The Effect of Boron Nitride on the Thermal and Mechanical Properties of Poly(3-hydroxybutyrate-co-3-hydroxyvalerate)
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Surface Modification of Boron Nitride
2.3. Preparation of Nanobiocomposites
2.4. Characterization of BN Nanoparticles and Composites
2.5. Thermal Properties of PHBV/BN Nanobiocomposites
2.6. Mechanical Properties of PHBV Nanobiocomposites
3. Results and Discussion
3.1. Morphological Characterization of BN Particles by SEM
3.2. SEM of Nanocomposites
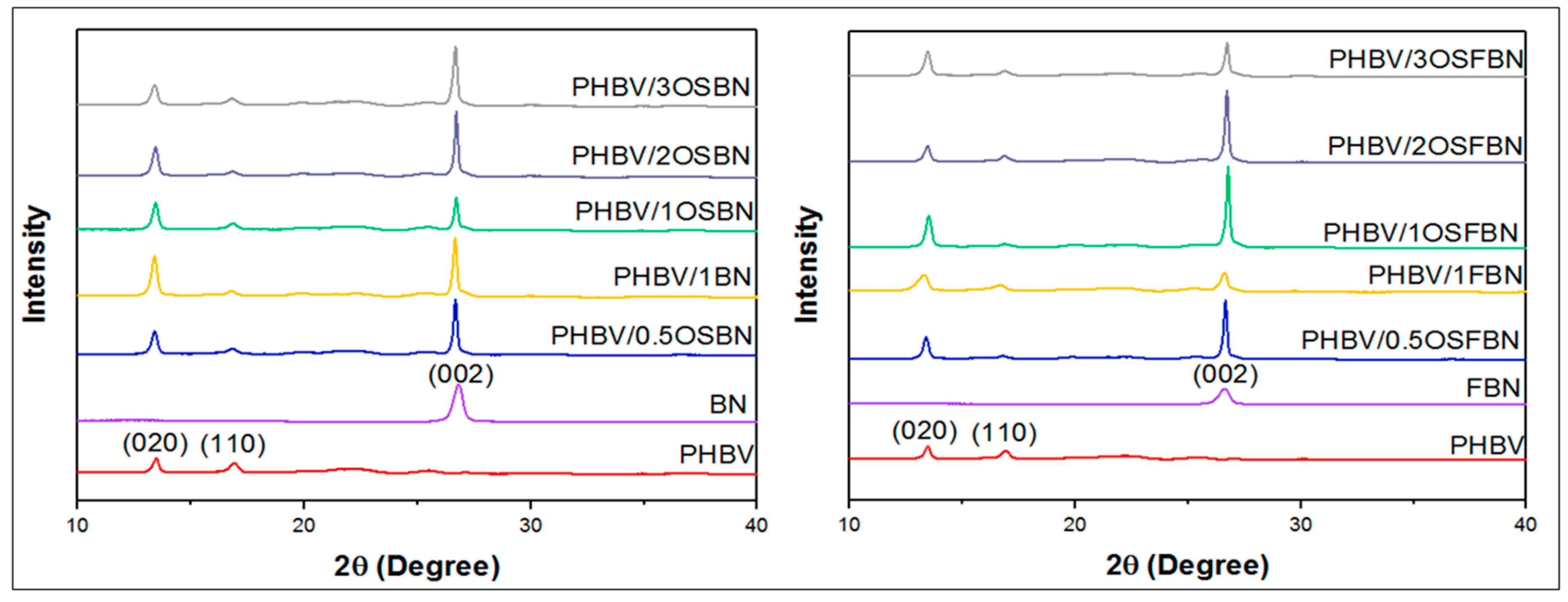
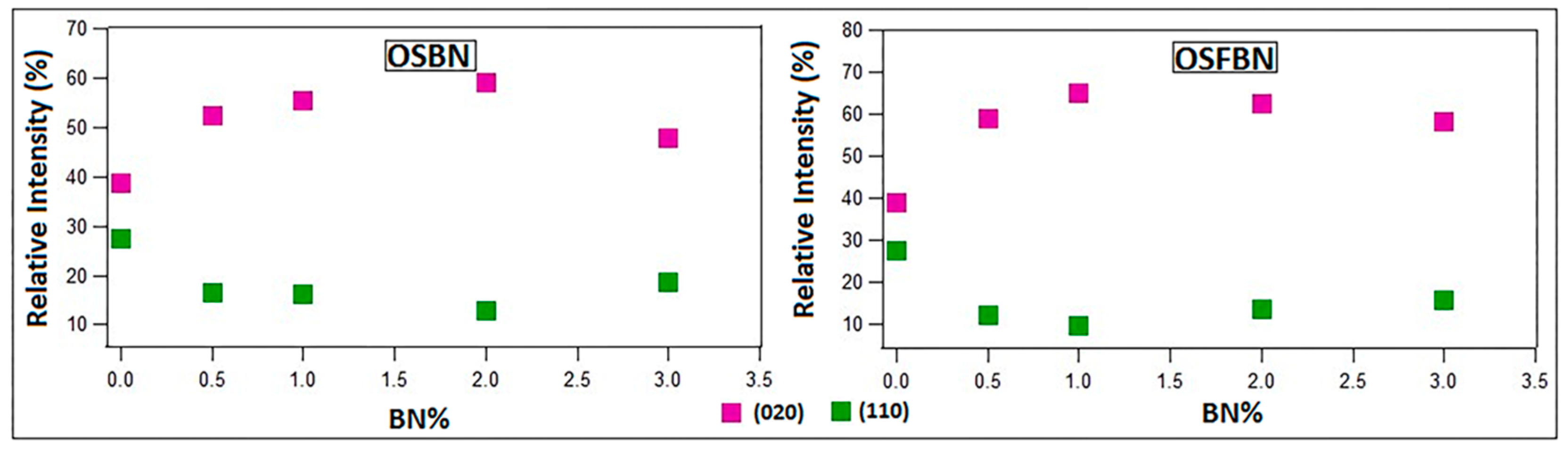
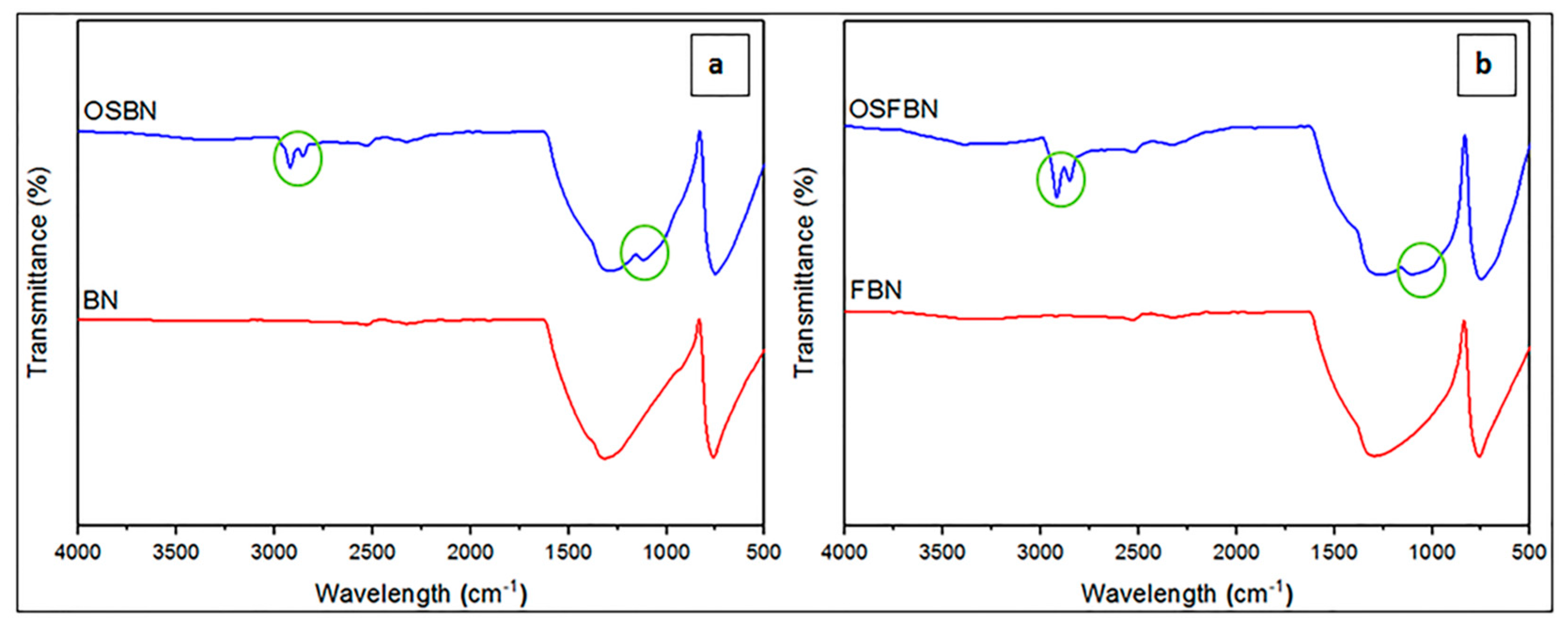
3.3. Characterization by XRD and FTIR
3.4. Thermal Stability of Nanocomposites
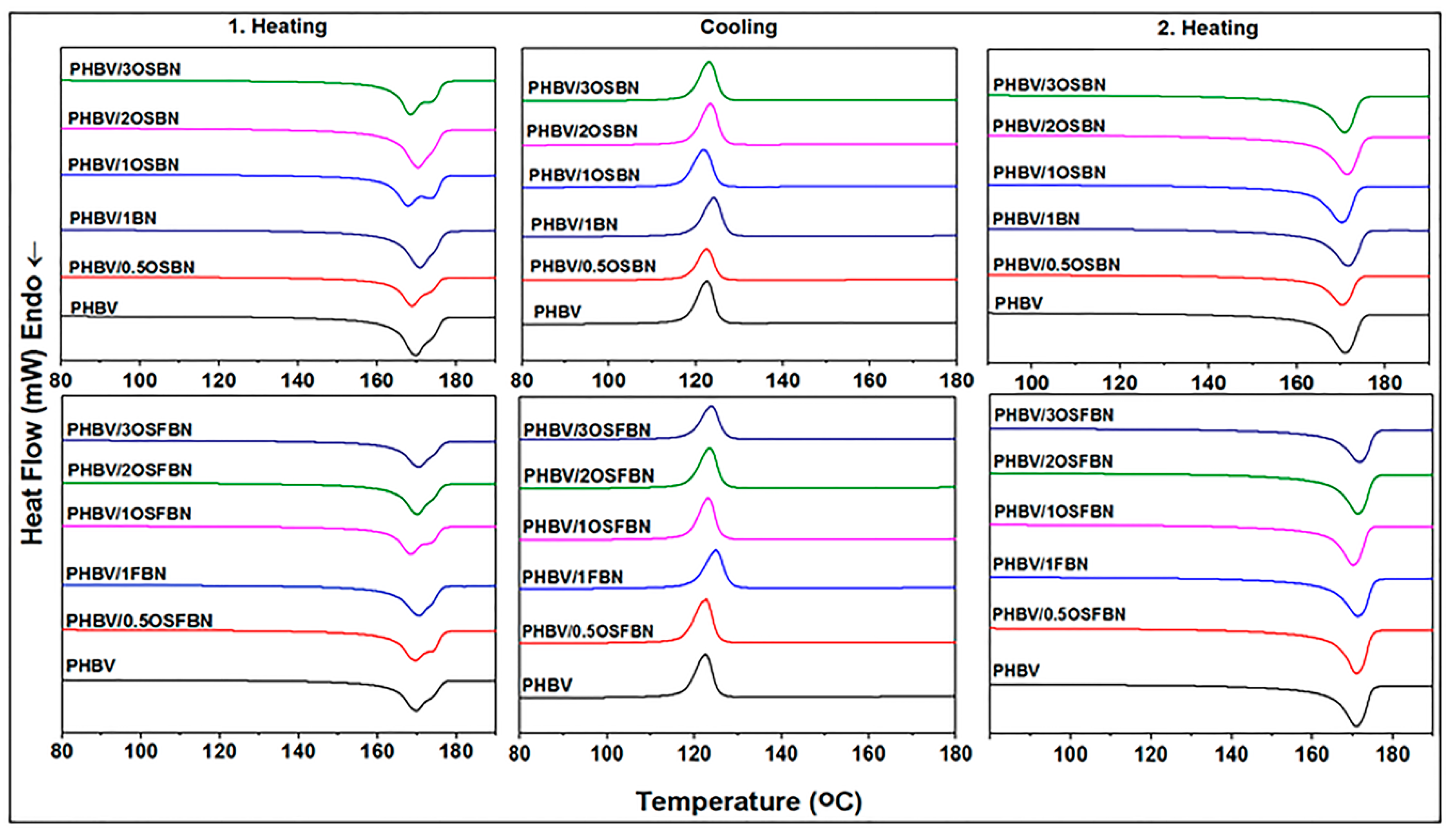
3.5. Thermal Properties of Composites
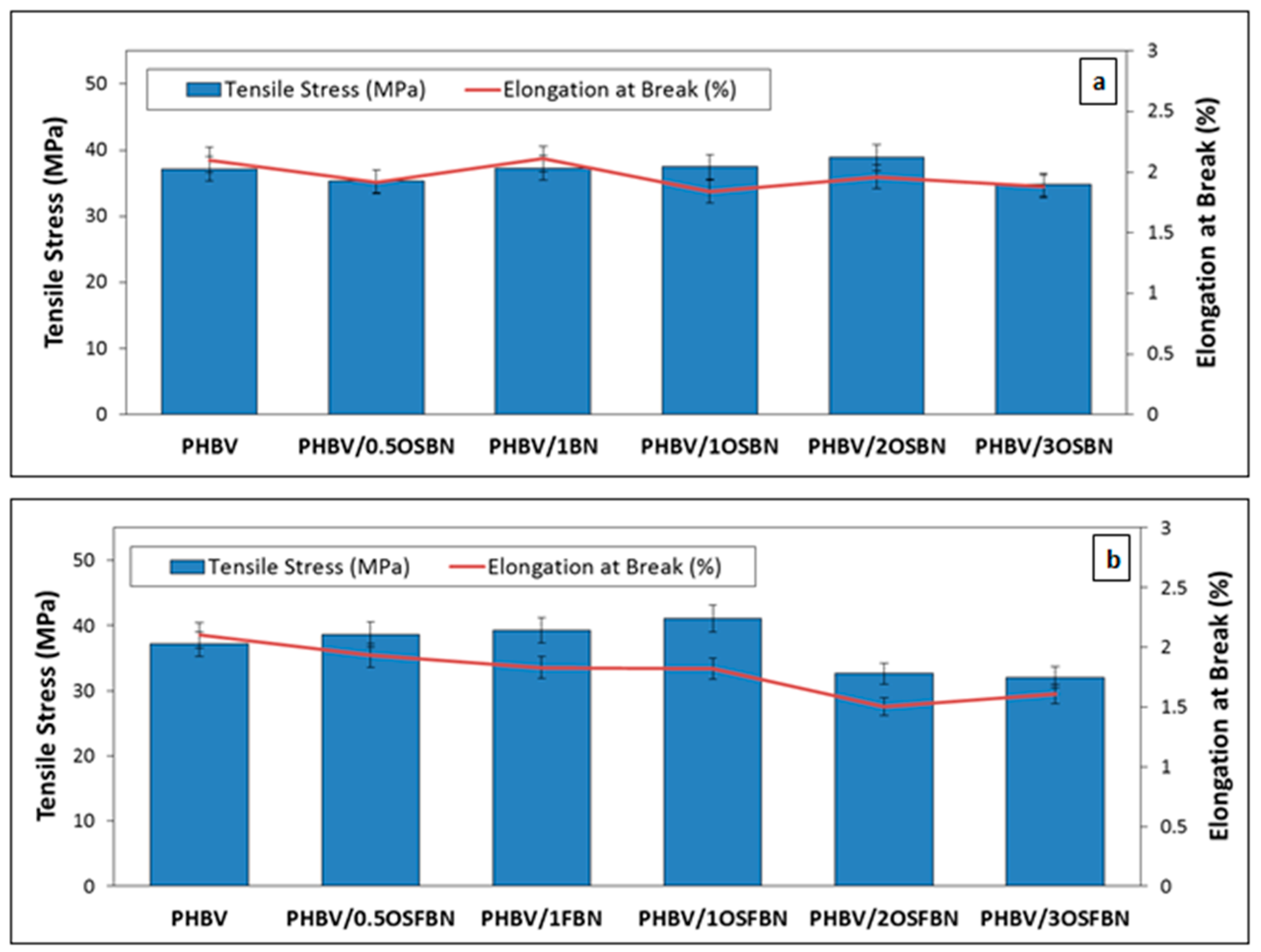
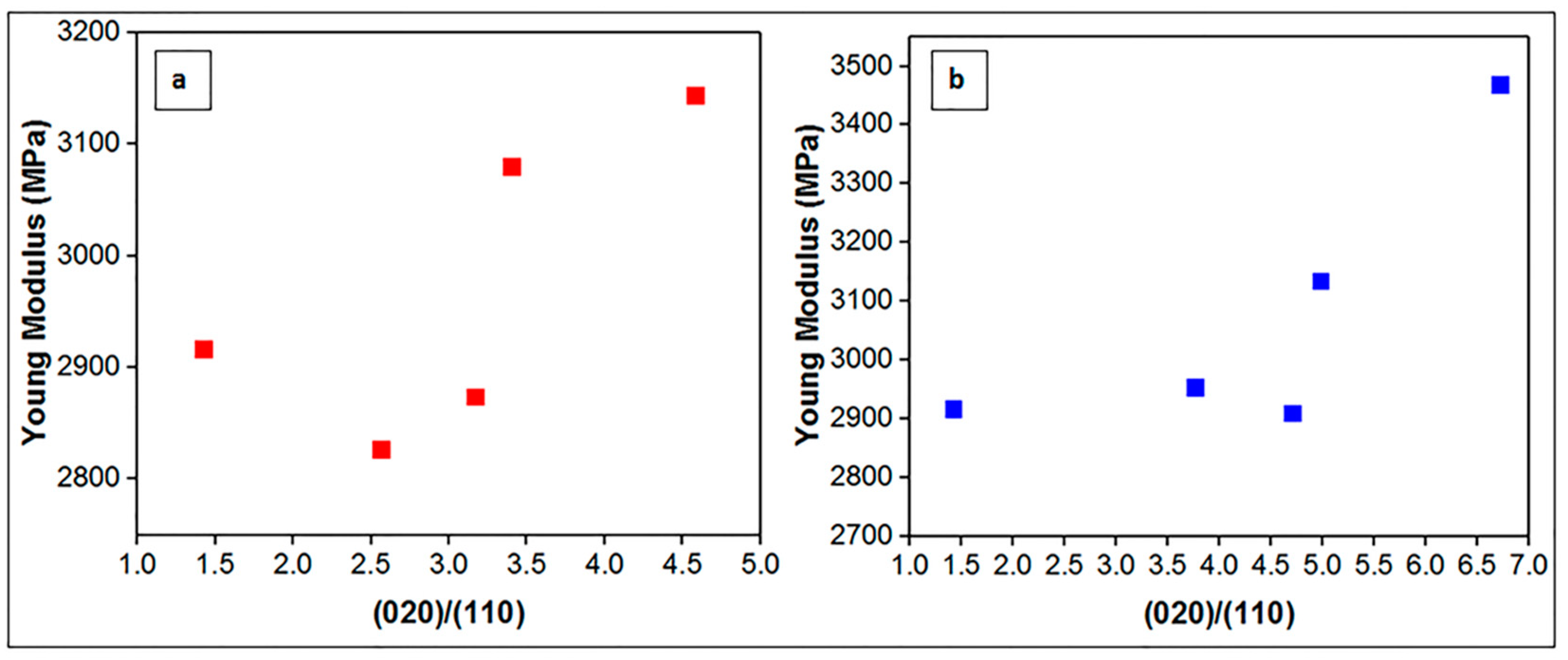
3.6. Mechanical Analysis Results
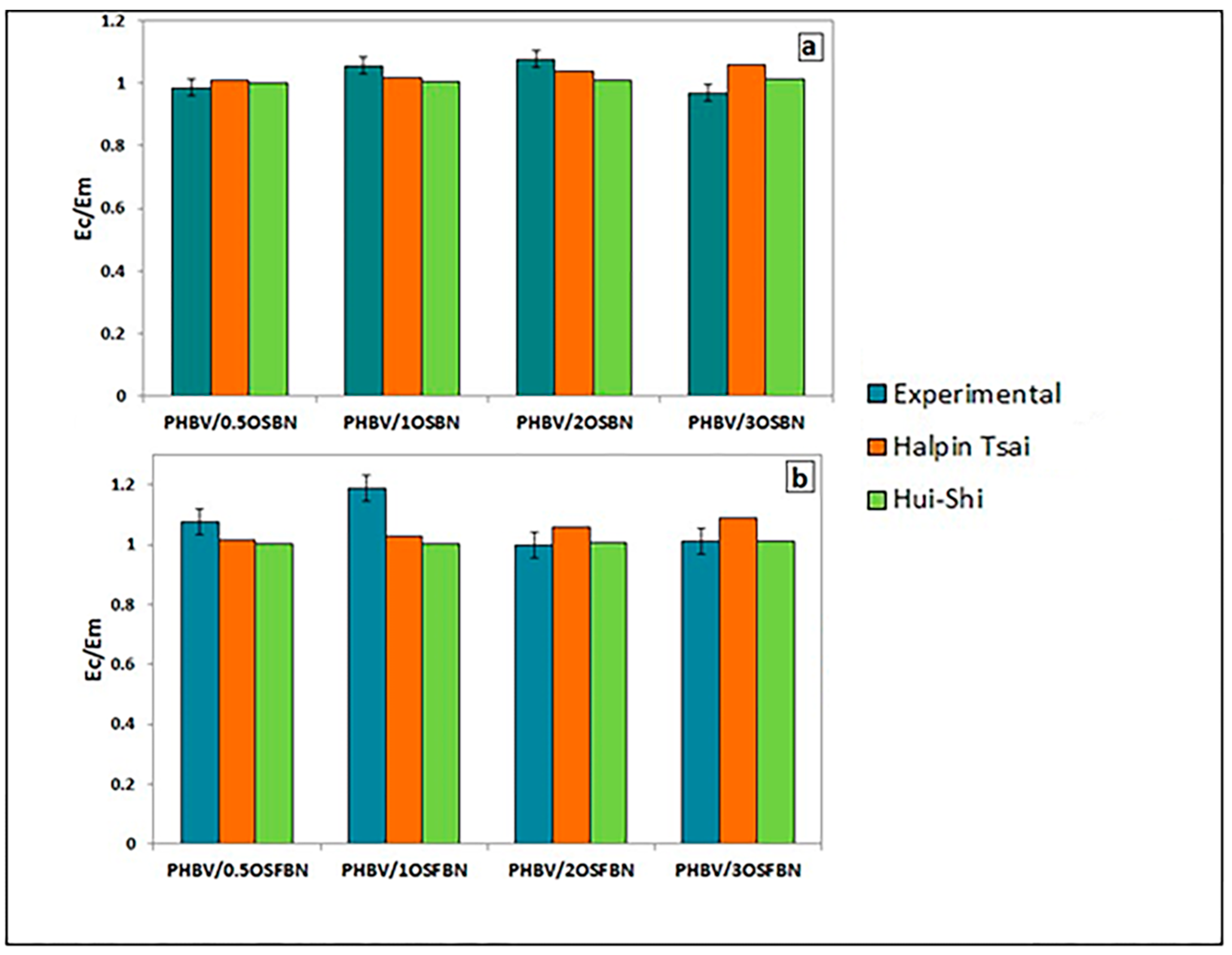
3.7. Mechanical Modelling
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Malmir, S.; Montero, B.; Rico, M.; Barral, L.; Bouza, R.; Farrag, Y. Effects of poly(3-hydroxybutyrate-co-hydroxyvalerate) microparticles on morphological, mechanical, thermal, and barrier properties in thermoplastic potato starch films. Carbohydr. Polym. 2018, 194, 357–364. [Google Scholar] [CrossRef] [PubMed]
- Williams, C.K.; Hillmyer, M.A. Polymers from renewable resources: A perspective for a special issue of polymer reviews. Polym. Rev. 2008, 48, 1–10. [Google Scholar] [CrossRef]
- Bordes, P.; Pollet, E.; Avérous, L. Nano-biocomposites: Biodegradable polyester/nanoclay systems. Prog. Polym. Sci. 2009, 34, 125–155. [Google Scholar] [CrossRef]
- Kushwah, B.S.; Kushwah, A.V.S.; Singh, V. Towards understanding polyhydroxyalkanoates and their use. J. Polym. Res. 2016, 23, 1–14. [Google Scholar] [CrossRef]
- Poirier, Y.; Nawrath, C.; Somerville, C. Production of polyhydroxyalkanoates, a family of biodegradable plastics and elastomers, in bacteria and plants. Nat. Biotechnol. 1995, 13, 142–150. [Google Scholar] [CrossRef]
- Silverman, T.; Naffakh, M.; Marco, C.; Ellis, G. Morphology and thermal properties of biodegradable poly (hydroxybutyrate-co-hydroxyvalerate)/tungsten disulphide inorganic nanotube nanocomposites. Mater. Chem. Phys. 2016, 170, 145–153. [Google Scholar] [CrossRef]
- Avella, M.; Martuscelli, E.; Raimo, M. Review Properties of blends and composites based on poly(3-hydroxy) butyrate (PHB) and poly (3-hydroxybutyrate-hydroxyvalerate)(PHBV) copolymers. J. Mater. Sci. 2000, 35, 523–545. [Google Scholar] [CrossRef]
