [CrossRef]
- Sun, S.; Song, P.; Cui, J.; Liang, S. Amorphous TiO2 nanostructures: Synthesis, fundamental properties and photocatalytic applications. Catal. Sci. Technol. 2019, 9, 4198–4215. [Google Scholar] [CrossRef]
- Guo, Z.; Wang, Q.; Shen, T.; Hou, X.; Kuang, J.; Liu, W.; Cao, W. Synthesis of 3D CQDs/urchin-like and yolk-shell TiO2 hierarchical structure with enhanced photocatalytic properties. Ceram. Int. 2019, 45, 5858–5865. [Google Scholar] [CrossRef]
- Oseghe, E.O.; Msagati, T.A.M.; Mamba, B.B.; Ofomaja, A.E. An efficient and stable narrow bandgap carbon dot-brookite composite over other CD-TiO2 polymorphs in rhodamine B degradation under LED light. Ceram. Int. 2019, 45, 14173–14181. [Google Scholar] [CrossRef]
- Xiong, H.; Wu, L.; Liu, Y.; Gao, T.; Li, K.; Long, Y.; Zhang, R.; Zhang, L.; Qiao, Z.A.; Huo, Q.; et al. Controllable synthesis of mesoporous TiO2 polymorphs with tunable crystal structure for enhanced photocatalytic H2 production. Adv. Energy Mater. 2019, 9, 1901634. [Google Scholar] [CrossRef]
- Liu, C.; Zhang, C.; Wang, J.; Xu, Q.; Chen, X.; Wang, C.; Xi, X.; Hou, W. N-doped CsTi2NbO7@g-C3N4 core-shell nanobelts with enhanced visible light photocatalytic activity. Mater. Lett. 2018, 217, 235–238. [Google Scholar] [CrossRef]
- Zhai, Z.; Hu, C.H.; Yang, X.Y.; Zhang, L.H.; Liu, C.; Fan, Y.N.; Hou, W.H. Nitrogen-doped mesoporous nanohybrids of TiO2 nanoparticles and HTiNbO5 nanosheets with a high visible-light photocatalytic activity and a good biocompatibility. J. Mater. Chem. 2012, 22, 19122–19131. [Google Scholar] [CrossRef]
- Zhai, Z.; Huang, Y.C.; Xu, L.; Yang, X.Y.; Hu, C.H.; Zhang, L.H.; Fan, Y.N.; Hou, W.H. Thermostable nitrogen-doped HTiNbO5 nanosheets with a high visible-light photocatalytic activity. Nano Res. 2011, 4, 635–647. [Google Scholar] [CrossRef]
- D’Arienzo, M.; Carbajo, J.; Bahamonde, A.; Crippa, M.; Polizzi, S.; Scotti, R.; Wahba, L.; Morazzoni, F. Photogenerated defects in shape-controlled TiO2 anatase nanocrystals: A probe to evaluate the role of crystal facets in photocatalytic processes. J. Am. Chem. Soc. 2011, 133, 17652–17661. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, H.; Tomita, O.; Higashi, M.; Nakada, A.; Abe, R. Improved visible-light activity of nitrogen-doped layered niobate photocatalysts by NH3-nitridation with KCl flux. Appl. Catal. B 2018, 232, 49–54. [Google Scholar] [CrossRef]
- Shown, I.; Samireddi, S.; Chang, Y.C.; Putikam, R.; Chang, P.H.; Sabbah, A.; Fu, F.Y.; Chen, W.F.; Wu, C.I.; Yu, T.Y.; et al. Carbon-doped SnS2 nanostructure as a high-efficiency solar fuel catalyst under visible light. Nat. Commun. 2018, 9, 169. [Google Scholar] [CrossRef]
- Ohno, T.; Akiyoshi, M.; Umebayashi, T.; Asai, K.; Mitsui, T.; Matsumura, M. Preparation of S-doped TiO2 photocatalysts and their photocatalytic activities under visible light. Appl. Catal. A 2004, 265, 115–121. [Google Scholar] [CrossRef]
- Liu, C.; Han, R.; Ji, H.; Sun, T.; Zhao, J.; Chen, N.; Chen, J.; Guo, X.; Hou, W.; Ding, W. S-doped mesoporous nanocomposite of HTiNbO5 nanosheets and TiO2 nanoparticles with enhanced visible light photocatalytic activity. Phys. Chem. Chem. Phys. 2016, 18, 801–810. [Google Scholar] [CrossRef]
- Liu, C.; Liang, J.; Han, R.; Wang, Y.; Zhao, J.; Huang, Q.; Chen, J.; Hou, W. S-doped Na2Ti6O13@TiO2 core-shell nanorods with enhanced visible light photocatalytic performance. Phys. Chem. Chem. Phys. 2015, 17, 15165–15172. [Google Scholar] [CrossRef]
