Preparation of Pd/C by Atmospheric-Pressure Ethanol Cold Plasma and Its Preparation Mechanism
Abstract
1. Introduction
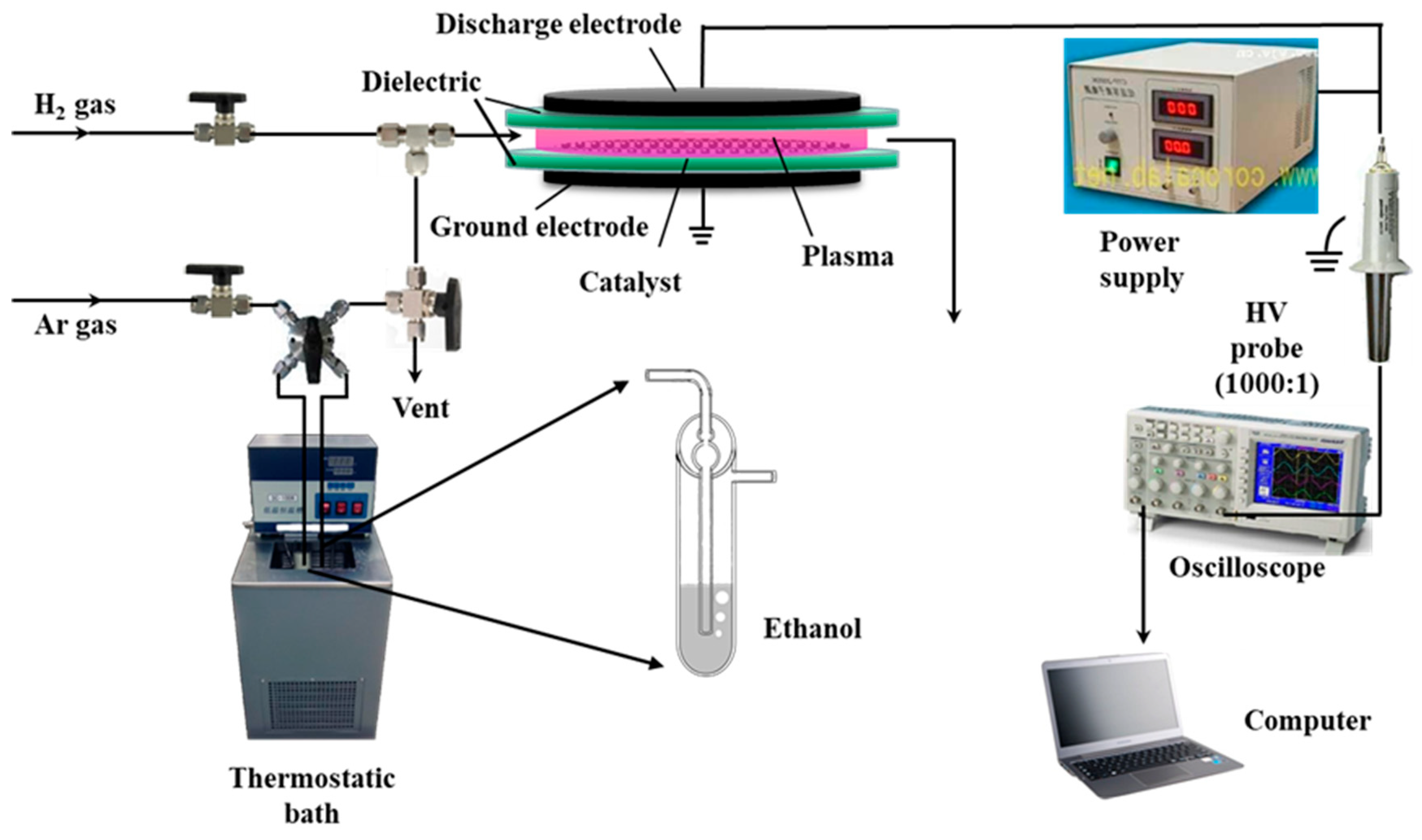
2. Experimental
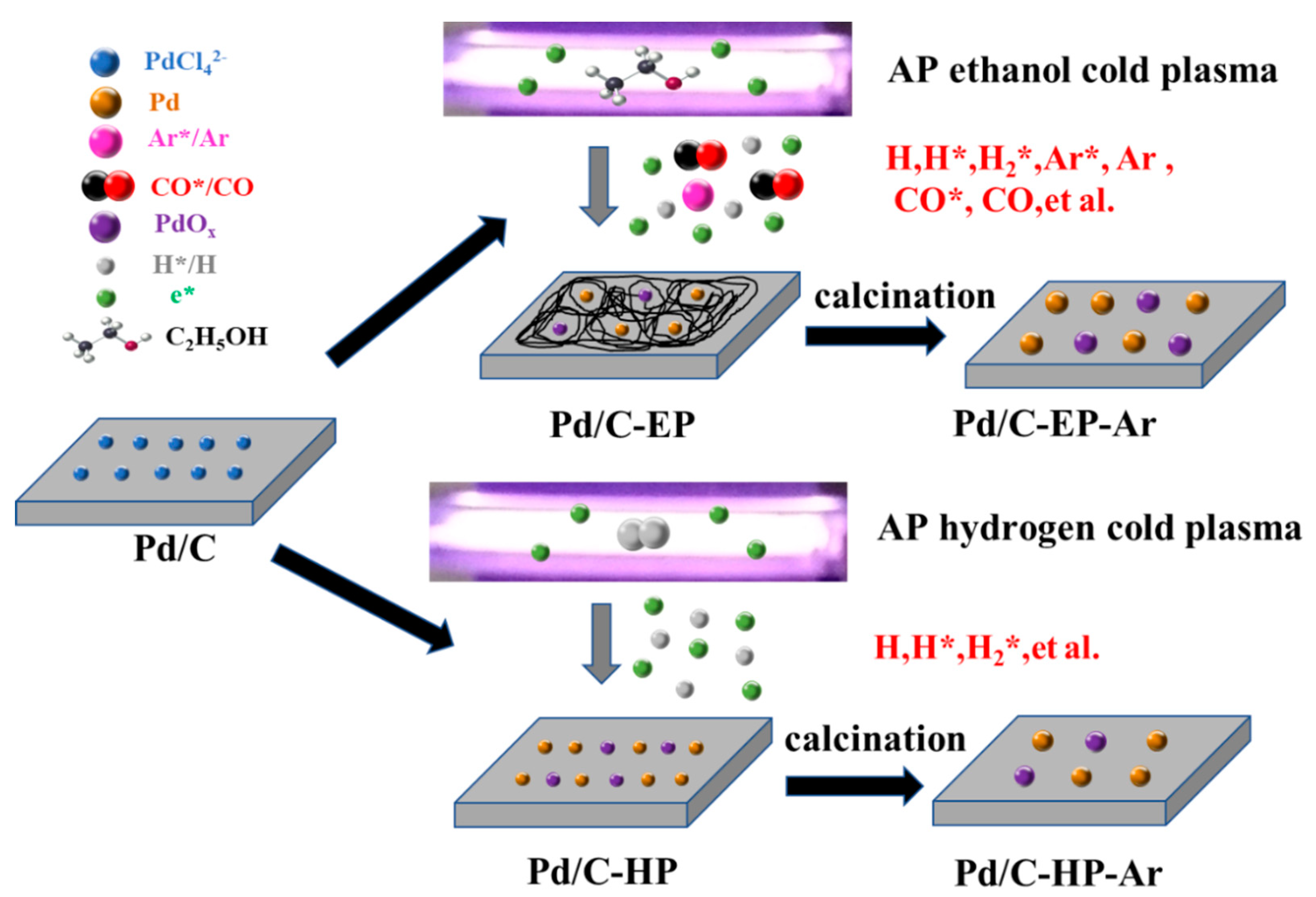
2.1. Catalysts Preparation
2.2. Catalysts Characterization and Activity Test
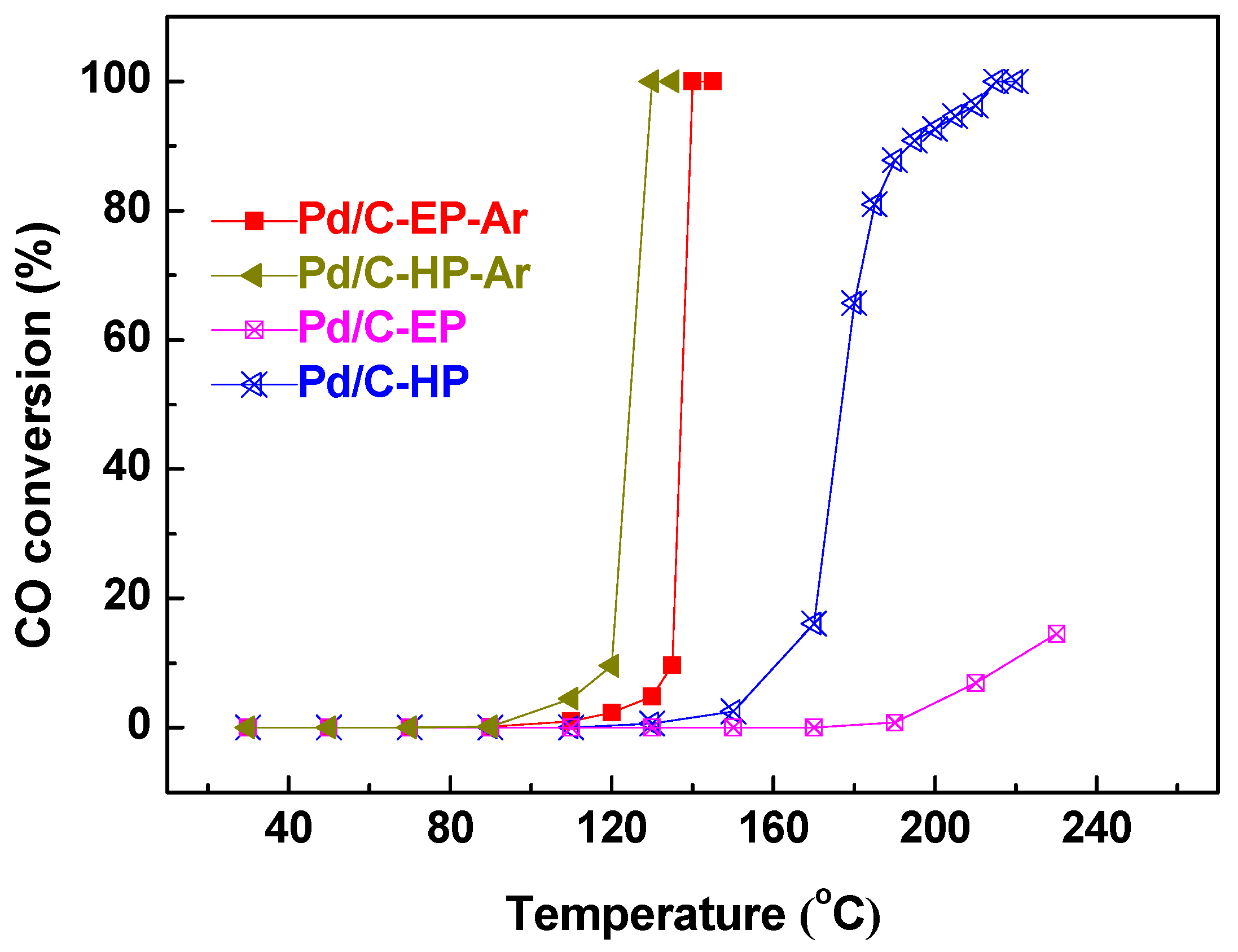
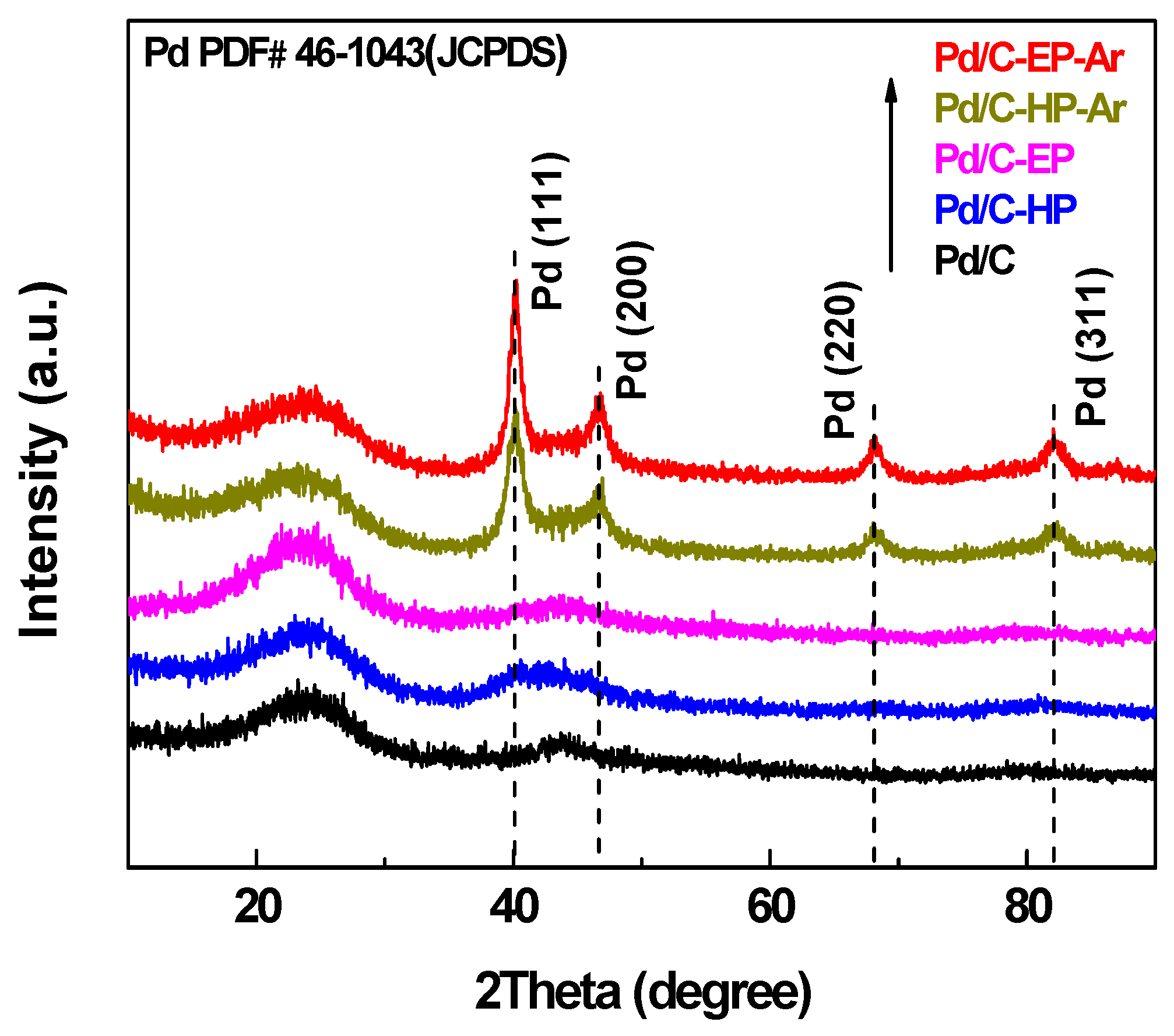
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Fihri, A.; Cha, D.; Bouhrara, M.; Almana, N.; Polshettiwar, V. Fibrous nano-silica (KCC-1) -supported palladium catalyst: Suzuki coupling reactions under sustainable conditions. ChemSusChem 2012, 5, 85–89. [Google Scholar] [CrossRef]
- Zeng, M.F.; Zhang, X.; Shao, L.J.; Qi, C.Z.; Zhang, X.M. Highly porous chitosan microspheres supported palladium catalyst for coupling reactions in organic and aqueous solutions. J. Organomet. Chem. 2012, 704, 29–37. [Google Scholar] [CrossRef]
- Li, Y.Z.; Yu, Y.; Wang, J.G.; Song, J.; Li, Q.; Dong, M.D.; Liu, C.J. CO oxidation over graphene supported palladium catalyst. Appl. Catal. B Environ. 2012, 125, 189–196. [Google Scholar] [CrossRef]
- Monguchi, Y.; Wakayama, F.; Ueda, S.; Ito, R.; Takada, H.; Inoue, H.; Nakamura, A.; Sawama, Y.; Sajiki, H. Amphipathic monolith-supported palladium catalysts for chemoselective hydrogenation and cross-coupling reactions. RSC Adv. 2017, 7, 1833–1840. [Google Scholar] [CrossRef]
