Scalable Fabrication of High-Performance Transparent Conductors Using Graphene Oxide-Stabilized Single-Walled Carbon Nanotube Inks
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Synthesis
2.2. rGO-SWNT Films
2.3. Material Characterization
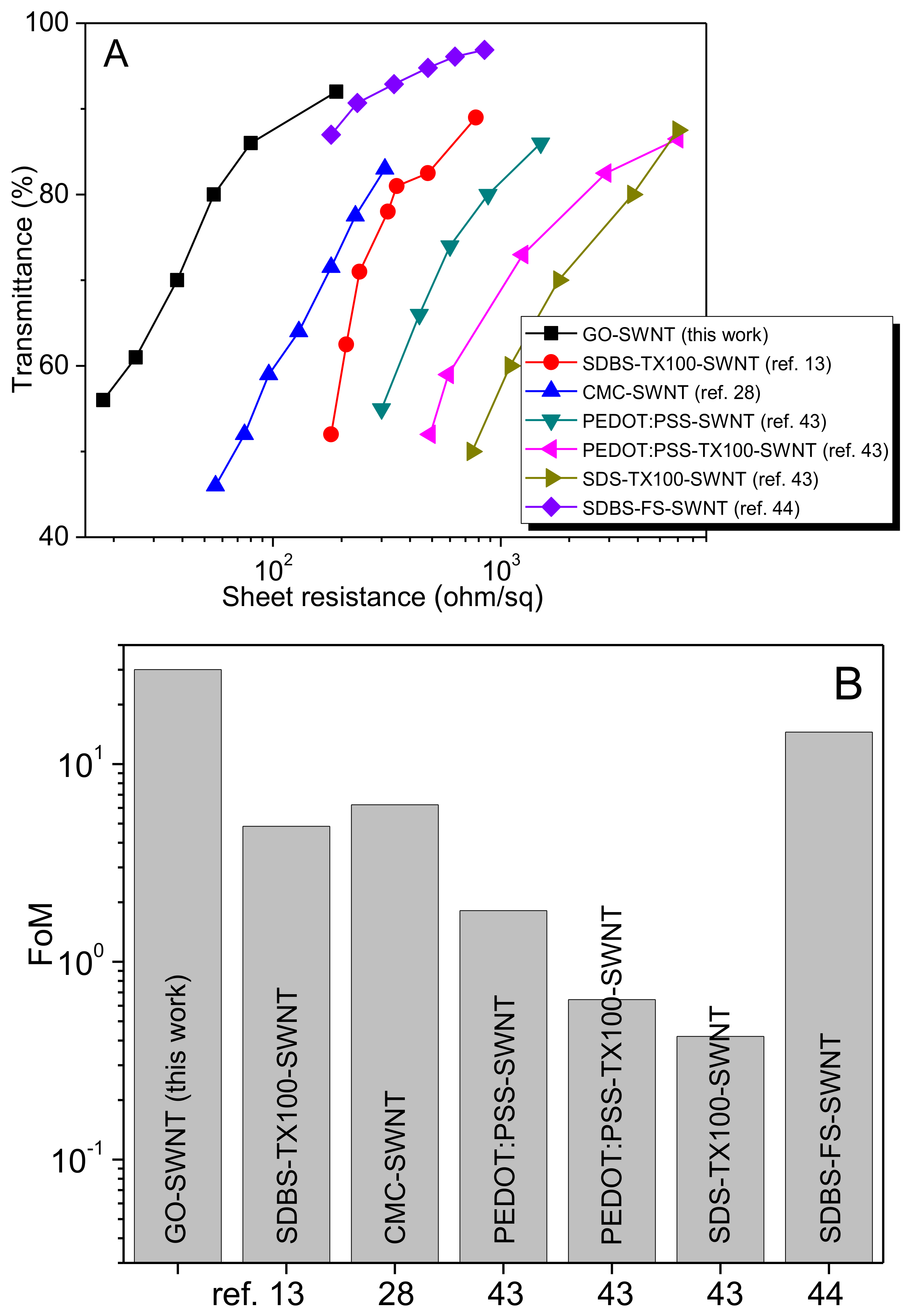
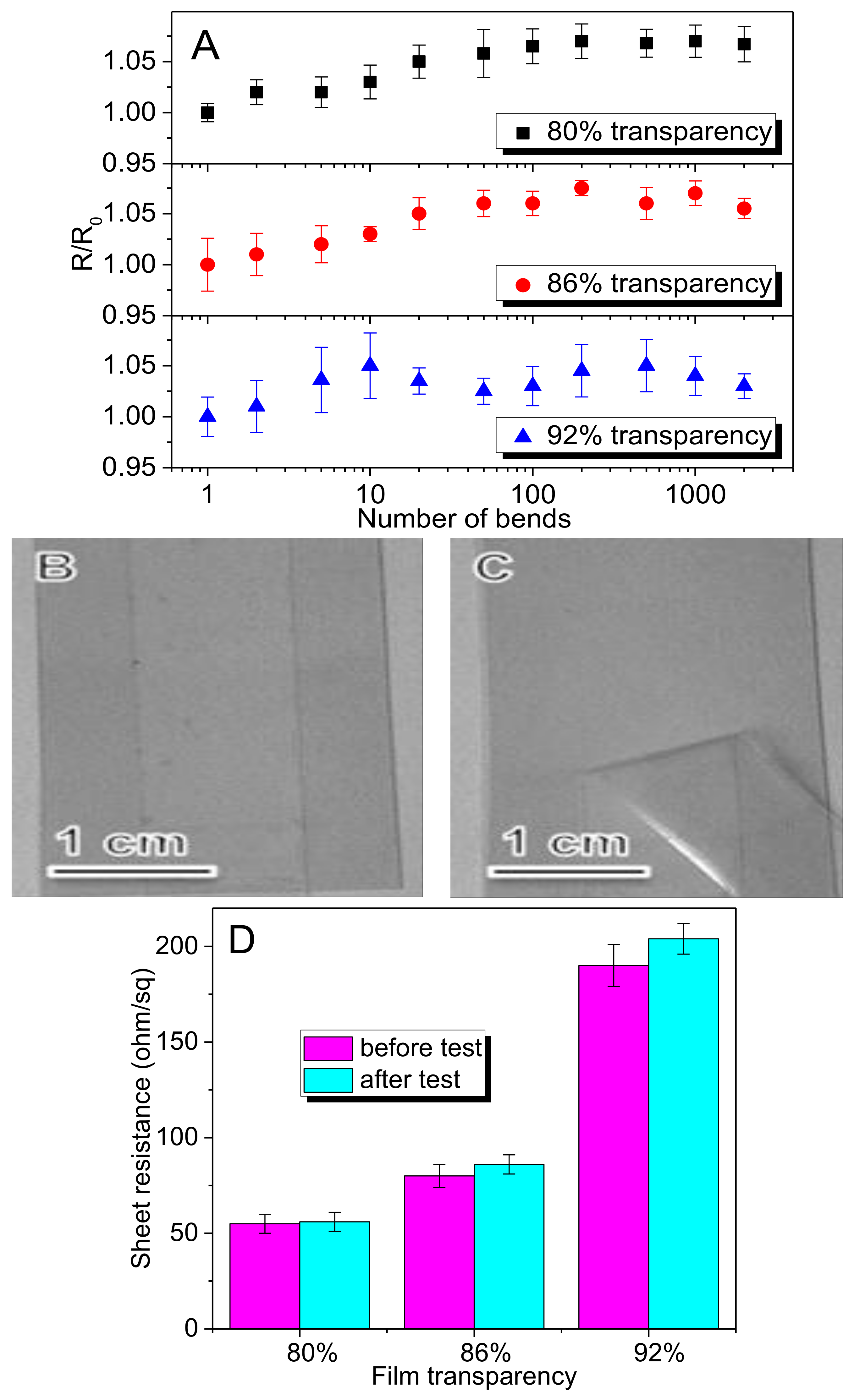
3. Results
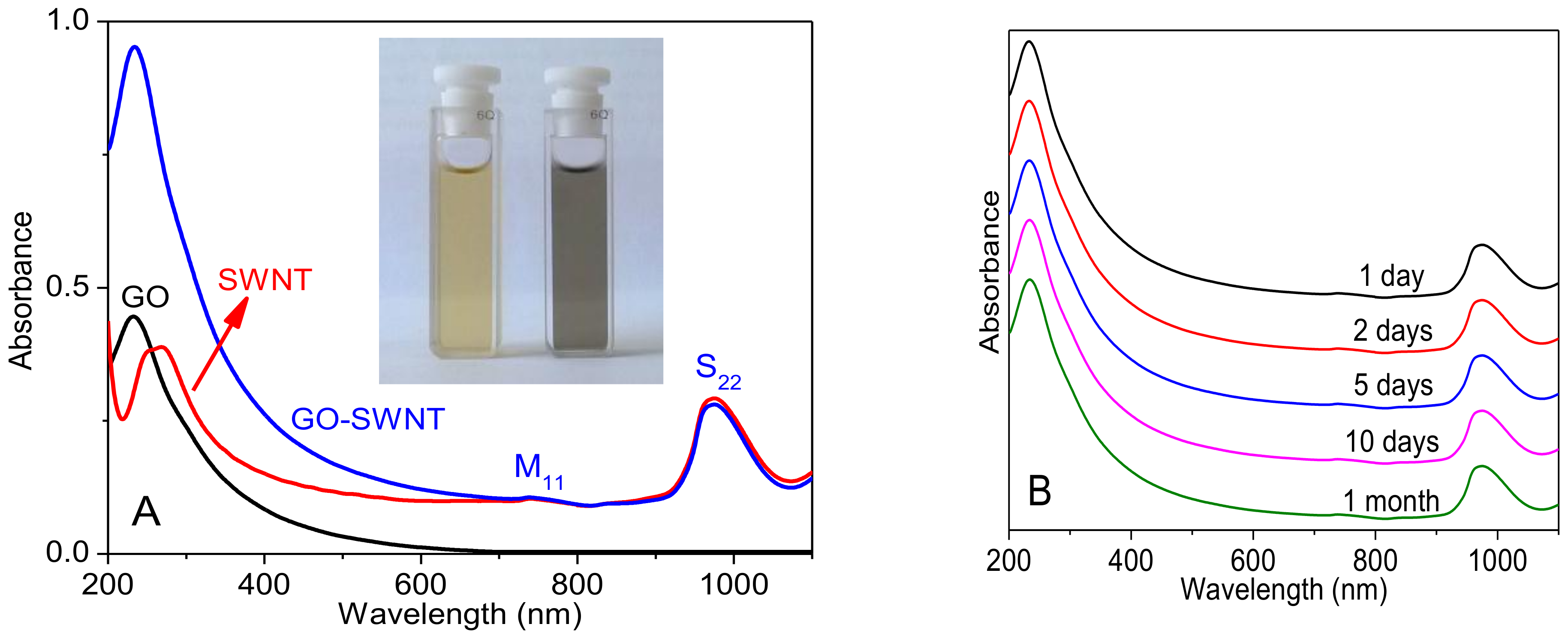
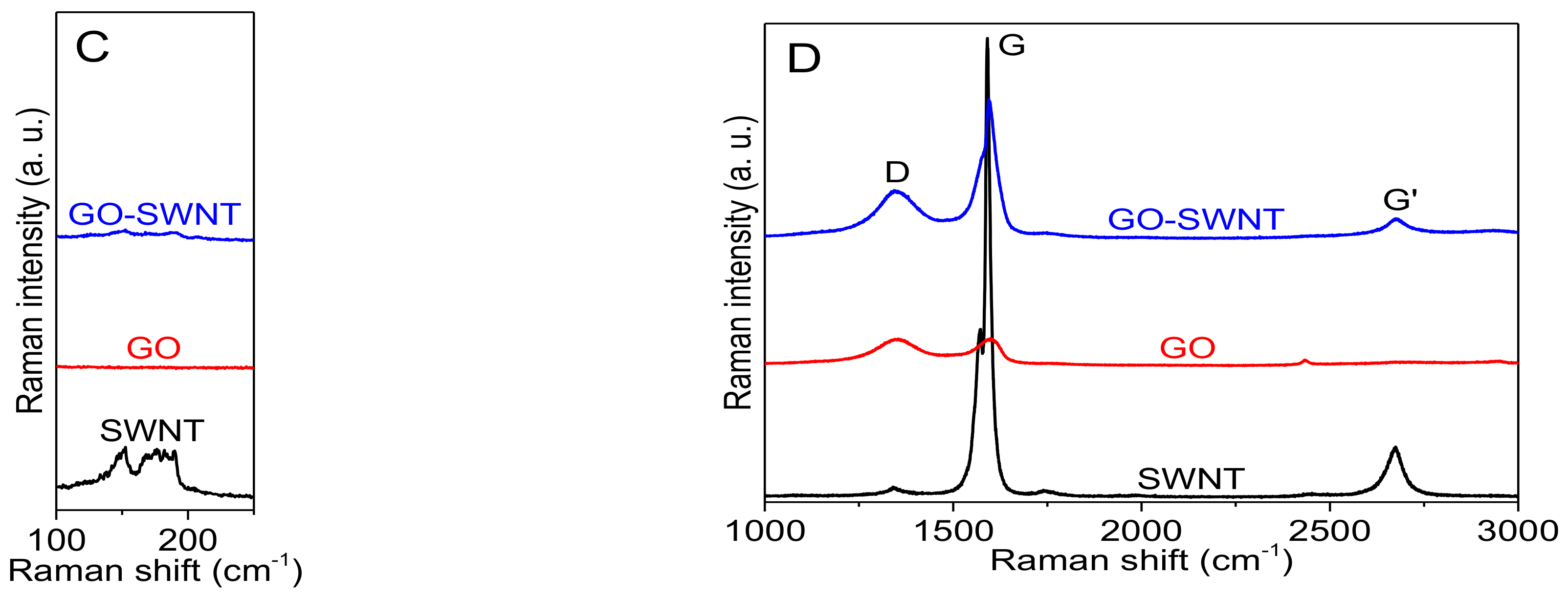
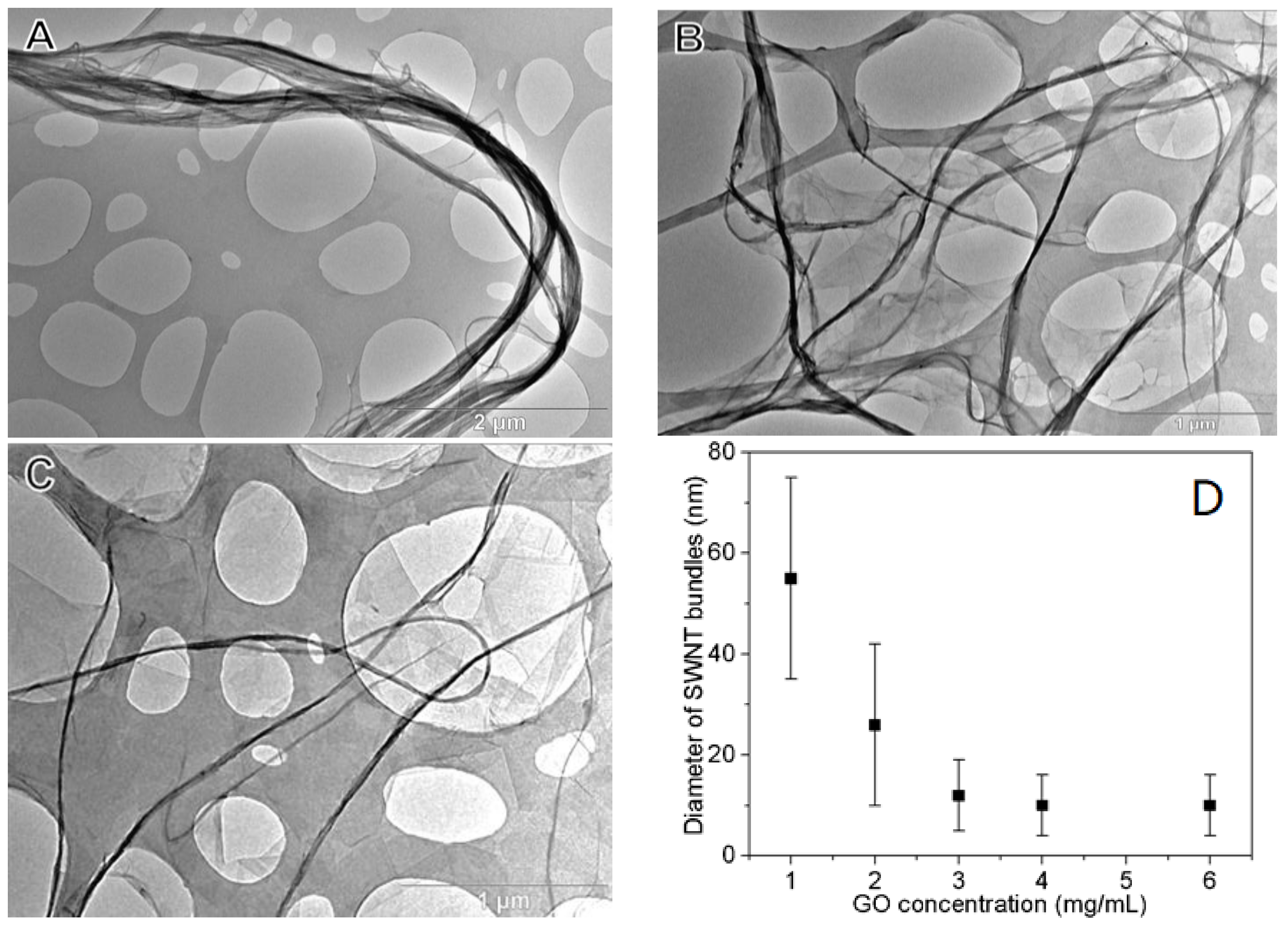
3.1. Dispersing SWNTs in Water Using GO
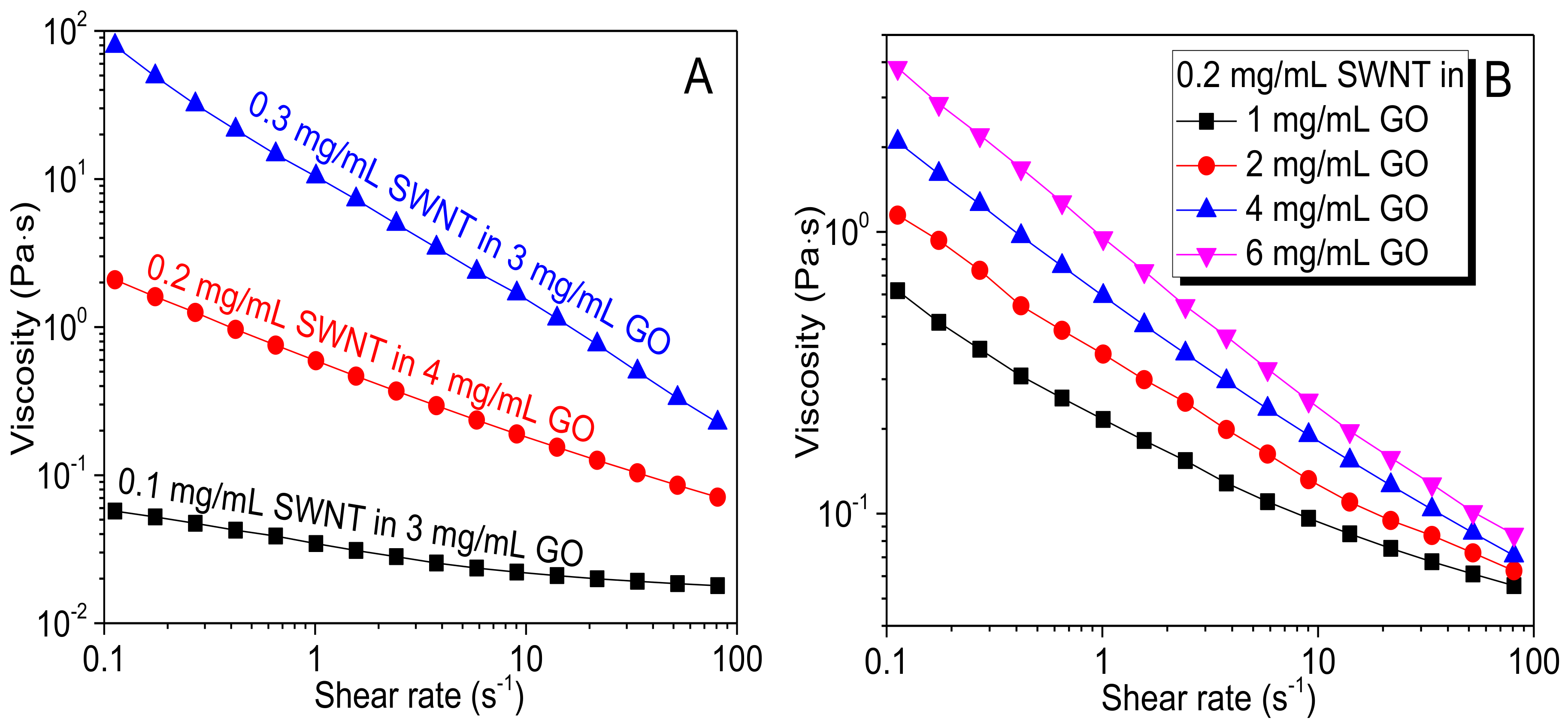
3.2. Wetting Behavior and Rheological Properties of GO-SWNT Dispersions
3.3. Fabrication & Characterization of rGO-SWNT Films
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- He, L.X.; Tjong, S.C. Nanostructured transparent conductive films: Fabrication, characterization and applications. Mater. Sci. Eng. R Rep. 2016, 109, 1–101. [Google Scholar] [CrossRef]
- Artukovic, E.; Kaempgen, M.; Hecht, D.S.; Roth, S.; Grüner, G. Transparent and flexible carbon nanotube transistors. Nano Lett. 2005, 5, 757–760. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhi, L.J.; Mullen, K. Transparent, conductive graphene electrodes for dye-sensitized solar cells. Nano Lett. 2008, 8, 323–327. [Google Scholar] [CrossRef] [PubMed]
- Hecht, D.S.; Hu, L.; Irvin, G. Emerging transparent electrodes based on thin films of carbon nanotubes, graphene, and metallic nanostructures. Adv. Mater. 2011, 23, 1482–1513. [Google Scholar] [CrossRef] [PubMed]
- Ye, S.R.; Rathmell, A.R.; Chen, Z.F.; Stewart, I.E.; Wiley, B.J. Metal nanowire networks: The next generation of transparent conductors. Adv. Mater. 2014, 26, 6670–6687. [Google Scholar] [CrossRef] [PubMed]