- Lagaron, J.M. Multifunctional and Nanoreinforced Polymers for Food Packaging, 1st ed.; Woodhead Publishing Elsevier Science: New York, NY, USA, 2011. [Google Scholar]
- Muller, K.; Bugnicourt, E.; Latorre, M.; Jorda, M.; Sanz, Y.E.; Lagaron, J.M.; Miesbauer, O.; Bianchin, A.; Hankin, S.; Bolz, U.; et al. Review on the Processing and Properties of Polymer Nanocomposites and Nanocoatings and Their Applications in the Packaging, Automotive and Solar Energy Fields. Nanomaterials 2017, 7, 74. [Google Scholar] [CrossRef] [PubMed]
- Fawaz, J.; Mittal, V. Synthesis of Polymer Nanocomposites: Review of Various Techniques, 1st ed.; John Wiley & Sons: Weinheim, Germany, 2014; pp. 1–26. [Google Scholar]
- Carli, L.N.; Crespo, J.S.; Mauler, R.S. PHBV nanocomposites based on organomodified montmorillonite and halloysite: The effect of clay type on the morphology and thermal and mechanical properties. Compos. Part A-Appl. Sci. Manuf. 2011, 42, 1601–1608. [Google Scholar] [CrossRef]
- Chen, G.X.; Hao, G.J.; Guo, T.Y.; Song, M.D.; Zhang, B.H. Structure and mechanical properties of poly(3-hydroxybutyrate-co-3-hydroxyvalerate) (PHBV)/clay nanocomposites. J. Mater. Sci. Lett. 2002, 21, 1587–1589. [Google Scholar] [CrossRef]
- Ten, E.; Turtle, J.; Bahr, D.; Jiang, L.; Wolcott, M. Thermal and mechanical properties of poly(3-hydroxybutyrate-co-3-hydroxyvalerate)/cellulose nanowhiskers composites. Polymer 2010, 51, 2652–2660. [Google Scholar] [CrossRef]
- Mook, C.W.; Wan, K.T.; Ok, P.O.; Keun, C.Y.; Woo, L.J. Preparation and characterization of poly(hydroxybutyrate-co-hydroxyvalerate)–organoclay nanocomposites. J. Appl. Polym. Sci. 2003, 90, 525–529. [Google Scholar] [CrossRef]
- Öner, M.; İlhan, B. Fabrication of poly(3-hydroxybutyrate-co-3-hydroxyvalerate) biocomposites with reinforcement by hydroxyapatite using extrusion processing. Mater. Sci. Eng. C 2016, 65, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Xiang, H.X.; Chen, S.H.; Cheng, Y.H.; Zhou, Z.; Zhu, M.F. Structural characteristics and enhanced mechanical and thermal properties of full biodegradable tea polyphenol/poly(3-hydroxybutyrate-co-3-hydroxyvalerate) composite films. eXPRESS Polym. Lett. 2013, 7, 778–786. [Google Scholar] [CrossRef]
- Luo, S.; Netravali, A.N. Mechanical and thermal properties of environment-friendly “green” composites made from pineapple leaf fibers and poly(hydroxybutyrate-co-valerate) resin. Poly. Compos. 1999, 20, 367–378. [Google Scholar] [CrossRef]
