Fabrication of ZnO/Red Phosphorus Heterostructure for Effective Photocatalytic H2 Evolution from Water Splitting
Abstract
1. Introduction
2. Experiments
2.1. Synthesis of ZnO/RP Heterojunction Photocatalyst
2.2. Characterizations
2.3. Photocatalytic H2 Evolution Experiments
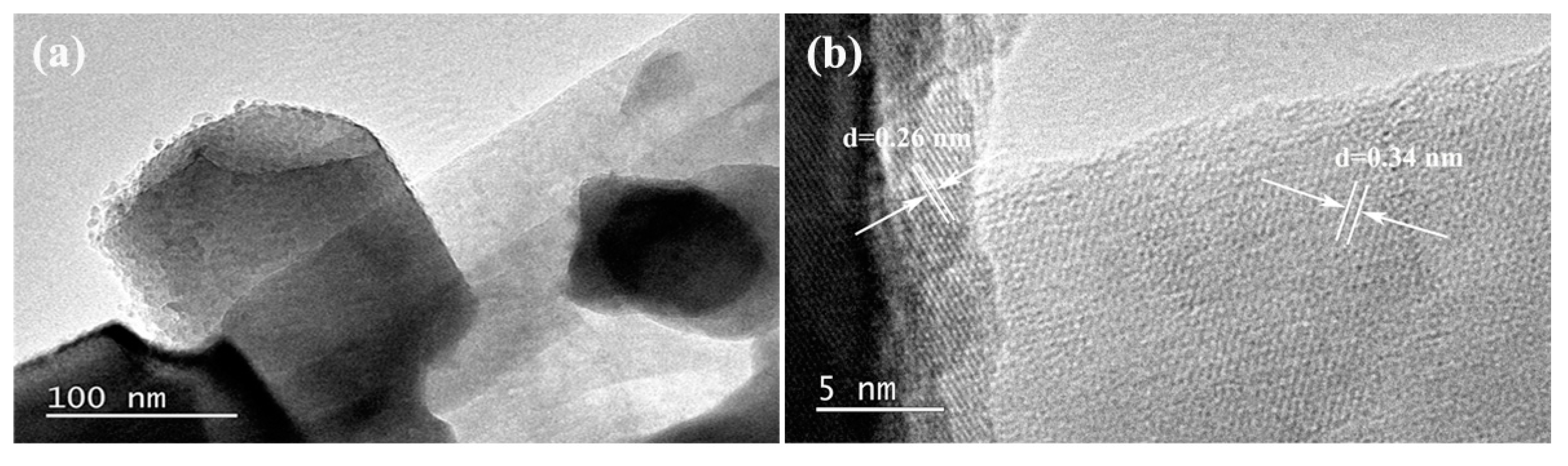
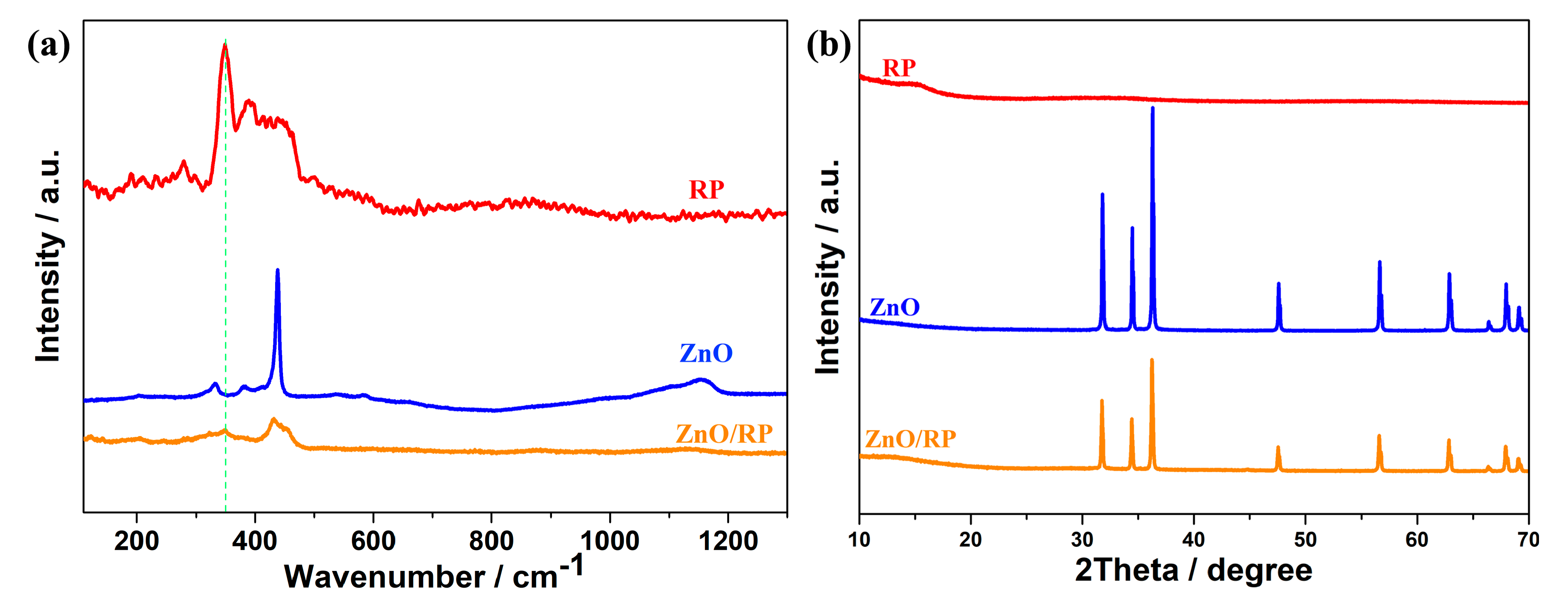
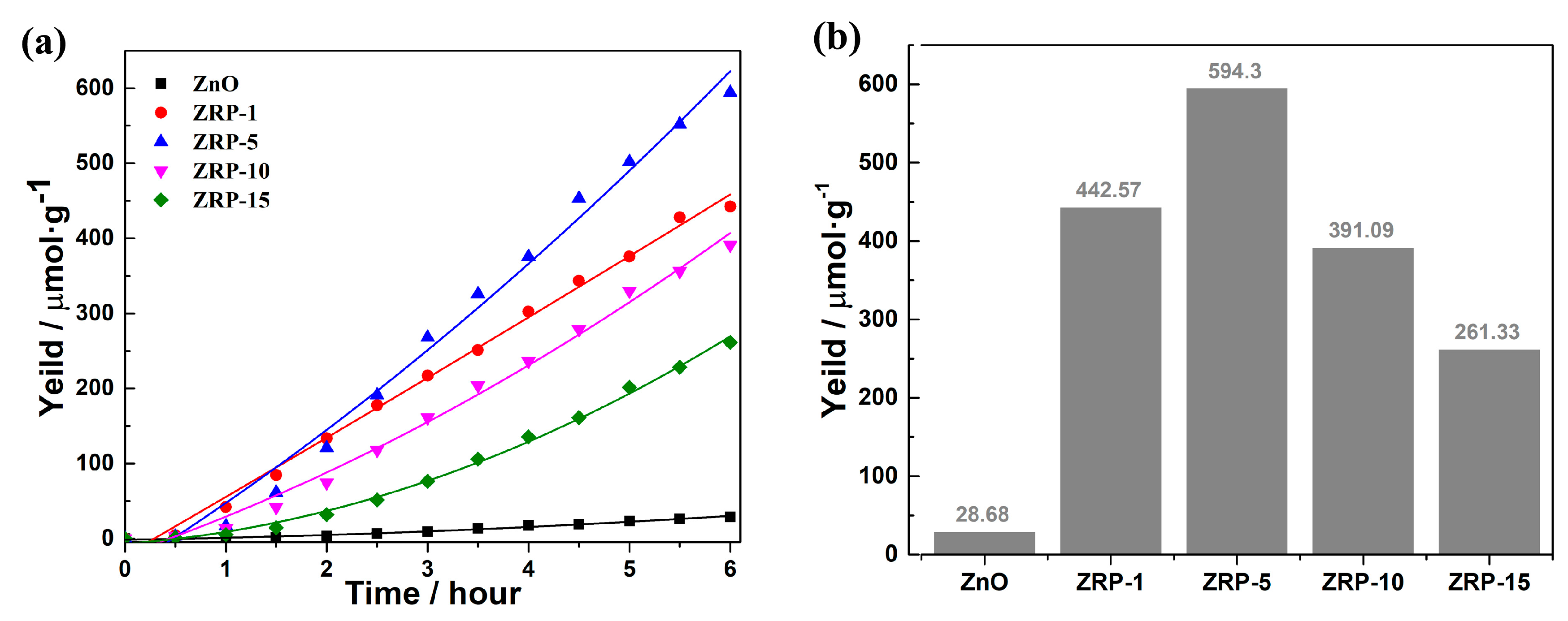
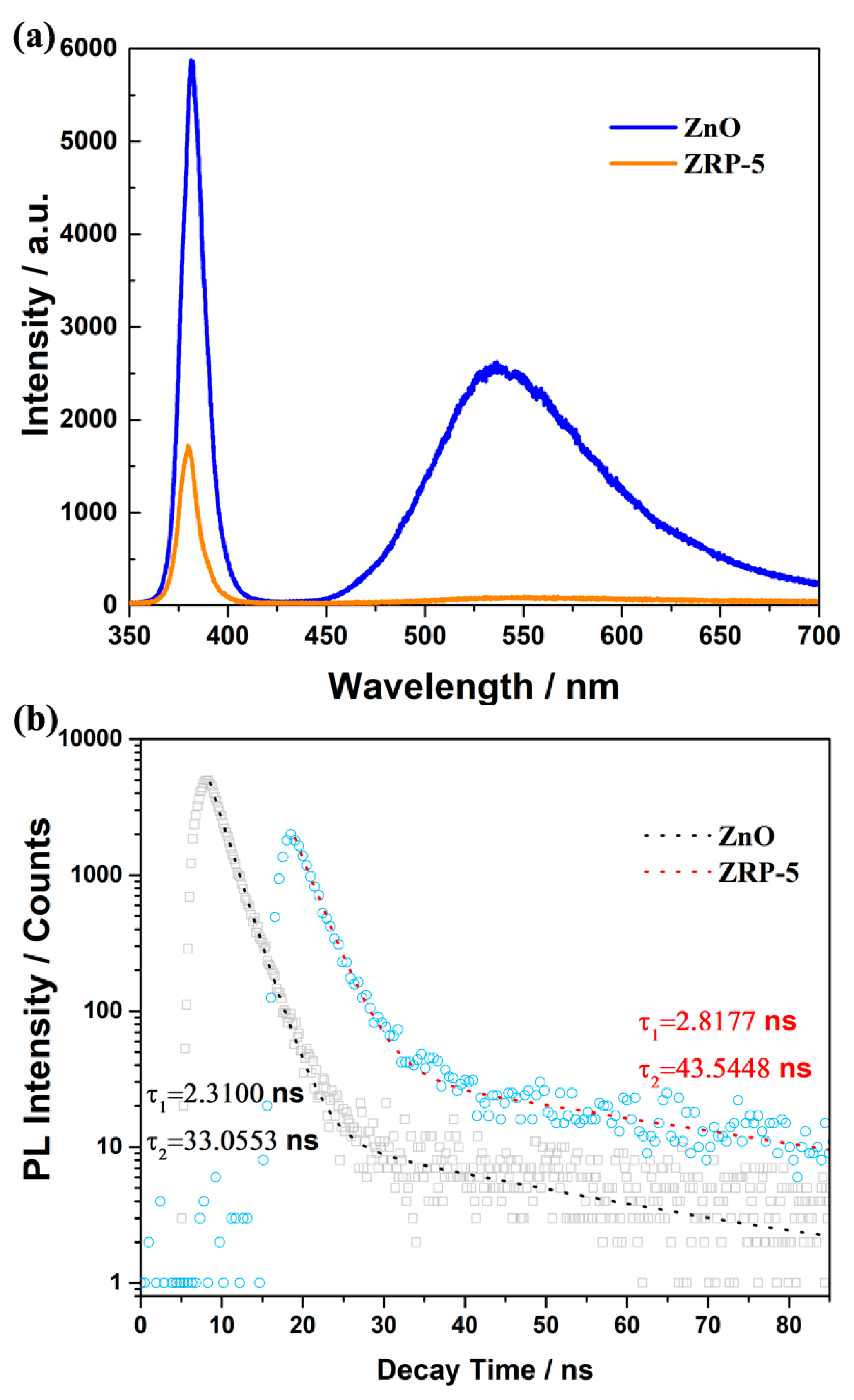
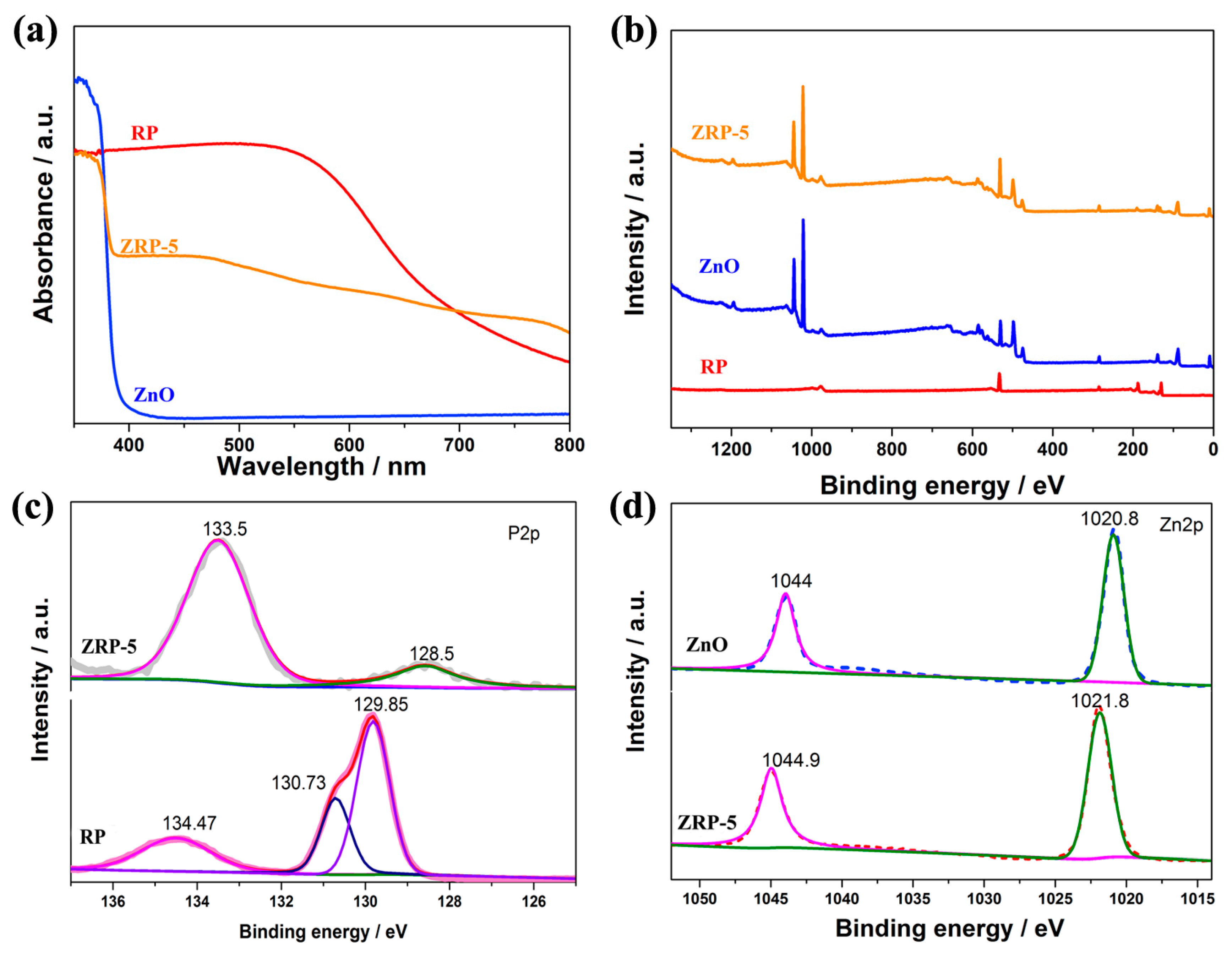
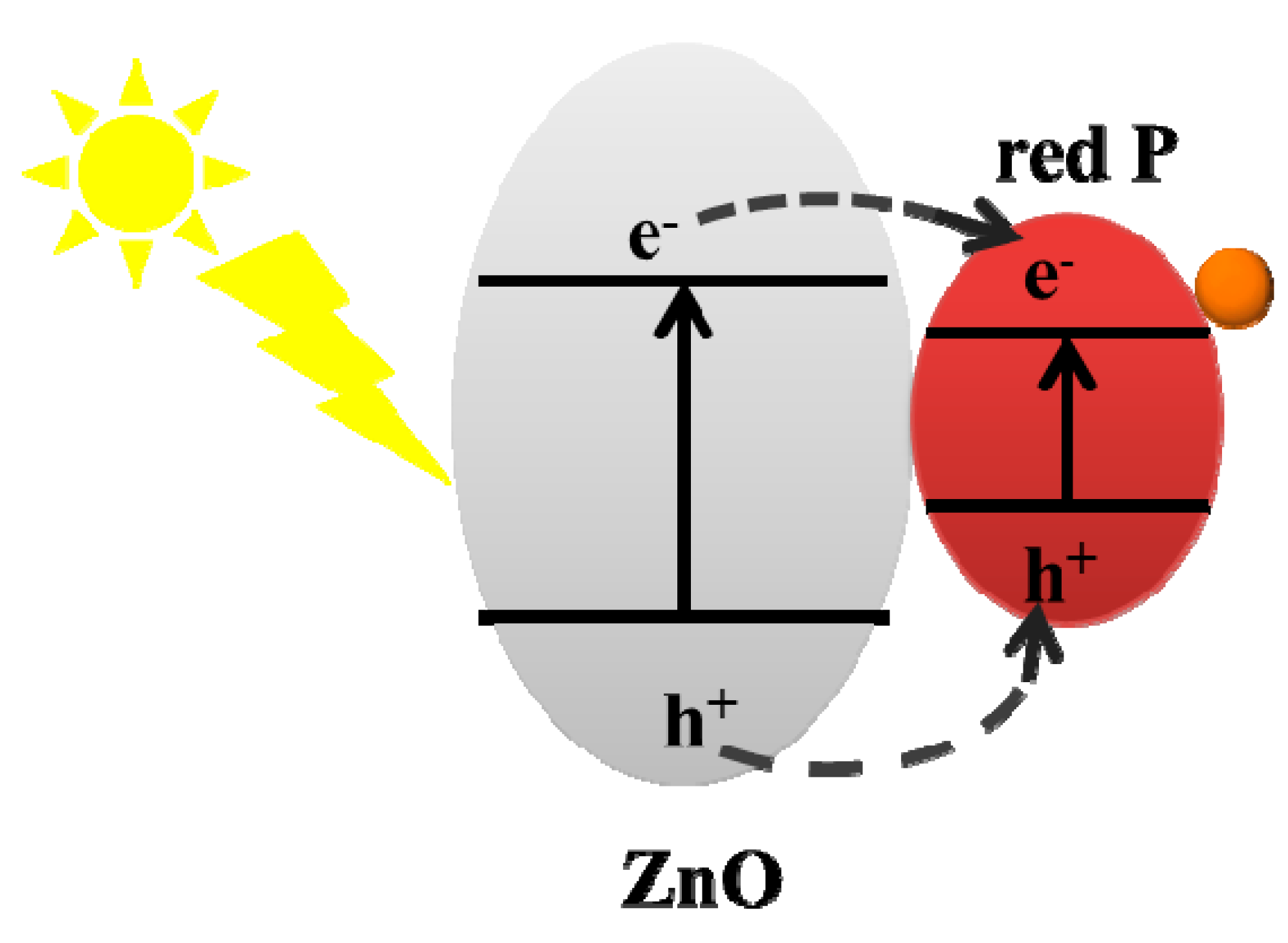
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Yu, C.; Yu, J.C.; Zhou, W.; Yang, K. WO3 Coupled P-TiO2 photocatalysts with mesoporous structure. Catal. Lett. 2010, 140, 172–183. [Google Scholar] [CrossRef]
- Ran, J.; Zhang, J.; Yu, J.; Jaroniec, M.; Qiao, S.Z. Earth-abundant cocatalysts for semiconductor-based photocatalytic water splitting. Chem. Soc. Rev. 2014, 43, 7787–7812. [Google Scholar] [CrossRef] [PubMed]
- Fujishima, A.; Honda, K. Electrochemical photolysis of water at a semiconductor electrode. Nature 1972, 238, 238–245. [Google Scholar] [CrossRef]
- Yang, Y.; Liu, G.; Irvine, J.T.; Cheng, H.M. Enhanced photocatalytic H2 production in Core-Shell engineered rutile TiO2. Adv. Mater. 2016, 28, 5850–5856. [Google Scholar] [CrossRef] [PubMed]