- Iijima, S.; Ichihashi, T. Single-shell carbon nanotubes of 1-nm diameter. Nature 1993, 363, 603–605. [Google Scholar] [CrossRef]
- Hu, L.; Hecht, D.S.; Grüner, G. Carbon nanotube thin films: Fabrication, properties, and applications. Chem. Rev. 2010, 110, 5790–5844. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Yim, J.; Wang, X.; Bradley, D.D.; Lee, S.; deMello, J.C. Spin-and spray-deposited single-walled carbon-nanotube electrodes for organic solar cells. Adv. Funct. Mater. 2010, 20, 2310–2316. [Google Scholar] [CrossRef]
- Jo, J.W.; Jung, J.W.; Lee, J.U.; Jo, W.H. Fabrication of highly conductive and transparent thin films from single-walled carbon nanotubes using a new non-ionic surfactant via spin coating. ACS Nano 2010, 4, 5382–5388. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Minami, N.; Zhu, W.; Kazaoui, S.; Azumi, R.; Matsumoto, M. Langmuir-Blodgett films of single-wall carbon nanotubes: Layer-by-layer deposition and in-plane orientation of tubes. Jpn. J. Appl. Phys. 2003, 42, 7629–7634. [Google Scholar] [CrossRef]
- Eda, G.; Lin, Y.Y.; Miller, S.; Chen, C.W.; Su, W.F.; Chhowalla, M. Transparent and conducting electrodes for organic electronics from reduced graphene oxide. Appl. Phys. Lett. 2008, 92, 233305. [Google Scholar] [CrossRef]
- Zhang, D.; Ryu, K.; Liu, X.; Polikarpov, E.; Ly, J.; Tompson, M.E.; Zhou, C. Transparent, conductive, and flexible carbon nanotube films and their application in organic light-emitting diodes. Nano Lett. 2006, 6, 1880–1886. [Google Scholar] [CrossRef] [PubMed]