- Du, J.; Zhao, G.; Pan; Zhuang, L.; Li, D.; Zhang, R. Crystallization and mechanical properties of reinforced PHBV composites using melt compounding: Effect of CNCs and CNFs. Carbohydr. Polym. 2017, 168, 255–262. [Google Scholar]
- Qiang, T.; Wang, J.; Wolcott, M.P. Facile Fabrication of 100% Bio-Based and Degradable Ternary Cellulose/PHBV/PLA Composites. Materials 2018, 11, 330. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Yu, H.Y.; Wang, C.; Yao, J. Effect of silver contents in cellulose nanocrystal/silver nanohybrids on PHBV crystallization and property improvements. Carbohydr. Polym. 2017, 173, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.J.; Zhang, Y.J.; Zhang, J.Q.; Gou, Q.T.; Wang, Z.B.; Chen, P.; Gu, Q. Crystallization behavior, thermal and mechanical properties of PHBV/graphene nanosheet composites. Chin. J. Polym. Sci. 2013, 31, 670–678. [Google Scholar] [CrossRef]
- Wang, D.; Chen, G.; Li, C.; Cheng, M.; Yang, W.; Wu, S.; Chen, P. Thermally induced graphene rotation on hexagonal boron nitride. Phys. Rev. Lett. 2016, 116, 126101–126105. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Hao, X.; Zhao, M.; Wu, Y.; Yang, J.; Tian, Y.; Qian, G. Exfoliation of hexagonal boron nitride by molten hydroxides. Adv. Mater. 2013, 25, 2200–2204. [Google Scholar] [CrossRef] [PubMed]
- Raman, C.; Meneghetti, P. Boron nitride finds new applications in thermoplastic compounds. Plast. Addit. Comp. 2008, 10, 26–31. [Google Scholar] [CrossRef]
- Zhi, C.; Bando, Y.; Tang, C.; Kuwahara, H.; Golberg, D. Large-scale fabrication of boron nitride nanosheets and their utilization in polymeric composites with improved thermal and mechanical properties. Adv. Mater. 2009, 21, 2889–2893. [Google Scholar] [CrossRef]
- Wattanakul, K.; Manuspiya, H.; Yanumet, N. Thermal conductivity and mechanical properties of BN-filled epoxy composite: Effects of filler content, mixing conditions and BN agglomerate size. J. Compos. Mater. 2011, 45, 1967–1980. [Google Scholar] [CrossRef]
- Pradhan, G.C.; Behera, L.; Swain, S.K. Effects of boron nitride nanopowder on thermal, chemical and gas barrier properties of starch. Chin. J. Polym. Sci. 2014, 32, 1311–1318. [Google Scholar] [CrossRef]
- Cheewawuttipong, W.; Fuoka, D.; Tanoue, S.; Uematsu, H.; Iemoto, Y. Thermal and mechanical properties of polypropylene/boron nitride composites. Energy Procedia 2013, 34, 808–817. [Google Scholar] [CrossRef]
