Preparation of a Sepia Melanin and Poly(ethylene-alt-maleic Anhydride) Hybrid Material as an Adsorbent for Water Purification
Abstract
:1. Introduction
2. Results and Discussion
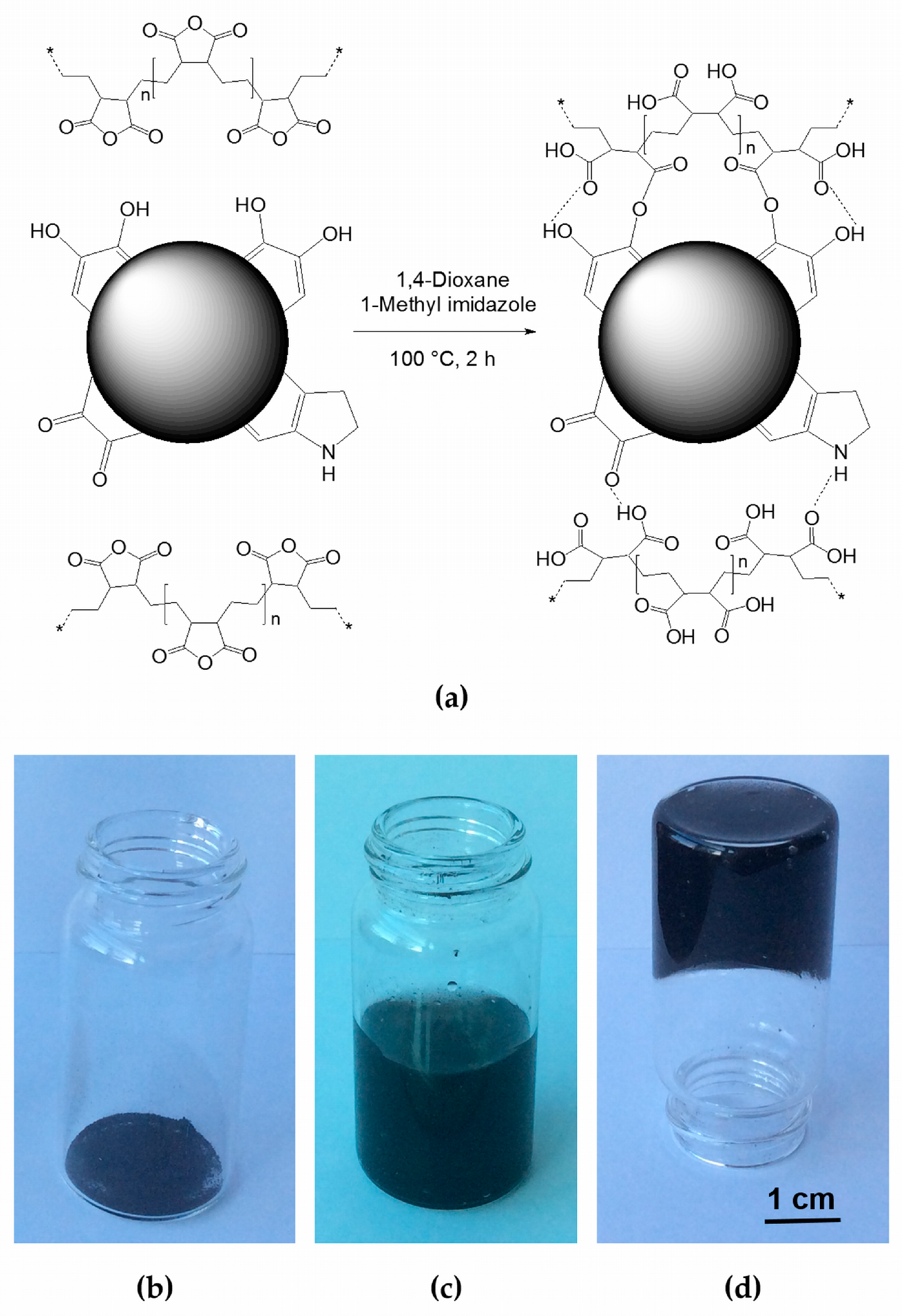
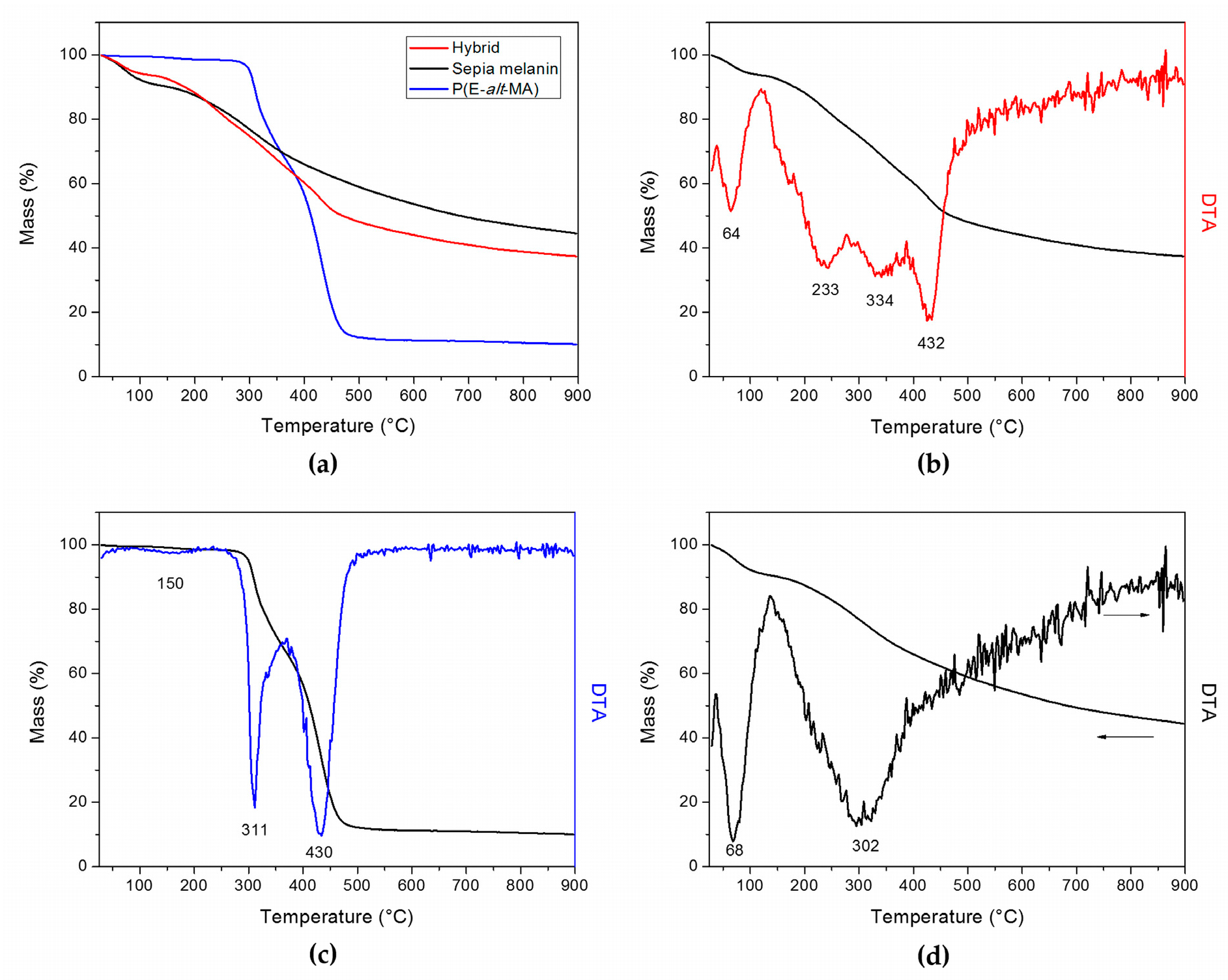
2.1. Sepia Melanin-P(E-alt-MA) Hybrid: Its Synthesis and Characterization
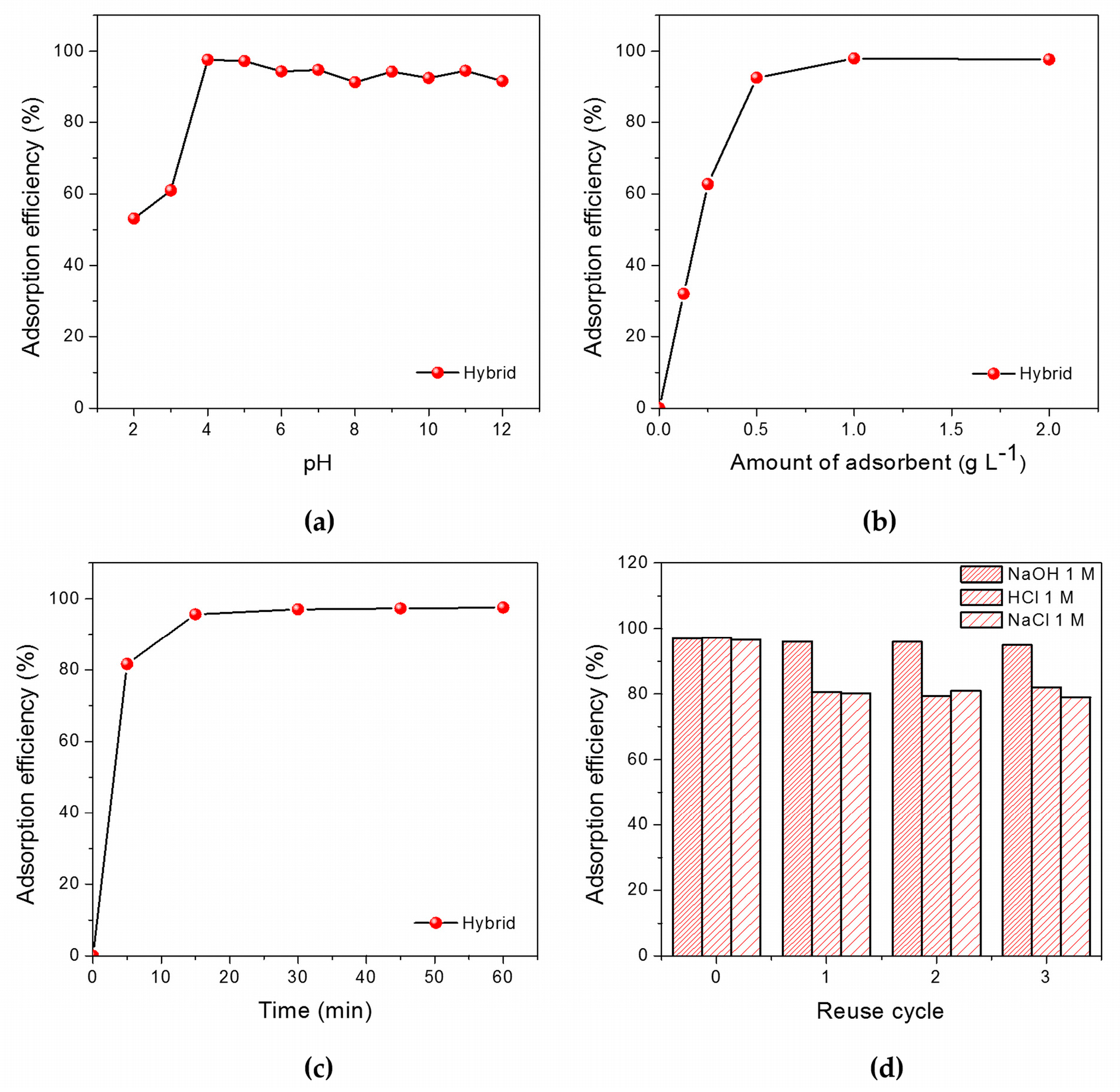
2.2. Evaluation of the Adsorption Efficiency
3. Materials and Methods
3.1. Synthesis of the Sepia Melanin-P(E-alt-MA) Hybrid
3.2. Adsorption Studies
3.2.1. Effect of pH on Adsorption Efficiency
3.2.2. Effect of Adsorbent Dosage
3.2.3. Effect of Adsorption Time
3.2.4. Reuse of Adsorbent
4. Conclusions
Supplementary Materials
Author Contributions
Conflicts of Interest
References
- Schwarzenbach, R.P.; Egli, T.; Hofstetter, T.B.; von Gunten, U.; Wehrli, B. Global Water Pollution and Human Health. Annu. Rev. Environ. Resour. 2010, 35, 109–136. [Google Scholar] [CrossRef]
- Wang, Q.; Yang, Z. Industrial water pollution, water environment treatment, and health risks in China. Environ. Pollut. 2016, 218, 358–365. [Google Scholar] [CrossRef] [PubMed]
- Shannon, M.; Bohn, P.W.; Elimelech, M.; Georgiadis, J.G.; Mariñas, B.J.; Mayes, A.M. Science and technology for water purification in the coming decades. Nature 2008, 452, 301–310. [Google Scholar] [CrossRef] [PubMed]
- Tuck, C.O.; Perez, E.; Horvath, I.T.; Sheldon, R.A.; Poliakoff, M. Valorization of Biomass: Deriving More Value from Waste. Science 2012, 337, 695–699. [Google Scholar] [CrossRef] [PubMed]
