Organic Phase Change Nanoparticles for in-Product Labeling of Agrochemicals
Abstract
:1. Introduction
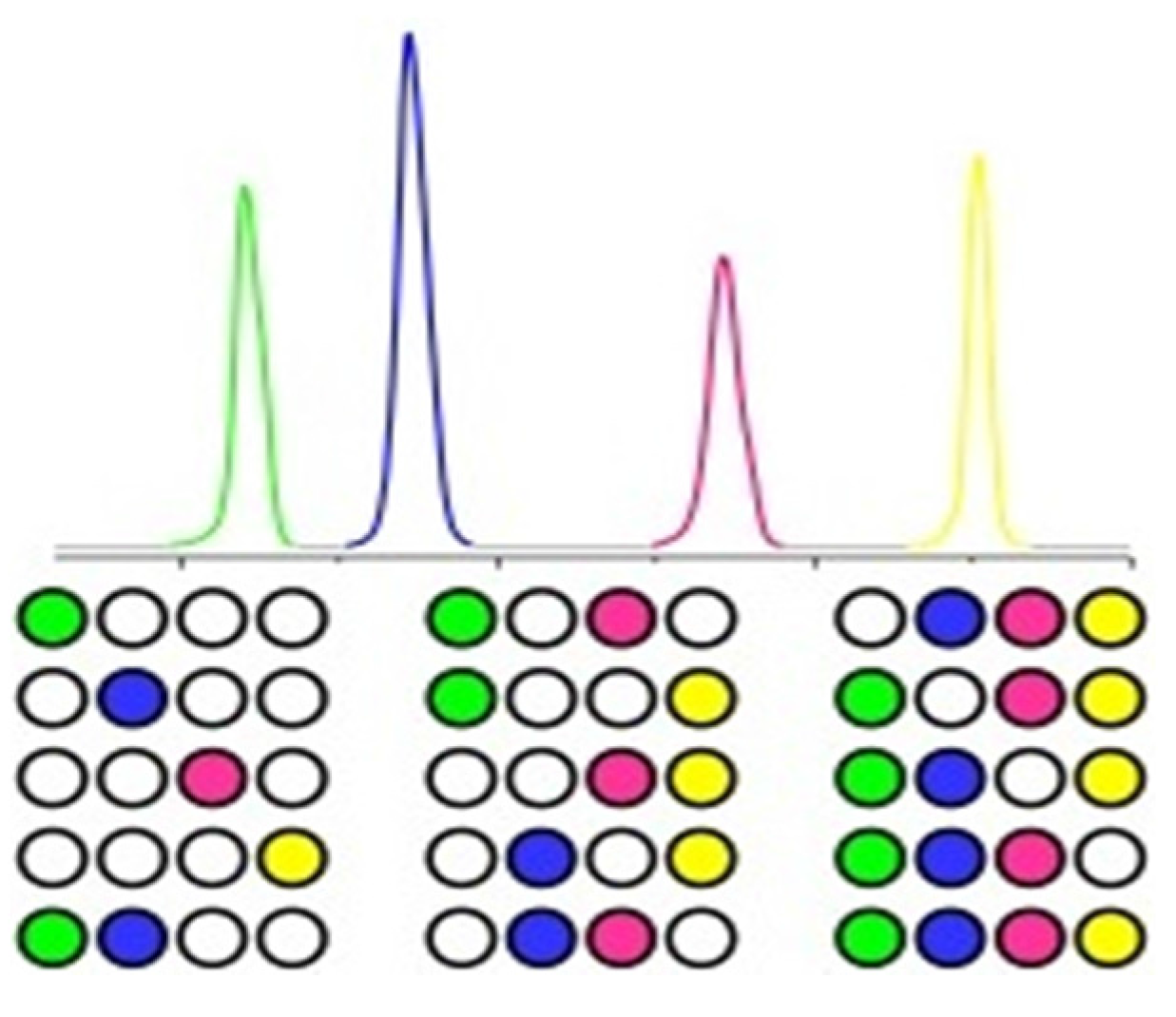
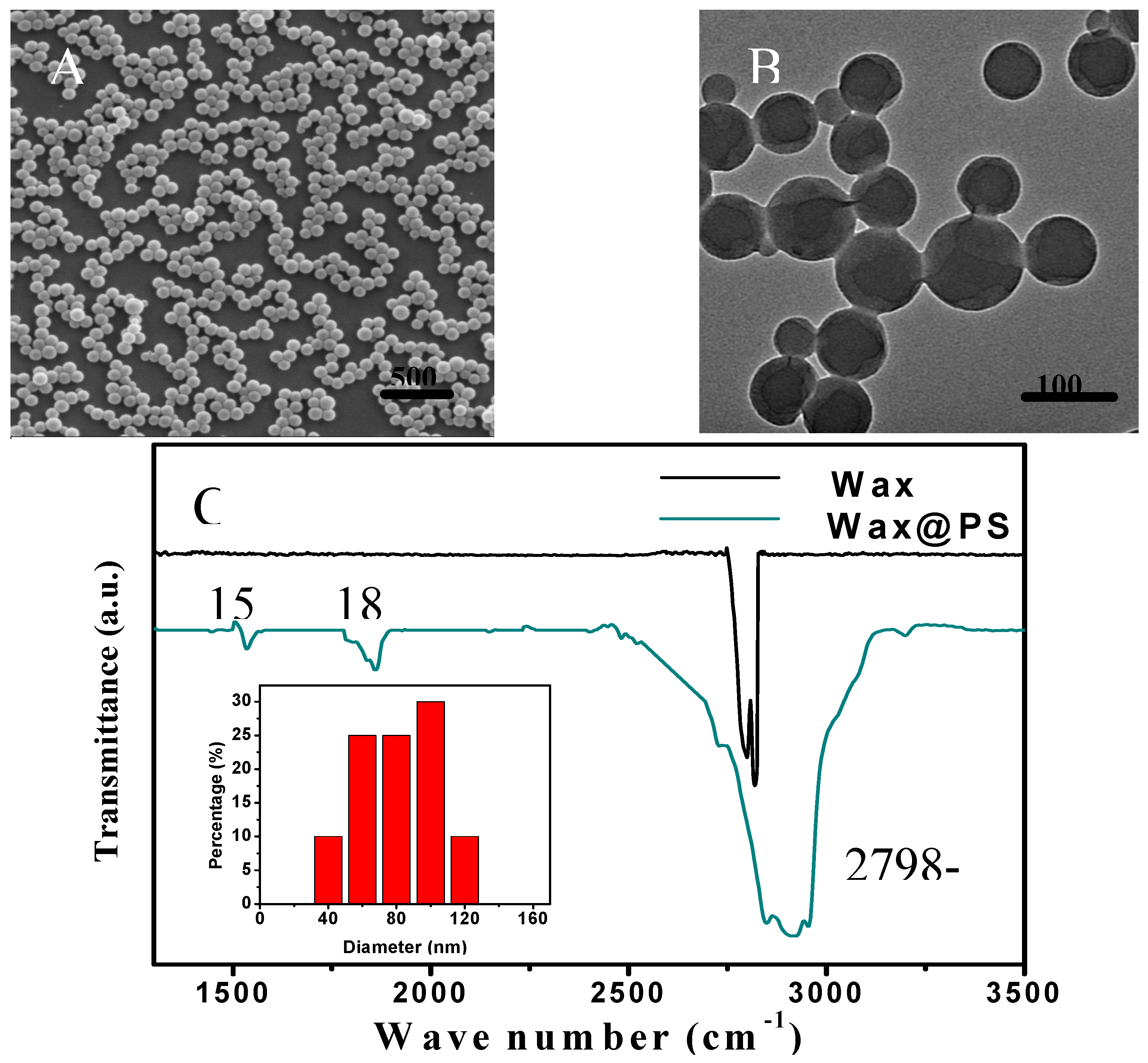
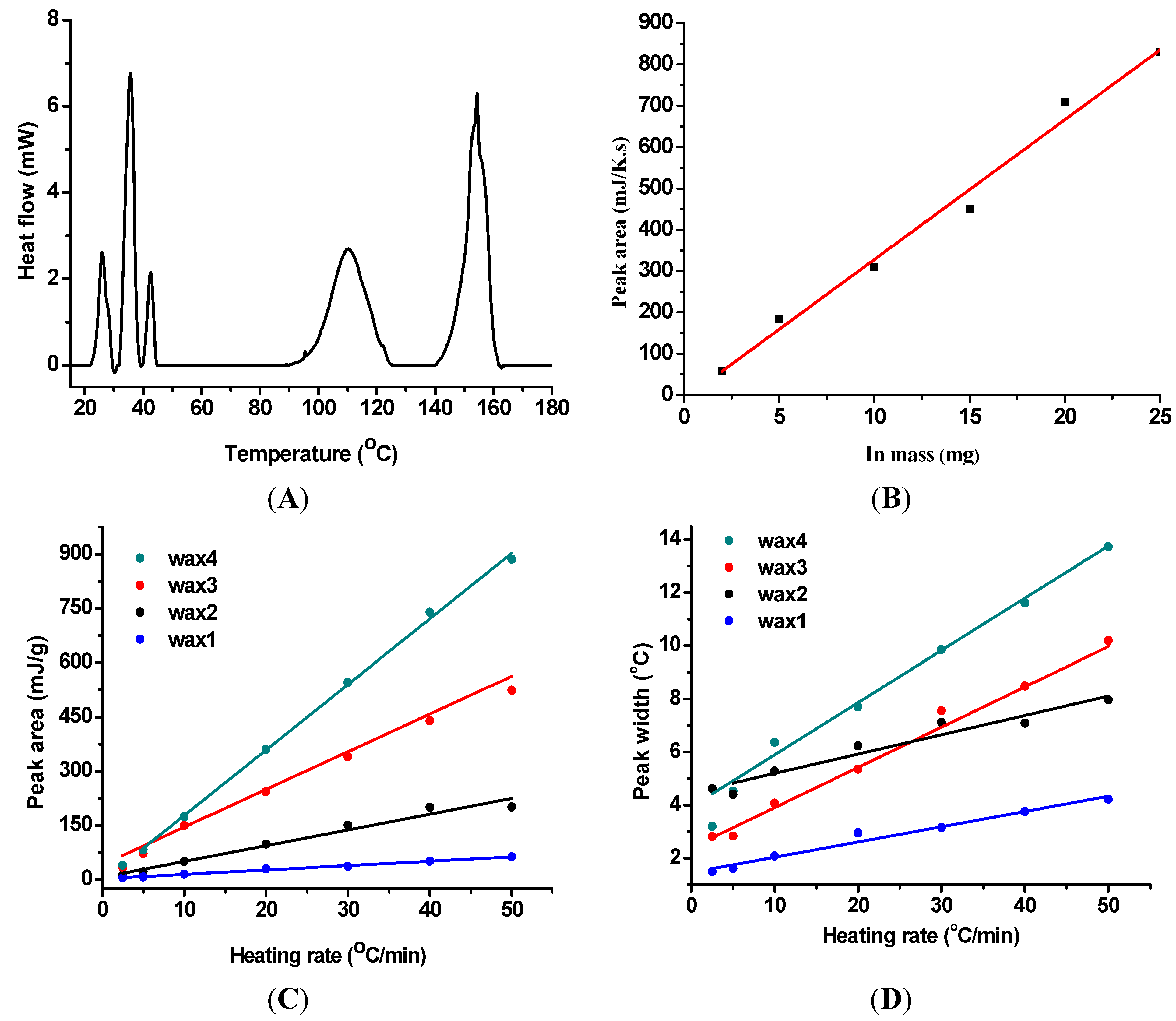
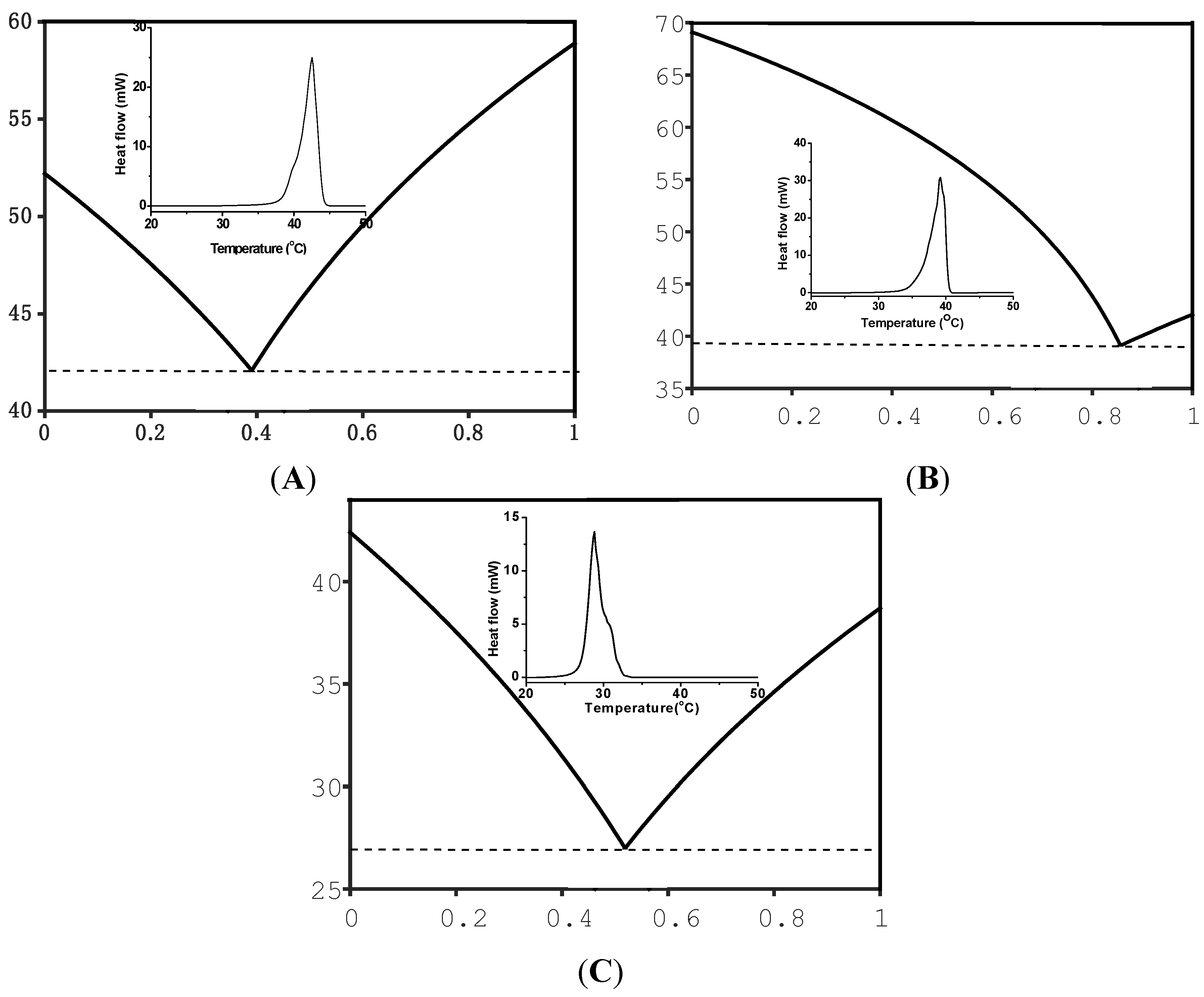
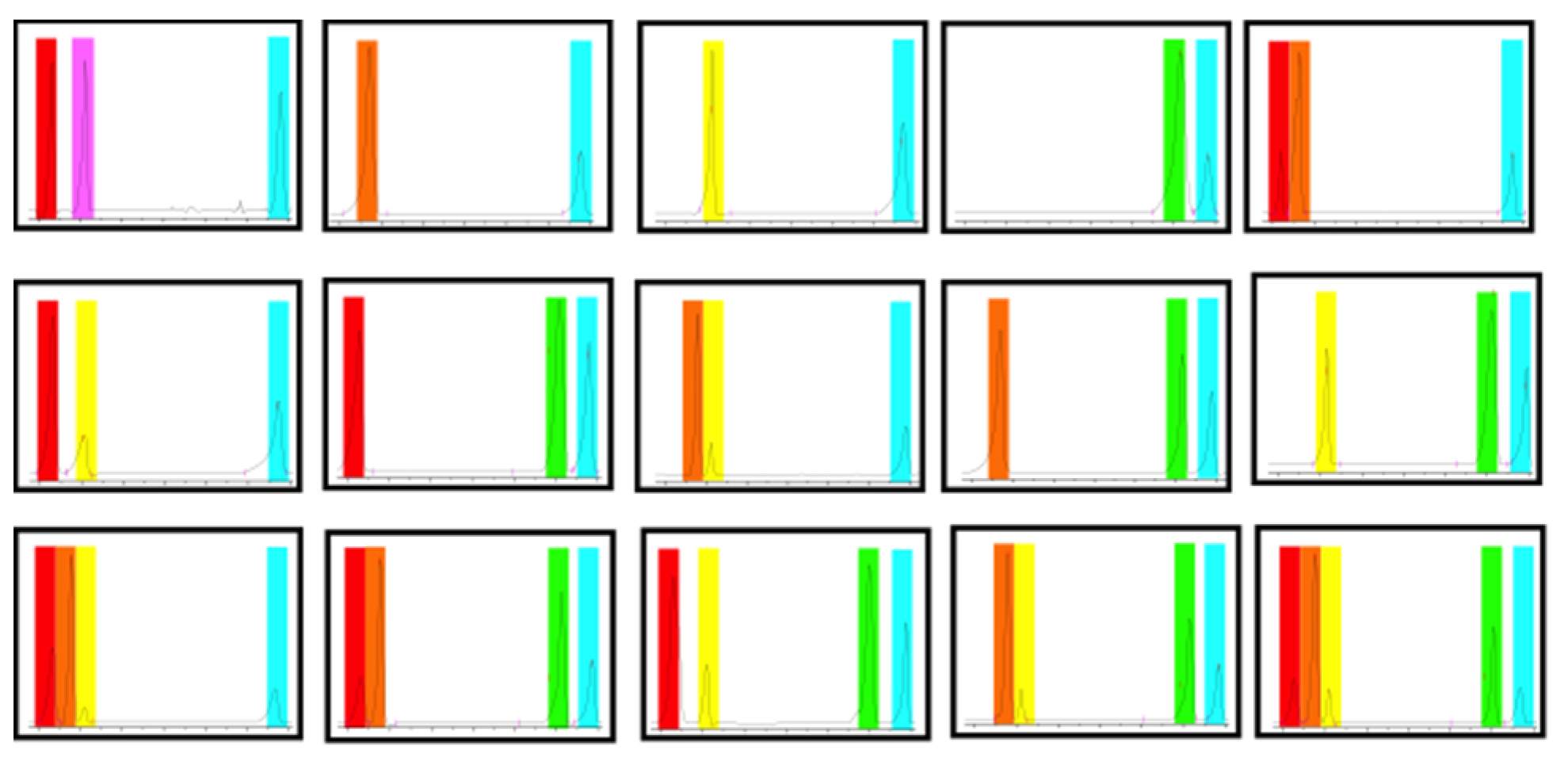
2. Results and Discussion
3. Experimental Section
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Weller, P.; Boner, M.; Foerstel, H.; Becker, H.; Peikert, B.; Dreher, W. Isotopic fingerprinting for the authenticity control of crop protection active compounds using the representative insecticide fipronil. J. Agric. Food Chem. 2011, 59, 4365–4370. [Google Scholar] [CrossRef]
- Karasali, H.; Kasiotis, K.M.; Machera, K.; Ambrus, A. Case study to illustrate an approach for detecting contamination and impurities in pesticide formulations. J. Agric. Food Chem. 2014, 62, 11347–11352. [Google Scholar] [CrossRef] [PubMed]
- Deisingh, A.K. Pharmaceutical counterfeiting. Analyst 2005, 130, 271–279. [Google Scholar] [CrossRef] [PubMed]
