Dendrimer-Functionalized Laponite Nanodisks as a Platform for Anticancer Drug Delivery
Abstract
:1. Introduction
2. Results and Discussion
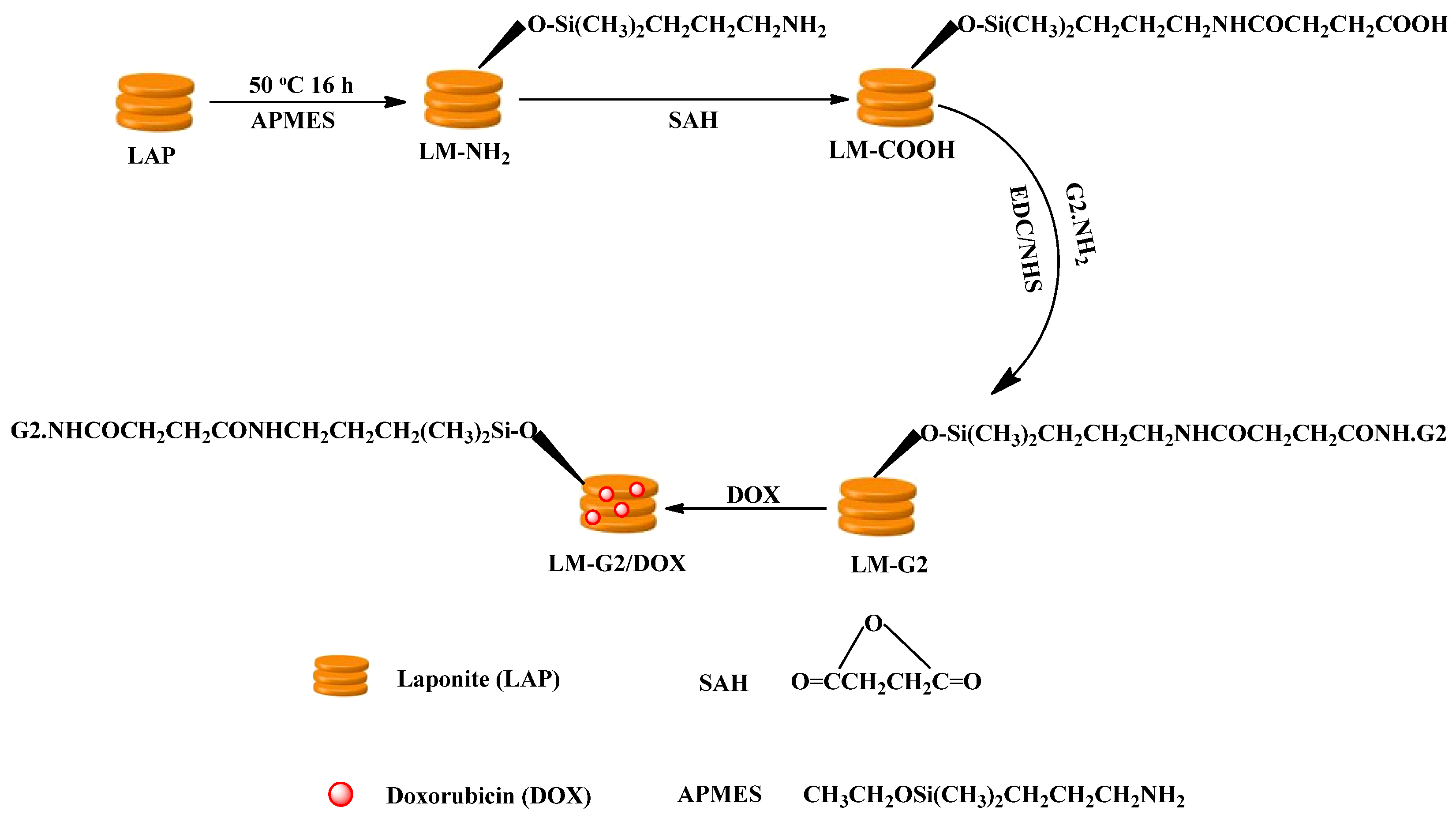
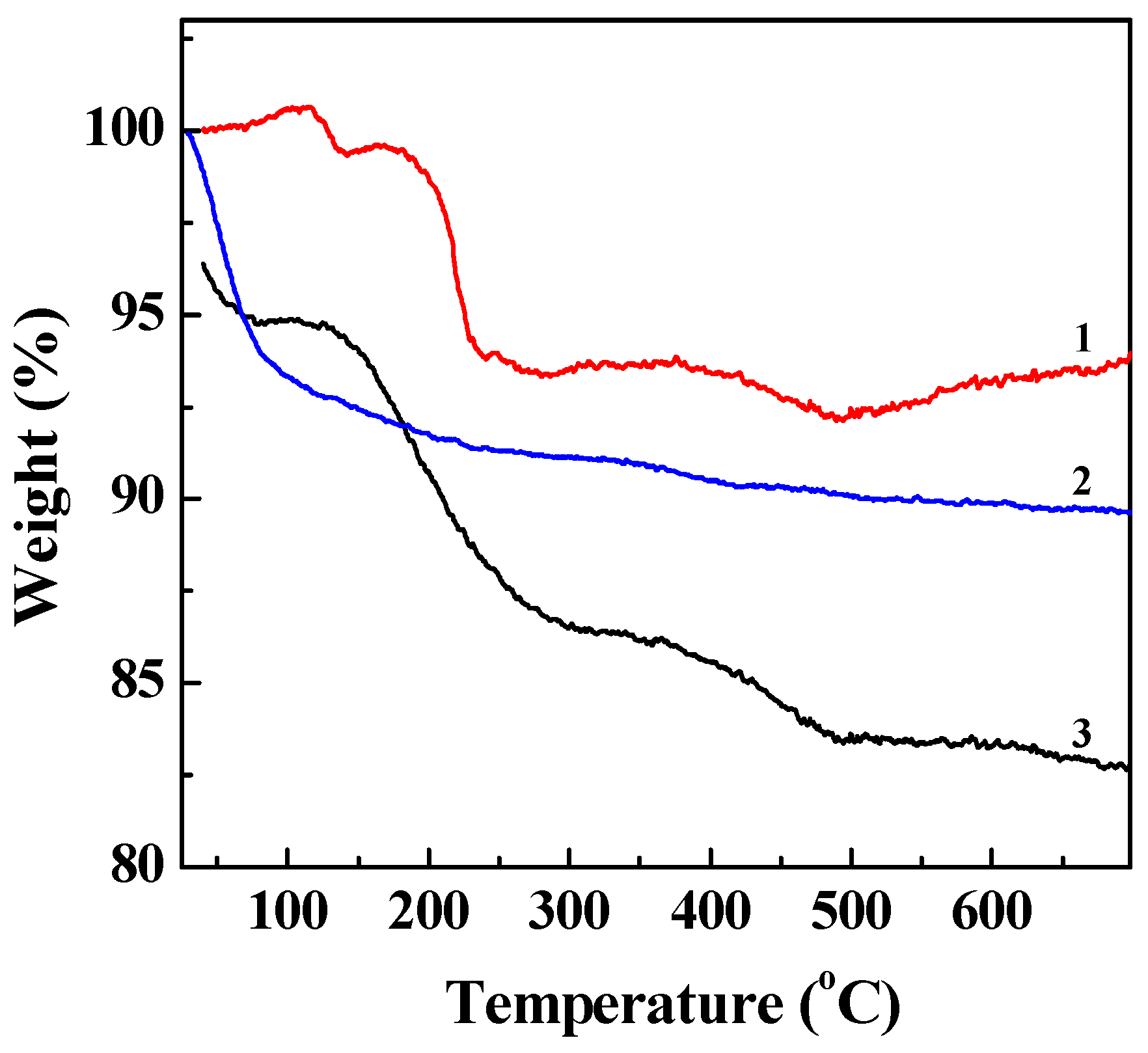
2.1. Synthesis and Characterization of LM-G2
| Materials | Zeta Potential (mV) | Hydrodynamic Size (nm) |
|---|---|---|
| LAP | −32.5 ± 0.5 | 74 ± 8.9 |
| LM-NH2 | −18.6 ± 1.8 | 225 ± 12.7 |
| LM-COOH | −19.6 ± 2.5 | 566 ± 5.3 |
| LM-G2 | +5.8 ± 0.5 | 869 ±18.2 |

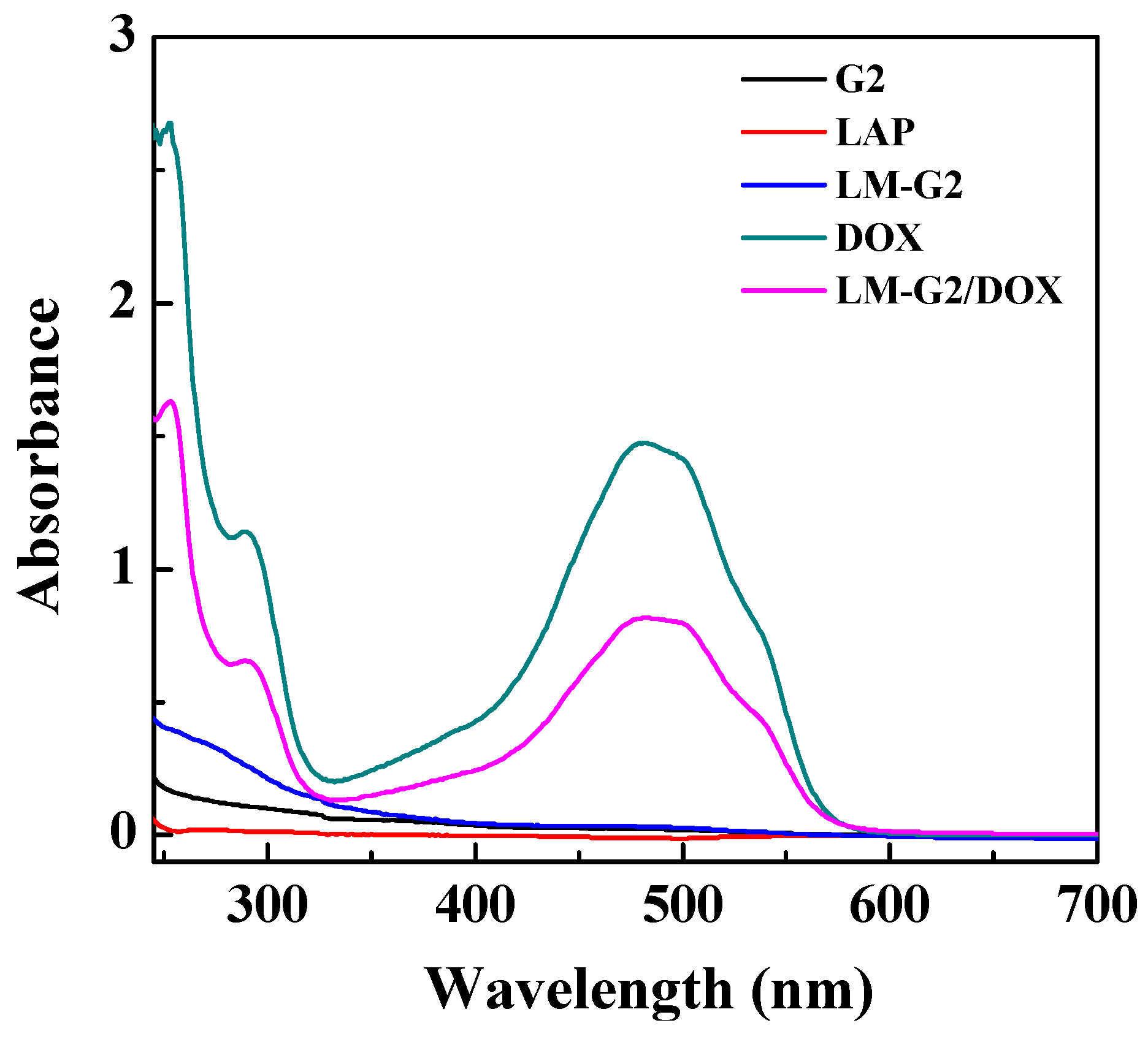
2.2. Loading Efficiency of DOX into LM-G2 Nanodisks
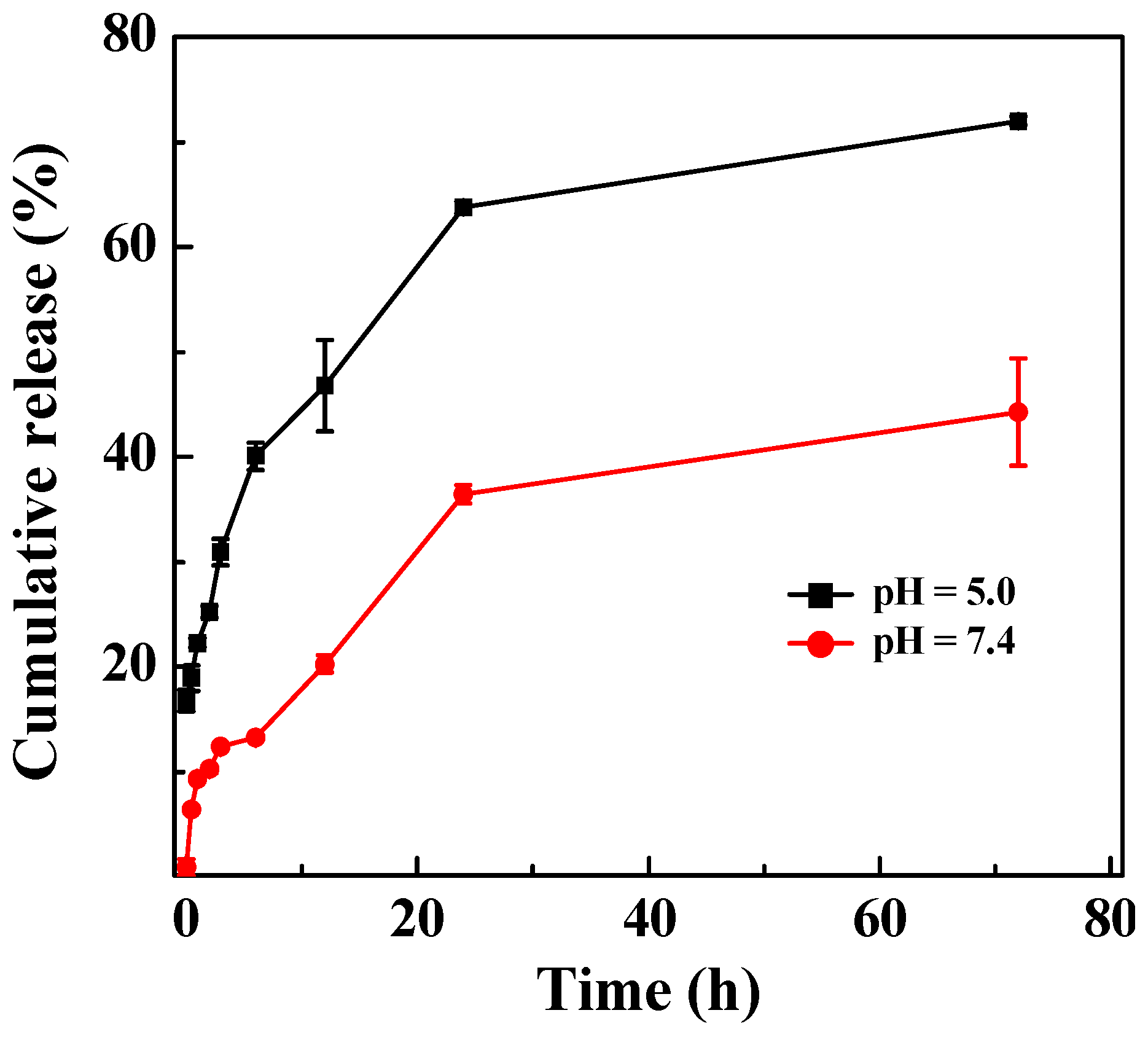
2.3. Release Profile of DOX from LM-G2/DOX Complexes
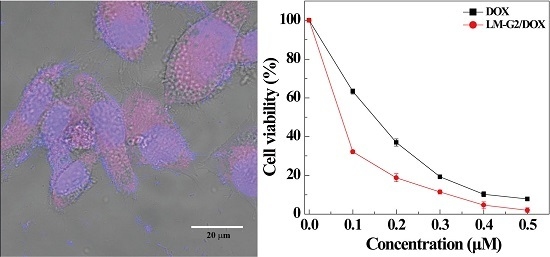
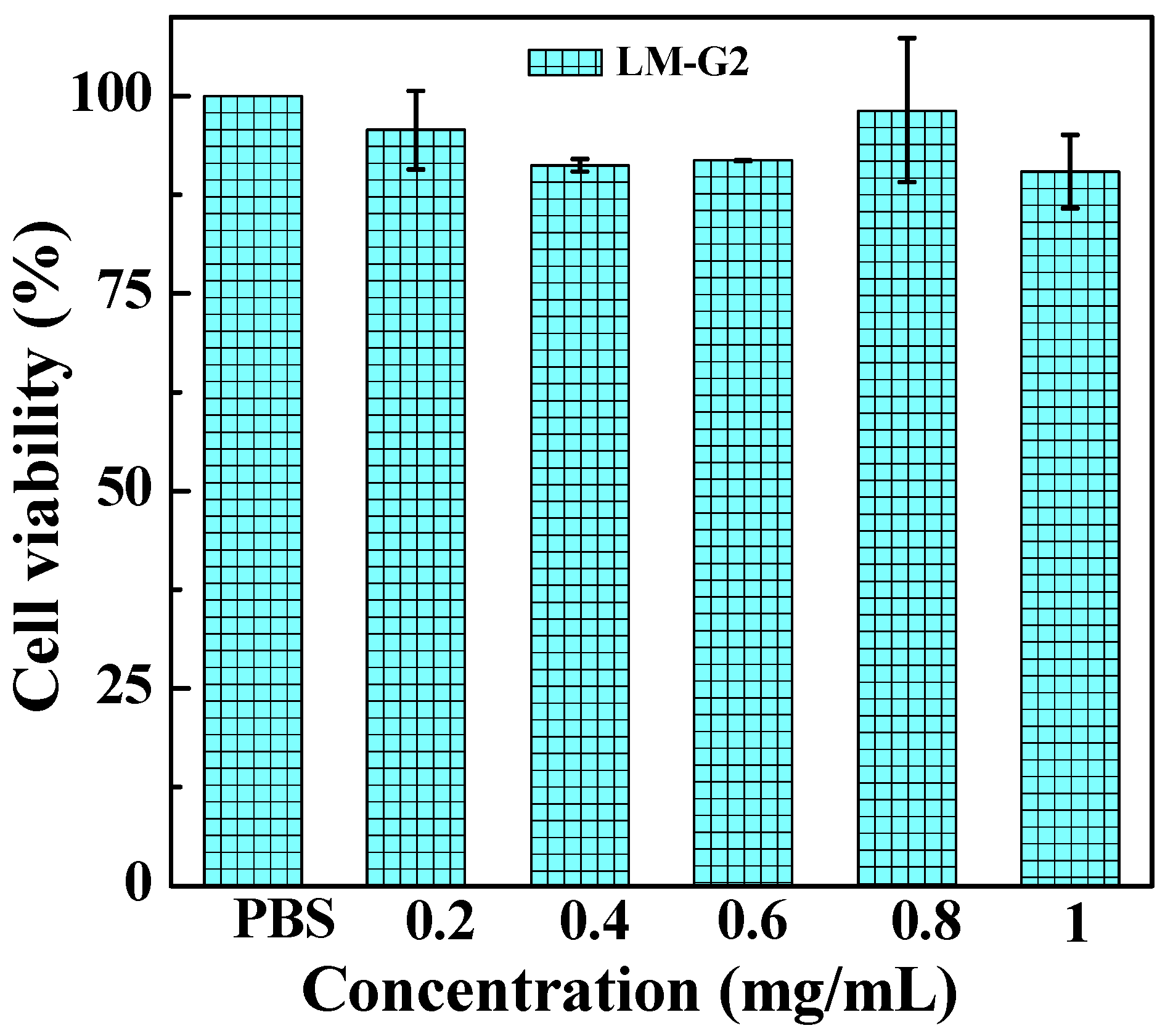
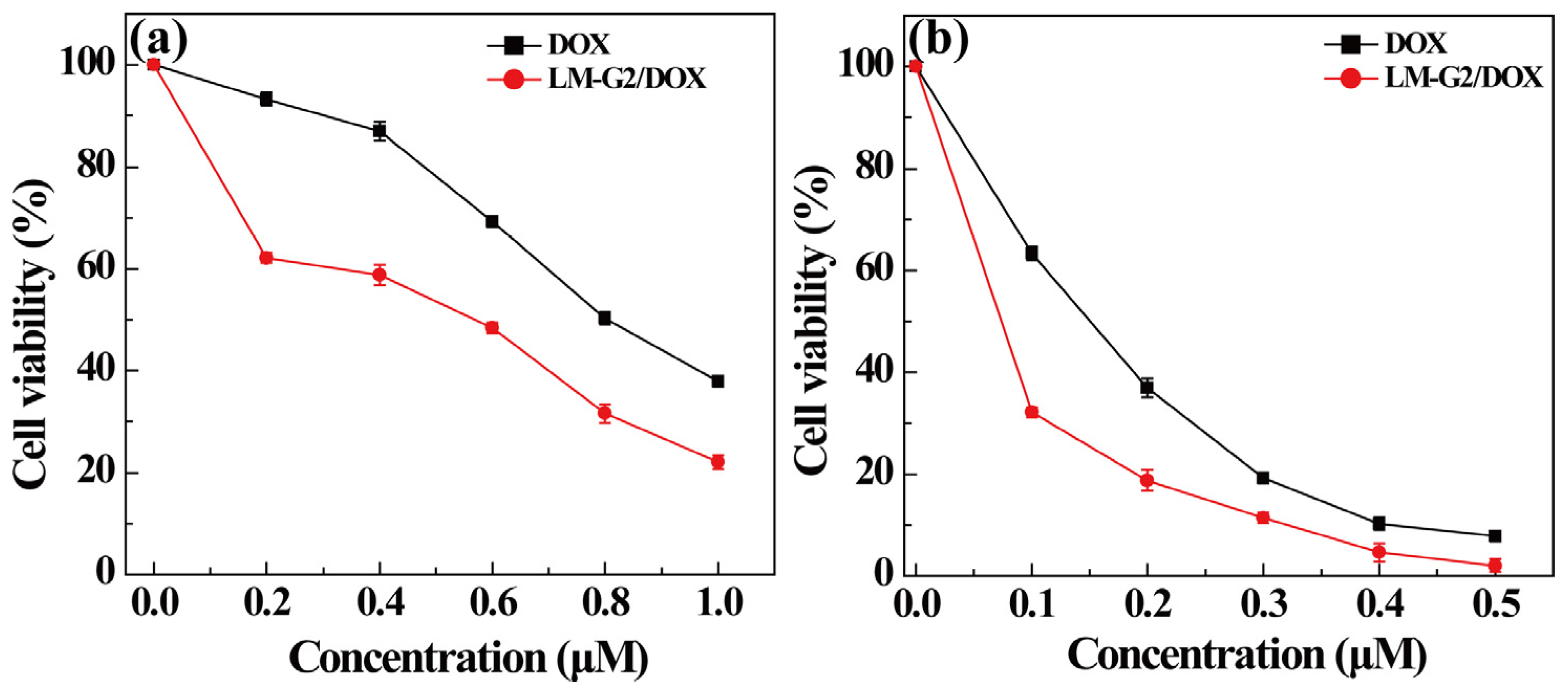
2.4. Antitumor Efficacy of the LM-G2/DOX Complexes
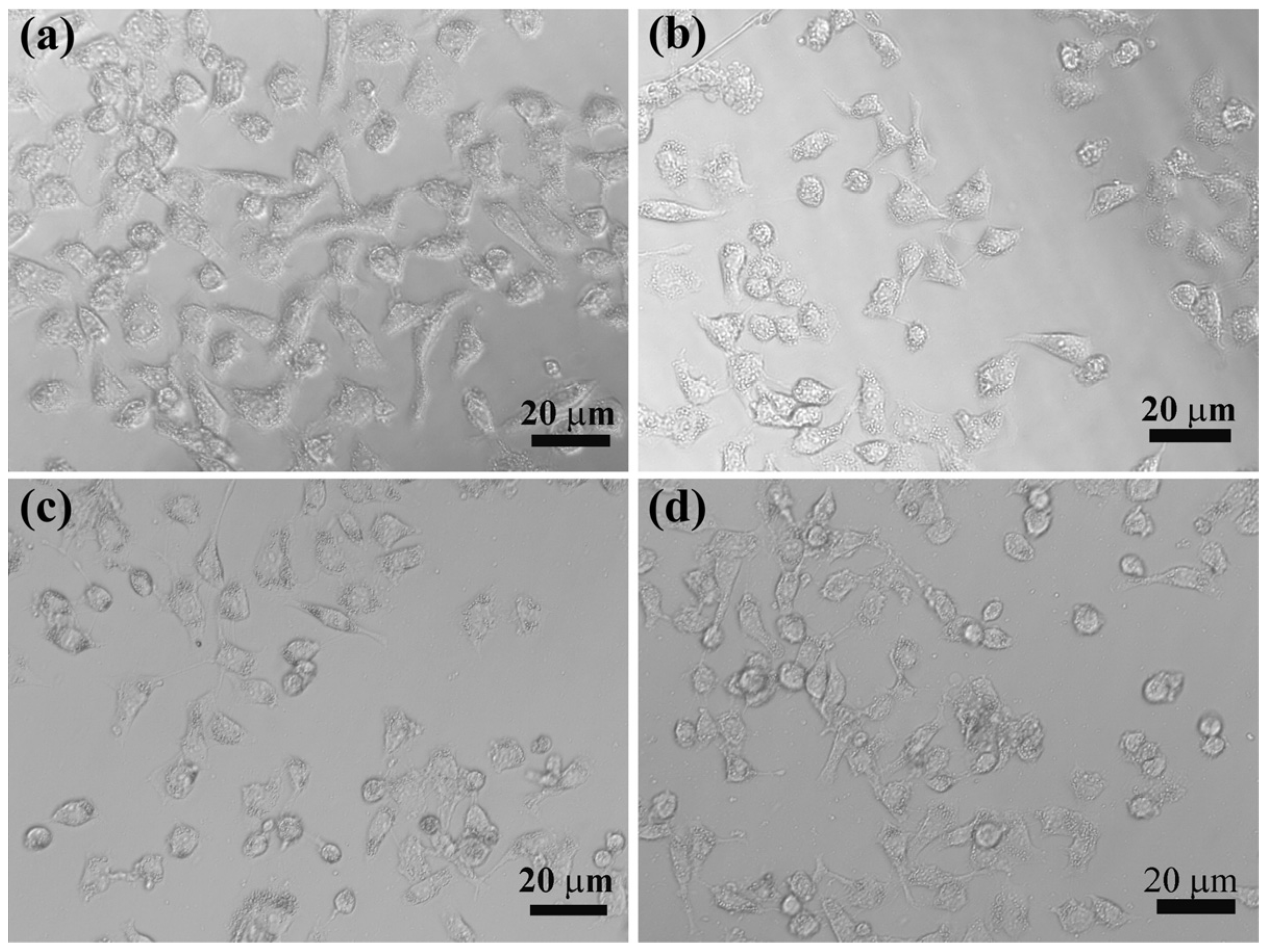
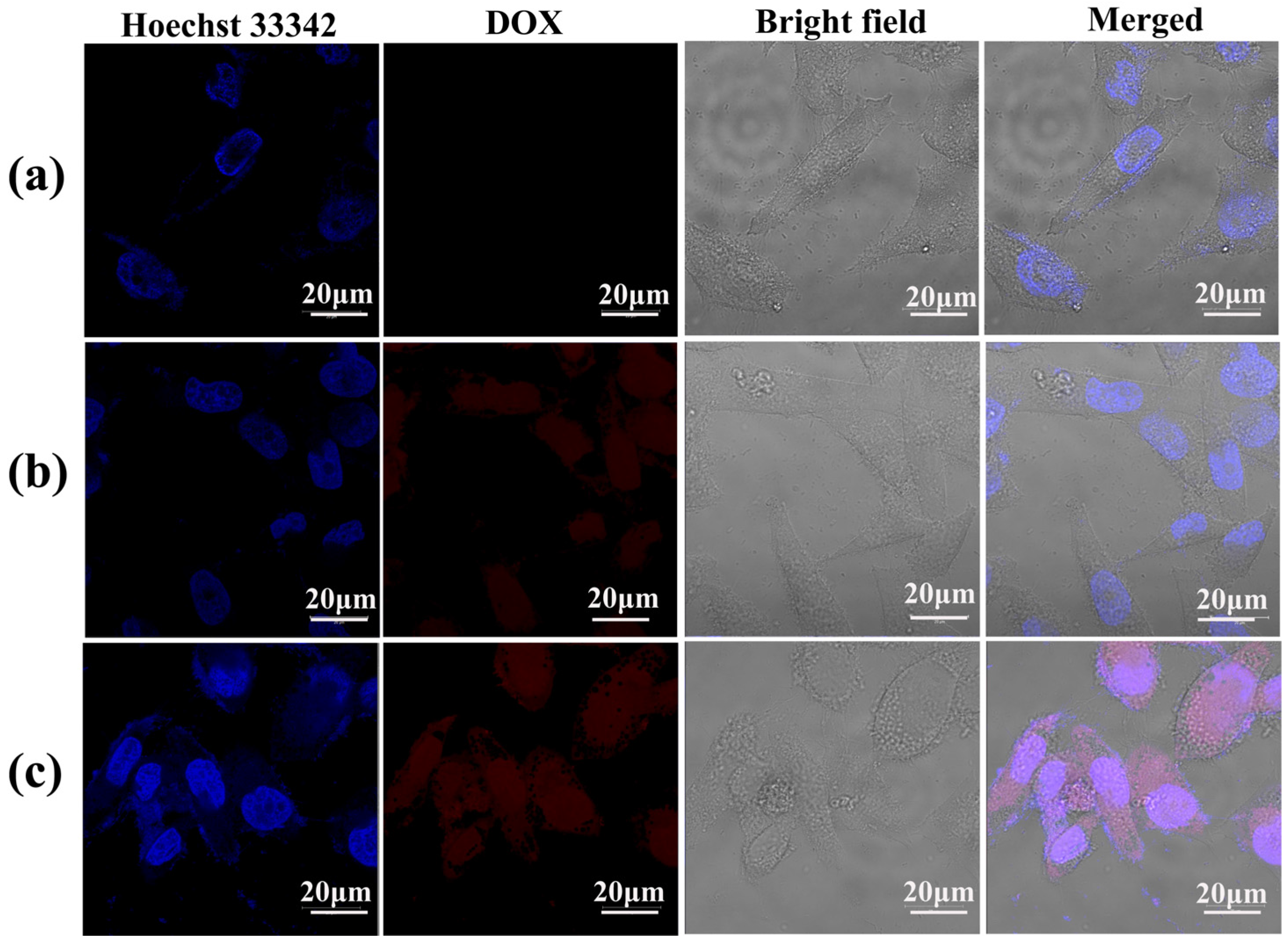
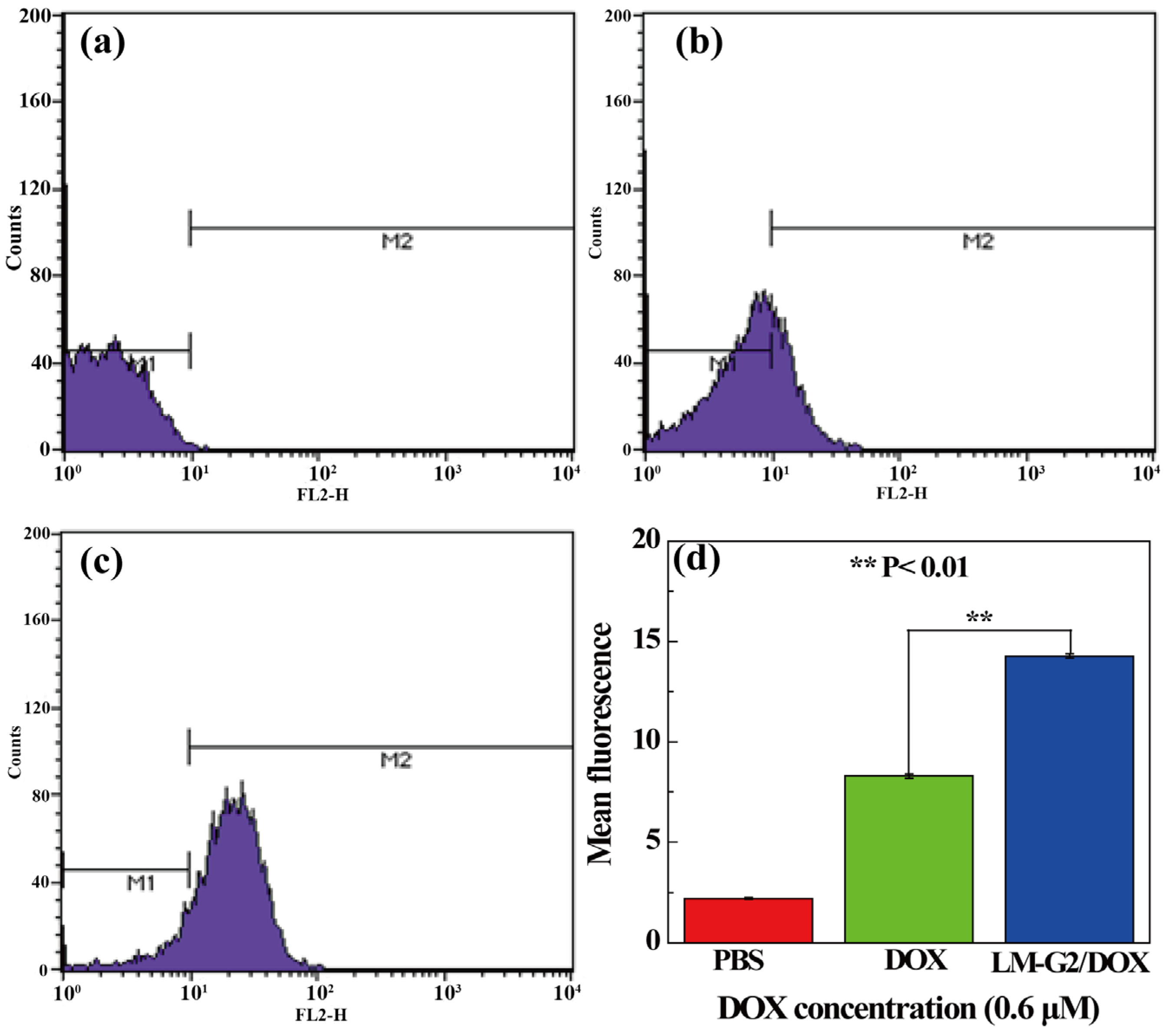
2.5. Intracellular Uptake of LM-G2/DOX Complexes
3. Experimental Section
3.1. Materials
3.2. Synthesis of LM-G2 Nanodisks
3.3. Characterization
3.4. Loading of DOX within the LM-G2 Nanodisks
3.5. In Vitro Drug Release Kinetics
3.6. Cell Culture and Cytotoxicity Assay
3.7. FCM Analysis
3.8. CLSM Imaging
