Can Pulsed Electromagnetic Fields Trigger On-Demand Drug Release from High-Tm Magnetoliposomes?
Abstract
:1. Introduction
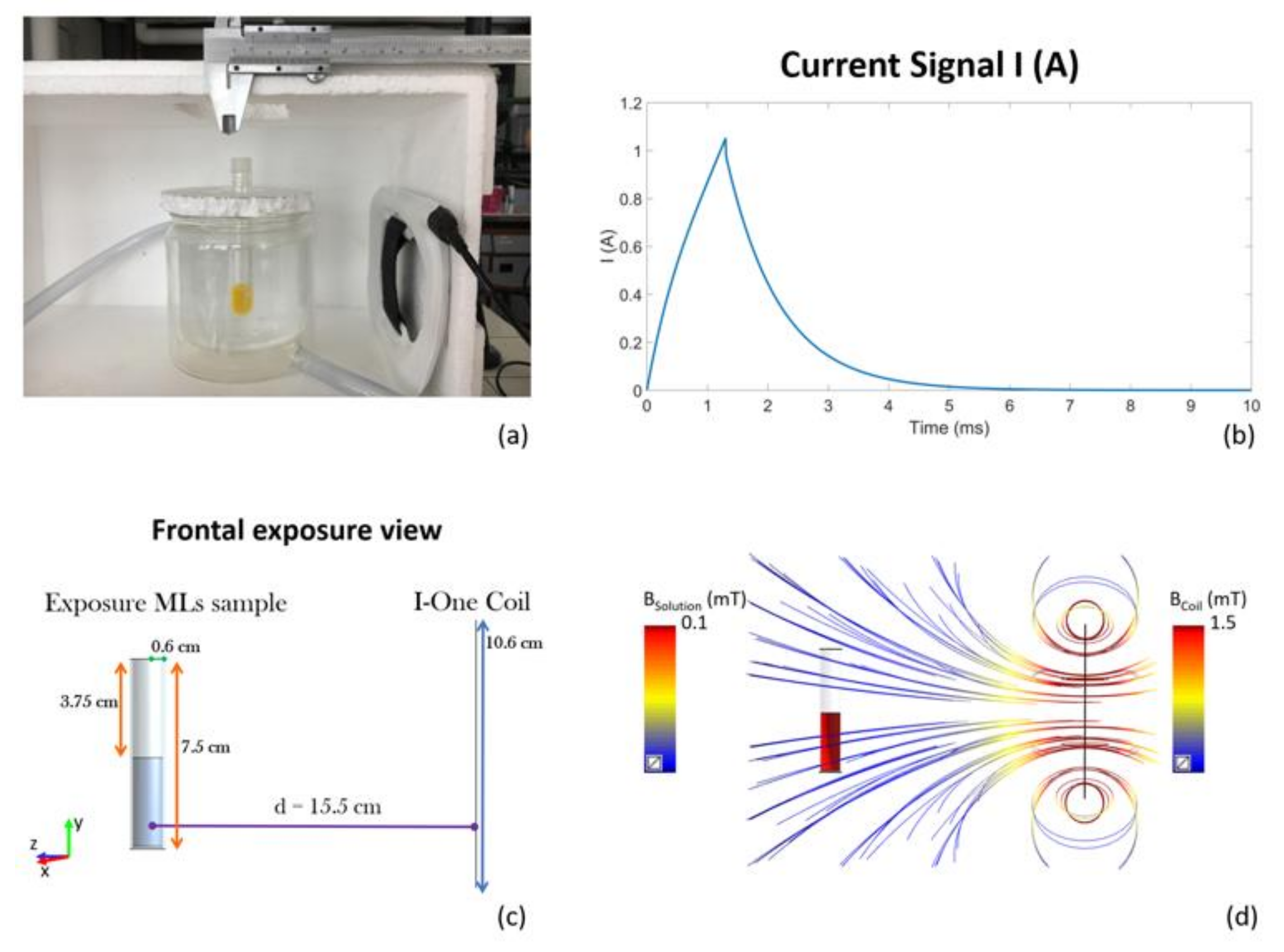
2. Materials and Methods
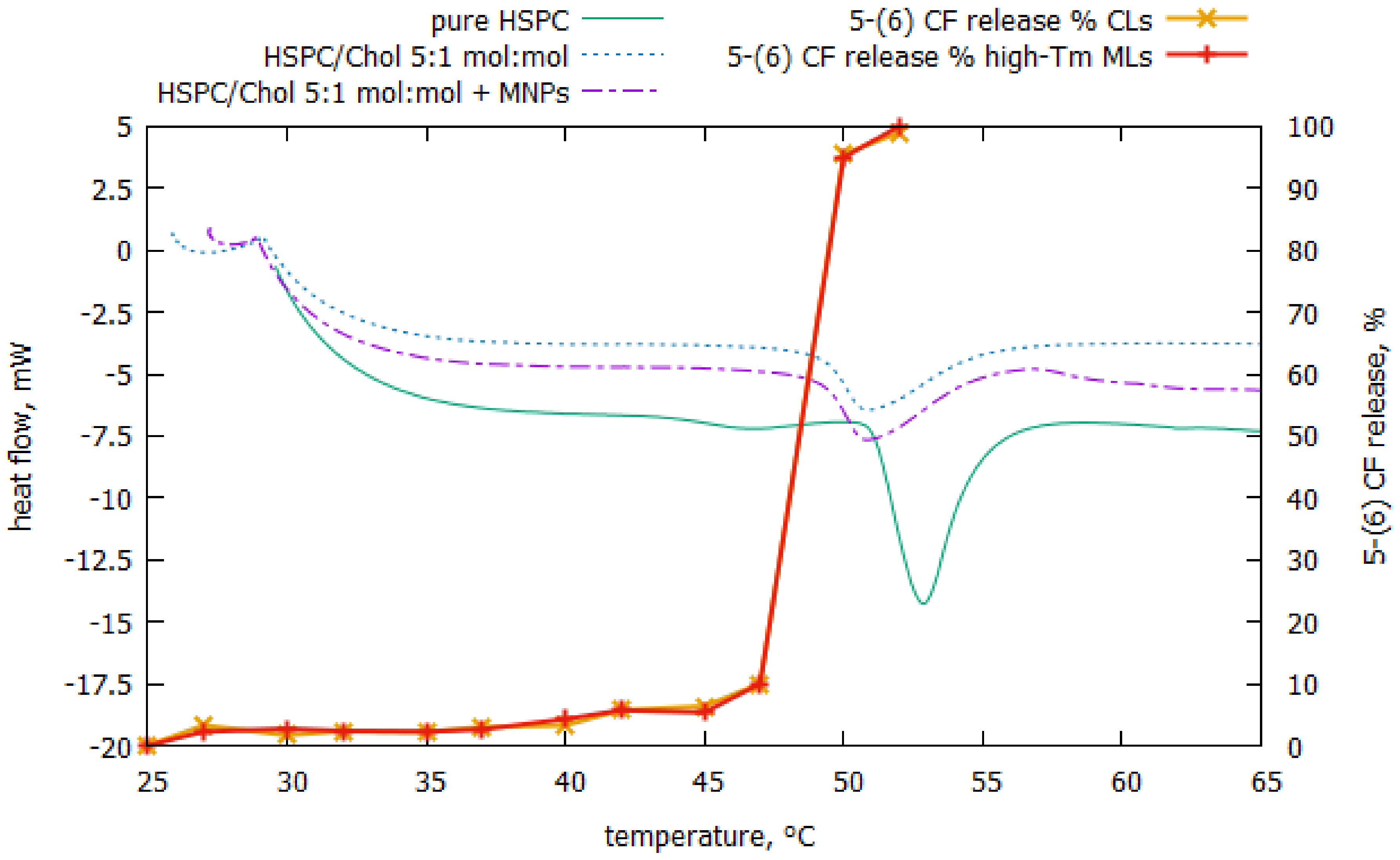
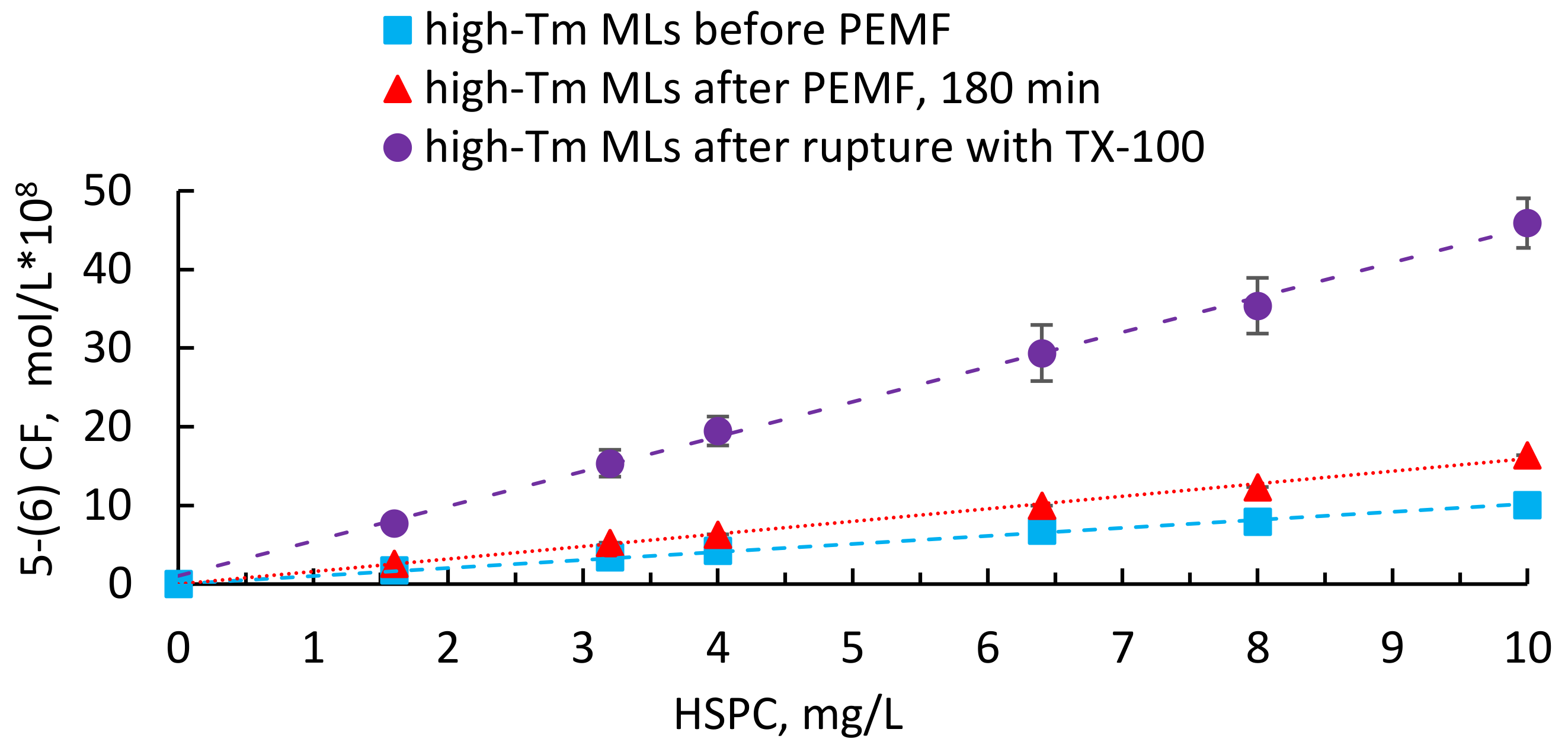
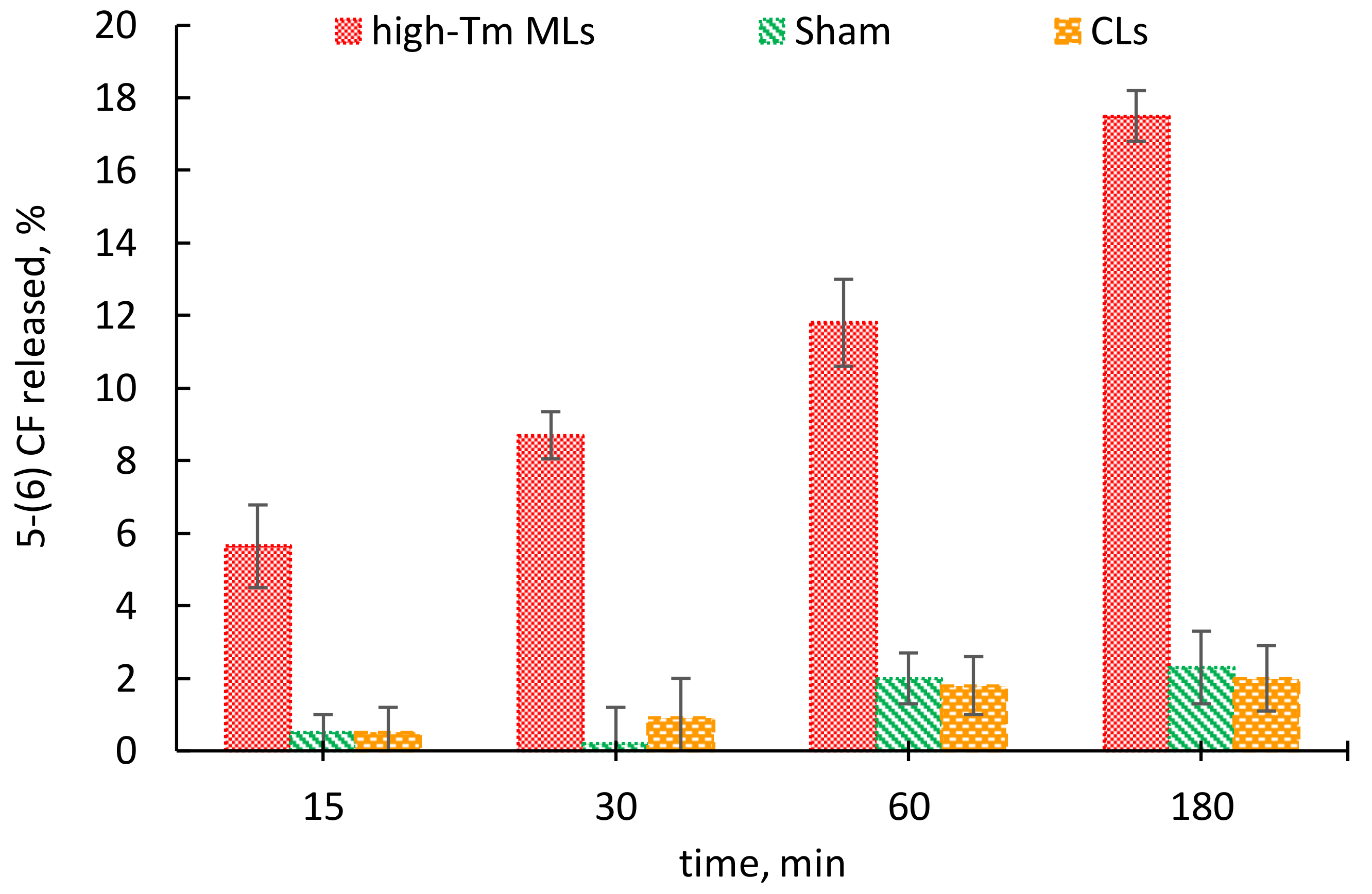
3. Results
4. Discussion
Supplementary Materials
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ganta, S.; Devalapally, H.; Shahiwala, A.; Amiji, M. A review of stimuli-responsive nanocarriers for drug and gene delivery. J. Control. Release 2008, 126, 187–204. [Google Scholar] [CrossRef] [PubMed]
- Mura, S.; Nicolas, J.; Couvreur, P. Stimuli-responsive nanocarriers for drug delivery. Nat. Mater. 2012, 12, 991–1003. [Google Scholar] [CrossRef] [PubMed]
- Leung, S.J.; Romanowski, M. Light-activated content release from liposomes. Theranostics 2012, 2, 1020–1036. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, A.; Kost, J.; Barenholz, Y. Ultrasound, liposomes and drug delivery: Principles for using ultrasound to control the release of drugs from liposomes. Chem. Phys. Lipids 2009, 162, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Karanth, H.; Murthy, R.S. pH-sensitive liposomes-principle and application in cancer therapy. J. Pharm. Pharmacol. 2007, 59, 469–483. [Google Scholar] [CrossRef] [PubMed]
