pH-Sensitive Poly(β-amino ester)s Nanocarriers Facilitate the Inhibition of Drug Resistance in Breast Cancer Cells
Abstract
:1. Introduction
2. Materials and Methods
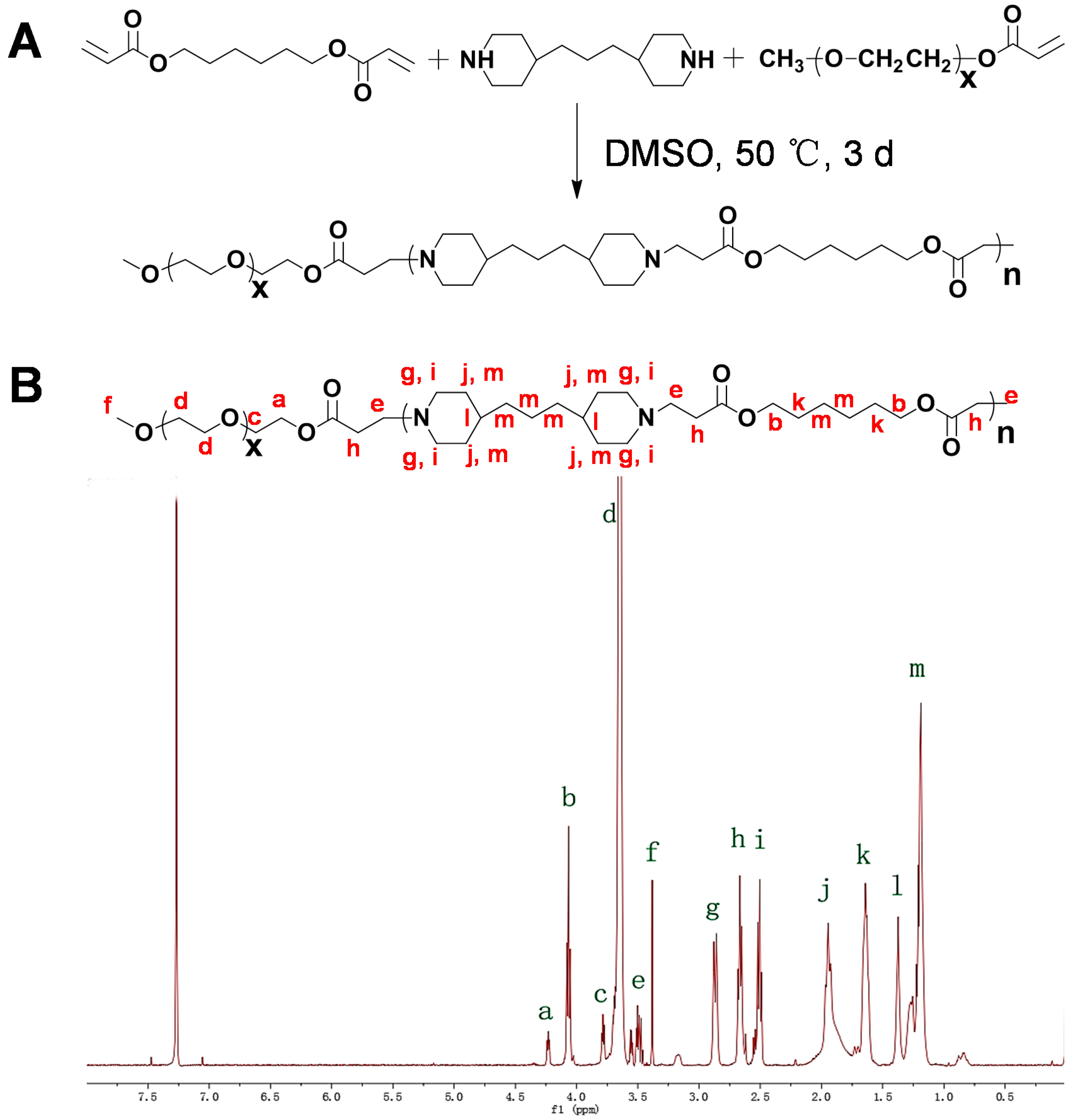
2.1. Synthesis and Characterization of PHP Copolymer
2.2. Preparation and Characterization of Micelles
2.3. Preparation and Characterization of DOX-Loaded Copolymer Micelles
2.4. pH-Sensitive Release of DOX from Micelles
2.5. Cytotoxicity Assays
2.6. Cellular Uptake and Intracellular Translocation of Nanoparticles
2.7. Rhodamine 123 (Rh123) Accumulation and Efflux
2.8. Mitochondrial Membrane Potential
2.9. Intracellular ATP Level Assay
2.10. Lactate Dehydrogenase (LDH) Assay
2.11. Statistics
3. Results
3.1. Synthesis of PHP Copolymer
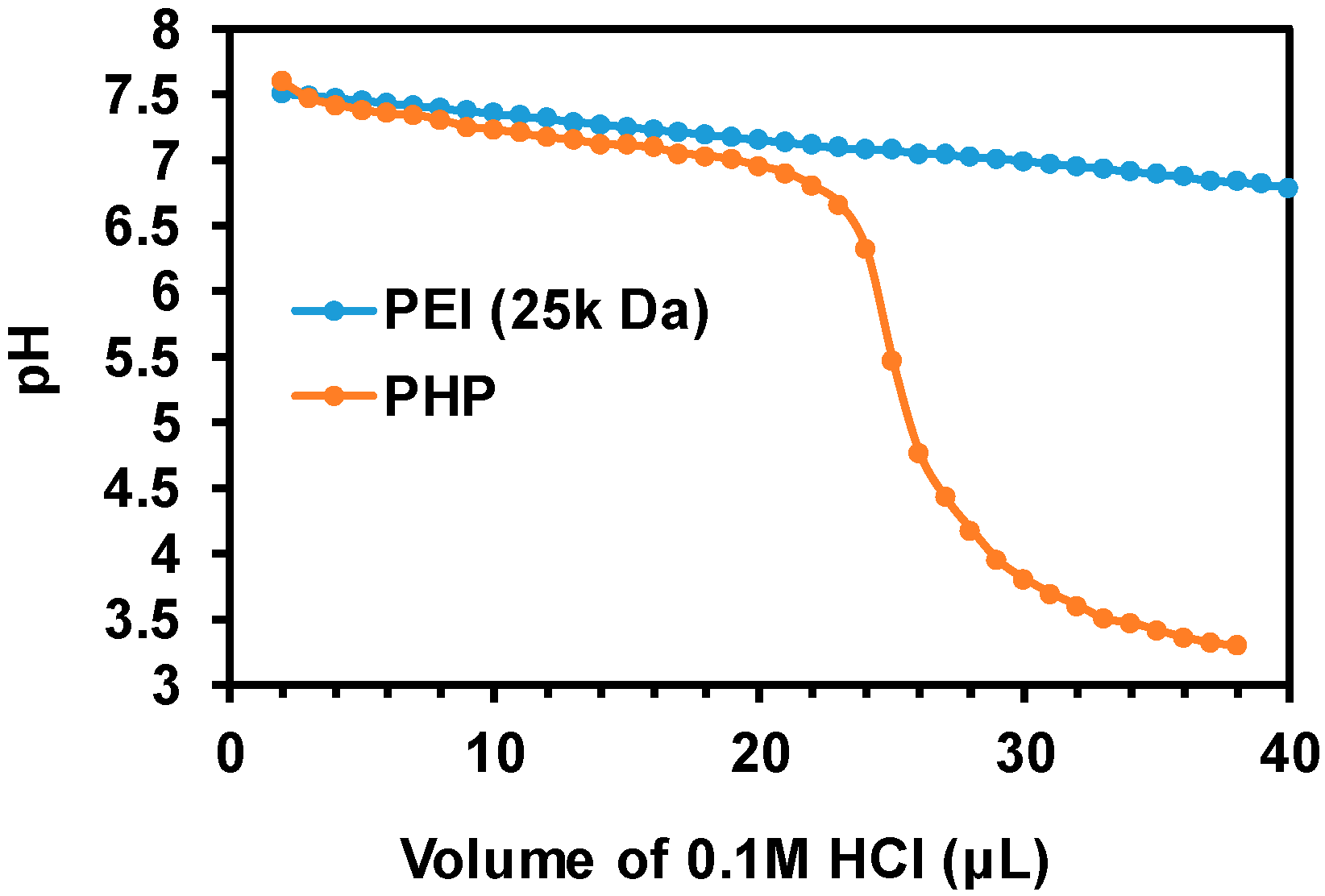
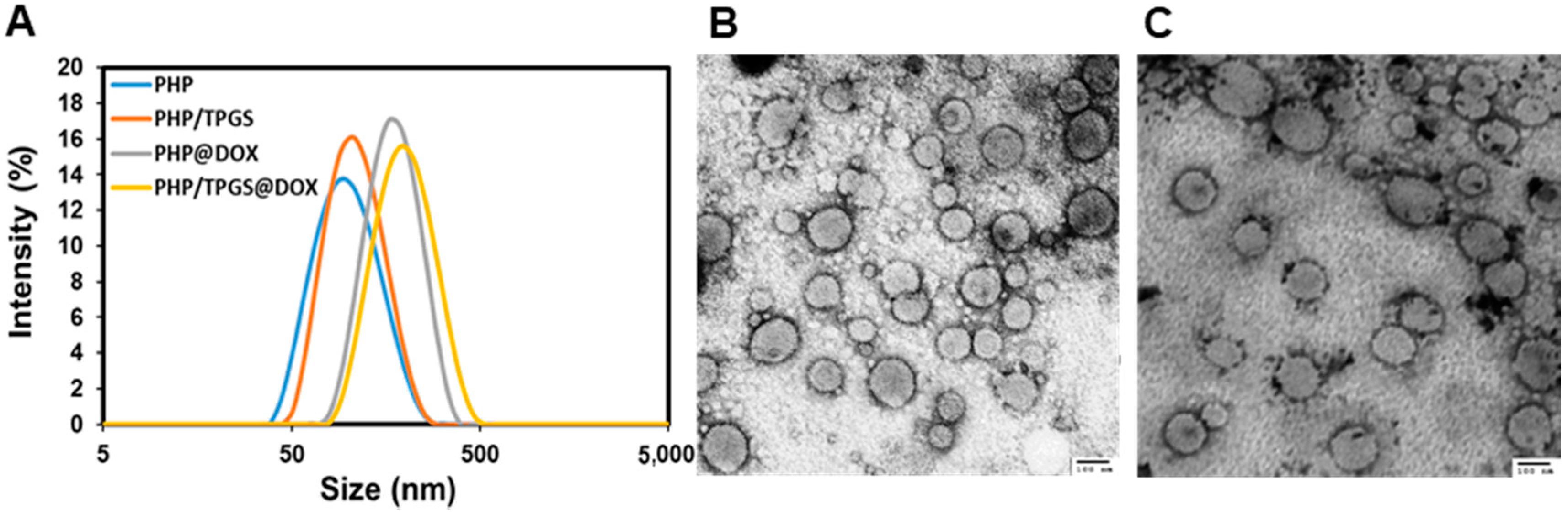
3.2. Characterization of PHP Micelles
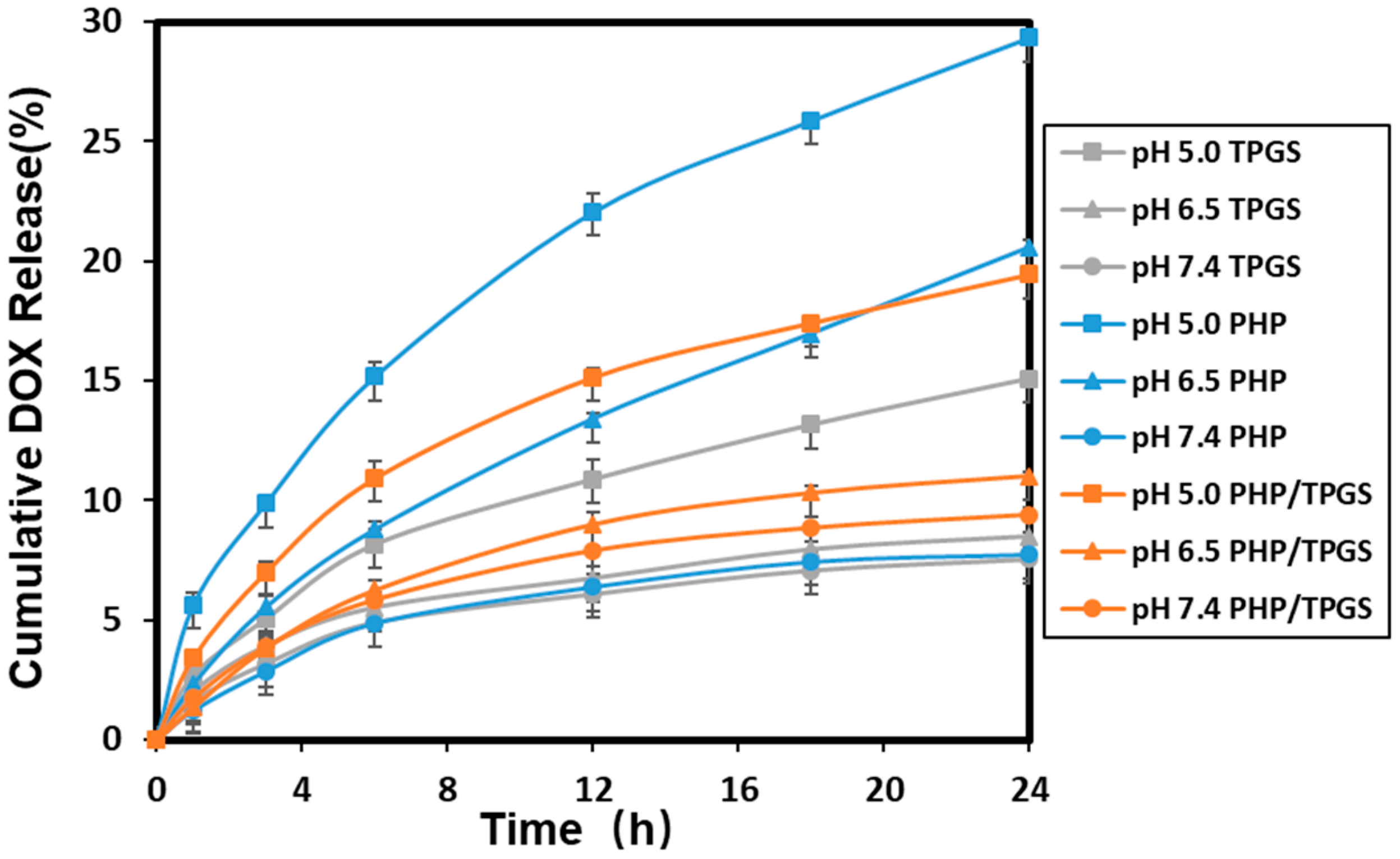
3.3. DOX Release Profiles from the PHP Micelles, TPGS and PHP/TPGS Amphiphilic Copolymer Micelles
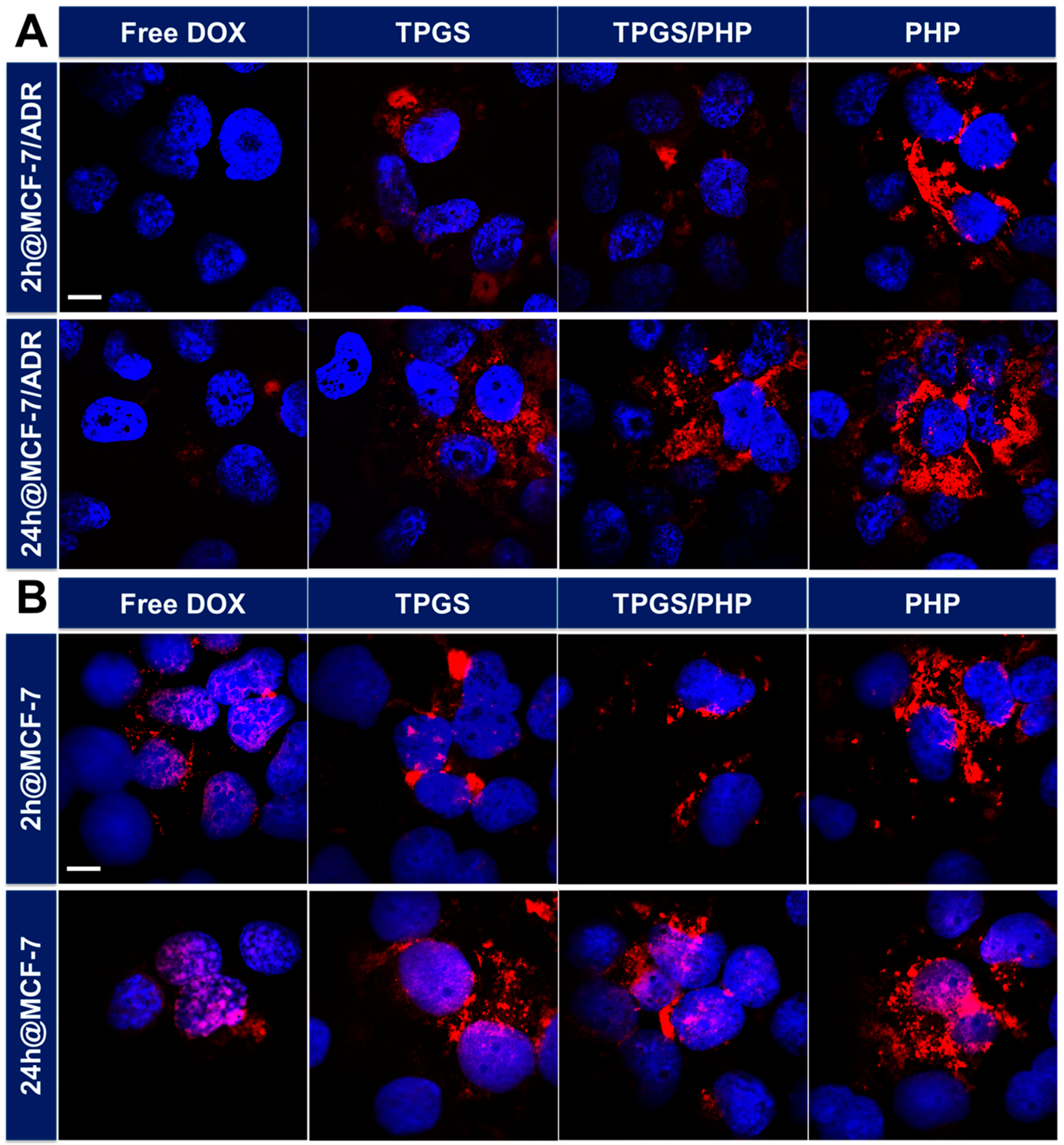
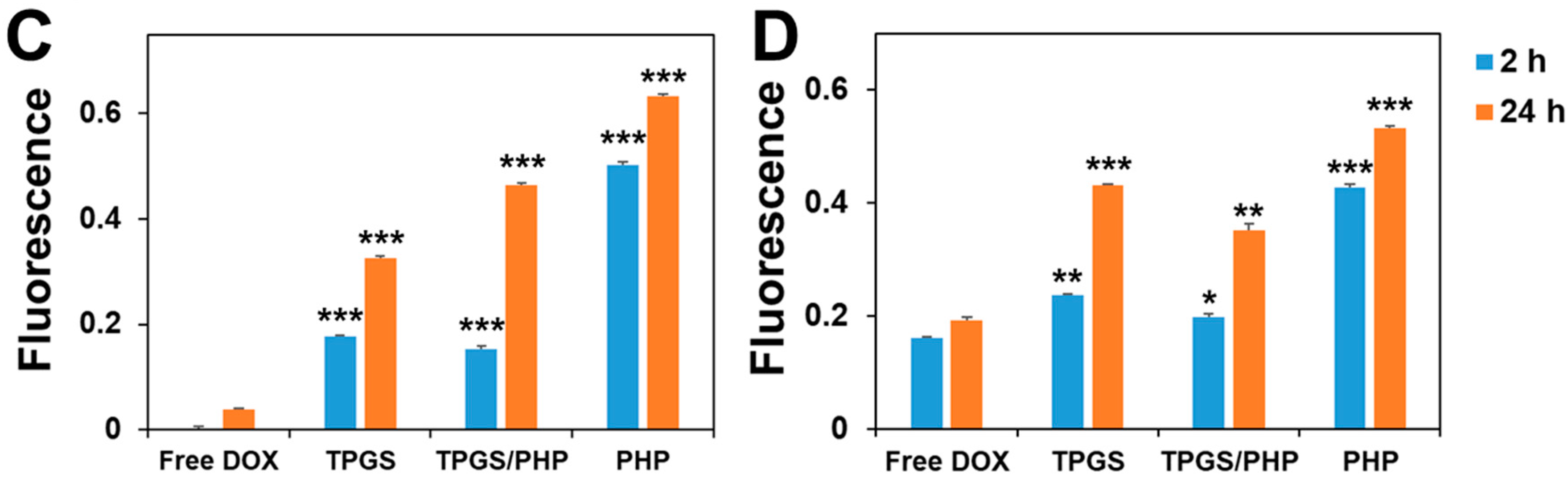
3.4. Cellular Uptake and Intracellular Distribution of DOX
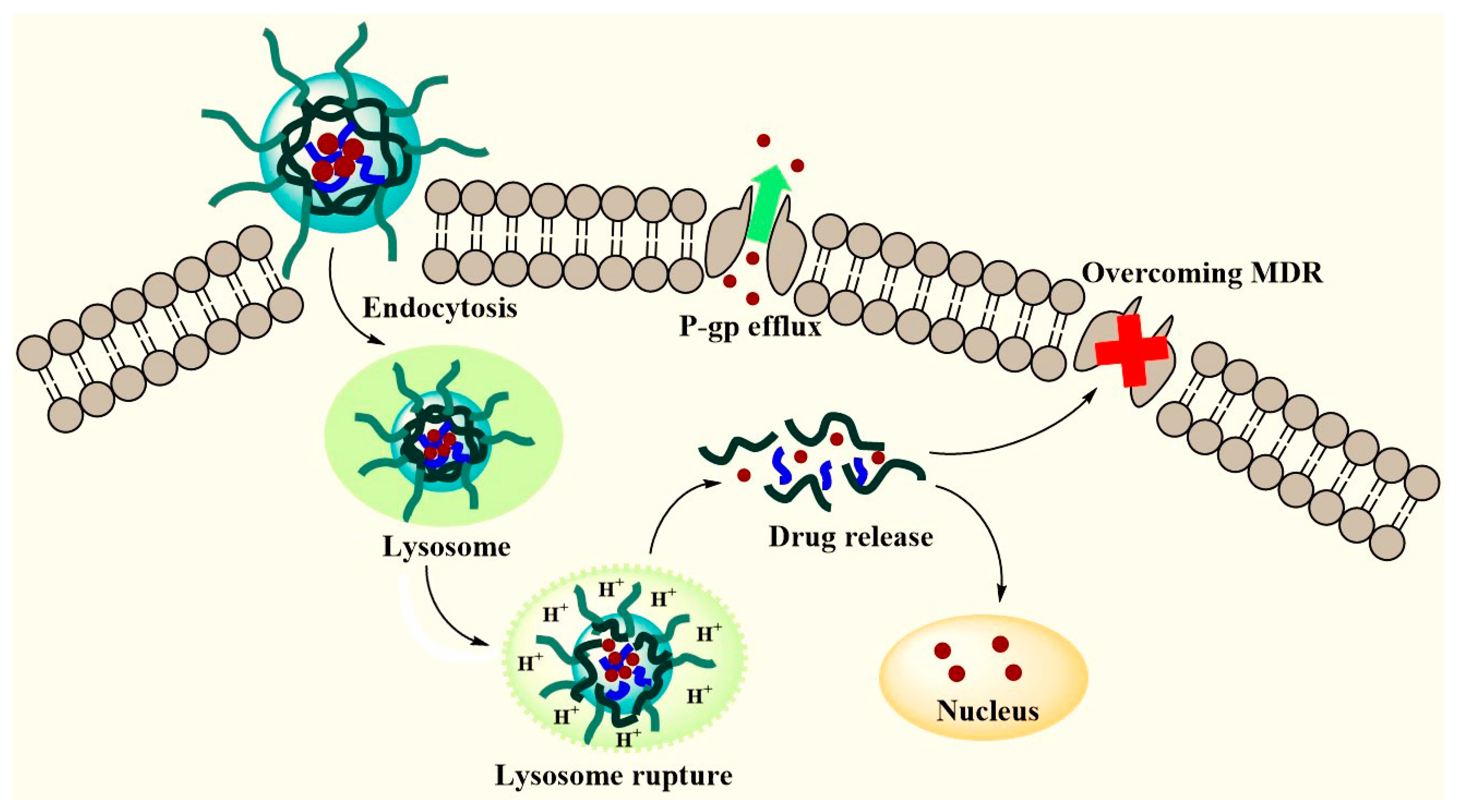
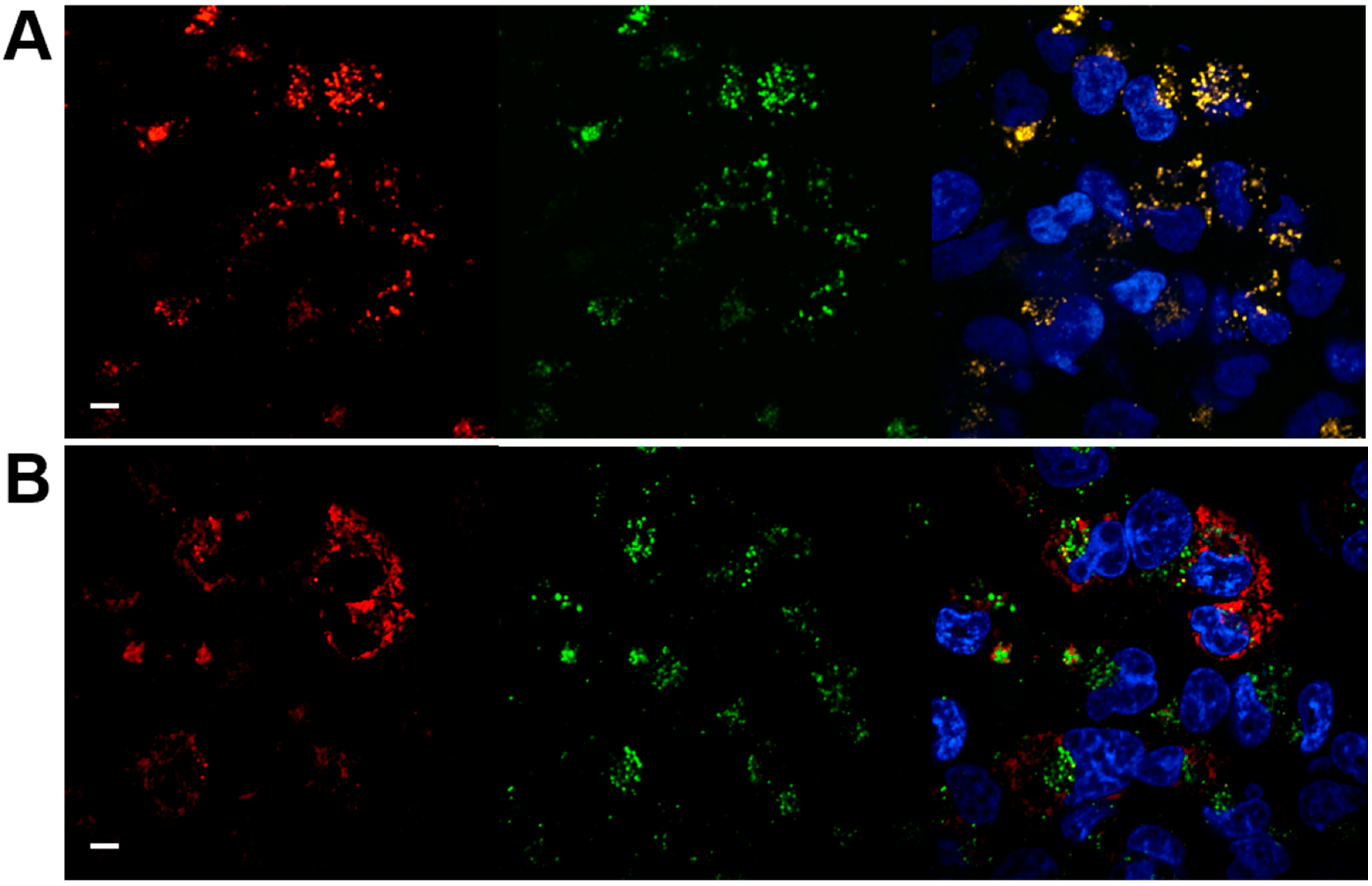
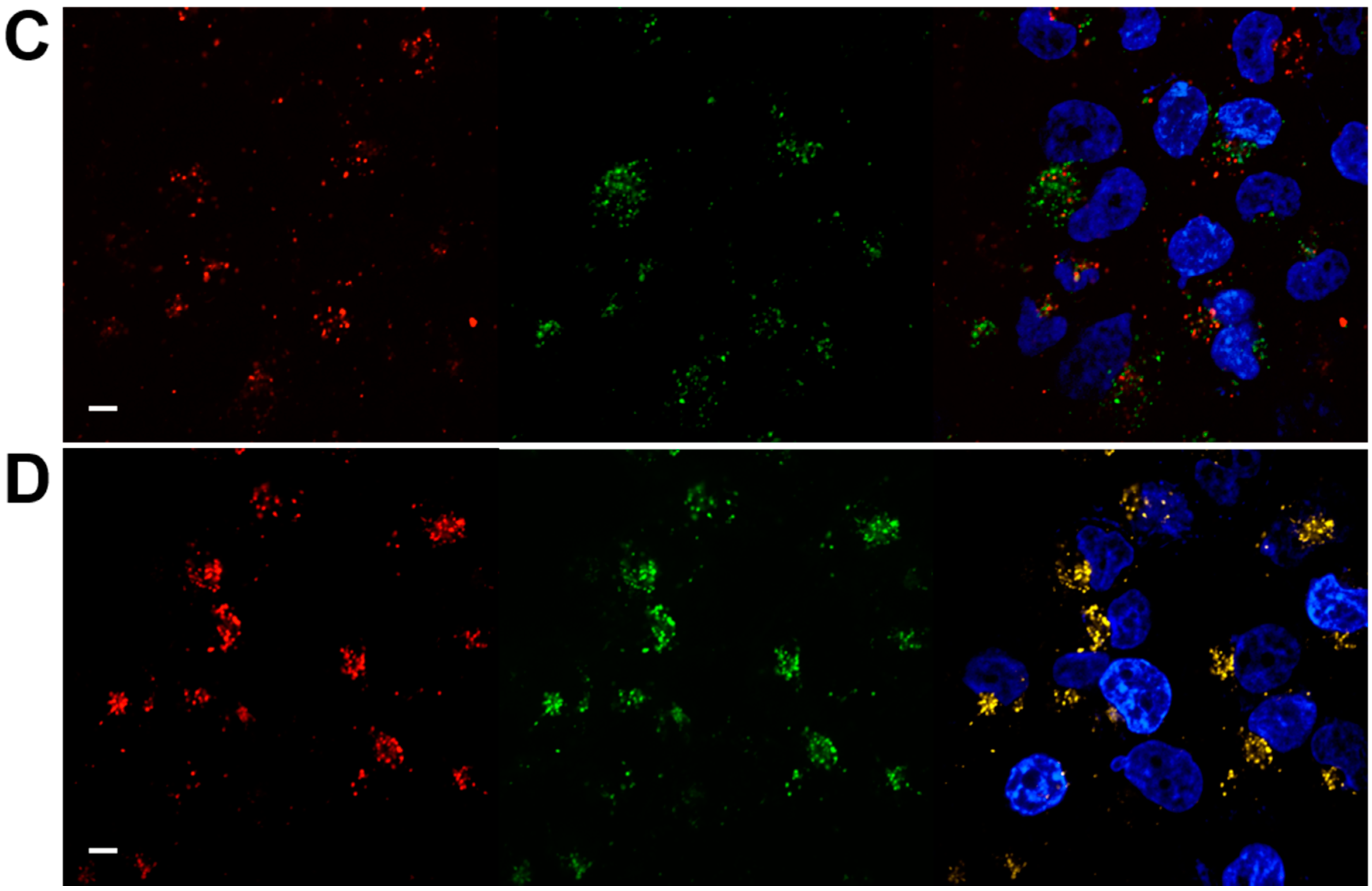
3.5. Intracellular Trafficking of Nanoparticles
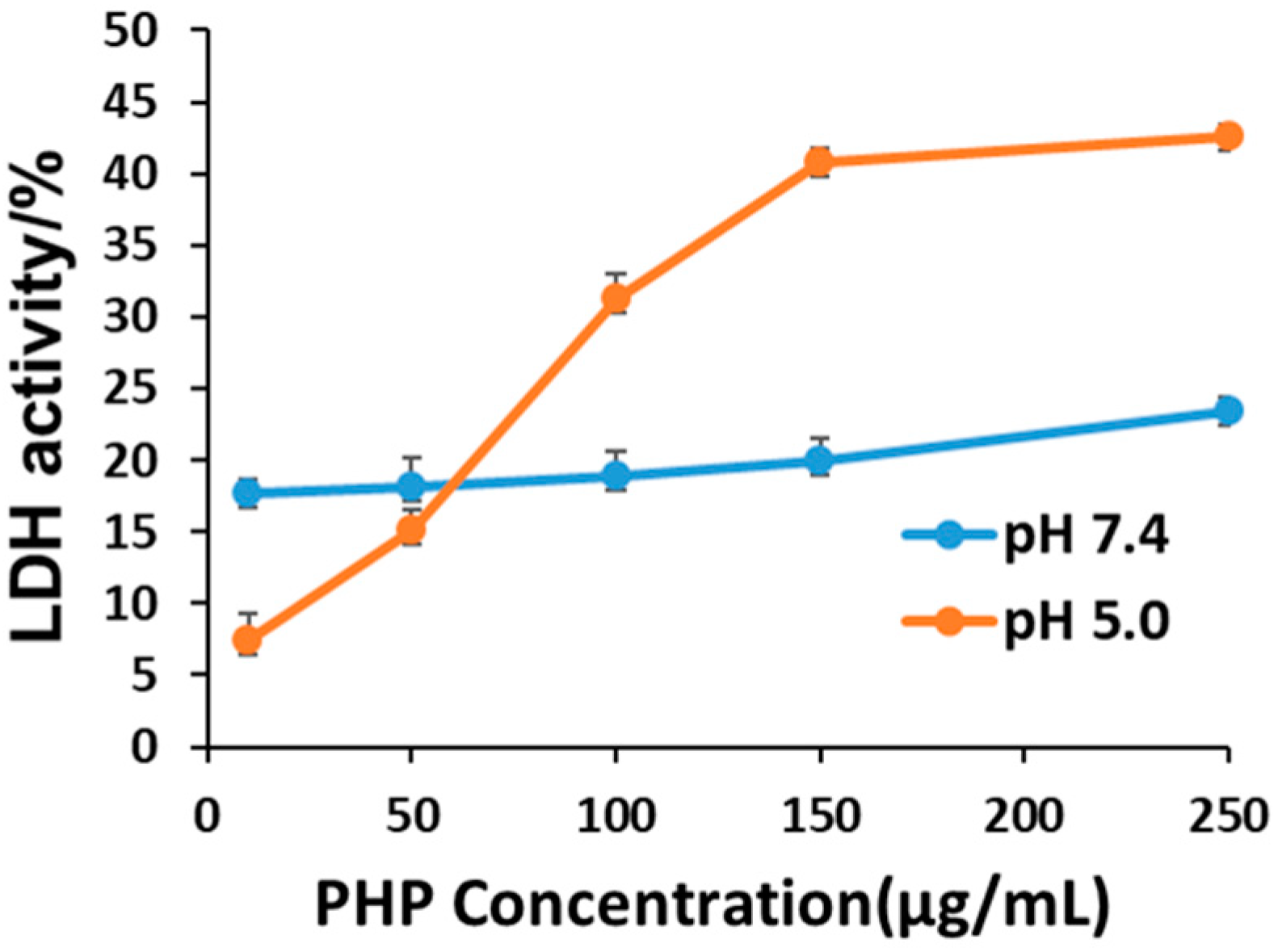
3.6. Membrane Destabilization by PHP Copolymers
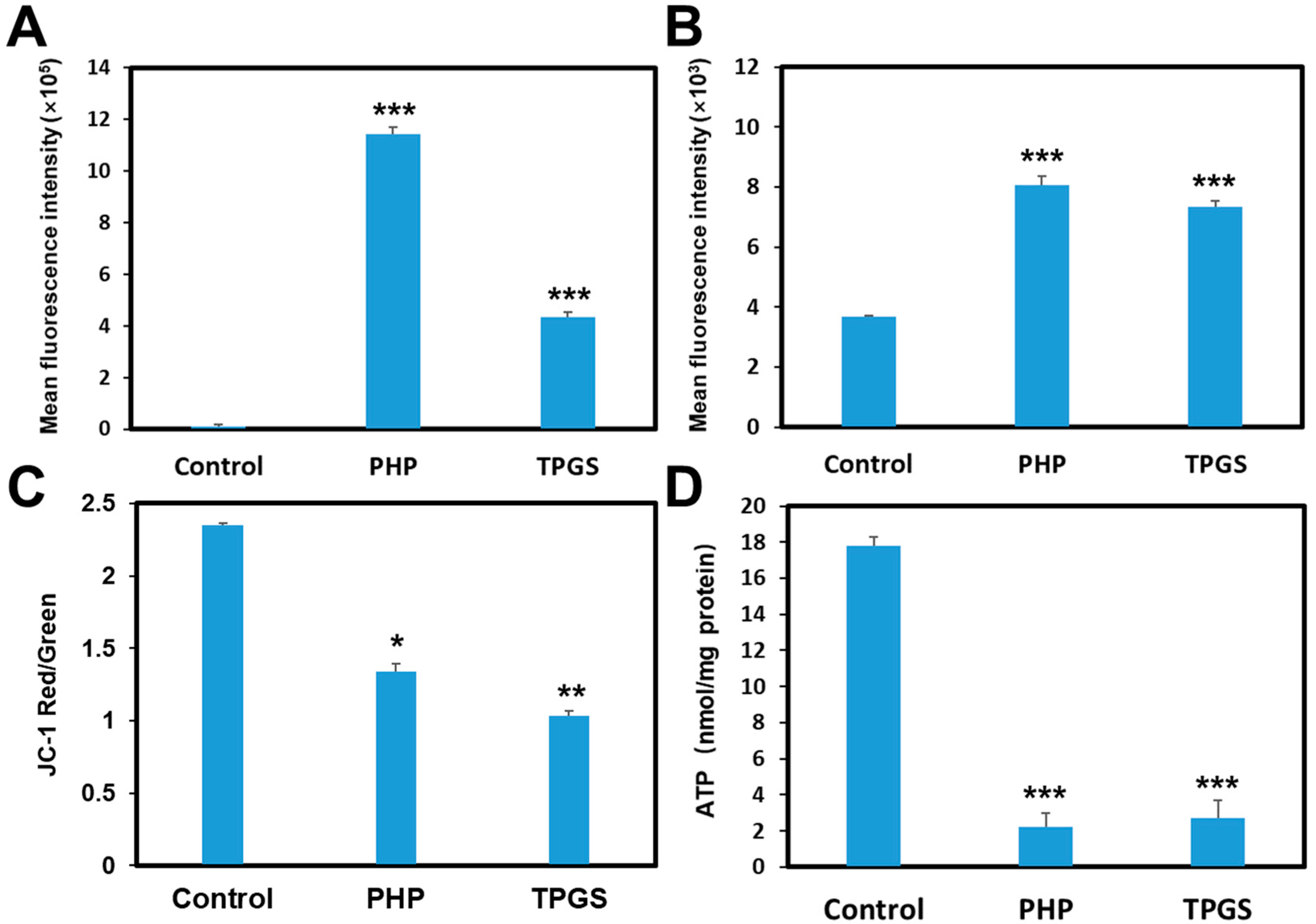
3.7. Inhibition of P-gp Efflux
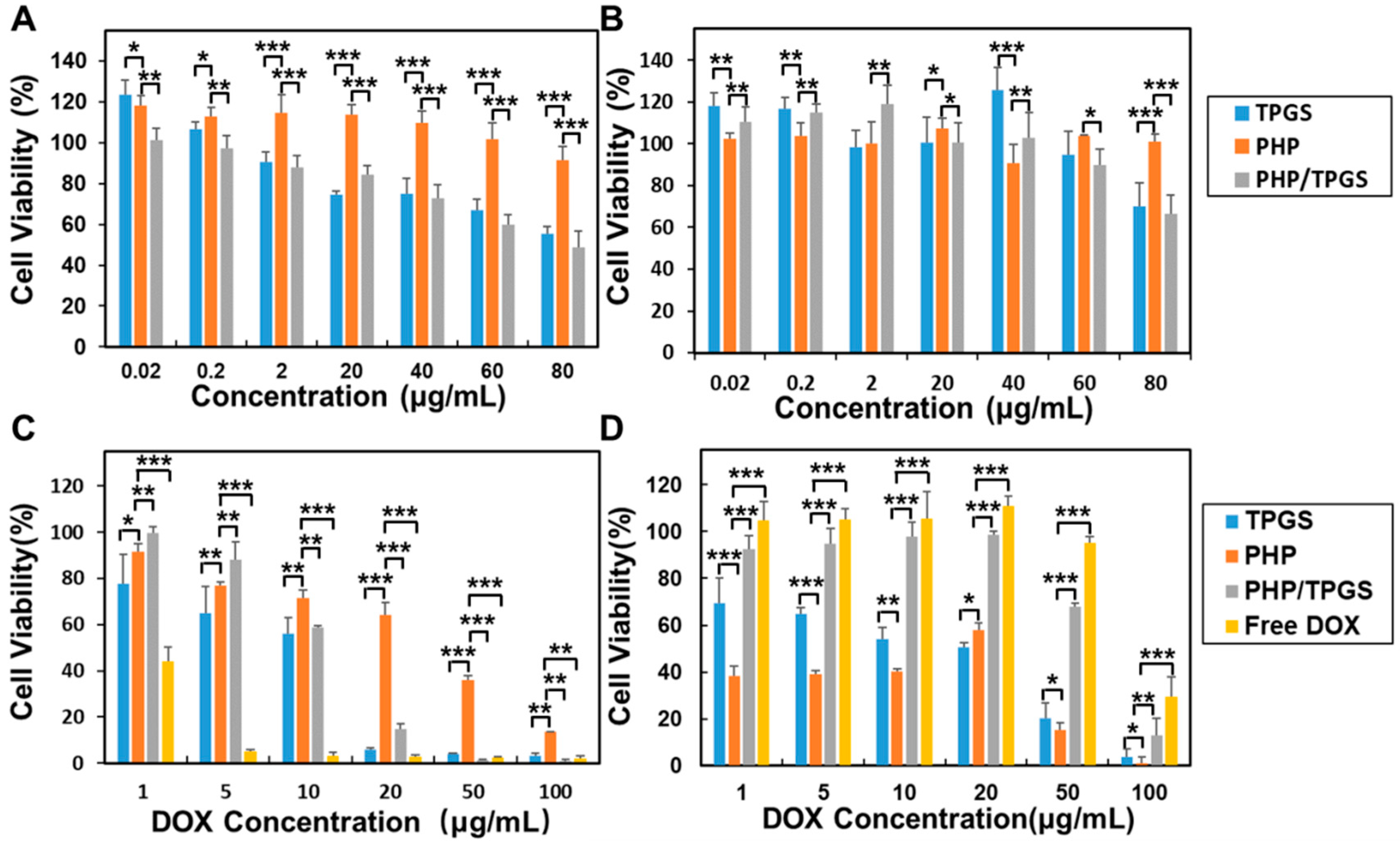
3.8. Cytotoxicity of DOX-Loaded PHP Micelles