- Jungwon, K.; Chulwee, L.; Wonyong, C. Platinized WO3 as an environmental photocatalyst that generates OH radicals under visible light. Environ. Sci. Technol. 2010, 44, 6849. [Google Scholar] [CrossRef]
- Kambur, A.; Pozan, G.S.; Boz, I. Preparation, characterization and photocatalytic activity of TiO2–ZrO2 binary oxide nanoparticles. Appl. Catal. B 2012, 115–116, 149–158. [Google Scholar] [CrossRef]
- Dodd, A.; McKinley, A.; Saunders, M.; Tsuzuki, T. Mechanochemical synthesis of nanocrystalline SnO2–ZnO photocatalysts. Nanotechnology 2006, 17, 692–698. [Google Scholar] [CrossRef]
- Zhang, N.; Fu, X.; Xu, Y.-J. A facile and green approach to synthesize Pt@CeO2 nanocomposite with tunable core-shell and yolk-shell structure and its application as a visible light photocatalyst. J. Mater. Chem. 2011, 21, 8152. [Google Scholar] [CrossRef]
- Li, Y.; Wang, L.; Liang, J.; Gao, F.; Yin, K.; Dai, P. Hierarchical heterostructure of ZnO@TiO2 hollow spheres for highly efficient photocatalytic hydrogen evolution. Nanoscale Res. Lett. 2017, 12, 531. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.M.; Adil, S.F.; Al-Mayouf, A. Metal oxides as photocatalysts. J. Saudi Chem. Soc. 2015, 19, 462–464. [Google Scholar] [CrossRef]