- Pradhan, P.; Giri, J.; Rieken, F.; Koch, C.; Mykhaylyk, O.; Döblinger, M.; Banerjee, R.; Bahadur, D.; Plank, C. Targeted temperature sensitive magnetic liposomes for thermo-chemotherapy. J. Control. Release 2010, 142, 108–121. [Google Scholar] [CrossRef] [PubMed]
- Preiss, M.R.; Bothun, G.D. Stimuli-responsive liposome-nanoparticle assemblies. Expert Opin. Drug Deliv. 2011, 8, 1025–1040. [Google Scholar] [CrossRef] [PubMed]
- Kurd, K.; Khandagi, A.A.; Davaran, S.; Akbarzadeh, A. Cisplatin release from dual-responsive magnetic nanocomposites. Artif. Cells Nanomed. Biotechnol. 2016, 44, 1031–1039. [Google Scholar] [CrossRef] [PubMed]
- Babincová, M.; Cicmanec, P.; Altanerová, V.; Altaner, C.; Babinec, P. AC-magnetic field controlled drug release from magnetoliposomes: Design of a method for site-specific chemotherapy. Bioelectrochemistry 2002, 55, 17–19. [Google Scholar] [CrossRef]
- Viroonchatapan, E.; Sato, H.; Ueno, M.; Adachi, I.; Tazawa, K.; Horikoshi, I. Release of 5-Fluorouracil from Thermosensitive Magnetoliposomes Induced by an Electromagnetic Field. J. Control. Release 1997, 46, 263–271. [Google Scholar] [CrossRef]
