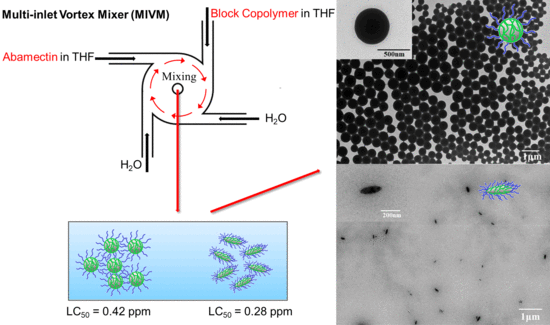
Spherical and Spindle-Like Abamectin-Loaded Nanoparticles by Flash Nanoprecipitation for Southern Root-Knot Nematode Control: Preparation and Characterization
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
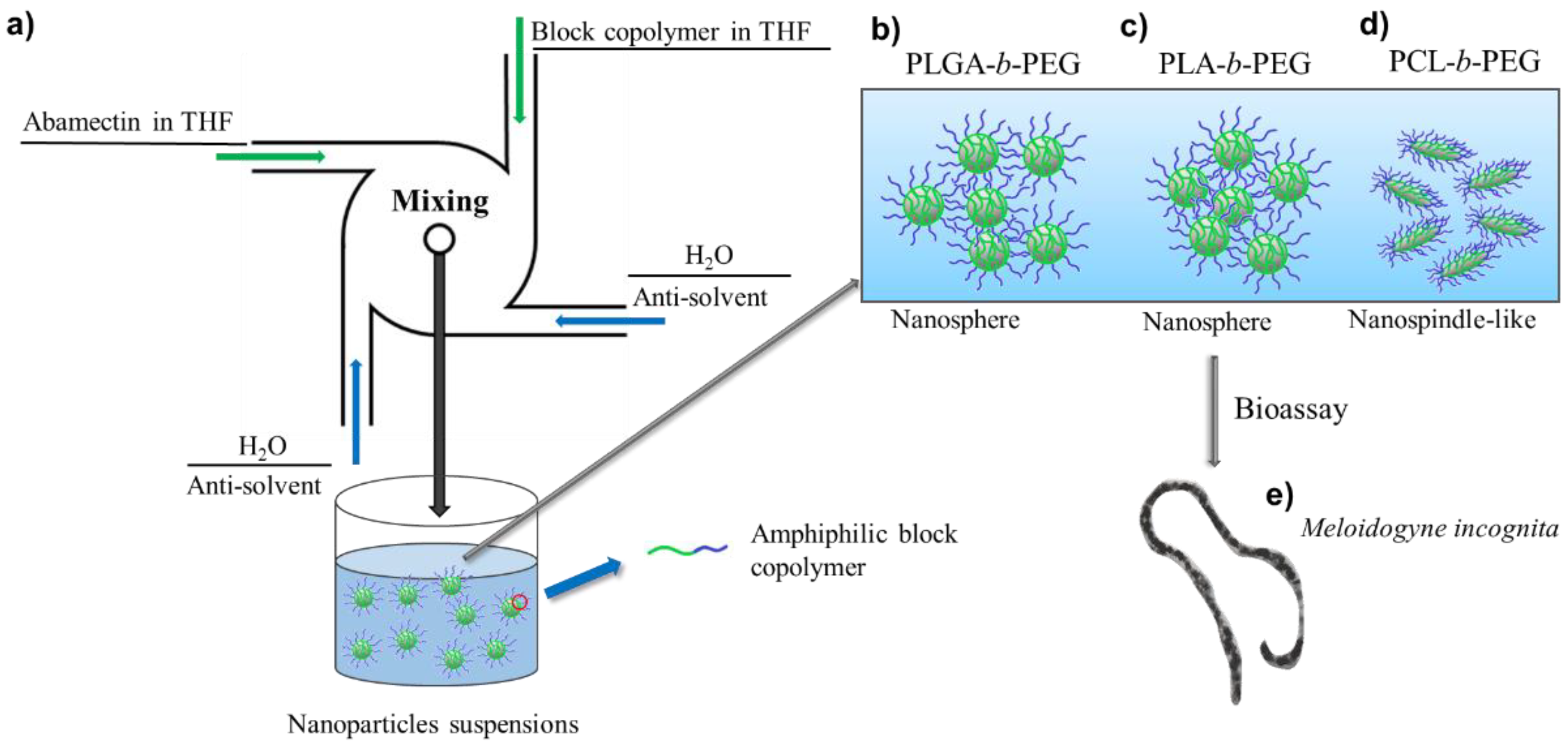
2.2. Preparation of Abm-Loaded Nanoparticles
2.3. Characterization
2.4. Drug Loading Capacity (DLC) and Encapsulation Efficiency (EE)
2.5. Biological Assay
3. Results and Discussion
3.1. Effect of BCP on Size and Morphology of Abm-Loaded Particles
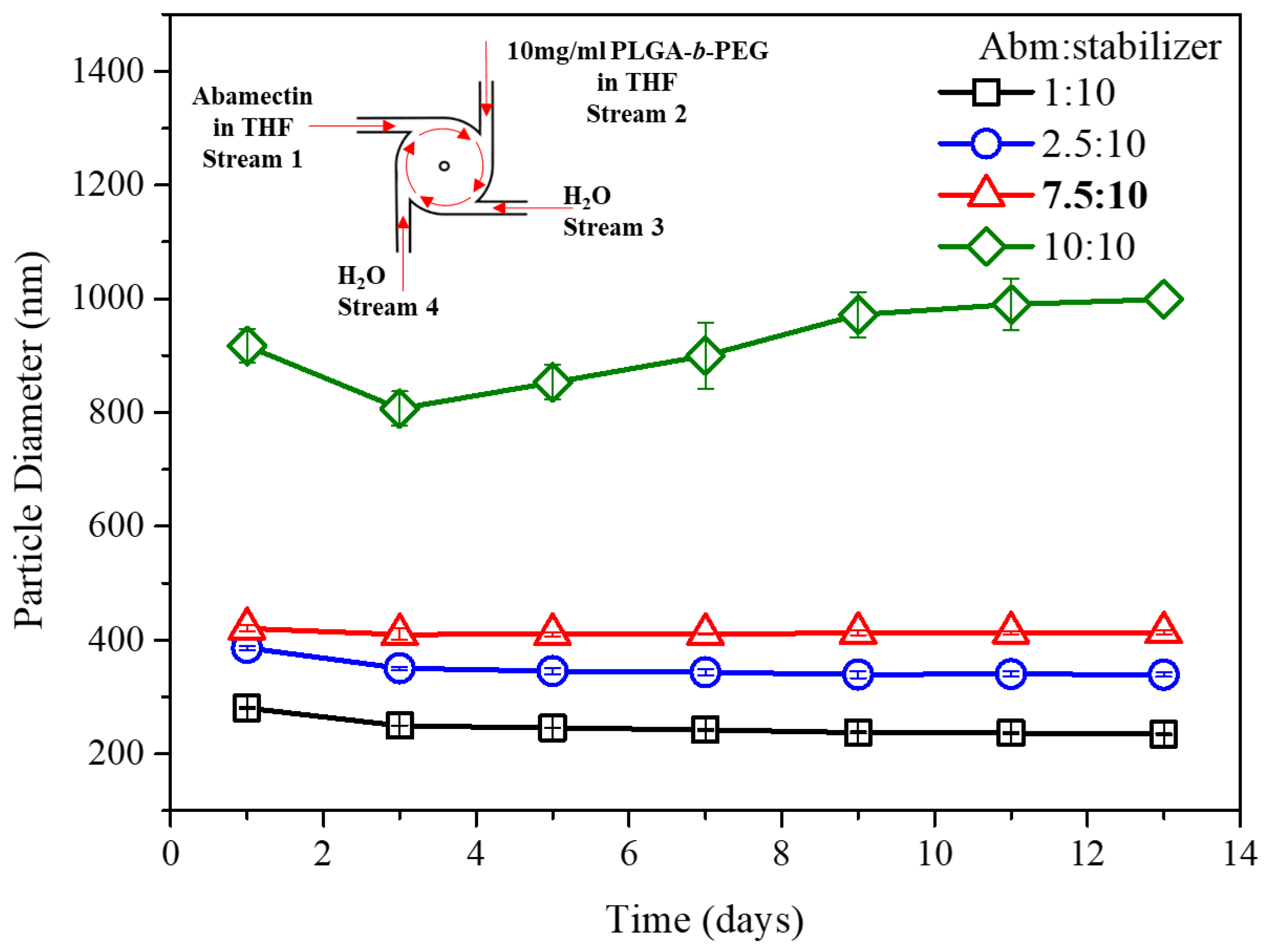
3.2. Effect of Abm-to-Stabilizer Feed Ratio on Particle Stability
3.3. The Amount of Abamectin Encapsulated in FNP-NPs
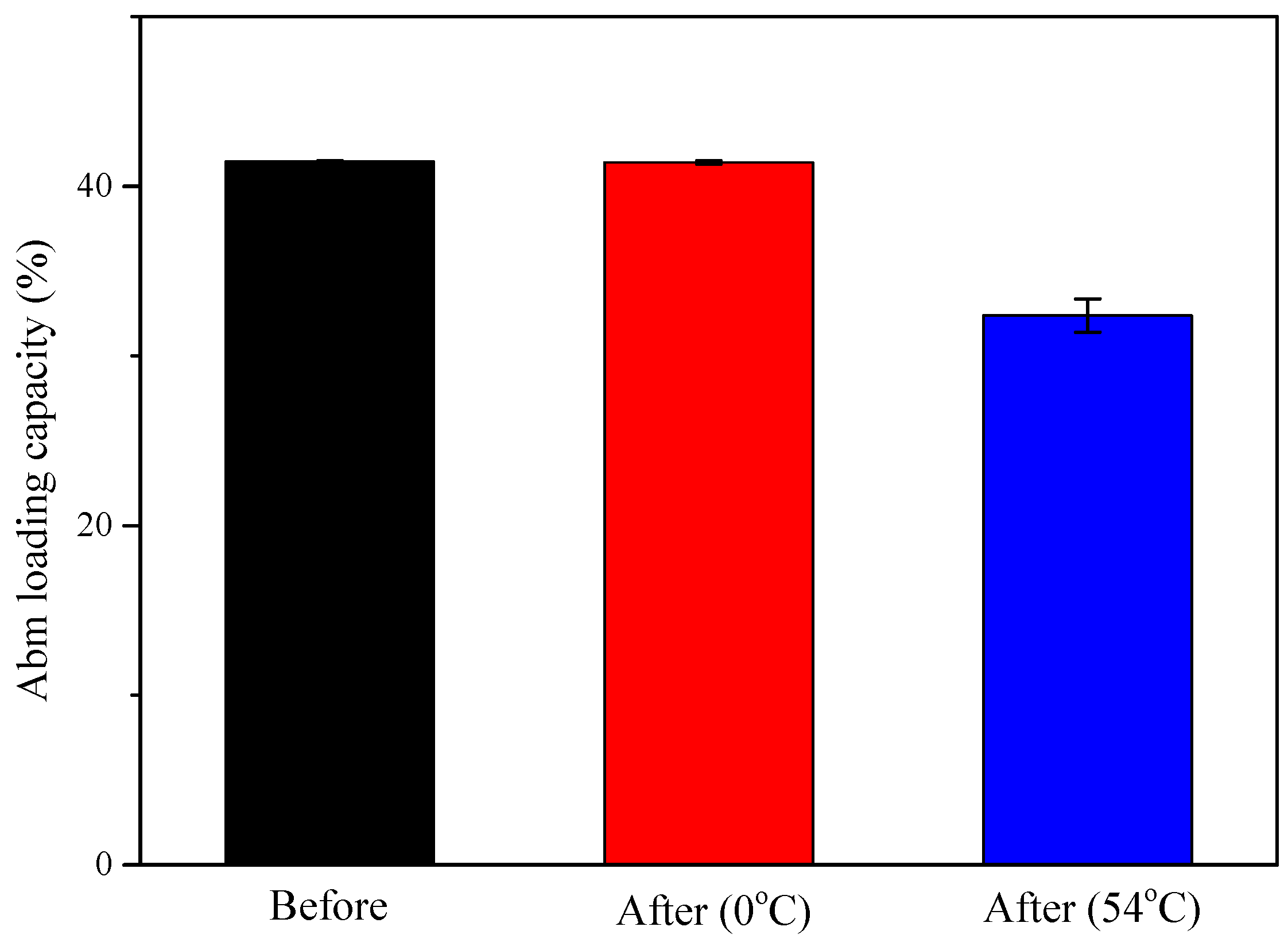
3.4. Effect of Temperature on Particle Stability
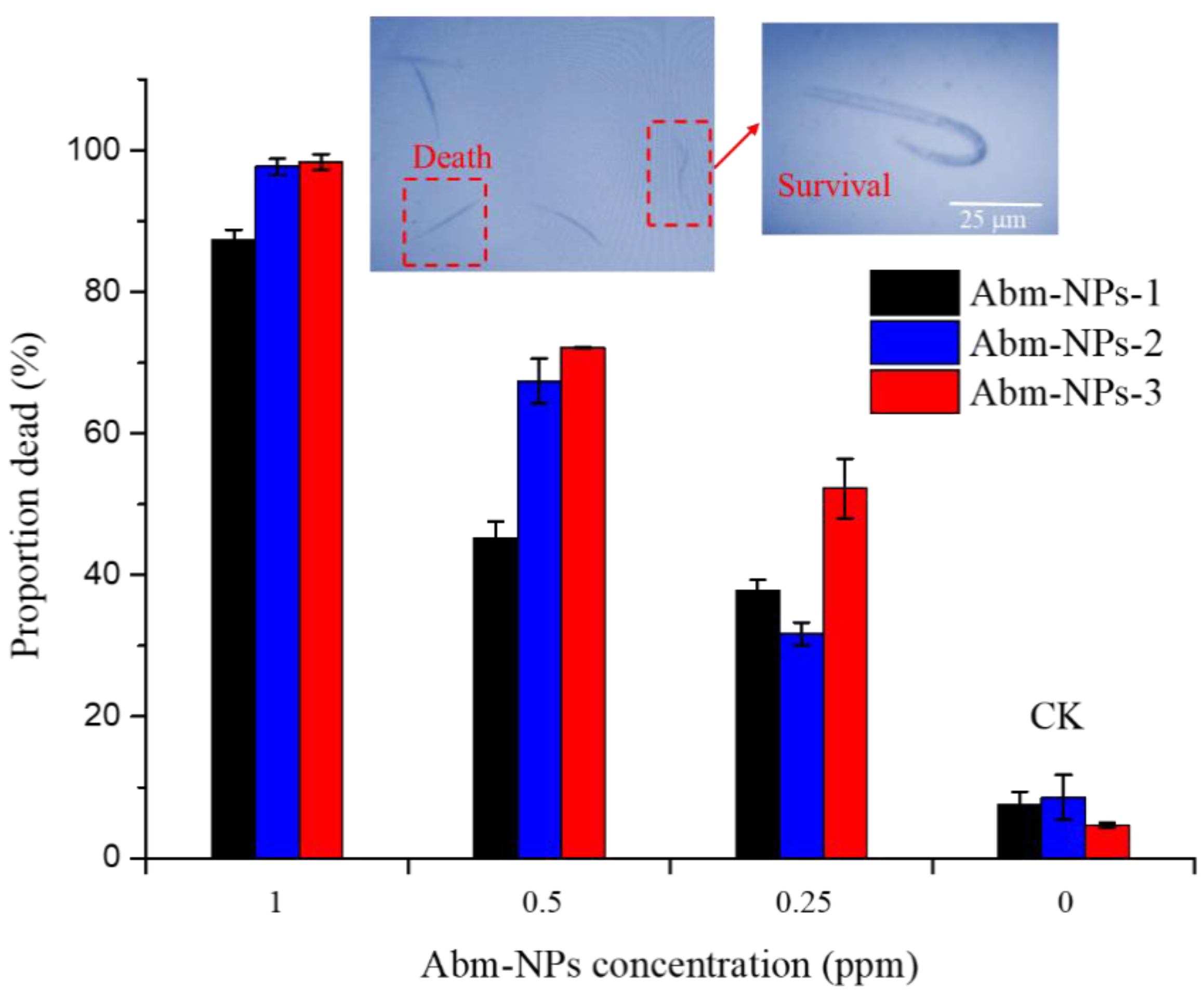
3.5. Toxicity of Abm-Loaded Particles to Meloidogyne incognita
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Abad, P.; Gouzy, J.; Aury, J.M.; Castagnone-Sereno, P.; Danchin, E.G.J.; Deleury, E.; Perfus-Barbeoch, L.; Anthouard, V.; Artiguenave, F.; Blok, V.C.; et al. Genome sequence of the metazoan plant-parasitic nematode Meloidogyne incognita. Nat. Biotechnol. 2008, 26, 909–915. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.T.; Haegeman, A.; Danchin, E.G.J.; Gaur, H.S.; Helder, J.; Jones, M.G.K.; Kikuchi, T.; Manzanilla-López, R.; Palomares-Rius, J.E.; Wesemael, W.M.L.; et al. Top 10 plant-parasitic nematodes in molecular plant pathology. Mol. Plant Pathol. 2013, 14, 946–961. [Google Scholar] [CrossRef] [PubMed]