- Al-Keisy, A.; Ren, L.; Xu, X.; Hao, W.; Dou, S.X.; Du, Y. Selective ferroelectric BiOI/Bi4Ti3O12 heterostructures for visible light-driven photocatalysis. J. Phys. Chem. C 2018, 123, 517–525. [Google Scholar] [CrossRef]
- Yang, J.; Liang, Y.; Li, K.; Yang, G.; Yin, S. One-step low-temperature synthesis of 0D CeO2 quantum dots/2D BiOX (X = Cl, Br) nanoplates heterojunctions for highly boosting photo-oxidation and reduction ability. Appl. Catal. B 2019, 250, 17–30. [Google Scholar] [CrossRef]
- Liu, C.; Xu, G.; Zhu, Y.; Xu, Q.; Yu, G.; Hou, H.; Xu, Q.; Xi, X.; Hou, W. In situ construction of layered K3Ti5NbO14/g-C3N4 composite for improving visible-light-driven photocatalytic performance. J. Mater. Sci. 2018, 29, 15859–15868. [Google Scholar] [CrossRef]
- Park, S.; Lee, J.M.; Jo, Y.K.; Kim, I.Y.; Hwang, S.J. A facile exfoliation-crystal growth route to multicomponent Ag2CO3/Ag-Ti5NbO14 nanohybrids with improved visible light photocatalytic activity. Dalton Trans. 2014, 43, 10566–10573. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Sun, T.; Wu, L.; Liang, J.; Huang, Q.; Chen, J.; Hou, W. N-doped Na2Ti6O13@TiO2 core–shell nanobelts with exposed {1 0 1} anatase facets and enhanced visible light photocatalytic performance. Appl. Catal. B 2015, 170, 17–24. [Google Scholar] [CrossRef]
- Wang, Y.; Jiang, W.; Luo, W.; Chen, X.; Zhu, Y. Ultrathin nanosheets g-C3N4@Bi2WO6 core-shell structure via low temperature reassembled strategy to promote photocatalytic activity. Appl. Catal. B 2018, 237, 633–640. [Google Scholar] [CrossRef]
- Wang, Q.; Wang, W.; Zhong, L.; Liu, D.; Cao, X.; Cui, F. Oxygen vacancy-rich 2D/2D BiOCl-g-C3N4 ultrathin heterostructure nanosheets for enhanced visible-light-driven photocatalytic activity in environmental remediation. Appl. Catal. B 2018, 220, 290–302. [Google Scholar] [CrossRef]
- Liu, G.; Yang, H.G.; Pan, J.; Yang, Y.Q.; Lu, G.Q.; Cheng, H.M. Titanium dioxide crystals with tailored facets. Chem. Rev. 2014, 114, 9559–9612. [Google Scholar] [CrossRef]
- Xia, Y.; Liang, S.; Wu, L.; Wang, X. Ultrasmall NiS decorated HNb3O8 nanosheeets as highly efficient photocatalyst for H2 evolution reaction. Catal. Today 2019, 330, 195–202. [Google Scholar] [CrossRef]
- Chen, Z.J.; Lin, B.Z.; Chen, Y.L.; Zhang, K.Z.; Li, B.; Zhu, H. Pillaring and photocatalytic properties of mesoporous α-Fe2O3/titanate nanocomposites via an exfoliation and restacking route. J. Phys. Chem. Solids 2010, 71, 841–847. [Google Scholar] [CrossRef]
- Du, G.H.; Yu, Y.; Chen, Q.; Wang, R.H.; Zhou, W.; Peng, L.M. Exfoliating KTiNbO5 particles into nanosheets. Chem. Phys. Lett. 2003, 377, 445–448. [Google Scholar] [CrossRef]
- Yu, J.C.; Ho, W.; Yu, J.; Yip, H.; Wong, P.K.; Zhao, J. Efficient visible-light-Induced photocatalytic disinfection on sulfur-doped nanocrystalline titania. Environ. Sci. Technol. 2005, 39, 1175–1179. [Google Scholar] [CrossRef] [PubMed]
- Nasir, M.; Xi, Z.; Xing, M.; Zhang, J.; Chen, F.; Tian, B.; Bagwasi, S. Study of synergistic effect of Ce- and S-codoping on the enhancement of visible-light photocatalytic activity of TiO2. J. Phys. Chem. C 2013, 117, 9520–9528. [Google Scholar] [CrossRef]
- El-Sheikh, S.M.; Zhang, G.; El-Hosainy, H.M.; Ismail, A.A.; O’Shea, K.E.; Falaras, P.; Kontos, A.G.; Dionysiou, D.D. High performance sulfur, nitrogen and carbon doped mesoporous anatase–brookite TiO2 photocatalyst for the removal of microcystin-LR under visible light irradiation. J. Hazard. Mater. 2014, 280, 723–733. [Google Scholar] [CrossRef] [PubMed]
