Chalcopyrite Nanoparticles as a Sustainable Thermoelectric Material
Abstract
:1. Introduction
2. Results and Discussion
2.1. Morphology

2.2. Crystalline Properties

2.3. Composition
| Sample | Cu:Fe Input (%) | ICP-OES | EDS (%) | |||
|---|---|---|---|---|---|---|
| Cu (%) | Fe (%) | Cu (%) | Fe (%) | |||
| B | 70:30 | 77 | 23 | 82 ± 3 | 18 ± 3 | |
| C | 50:50 | 54 | 46 | 54 ± 1 | 46 ± 1 | |
| D | 30:70 | 36 | 64 | 51 ± 8 | 49 ± 8 | Area 1 |
| 20 ± 5 | 80 ± 5 | Area 2 | ||||
2.4. Seebeck Coefficient Measurement
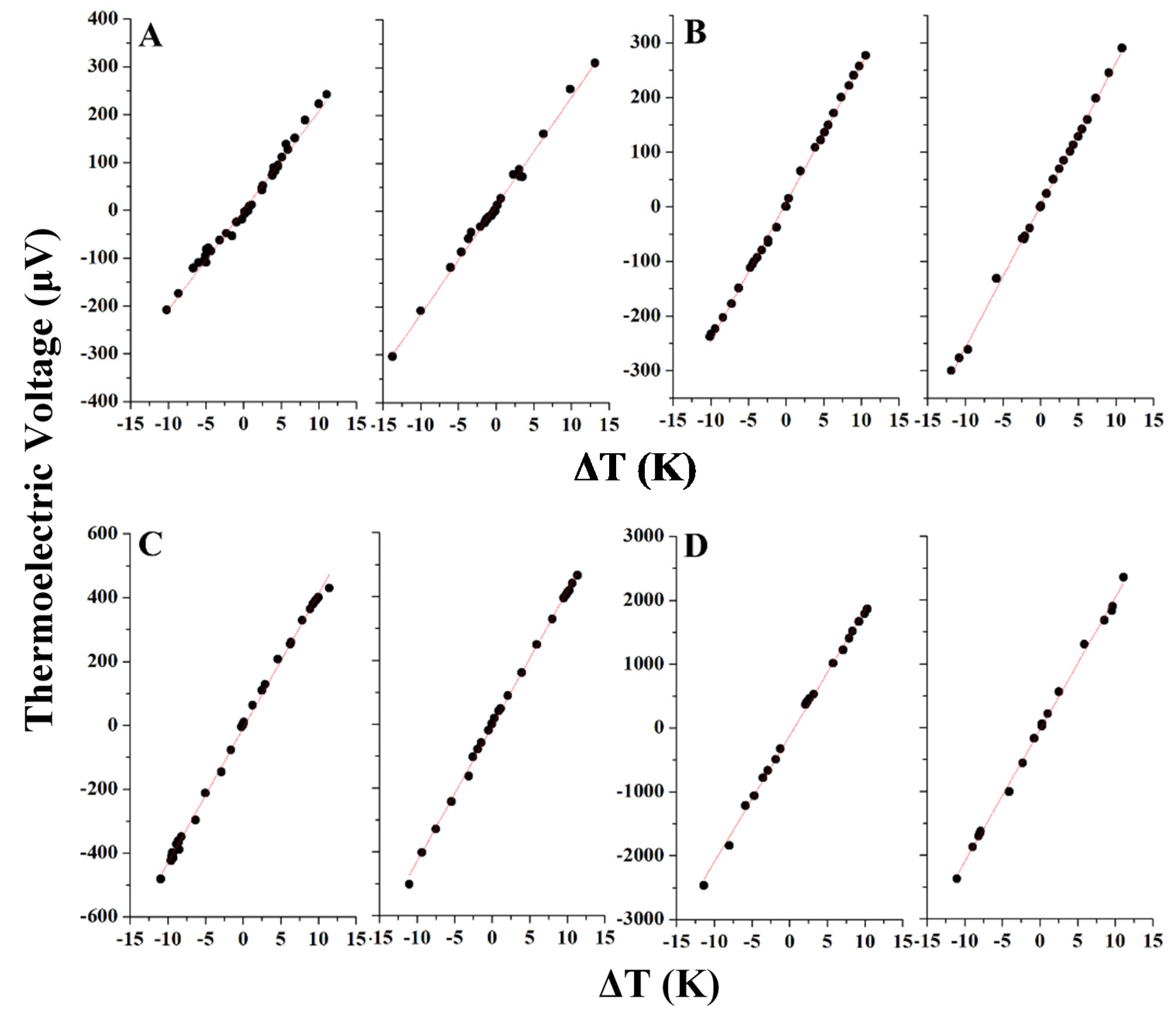
| Sample | Seebeck Coefficient (μV/K) | Electrical Conductivity (S/m) | Power Factor (μW/mK2) |
|---|---|---|---|
| A | +22 ± 1 | 9304 ± 539 | 4.5 |
| B | +26 ± 1 | 2850 ± 139 | 1.9 |
| C | +43 ± 1 | 1519 ± 24 | 2.8 |
| D | +203 ± 7 | 2.2 ± 0.4 | 0.1 |
2.5. Discussion
3. Experimental Section
3.1. Chemicals
3.2. Instrumentation
3.3. Nanoparticle Synthetic Technique
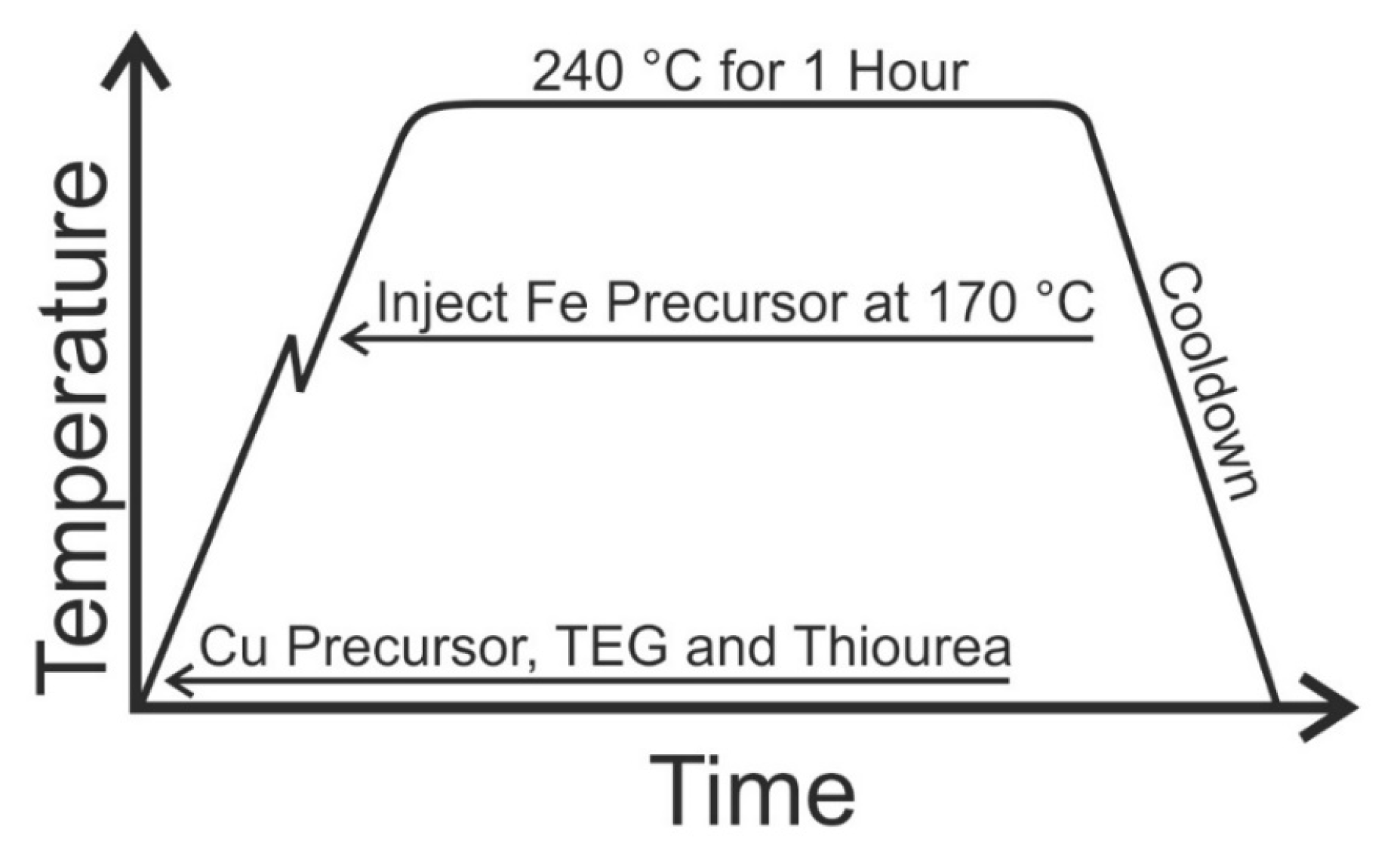
| Sample | Copper Nitrate (mmol) | Iron Sulfate (mmol) | Cu:Fe (%) |
|---|---|---|---|
| A | 10 | 0 | 100:0 |
| B | 7 | 3 | 70:30 |
| C | 5 | 5 | 50:50 |
| D | 3 | 7 | 30:70 |
4. Conclusions
Supplementary Materials
Author Contributions
Conflicts of Interest
References and Notes
- Yaldagard, M.; Jahanshahi, M.; Seghatoleslami, N. Carbonaceous nanostructured support materials for low temperature fuel cell electrocatalysts—A review. World J. Nano Sci. Eng. 2013, 3, 121–153. [Google Scholar] [CrossRef]
- Guo, C.F.; Sun, T.; Cao, F.; Liu, Q.; Ren, Z. Metallic nanostructures for light trapping in energy-harvesting devices. Light Sci. Appl. 2014, 3. [Google Scholar] [CrossRef]
- Okada, T.; Saiki, T.; Taniguchi, S.; Ueda, T.; Nakamura, K.; Nishikawa, Y.; Iida, Y. Hydrogen production using reduced-iron nanoparticles by laser ablation in liquids. ISRN Renew. Energy 2013, 2013. [Google Scholar] [CrossRef]