- Jaouannet, M.; Magliano, M.; Arguel, M.J.; Gourgues, M.; Evangelisti, E.; Abad, P.; Rosso, M.N. The root-knot nematode calreticulin Mi-CRT is a key effector in plant defense suppression. Mol. Plant Microbe Interact. 2013, 26, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Desaeger, J.A.; Seebold, K.W.; Csinos, A.S. Effect of application timing and method on efficacy and phytotoxicity of 1,3-D, chloropicrin and metam-sodium combinations in squash plasticulture. Pest Manag. Sci. 2010, 64, 230–238. [Google Scholar] [CrossRef] [PubMed]
- Stirling, A.M.; Stirling, G.R.; Macrae, I.C. Microbial degradation of Fenamiphos after repeated application to a tomato-growing soil. Nematologica 1992, 38, 245–254. [Google Scholar] [CrossRef]
- Giannakou, I.O.; Karpouzas, D.G. Evaluation of chemical and integrated strategies as alternatives to methyl bromide for the control of root-knot nematodes in Greece. Pest Manag. Sci. 2003, 59, 883–892. [Google Scholar] [CrossRef] [PubMed]
- Sasser, J.N.; Kirkpatrick, T.L.; Dybas, R.A. Efficacy of avermectins for root-knot control in tobacco. Plant Dis. 1982, 66, 691–693. [Google Scholar] [CrossRef]
- Shi, Y.; Zheng, T.; Shang, Q. Preparation of acrylic/acrylate copolymeric surfactants by emulsion polymerization used in pesticide oil-in-water emulsions. J. Appl. Polym. Sci. 2012, 123, 3117–3127. [Google Scholar] [CrossRef]
- Guimaraes, J.R.; Barbosa, I.M.; Maniero, M.G.; Rath, S. Abamectin degradation by Advanced Oxidation Processes: Evaluation of toxicity reduction using Daphnia similis. J. Adv. Oxid. Tech. 2014, 17, 82–92. [Google Scholar]
- Erzen, N.K.; Kolar, L.; Flajs, V.C.; Kužner, J.; Marc, I.; Pogačnik, M. Degradation of abamectin and doramectin on sheep grazed pasture. Ecotoxicology 2005, 14, 627–635. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.W.; Li, R.; Zhou, W.Y.; Sun, J.Q.; Guo, P.; Ma, J.P.; Li, S.P. Isolation and characterization of an abamectin-degrading Burkholderiacepacia-like GB-01 strain. Biodegradation 2010, 21, 441–452. [Google Scholar] [CrossRef] [PubMed]