- Sun, G.; Bi, J.; Wang, W.; Zhang, J. Microstructure and mechanical properties of boron nitride nanosheets-reinforced fused silica composites. J. Eur. Ceram. Soc. 2017, 37, 3195–3202. [Google Scholar] [CrossRef]
- Kisku, S.K.; Swain, S.K. Synthesis and characterization of chitosan/boron nitride composites. J. Am. Ceram. Soc. 2012, 95, 2753–2757. [Google Scholar] [CrossRef]
- Zhou, W.; Zuo, J.; Zhang, X.; Zhou, A. Thermal, electrical and mechanical properties of hexagonal boron nitride-reinforced epoxy composites. J. Compos. Mater. 2014, 48, 2517–2526. [Google Scholar] [CrossRef]
- Li, S.; Yang, T.; Zou, H.; Liang, M.; Chen, Y. Enhancement in thermal conductivity and mechanical properties via large-scale fabrication of boron nitride nanosheet. High Perform. Polym. 2016, 29, 315–327. [Google Scholar] [CrossRef]
- Biscarat, J.; Bechelany, M.; Pochat-Bohatier, C.; Miele, P. Graphene-like BN/gelatin nanobiocomposites forgas barrier applications. Nanoscale 2015, 7, 613–618. [Google Scholar] [CrossRef] [PubMed]
- Öner, M.; Çöl, A.A.; Pochat-Bohatier, C.; Bechelany, M. Effect of incorporation of boron nitride nanoparticles on the oxygen barrier and thermalproperties of poly(3-hydroxybutyrate-cohydroxyvalerate). RSC Adv. 2016, 6, 90973–90981. [Google Scholar] [CrossRef]
- Öner, M.; Keskin, G.; Kızıl, G.; Pochat-Bohatier, C.; Bechelany, M. Development of poly(3-hydroxybutyrate-co-3-hydroxyvalerate)/boron nitride bionanocomposites with enhanced barrier properties. Polym. Compos. in press. [CrossRef]
- Yu, H.; Sun, B.; Zhang, D.; Chen, G.; Yang, X.; Yao, J. Reinforcement of biodegradable poly(3-hydroxybutyrate-co-3-hydroxyvalerate) with cellulose nanocrystal/silver nanohybrids as bifunctional nanofillers. J. Mater. Chem. B 2014, 2, 8479–8489. [Google Scholar] [CrossRef]
- Huang, W.; Wang, Y.; Ren, L.; Du, C.; Shi, X. A novel PHBV/HA microsphere releasing system loaded with alendronate. Mater. Sci. Eng. C 2009, 29, 2221–2225. [Google Scholar] [CrossRef]
- Ambrosio-Martín, J.; Gorrasi, G.; Lopez-Rubio, A.; Fabra, M.J.; Mas, L.C.; López-Manchado, M.A.; Lagaron, J.M. On the use of ball milling to develop poly(3-hydroxybutyrate-co-3-hydroxyvalerate)-graphene nanocomposites (II)—Mechanical, barrier, and electrical properties. J. Appl. Polym. Sci. 2015, 132, 42217–42225. [Google Scholar] [CrossRef]
- Yu, H.; Yan, C.; Yao, J. Fully biodegradable food packaging materials based on functionalized cellulose nanocrystals/poly(3-hydroxybutyrate-co-3-hydroxyvalerate) nanocomposites. RSC Adv. 2014, 4, 59792–59802. [Google Scholar] [CrossRef]