- Riley, P.A. Melanin. Int. J. Biochem. Cell Biol. 1997, 29, 1235–1239. [Google Scholar] [CrossRef]
- Piattelli, M.; Nicolaus, R.A. The structure of melanins and melanogenesis—I: The structure of melanin in Sepia. Tetrahedron 1961, 15, 66–75. [Google Scholar] [CrossRef]
- Pezzella, A.; D’Ischia, M.; Napolitano, A.; Palumbo, A.; Prota, G. An integrated approach to the structure of sepia melanin. Evidence for a high proportion of degraded 5,6-Dihydroxyindole-2-carboxylic acid units in the pigment backbone. Tetrahedron 1997, 53, 8281–8286. [Google Scholar] [CrossRef]
- Hong, L.; Simon, J.D. Current understanding of the binding sites, capacity, affinity, and biological significance of metals in melanin. J. Phys. Chem. B 2007, 111, 7938–7947. [Google Scholar] [CrossRef] [PubMed]
- Sono, K.; Lye, D.; Moore, C.A.; Boyd, W.C.; Gorlin, T.A.; Belitsky, J.M. Melanin-based coatings as lead-binding agents. Bioinorg. Chem. Appl. 2012, 2012, 361803. [Google Scholar] [CrossRef] [PubMed]
- Bikiaris, D.; Prinos, J.; Koutsopoulos, K.; Vouroutzis, N.; Pavlidou, E.; Frangis, N.; Panayiotou, C. LDPE/plasticized starch blends containing PE-g-MA copolymer as compatibilizer. Polym. Degrad. Stab. 1998, 59, 287–291. [Google Scholar] [CrossRef]
- Di Corato, R.; Quarta, A.; Piacenza, P.; Ragusa, A.; Figuerola, A.; Buonsanti, R.; Cingolani, R.; Manna, L.; Pellegrino, T. Water solubilization of hydrophobic nanocrystals by means of poly(maleic anhydride-alt-1-octadecene). J. Mater. Chem. 2008, 18, 1991–1996. [Google Scholar] [CrossRef]