- Dan, B.; Irvin, G.C.; Pasquali, M. Continuous and scalable fabrication of transparent conducting carbon nanotube films. ACS Nano 2009, 3, 835–843. [Google Scholar] [CrossRef] [PubMed]
- He, L.X.; Tjong, S.C. Aqueous graphene oxide-dispersed carbon nanotubes as inks for the scalable production of all-carbon transparent conductive films. J. Mater. Chem. C 2016, 4, 7043–7051. [Google Scholar] [CrossRef]
- Zhou, M.; Wang, Y.; Zhai, Y.; Zhai, J.; Ren, W.; Wang, F.; Dong, S. Controlled synthesis of large-area and patterned electrochemically reduced graphene oxide films. Chem. Eur. J. 2009, 15, 6116–6120. [Google Scholar] [CrossRef] [PubMed]
- He, L.X.; Tjong, S.C. Low percolation threshold of graphene/polymer composites prepared by solvothermal reduction of graphene oxide in the polymer solution. Nanoscale Res. Lett. 2013, 8, 132. [Google Scholar] [CrossRef] [PubMed]
- He, L.X.; Tjong, S.C. Effect of temperature on electrical conduction behavior of polyvinylidene fluoride nanocomposites with carbon nanotubes and nanofibers. Curr. Nanosci. 2010, 6, 520–524. [Google Scholar] [CrossRef]
- Hu, L.B.; Choi, J.W.; Yang, Y.; Jeong, S.; La Mantia, F.; Cui, L.F.; Cui, Y. Highly conductive paper for energy-storage devices. Proc. Natl. Acad. Sci. USA 2009, 106, 21490–21494. [Google Scholar] [CrossRef] [PubMed]
- Hummers, W.S.; Offeman, R.E. Preparation of graphitic oxide. J. Am. Chem. Soc. 1958, 80, 1339. [Google Scholar] [CrossRef]
- Dreyer, D.R.; Park, S.; Bielawski, C.W.; Ruoff, R.S. The chemistry of graphene oxide. Chem. Soc. Rev. 2010, 39, 228–240. [Google Scholar] [CrossRef] [PubMed]
- Cote, L.J.; Kim, F.; Huang, J.X. Langmuir-Blodgett assembly of graphite oxide single layers. J. Am. Chem. Soc. 2009, 131, 1043–1049. [Google Scholar] [CrossRef] [PubMed]