- Khot, L.R.; Sankaran, S.; Maja, J.M.; Ehsania, R.; Schusterb, E.W. Applications of nanomaterials in agricultural production and crop protection: A review. Crop Prot. 2012, 35, 64–70. [Google Scholar] [CrossRef]
- Mattos, B.D.; Tardy, B.L.; Magalhães, W.L.E.; Rojas, O.J. Controlled release for crop and wood protection: Recent progress toward sustainable and safe nanostructured biocidal systems. J. Control. Release 2017, 262, 139–150. [Google Scholar] [CrossRef] [PubMed]
- Iavicoli, I.; Leso, V.; Beezhold, D.H.; Shvedova, A.A. Nanotechnology in agriculture: Opportunities, toxicological implications, and occupational risks. Toxicol. Appl. Pharmacol. 2017, 329, 96–111. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, J.L.; Campos, E.V.; Bakshi, M.; Abhilash, P.C.; Fraceto, L.F. Application of nanotechnology for the encapsulation of botanical insecticides for sustainable agriculture: Prospects and promises. Biotechnol. Adv. 2014, 32, 1550–1561. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, A.; Wang, C.; Cui, B.; Sun, C.; Zhao, X.; Zeng, Z.; Shen, Y.; Gao, F.; Liu, G.; et al. Synthesis and characterization of emamectin-benzoate slow-release microspheres with different surfactants. Sci. Rep. 2017, 7, 12761. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Wang, Y.; Zhao, X.; Sun, C.; Cui, B.; Gao, F.; Zeng, Z.; Cui, H. Preparation and Physicochemical Characteristics of Thermo-Responsive Emamectin Benzoate Microcapsules. Polymers 2017, 9, 418. [Google Scholar] [CrossRef]
- Liu, B.; Wang, Y.; Yang, F.; Wang, X.; Shen, H.; Cui, H.; Wu, D. Construction of a controlled-release delivery system for pesticides using biodegradable PLA-based microcapsules. Colloids Surf. B 2016, 144, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, E.; Kuroda, K. Colloidal Mesoporous Silica Nanoparticles. Bull. Chem. Soc. Jpn. 2016, 89, 501–539. [Google Scholar] [CrossRef]
