Lipid-Based Drug Delivery Systems: Concepts and Recent Advances in Transdermal Applications
Abstract
1. Introduction
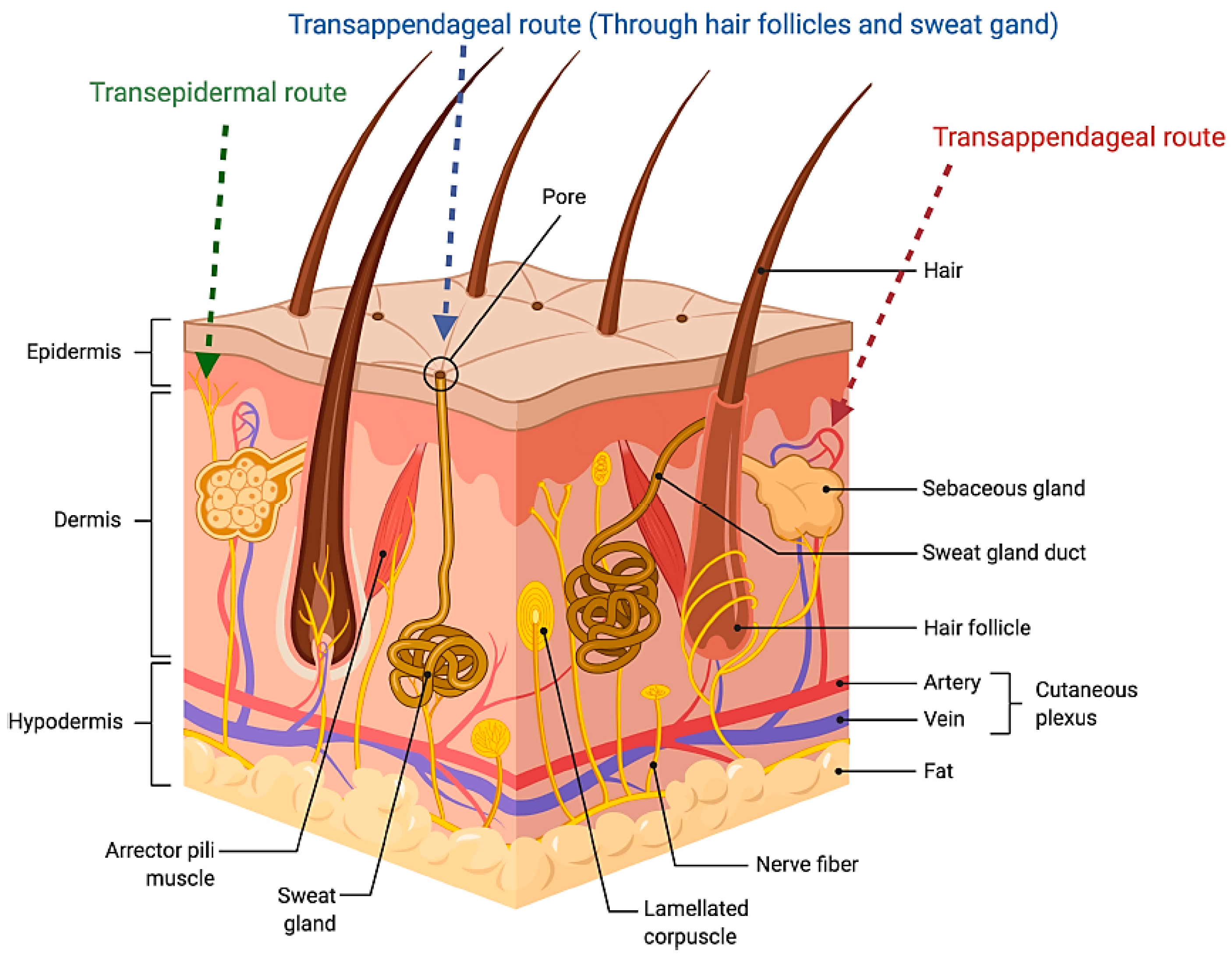
2. Anatomy of the Skin: Challenges for Formulation Strategies
3. Lipid-Based Systems as a Strategy for Permeation Enhancement
4. Types of Lipid-Based Drug Delivery Systems for Transdermal Administration
4.1. Liposomes
4.2. Solid Lipid Nanoparticles (SLNs)
4.3. Nanostructured Lipid Carriers (NLCs)
4.4. Ethosomes and Transfersomes
5. Fast Clinical Translation of Lipid-Based Formulations for Transdermal Administration
6. Quality by Design Approach for the Design and the Development of Lipid-Based Drug Delivery Systems
7. Preparation and Characterization Methods of Lipid-Based Drug Delivery Systems
8. Regulatory Aspects of Lipid-Based Drug Delivery Systems
8.1. Characterization Requirements by Regulatory Framework
8.2. Challenges and Limitations of Regulatory Framework
9. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Alkilani, A.Z.; Hamed, R.; Musleha, B.; Sharaire, Z. Breaking Boundaries: The Advancements in Transdermal Delivery of Antibiotics. Drug Deliv. 2024, 31, 2304251. [Google Scholar] [CrossRef]
- Sala, M.; Diab, R.; Elaissari, A.; Fessi, H. Lipid Nanocarriers as Skin Drug Delivery Systems: Properties, Mechanisms of Skin Interactions and Medical Applications. Int. J. Pharm. 2018, 535, 1–17. [Google Scholar] [CrossRef]
- Akl, M.A.; Eldeen, M.A.; Kassem, A.M. Beyond Skin Deep: Phospholipid-Based Nanovesicles as Game-Changers in Transdermal Drug Delivery. AAPS PharmSciTech 2024, 25, 184. [Google Scholar] [CrossRef] [PubMed]
- Pradhan, M.; Srivastava, S.; Singh, D.; Saraf, S.; Saraf, S.; Singh, M.R. Perspectives of Lipid-Based Drug Carrier Systems for Transdermal Delivery. Crit. Rev. Ther. Drug Carrier Syst. 2018, 35, 331–367. [Google Scholar] [CrossRef] [PubMed]
- Prausnitz, M.R.; Langer, R. Transdermal Drug Delivery. Nat. Biotechnol. 2008, 26, 1261–1268. [Google Scholar] [CrossRef]
- Phatale, V.; Vaiphei, K.K.; Jha, S.; Patil, D.; Agrawal, M.; Alexander, A. Overcoming Skin Barriers through Advanced Transdermal Drug Delivery Approaches. J. Control. Release 2022, 351, 361–380. [Google Scholar] [CrossRef]
- Pielenhofer, J.; Sohl, J.; Windbergs, M.; Langguth, P.; Radsak, M.P. Current Progress in Particle-Based Systems for Transdermal Vaccine Delivery. Front. Immunol. 2020, 11, 266. [Google Scholar] [CrossRef] [PubMed]
- De Jong, W.H.; Borm, P.J. Drug Delivery and Nanoparticles: Applications and Hazards. Int. J. Nanomed. 2008, 3, 133–149. [Google Scholar] [CrossRef]
- Singh, A.P.; Biswas, A.; Shukla, A.; Maiti, P. Targeted Therapy in Chronic Diseases Using Nanomaterial-Based Drug Delivery Vehicles. Signal Transduct. Target. Ther. 2019, 4, 33. [Google Scholar] [CrossRef]
- Wang, X.; Li, C.; Wang, Y.; Chen, H.; Zhang, X.; Luo, C.; Zhou, W.; Li, L.; Teng, L.; Yu, H.; et al. Smart Drug Delivery Systems for Precise Cancer Therapy. Acta Pharm. Sin. B 2022, 12, 4098–4121. [Google Scholar] [CrossRef]
- Mitchell, M.J.; Billingsley, M.M.; Haley, R.M.; Wechsler, M.E.; Peppas, N.A.; Langer, R. Engineering Precision Nanoparticles for Drug Delivery. Nat. Rev. Drug Discov. 2021, 20, 101–124. [Google Scholar] [CrossRef]
- Despotopoulou, D.; Lagopati, N.; Pispas, S.; Gazouli, M.; Demetzos, C.; Pippa, N. The Technology of Transdermal Delivery Nanosystems: From Design and Development to Preclinical Studies. Int. J. Pharm. 2022, 611, 121290. [Google Scholar] [CrossRef]
- Chen, Y.; Feng, X. Gold Nanoparticles for Skin Drug Delivery. Int. J. Pharm. 2022, 625, 122122. [Google Scholar] [CrossRef]