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Powell, J.; McConkey, C.C. The rising trend in oesophageal adenocarcinoma and gastric cardia. Eur. J. Cancer Prev. 1992, 1, 265–269. [Google Scholar] [CrossRef] [PubMed]
- Peer, D.; Karp, J.M.; Hong, S.P.; FaroKhzad, O.C.; Margalit, R.; Langer, R. Nanocarriers as an emerging platform for cancer therapy. Nat. Nanotechnol. 2007, 2, 751–760. [Google Scholar] [CrossRef] [PubMed]
- Maeda, H.; Bharate, G.Y.; Daruwalla, J. Polymeric drugs for efficient tumor-targeted drug delivery based on EPR-effect. Eur. J. Pharm. Biopharm. 2009, 71, 409–419. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Cao, X.; Guo, R.; Shen, M.; Zhang, M.; Zhu, M.; Shi, X. Targeted delivery of doxorubicin into cancer cells using a folic acid-dendrimer conjugate. Polym. Chem. 2011, 2, 1754–1760. [Google Scholar] [CrossRef]
- Liu, Z.; Fan, A.C.; Rakhra, K.; Sherlock, S.; Goodwin, A.; Chen, X.; Yang, Q.; Felsher, D.W.; Dai, H. Supramolecular stacking of doxorubicin on carbon nanotubes for in vivo cancer therapy. Angew. Chem. Int. Ed. 2009, 48, 7668–7672. [Google Scholar] [CrossRef] [PubMed]
- Perche, F.; Patel, N.R.; Torchilin, V.P. Accumulation and toxicity of antibody-targeted doxorubicin-loaded PEG-PE micelles in ovarian cancer cell spheroid model. J. Control. Release 2012, 164, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Szebeni, J.; Bedocs, P.; Urbanics, R.; Buenger, R.; Rosivall, L.; Toth, M.; Barenholz, Y. Prevention