- Blaser, H.U.; Indolese, A.; Schnyder, A.; Steiner, H.; Studer, M. Supported palladium catalysts for fine chemicals synthesis. J. Mol. Catal. A Chem. 2001, 173, 3–18. [Google Scholar] [CrossRef]
- Zhao, F.Y.; Bhanage, B.M.; Shirai, M.; Arai, M. Heck reactions of iodobenzene and methyl acrylate with conventional supported palladium catalysts in the presence of organic and/or inorganic bases without ligands. Chem. Eur. J. 2000, 6, 843–848. [Google Scholar] [CrossRef]
- Sekizawa, K.; Widjaja, H.; Maeda, S.; Ozawab, Y.; Eguchia, K. Low temperature oxidation of methane over Pd catalyst supported on metal oxides. Catal. Today 2000, 59, 69–74. [Google Scholar] [CrossRef]
- Xiao, L.H.; Sun, K.P.; Xu, X.L.; Li, X.N. Low-temperature catalytic combustion of methane over Pd/CeO2 prepared by deposition-precipitation method. Catal. Commun. 2005, 6, 796–801. [Google Scholar] [CrossRef]
- Vichaphund, S.; Aht-ong, D.; Sricharoenchaikul, V.; Atong, D. Production of aromatic compounds from catalytic fast pyrolysis of Jatropha residues using metal/HZSM-5 prepared by ion-exchange and impregnation methods. Renew. Energy 2015, 79, 28–37. [Google Scholar] [CrossRef]