- Benson, H.A.E. Skin Structure, Function, and Permeation. In Topical and Transdermal Drug Delivery; Benson, H.A.E., Watkinson, A.C., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2011; pp. 1–22. [Google Scholar]
- Yu, Y.Q.; Yang, X.; Wu, X.F.; Fan, Y.B. Enhancing Permeation of Drug Molecules Across the Skin via Delivery in Nanocarriers: Novel Strategies for Effective Transdermal Applications. Front. Bioeng. Biotechnol. 2021, 9, 646554. [Google Scholar] [CrossRef] [PubMed]
- Lawton, S. Skin 1: The Structure and Functions of the Skin. Nurs. Times 2019, 115, 30–33. [Google Scholar]
- Lane, M.E.; Santos, P.; Watkinson, A.C.; Hadgraft, J. Passive Skin Permeation Enhancement. In Topical and Transdermal Drug Delivery; Benson, H.A.E., Watkinson, A.C., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2012; pp. 23–42. [Google Scholar]
- Khan, M.S.; Mohapatra, S.; Gupta, V.; Ali, A.; Naseef, P.P.; Kurunian, M.S.; Alshadidi, A.A.F.; Alam, M.S.; Mirza, M.A.; Iqbal, Z. Potential of Lipid-Based Nanocarriers against Two Major Barriers to Drug Delivery—Skin and Blood–Brain Barrier. Membranes 2023, 13, 343. [Google Scholar] [CrossRef]
- Kalave, S.; Chatterjee, B.; Shah, P.; Misra, A. Transdermal Delivery of Macromolecules Using Nano Lipid Carriers. Curr. Pharm. Des. 2021, 27, 4330–4340. [Google Scholar] [CrossRef]
- Chacko, I.A.; Ghate, V.M.; Dsouza, L.; Lewis, S.A. Lipid Vesicles: A Versatile Drug Delivery Platform for Dermal and Transdermal Applications. Colloids Surf. B Biointerfaces 2020, 195, 111262. [Google Scholar] [CrossRef]
- Dobreva, M.; Stefanov, S.; Andonova, V. Natural Lipids as Structural Components of Solid Lipid Nanoparticles and Nanostructured Lipid Carriers for Topical Delivery. Curr. Pharm. Des. 2020, 26, 4524–4535. [Google Scholar] [CrossRef]
- Hua, S. Lipid-Based Nano-Delivery Systems for Skin Delivery of Drugs and Bioactives. Front. Pharmacol. 2015, 6, 219. [Google Scholar] [CrossRef]
- Guo, F.; Wang, J.; Ma, M.; Tan, F.; Li, N. Skin targeted lipid vesicles as novel nano carrier of ketoconazole: Characterization, in vitro and in vivo evaluation. J. Mater. Sci. Mater. Med. 2015, 26, 175. [Google Scholar] [CrossRef]
- Gillet, A.; Compère, P.; Lecomte, F.; Hubert, P.; Ducat, E.; Evrard, B.; Piel, G. Skin penetration behaviour of liposomes as a function of their composition. Eur. J. Pharm. Biopharm. 2011, 79, 43–53. [Google Scholar] [CrossRef]
- Rangsimawong, W.; Opanasopit, P.; Rojanarata, T.; Ngawhirunpat, T. Terpene-Containing PEGylated Liposomes as Transdermal Carriers of a Hydrophilic Compound. Biol. Pharm. Bull. 2014, 37, 1936–1943. [Google Scholar] [CrossRef]
- Zhang, J.P.; Wei, Y.H.; Zhou, Y.; Li, Y.Q.; Wu, X.A. Ethosomes, binary ethosomes and transfersomes of terbinafine hydrochloride: A comparative study. Arch. Pharm. Res. 2012, 35, 109–117. [Google Scholar] [CrossRef]
- Pena-Rodríguez, E.; Moreno, M.C.; Blanco-Fernandez, B.; González, J.; Fernández-Campos, F. Epidermal Delivery of Retinyl Palmitate Loaded Transfersomes: Penetration and Biodistribution Studies. Pharmaceutics 2020, 12, 112. [Google Scholar] [CrossRef]
- Touitou, E.; Dayan, N.; Bergelson, L.; Godin, B.; Eliaz, M. Ethosomes—Novel Vesicular Carriers for Enhanced Delivery: Characterization and Skin Penetration Properties. J. Control. Release 2000, 65, 403–418. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Wu, L.; Wu, D.; Shi, D.; Wang, T.; Zhu, X. Mechanism investigation of ethosomes transdermal permeation. Int. J. Nanomed. 2017, 12, 3357–3364. [Google Scholar] [CrossRef] [PubMed]
- Pardeike, J.; Hommoss, A.; Müller, R.H. Lipid nanoparticles (SLN, NLC) in cosmetic and pharmaceutical dermal products. Int. J. Pharm. 2009, 366, 170–184. [Google Scholar] [CrossRef] [PubMed]