of infusion reactions to pegylated liposomal doxorubicin via tachyphylaxis induction by placebo vesicles: A porcine model. J. Control. Release 2012, 160, 382–387. [Google Scholar] [CrossRef] [PubMed]
- Cuong, N.V.; Jiang, J.; Li, Y.; Chen, J.R.; Jwo, S.C.; Hsieh, M.F. Doxorubicin-loaded PEG-PCL-PEG micelle using xenograft model of nude mice: Effect of multiple administration of micelle on the suppression of human breast cancer. Cancers 2010, 3, 61–78. [Google Scholar] [PubMed]
- Lee, C.C.; Gillies, E.R.; Fox, M.E.; Guillaudeu, S.J.; Frechet, J.M.J.; Dy, E.E.; Szoka, C. A single dose of doxorubicin-functionalized bow-tie dendrimer cures mice bearing C-26 colon carcinomas. Proc. Natl. Acad. Sci. USA 2006, 103, 16649–16654. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Zheng, F.; Huang, Y.; Fang, Y.; Shen, M.; Zhu, M.; Shi, X. Encapsulation of amoxicillin within laponite-doped poly(lactic-co-glycolic acid) nanofibers: Preparation, characterization, and antibacterial activity. ACS Appl. Mater. Interfaces 2012, 4, 6393–6401. [Google Scholar] [CrossRef] [PubMed]
- Jung, K.; Kim, H.-K.; Choy, Y.B.; Hwang, S.-J.; Choy, J.-H. Itraconazole-laponite: Kinetics and mechanism of drug release. Appl. Clay Sci. 2008, 40, 99–107. [Google Scholar] [CrossRef]