- Pompe, T.; Zschoche, S.; Herold, N.; Salchert, K.; Gouzy, M.F.; Sperling, C.; Werner, C. Maleic anhydride copolymers—A versatile platform for molecular biosurface engineering. Biomacromolecules 2003, 4, 1072–1079. [Google Scholar] [CrossRef] [PubMed]
- Yoo, S.; Yi, M.H.; Kim, Y.H.; Jang, K.S. One-pot surface modification of poly(ethylene-alt-maleic anhydride) gate insulators for low-voltage DNTT thin-film transistors. Org. Electron. 2016, 33, 263–268. [Google Scholar] [CrossRef]
- Rivas, B.L.; Pooley, S.A.; Pereira, E.; Montoya, E.; Cid, R. Poly(ethylene-alt-maleic acid) as complexing reagent to separate metal ions using membrane filtration. J. Appl. Polym. Sci. 2006, 101, 2057–2061. [Google Scholar] [CrossRef]
- Masoumi, A.; Ghaemy, M. Adsorption of heavy metal ions and azo dyes by crosslinked nanochelating resins based on poly(methylmethacrylate-co-maleic anhydride). Express Polym. Lett. 2014, 8, 187–196. [Google Scholar] [CrossRef]
- Kabiri, K.; Omidian, H.; Zohuriaan-Mehr, M.J.; Doroudiani, S. Superabsorbent hydrogel composites and nanocomposites: A review. Polym. Compos. 2011, 32, 277–289. [Google Scholar] [CrossRef]
- Kopeĉek, J. Hydrogels: From soft contact lenses and implants to self-assembled nanomaterials. J. Polym. Sci. Part A Polym. Chem. 2009, 47, 5929–5946. [Google Scholar] [CrossRef] [PubMed]
- Watt, A.A.R.; Bothma, J.P.; Meredith, P. The supramolecular structure of melanin. Soft Matter 2009, 5, 3754. [Google Scholar] [CrossRef]
- Chedekel, M.R.; Ahene, A.B.; Zeise, L. Melanin Standard Method: Empirical Formula 2. Pigment Cell Res. 1992, 5, 240–246. [Google Scholar] [CrossRef] [PubMed]