- Lee, K.M.; Lai, C.W.; Ngai, K.S.; Juan, J.C. Recent developments of zinc oxide based photocatalyst in water treatment technology: A review. Water Res. 2016, 88, 428–448. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.J.; Pereira, L.M.; Menghini, M.; Temst, K.; Vantomme, A.; Locquet, J.P.; Van Haesendonck, C. Tuning quantum corrections and magnetoresistance in ZnO nanowires by ion implantation. Nano Lett. 2012, 12, 666–672. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.J.; Ye, Z.Z.; Lu, Y.F.; Lu, J.G.; Sun, L.; Xu, W.Z.; Zhu, L.P.; Zhao, B.H.; Che, Y. ZnMgO quantum dots grown by low-pressure metal organic chemical vapor deposition. Appl. Phys. Lett. 2007, 90, 012111. [Google Scholar] [CrossRef]
- Ma, D.; Shi, J.W.; Zou, Y.; Fan, Z.; Ji, X.; Niu, C. Highly efficient photocatalyst based on a CdS Quantum Dots/ZnO nanosheets 0D/2D heterojunction for hydrogen evolution from water splitting. ACS. Appl. Mater. Inter. 2017, 9, 25377–25386. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Liu, G.; Chen, Z.G.; Li, F.; Wang, L.; Lu, G.Q.; Cheng, H.M. Enhanced photocatalytic hydrogen evolution by prolonging the lifetime of carriers in ZnO/CdS heterostructures. Chem. Commun. 2009, 23, 3452–3454. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Cheng, H.; Zong, R.; Zhu, Y. Photocorrosion Suppression of ZnO Nanoparticles via Hybridization with Graphite-like Carbon and Enhanced Photocatalytic Activity. J. Phys. Chem. C 2009, 113, 2368–2374. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, L.; Chen, Z.; Hu, J.; Li, S.; Wang, Z.; Liu, J.; Wang, X. Semiconductor heterojunction photocatalysts: Design, construction, and photocatalytic performances. Chem. Soc. Rev. 2014, 43, 5234–5244. [Google Scholar] [CrossRef] [PubMed]
- Fujishima, A.; Zhang, X.; Tryk, D. Heterogeneous photocatalysis: From water photolysis to applications in environmental clean up. Int. J. Hydrogen Energy 2007, 32, 2664–2672. [Google Scholar] [CrossRef]
- Low, J.; Cao, S.; Yu, J.; Wageh, S. Two-dimensional layered composite photocatalysts. Chem. Commun. 2014, 50, 10768–10777. [Google Scholar] [CrossRef] [PubMed]
- Low, J.; Yu, J.; Jaroniec, M.; Wageh, S.; Al-Ghamdi, A.A. Heterojunction Photocatalysts. Adv. Mater. 2017, 29, 101694. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, Q.; Zhan, X.; Wang, F.; Safdar, M.; He, J. Visible light driven type II heterostructures and their enhanced photocatalysis properties: A review. Nanoscale 2013, 5, 8326–8339. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Shen, Z.; Yu, J.C. Phosphorus containing materials for photocatalytic hydrogen evolution. Green Chem. 2017, 19, 588–613. [Google Scholar] [CrossRef]