- Müller, R.H.; Radtke, M.; Wissing, S.A. Solid lipid nanoparticles (SLN) and nanostructured lipid carriers (NLC) in cosmetic and dermatological preparations. Adv. Drug Deliv. Rev. 2002, 54 (Suppl. S1), S131–S155. [Google Scholar] [CrossRef] [PubMed]
- Moghddam, S.M.; Ahad, A.; Aqil, M.; Imam, S.S.; Sultana, Y.; Ali, A. Optimization of nanostructured lipid carriers for topical delivery of nimesulide using Box–Behnken design approach. Artif. Cells Nanomed. Biotechnol. 2017, 45, 617–624. [Google Scholar] [CrossRef]
- Hosny, K.M.; Aldawsari, H.M. Avanafil Liposomes as Transdermal Drug Delivery for Erectile Dysfunction Treatment: Preparation, Characterization, and In Vitro, Ex Vivo and In Vivo Studies. Trop. J. Pharm. Res. 2015, 14, 559–565. [Google Scholar] [CrossRef][Green Version]
- Bi, Y. Liposomal Vitamin D3 as an Anti-Aging Agent for the Skin. Pharmaceutics 2019, 11, 311. [Google Scholar] [CrossRef]
- Kapoor, M.S.; D’Souza, A.; Aibani, N.; Nair, S.S.; Sandbhor, P.; Kumari, D.; Banerjee, R. Stable Liposome in Cosmetic Platforms for Transdermal Folic Acid Delivery for Fortification and Treatment of Micronutrient Deficiencies. Sci. Rep. 2018, 8, 16122. [Google Scholar] [CrossRef]
- Franzè, S.; Angelo, L.; Casiraghi, A.; Minghetti, P.; Cilurzo, F. Design of Liposomal Lidocaine/Cannabidiol Fixed Combinations for Local Neuropathic Pain Treatment. Pharmaceutics 2022, 14, 1915. [Google Scholar] [CrossRef]
- Rostamkalaei, S.S.; Akbari, J.; Saeedi, M.; Morteza-Semnani, K.; Nokhodchi, A. Topical Gel of Metformin Solid Lipid Nanoparticles: A Hopeful Promise as a Dermal Delivery System. Colloids Surf. B Biointerfaces 2019, 175, 150–157. [Google Scholar] [CrossRef]
- Butani, D.; Yewale, C.; Misra, A. Topical Amphotericin B Solid Lipid Nanoparticles: Design and Development. Colloids Surf. B Biointerfaces 2016, 139, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Garg, A.; Singh, S. Targeting of Eugenol-Loaded Solid Lipid Nanoparticles to the Epidermal Layer of Human Skin. Nanomedicine 2014, 9, 1223–1238. [Google Scholar] [CrossRef] [PubMed]
- Geetha, T.; Kapila, M.; Prakash, O.; Deol, P.K.; Kakkar, V.; Kaur, I.P. Sesamol-Loaded Solid Lipid Nanoparticles for Treatment of Skin Cancer. J. Drug Target. 2015, 23, 159–169. [Google Scholar] [CrossRef]
- Shrotriya, S.N.; Vidhate, B.; Shukla, M.S. Formulation and Development of Silybin-Loaded Solid Lipid Nanoparticle Enriched Gel for Irritant Contact Dermatitis. Drug Deliv. Transl. Res. 2017, 41, 164–173. [Google Scholar] [CrossRef]
- Fitriani, E.W.; Avanti, C.; Rosana, Y.; Surini, S. Nanostructured Lipid Carriers: A Prospective Dermal Drug Delivery System for Natural Active Ingredients. Pharmacia 2024, 71, 1–15. [Google Scholar] [CrossRef]
- Agrawal, Y.; Petkar, K.C.; Sawant, K.K. Development, Evaluation and Clinical Studies of Acitretin Loaded Nanostructured Lipid Carriers for Topical Treatment of Psoriasis. Int. J. Pharm. 2010, 401, 93–102. [Google Scholar] [CrossRef]
- Kawadkar, J.; Pathak, A.; Kishore, R.; Chauhan, M.K. Formulation, Characterization and In Vitro-In Vivo Evaluation of Flurbiprofen-Loaded Nanostructured Lipid Carriers for Transdermal Delivery. Drug Dev. Ind. Pharm. 2013, 39, 569–578. [Google Scholar] [CrossRef]
- Abdel-Salam, F.S.; Mahmoud, A.A.; Ammar, H.O.; Elkheshen, S.A. Nanostructured Lipid Carriers as Semisolid Topical Delivery Formulations for Diflucortolone Valerate. J. Liposome Res. 2017, 27, 41–55. [Google Scholar] [CrossRef]
- Baghel, S.; Nair, V.S.; Pirani, A.; Sravani, A.B.; Bhemisetty, B.; Ananthamurthy, K.; Aranjani, J.M.; Lewis, S.A. Luliconazole-Loaded Nanostructured Lipid Carriers for Topical Treatment of Superficial Tinea Infections. Dermatol. Ther. 2020, 33, e13959. [Google Scholar] [CrossRef]