- Bridelli, M.G.; Crippa, P.R. Infrared and water sorption studies of the hydration structure and mechanism in natural and synthetic melanin. J. Phys. Chem. B 2010, 114, 9381–9390. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Marín, A.M.; Sánchez, C.I. Thermal and mass spectroscopic characterization of a sulphur-containing bacterial melanin from Bacillus subtilis. J. Non-Cryst. Solids 2010, 356, 1576–1580. [Google Scholar] [CrossRef]
- Fu, J.; Chen, Z.; Wang, M.; Liu, S.; Zhang, J.; Zhang, J.; Han, R.; Xu, Q. Adsorption of methylene blue by a high-efficiency adsorbent (polydopamine microspheres): Kinetics, isotherm, thermodynamics and mechanism analysis. Chem. Eng. J. 2015, 259, 53–61. [Google Scholar] [CrossRef]
- Rice, L.; Wainwright, M.; Phoemix, D.A. Phenothiazine photosensitizers. III. Activity of methylene blue derivatives against pigmented melanoma cell lines. J. Chemother. 2000, 12, 94–104. [Google Scholar] [CrossRef] [PubMed]
- Delben, F.; Paoletti, S.; Porasso, R.D.; Benegas, J.C. Potentiometric titrations of maleic acid copolymers in dilute aqueous solution: Experimental results and theoretical interpretation. Macromol. Chem. Phys. 2006, 207, 2299–2310. [Google Scholar] [CrossRef]
- Zhang, J.; Cai, J.; Deng, Y.; Chen, Y.; Ren, G. Characterization of melanin produced by a wild-type strain of Bacillus cereus. Front. Biol. China 2007, 2, 26–29. [Google Scholar] [CrossRef]

| P(E-alt-MA) | Sepia Melanin | Hybrid | |||||
|---|---|---|---|---|---|---|---|
| C (%) | O (%) | C (%) | O (%) | N (%) | C (%) | O (%) | N (%) |