- Gupta, A.K.; Naregalkar, R.R.; Vaidya, V.D.; Gupta, M. Recent advances on surface engineering of magnetic iron oxide nanoparticles and their biomedical applications. Nanomedicine 2010, 2, 23–39. [Google Scholar] [CrossRef] [PubMed]
- Nappini, S.; Fogli, S.; Castroflorio, B.; Bonini, M.; Bombelli, F.B.; Baglioni, P. Magnetic field responsive drug release from magnetoliposomes in biological fluids. J. Mater. Chem. B 2016, 4, 716–725. [Google Scholar] [CrossRef] [Green Version]
- Golovin, Y.I.; Gribanovsky, S.L.; Golovin, D.Y.; Klyachko, N.L.; Majouga, A.G.; Master, А.M.; Kabanov, A.V. Towards nanomedicines of the future: Remote magneto-mechanical actuation of nanomedicines by alternating magnetic fields. J. Control. Release 2015, 219, 43–60. [Google Scholar] [CrossRef] [PubMed]
- Dobson, J. Remote control of cellular behaviour with magnetic nanoparticles. Nat. Nanotechnol. 2008, 3, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Klyachko, N.L.; Sokolsky-Papkov, M.; Pothayee, N.; Efremova, M.V.; Gulin, D.A.; Pothayee, N.; Kabanov, A.V. Changing the Enzyme Reaction Rate in Magnetic Nanosuspensions by a Non-Heating Magnetic Field. Angew. Chem. Int. Ed. 2012, 51, 12016–12019. [Google Scholar] [CrossRef] [PubMed]