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Yang, M.Y.; Yu, L.X.; Guo, R.W.; Dong, A.J.; Lin, C.G.; Zhang, J.H. A Modular Coassembly Approach to All-in-One Multifunctional Nanoplatform for Synergistic Codelivery of Doxorubicin and Curcumin. Nanomaterials 2018, 8, 167. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.; Xie, P.L.; Luo, M.M.; Li, Q.; Li, L.; Zhang, J.Z.; Zheng, Q.X.; Chen, H.; Nan, K.H. Efficiency against Multidrug Resistance by Co-Delivery of Doxorubicin and Curcumin with a Legumain-Sensitive Nanocarrier. Nano Res. 2018, 11, 3619–3635. [Google Scholar] [CrossRef]
- Wu, M.; Lin, X.Y.; Tan, X.H.; Li, J.; Wei, Z.W.; Zhang, D.; Zheng, Y.S.; Zheng, A.X.; Zhao, B.X.; Zeng, Y.Y.; et al. Photoresponsive Nanovehicle for Two Independent Wavelength Light-Triggered Sequential Release of P-Gp Shrna and Doxorubicin to Optimize and Enhance Synergistic Therapy of Multidrug-Resistant Cancer. ACS Appl. Mater. Interfaces 2018, 10, 19416–19427. [Google Scholar] [CrossRef] [PubMed]
- Robey, R.W.; Pluchino, K.M.; Hall, M.D.; Fojo, A.T.; Bates, S.E.; Gottesman, M.M. Revisiting the Role of Abc Transporters in Multidrug-Resistant Cancer. Nat. Rev. Cancer 2018, 18, 452–464. [Google Scholar] [CrossRef] [PubMed]
- Don, T.M.; Lu, K.Y.; Lin, L.J.; Hsu, C.H.; Wu, J.Y.; Mi, F.L. Temperature/Ph/Enzyme Triple-Responsive Cationic Protein/Paa-B-Pnipaam Nanogels for Controlled Anticancer Drug and Photosensitizer Delivery against Multidrug Resistant Breast Cancer Cells. Mol. Pharm. 2017, 14, 4648–4660. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.Y.; He, W.X.; Yang, S.F.; Zhao, D.J.; Li, Z.H.; Luan, Y.X. Co-Delivery of Docetaxel and Verapamil by Reduction-Sensitive Peg-Plga-Ss-Dtx Conjugate Micelles to Reverse the Multi-Drug Resistance of Breast Cancer. Colloid