- Hu, S.; Scudiero, L.; Ha, S. Electronic effect on oxidation of formic acid on supported Pd–Cu bimetallic surface. Electrochim. Acta 2012, 83, 354–358. [Google Scholar] [CrossRef]
- Yang, M.M.; Wang, Z.Y.; Wang, W.; Liu, C.J. Synthesis of AuPd alloyed nanoparticles via room-temperature electron reduction with argon glow discharge as electron source. Nanoscale Res. Lett. 2014, 9, 405. [Google Scholar] [CrossRef]
- Ouyang, L.Z.; Cao, Z.J.; Wang, H.; Hu, R.Z.; Zhu, M. Application of dielectric barrier discharge plasma-assisted milling in energy storage materials-A review. J. Alloys Compd. 2017, 691, 422–435. [Google Scholar] [CrossRef]
- Deng, X.Q.; Zhu, B.; Li, X.S.; Liu, J.L.; Zhu, X.B.; Zhu, A.M. Visible-light photocatalytic oxidation of CO over plasmonic Au/TiO2: Unusual features of oxygen plasma activation. Appl. Catal. B Environ. 2016, 188, 48–55. [Google Scholar] [CrossRef]
- Di, L.B.; Xu, W.J.; Zhan, Z.B.; Zhang, X.L. Synthesis of alumina supported Pd–Cu alloy nanoparticles for CO oxidation via a fast and facile method. RSC Adv. 2015, 5, 71854–71858. [Google Scholar] [CrossRef]
- Gallon, H.J.; Tu, X.; Twigg, M.V.; Whitehead, J.C. Plasma-assisted methane reduction of a NiO catalyst low temperature activation of methane and formation of carbon nanofibres. Appl. Catal. B Environ. 2011, 106, 616–620. [Google Scholar] [CrossRef]