- Nakamura, M.; Tahara, Y.; Fukata, S.; Zhang, M.; Yang, M.; Iijima, S.; Yudasaka, M. Significance of Optimization of Phospholipid Poly(Ethylene Glycol) Quantity for Coating Carbon Nanohorns to Achieve Low Cytotoxicity. Bull. Chem. Soc. Jpn. 2017, 90, 662–666. [Google Scholar] [CrossRef]
- Tang, Q.; Liu, J.; Shrestha, L.K.; Ariga, K.; Ji, Q. Antibacterial Effect of Silver-Incorporated Flake-Shell Nanoparticles under Dual-Modality. ACS Appl. Mater. Interfaces 2016, 8, 18922–18929. [Google Scholar] [CrossRef] [PubMed]
- Li, B.L.; Setyawati, M.I.; Chen, L.; Xie, J.; Ariga, K.; Lim, C.-T.; Garaj, S.; Leong, D.T. Directing Assembly and Disassembly of 2D MoS2 Nanosheets with DNA for Drug Delivery. ACS Appl. Mater. Interfaces 2017, 9, 15286–15296. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Cui, H.; Sun, C.; Zhao, X.; Cui, B. Construction and evaluation of controlled-release delivery system of Abamectin using porous silica nanoparticles as carriers. Nanoscale Res. Lett. 2014, 9, 655. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Yao, J.; Liang, J.; Zeng, Z.; Cui, B.; Zhao, X.; Sun, C.; Wang, Y.; Liu, G.; Cui, H. Development of functionalized abamectinpoly (lactic acid) nanoparticles with regulatable adhesion to enhance foliar retention. RSC Adv. 2017, 7, 11271–11280. [Google Scholar] [CrossRef]
- Cao, J.; Guenther, R.H.; Sit, T.L.; Lommel, S.A.; Opperman, C.H.; Willoughby, J.A. Development of Abamectin loaded plant virus nanoparticles for efficacious plant parasitic nematode control. ACS Appl. Mater. Interfaces 2015, 7, 9546–9553. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Xu, Y.; Wang, J.; Liu, M.; Yuan, Z.; Chen, K.; Li, L.; Prud’Homme, R.K.; Guo, X. Biocompatible nanoparticle based on dextran-b-poly(l-lactide) block copolymer formed by flash nanoprecipitation. Chem. Lett. 2015, 44, 1688–1690. [Google Scholar] [CrossRef]