Surf. B-Biointerfaces 2017, 151, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Suo, X.B.; Eldridge, B.N.; Zhang, H.; Mao, C.Q.; Min, Y.Z.; Sun, Y.; Singh, R.; Ming, X. P-Glycoprotein-Targeted Photothermal Therapy of Drug-Resistant Cancer Cells Using Antibody-Conjugated Carbon Nanotubes. ACS Appl. Mater. Interfaces 2018, 10, 33464–33473. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.X.; Wen, K.K.; Bi, Y.; Lu, H.R.; Chen, J.; Hu, Y.; Chai, Z.F. The Application of Stimuli-Responsive Nanocarriers for Targeted Drug Delivery. Curr. Top. Med. Chem. 2017, 17, 2319–2334. [Google Scholar] [CrossRef] [PubMed]
- Mura, S.; Nicolas, J.; Couvreur, P. Stimuli-Responsive Nanocarriers for Drug Delivery. Nat. Mater. 2013, 12, 991–1003. [Google Scholar] [CrossRef] [PubMed]
- Meng, F.H.; Zhong, Y.A.; Cheng, R.; Deng, C.; Zhong, Z.Y. Ph-Sensitive Polymeric Nanoparticles for Tumor-Targeting Doxorubicin Delivery: Concept and Recent Advances. Nanomedicine 2014, 9, 487–499. [Google Scholar] [CrossRef] [PubMed]
- Kydd, J.; Jadia, R.; Velpurisiva, P.; Gad, A.; Paliwal, S.; Rai, P. Targeting Strategies for the Combination Treatment of Cancer Using Drug Delivery Systems. Pharmaceutics 2017, 9, 46. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Li, X.; Gao, L.; Hu, Y.; Zhong, W.; Xing, M.M. A Facile Strategy for in Situ Controlled Delivery of Doxorubicin with a Ph-Sensitive Injectable Hydrogel. Nano Life 2014, 4, 1441001. [Google Scholar] [CrossRef]