- Sato, H.; Murakami, R.; Padermshoke, A.; Hirose, F.; Senda, K.; Noda, I.; Ozaki, Y. Infrared Spectroscopy Studies of CH–O Hydrogen Bondings and Thermal Behavior of Biodegradable Poly(hydroxyalkanoate). Macromolecules 2004, 37, 7203–7213. [Google Scholar] [CrossRef]
- Suthar, V.; Pratap, A.; Raval, H. Studies on poly(hydroxy alkanoates)/(ethylcellulose) blends. Bull. Mater. Sci. 2000, 23, 215–219. [Google Scholar] [CrossRef]
- Singh, S.; Mohanty, A.K.; Sugie, T.; Takai, Y.; Hamada, H. Renewable resource based biocomposites from natural fiber and polyhydroxybutyrate-co-valerate (PHBV) bioplastic. Compos. Part A-Appl. Sci. Manuf. 2008, 39, 875–888. [Google Scholar] [CrossRef]
- Yu, L.; Gao, B.; Chen, Z.; Sun, C.; Cui, D.; Wang, C.; Wang, Q.; Jiang, M. In Situ FTIR Investigation on Phase Transformations in BN Nanoparticles. Chin. Sci. Bull. 2005, 50, 2827–2831. [Google Scholar]
- Shi, L.; Gu, Y.; Chen, L.; Qian, Y.; Yang, Z.; Ma, J. Synthesis and Morphology Control of Nanocrystalline Boron Nitride. J. Solid State Chem. 2004, 177, 721–724. [Google Scholar] [CrossRef]
- Soliveri, G.; Pifferi, V.; Annunziata, R.; Rimoldi, L.; Aina, V.; Cerrato, G.; Meroni, D. Alkylsilane–SiO2 Hybrids. A Concerted Picture of Temperature Effects in Vapor Phase Functionalization. J. Phys. Chem. C 2015, 119, 15390–15400. [Google Scholar] [CrossRef]
- Paul, B.; Martens, W.N.; Frost, R.L. Organosilane grafted acid-activated beidellite clay for the removal of non-ionic alachlor and anionic imazaquin. Appl. Surf. Sci. 2011, 257, 5552–5558. [Google Scholar] [CrossRef]
- Li, J.; Lai, M.F.; Liu, J.J. Effect of poly (propylene carbonate) on the crystallization and melting behavior of poly(hydroxybutyrate-co-hydroxyvalerate). J. Appl. Polym. Sci. 2004, 92, 2514–2521. [Google Scholar] [CrossRef]
- Camargo, F.A.; Innocentini-Mei, L.H.; Lemes, A.P.; Moraes, S.G.; Durán, N. Processing and characterization of composites of poly (3-hydroxybutyrate-co-hydroxyvalerate) and lignin from sugar cane bagasse. J. Compos. Mater. 2012, 46, 417–425. [Google Scholar] [CrossRef]
- Bhardwaj, R.; Mohanty, A.K.; Drzal, L.T.; Pourboghrat, F.; Misra, M. Renewable resource-based green composites from recycled cellulose fiber and poly(3-hydroxybutyrate-co-3-hydroxyvalerate) bioplastic. Biomacromolecules 2006, 7, 2044–2051. [Google Scholar] [CrossRef] [PubMed]