- Zhang, J.S.; Di, L.B.; Yu, F.; Duan, D.Z.; Zhang, X.L. Atmospheric-pressure cold plasma activating Au/P25 for CO oxidation: Effect of working gas. Nanomaterials 2018, 8, 742. [Google Scholar] [CrossRef]
- Ovanesyan, R.A.; Hausmann, D.M.; Agarwal, S. Low-temperature conformal atomic layer deposition of SiNx films using Si2Cl6 and NH3 plasma. ACS Appl. Mater. Interfaces 2015, 7, 10806–10813. [Google Scholar] [CrossRef]
- Guo, Z.F.; Yi, Y.H.; Wang, L.; Yang, J.H.; Guo, H.C. Pt/TS-1 catalyst promoted C–N coupling reaction in CH4–NH3 plasma for HCN synthesis at low temperature. ACS Catal. 2018, 8, 10219–10224. [Google Scholar] [CrossRef]
- Du, C.M.; Ma, D.Y.; Wu, J.; Lin, Y.C.; Xiao, W.; Ruan, J.J.; Huang, D.W. Plasma-catalysis reforming for H2 production from ethanol. Int. J. Hydrog. Energy 2015, 40, 15398–15410. [Google Scholar] [CrossRef]
- Xu, W.Q.; Liu, Z.Y.; Johnston-Peck, A.C.; Senanayake, S.D.; Zhou, G.; Stacchiola, D.; Stach, E.A.; Rodriguez, J.A. Steam reforming of ethanol on Ni/CeO2: Reaction pathway and interaction between Ni and the CeO2 support. ACS Catal. 2013, 3, 975–984. [Google Scholar] [CrossRef]
