MxCy-Type Nanocarbide Crystallization in CrMnFeCoNi, CrMnFeCoNiV0.5, and CrMnFeCoNiMo0.5 HEAs Manufactured Through Powder Metallurgy
Abstract
1. Introduction
2. Materials and Methods
2.1. High-Energy Ball Milling
2.2. HEA Characterization
2.3. Mathematical Models
3. Results and Discussions
3.1. Characterization of Powders Obtained Through HEBM
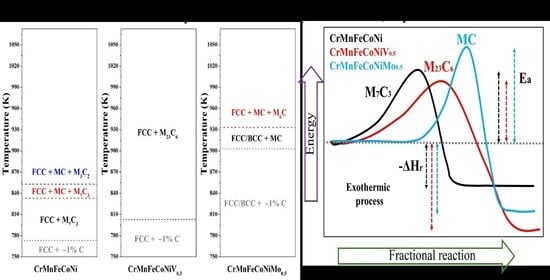
3.2. Structural Evolution at High Temperatures
3.3. Effect of V and Mo on the Fundamental Thermodynamic Parameters and Kinetics of MxCy Crystallization in HEAs
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yeh, J.-W.; Lin, S.-J.; Chin, T.-S.; Gan, J.-Y.; Chen, S.-K.; Shun, T.-T.; Tsau, C.-H.; Chou, S.-Y. Formation of simple crystal structures in Cu-co-Ni-Cr-Al-Fe-Ti-V alloys with multiprincipal metallic elements. Metall. Mater. Trans. A 2004, 35, 2533–2536. [Google Scholar] [CrossRef]
- Cantor, B.; Chang, I.; Knight, P.; Vincent, A. Microstructural development in equiatomic multicomponent alloys. Mater. Sci. Eng. A 2004, 375, 213–218. [Google Scholar] [CrossRef]
- Han, S.Z.; Choi, E.-A.; Lim, S.H.; Kim, S.; Lee, J. Alloy design strategies to increase strength and its trade-offs together. Prog. Mater. Sci. 2021, 117, 100720. [Google Scholar] [CrossRef]
- Angelo, P.; Subramanian, R.; Ravisankar, B. Powder Metallurgy: Science, Technology and Applications; PHI Learning Pvt. Ltd.: Delhi, India, 2022. [Google Scholar]
- Baláž, P. High-energy milling. In Mechanochemistry in Nanoscience and Minerals Engineering; Springer: Berlin/Heidelberg, Germany, 2008; pp. 103–132. [Google Scholar] [CrossRef]
- Kundin, J.; Lukianova, O.; Esakkiraja, N.; Paul, A.; Divinski, S.; Steinbach, I. Carbon effect on thermo-kinetics of Co-Cr-Fe-Mn-Ni high entropy alloys: A computational study validated by interdiffusion experiments. Acta Mater. 2023, 261, 119358. [Google Scholar] [CrossRef]
- Ravi, R.; Bakshi, S.R. Microstructural evolution and wear behavior of carbon added CoCrFeMnNi multi-component alloy fabricated by mechanical alloying and spark plasma sintering. J. Alloys Compd. 2021, 883, 160879. [Google Scholar] [CrossRef]
- Wang, X.; Luo, G.; Wei, Q.; Sun, Y.; Huang, W.; Peng, J.; Zhang, J.; Shen, Q. Superior high-temperature strength of a carbide-reinforced high-entropy alloy with ultrafine eutectoid structure. Scr. Mater. 2025, 255, 116393. [Google Scholar] [CrossRef]
- Chen, X.; Wang, F.; Zhang, X.; Hu, S.; Liu, X.; Humphry-Baker, S.; Gao, M.C.; He, L.; Lu, Y.; Cui, B. Novel refractory high-entropy metal-ceramic composites with superior mechanical properties. Int. J. Refract. Met. Hard Mater. 2024, 119, 106524. [Google Scholar] [CrossRef]
- Kaushik, N.; Meena, A.; Mali, H.S. High entropy alloy synthesis, characterization, manufacturing & potential applications: A review. Mater. Manuf. Process. 2022, 37, 1085–1109. [Google Scholar] [CrossRef]