- Qin, Y.P.; Chen, J.; Bi, Y.; Xu, X.H.; Zhou, H.; Gao, J.M.; Hu, Y.; Zhao, Y.L.; Chai, Z.F. Near-Infrared Light Remote-Controlled Intracellular Anti-Cancer Drug Delivery Using Thermo/Ph Sensitive Nanovehicle. Acta Biomater. 2015, 17, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Bi, Y.; Chen, J.; Peng, L.R.; Wen, K.K.; Ji, P.; Ren, W.F.; Li, X.Q.; Zhang, N.; Gao, J.M.; et al. Near-Infrared Light-Triggered Switchable Nanoparticles for Targeted Chemo/Photothermal Cancer Therapy. ACS Appl. Mater. Interfaces 2016, 8, 15103–15112. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Bi, Y.; Wang, X.; Wang, M.; Zhou, M.X.; Lu, H.R.; Gao, J.M.; Chen, J.; Hu, Y. Near-Infrared Guided Thermal-Responsive Nanomedicine against Orthotopic Superficial Bladder Cancer. ACS Biomater. Sci. Eng. 2017, 3, 3628–3634. [Google Scholar] [CrossRef]
- Chen, G.C.; Xie, Y.S.; Peltier, R.; Lei, H.P.; Wang, P.; Chen, J.; Hu, Y.; Wang, F.; Yao, X.; Sun, H.Y. Peptide-Decorated Gold Nanoparticles as Functional Nano-Capping Agent of Mesoporous Silica Container for Targeting Drug Delivery. ACS Appl. Mater. Interfaces 2016, 8, 11204–11209. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.; Yin, Q.; Su, J.; Sun, H.; Meng, Q.; Chen, Y.; Chen, L.; Huang, Y.; Gu, W.; Xu, M.; et al. Inhibition of Metastasis and Growth of Breast Cancer by Ph-Sensitive Poly (Beta-Amino Ester) Nanoparticles Co-Delivering Two Sirna and Paclitaxel. Biomaterials 2015, 48, 1–15. [Google Scholar] [CrossRef] [PubMed]
- An, T.; Zhang, C.; Han, X.; Wan, G.Y.; Wang, D.; Yang, Z.; Wang, Y.; Zhang, L.Y.; Wang, Y.S. Hyaluronic Acid-Coated Poly(Beta-Amino) Ester Nanoparticles as Carrier of Doxorubicin for Overcoming Drug Resistance in Breast Cancer Cells. RSC Adv. 2016, 6, 38624–38636. [Google Scholar] [CrossRef]