- Hou, T.F.; Yu, B.; Zhang, S.Y.; Xu, T.K.; Wang, D.Z.; Cai, W.J. Hydrogen production from ethanol steam reforming over Rh/CeO2 catalyst. Catal. Commun. 2015, 58, 137–140. [Google Scholar] [CrossRef]
- Wang, B.; Sun, B.; Zhu, X.M.; Yan, Z.Y.; Liu, Y.J.; Liu, H.; Liu, Q. Hydrogen production from alcohol solution by microwave discharge in liquid. Int. J. Hydrog. Energy 2016, 41, 7280–7291. [Google Scholar] [CrossRef]
- Wu, Y.W.; Chung, W.C.; Chang, M.B. Modification of Ni/γ-Al2O3 catalyst with plasma for steam reforming of ethanol to generate hydrogen. Int. J. Hydrog. Energy 2015, 40, 8071–8080. [Google Scholar] [CrossRef]
- Di, L.B.; Li, Z.; Zhang, X.L.; Wang, H.Y.; Fan, Z.Y. Reduction of supported metal ions by a safe atmospheric pressure alcohol cold plasma method. Catal. Today 2019, 337, 55–62. [Google Scholar] [CrossRef]
- Di, L.B.; Li, Z.; Lee, B.; Park, D.W. An alternative atmospheric-pressure cold plasma method for synthesizing Pd/P25 catalysts with the assistance of ethanol. Int. J. Hydrog. Energy 2017, 42, 11372–11378. [Google Scholar] [CrossRef]
- Li, Z.; Zhang, X.L.; Zhang, Y.Z.; Duan, D.Z.; Di, L.B. Hydrogen cold plasma for synthesizing Pd/C catalysts: The effect of support-metal ion interaction. Plasma Sci. Technol. 2018, 20, 014016. [Google Scholar] [CrossRef]