- Akane, O.; Arimitsu, U. Twenty years of polymer-clay nanocomposites. Macromol. Mater. Eng. 2006, 291, 1449–1476. [Google Scholar]
- Herrera, N.N.; Letoffe, J.M.; Reymond, J.P.; Bourgeat-Lami, E. Silylation of laponite clay particles with monofunctional and trifunctional vinyl alkoxysilanes. J. Mater. Chem. 2005, 15, 863–871. [Google Scholar] [CrossRef]
- Wang, S.; Wu, Y.; Guo, R.; Huang, Y.; Wen, S.; Shen, M.; Wang, J.; Shi, X. Laponite nanodisks as an efficient platform for doxorubicin delivery to cancer cells. Langmuir 2013, 29, 5030–5036. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Guo, R.; Wen, S.; Shen, M.; Zhu, M.; Wang, J.; Shi, X. Folic acid-modified laponite nanodisks for targeted anticancer drug delivery. J. Mater. Chem. B 2014, 2, 7410–7418. [Google Scholar] [CrossRef]
- Chen, G.; Li, D.; Li, J.; Cao, X.; Wang, J.; Shi, X.; Guo, R. Targeted doxorubicin delivery to hepatocarcinoma cells by lactobionic acid-modified laponite nanodisks. New J. Chem. 2015, 39, 2847–2855. [Google Scholar]
- Tomalia, D.; Baker, H.; Dewald, J.; Hall, M.; Kallos, G.; Martin, S.; Roeck, J.; Ryder, J.; Smith, P. A new class of polymers: Starburst-dendritic macromolecules. Polym. J. 1985, 17, 117–132. [Google Scholar] [CrossRef]
- Tomalia, D.A.; Frechet, J.M.J. Dendrimers and Other Dendritic Polymers; John Wiley & Sons: New York, NY, USA, 2001. [Google Scholar]