- Lai, M.; Li, J.; Yang, J.; Liu, J.; Tong, X.; Cheng, H. The morphology and thermal properties of multi-walled carbon nanotube and poly(hydroxybutyrate-co hydroxyvalerate) composite. Polym. Int. 2004, 53, 1479–1484. [Google Scholar] [CrossRef]
- Avella, M.; La, R.G.; Martuscelli, E.; Raimo, M. Poly(3-hydroxybutyrate-co-3-hydroxyvalerate) and wheat straw fibre composites: Thermal, mechanical properties and biodegradation behavior. J. Mater. Sci. 2000, 35, 829–836. [Google Scholar] [CrossRef]
- Hsu, S.F.; Wu, T.M.; Liao, C.S. Nonisothermal crystallization behavior and crystalline structure of poly(3-hydroxybutyrate)/layered double hydroxide nanocomposites. J. Polym. Sci. Part B: Polym. Phys. 2007, 45, 995–1002. [Google Scholar] [CrossRef]
- Cretois, R.; Follain, N.; Dargent, E.; Soulestin, J.; Bourbigot, S.; Marais, S.; Lebrun, L. Microstructure and barrier properties of PHBV/organoclays bionanocomposites. J. Membr. Sci. 2014, 467, 56–66. [Google Scholar] [CrossRef]
- Sanchez-Garcia, M.D.; Gimenez, E.; Lagaron, J.M. Novel PET nanocomposites of interest in food packaging applications and comparative barrier performance with biopolyester nanocomposites. J. Plast. Film Sheet. 2007, 23, 133–148. [Google Scholar] [CrossRef]
- Hassaini, L.; Kaci, M.; Touati, N.; Pillin, I.; Kervoelen, A.; Bruzaud, S. Valorization of olive husk flour as a filler for biocomposites based on poly (3-hydroxybutyrate-co-3-hydroxyvalerate): Effects of silane treatment. Polym. Test. 2017, 59, 430–440. [Google Scholar] [CrossRef]
- Pokharel, P.; Bae, H.; Lim, J.; Yong, L.K.; Choi, S. Effects of titanate treatment on morphology and mechanical properties of graphene nanoplatelets/high density polyethylene nanocomposites. J. Appl. Polym. Sci. 2015, 132, 42073–42085. [Google Scholar] [CrossRef]
- Lu, Y.L.; Zhang, G.B.; Feng, M.; Zhang, Y.; Yang, M.S.; Shen, D.T. Hydrogen bonding in polyamide 66/clay nanocomposite. J. Polym. Sci. Part B-Polym. Phys. 2003, 41, 2313–2321. [Google Scholar] [CrossRef]
- Evans, J.R.G.; Chen, B. Poly(epsilon-caprolactone)-clay nanocomposites: Structure and mechanical properties. Macromolecules 2006, 39, 747–754. [Google Scholar]
- Yu, W.; Lan, C.H.; Wang, S.J.; Fang, P.F.; Sun, Y.M. Influence of zinc oxide nanoparticles on the crystallization behavior of electrospun poly(3-hydroxybutyrate-co-3-hydroxyvalerate) nanofibers. Polymer 2010, 51, 2403–2409. [Google Scholar] [CrossRef]
- Bhattacharya, M. Polymer Nanocomposites—A comparison between Carbon Nanotubes, Graphene and Clay as Nanofillers. Materials 2016, 9, 262. [Google Scholar] [CrossRef] [PubMed]