- Chen, Y.J.; Yue, Q.X.; De, G.J.; Wang, J.; Li, Z.Z.; Xiao, S.M.; Yu, H.T.; Ma, H.; Sui, F.; Zhao, Q.H. Inhibition of Breast Cancer Metastasis by Paclitaxel-Loaded Ph Responsive Poly(Beta-Amino Ester) Copolymer Micelles. Nanomedicine 2017, 12, 147–164. [Google Scholar] [CrossRef] [PubMed]
- Song, W.; Tang, Z.; Li, M.; Lv, S.; Yu, H.; Ma, L.; Zhuang, X.; Huang, Y.; Chen, X. Tunable Ph-Sensitive Poly(Beta-Amino Ester)S Synthesized from Primary Amines and Diacrylates for Intracellular Drug Delivery. Macromol. Biosci. 2012, 12, 1375–1383. [Google Scholar] [CrossRef] [PubMed]
- Ottemann, B.M.; Helmink, A.J.; Zhang, W.; Mukadam, I.; Woldstad, C.; Hilaire, J.R.; Liu, Y.; McMillan, J.M.; Edagwa, B.J.; Mosley, R.L.; et al. Bioimaging Predictors of Rilpivirine Biodistribution and Antiretroviral Activities. Biomaterials 2018, 185, 174–193. [Google Scholar] [CrossRef] [PubMed]
- Kopecka, J.; Campia, I.; Olivero, P.; Pescarmona, G.; Ghigo, D.; Bosia, A.; Riganti, C. A Ldl-Masked Liposomal-Doxorubicin Reverses Drug Resistance in Human Cancer Cells. J. Control. Release 2011, 149, 196–205. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Hao, J.; Li, Y.; Zhang, Z.; Sha, X.; Han, L.; Fang, X. Poly(Caprolactone)-Modified Pluronic P105 Micelles for Reversal of Paclitaxcel-Resistance in Skov-3 Tumors. Biomaterials 2012, 33, 4741–4751. [Google Scholar] [CrossRef] [PubMed]
- Han, M.; Diao, Y.Y.; Jiang, H.L.; Ying, X.Y.; Chen, D.W.; Liang, W.Q.; Gao, J.Q. Molecular Mechanism Study of Chemosensitization of Doxorubicin-Resistant Human Myelogenous Leukemia Cells Induced by a Composite Polymer Micelle. Int. J. Pharm. 2011, 420, 404–411. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Qiu, X.; Ouyang, J.; Kong, J.; Zhong, W.; Xing, M.M. Ph and Reduction Dual-Sensitive Copolymeric Micelles for Intracellular Doxorubicin Delivery. Biomacromolecules 2011, 12, 3601–3611. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Huang, S.W.; Liu, M.; Zhuo, R.X. Synthesis and Degradation of Poly(Beta-Aminoester) with Pendant Primary Amine. Polymer 2007, 48, 675–681. [Google Scholar] [CrossRef]
- Ke, W.; Yu, P.; Wang, J.; Wang, R.; Guo, C.; Zhou, L.; Li, C.; Li, K. Mcf-7/Adr Cells (Re-Designated Nci/Adr-Res) Are Not Derived from Mcf-7 Breast Cancer Cells: A Loss for Breast Cancer Multidrug-Resistant Research. Med. Oncol. 2011, 28 (Suppl. 1), S135–S141. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, H.P.; Xie, J.; Zhou, M.X.; Huang, H.; Lu, H.R.; Chai, Z.F.; Chen, J.; Hu, Y. Facile Construction of Mitochondria-Targeting Nanoparticles for Enhanced Phototherapeutic Effects. Biomater. Sci. 2017, 5, 1022–1031. [Google Scholar] [CrossRef] [PubMed]
- Mozhi, A.; Ahmad, I.; Kaleem, Q.M.; Tuguntaev, R.G.; Eltahan, A.S.; Wang, C.; Yang, R.; Li, C.; Liang, X.J. Nrp-1 Receptor Targeting Peptide-Functionalized Tpgs Micellar Nanosystems to Deliver 10-Hydroxycampothecin for Enhanced Cancer Chemotherapy. Int. J. Pharm. 2018, 547, 582–592. [Google Scholar] [CrossRef] [PubMed]
- Mi, Y.; Liu, Y.; Feng, S.S. Formulation of Docetaxel by Folic Acid-Conjugated D-Alpha-Tocopheryl Polyethylene Glycol Succinate 2000 (Vitamin E Tpgs(2k)) Micelles for Targeted and Synergistic Chemotherapy. Biomaterials 2011, 32, 4058–4066. [Google Scholar] [CrossRef] [PubMed]
- Alakhova, D.Y.; Kabanov, A.V. Pluronics and Mdr Reversal: An Update. Mol. Pharm. 2014, 11, 2566–2578. [Google Scholar] [CrossRef] [PubMed]