- Kishore, S.; Nelson, J.A.; Adair, J.H.; Eklund, P.C. Hydrogen storage in spherical and platelet palladium nanoparticles. J. Alloy. Compd. 2005, 389, 234–242. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, H.; Kraemer, S.; Shi, Y.; Zhang, F.; Snedaker, M.; Ding, K.; Moskovits, M.; Snyder, G.J.; Stucky, G.D. Surfactant-free synthesis of Bi2Te3-Te micro-nano heterostructure with enhanced thermoelectric figure of merit. ACS Nano 2011, 4, 3158–3165. [Google Scholar] [CrossRef] [PubMed]
- Poudel, B.; Hao, Q.; Ma, Y.; Lan, Y.; Minnich, A.; Yu, B.; Yan, X.; Wang, D.; Muto, A.; Vashaee, D.; et al. High-thermoelectric performance of nanostructured bismuth antimony telluride bulk alloys. Science 2008, 320, 634–638. [Google Scholar] [CrossRef] [PubMed]
- Heremans, J.P.; Jovovic, V.; Toberer, E.S.; Saramat, A.; Kurosaki, K.; Charoenphakdee, A.; Yamanaka, S.; Snyder, G.J. Enhancement of thermoelectric efficiency in PbTe by distortion of the electronic density of states. Science 2008, 321, 554–557. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, A.P.; Godart, C. New promising bulk thermoelectrics: Intermetallics, pnictides and chalcogenides. Eur. Phys. J. B 2014, 87. [Google Scholar] [CrossRef]
- Zhao, L.-D.; Hao, S.; Lo, S.-H.; Wu, C.-I.; Zhou, X.; Lee, Y.; Li, H.; Biswas, K.; Hogan, T.P.; Uher, C.; et al. High thermoelectric performance via hierarchical compositionally alloyed nanostructures. J. Am. Chem. Soc. 2013, 135, 7364–7370. [Google Scholar] [CrossRef] [PubMed]
- Liang, D.; Ma, R.; Jiao, S.; Pang, G.; Feng, S. A facile synthetic approach for copper iron sulfide nanocrystals with enhanced thermoelectric performance. Nanoscale 2012, 4, 6265–6268. [Google Scholar] [CrossRef] [PubMed]
- Rowe, D.M. CRC Handbook of Thermoelectrics; CRC Press: Boca Raton, FL, USA, 1995; p. 407. [Google Scholar]