- Spera, R.; Apollonio, F.; Liberti, M.; Paffi, A.; Merla, C.; Pinto, R.; Petralito, S. Controllable release from high-transition temperature magnetoliposomes by low-level magnetic stimulation. Colloids Surf. B Biointerfaces 2015, 131, 136–140. [Google Scholar] [CrossRef] [PubMed]
- Spera, R.; Petralito, S.; Liberti, M.; Merla, C.; D’Inzeo, G.; Pinto, R.; Apollonio, F. Controlled release from magnetoliposomes aqueous suspensions exposed to a low intensity magnetic field. Bioelectromagnetics 2014, 35, 309–312. [Google Scholar] [CrossRef] [PubMed]
- Varani, K.; Gessi, S.; Merighi, S.; Iannotta, V.; Cattabriga, E.; Spisani, S.; Cadossi, R.; Borea, P.A. Effect of low frequency electromagnetic fields on a2a adenosine receptors in human neutrophils. Br. J. Pharmacol. 2002, 136, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Varani, K.; De Mattei, M.; Vincenzi, F.; Gessi, S.; Merighi, S.; Pellati, A.; Ongaro, A.; Caruso, A.; Cadossi, R.; Borea, P.A. Characterization of adenosine receptors in bovine chondrocytes and fibroblast-like synoviocytes exposed to low frequency low energy pulsed electromagnetic fields. Osteoarthr. Cartil. 2008, 16, 292–304. [Google Scholar] [CrossRef] [PubMed]