- Cuong Viet, P.; Cho, C.-W. Application of D-Alpha-Tocopheryl Polyethylene Glycol 1000 Succinate (Tpgs) in Transdermal and Topical Drug Delivery Systems (Tdds). J. Pharm. Investig. 2017, 47, 111–121. [Google Scholar] [CrossRef]
- Zhang, Z.; Tan, S.; Feng, S.S. Vitamin E Tpgs as a Molecular Biomaterial for Drug Delivery. Biomaterials 2012, 33, 4889–4906. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Fu, Q.; Wang, Y.; Racette, K.; Wang, D.; Liu, F. Vitamin E Reverses Multidrug Resistance in Vitro and in Vivo. Cancer Lett. 2013, 336, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Collnot, E.M.; Baldes, C.; Schaefer, U.F.; Edgar, K.J.; Wempe, M.F.; Lehr, C.M. Vitamin E Tpgs P-Glycoprotein Inhibition Mechanism: Influence on Conformational Flexibility, Intracellular Atp Levels, and Role of Time and Site of Access. Mol. Pharm. 2010, 7, 642–651. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Lee, S.H.; Gan, C.W.; Feng, S.S. In Vitro and in Vivo Investigation on Pla-Tpgs Nanoparticles for Controlled and Sustained Small Molecule Chemotherapy. Pharm. Res. 2008, 25, 1925–1935. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Chen, H.; Zheng, Y.; Song, X.; Liu, R.; Liu, K.; Zeng, X.; Mei, L. Nanoformulation of D-Alpha-Tocopheryl Polyethylene Glycol 1000 Succinate-B-Poly(Epsilon-Caprolactone-Ran-Glycolide) Diblock Copolymer for Breast Cancer Therapy. Integr. Biol. 2011, 3, 993–1002. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Sun, J.; Chen, Q.; Gao, Y.; Li, L.; Li, H.; Leng, D.; Wang, Y.; Sun, Y.; Jing, Y.; et al. Star-Shape Copolymer of Lysine-Linked Di-Tocopherol Polyethylene Glycol 2000 Succinate for Doxorubicin Delivery with Reversal of Multidrug Resistance. Biomaterials 2012, 33, 6877–6888. [Google Scholar] [CrossRef] [PubMed]