- Perr, J.M.; Furton, K.G.; Almirall, J.R. Solid phase microextraction ion mobility spectrometer interface for explosive and taggant detection. J. Sep. Sci. 2005, 28, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Duong, B.; Liu, H.; Li, C.; Deng, W.; Ma, L.; Su, M. Printed multilayer microtaggants with phase change nanoparticles for enhanced labeling security. ACS Appl. Mater. Interface 2014, 6, 8909–8912. [Google Scholar] [CrossRef] [PubMed]
- Abad, E.; Palacio, F.; Nuin, M.; de Zarate, A.G.; Juarros, A.; Gómez, J.; Marco, S. RFID smart tag for traceability and cold chain monitoring of foods: Demonstration in an intercontinental fresh fish logistic chain. J. Food Eng. 2009, 93, 394–399. [Google Scholar] [CrossRef]
- Sanchez-Romaguera, V.; Ziai, M.A.; Oyeka, D.; Barbosa, S.; Wheeler, J.S.; Batchelor, J.C.; Parker, E.A.; Yeates, S.G. Towards inkjet-printed low cost passive UHF RFID skin mounted tattoo paper tags based on silver nanoparticle inks. J. Mater. Chem. C 2013, 1, 6395–6402. [Google Scholar] [CrossRef] [Green Version]
- Maind, S.; Kumar, S.; Chattopadhyay, N.; Gandhi, C.; Sudersanan, M. Analysis of Indian blue ballpoint pen inks tagged with rare-earth thenoyltrifluoroacetonates by inductively coupled plasma-mass spectrometry and instrumental neutron activation analysis. Forensic Sci. Int. 2006, 159, 32–42. [Google Scholar] [CrossRef] [PubMed]