- Wang, M.; Yang, N.; Guo, Z.; Gu, K.; Shao, A.; Zhu, W.; Xu, Y.; Wang, J.; Prud’Homme, R.K.; Guo, X. Facile Preparation of AIE-active fluorescent nanoparticles through flash nanoprecipitation. Ind. Eng. Chem. Res. 2015, 54, 4683–4688. [Google Scholar] [CrossRef]
- Johnson, B.K.; Prud’Homme, R.K. Mechanism for rapid self-assembly of block copolymer nanoparticles. Phys. Rev. Lett. 2003, 91, 118302. [Google Scholar] [CrossRef] [PubMed]
- Saad, W.S.; Prud’Homme, R.K. Principles of nanoparticle formation by Flash Nanoprecipitation. Nano Today 2016, 11, 212–227. [Google Scholar] [CrossRef]
- Liu, Y.; Cheng, C.; Liu, Y.; Prud’Homme, R.K.; Fox, R.O. Mixing in a multi-inlet vortex mixer (MIVM) for flash nano-precipitation. Chem. Eng. Sci. 2008, 63, 2829–2842. [Google Scholar] [CrossRef]
- Russ, R.; Liu, Y.; Prud’homme, R.K. Optimized descriptive model for micromixing in avortexmixer. Chem. Eng. Commun. 2010, 197, 1068–1075. [Google Scholar] [CrossRef]
- Fu, Z.; Li, L.; Wang, M.; Guo, X. Size control of drug nanoparticles stabilized by mPEG-b-PCL during flash nanoprecipitation. Colloid Polym. Sci. 2018, 296, 935–940. [Google Scholar] [CrossRef]
- Zhu, Z. Effects of amphiphilic diblock copolymer on drug nanoparticle formation and stability. Biomaterials 2013, 34, 10238–10248. [Google Scholar] [CrossRef] [PubMed]
- Pustulka, K.M.; Wohl, A.R.; Lee, H.S.; Michel, A.R.; Han, J.; Hoye, T.R.; McCormick, A.V.; Panyam, J.; Macosko, C.W. Flash nanoprecipitation: Particle structure and stability. Mol. Pharm. 2013, 10, 4367–4377. [Google Scholar] [CrossRef] [PubMed]
- Tuncelli, G.; Ay, A.N.; Zümreoglu-Karan, B. 5-Fluorouracil intercalated iron oxide@layered double hydroxide core-shell nano-composites with isotropic and anisotropic architectures for shape-selective drug delivery applications. Mater. Sci. Eng. C 2015, 55, 562–568. [Google Scholar] [CrossRef] [PubMed]
