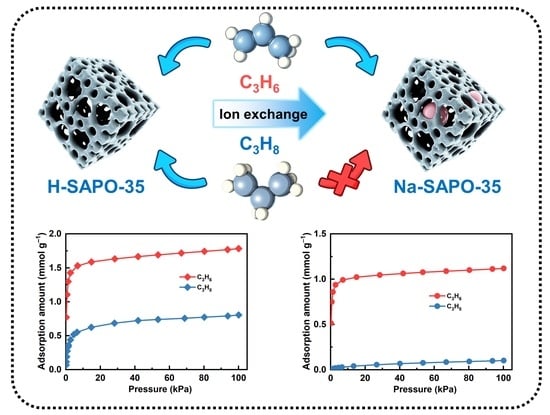
The Molecular Sieving of Propylene and Propane on SAPO-35 Molecular Sieve
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
2.2. Synthesis and Modification of SAPO-35
2.3. Breakthrough Experiments
2.4. Theoretical Calculation Method
2.5. Characterizations
3. Results and Discussion
3.1. Characterization of Adsorbents
3.2. Equilibrium Adsorption Performance
3.3. Regeneration Performance
3.4. Adsorption Thermodynamics
3.5. Breakthrough Experiment
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Zhang, K.Q.; Wang, R. A critical review on new and efficient adsorbents for CO2 capture. Chem. Eng. J. 2024, 485, 149495. [Google Scholar] [CrossRef]
- Rogg, S.; Hess, C. CO2 as a soft oxidant for propane oxidative dehydrogenation: A mechanistic study using operando UV Raman spectroscopy. J. CO2 Util. 2021, 50, 101604. [Google Scholar] [CrossRef]
- Zhou, W.; Jiang, Y.L.; Sun, Z.G.; Zhou, S.Q.; Xing, E.R.; Hai, Y.; Chen, G.H.; Zhao, Y.T. Support effect of Ga-based catalysts in the CO2-assisted oxidative dehydrogenation of propane. Catalysts 2023, 13, 896. [Google Scholar] [CrossRef]
- Hong, A.N.; Yang, H.J.; Li, T.; Wang, Y.; Wang, Y.X.; Jia, X.X.; Zhou, A.; Kusumoputro, E.; Li, J.P.; Bu, X.H.; et al. Pore-space partition and optimization for propane-selective high-performance propane/propylene separation. ACS Appl. Mater. Interfaces 2021, 13, 52160–52166. [Google Scholar] [CrossRef] [PubMed]
- Sun, N.; Yu, H.Y.; Potapov, A.S.; Sun, Y.G. Separation of propylene and propane using metal-organic frameworks. Comments Inorg. Chem. 2024, 44, 203–233. [Google Scholar] [CrossRef]
- Jarvelin, H.; Fair, J.R. Adsorptive separation of propylene propane mixtures. Ind. Eng. Chem. Res. 1993, 32, 2201–2207. [Google Scholar] [CrossRef]
- van Miltenburg, A.; Zhu, W.; Kapteijn, F.; Moulijn, J.A. Adsorptive separation of light olefin/paraffin mixtures. Chem. Eng. Res. Des. 2006, 84, 350–354. [Google Scholar] [CrossRef]
- Yu, L.; Han, X.; Wang, H.; Ullah, S.; Xia, Q.B.; Li, W.Y.; Li, J.N.; da Silva, I.; Manuel, P.; Rudic, S.; et al. Pore distortion in a metal-organic framework for regulated separation of propane and propylene. J. Am. Chem. Soc. 2021, 143, 19300–19305. [Google Scholar] [CrossRef]
- Wang, Y.X.; Zhao, D. Beyond equilibrium: Metal-organic frameworks for molecular sieving and kinetic gas separation. Cryst. Growth Des. 2017, 17, 2291–2308. [Google Scholar] [CrossRef]
- Li, J.R.; Kuppler, R.J.; Zhou, H.C. Selective gas adsorption and separation in metal-organic frameworks. Chem. Soc. Rev. 2009, 38, 1477–1504. [Google Scholar] [CrossRef]