| 58 ± 0.73 | 41 ± 0.99 | 53 ± 1.3 | 35 ± 1.4 | 11 ± 2.5 | 58 ± 3.0 | 35 ± 3.9 | 7.2 ± 2.7 |
| C/O 1.4 | C/O 1.5 | C/O 1.7 | |||||
| C/N 5 | C/N 8 | ||||||
| Sample | Residual Mass (%) |
|---|---|
| P(E-alt-MA) | 10.03 |
| Sepia melanin | 44.46 |
| Hybrid | 37.40 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Panzarasa, G.; Osypova, A.; Consolati, G.; Quasso, F.; Soliveri, G.; Ribera, J.; Schwarze, F.W.M.R. Preparation of a Sepia Melanin and Poly(ethylene-alt-maleic Anhydride) Hybrid Material as an Adsorbent for Water Purification. Nanomaterials 2018, 8, 54. https://doi.org/10.3390/nano8020054
Panzarasa G, Osypova A, Consolati G, Quasso F, Soliveri G, Ribera J, Schwarze FWMR. Preparation of a Sepia Melanin and Poly(ethylene-alt-maleic Anhydride) Hybrid Material as an Adsorbent for Water Purification. Nanomaterials. 2018; 8(2):54. https://doi.org/10.3390/nano8020054
Chicago/Turabian StylePanzarasa, Guido, Alina Osypova, Giovanni Consolati, Fiorenza Quasso, Guido Soliveri, Javier Ribera, and Francis W. M. R. Schwarze. 2018. "Preparation of a Sepia Melanin and Poly(ethylene-alt-maleic Anhydride) Hybrid Material as an Adsorbent for Water Purification" Nanomaterials 8, no. 2: 54. https://doi.org/10.3390/nano8020054
APA StylePanzarasa, G., Osypova, A., Consolati, G., Quasso, F., Soliveri, G., Ribera, J., & Schwarze, F. W. M. R. (2018). Preparation of a Sepia Melanin and Poly(ethylene-alt-maleic Anhydride) Hybrid Material as an Adsorbent for Water Purification. Nanomaterials, 8(2), 54. https://doi.org/10.3390/nano8020054