- Geng, Y.; Dalhaimer, P.; Cai, S.; Tsai, R.; Tewari, M.; Minko, T.; Discher, D.E. Shape effects of filaments versus spherical particles in flow and drug delivery. Nat. Nanotechnol. 2007, 2, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Qie, R.; Li, W.; Hong, N.; Li, Y.; Li, C.; Wang, R.; Shi, Y.; Guo, X.; Jia, X. Preparation of avermectin microcapsules with anti-photodegradation and slow-release by the assembly of lignin derivatives. New J. Chem. 2017, 41, 3190–3195. [Google Scholar] [CrossRef]
- Li, D.; Liu, B.; Yang, F.; Wang, X.; Shen, H.; Wu, D. Preparation of uniform starch microcapsules by premix membrane emulsion for controlled release of avermectin. Carbohydr. Polym. 2016, 136, 341–349. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Cui, H.; Wang, Y.; Sun, C.; Cui, B.; Zeng, Z. Development strategies and prospects of nano-based smart pesticide formulation. J. Agric. Food Chem. 2017. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.H.; Hua, M.Y.; Lee, R.S. Synthesis and characterization of poly(ethylene glycol)-b-poly(ε-caprolactone) copolymers with functional side groups on the polyester block. J. Appl. Polym. Sci. 2012, 125, 2902–2913. [Google Scholar] [CrossRef]
- Wang, J.; Zhu, W.; Peng, B.; Chen, Y. A facile way to prepare crystalline platelets of block copolymers by crystallization-driven self-assembly. Polymer 2013, 54, 6760–6767. [Google Scholar] [CrossRef]
- Rytting, E.; Nguyen, J.; Wang, X.; Kissel, T. Biodegradable polymeric nanocarriers for pulmonary drug delivery. Expert Opin. Drug Deliv. 2008, 5, 629–639. [Google Scholar] [CrossRef] [PubMed]
- Houchin, M.L.; Topp, E.M. Physical properties of PLGA films during polymer degradation. J. Appl. Polym. Sci. 2009, 114, 2848–2854. [Google Scholar] [CrossRef]