- Qi, B.; Di, L.D.; Xu, W.J.; Zhang, X.L. Dry plasma reduction to prepare a high performance Pd/C catalyst at atmospheric pressure for CO oxidation. J. Mater. Chem. A 2014, 2, 11885–11890. [Google Scholar] [CrossRef]
- Lazzarini, A.; Piovano, A.; Pellegrini, R.; Leofanti, G.; Agostini, G.; Rudić, S.; Chierotti, M.R.; Gobetto, R.; Battiato, A.; Spoto, G.; et al. A comprehensive approach to investigate the structural and surface properties of activated carbons and related Pd-based catalysts. Catal. Sci. Technol. 2016, 6, 4910–4922. [Google Scholar] [CrossRef]
- Di, L.B.; Duan, D.Z.; Park, D.W.; Ahn, W.S.; Lee, B.J.; Zhang, X.L. Cold plasma for synthesizing high performance bimetallic PdCu catalysts: Effect of reduction sequence and Pd/Cu atomic ratios. Top. Catal. 2017, 12–14, 925–933. [Google Scholar] [CrossRef]
- Di, L.B.; Zhang, J.S.; Zhang, X.L. A review on the recent progress, challenges, and perspectives of atmospheric-pressure cold plasma for preparation of supported metal catalysts. Plasma Processes Polym. 2018, 15, 1700234. [Google Scholar] [CrossRef]
- Fang, G.D.; Dionysiou, D.D.; Wang, Y.; Al-Abed, S.R.; Zhou, D.M. Sulfate radical-based degradation of polychlorinated biphenyls: Effects of chloride ion and reaction kinetics. J. Hazard. Mater. 2012, 227–228, 394–401. [Google Scholar] [CrossRef]