- Thomas, S.W., III; Amara, J.P.; Bjork, R.E.; Swager, T.M. Amplifying fluorescent polymer sensors for the explosives taggant 2,3-dimethyl-2,3-dinitrobutane (DMNB). Chem. Comm. 2005, 36, 4572–4574. [Google Scholar] [CrossRef] [PubMed]
- Naddo, T.; Yang, X.; Moore, J.S.; Zang, L. Highly responsive fluorescent sensing of explosives taggant with an organic nanofibril film. Sensors Actuat. B 2008, 134, 287–291. [Google Scholar] [CrossRef]
- Huang, C.; Lucas, B.; Vervaet, C.; Braeckmans, K.; van Calenbergh, S.; Karalic, I.; Vandewoestyne, M.; Deforce, D.; Demeester, J.; de Smedt, S.C. Unbreakable codes in electrospun fibers: Digitally encoded polymers to stop medicine counterfeiting. Adv. Mater. 2010, 22, 2657–2662. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Bae, H.J.; Kim, J.; Shin, S.; Choi, S.E.; Lee, S.H.; Kwon, S.; Park, W. Lithographically encoded polymer microtaggant using high-capacity and error-correctable QR code for anti-counterfeiting of drugs. Adv. Mater. 2012, 24, 5924–5929. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Hong, Y.; Zhang, M.; Su, M. Encoding and decoding nanoscale thermal barcodes for ultrahigh capacity identification systems. Appl. Phys. Lett. 2009, 95, 233101. [Google Scholar] [CrossRef]