- Mendes, I.T.; Ruela, A.L.M.; Carvalho, F.C.; Freitas, J.T.J.; Bonfilio, R.; Pereira, G.R. Development and Characterization of Nanostructured Lipid Carrier-Based Gels for the Transdermal Delivery of Donepezil. Colloids Surf. B Biointerfaces 2019, 177, 274–281. [Google Scholar] [CrossRef] [PubMed]
- Paiva-Santos, A.C.; Silva, A.L.; Guerra, C.; Peixoto, D.; Pereira-Silva, M.; Zeinali, M.; Mascarenhas-Melo, F.; Castro, R.; Veiga, F. Ethosomes as Nanocarriers for the Development of Skin Delivery Formulations. Pharm. Res. 2021, 38, 947–970. [Google Scholar] [CrossRef] [PubMed]
- Dubey, V.; Mishra, D.; Dutta, T.; Nahar, M.; Saraf, D.K.; Jain, N.K. Dermal and Transdermal Delivery of an Anti-Psoriatic Agent via Ethanolic Liposomes. J. Control. Release 2007, 123, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Godin, B.; Touitou, E. Erythromycin Ethosomal Systems: Physicochemical Characterization and Enhanced Antibacterial Activity. Curr. Drug Deliv. 2005, 2, 269–275. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhang, G.Q.; Wu, X.A. Synergistic Penetration of Ethosomes and Lipophilic Prodrug on the Transdermal Delivery of Acyclovir. Arch. Pharm. Res. 2010, 33, 567–574. [Google Scholar] [CrossRef]
- Liu, X.; Liu, H.; Liu, J.; He, Z.; Ding, C.; Huang, G.; Zhou, W.; Zhou, L. Preparation of a Ligustrazine Ethosome Patch and Its Evaluation In Vitro and In Vivo. Int. J. Nanomed. 2011, 6, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Cevc, G.; Blume, G. Biological Activity and Characteristics of Triamcinolone Acetonide Formulated with the Self-Regulating Drug Carriers, Transfersomes. Biochim. Biophys. Acta Biomembr. 2003, 1614, 156–164. [Google Scholar] [CrossRef] [PubMed]
- Jain, S.; Jain, P.; Umamaheshwari, R.B.; Jain, N.K. Transfersomes--a novel vesicular carrier for enhanced transdermal delivery: Development, characterization, and performance evaluation. Drug Dev. Ind. Pharm. 2003, 29, 1013–1026. [Google Scholar] [CrossRef] [PubMed]
- Cevc, G. Transdermal Drug Delivery of Insulin with Ultradeformable Carriers. Clin. Pharmacokinet. 2003, 42, 461–474. [Google Scholar] [CrossRef]
- Pierre, M.B.R.; Costa, I.M. Liposomal Systems as Drug Delivery Vehicles for Dermal and Transdermal Applications. Arch. Dermatol. Res. 2011, 303, 607–621. [Google Scholar] [CrossRef] [PubMed]
- Yarosh, D.; Klein, J.; O’Connor, A.; Hawk, J.; Rafal, E.; Wolf, P. Effect of Topically Applied T4 Endonuclease V in Liposomes on Skin Cancer in Xeroderma Pigmentosum: A Randomised Study. Lancet 2001, 357, 926–929. [Google Scholar] [CrossRef]
- Yarosh, D.B.; O’Connor, A.; Alas, L.; Potten, C.; Wolf, P. Photoprotection by Topical DNA Repair Enzymes: Molecular Correlates of Clinical Studies. Photochem. Photobiol. 1999, 69, 136–140. [Google Scholar] [CrossRef]
- Montoto, S.S.; Muraca, G.; Ruiz, M.E. Solid Lipid Nanoparticles for Drug Delivery: Pharmacological and Biopharmaceutical Aspects. Front. Mol. Biosci. 2020, 7, 587997. [Google Scholar] [CrossRef]
- Jyothi, V.G.S.S.; Ghouse, S.M.; Khatri, D.K.; Nanduri, S.; Singh, S.B.; Madan, J. Lipid Nanoparticles in Topical Dermal Drug Delivery: Does Chemistry of Lipid Persuade Skin Penetration? J. Drug Deliv. Sci. Technol. 2022, 69, 103176. [Google Scholar] [CrossRef]
- Aggarwal, N.; Goindi, S. Preparation and In Vivo Evaluation of Solid Lipid Nanoparticles of Griseofulvin for Dermal Use. J. Biomed. Nanotechnol. 2013, 9, 564–576. [Google Scholar] [CrossRef]
- Jain, S.; Jain, S.; Khare, P.; Gulbake, A.; Bansal, D.; Jain, S.K. Design and development of solid lipid nanoparticles for topical delivery of an anti-fungal agent. Drug Deliv. 2010, 17, 443–451. [Google Scholar] [CrossRef]