- Bao, L.S.; Zhang, X.F.; Yu, L.; Zhu, G.S. Study on the influence of chloride ion content on pavement performance of base material with fly-ash-flushed-by-seawater. Adv. Mater. Res. 2011, 194–196, 993–1000. [Google Scholar] [CrossRef]
- Tan, E.Z.; Yin, P.G.; Guo, L. SERS-activity of immobilized silver nanoparticles: Effect of chloride ion. J. Light Scatt. 2010, 22, 305–308. [Google Scholar] [CrossRef]
- Hsieh, Y.C.; Senanayake, S.D.; Zhang, Y.; Xu, W.Q.; Polyansky, D.E. Effect of chloride anions on the synthesis and enhanced catalytic activity of silver nanocoral electrodes for CO2 electroreduction. ACS Catal. 2015, 46, 2584–2592. [Google Scholar] [CrossRef]
- Di, L.B.; Zhang, X.L.; Lee, B.; Lu, P.; Ahn, W.S.; Park, D.W. Feasibility of atmospheric-pressure CO cold plasma for reduction of supported metal ions. Plasma Chem. Plasma Process. 2017, 37, 1535–1549. [Google Scholar] [CrossRef]
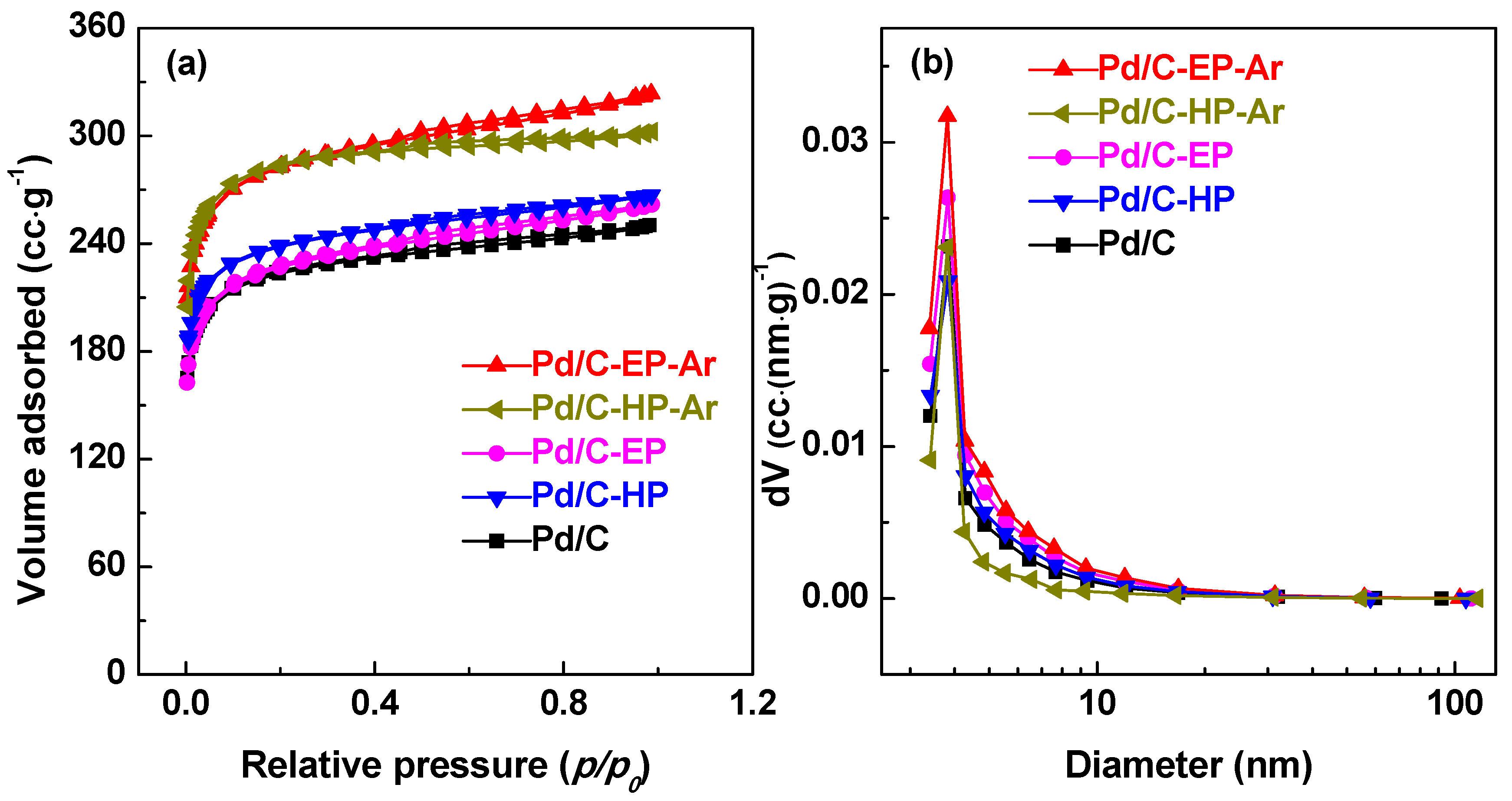
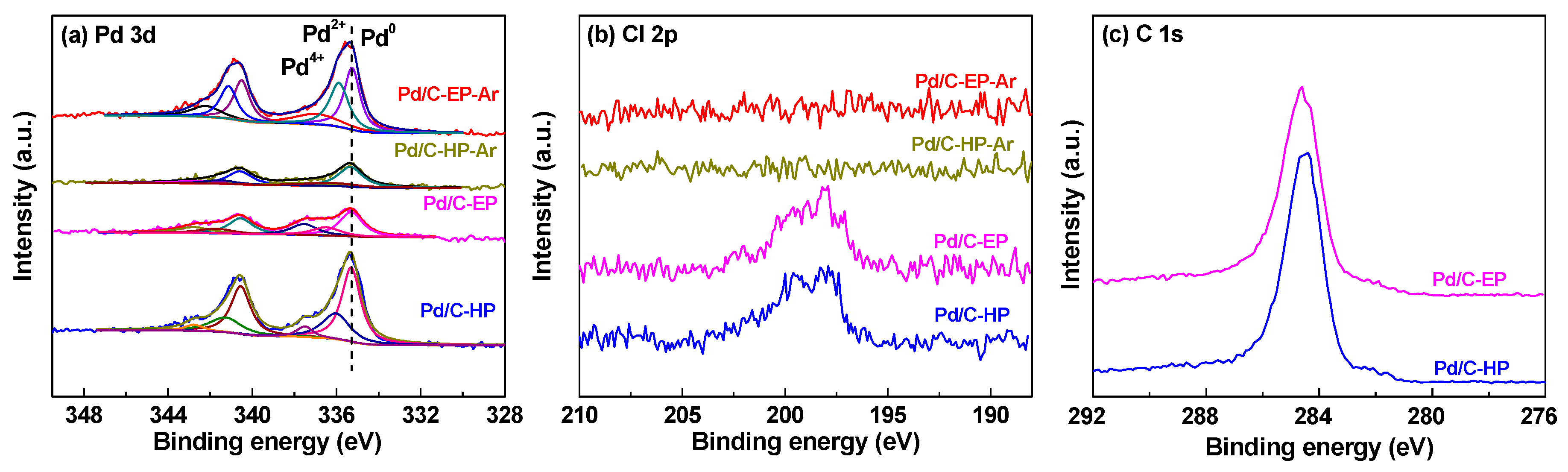
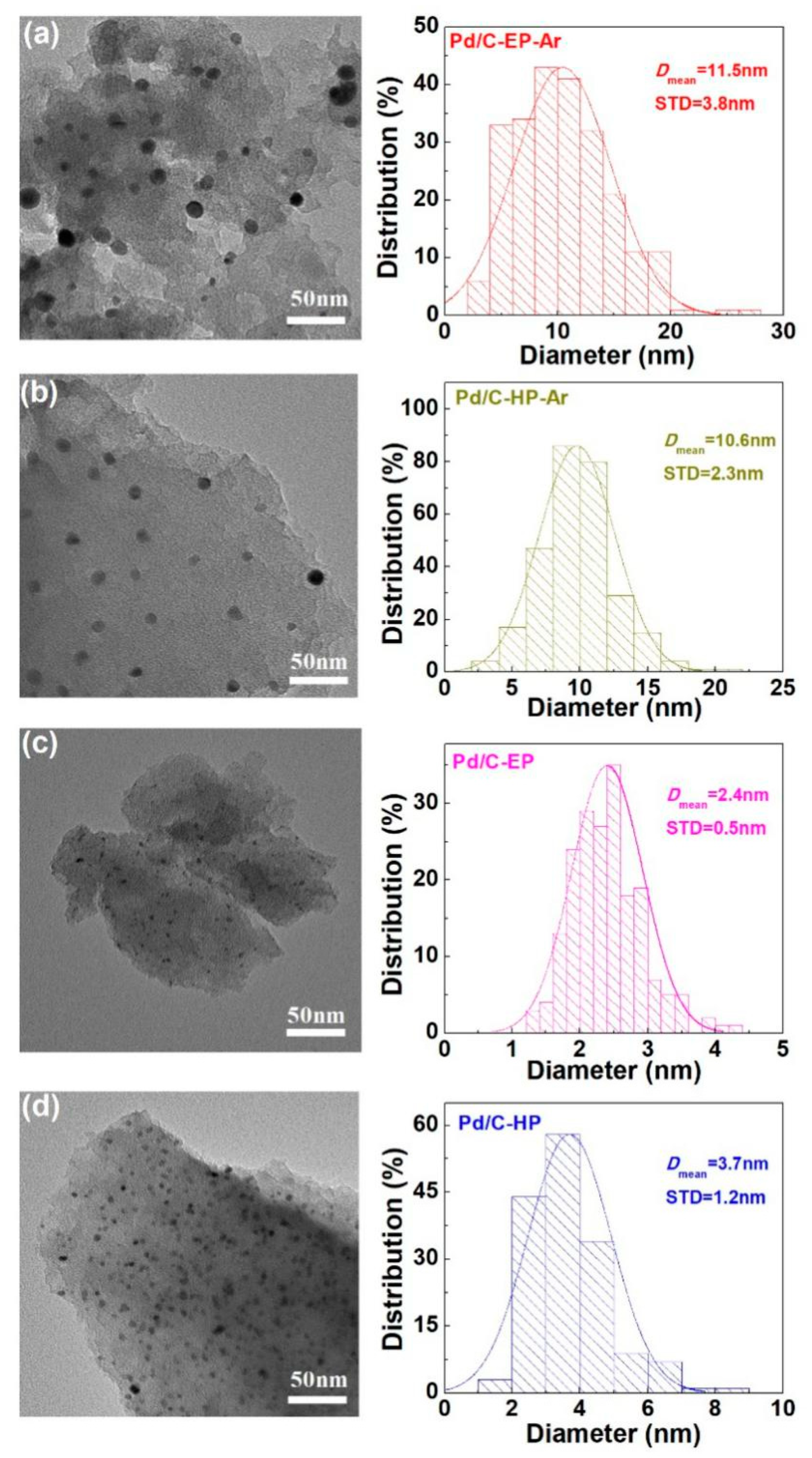
| Samples | Pore Diameter (nm) | Pore Volume (cm3·g−1) | Specific Surface Area (m2·g−1) |
|---|---|---|---|
| Pd/C-EP-Ar | 3.82 | 0.50 | 874 |
| Pd/C-HP-Ar | 3.82 | 0.47 | 871 |
| Pd/C-EP | 3.83 | 0.41 | 703 |
| Pd/C-HP | 3.83 | 0.41 | 736 |