- Cadek, M.; Coleman, J.N.; Ryan, K.P.; Nicolosi, V.; Bister, G.; Fonseca, A.; Nagy, J.B.; Szostak, K.; Beguin, F.; Blau, W.J. Reinforcement of polymers with carbon nanotubes: The role of surface area. Nano Lett. 2004, 4, 353–356. [Google Scholar] [CrossRef]
- Carotenuto, G.; De Nicola, S.; Palomba, M.; Pullini, D.; Horsewell, A.; Hansen, T.W.; Nicolais, L. Mechanical properties of low-density polyethylene filled by graphite nanoplatelets. Nanotechnology 2012, 23, 48570–48713. [Google Scholar] [CrossRef] [PubMed]
- Xiang, H.; Li, L.; Wang, S.; Wang, R.; Cheng, Y.; Zhou, Z.; Zhu, M. Natural polyphenol tannic acid reinforced poly(3-hydroxybutyrate-co-3-hydroxyvalerate) composite films with enhanced tensile strength and fracture toughness. Polym. Compos. 2015, 36, 2303–2308. [Google Scholar] [CrossRef]
- Dong, Y.; Bhattacharyya, D. A simple micromechanical approach to predict mechanical behaviour of polypropylene/organoclay nanocomposites based on representative volume element (RVE). Comput. Mater. Sci. 2010, 49, 1–8. [Google Scholar] [CrossRef]
- Colemn, J.N.; Khan, U.; Blau, W.J.; Gun’ko, Y.K. Small but strong: A review of the mechanical properties of carbon nanotube–polymer composites. Carbon 2006, 44, 1624–1652. [Google Scholar] [CrossRef]





| Sample | Boron Nitride Content (wt.%) |
|---|---|
| PHBV | - |
| BORTEK | |
| PHBV/0.5OSBN | 0.5 |
| PHBV/1BN | 1 |
| PHBV/1OSBN | 1 |
| PHBV/2OSBN | 2 |
| PHBV/3OSBN | 3 |
| SAINT GOBAIN (PHPP325B) | |
| PHBV/0.5OSFBN | 0.5 |
| PHBV/1FBN | 1 |
| PHBV/1OSFBN | 1 |
| PHBV/2OSFBN | 2 |
| PHBV/3OSFBN | 3 |
| (020)/(110) | |
|---|---|
| PHBV | 1.42 |
| PHBV/0.5OSBN | 3.17 |
| PHBV/1OSBN | 3.40 |
| PHBV/2OSBN | 4.58 |
| PHBV/3OSBN | 2.56 |
| PHBV/0.5OSFBN | 4.98 |
| PHBV/1OSFBN | 6.72 |
| PHBV/2OSFBN | 4.70 |
| PHBV/3OSFBN | 3.76 |
| Sample | Ti (°C) | T10 (°C) | T50 (°C) | Tmax (°C) | Char (%) |
|---|---|---|---|---|---|
| PHBV | 234.45 | 243.50 | 256.04 | 275.08 | 1.81 |
| PHBV/0.5OSBN | 251.90 | 271.35 | 283.39 | 293.90 | 1.82 |
| PHBV/1BN | 250.30 | 271.97 | 282.97 | 292.30 | 2.03 |
| PHBV/1OSBN | 252.70 | 270.67 | 282.10 | 294.70 | 2.23 |
| PHBV/2OSBN | 253.50 | 271.53 | 282.73 | 295.50 | 2.88 |
| PHBV/3OSBN | 254.30 | 271.06 | 282.30 | 295.50 | 3.97 |
| PHBV/0.5OSFBN | 248.92 | 269.00 | 279.45 | 289.44 | 2.17 |
| PHBV/1FBN | 247.09 | 268.18 | 278.92 | 289.54 | 2.53 |
| PHBV/1OSFBN | 251.10 | 267.82 | 278.37 | 289.90 | 2.62 |
| PHBV/2OSFBN | 251.90 | 269.40 | 279.50 | 291.06 | 3.06 |
| PHBV/3OSFBN | 253.45 | 270.17 | 280.11 | 294.17 | 3.75 |