- Montenegro, L.; Sinico, C.; Castangia, I.; Carbone, C.; Puglisi, G. Idebenone-Loaded Solid Lipid Nanoparticles for Drug Delivery to the Skin: In Vitro Evaluation. Int. J. Pharm. 2012, 434, 169–174. [Google Scholar] [CrossRef]
- Jeon, H.S.; Seo, J.E.; Kim, M.S.; Kang, M.H.; Oh, D.H.; Jeon, S.O.; Jeong, S.H.; Choi, Y.W.; Lee, S. A Retinyl Palmitate-Loaded Solid Lipid Nanoparticle System: Effect of Surface Modification with Dicetyl Phosphate on Skin Permeation In Vitro and Antiwrinkle Effect In Vivo. Int. J. Pharm. 2013, 452, 311–320. [Google Scholar] [CrossRef]
- Pradhan, M.; Singh, D.; Murthy, S.N.; Singh, M.R. Design, Characterization and Skin Permeating Potential of Fluocinolone Acetonide Loaded Nanostructured Lipid Carriers for Topical Treatment of Psoriasis. Steroids 2015, 101, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, M.K.; Sharma, P.K. Optimization and Characterization of Rivastigmine Nanolipid Carrier Loaded Transdermal Patches for the Treatment of Dementia. Chem. Phys. Lipids 2019, 224, 104794. [Google Scholar] [CrossRef]
- Zhan, B.; Wang, J.; Li, H.; Xiao, K.; Fang, X.; Shi, Y.; Jia, Y. Ethosomes: A Promising Drug Delivery Platform for Transdermal Application. Chemistry 2024, 6, 993–1019. [Google Scholar] [CrossRef]
- Fang, Y.P.; Huang, Y.B.; Wu, P.C.; Tsai, Y.H. Topical Delivery of 5-Aminolevulinic Acid-Encapsulated Ethosomes in a Hyperproliferative Skin Animal Model Using the CLSM Technique to Evaluate the Penetration Behavior. Eur. J. Pharm. Biopharm. 2009, 73, 391–398. [Google Scholar] [CrossRef]
- Bhalaria, M.K.; Naik, S.; Misra, A.N. Ethosomes: A Novel System for Antifungal Drugs in the Treatment of Topical Fungal Disease. Indian J. Exp. Biol. 2009, 47, 368–375. [Google Scholar]
- Cevc, G.; Blume, G. New, Highly Efficient Formulation of Diclofenac for the Topical, Transdermal Administration in Ultradeformable Drug Carriers, Transfersomes. Biochim. Biophys. Acta Biomembr. 2001, 1514, 191–205. [Google Scholar] [CrossRef]
- Ghanbarzadeh, S.; Arami, S. Enhanced Transdermal Delivery of Diclofenac Sodium via Conventional Liposomes, Ethosomes, and Transfersomes. BioMed Res. Int. 2013, 2013, 616810. [Google Scholar] [CrossRef] [PubMed]
- Sharma, G.; Goyal, H.; Thakur, K.; Raza, K.; Katare, O.P. Novel Elastic Membrane Vesicles (EMVs) and Ethosomes-Mediated Effective Topical Delivery of Aceclofenac: A New Therapeutic Approach for Pain and Inflammation. Drug Deliv. 2016, 23, 3135–3145. [Google Scholar] [CrossRef] [PubMed]
- Botelho, M.A.; Queiroz, D.B.; Barros, G.; Guerreiro, S.; Fechine, P.; Umbelino, S.; Lyra, A.; Borges, B.; Freitas, A.; Queiroz, D.C.; et al. Nanostructured Transdermal Hormone Replacement Therapy for Relieving Menopausal Symptoms: A Confocal Raman Spectroscopy Study. Clinics 2014, 69, 75–82. [Google Scholar] [CrossRef]
- Pathak, P.; Nagarsenker, M. Formulation and Evaluation of Lidocaine Lipid Nanosystems for Dermal Delivery. AAPS PharmSciTech 2009, 10, 985–992. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Xiao, Z.; Wang, C.; Chen, G.; Zhang, Y.; Zhang, X.; Han, X.; Wang, J.; Ye, X.; Prausnitz, M.R.; et al. Microneedle Patches Loaded with Nanovesicles for Glucose Transporter-Mediated Insulin Delivery. ACS Nano 2022, 16, 18223–18231. [Google Scholar] [CrossRef]
- Khan, A.; Naquvi, K.J.; Haider, M.F.; Khan, M.A. Quality by Design—Newer Technique for Pharmaceutical Product Development. Intell. Pharm. 2024, 2, 122–129. [Google Scholar] [CrossRef]
- Verma, D.D.; Verma, S.; Blume, G.; Fahr, A. Particle Size of Liposomes Influences Dermal Delivery of Substances into Skin. Int. J. Pharm. 2003, 258, 141–151. [Google Scholar] [CrossRef]
- Abdel-Mottaleb, M.M.A.; Mortada, N.D.; El-Shamy, A.A.; Awad, G.A. Physically Cross-Linked Polyvinyl Alcohol for the Topical Delivery of Fluconazole. Drug Dev. Ind. Pharm. 2009, 35, 311–320. [Google Scholar] [CrossRef]