- Varani, K.; Vincenzi, F.; Ravani, A.; Pasquini, S.; Merighi, S.; Gessi, S.; Setti, S.; Cadossi, M.; Borea, P.A.; Cadossi, R. Adenosine receptors as a biological pathway for the anti-inflammatory and beneficial effects of low frequency low energy pulsed electromagnetic fields. Mediators Inflamm. 2017, 2017, 2740963. [Google Scholar] [CrossRef] [PubMed]
- Martina, M.S.; Fortin, J.P.; Menager, C.; Clement, O.; Barratt, G.; Grabielle-Madelmont, C.; Gazeau, F.; Cabuil, V.; Lesieur, S. Generation of Superparamagnetic Liposomes Revealed as Highly Efficient MRI Contrast Agents for In Vivo Imaging. J. Am. Chem. Soc. 2005, 127, 10676–10685. [Google Scholar] [CrossRef] [PubMed]
- Belikov, V.G.; Kuregyan, A.G.; Ismailova, G.K. Standardization of magnetite. Pharm. Chem. J. 2002, 36, 333–336. [Google Scholar] [CrossRef]
- Kitayama, H.; Takechi, Y.; Tamai, N.; Matsuki, H.; Yomota, C.; Saito, H. Thermotropic Phase Behavior of Hydrogenated Soybean Phosphatidylcholine–Cholesterol Binary Liposome Membrane. Chem. Pharm. Bull. 2014, 62, 58–63, doiorg/101248/cpbc13. [Google Scholar] [CrossRef] [PubMed]
- Veronesi, F.; Torricelli, P.; Giavaresi, G.; Sartori, M.; Cavani, F.; Setti, S.; Cadossi, M.; Ongaro, A.; Fini, M. In vivo effect of two different pulsed electromagnetic field frequencies on osteoarthritis. J. Orthop. Res. 2014, 32, 677–685. [Google Scholar] [CrossRef] [PubMed]