- Hu, Z.; Yuan, L.; Liu, Z.; Shen, Z.; Yu, J.C. An elemental phosphorus photocatalyst with a record high hydrogen evolution efficiency. Angew. Chem. Int. Ed. 2016, 55, 9580–9585. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Ng, W.K.H.; Yu, J.C.; Zhu, H.; Li, C.; Zhang, L.; Liu, Z.; Li, Q. Red phosphorus: An elemental photocatalyst for hydrogen formation from water. Appl. Catal. B 2012, 111–112, 409–414. [Google Scholar] [CrossRef]
- Shen, Z.; Sun, S.; Wang, W.; Liu, J.; Liu, Z.; Yu, J.C. A black–red phosphorus heterostructure for efficient visible-light-driven photocatalysis. J. Mater. Chem. A 2015, 3, 3285–3288. [Google Scholar] [CrossRef]
- Yuan, Y.-P.; Cao, S.-W.; Liao, Y.-S.; Yin, L.-S.; Xue, C. Red phosphor/g-C3N4 heterojunction with enhanced photocatalytic activities for solar fuels production. Appl. Catal. B 2013, 140–141, 164–168. [Google Scholar] [CrossRef]
- Shi, Z.; Dong, X.; Dang, H. Facile fabrication of novel red phosphorus-CdS composite photocatalysts for H2 evolution under visible light irradiation. Int. J. Hydrogen Energy 2016, 41, 5908–5915. [Google Scholar] [CrossRef]
- Qi, K.; Yang, J.; Fu, J.; Wang, G.; Zhu, L.; Liu, G.; Zheng, W. Morphology-controllable ZnO rings: Ionic liquid-assisted hydrothermal synthesis, growth mechanism and photoluminescence properties. CrystEngComm 2013, 15, 6729. [Google Scholar] [CrossRef]
- Chang, W.C.; Tseng, K.W.; Tuan, H.Y. Solution Synthesis of Iodine-Doped Red Phosphorus Nanoparticles for Lithium-Ion Battery Anodes. Nano Lett. 2017, 17, 1240–1247. [Google Scholar] [CrossRef] [PubMed]