- Peralta, D.; Chaplais, G.; Simon-Masseron, A.; Barthelet, K.; Chizallet, C.; Quoineaud, A.A.; Pirngruber, G.D. Comparison of the behavior of metal-organic frameworks and zeolites for hydrocarbon separations. J. Am. Chem. Soc. 2012, 134, 8115–8126. [Google Scholar] [CrossRef]
- Ren, Y.X.; Liang, X.; Dou, H.Z.; Ye, C.M.; Guo, Z.Y.; Wang, J.Y.; Pan, Y.C.; Wu, H.; Guiver, M.D.; Jiang, Z.Y. Membrane-based olefin/paraffin separations. Adv. Sci. 2020, 7, 2001398. [Google Scholar] [CrossRef]
- Da Silva, F.A.; Rodrigues, A.E. Adsorption equilibria and kinetics for propylene and propane over 13X and 4A zeolite pellets. Ind. Eng. Chem. Res. 1999, 38, 2051–2057. [Google Scholar] [CrossRef]
- Rege, S.U.; Padin, J.; Yang, R.T. Olefin/paraffin separations by adsorption: π-complexation vs. kinetic separation. AIChE J. 1998, 44, 799–809. [Google Scholar] [CrossRef]
- Padin, J.; Rege, S.U.; Yang, R.T.; Cheng, L.S. Molecular sieve sorbents for kinetic separation of propane/propylene. Chem. Eng. Sci. 2000, 55, 4525–4535. [Google Scholar] [CrossRef]
- van den Bergh, J.; Zhu, W.; Gascon, J.; Moulijn, J.A.; Kapteijn, F. Separation and permeation characteristics of a DD3R zeolite membrane. J. Membr. Sci. 2008, 316, 35–45. [Google Scholar] [CrossRef]
- Olson, D.H.; Camblor, M.A.; Villaescusa, L.A.; Kuehl, G.H. Light hydrocarbon sorption properties of pure silica Si-CHA and ITQ-3 and high silica ZSM-58. Microporous Mesoporous Mater. 2004, 67, 27–33. [Google Scholar] [CrossRef]
- López, J.M.; Navarro, M.V.; García, T.; Murillo, R.; Mastral, A.M.; Varela-Gandía, F.J.; Lozano-Castelló, D.; Bueno-López, A.; Cazorla-Amorós, D. Screening of different zeolites and silicoaluminophosphates for the retention of propene under cold start conditions. Microporous Mesoporous Mater. 2010, 130, 239–247. [Google Scholar] [CrossRef]
- Pérez-Botella, E.; Martínez-Franco, R.; González-Camuñas, N.; Cantín, A.; Palomino, M.; Moliner, M.; Valencia, S.; Rey, F. Unusually low heat of adsorption of CO2 on AlPO and SAPO molecular sieves. Front. Chem. 2020, 8, 588712. [Google Scholar] [CrossRef]
- Tong, Y.S.; Xing, J.C.; Lou, C.Y.; Yuan, D.H.; Huang, W.; Chen, Z.A.; Liu, Z.M.; Xu, Y.P. Efficient separation of propylene and propane on SAPO-17 molecular sieve. Can. J. Chem. 2021, 99, 570–575. [Google Scholar] [CrossRef]
- IZA Structure Commission. Available online: https://europe.iza-structure.org/IZA-SC/framework.php?ID=134 (accessed on 19 November 2025).