- Devi, L.G.; Kavitha, R. Enhanced photocatalytic activity of sulfur doped TiO2 for the decomposition of phenol: A new insight into the bulk and surface modification. Mater. Chem. Phys. 2014, 143, 1300–1308. [Google Scholar] [CrossRef]
- Song, Y.; Wang, H.; Xiong, J.; Guo, B.; Liang, S.; Wu, L. Photocatalytic hydrogen evolution over monolayer H1.07Ti1.73O4·H2O nanosheets: Roles of metal defects and greatly enhanced performances. Appl. Catal. B 2018, 221, 473–481. [Google Scholar] [CrossRef]
- Tan, S.; Xing, Z.; Zhang, J.; Li, Z.; Wu, X.; Cui, J.; Kuang, J.; Zhu, Q.; Zhou, W. Ti3+-TiO2/g-C3N4 mesostructured nanosheets heterojunctions as efficient visible-light-driven photocatalysts. J. Catal. 2018, 357, 90–99. [Google Scholar] [CrossRef]
- Liu, C.; Zhu, H.; Zhu, Y.; Dong, P.; Hou, H.; Xu, Q.; Chen, X.; Xi, X.; Hou, W. Ordered layered N-doped KTiNbO5/g-C3N4 heterojunction with enhanced visible light photocatalytic activity. Appl. Catal. B 2018, 228, 54–63. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, H.; Tu, W.; Liu, Y.; Tan, Y.Z.; Yuan, X.; Chew, J.W. Quasi-polymeric construction of stable perovskite-type LaFeO3/g-C3N4 heterostructured photocatalyst for improved Z-scheme photocatalytic activity via solid p-n heterojunction interfacial effect. J. Hazard. Mater. 2018, 347, 412–422. [Google Scholar] [CrossRef]
- Peng, Z.; Jiang, Y.; Wang, X.; Xu, R.H.; Xiao, Y.; Jing, X.; Zhang, J.; Liu, Y.; Ni, L. Novel CdIn2S4 nano-octahedra/TiO2 hollow hybrid heterostructure: In-situ synthesis, synergistic effect and enhanced dual-functional photocatalytic activities. Ceram. Int. 2019, 45, 15942–15953. [Google Scholar] [CrossRef]
- Yan, Y.; Yang, M.; Shi, H.; Wang, C.; Fan, J.; Liu, E.; Hu, X. CuInS2 sensitized TiO2 for enhanced photodegradation and hydrogen production. Ceram. Int. 2019, 45, 6093–6101. [Google Scholar] [CrossRef]
- Zhou, H.; Zhong, S.; Shen, M.; Yao, Y. Composite soft template-assisted construction of a flower-like β-Bi2O3/Bi2O2CO3 heterojunction photocatalyst for the enhanced simulated sunlight photocatalytic degradation of tetracycline. Ceram. Int. 2019, 45, 15036–15047. [Google Scholar] [CrossRef]
- Xu, J.; Yan, X.; Qi, Y.; Fu, Y.; Wang, C.; Wang, L. Novel phosphidated MoS2 nanosheets modified CdS semiconductor for an efficient photocatalytic H2 evolution. Chem. Eng. J. 2019, 375, 122053. [Google Scholar] [CrossRef]
- Zhong, W.; Shen, S.; He, M.; Wang, D.; Wang, Z.; Lin, Z.; Tu, W.; Yu, J. The pulsed laser-induced Schottky junction via in-situ forming Cd clusters on CdS surfaces toward efficient visible light-driven photocatalytic hydrogen evolution. Appl. Catal. B 2019, 258, 117967. [Google Scholar] [CrossRef]
- Wei, Z.; Liu, J.; Fang, W.; Xu, M.; Qin, Z.; Jiang, Z.; Shangguan, W. Photocatalytic hydrogen evolution with simultaneous antibiotic wastewater degradation via the visible-light-responsive bismuth spheres-g-C3N4 nanohybrid: Waste to energy insight. Chem. Eng. J. 2019, 358, 944–954. [Google Scholar] [CrossRef]
- Zhao, L.; Qi, Y.; Song, L.; Ning, S.; Ouyang, S.; Xu, H.; Ye, J. Solar-driven water–gas shift reaction over CuOx/Al2O3 with 1.1 % of light-to-energy storage. Angew. Chem. Int. Ed. 2019, 58, 7708–7712. [Google Scholar] [CrossRef] [PubMed]