- Omelczuk, M.O.; McGinity, J.W. The influence of polymer glass transition temperature and molecular weight on drug release from tablets containing poly(dl-lactic acid). Pharm. Res. 1992, 9, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Ghormade, V.; Deshpande, M.V.; Paknikar, K.M. Perspectives for nano-biotechnology enabled protection and nutrition of plants. Biotechnol. Adv. 2011, 29, 792–803. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Adamson, D.H.; Prud’Homme, R.K. Fluorescent polymeric nanoparticles: Aggregation and phase behavior of pyrene and amphotericin B molecules in nanoparticle cores. Small 2010, 6, 2907–2914. [Google Scholar] [CrossRef] [PubMed]
- D’Addio, S.M.; Prud’Homme, R.K. Controlling drug nanoparticle formation by rapid precipitation. Adv. Drug Deliv. Rev. 2011, 63, 417–426. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Tong, Z.; Prud’Homme, R.K. Stabilized polymeric nanoparticles for controlled and efficient release of bifenthrin. Pest Manag. Sci. 2008, 64, 808–812. [Google Scholar] [CrossRef] [PubMed]
- Allen, C.; Maysinger, D.; Eisenberg, A. Nano-engineering block copolymer aggregates for drug delivery. Colloids Surf. B 1999, 16, 3–27. [Google Scholar] [CrossRef]
- Faske, T.R.; Starr, J.L. Sensitivity of Meloidogyne incognita and Rotylenchulus reniformis to Abamectin. J. Nematol. 2006, 38, 240–244. [Google Scholar] [PubMed]

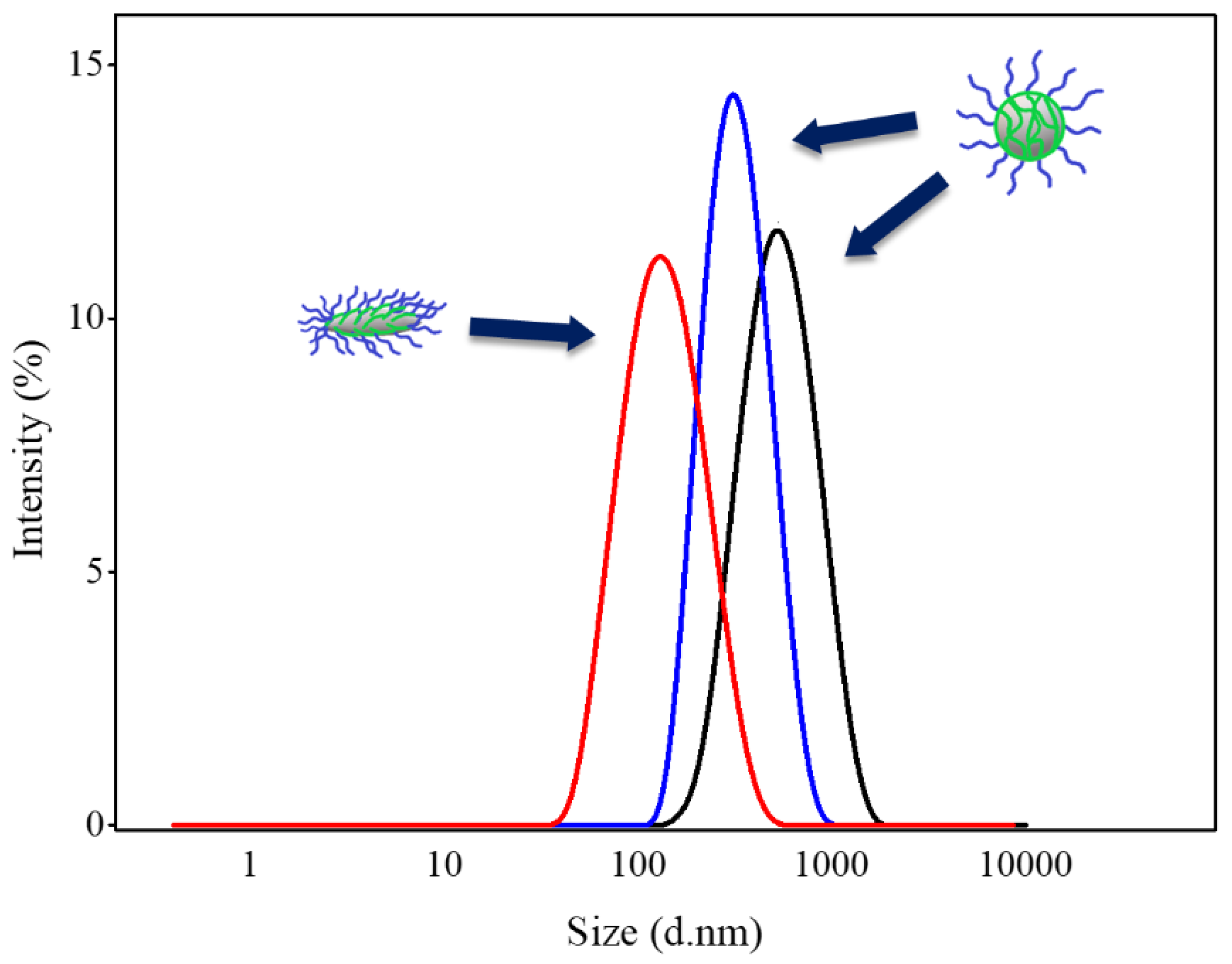
| Ratio of Abamectin to Stabilizer | Particle Diameter (nm) | PDI |
|---|---|---|
| 1:10 | 252 ± 1 | 0.25 ± 0.01 |
| 2.5:10 | 355 ± 3 | 0.14 ± 0.04 |
| 7.5:10 | 414 ± 5 | 0.19 ± 0.07 |
| 10:10 | 898 ± 30 | 0.53 ± 0.20 |
| Stabilizer | DLC (%) | EE (%) | |
|---|---|---|---|
| Abm-NPs-1 | PLGA-b-PEG | 41.46 ± 0.05 | 96.74 ± 0.12 |
| Abm-NPs-2 | PLA-b-PEG | 40.97 ± 0.05 | 95.60 ± 0.11 |