- Qiu, L.; Yang, X.; Gou, X.; Yang, W.; Ma, Z.F.; Wallace, G.G.; Li, D. Dispersing carbon nanotubes with graphene oxide in water and synergistic effects between graphene derivatives. Chem. Eur. J. 2010, 16, 10653–10658. [Google Scholar] [CrossRef] [PubMed]
- Pei, S.; Cheng, H.M. The reduction of graphene oxide. Carbon 2012, 50, 3210–3228. [Google Scholar] [CrossRef]
- Tian, L.; Meziani, M.J.; Lu, L.; Kong, C.Y.; Cao, L.; Thorne, T.J.; Sun, Y.P. Graphene oxides for homogeneous dispersion of carbon nanotubes. ACS Appl. Mater. Interfaces 2010, 2, 3217–3222. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Q.; Zhang, B.; Lin, X.; Shen, X.; Yousefi, N.; Huang, Z.D.; Li, Z.; Kim, J.K. Highly transparent and conducting ultralarge graphene oxide/single-walled carbon nanotube hybrid films produced by Langmuir-Blodgett assembly. J. Mater. Chem. 2012, 22, 25072–25082. [Google Scholar] [CrossRef]
- Gorkina, A.L.; Tsapenko, A.P.; Gilshteyn, A.P.; Koltsova, T.S.; Larionova, T.V.; Talyzin, A.; Anisimov, A.S.; Anoshkin, I.V.; Kauppinen, E.I.; Tolochko, O.V.; et al. Transparent and conductive hybrid graphene/carbon nanotube films. Carbon 2016, 100, 501–507. [Google Scholar] [CrossRef]
- Hecht, D.S.; Kaner, R.B. Solution-processed transparent electrodes. MRS Bull. 2011, 36, 749–755. [Google Scholar] [CrossRef]
- Li, X.; Gittleson, F.; Carmo, M.; Sekol, R.C.; Taylor, A.D. Scalable fabrication of multifunctional freestanding carbon nanotube/polymer composite thin films for energy conversion. ACS Nano 2012, 6, 1347–1356. [Google Scholar] [CrossRef] [PubMed]