- Gobbi, A.; Karnatzikos, G.; Chaurasia, S. Pulsed Electromagnetic Fields for the Treatment of Symptomatic Patellofemoral Cartilage Lesions of the Knee. In The Patellofemoral Joint; Gobbi, A., Mendes, J.E., Nakamura, N., Eds.; Springer: Heidelberg/Berlin, Germany, 2014; pp. 261–266. ISBN 978-3-642-54965-6. [Google Scholar]
- Van den Hoven, J.M.; Van Tomme, S.R.; Metselaar, J.M.; Nuijen, B.; Beijnen, J.H.; Storm, G. Liposomal Drug Formulations in the Treatment of Rheumatoid Arthritis. Mol. Pharm. 2011, 8, 1002–1015. [Google Scholar] [CrossRef] [PubMed]
- Chuang, S.Y.; Lin, C.H.; Huang, T.H.; Fang, J.Y. Lipid-Based Nanoparticles as a Potential Delivery Approach in the Treatment of Rheumatoid Arthritis. Nanomaterials 2018, 8, 42–58. [Google Scholar] [CrossRef] [PubMed]
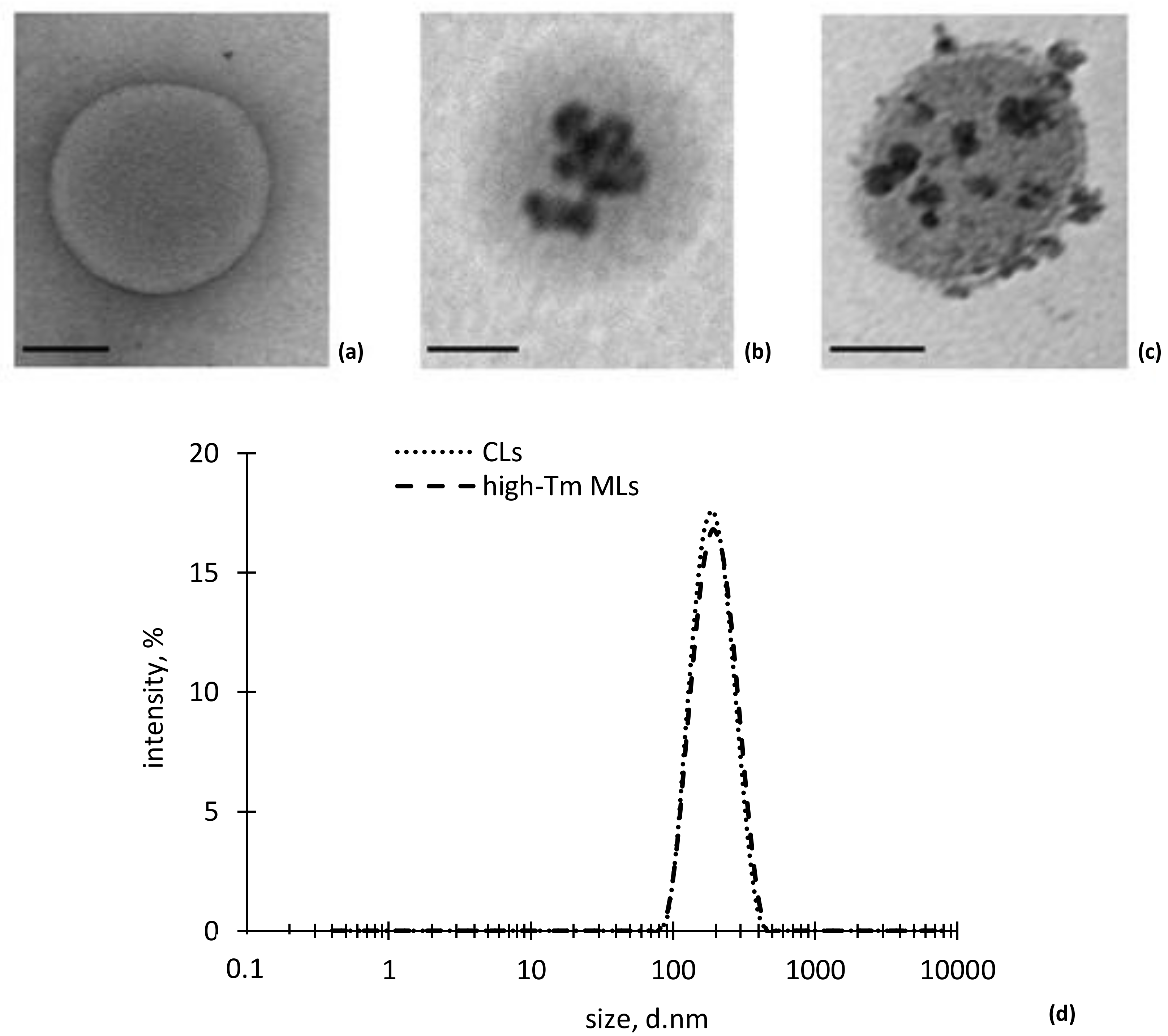
| Sample | CLs | High-Tm MLs |
|---|---|---|
| Hydrodynamic diameter (nm) | 220.9 ± 22.4 | 240.9 ± 26.6 |
| PdI | 0.046 ± 0.028 | 0.131 ± 0.031 |
| ζ-potential (mV) | −10.28 ± 1.43 | −15.42 ± 1.51 |
| 5-(6) CF loading efficiency (µL/mg HSPC) | 2.29 ± 0.26 | 1.84 ± 0.13 |