- Alshataif, Y.A.; Sivasankaran, S.; Al-Mufadi, F.A.; Alaboodi, A.S.; Ammar, H.R. Manufacturing methods, microstructural and mechanical properties evolutions of high-entropy alloys: A review. Met. Mater. Int. 2020, 26, 1099–1133. [Google Scholar] [CrossRef]
- Martinez Garcia, A.; González, S.; Mendoza Duarte, J.M.; Gómez Esparza, C.D.; Ruiz Esparza Rodríguez, M.A.; Hurtado Macías, A.; Juarez Arellano, E.A.; Gutiérrez Castañeda, E.J.; Atanacio Sánchez, X.; Garay Reyes, C.G.; et al. Relationship Between Structure/Microstructure and Hardness of CrMnFeCoNiX0.5 High-Entropy Alloys with Refractory Metals X= V and Mo Obtained by Mechanical Alloying. Coatings 2026, 16, 491. [Google Scholar] [CrossRef]
- Yong, X.; Liu, X.; Yang, M.; Zhou, X. First-Principles Calculate the Stability, Mechanical Properties and Electronic Structure of Carbide MC, M2C and M6C in M50NiL Steel. Materials 2024, 17, 3498. [Google Scholar] [CrossRef]
- ASTM E2890; Test Method for Kinetic Parameters for Thermally Unstable Materials by Differential Scanning Calorimetry Using the Kissinger Method. ASTM: West Conshohocken, PA, USA, 2018.
- ASTM E698; Standard Test Method for Kinetic Parameters for Thermally Unstable Materials Using Differential Scanning Calorimetry and the Flynn/Wall/Ozawa Method. ASTM: West Conshohocken, PA, USA, 2018.
- Guía-Tello, J.; Garay-Reyes, C.; Ruiz-Esparza-Rodríguez, M.; Hernández, L.G.; Santillán, J.A.; Estrada-Guel, I.; Martínez-Sánchez, R. Effect of plastic deformation on the precipitation reaction in 2024 alloys. Mater. Chem. Phys. 2021, 271, 124927. [Google Scholar] [CrossRef]
- Brown, M.E. Introduction to Thermal Analysis: Techniques and Applications; Springer: Dordrecht, The Netherlands, 2001. [Google Scholar] [CrossRef]
- Park, J.; Ryu, W.; Kang, Y. DSC study on carbide precipitation reaction in Inconel 600. J. Nucl. Mater. 1994, 209, 221–225. [Google Scholar] [CrossRef]
- Dépinoy, S.; Toffolon-Masclet, C.; Urvoy, S.; Roubaud, J.; Marini, B.; Roch, F.; Kozeschnik, E.; Gourgues-Lorenzon, A.-F. Carbide precipitation in 2.25Cr-1Mo bainitic steel: Effect of heating and isothermal tempering conditions. Metall. Mater. Trans. A 2017, 48, 2164–2178. [Google Scholar] [CrossRef]
- Ravikanth, K.; Verma, A.; Singh, J.; Vishwanadh, B.; Rai, S.; Karri, M. Effect of prior microstructure on carbide precipitation in a Cr-Mo-V pressure vessel steel at 650 °C. J. Nucl. Mater. 2024, 602, 155359. [Google Scholar] [CrossRef]
- Shatynski, S.R. The thermochemistry of transition metal carbides. Oxid. Met. 1979, 13, 105–118. [Google Scholar] [CrossRef]
- Vaidya, M.; Karati, A.; Marshal, A.; Pradeep, K.; Murty, B. Phase evolution and stability of nanocrystalline CoCrFeNi and CoCrFeMnNi high entropy alloys. J. Alloys Compd. 2019, 770, 1004–1015. [Google Scholar] [CrossRef]