- D’Amato, C.A.; Giovannetti, R.; Zannotti, M.; Rommozzi, E.; Minicucci, M.; Gunnella, R.; Di Cicco, A. Band gap implications on nano-TiO2 surface modification with ascorbic acid for visible light-active polypropylene coated photocatalyst. Nanomaterials 2018, 8, 599. [Google Scholar] [CrossRef] [PubMed]
- Gong, C.; Du, J.; Li, X.; Yu, Z.; Ma, J.; Qi, W.; Zhang, K.; Yang, J.; Luo, M.; Peng, H. One-step acidic hydrothermal preparation of dendritic rutile TiO2 nanorods for Photocatalytic Performance. Nanomaterials 2018, 8, 683. [Google Scholar] [CrossRef] [PubMed]
- Ma, D.; Shi, J.-W.; Zou, Y.; Fan, Z.; Ji, X.; Niu, C.; Wang, L. Rational design of CdS@ZnO core-shell structure via atomic layer deposition for drastically enhanced photocatalytic H2 evolution with excellent photostability. Nano Energy 2017, 39, 183–191. [Google Scholar] [CrossRef]
- Yu, Z.B.; Xie, Y.P.; Liu, G.; Lu, G.Q.; Ma, X.L.; Cheng, H.-M. Self-assembled CdS/Au/ZnO heterostructure induced by surface polar charges for efficient photocatalytic hydrogen evolution. J. Mater. Chem. A 2013, 1, 2773. [Google Scholar] [CrossRef]
- Moon, S.C.; Mametsuka, H.; Tabata, S.; Suzuki, E. Photocatalytic production of hydrogen from water using TiO2 and B/TiO2. Catal. Today 2000, 58, 125–132. [Google Scholar] [CrossRef]