- Ge, Z.-H.; Zhang, B.-P.; Chen, Y.-X.; Yu, Z.-X.; Liu, Y.; Li, J.-F. Synthesis and transport property of Cu1.8S as a promising thermoelectric compound. Chem. Commun. 2011, 47, 12697–12699. [Google Scholar] [CrossRef] [PubMed]
- Mott, D.; Yin, J.; Engelhard, M.; Loukrakpam, R.; Chang, P.; Miller, G.; Bae, I.-T.; Das, N.C.; Wang, C.; Luo, J.; et al. From ultrafine thiolate-capped copper nanoclusters toward copper sulfide nanodiscs: A thermally activated evolution route. Chem. Mater. 2010, 22, 261–271. [Google Scholar] [CrossRef]
- Li, S.; Wang, H.Z.; Xu, W.W.; Si, H.L.; Tao, X.J.; Lou, S.; Du, Z.; Li, L.S. Synthesis and assembly of monodisperse spherical Cu2S nanocrystals. J. Colloid Interf. Sci. 2009, 330, 483–487. [Google Scholar] [CrossRef] [PubMed]
- Suekuni, K.; Tomizawa, Y.; Ozaki, T.; Koyano, M. Systematic study of electronic and magnetic properties for Cu12–xTMxSb4S13 (TM = Mn, Fe, Co, Ni, and Zn) tetrahedrite. J. Appl. Phys. 2014, 115. [Google Scholar] [CrossRef]
- Sigman, M.B.; Ghezelbash, A.; Hanrath, T.; Saunders, A.E.; Lee, F.; Korgel, B.A. Solventless synthesis of monodisperse Cu2S nanorods, nanodisks, and nanoplatelets. J. Am. Chem. Soc. 2003, 125, 16050–16057. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, Z.; Peng, Q.; Zhang, B.; Li, Y. Controllable synthesis of Cu2S nanocrystals and their assembly into a superlattice. J. Am. Chem. Soc. 2008, 130, 10482–10483. [Google Scholar] [CrossRef] [PubMed]
- Mansour, B.A. Electrical and thermoelectric properties of in and Cd doped Cu1.8S. Phys. Status Solidi A 1993, 136, 153–159. [Google Scholar] [CrossRef]
- Nath, S.K.; Kalita, P.K. Chemical synthesis of copper sulfide nanoparticles embedded in PVA matrix. Nanosci. Nanotechnol. Int. J. 2012, 2, 8–12. [Google Scholar]
- Kumar, P.; Gusain, M.; Kumar, P.S.; Uma, S.; Nagarajan, R. A simple one pot synthesis of cubic Cu5FeS4. RSC Adv. 2014, 4, 52633–52636. [Google Scholar] [CrossRef]