- Adib, Z.M.; Ghanbarzadeh, S.; Kouhsoltani, M.; Yari Khosroshahi, A.; Hamishehkar, H. The Effect of Particle Size on the Deposition of Solid Lipid Nanoparticles in Different Skin Layers: A Histological Study. Adv. Pharm. Bull. 2016, 6, 31. [Google Scholar] [CrossRef]
- Song, C.K.; Balakrishnan, P.; Shim, C.K.; Chung, S.J.; Chong, S.; Kim, D.D. A Novel Vesicular Carrier, Transethosome, for Enhanced Skin Delivery of Voriconazole: Characterization and In Vitro/In Vivo Evaluation. Colloids Surf. B Biointerfaces 2012, 92, 299–304. [Google Scholar] [CrossRef]
- Gomathi, T.; Rajeshwari, K.; Kanchana, V.; Sudha, P.N.; Parthasarathy, K. Impact of Nanoparticle Shape, Size, and Properties of the Sustainable Nanocomposites. In Sustainable Polymer Composites and Nanocomposites; Inamuddin, S., Thomas, S., Mavinkere Rangappa, S., Asiri, A.M., Eds.; Springer: Berlin/Heidelberg, Germany, 2019; pp. 313–336. [Google Scholar]
- Akombaetwa, N.; Ilangala, A.B.; Thom, L.; Memvanga, P.B.; Witika, B.A.; Buya, A.B. Current Advances in Lipid Nanosystems Intended for Topical and Transdermal Drug Delivery Applications. Pharmaceutics 2023, 15, 656. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Su, W.; Li, Q.; Li, C.; Wang, H.; Li, Y.; Cao, Y.; Chang, J.; Zhang, L. Preparation and Evaluation of Lidocaine Hydrochloride-Loaded TAT-Conjugated Polymeric Liposomes for Transdermal Delivery. Int. J. Pharm. 2013, 441, 748–756. [Google Scholar] [CrossRef] [PubMed]
- Jin, S.E.; Kim, C.K. Charge-Mediated Topical Delivery of Plasmid DNA with Cationic Lipid Nanoparticles to the Skin. Colloids Surf. B Biointerfaces 2014, 116, 582–590. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Li, Y.; Chen, X.; Zhou, Z.; Pang, J.; Luo, X.; Kong, M. A Surface Charge Dependent Enhanced Th1 Antigen-Specific Immune Response in Lymph Nodes by Transfersome-Based Nanovaccine-Loaded Dissolving Microneedle-Assisted Transdermal Immunization. J. Mater. Chem. B 2019, 7, 4854–4866. [Google Scholar] [CrossRef]
- Zhou, M.; Zhang, R.; Huang, M.; Lu, W.; Song, S. pH-Sensitive Imidazole-Modified Liposomes for Enhanced Transdermal Delivery. Pharmaceuticals 2022, 15, 306. [Google Scholar] [CrossRef]
- Pathan, I.B.; Jaware, B.P.; Shelke, S.; Ambekar, W. Curcumin Loaded Ethosomes for Transdermal Application: Formulation, Optimization, In-Vitro and In-Vivo Study. J. Drug Deliv. Sci. Technol. 2018, 44, 49–57. [Google Scholar] [CrossRef]
- Yang, Y.; Yan, S.; Yu, B.; Gao, C. Hydrophobically Modified Inulin Based Nanoemulsions for Enhanced Stability and Transdermal Delivery of Retinyl Propionate. Colloids Surf. A Physicochem. Eng. Asp. 2022, 653, 129883. [Google Scholar] [CrossRef]
- Jain, S.; Patel, N.; Madan, P.; Lin, S. Quality by Design Approach for Formulation, Evaluation and Statistical Optimization of Diclofenac-Loaded Ethosomes via Transdermal Route. Pharm. Dev. Technol. 2015, 20, 473–489. [Google Scholar] [CrossRef]
- Zagalo, D.M.; Silva, B.; Silva, C.; Simões, S. A Quality by Design (QbD) Approach in Pharmaceutical Development of Lipid-Based Nanosystems: A Systematic Review. J. Drug Deliv. Sci. Technol. 2022, 70, 103207. [Google Scholar] [CrossRef]
- Mendes, M.; Nunes, S.C.C.; Sousa, J.J.; Pais, A.A.C.C.; Vitorino, C. Expanding Transdermal Delivery with Lipid Nanoparticles: A New Drug-in-NLC-in-Adhesive Design. Mol. Pharm. 2017, 14, 2099–2115. [Google Scholar] [CrossRef] [PubMed]
- Arora, D.; Nanda, S. Quality by Design Driven Development of Resveratrol Loaded Ethosomal Hydrogel for Improved Dermatological Benefits via Enhanced Skin Permeation and Retention. Int. J. Pharm. 2019, 567, 118448. [Google Scholar] [CrossRef]