- Gao, J.; Zhao, Q.; Sun, Y.; Li, G.; Zhang, J.; Yu, D. A Novel Way for Synthesizing Phosphorus-Doped Zno Nanowires. Nanoscale Res. Lett. 2011, 6, 45. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Zhang, Y.; Dong, F.; Reshak, A.H.; Ye, L.; Pinna, N.; Zeng, C.; Zhang, T.; Huang, H. Chlorine intercalation in graphitic carbon nitride for efficient photocatalysis. Appl. Catal. B 2017, 203, 465–474. [Google Scholar] [CrossRef]
- Xu, T.; Zhang, L.; Cheng, H.; Zhu, Y. Significantly enhanced photocatalytic performance of ZnO via graphene hybridization and the mechanism study. Appl. Catal. B 2011, 101, 382–387. [Google Scholar] [CrossRef]
- Yang, C.; Qin, J.; Xue, Z.; Ma, M.; Zhang, X.; Liu, R. Rational design of carbon-doped TiO2 modified g-C3N4 via in-situ heat treatment for drastically improved photocatalytic hydrogen with excellent photostability. Nano Energy 2017, 41, 1–9. [Google Scholar] [CrossRef]
- Zhang, Z.; Huang, J.; Fang, Y.; Zhang, M.; Liu, K.; Dong, B. A nonmetal plasmonic Z-Scheme photocatalyst with UV-to NIR-Driven photocatalytic protons reduction. Adv. Mater. 2017, 29. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Zhang, X.; Sun, Y.; Zhao, Q.; Yu, D. Compensation mechanism in N-doped ZnO nanowires. Nanotechnology 2010, 21, 245703. [Google Scholar] [CrossRef] [PubMed]