| Fe3O4 loading efficiency (g/mmol HSPC) | - | 0.2 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nardoni, M.; Della Valle, E.; Liberti, M.; Relucenti, M.; Casadei, M.A.; Paolicelli, P.; Apollonio, F.; Petralito, S. Can Pulsed Electromagnetic Fields Trigger On-Demand Drug Release from High-Tm Magnetoliposomes? Nanomaterials 2018, 8, 196. https://doi.org/10.3390/nano8040196
Nardoni M, Della Valle E, Liberti M, Relucenti M, Casadei MA, Paolicelli P, Apollonio F, Petralito S. Can Pulsed Electromagnetic Fields Trigger On-Demand Drug Release from High-Tm Magnetoliposomes? Nanomaterials. 2018; 8(4):196. https://doi.org/10.3390/nano8040196
Chicago/Turabian StyleNardoni, Martina, Elena Della Valle, Micaela Liberti, Michela Relucenti, Maria Antonietta Casadei, Patrizia Paolicelli, Francesca Apollonio, and Stefania Petralito. 2018. "Can Pulsed Electromagnetic Fields Trigger On-Demand Drug Release from High-Tm Magnetoliposomes?" Nanomaterials 8, no. 4: 196. https://doi.org/10.3390/nano8040196
APA StyleNardoni, M., Della Valle, E., Liberti, M., Relucenti, M., Casadei, M. A., Paolicelli, P., Apollonio, F., & Petralito, S. (2018). Can Pulsed Electromagnetic Fields Trigger On-Demand Drug Release from High-Tm Magnetoliposomes? Nanomaterials, 8(4), 196. https://doi.org/10.3390/nano8040196