- Svenson, S. Dendrimers as versatile platform in drug delivery applications. Eur. J. Pharm. Biopharm. 2009, 71, 445–462. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Shi, X. Dendrimer-based nanodevices for targeted drug delivery applications. J. Mater. Chem. B 2013, 1, 4199–4211. [Google Scholar] [CrossRef]
- Shi, X.; Lee, I.; Chen, X.; Shen, M.; Xiao, S.; Zhu, M.; Baker, J.R.; Wang, S.H. Influence of dendrimer surface charge on the bioactivity of 2-methoxyestradiol complexed with dendrimers. Soft Matter 2010, 6, 2539–2545. [Google Scholar] [CrossRef] [PubMed]
- Kukowska-Latallo, J.F.; Candido, K.A.; Cao, Z.Y.; Nigavekar, S.S.; Majoros, I.J.; Thomas, T.P.; Balogh, L.P.; Khan, M.K.; Baker, J.R. Nanoparticle targeting of anticancer drug improves therapeutic response in animal model of human epithelial cancer. Cancer Res. 2005, 65, 5317–5324. [Google Scholar] [CrossRef] [PubMed]
- Bhadra, D.; Bhadra, S.; Jain, S.; Jain, N.K. A pegylated dendritic nanoparticulate carrier of fluorouracil. Int. J. Pharm. 2003, 257, 111–124. [Google Scholar] [CrossRef]
- Noriega-Luna, B.; Godinez, L.A.; Rodriguez, F.J.; Rodriguez, A.; Zaldivar-Lelo de Larrea, G.; Sosa-Ferreyra, C.F.; Mercado-Curiel, R.F.; Manriquez, J.; Bustos, E. Applications of dendrimers in drug delivery agents, diagnosis, therapy, and detection. J. Nanomater. 2014, 2014, 1–19. [Google Scholar] [CrossRef]