- Wang, M.; Kong, T.; Jing, X.; Hung, Y.-K.; Sun, D.; Lin, L.; Zheng, Y.; Huang, J.; Li, Q. Fabrication of Au nanowire/Pichia pastoris cell composites with hexadecyltrimethylammonium bromides as a platform for SERS detection: A microorganism-mediated approach. Ind. Eng. Chem. Res. 2012, 51, 16651–16659. [Google Scholar] [CrossRef]
- Huang, M.H.; Mao, S.; Feick, H.; Yan, H.; Wu, Y.; Kind, H.; Weber, E.; Russo, R.; Yang, P. Room-temperature ultraviolet nanowire nanolasers. Science 2001, 292, 1897–1899. [Google Scholar] [CrossRef] [PubMed]
- Treacy, M.J.; Ebbesen, T.; Gibson, J. Exceptionally high Young’s modulus observed for individual carbon nanotubes. Nature 1996, 381, 678–680. [Google Scholar] [CrossRef]
- Alivisatos, P. The use of nanocrystals in biological detection. Nat. Biotechnol. 2004, 22, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.C.; Jin, R.; Mirkin, C.A. Nanoparticles with Raman spectroscopic fingerprints for DNA and RNA detection. Science 2002, 297, 1536–1540. [Google Scholar] [CrossRef] [PubMed]