- Schmuecker, S.M.; Clouser, D.; Kraus, T.J.; Leonard, B.M. Synthesis of metastable chromium carbide nanomaterials and their electrocatalytic activity for the hydrogen evolution reaction. Dalton Trans. 2017, 46, 13524–13530. [Google Scholar] [CrossRef] [PubMed]
- Rahimi, R.; Volkova, O.; Biermann, H.; Mola, J. Thermal analysis of the formation and dissolution of Cr-rich carbides in Al-alloyed stainless steels. Adv. Eng. Mater. 2019, 21, 1800658. [Google Scholar] [CrossRef]
- Huang, M.; Zhou, Z.; Cui, C.; Zhang, R.; Shi, Z.; Wang, X.; Zhou, Y.; Sun, X. Formation and evolution behavior of M6C carbide in a Ni-W-Cr superalloy. Mater. Charact. 2023, 204, 113211. [Google Scholar] [CrossRef]
- Saumitra, V. Microstructural evolution and precipitation kinetics of carbides in L-PBF processed Inconel 718 during long-term homogenization. Mater. Today Commun. 2025, 42, 111192. [Google Scholar] [CrossRef]
- Oppermann, F.; Kies, F.; Noori, M.; Pradeep, K.; Hallstedt, B.; Haase, C. Influence of vanadium-carbide precipitation on the deformation behavior of Fe-30Mn-10Co-10Cr-2.5V-1.5C multi-principal element alloy. Mater. Des. 2024, 241, 112928. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, H.; Guo, C.; Liu, W.; Yang, Z.; Sun, X.; Zhang, Z.; Jiang, F. Effect of molybdenum addition on the precipitation of carbides in the austenite matrix of titanium micro-alloyed steels. J. Mater. Sci. 2016, 51, 4996–5007. [Google Scholar] [CrossRef]
- Benarosch, A.; Toffolon-Masclet, C.; Joubert, J.; Marini, B.; Meslin, E.; Trzaska, Z.; Thiaudiére, D.; Mocuta, C.; Guillot, I. Experimental study and modelling of carbide precipitation sequence in Fe based model alloys containing C, Mo and Mn. Acta Mater. 2024, 268, 1197. [Google Scholar] [CrossRef]





| Sample | Elemental Analysis/wt.% | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Cr | Co | Mn | Fe | Ni | V | Mo | C | O | |
| High-Energy Ball Milling 15 h | |||||||||
| CoCrFeMnNi | 18.28 | 22.39 | 18.75 | 18.48 | 21.29 | ---- | 0.79 | 0.01 | |
| CoCrFeMnNiV0.5 | 16.33 | 19.78 | 17.26 | 17.02 | 21.03 | 7.68 | ---- | 0.89 | 0.01 |
| CoCrFeMnNiMo0.5 | 15.42 | 18.70 | 16.29 | 16.14 | 17.66 | ---- | 14.88 | 0.91 | 0.01 |
| Scan Points | Elemental Analyses/wt.% | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Cr | Co | Mn | Fe | Ni | V | Mo | C | O | |
| 1 | 22.7 | 16.8 | 15.7 | 16.7 | 15.6 | 9.8 | 2.6 | ||
| 2 | 19.5 | 17.5 | 16.3 | 17.7 | 16.4 | 9.9 | 2.3 | ||
| 3 | 18.5 | 16.3 | 14.0 | 13.5 | 16.0 | 7.1 | 9.5 | 1.7 | |
| 4 | 21.3 | 13.2 | 12.4 | 14.2 | 12.9 | 8.0 | 15.3 | 2.6 | |
| 5 | 11.7 | 9.1 | 7.3 | 15.5 | 6.4 | 41.0 | 11.2 | 2.0 | |
| 6 | 16.8 | 13.3 | 13.9 | 14.2 | 14.2 | 9.1 | 16.9 | 1.6 | |