- Sivadasan, D.; Madkhali, O.A. The Design Features, Quality by Design Approach, Characterization, Therapeutic Applications, and Clinical Considerations of Transdermal Drug Delivery Systems—A Comprehensive Review. Pharmaceuticals 2024, 17, 1346. [Google Scholar] [CrossRef]
- Kaur, P.; Muskan; Kriplani, P. Quality by Design for Niosome-Based Nanocarriers to Improve Transdermal Drug Delivery from Lab to Industry. Int. J. Pharm. 2024, 666, 124747. [Google Scholar] [CrossRef] [PubMed]
- Cheng, T.; Tai, Z.; Shen, M.; Li, Y.; Yu, J.; Wang, J.; Zhu, Q.; Chen, Z. Advance and Challenges in the Treatment of Skin Diseases with the Transdermal Drug Delivery System. Pharmaceutics 2023, 15, 2165. [Google Scholar] [CrossRef] [PubMed]
- Schlich, M.; Musazzi, U.M.; Campani, V.; Biondi, M.; Franzé, S.; Lai, F.; De Rosa, G.; Sinico, C.; Cilurzo, F. Design and Development of Topical Liposomal Formulations in a Regulatory Perspective. Drug Deliv. Transl. Res. 2022, 12, 1811–1828. [Google Scholar] [CrossRef] [PubMed]
- Food and Drug Administration (FDA). Drug Products, Including Biological Products, That Contain Nanomaterials—Guidance for Industry; FDA: Silver Spring, MD, USA, 2017. [Google Scholar]
- European Medicines Agency (EMA). Reflection Paper on Surface Coating: General Issues for Consideration Regarding Parenteral Administration of Coated Nanomedicine Products; EMA: Amsterdam, The Netherlands, 2013. [Google Scholar]
- European Medicine Agency (EMA). Reflection Paper on the Data Requirements for Intravenous Liposomal Products Developed with Reference to an Innovator Liposomal Product; EMA: Amsterdam, The Netherlands, 2013. [Google Scholar]
- Food and Drug Administration (FDA). Liposome Drug Products Chemistry, Manufacturing, and Controls; Human Pharmacokinetics and Bioavailability, and Labeling Documentation Guidance for Industry; FDA: Silver Spring, MD, USA, 2018. [Google Scholar]
- European Medicines Agency (EMA). Draft Guideline on Quality and Equivalence of Topical Products; EMA: Amsterdam, The Netherlands, 2018. [Google Scholar]
- Food and Drug Administration (FDA). Transdermal and Topical Delivery Systems—Product Development and Quality Considerations; FDA: Silver Spring, MD, USA, 2019. [Google Scholar]







| Vesicular Type | Physicochemical Characteristics | API | Application | Key Findings | Reference |
|---|---|---|---|---|---|
| Liposomes |
| Avanafil | Erectile dysfunction | Four-fold increase in permeation and seven-fold in bioavailability compared to the conventional suspensions | [33] |
| Vitamin D3 | UV-protective, antiaging, and regenerative agents | Increased retention of Vitamin D3 in skin; improved skin appearance | [34] | |
| Folic acid | UV-protective, antiaging, and regenerative agents | Liposome stabilization at room temperature; transdermal penetration enhancement | [35] | |
| Lidocaine (LD)/cannabidiol (CBD) | Pain relief | Improved skin penetration; 1-year storage stability | [36] | |
| Solid Lipid Nanoparticles (SLNs) |
| Cyclosporine A and calcipotriol | Psoriasis and inflammation treatment | Low scores of eye inflammation and psoriatic symptoms | [15] |
| Metformin | Topical anti-inflammatory | Enhanced penetration to deeper skin layers | [37] | |
| Amphotericin B | Antifungal therapy | Enhanced efficacy and reduced adverse events | [38] | |
| Eugenol | Antifungal therapy | Enhanced efficacy and reduced adverse events | [39] | |
| Fluconazole | Antifungal therapy | Enhanced efficacy and reduced adverse events | ||
| Sesamol | Antioxidant activity for thetreatment of skin cancer | Enhanced bioavailability up to the desired anticancer effect | [40] | |