- Elghanian, R.; Storhoff, J.J.; Mucic, R.C.; Letsinger, R.L.; Mirkin, C.A. Selective colorimetric detection of polynucleotides based on the distance-dependent optical properties of gold nanoparticles. Science 1997, 277, 1078–1081. [Google Scholar] [CrossRef] [PubMed]
- Han, M.; Gao, X.; Su, J.Z.; Nie, S. Quantum-dot-tagged microbeads for multiplexed optical coding of biomolecules. Nat. Biotechnol. 2001, 19, 631–635. [Google Scholar] [CrossRef] [PubMed]
- Freeman, R.; Willner, I. NAD+/NADH-sensitive quantum dots: Applications to probe NAD+-dependent enzymes and to sense the RDX explosive. Nano Lett. 2008, 9, 322–326. [Google Scholar] [CrossRef] [PubMed]
- Klimov, V.; Mikhailovsky, A.; Xu, S.; Malko, A.; Hollingsworth, J.; Leatherdale, C.; Eisler, H.-J.; Bawendi, M. Optical gain and stimulated emission in nanocrystal quantum dots. Science 2000, 290, 314–317. [Google Scholar] [CrossRef] [PubMed]
- Nie, S.; Emory, S.R. Probing single molecules and single nanoparticles by surface-enhanced Raman scattering. Science 1997, 275, 1102–1106. [Google Scholar] [CrossRef] [PubMed]
- Nicewarner-Pena, S.R.; Freeman, R.G.; Reiss, B.D.; He, L.; Peña, D.J.; Walton, I.D.; Cromer, R.; Keating, C.D.; Natan, M.J. Submicrometer metallic barcodes. Science 2001, 294, 137–141. [Google Scholar] [CrossRef] [PubMed]