- Guo, R.; Yao, Y.; Cheng, G.; Wang, S.; Li, Y.; Shen, M.; Zhang, Y.; Baker, J.R., Jr.; Wang, J.; Shi, X. Synthesis of glycoconjugated poly(amindoamine) dendrimers for targeting human liver cancer cells. RSC Adv. 2012, 2, 99–102. [Google Scholar] [CrossRef]
- Cheng, Y.; Zhao, L.; Li, Y.; Xu, T. Design of biocompatible dendrimers for cancer diagnosis and therapy: Current status and future perspectives. Chem. Soc. Rev. 2011, 40, 2673–2703. [Google Scholar] [CrossRef] [PubMed]
- Pan, G.F.; Lemmouchi, Y.; Akala, E.O.; Bakare, O. Studies on pegylated and drug-loaded pamam dendrimers. J. Bioact. Compat. Polym. 2005, 20, 113–128. [Google Scholar] [CrossRef]
- Milowska, K.; Rybczynska, A.; Mosiolek, J.; Durdyn, J.; Szewczyk, E.M.; Katir, N.; Brahmi, Y.; Majoral, J.P.; Bousmina, M.; Bryszewska, M. Biological activity of mesoporous dendrimer-coated titanium dioxide: Insight on the role of the surface-interface composition and the framework crystallinity. ACS Appl. Mater. Interfaces 2015, 7, 19994–20003. [Google Scholar] [CrossRef] [PubMed]
- Azzouz, A.; Nousir, S.; Bouazizi, N.; Roy, R. Metal–inorganic–organic matrices as efficient sorbents for hydrogen storage. Chem. Sus. Chem. 2015, 8, 800–803. [Google Scholar] [CrossRef] [PubMed]