- Geng, H.Z.; Lee, D.S.; Kim, K.K.; Kim, S.J.; Bae, J.J.; Lee, Y.H. Effect of carbon nanotube types in fabricating flexible transparent conducting films. J. Korean Phys. Soc. 2008, 53, 979–985. [Google Scholar] [CrossRef]
- Yu, L.; Shearer, C.; Shapter, J. Recent development of carbon nanotube transparent conductive films. Chem. Rev. 2016, 116, 13413–13453. [Google Scholar] [CrossRef] [PubMed]
- Imholt, T.J.; Dyke, C.A.; Hasslacher, B.; Perez, J.M.; Roberts, J.A.; Scott, J.B.; Wadhawan, A.; Ye, Z.; Tour, J.M. Nanotubes in microwave fields: Light emission, intense heat, outgassing, and reconstruction. Chem. Mater. 2003, 15, 3969–3970. [Google Scholar] [CrossRef]
- Voiry, D.; Yang, J.; Kupferberg, J.; Fullon, R.; Lee, C.; Jeong, H.Y.; Shin, H.S.; Chhowalla, M. High-quality graphene via microwave reduction of solution-exfoliated graphene oxide. Science 2016, 353, 1413–1416. [Google Scholar] [CrossRef] [PubMed]
- Zou, J.H.; Liu, L.W.; Chen, H.; Khondaker, S.I.; McCullough, R.D.; Huo, Q.; Zhai, L. Dispersion of pristine carbon nanotubes using conjugated block copolymers. Adv. Mater. 2008, 20, 2055–2060. [Google Scholar] [CrossRef]
- Zhang, L.; Xia, J.; Zhao, Q.; Liu, L.; Zhang, Z. Functional graphene oxide as a nanocarrier for controlled loading and targeted delivery of mixed anticancer drugs. Small 2010, 6, 537–544. [Google Scholar] [CrossRef] [PubMed]
- Hamedi, M.M.; Hajian, A.; Fall, A.B.; Håkansson, K.; Salajkova, M.; Lundell§, F.; Wågberg, L.; Berglund, L.A. Highly conducting, strong nanocomposites based on nanocellulose-assisted aqueous dispersions of single-wall carbon nanotubes. ACS Nano 2014, 8, 2467–2476. [Google Scholar] [CrossRef] [PubMed]