- Davis, J. Microbial decomposition of hydrocarbons. Ind. Eng. Chem. 1956, 48, 1444–1448. [Google Scholar] [CrossRef]
- Jiang, H.; Moon, K.; Dong, H.; Hua, F.; Wong, C. Size-dependent melting properties of tin nanoparticles. Chem. Phys. Lett. 2006, 429, 492–496. [Google Scholar] [CrossRef]
- Duong, B.; Liu, H.; Ma, L.; Su, M. Covert thermal barcodes based on phase change nanoparticles. Sci. Rep. 2014, 4, 5170–5175. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.; Ding, S.; Wu, W.; Hu, J.; Voevodin, A.A.; Gschwender, L.; Snyder, E.; Chow, L.; Su, M. Enhancing heat capacity of colloidal suspension using nanoscale encapsulated phase-change materials for heat transfer. ACS Appl. Mater. Interface 2010, 2, 1685–1691. [Google Scholar] [CrossRef] [PubMed]
- Müller, B. Influence of experimental variables on curves in differential scanning calorimetry. Part I. Study design and results of calibration checks. Thermochim. Acta 1981, 49, 151–161. [Google Scholar]
- Rubin, G.; Finel, A. A review on phase change energy storage: Materials and applications. J. Phys.Condens. Mater. 1993, 5, 9105. [Google Scholar] [CrossRef]