| Pd/C | 3.84 | 0.39 | 690 |
| Catalyst | Pd Composition (%) | Pd/C Atomic Ratio | ||
|---|---|---|---|---|
| Pd0 | Pd2+ | Pd4+ | ||
| Pd/C-EP-Ar | 42.6 | 36.2 | 21.2 | 0.023 |
| Pd/C-HP-Ar | 58.7 | 19.8 | 21.5 | 0.010 |
| Pd/C-EP | 52.1 | 21.4 | 26.5 | 0.013 |
| Pd/C-HP | 61.4 | 31.1 | 7.5 | 0.026 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Z.; Zhang, J.; Wang, H.; Li, Z.; Zhang, X.; Di, L. Preparation of Pd/C by Atmospheric-Pressure Ethanol Cold Plasma and Its Preparation Mechanism. Nanomaterials 2019, 9, 1437. https://doi.org/10.3390/nano9101437
Li Z, Zhang J, Wang H, Li Z, Zhang X, Di L. Preparation of Pd/C by Atmospheric-Pressure Ethanol Cold Plasma and Its Preparation Mechanism. Nanomaterials. 2019; 9(10):1437. https://doi.org/10.3390/nano9101437
Chicago/Turabian StyleLi, Zhuang, Jingsen Zhang, Hongyang Wang, Zhihui Li, Xiuling Zhang, and Lanbo Di. 2019. "Preparation of Pd/C by Atmospheric-Pressure Ethanol Cold Plasma and Its Preparation Mechanism" Nanomaterials 9, no. 10: 1437. https://doi.org/10.3390/nano9101437
APA StyleLi, Z., Zhang, J., Wang, H., Li, Z., Zhang, X., & Di, L. (2019). Preparation of Pd/C by Atmospheric-Pressure Ethanol Cold Plasma and Its Preparation Mechanism. Nanomaterials, 9(10), 1437. https://doi.org/10.3390/nano9101437