- Keunings, R.; Bousfield, D. Analysis of surface-tension driven leveling in viscoelastic films. J. Non-Newton. Fluid Mech. 1987, 22, 219–233. [Google Scholar] [CrossRef]
- Redon, C.; Brochard-Wyart, F.; Rondelez, F. Dynamics of dewetting. Phys. Rev. Lett. 1991, 66, 715–718. [Google Scholar] [CrossRef] [PubMed]
- Cohen, E.D.; Gutoff, E.B. Modern Coating and Drying Technology; VCH: New York, NY, USA, 1992; pp. 6–7. [Google Scholar]
- Nijenhuis, K.; McKinley, G.H.; Spiegelberg, S.; Barnes, H.A.; Aksel, N.; Heymann, L.A.; Odell, J. Thixotropy, rheopexy and yield Stress. In Springer Handbook of Experimental Fluid Mechanics; Tropea, C., Yarin, A.L., Foss, J.F., Eds.; Springer: Berlin, Germany, 2007; pp. 661–679. [Google Scholar]
- Irgens, F. Rheology and Non-Newtonian Fluids; Springer: Berlin, Germany, 2014; pp. 1–16. [Google Scholar]
- Meng, Y.Z.; Tjong, S.C. Rheology and morphology of compatibilized polyamide 6 blends containing liquid crystalline copolyesters. Polymer 1998, 39, 99–107. [Google Scholar] [CrossRef]
- Tjong, S.C.; Meng, Y.Z. Morphology and mechanical characteristics of compatibilized polyamide 6-liquid crystalline polymer composites. Polymer 1997, 38, 4609–4615. [Google Scholar] [CrossRef]
- Tjong, S.C.; Liu, S.L.; Li, R.K.Y. Mechanical properties of injection moulded blends of polypropylene with thermotropic liquid crystalline polymer. J. Mater. Sci. 1996, 31, 479–484. [Google Scholar] [CrossRef]
- Meng, Y.Z.; Tjong, S.C.; Hay, A.S.; Wang, S.J. Synthesis and proton conductivities of phosphonic acid containing poly-(arylene ether)s. J. Polym. Sci. A Polym. Chem. 2001, 39, 3218–3226. [Google Scholar] [CrossRef]