- Dantas, S.; Struckhoff, K.C.; Thommes, M.; Neimark, A.V. Phase behavior and capillary condensation hysteresis of carbon dioxide in mesopores. Langmuir 2019, 35, 11291–11298. [Google Scholar] [CrossRef]
- Gesikiewicz-Puchalska, A.; Zgrzebnicki, M.; Michalkiewicz, B.; Kalamaga, A.; Narkiewicz, U.; Morawski, A.W.; Wrobel, R. Changes in porous parameters of the ion exchanged X zeolite and their effect on CO2 adsorption. Molecules 2021, 26, 7520. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Chen, H.W.; Wang, C.R.; Ye, Y.; Li, L.B.; Song, X.W.; Yu, J.H. Achieving highly selective CO2 adsorption on SAPO-35 zeolites by template-modulating the framework silicon content. Chem. Sci. 2022, 13, 5687–5692. [Google Scholar] [CrossRef] [PubMed]
- Agliullin, M.R.; Kolyagin, Y.G.; Serebrennikov, D.V.; Grigor’eva, N.G.; Dmitrenok, A.S.; Maistrenko, V.N.; Dib, E.; Mintova, S.; Kutepov, B.I. Acid properties and morphology of SAPO-11 molecular sieve controlled by silica source. Microporous Mesoporous Mater. 2022, 338, 111962. [Google Scholar] [CrossRef]
- Yang, G.P.; Du, X.S.; Ran, J.Y.; Wang, X.M.; Chen, Y.R.; Zhang, L.; Rac, V.; Rakic, V.; Crittenden, J. Irregular influence of alkali metals on Cu-SAPO-34 catalyst for selective catalytic reduction of NOx with ammonia. J. Hazard. Mater. 2020, 387, 122007. [Google Scholar] [CrossRef]
- Zibrowius, B.; Loffler, E.; Hunger, M. Multinuclare MAS NMR and IR spectroscopic study of silicon incorporation into SAPO-5, SAPO-31, and SAPO-34 molecular sieves. Zeolites 1992, 12, 167–174. [Google Scholar] [CrossRef]
- Buchholz, A.; Wang, W.; Arnold, A.; Xu, M.; Hunger, M. Successive steps of hydration and dehydration of silicoaluminophosphates H-SAPO-34 and H-SAPO-37 investigated by in situ CF MAS NMR spectroscopy. Microporous Mesoporous Mater. 2003, 57, 157–168. [Google Scholar] [CrossRef]
- Abdi, H.; Maghsoudi, H. All-silica DD3R zeolite for adsorptive separation of propylene from propane: Equilibrium and kinetic data. Microporous Mesoporous Mater. 2020, 307, 110513. [Google Scholar] [CrossRef]
- Xiong, Y.; Tian, T.; L’Hermitte, A.; Mendez, A.S.J.; Danaci, D.; Platero-Prats, A.E.; Petit, C. Using silver exchange to achieve high uptake and selectivity for propylene/propane separation in zeolite Y. Chem. Eng. J. 2022, 446, 137104. [Google Scholar] [CrossRef]
- Min, J.G.; Luna-Triguero, A.; Byun, Y.; Balestra, S.R.G.; Vicent-Luna, J.M.; Calero, S.; Hong, S.B.; Camblor, M.A. Stepped propane adsorption in pure-silica ITW zeolite. Langmuir 2018, 34, 4774–4779. [Google Scholar] [CrossRef]
- Maruyama, H.; Seki, H. Adsorption modeling by two sites Langmuir type isotherm for adsorption of bisphenol-A and diethyl phthalate onto activated carbon in single and binary system. Sep. Sci. Technol. 2022, 57, 1535–1542. [Google Scholar] [CrossRef]
- Min, J.G.; Kemp, K.C.; Kencana, K.S.; Mukti, R.R.; Hong, S.B. Dealuminated Cs-ZK-5 zeolite for propylene/propane separation. Chem. Eng. J. 2021, 413, 127422. [Google Scholar] [CrossRef]
- Boronat, M.; Viruela, P.; Corma, A. A theoretical study on the mechanism of the superacid-catalyzed unimolecular isomerization of n-alkanes and n-alkenes. Comparison between ab initio and density functional results. J. Phys. Chem. 1996, 100, 16514–16521. [Google Scholar] [CrossRef]
- Biscardi, J.A.; Iglesia, E. Isotopic tracer studies of propane reactions on H-ZSM5 zeolite. J. Phys. Chem. B 1998, 102, 9284–9289. [Google Scholar] [CrossRef]
- Kiricsi, I.; Förster, H.; Tasi, G.; Nagy, J.B. Generation, characterization, and transformations of unsaturated carbenium ions in zeolites. Chem. Rev. 1999, 99, 2085–2114. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, A.K.; Kydd, R.A. A Fourier-transform infrared spectral study of propene reactions on acidic zeolites. J. Catal. 1986, 100, 185–195. [Google Scholar] [CrossRef]