- Li, P.Y.; Lai, P.S.; Hung, W.C.; Syu, W.J. Poly(l-Lactide)-Vitamin E Tpgs Nanoparticles Enhanced the Cytotoxicity of Doxorubicin in Drug-Resistant Mcf-7 Breast Cancer Cells. Biomacromolecules 2010, 11, 2576–2582. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Tan, S.; Guo, Y.; Huang, J.; Chu, M.; Liu, H.; Zhang, Z. Ph-Sensitive Docetaxel-Loaded D-Alpha-Tocopheryl Polyethylene Glycol Succinate-Poly(Beta-Amino Ester) Copolymer Nanoparticles for Overcoming Multidrug Resistance. Biomacromolecules 2013, 14, 2636–2646. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, M.; Zhang, X.; Xie, J.; Qi, R.; Lu, H.; Leporatti, S.; Chen, J.; Hu, Y. pH-Sensitive Poly(β-amino ester)s Nanocarriers Facilitate the Inhibition of Drug Resistance in Breast Cancer Cells. Nanomaterials 2018, 8, 952. https://doi.org/10.3390/nano8110952
Zhou M, Zhang X, Xie J, Qi R, Lu H, Leporatti S, Chen J, Hu Y. pH-Sensitive Poly(β-amino ester)s Nanocarriers Facilitate the Inhibition of Drug Resistance in Breast Cancer Cells. Nanomaterials. 2018; 8(11):952. https://doi.org/10.3390/nano8110952
Chicago/Turabian StyleZhou, Mengxue, Xingcai Zhang, Jin Xie, Rongxiang Qi, Huiru Lu, Stefano Leporatti, Jun Chen, and Yi Hu. 2018. "pH-Sensitive Poly(β-amino ester)s Nanocarriers Facilitate the Inhibition of Drug Resistance in Breast Cancer Cells" Nanomaterials 8, no. 11: 952. https://doi.org/10.3390/nano8110952
APA StyleZhou, M., Zhang, X., Xie, J., Qi, R., Lu, H., Leporatti, S., Chen, J., & Hu, Y. (2018). pH-Sensitive Poly(β-amino ester)s Nanocarriers Facilitate the Inhibition of Drug Resistance in Breast Cancer Cells. Nanomaterials, 8(11), 952. https://doi.org/10.3390/nano8110952