- Zhong, W.; Tu, W.; Feng, S.; Xu, A. Photocatalytic H2 evolution on CdS nanoparticles by loading FeSe nanorods as co-catalyst under visible light irradiation. J. Alloys Compd. 2019, 772, 669–674. [Google Scholar] [CrossRef]
- Li, X.; Yang, G.; Li, S.; Xiao, N.; Li, N.; Gao, Y.; Lv, D.; Ge, L. Novel dual co-catalysts decorated Au@HCS@PdS hybrids with spatially separated charge carriers and enhanced photocatalytic hydrogen evolution activity. Chem. Eng. J. 2010, 379, 122350. [Google Scholar] [CrossRef]
- Zhao, Z.; Wu, J.; Zheng, Y.Z.; Li, N.; Li, X.; Tao, X. Ni3C-Decorated MAPbI3 as visible-light photocatalyst for H2 evolution from HI splitting. ACS Catal. 2019, 9, 8144–8152. [Google Scholar] [CrossRef]
- Cao, S.; Shen, B.; Tong, T.; Fu, J.; Yu, J. 2D/2D Heterojunction of ultrathin MXene/Bi2WO6 nanosheets for improved photocatalytic CO2 reduction. Adv. Funct. Mater. 2018, 28, 1800136. [Google Scholar] [CrossRef]
- Liang, X.; Zhang, Y.; Li, D.; Wen, B.; Jiang, D.; Chen, M. 2D/2D BiOCl/K+Ca2Nb3O10− heterostructure with Z-scheme charge carrier transfer pathways for tetracycline degradation under simulated solar light. Appl. Surf. Sci. 2019, 466, 863–873. [Google Scholar] [CrossRef]
- Wang, K.; Li, Y.; Zhang, G.; Li, J.; Wu, X. 0D Bi nanodots/2D Bi3NbO7 nanosheets heterojunctions for efficient visible light photocatalytic degradation of antibiotics: Enhanced molecular oxygen activation and mechanism insight. Appl. Catal. B 2019, 240, 39–49. [Google Scholar] [CrossRef]
- Liu, C.; Xu, Q.; Zhang, Q.; Zhu, Y.; Ji, M.; Tong, Z.; Hou, W.; Zhang, Y.; Xu, J. Layered BiOBr/Ti3C2 MXene composite with improved visible-light photocatalytic activity. J. Mater. Sci. 2019, 54, 2458–2471. [Google Scholar] [CrossRef]
- Yang, X.; Qian, F.; Wang, Y.; Li, M.; Lu, J.; Li, Y.; Bao, M. Constructing a novel ternary composite (C16H33(CH3)3N)4W10O32/g-C3N4/rGO with enhanced visible-light-driven photocatalytic activity for degradation of dyes and phenol. Appl. Catal. B 2017, 200, 283–296. [Google Scholar] [CrossRef]
- Li, Y.; Lv, K.; Ho, W.; Dong, F.; Wu, X.; Xia, Y. Hybridization of rutile TiO2 (rTiO2) with g-C3N4 quantum dots (CN QDs): An efficient visible-light-driven Z-scheme hybridized photocatalyst. Appl. Catal. B 2017, 202, 611–619. [Google Scholar] [CrossRef]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, C.; Gao, X.; Han, Z.; Sun, Y.; Feng, Y.; Yu, G.; Xi, X.; Zhang, Q.; Zou, Z. Core-Shell Heterostructured and Visible-Light-Driven Titanoniobate/TiO2 Composite for Boosting Photodegradation Performance. Nanomaterials 2019, 9, 1503. https://doi.org/10.3390/nano9101503
Liu C, Gao X, Han Z, Sun Y, Feng Y, Yu G, Xi X, Zhang Q, Zou Z. Core-Shell Heterostructured and Visible-Light-Driven Titanoniobate/TiO2 Composite for Boosting Photodegradation Performance. Nanomaterials. 2019; 9(10):1503. https://doi.org/10.3390/nano9101503
Chicago/Turabian StyleLiu, Chao, Xin Gao, Zitong Han, Yao Sun, Yue Feng, Guiyun Yu, Xinguo Xi, Qinfang Zhang, and Zhigang Zou. 2019. "Core-Shell Heterostructured and Visible-Light-Driven Titanoniobate/TiO2 Composite for Boosting Photodegradation Performance" Nanomaterials 9, no. 10: 1503. https://doi.org/10.3390/nano9101503
APA StyleLiu, C., Gao, X., Han, Z., Sun, Y., Feng, Y., Yu, G., Xi, X., Zhang, Q., & Zou, Z. (2019). Core-Shell Heterostructured and Visible-Light-Driven Titanoniobate/TiO2 Composite for Boosting Photodegradation Performance. Nanomaterials, 9(10), 1503. https://doi.org/10.3390/nano9101503