- Kumar, P.; Uma, S.; Nagarajan, R. Precursor driven one pot synthesis of wurtzite and chalcopyrite CuFeS2. Chem. Commun. 2013, 49, 7316–7318. [Google Scholar] [CrossRef] [PubMed]
- Reference pattern accessed from the International Centre for Diffraction Data database 2013, card number 00-001-0842 for tetragonal copper iron disulfide, card number 01-073-1667 for cubic Cu5FeS4 and card number 00-004-0861 for cubic Cu1.8S.
- Li, J.; Tan, Q.; Li, J.F. Synthesis and property evaluation of CuFeS2x as earth-abundant and environmentally-friendly thermoelectric materials. J. Alloy. Compd. 2013, 551, 143–149. [Google Scholar] [CrossRef]
- Verma, S.; Singh, M.; Ahuja, D.; Shimose, H.; Nishino, S.; Miyata, M.; Mott, D.; Koyano, M.; Maenosono, S. Chalcopyrite nanocomposite material for sustainable thermoelectrics. Jpn. J. Appl. Phys. 2014, 53. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Singh, M.; Miyata, M.; Nishino, S.; Mott, D.; Koyano, M.; Maenosono, S. Chalcopyrite Nanoparticles as a Sustainable Thermoelectric Material. Nanomaterials 2015, 5, 1820-1830. https://doi.org/10.3390/nano5041820
Singh M, Miyata M, Nishino S, Mott D, Koyano M, Maenosono S. Chalcopyrite Nanoparticles as a Sustainable Thermoelectric Material. Nanomaterials. 2015; 5(4):1820-1830. https://doi.org/10.3390/nano5041820
Chicago/Turabian StyleSingh, Maninder, Masanobu Miyata, Shunsuke Nishino, Derrick Mott, Mikio Koyano, and Shinya Maenosono. 2015. "Chalcopyrite Nanoparticles as a Sustainable Thermoelectric Material" Nanomaterials 5, no. 4: 1820-1830. https://doi.org/10.3390/nano5041820
APA StyleSingh, M., Miyata, M., Nishino, S., Mott, D., Koyano, M., & Maenosono, S. (2015). Chalcopyrite Nanoparticles as a Sustainable Thermoelectric Material. Nanomaterials, 5(4), 1820-1830. https://doi.org/10.3390/nano5041820