| HEA | β K/min | Tp K | Kissinger’s Model kJ/mol | Ozawa’s Model kJ/mol | ΔHr J/g |
|---|---|---|---|---|---|
| CoCrFeMnNi | 5 | 810.84 | r = 0.998 Ea = 238.2 ± 10.1 k1 = 23.5 ± 1.5 | r = 0.998 Ea = 251.9 ± 20.2 k2 = 38.9 ± 1.5 | −51.223 |
| 10 | 827.73 | −34.631 | |||
| 15 | 835.32 | −33.192 | |||
| 20 | 842.26 | −58.234 | |||
| CoCrFeMnNiV0.5 | 5 | 884.26 | r = 0.863 Ea = 188.3 ± 78.0 k1 = 13.9 ± 10.4 | r = 0.879 Ea = 203.3 ± 77.9 k2 = 29.6 ± 10.4 | −267.606 |
| 10 | 895.01 | −176.531 | |||
| 15 | 898.60 | −120.666 | |||
| 20 | 925.97 | −330.536 | |||
| CoCrFeMnNiMo0.5 | 5 | 924.07 | r = 0.981 Ea = 326.2 k1 = 30.4 ± 5.8 | r = 0.983 Ea = 341.8 k2 = 46.1 ± 5.8 | −96.238 |
| 10 | 940.25 | −80.423 | |||
| 15 | 950.49 | −104.746 | |||
| 20 | 951.13 | −29.204 |
| HEA | kc | n | r2 |
|---|---|---|---|
| CoCrFeMnNi | 0.0348 | 4.485 | 0.999 |
| CoCrFeMnNiV0.5 | 0.0350 | 2.319 | 0.999 |
| CoCrFeMnNiMo0.5 | 0.0389 | 4.521 | 0.999 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Garcia, A.M.; Garay Reyes, C.G.; Juárez Arellano, E.A.; Ruiz Esparza Rodríguez, M.A.; Mendoza Duarte, J.M.; López López, I.I.; Guía Tello, J.C.; Rodríguez Cabriales, G.; González, S.; Gutiérrez Castañeda, E.J.; et al. MxCy-Type Nanocarbide Crystallization in CrMnFeCoNi, CrMnFeCoNiV0.5, and CrMnFeCoNiMo0.5 HEAs Manufactured Through Powder Metallurgy. Nanomaterials 2026, 16, 592. https://doi.org/10.3390/nano16100592
Garcia AM, Garay Reyes CG, Juárez Arellano EA, Ruiz Esparza Rodríguez MA, Mendoza Duarte JM, López López II, Guía Tello JC, Rodríguez Cabriales G, González S, Gutiérrez Castañeda EJ, et al. MxCy-Type Nanocarbide Crystallization in CrMnFeCoNi, CrMnFeCoNiV0.5, and CrMnFeCoNiMo0.5 HEAs Manufactured Through Powder Metallurgy. Nanomaterials. 2026; 16(10):592. https://doi.org/10.3390/nano16100592
Chicago/Turabian StyleGarcia, Alfredo Martinez, Carlos Gamaliel Garay Reyes, Erick Adrián Juárez Arellano, Marco Antonio Ruiz Esparza Rodríguez, José Manuel Mendoza Duarte, Irving Ignacio López López, Juan Carlos Guía Tello, Gustavo Rodríguez Cabriales, Sergio González, Emmanuel José Gutiérrez Castañeda, and et al. 2026. "MxCy-Type Nanocarbide Crystallization in CrMnFeCoNi, CrMnFeCoNiV0.5, and CrMnFeCoNiMo0.5 HEAs Manufactured Through Powder Metallurgy" Nanomaterials 16, no. 10: 592. https://doi.org/10.3390/nano16100592
APA StyleGarcia, A. M., Garay Reyes, C. G., Juárez Arellano, E. A., Ruiz Esparza Rodríguez, M. A., Mendoza Duarte, J. M., López López, I. I., Guía Tello, J. C., Rodríguez Cabriales, G., González, S., Gutiérrez Castañeda, E. J., & Martínez Sánchez, R. (2026). MxCy-Type Nanocarbide Crystallization in CrMnFeCoNi, CrMnFeCoNiV0.5, and CrMnFeCoNiMo0.5 HEAs Manufactured Through Powder Metallurgy. Nanomaterials, 16(10), 592. https://doi.org/10.3390/nano16100592