- Farid, M.M.; Khudhair, A.M.; Razack, S.A.K.; Al-Hallaj, S. A review on phase change energy storage: Materials and applications. Energ. Convers. Manag. 2004, 45, 1597–1615. [Google Scholar] [CrossRef]
- Yuan, Y.; Tao, W.; Cao, X.; Li, B. Theoretic prediction of melting temperature and latent heat for a fatty acid eutectic mixture. J. Chem. Eng. Data 2011, 56, 2889–2891. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, M.; Duong, B.; Su, M. Organic Phase Change Nanoparticles for in-Product Labeling of Agrochemicals. Nanomaterials 2015, 5, 1810-1819. https://doi.org/10.3390/nano5041810
Wang M, Duong B, Su M. Organic Phase Change Nanoparticles for in-Product Labeling of Agrochemicals. Nanomaterials. 2015; 5(4):1810-1819. https://doi.org/10.3390/nano5041810
Chicago/Turabian StyleWang, Miao, Binh Duong, and Ming Su. 2015. "Organic Phase Change Nanoparticles for in-Product Labeling of Agrochemicals" Nanomaterials 5, no. 4: 1810-1819. https://doi.org/10.3390/nano5041810
APA StyleWang, M., Duong, B., & Su, M. (2015). Organic Phase Change Nanoparticles for in-Product Labeling of Agrochemicals. Nanomaterials, 5(4), 1810-1819. https://doi.org/10.3390/nano5041810