- Nicholas, A.; Parra-Vasquez, G.; Stepanek, I.; Davis, V.A.; Moore, V.C.; Haroz, E.H.; Shaver, J.; Hauge, R.H.; Smalley, R.E.; Pasquali, M. Simple length determination of single-walled carbon nanotubes by viscosity measurements in dilute suspensions. Macromolecules 2007, 40, 4043–4047. [Google Scholar]
- Satas, D.; Tracton, A.A. Coatings Technology Handbook, 2nd ed.; Marcel Dekker: New York, NY, USA, 2000; p. 132. [Google Scholar]
- Jackson, R.; Domercq, B.; Jain, R.; Kippelen, B.; Graham, S. Stability of doped transparent carbon nanotube electrodes. Adv. Funct. Mater. 2008, 18, 2548–2554. [Google Scholar] [CrossRef]
- Zhang, M.; Fang, S.; Zakhidov, A.A.; Lee, S.B.; Aliev, A.E.; Williams, C.D.; Atkinson, K.R.; Baughman, R.H. Strong, transparent, multifunctional, carbon nanotube sheets. Science 2005, 309, 1215–1219. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.Y.; Chen, T.H.; Chang, S.C.; Cheng, S.Y.; Chin, T.S. Strong carbon-nanotube-polymer bonding by microwave irradiation. Adv. Funct. Mater. 2007, 17, 1979–1983. [Google Scholar] [CrossRef]
- Wang, C.Y.; Chen, T.H.; Chang, S.C.; Chin, T.S.; Cheng, S.Y. Flexible field emitter made of carbon nanotubes microwave welded onto polymer substrates. Appl. Phys. Lett. 2007, 90, 103111. [Google Scholar] [CrossRef]
- Shim, H.C.; Kwak, Y.K.; Han, C.S.; Kim, S. Enhancement of adhesion between carbon nanotubes and polymer substrates using microwave irradiation. Scr. Mater. 2009, 61, 32–35. [Google Scholar] [CrossRef]
- Han, J.T.; Kim, D.; Kim, J.S.; Seol, S.K.; Jeong, S.Y.; Jeong, H.J.; Chang, W.S.; Lee, G.W.; Jung, S. Self-passivation of transparent single-walled carbon nanotube films on plastic substrates by microwave-induced rapid nanowelding. Appl. Phys. Lett. 2012, 100, 163120. [Google Scholar]