| Silybin | Antioxidant activity against irritant contact dermatitis | Enhanced skin delivery and therapeutic efficacy in treating irritant contact dermatitis compared to conventional formulations | [41] | |
| Nanostructured Lipid Carriers (NLCs) |
| Calcipotriol and methotrexate | Psoriasis treatment | Enhanced skin permeation and negligible irritation | [42] |
| Acitretin | Psoriasis treatment | Improved therapeutic response (reduced erythema) with mild adverse effects versus commercial gel | [43] | |
| Flurbiprofen | Treatment of osteoarthritis, rheumatoidarthritis | Reduction in erythema along with maintenance of the anti-inflammatory effect versus the commercial gel | [44] | |
| Difluctolone valerate | Treatment of skin diseases such as psoriasis, acne, tinea, ulcers, and discoid lupus erythematosus | High targeting of the API in the stratum corneum (SC) and limited systemic side effects versus Nerisone® cream | [45] | |
| Luliconazole | Antifungal therapy | Enhanced antifungal activity with no irritation compared to the commercial formulation | [46] | |
| Donepezil | Alzheimer disease | Higher skin permeation with limited adverse effects | [47] | |
| Ethosomes |
| Psoralen | Psoriasis treatment | 6.56× higher deposition vs. control | [48] |
| Methotrexate | Psoriasis and cancer therapy | Improved efficacy, lower toxicity | [49] | |
| Erythromycin | Anti-bacterial treatment | Significantly improved anti-bacterial action vs. free drug in in vitro tests; non-toxic to dermal cultured fibroblasts | [50] | |
| Acyclovir | Anti-viral treatment | Enhanced permeability of ethosome formulations | [51] | |
| Ligustrazine | Pulmonary treatment | AUC 2.09-fold increase vs. oral; 209% relative bioavailability | [52] | |
| Transfersomes |
| Triamcinolone acetonide | Corticosteroid | 25× lower dose for same efficacy | [53] |
| Dexamethasone | Corticosteroid | Improved transdermal delivery, prolonged release with no lag time | [54] | |
| No data available due to patent restrictions | Insulin | Diabetes | Comparable levels to subcutaneous injections | [55] |
| Lipid-Based Formulation | API | Indication | Added Value | Clinical Status | Reference |
|---|---|---|---|---|---|
| Liposomes + microneedles | Insulin | Diabetes | Preclinical studies | [75] | |
| SLNs | Lidocaine | Local anesthesia | Longer local anesthesia | Preclinical studies | [74] |
| Transfersomes | Diclofenac | Anti-inflammatory | Increased tissue concentration and permeation | Clinical study | [71,72] |
| NLCs | Estradiol | Hormone therapy | 92.5% symptom relief, hormone normalization | Preclinical studies | [73] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Antonara, L.; Triantafyllopoulou, E.; Chountoulesi, M.; Pippa, N.; Dallas, P.P.; Rekkas, D.M. Lipid-Based Drug Delivery Systems: Concepts and Recent Advances in Transdermal Applications. Nanomaterials 2025, 15, 1326. https://doi.org/10.3390/nano15171326
Antonara L, Triantafyllopoulou E, Chountoulesi M, Pippa N, Dallas PP, Rekkas DM. Lipid-Based Drug Delivery Systems: Concepts and Recent Advances in Transdermal Applications. Nanomaterials. 2025; 15(17):1326. https://doi.org/10.3390/nano15171326
Chicago/Turabian StyleAntonara, Lefkothea, Efstathia Triantafyllopoulou, Maria Chountoulesi, Natassa Pippa, Paraskevas P. Dallas, and Dimitrios M. Rekkas. 2025. "Lipid-Based Drug Delivery Systems: Concepts and Recent Advances in Transdermal Applications" Nanomaterials 15, no. 17: 1326. https://doi.org/10.3390/nano15171326
APA StyleAntonara, L., Triantafyllopoulou, E., Chountoulesi, M., Pippa, N., Dallas, P. P., & Rekkas, D. M. (2025). Lipid-Based Drug Delivery Systems: Concepts and Recent Advances in Transdermal Applications. Nanomaterials, 15(17), 1326. https://doi.org/10.3390/nano15171326