- El Kadib, A.; Katir, N.; Bousmina, M.; Majoral, J.P. Dendrimer–silica hybrid mesoporous materials. New J. Chem. 2012, 36, 241–255. [Google Scholar] [CrossRef]
- Wheeler, P.A.; Wang, J.; Baker, J.; Mathias, L.J. Synthesis and characterization of covalently functionalized laponite clay. Chem. Mater. 2005, 17, 3012–3018. [Google Scholar] [CrossRef]
- Grabchev, I.; Petkov, C.; Bojinov, V. Infrared spectral characterization of poly(amidoamine) dendrimers peripherally modified with 1,8-naphthalimides. Dyes Pigm. 2004, 62, 229–234. [Google Scholar] [CrossRef]
- Nazarpoor, Z.; Ma, S.; Fanson, P.T.; Alexeev, O.S.; Amiridis, M.D. Decomposition of poly(amidoamine) (PAMAM) dendrimers under O2 plasma conditions. Polym. Degrad. Stable 2012, 97, 439–451. [Google Scholar] [CrossRef]
- Zheng, F.; Wang, S.; Shen, M.; Zhu, M.; Shi, X. Antitumor efficacy of doxorubicin-loaded electrospun nano-hydroxyapatite-poly(lactic-co-glycolic acid) composite nanofibers. Polym. Chem. 2013, 4, 933–941. [Google Scholar] [CrossRef]
- Chen, F.; Zhang, L.; Chen, Q.; Zhang, Y.; Zhang, Z. Synthesis of a novel magnetic drug delivery system composed of doxorubicin-conjugated Fe3O4 nanoparticle cores and a peg-functionalized porous silica shell. Chem. Commun. 2010, 46, 8633–8635. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zheng, L.; Peng, C.; Guo, R.; Shen, M.; Shi, X.; Zhang, G. Computed tomography imaging of cancer cells using acetylated dendrimer-entrapped gold nanoparticles. Biomaterials 2011, 32, 2979–2988. [Google Scholar] [CrossRef] [PubMed]