- Do, D.D.; Nicholson, D.; Do, H.D. On the Henry constant and isosteric heat at zero loading in gas phase adsorption. J. Colloid Interface Sci. 2008, 324, 15–24. [Google Scholar] [CrossRef]
- Siderius, D.W.; Hatch, H.W.; Shen, V.K. Temperature extrapolation of Henry’s law constants and the isosteric heat of adsorption. J. Phys. Chem. B 2022, 126, 7999–8009. [Google Scholar] [CrossRef]
- Grande, C.A.; Gascon, J.; Kapteijn, F.; Rodrigues, A.E. Propane/propylene separation with Li-exchanged zeolite 13X. Chem. Eng. J. 2010, 160, 207–214. [Google Scholar] [CrossRef]
- Gao, S.S.; Liu, Z.Q.; Xu, S.T.; Zheng, A.M.; Wu, P.F.; Li, B.; Yuan, X.S.; Wei, Y.X.; Liu, Z.M. Cavity-controlled diffusion in 8-membered ring molecular sieve catalysts for shape selective strategy. J. Catal. 2019, 377, 51–62. [Google Scholar] [CrossRef]
- Zhou, X.P.; Falconer, J.L. Competitive adsorption between propylene and propane on zeolite 5A and the influence of organic phosphonic acid coatings. Sep. Purif. Technol. 2024, 346, 127435. [Google Scholar] [CrossRef]
- Xie, Y.; Shi, Y.S.; Morales, E.M.C.; El Karch, A.; Wang, B.; Arman, H.; Tan, K.; Chen, B.L. Optimal binding affinity for sieving separation of propylene from propane in an oxyfluoride anion-based metal-organic framework. J. Am. Chem. Soc. 2023, 145, 2386–2394. [Google Scholar] [CrossRef]
- Zhu, W.; Kapteijn, F.; Moulijn, J.A.; den Exter, M.C.; Jansen, J.C. Shape selectivity in adsorption on the all-silica DD3R. Langmuir 2000, 16, 3322–3329. [Google Scholar] [CrossRef]
- Barrer, R.M.; Lee, J.A. Hydrocarbons in Zeolite L. I. Sorption isotherms and enthalpies. Surf. Sci. 1968, 12, 341–353. [Google Scholar] [CrossRef]
- Olson, D.H.; Yang, X.B.; Camblor, M.A. ITQ-12: A zeolite having temperature dependent adsorption selectivity and potential for propene separation. J. Phys. Chem. B 2004, 108, 11044–11048. [Google Scholar] [CrossRef]
- Hedin, N.; DeMartin, G.J.; Strohmaier, K.G.; Reyes, S.C. PFG NMR self-diffusion of propylene in ITQ-29, CaA and NaCaA: Window size and cation effects. Microporous Mesoporous Mater. 2007, 98, 182–188. [Google Scholar] [CrossRef]
- Palomino, M.; Cantín, A.; Corma, A.; Leiva, S.; Rey, F.; Valencia, S. Pure silica ITQ-32 zeolite allows separation of linear olefins from paraffins. Chem. Commun. 2007, 12, 1233–1235. [Google Scholar] [CrossRef]
- Maghsoudi, H.; Abdi, H.; Aidani, A. Temperature- and pressure-dependent adsorption equilibria and diffusivities of propylene and propane in pure-silica Si-CHA zeolite. Ind. Eng. Chem. Res. 2020, 59, 1682–1692. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tong, Y.; Hu, K.; Yang, Q.; Liu, H.; Yuan, D.; Wang, J.; Lv, M.; Wang, H.; Tian, Z.; Xu, Y.; et al. The Molecular Sieving of Propylene and Propane on SAPO-35 Molecular Sieve. Nanomaterials 2025, 15, 1820. https://doi.org/10.3390/nano15231820
Tong Y, Hu K, Yang Q, Liu H, Yuan D, Wang J, Lv M, Wang H, Tian Z, Xu Y, et al. The Molecular Sieving of Propylene and Propane on SAPO-35 Molecular Sieve. Nanomaterials. 2025; 15(23):1820. https://doi.org/10.3390/nano15231820
Chicago/Turabian StyleTong, Yansi, Kadi Hu, Qihao Yang, Hao Liu, Danhua Yuan, Jungang Wang, Mengting Lv, Hailong Wang, Ziqi Tian, Yunpeng Xu, and et al. 2025. "The Molecular Sieving of Propylene and Propane on SAPO-35 Molecular Sieve" Nanomaterials 15, no. 23: 1820. https://doi.org/10.3390/nano15231820
APA StyleTong, Y., Hu, K., Yang, Q., Liu, H., Yuan, D., Wang, J., Lv, M., Wang, H., Tian, Z., Xu, Y., & Chen, L. (2025). The Molecular Sieving of Propylene and Propane on SAPO-35 Molecular Sieve. Nanomaterials, 15(23), 1820. https://doi.org/10.3390/nano15231820