- Jing, L.; Zhu, R.; Phillips, D.L.; Yu, J.C. Effective prevention of charge trapping in graphitic carbon nitride with nanosized red phosphorus modification for superior photo(electro)catalysis. Adv. Funct. Mater. 2017, 27, 1703484. [Google Scholar] [CrossRef]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, J.; Huang, S.; Long, Y.; Wu, J.; Li, H.; Li, Z.; Zeng, Y.-J.; Ruan, S. Fabrication of ZnO/Red Phosphorus Heterostructure for Effective Photocatalytic H2 Evolution from Water Splitting. Nanomaterials 2018, 8, 835. https://doi.org/10.3390/nano8100835
Chen J, Huang S, Long Y, Wu J, Li H, Li Z, Zeng Y-J, Ruan S. Fabrication of ZnO/Red Phosphorus Heterostructure for Effective Photocatalytic H2 Evolution from Water Splitting. Nanomaterials. 2018; 8(10):835. https://doi.org/10.3390/nano8100835
Chicago/Turabian StyleChen, Jiaqi, Shaolong Huang, Yaojia Long, Jiahao Wu, Hui Li, Zhao Li, Yu-Jia Zeng, and Shuangchen Ruan. 2018. "Fabrication of ZnO/Red Phosphorus Heterostructure for Effective Photocatalytic H2 Evolution from Water Splitting" Nanomaterials 8, no. 10: 835. https://doi.org/10.3390/nano8100835
APA StyleChen, J., Huang, S., Long, Y., Wu, J., Li, H., Li, Z., Zeng, Y.-J., & Ruan, S. (2018). Fabrication of ZnO/Red Phosphorus Heterostructure for Effective Photocatalytic H2 Evolution from Water Splitting. Nanomaterials, 8(10), 835. https://doi.org/10.3390/nano8100835