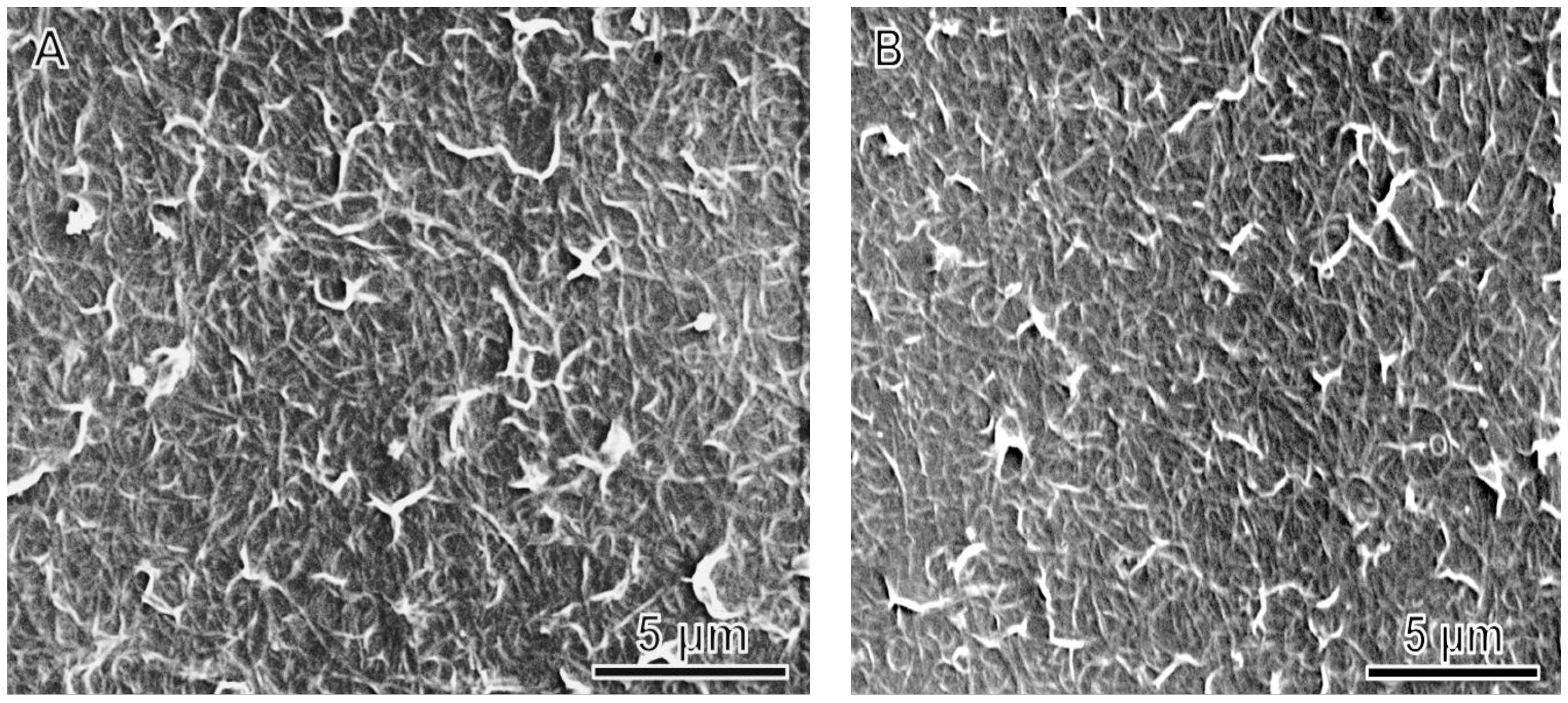
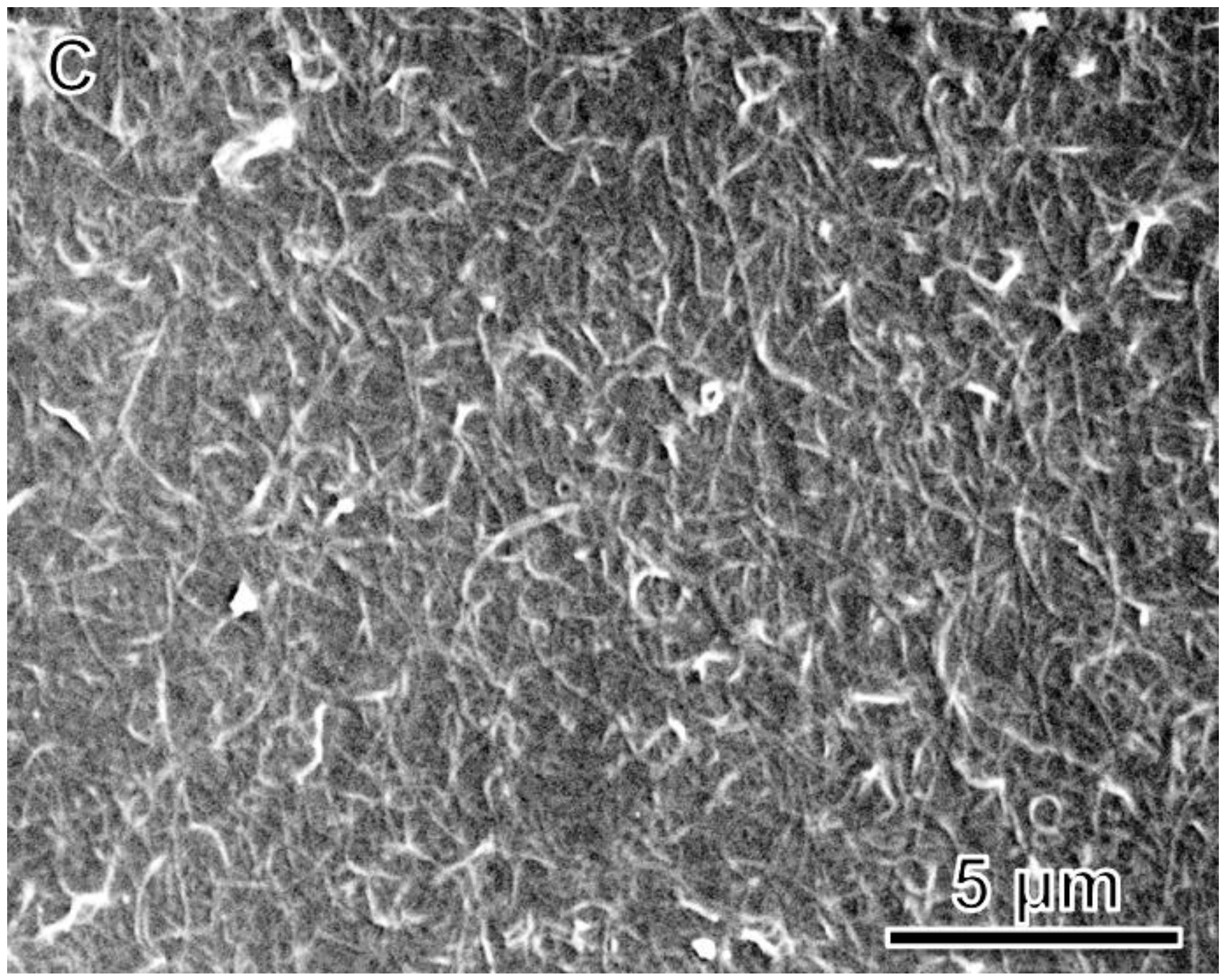
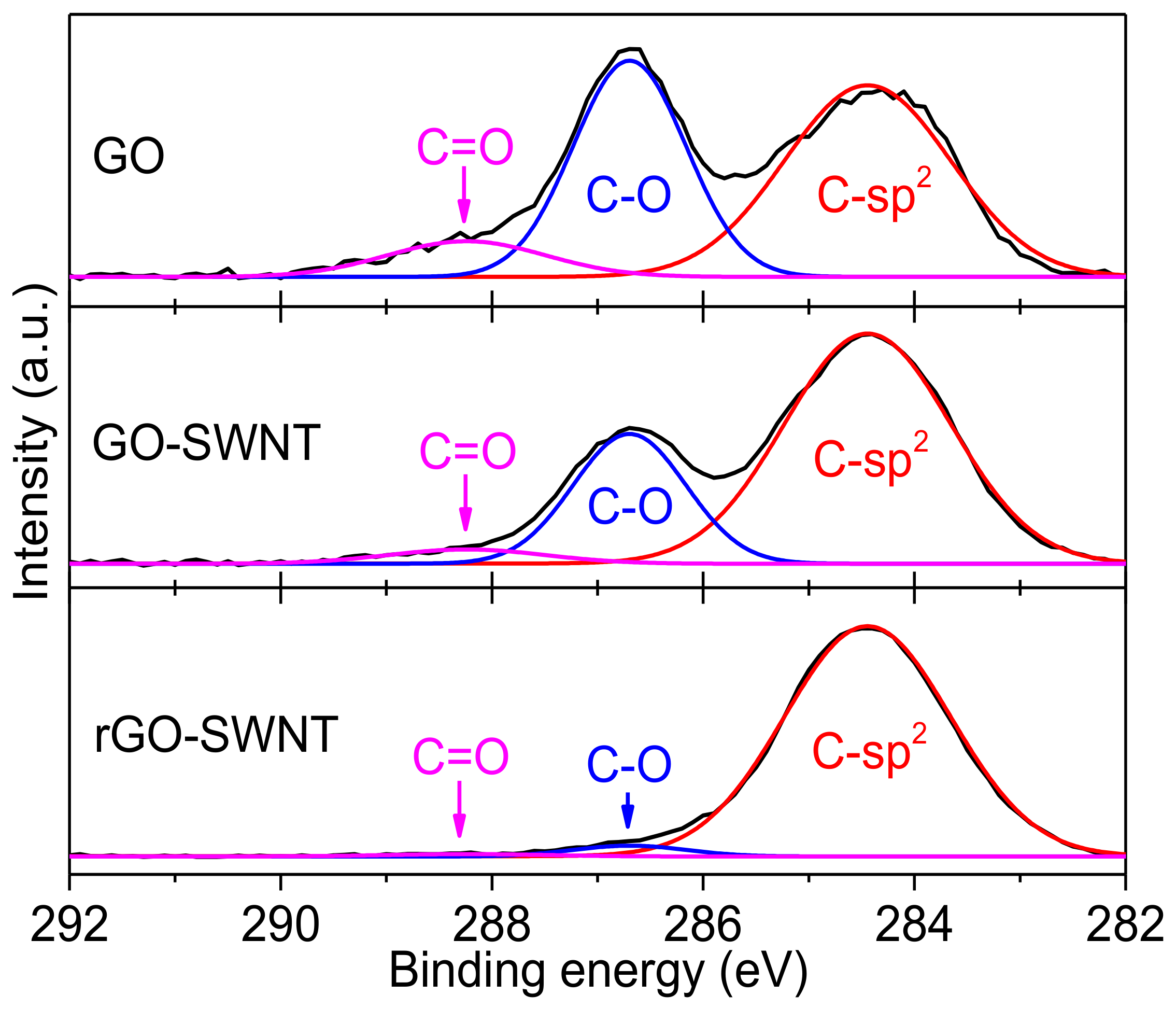
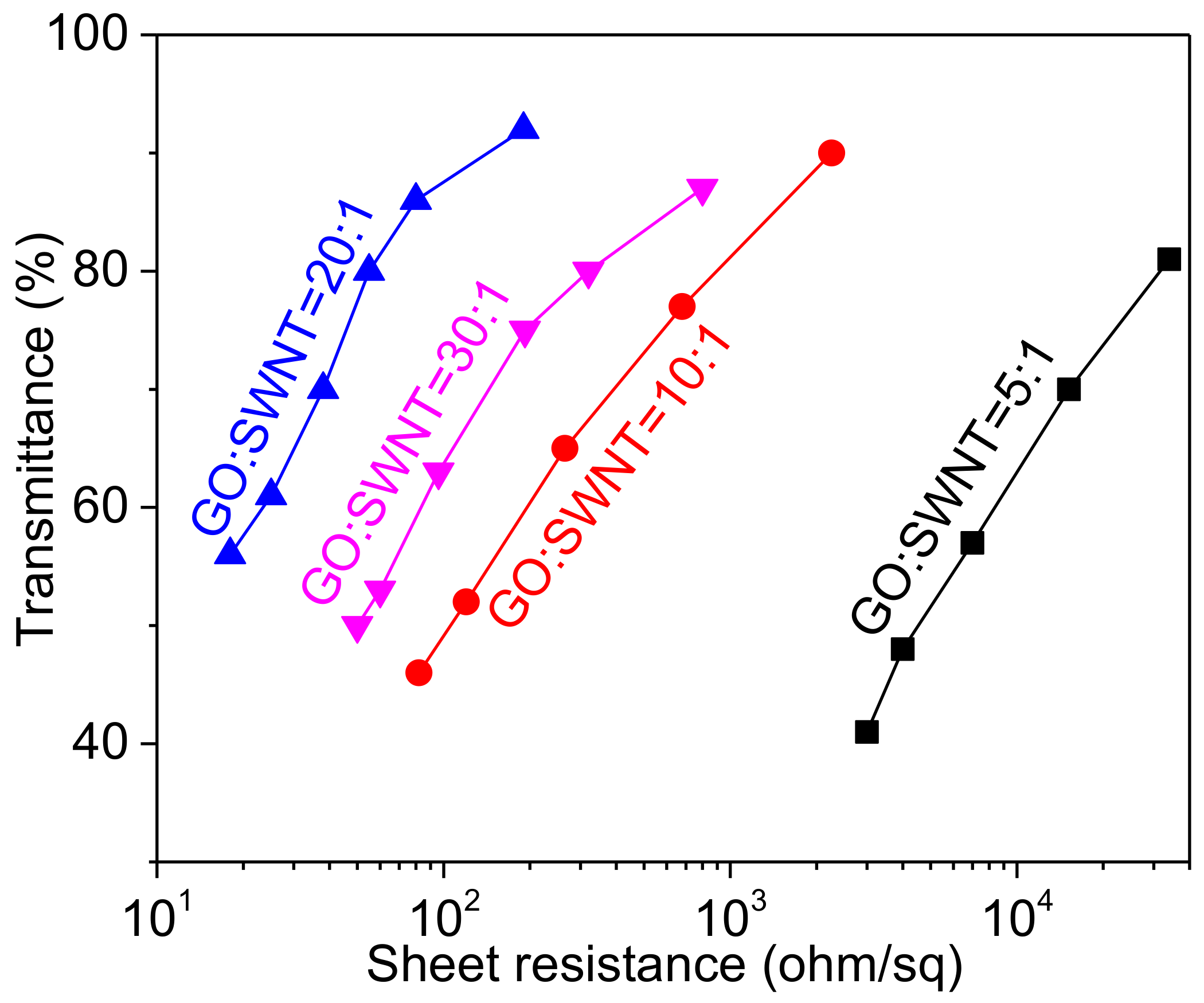


| Ink Composition | tlevel (s) | tdry (s) | tdewet (s) |
|---|---|---|---|
| 0.2 mg/mL SWNT + 1 mg/mL GO | 8.33 × 10−4 | 600 | 3.32 × 103 |
| 0.2 mg/mL SWNT + 2 mg/mL GO | 1.10 × 10−3 | 500 | 6.16 × 103 |
| 0.2 mg/mL SWNT + 4 mg/mL GO | 1.41 × 10−3 | 400 | 1.12 × 104 |
| 0.2 mg/mL SWNT + 6 mg/mL GO | 1.83 × 10−3 | 300 | 2.05 × 104 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, L.; Liao, C.; Tjong, S.C. Scalable Fabrication of High-Performance Transparent Conductors Using Graphene Oxide-Stabilized Single-Walled Carbon Nanotube Inks. Nanomaterials 2018, 8, 224. https://doi.org/10.3390/nano8040224
He L, Liao C, Tjong SC. Scalable Fabrication of High-Performance Transparent Conductors Using Graphene Oxide-Stabilized Single-Walled Carbon Nanotube Inks. Nanomaterials. 2018; 8(4):224. https://doi.org/10.3390/nano8040224
Chicago/Turabian StyleHe, Linxiang, Chengzhu Liao, and Sie Chin Tjong. 2018. "Scalable Fabrication of High-Performance Transparent Conductors Using Graphene Oxide-Stabilized Single-Walled Carbon Nanotube Inks" Nanomaterials 8, no. 4: 224. https://doi.org/10.3390/nano8040224
APA StyleHe, L., Liao, C., & Tjong, S. C. (2018). Scalable Fabrication of High-Performance Transparent Conductors Using Graphene Oxide-Stabilized Single-Walled Carbon Nanotube Inks. Nanomaterials, 8(4), 224. https://doi.org/10.3390/nano8040224