| Sample | First Heating | Cooling | Second Heating | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Tm1 (°C) | Tm2 (°C) | ΔHm1 (j/g) | Xc (%) | Tc1 (°C) | ΔHc (j/g) | Tm1 (°C) | ΔHm2 (j/g) | Xc (%) | |
| PHBV | 170 | 173 | 88 | 60 | 122 | 86 | 171 | 100 | 68 |
| PHBV/0.5OSBN | 169 | 174 | 87 | 60 | 122 | 88 | 170 | 98 | 67 |
| PHBV/1BN | 171 | - | 81 | 57 | 124 | 84 | 172 | 95 | 65 |
| PHBV/1OSBN | 166 | 173 | 86 | 60 | 121 | 91 | 168 | 100 | 69 |
| PHBV/2OSBN | 171 | - | 86 | 60 | 122 | 87 | 170 | 100 | 70 |
| PHBV/3OSBN | 168 | 175 | 83 | 56 | 123 | 81 | 164 | 90 | 64 |
| PHBV/0.5OSFBN | 170 | 175 | 87 | 60 | 123 | 89 | 171 | 99 | 68 |
| PHBV/1FBN | 171 | - | 87 | 60 | 125 | 89 | 171 | 100 | 69 |
| PHBV/1OSFBN | 169 | 173 | 89 | 61 | 122 | 90 | 169 | 102 | 71 |
| PHBV/2OSFBN | 170 | - | 85 | 59 | 124 | 84 | 172 | 95 | 67 |
| PHBV/3OSFBN | 171 | - | 82 | 58 | 124 | 81 | 172 | 91 | 65 |
| Model | Array Type | Formula |
|---|---|---|
| Halpin–Tsai Model | Random array | |
| Hui–Shia Model | Regular array |
| Sample | Halpin–Tsai Deviation (%) | Hui–Shia Deviation (%) |
|---|---|---|
| PHBV/0.5OSBN | 2.480 | 1.743 |
| PHBV/1OSBN | 3.447 | 4.829 |
| PHBV/2OSBN | 3.575 | 6.306 |
| PHBV/3OSBN | 9.353 | 4.757 |
| PHBV/0.5OSFBN | 5.578 | 6.730 |
| PHBV/1OSFBN | 13.473 | 15.560 |
| PHBV/2OSFBN | 6.133 | 1.123 |
| PHBV/3OSFBN | 7.460 | 0.011 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Öner, M.; Kızıl, G.; Keskin, G.; Pochat-Bohatier, C.; Bechelany, M. The Effect of Boron Nitride on the Thermal and Mechanical Properties of Poly(3-hydroxybutyrate-co-3-hydroxyvalerate). Nanomaterials 2018, 8, 940. https://doi.org/10.3390/nano8110940
Öner M, Kızıl G, Keskin G, Pochat-Bohatier C, Bechelany M. The Effect of Boron Nitride on the Thermal and Mechanical Properties of Poly(3-hydroxybutyrate-co-3-hydroxyvalerate). Nanomaterials. 2018; 8(11):940. https://doi.org/10.3390/nano8110940
Chicago/Turabian StyleÖner, Mualla, Gülnur Kızıl, Gülşah Keskin, Celine Pochat-Bohatier, and Mikhael Bechelany. 2018. "The Effect of Boron Nitride on the Thermal and Mechanical Properties of Poly(3-hydroxybutyrate-co-3-hydroxyvalerate)" Nanomaterials 8, no. 11: 940. https://doi.org/10.3390/nano8110940
APA StyleÖner, M., Kızıl, G., Keskin, G., Pochat-Bohatier, C., & Bechelany, M. (2018). The Effect of Boron Nitride on the Thermal and Mechanical Properties of Poly(3-hydroxybutyrate-co-3-hydroxyvalerate). Nanomaterials, 8(11), 940. https://doi.org/10.3390/nano8110940