| Abm-NPs-3 | PCL-b-PEG | 40.76 ± 0.03 | 95.10 ± 0.07 |
| Population | Insecticide | Stabilizer | LC50 (95% CL a) (ppm) | Fit of Probit Line | ||||
|---|---|---|---|---|---|---|---|---|
| Slope ± SE | χ2 | DF | P | n b | ||||
| Lab | Abm-NPs-1 | PLGA-b-PEG | 0.42 (0.32–0.62) | 5.76 ± 0.33 | 5.95 | 3 | 0.11 | 252 |
| Lab | Abm-NPs-2 | PLA-b-PEG | 0.37 (0.32–0.48) | 6.72 ± 0.59 | 0.79 | 3 | 0.84 | 320 |
| Lab | Abm-NPs-3 | PCL-b-PEG | 0.28 (0.23–0.33) | 6.57 ± 0.33 | 3.45 | 3 | 0.32 | 352 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fu, Z.; Chen, K.; Li, L.; Zhao, F.; Wang, Y.; Wang, M.; Shen, Y.; Cui, H.; Liu, D.; Guo, X. Spherical and Spindle-Like Abamectin-Loaded Nanoparticles by Flash Nanoprecipitation for Southern Root-Knot Nematode Control: Preparation and Characterization. Nanomaterials 2018, 8, 449. https://doi.org/10.3390/nano8060449
Fu Z, Chen K, Li L, Zhao F, Wang Y, Wang M, Shen Y, Cui H, Liu D, Guo X. Spherical and Spindle-Like Abamectin-Loaded Nanoparticles by Flash Nanoprecipitation for Southern Root-Knot Nematode Control: Preparation and Characterization. Nanomaterials. 2018; 8(6):449. https://doi.org/10.3390/nano8060449
Chicago/Turabian StyleFu, Zhinan, Kai Chen, Li Li, Fang Zhao, Yan Wang, Mingwei Wang, Yue Shen, Haixin Cui, Dianhua Liu, and Xuhong Guo. 2018. "Spherical and Spindle-Like Abamectin-Loaded Nanoparticles by Flash Nanoprecipitation for Southern Root-Knot Nematode Control: Preparation and Characterization" Nanomaterials 8, no. 6: 449. https://doi.org/10.3390/nano8060449
APA StyleFu, Z., Chen, K., Li, L., Zhao, F., Wang, Y., Wang, M., Shen, Y., Cui, H., Liu, D., & Guo, X. (2018). Spherical and Spindle-Like Abamectin-Loaded Nanoparticles by Flash Nanoprecipitation for Southern Root-Knot Nematode Control: Preparation and Characterization. Nanomaterials, 8(6), 449. https://doi.org/10.3390/nano8060449