- Lesniak, W.; Bielinska, A.U.; Sun, K.; Janczak, K.W.; Shi, X.; Baker, J.R.; Balogh, L.P. Silver/dendrimer nanocomposites as biomarkers: Fabrication, characterization, in vitro toxicity, and intracellular detection. Nano Lett. 2005, 5, 2123–2130. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mustafa, R.; Luo, Y.; Wu, Y.; Guo, R.; Shi, X. Dendrimer-Functionalized Laponite Nanodisks as a Platform for Anticancer Drug Delivery. Nanomaterials 2015, 5, 1716-1731. https://doi.org/10.3390/nano5041716
Mustafa R, Luo Y, Wu Y, Guo R, Shi X. Dendrimer-Functionalized Laponite Nanodisks as a Platform for Anticancer Drug Delivery. Nanomaterials. 2015; 5(4):1716-1731. https://doi.org/10.3390/nano5041716
Chicago/Turabian StyleMustafa, Rania, Yu Luo, Yilun Wu, Rui Guo, and Xiangyang Shi. 2015. "Dendrimer-Functionalized Laponite Nanodisks as a Platform for Anticancer Drug Delivery" Nanomaterials 5, no. 4: 1716-1731. https://doi.org/10.3390/nano5041716
APA StyleMustafa, R., Luo, Y., Wu, Y., Guo, R., & Shi, X. (2015). Dendrimer-Functionalized Laponite Nanodisks as a Platform for Anticancer Drug Delivery. Nanomaterials, 5(4), 1716-1731. https://doi.org/10.3390/nano5041716