Nanomaterial Functionalized Carbon Fiber-Reinforced Composites with Energy Storage Capabilities
Abstract
1. Introduction
2. Experimental Work
2.1. Materials
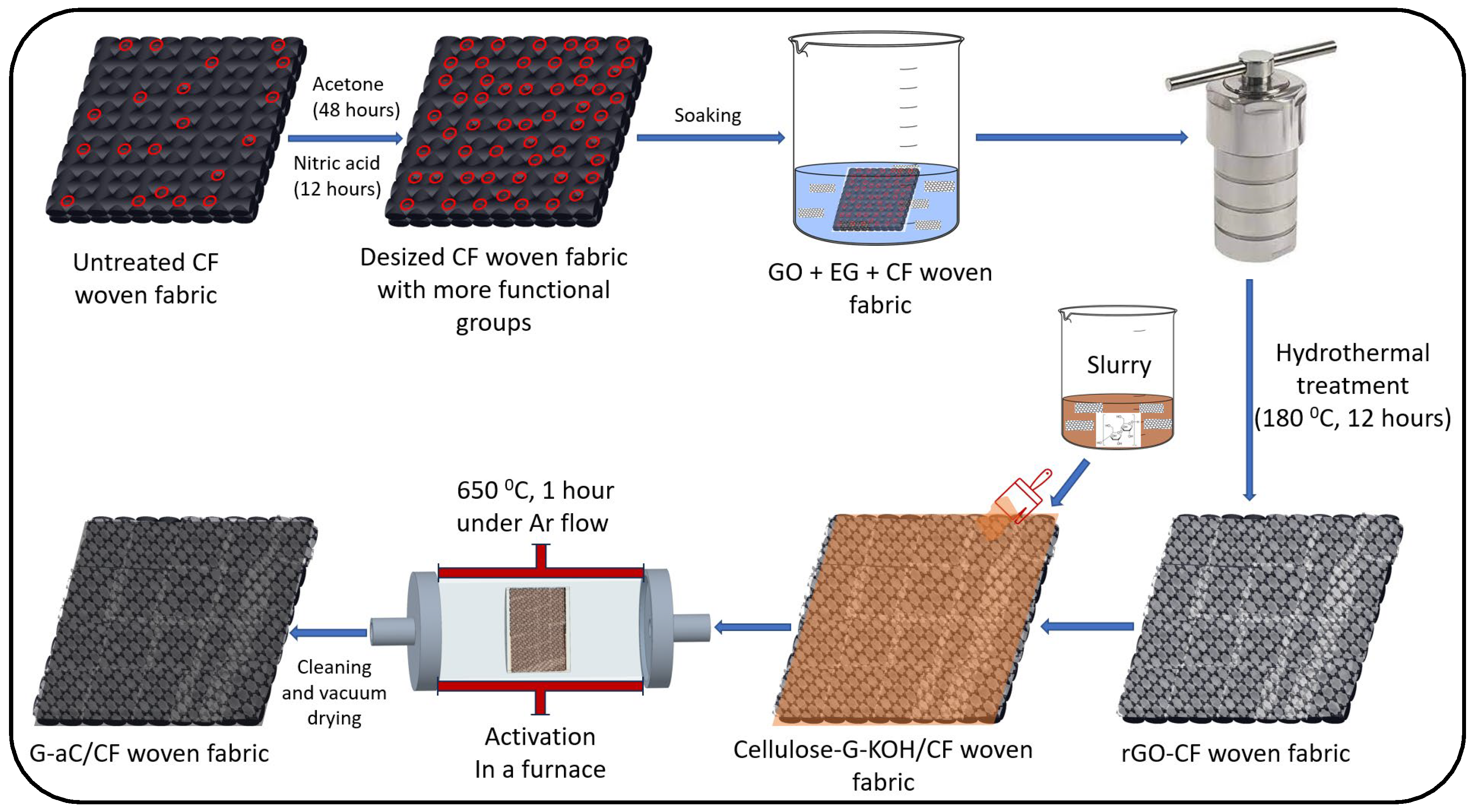
2.2. Graphene Oxide Coating on Carbon Fiber Fabrics (rGO/CF)
2.3. Graphene, Cellulose, and KOH Slurry Preparation
2.4. Preparation of CF Fabric Electrodes with Graphene and Activated Carbon
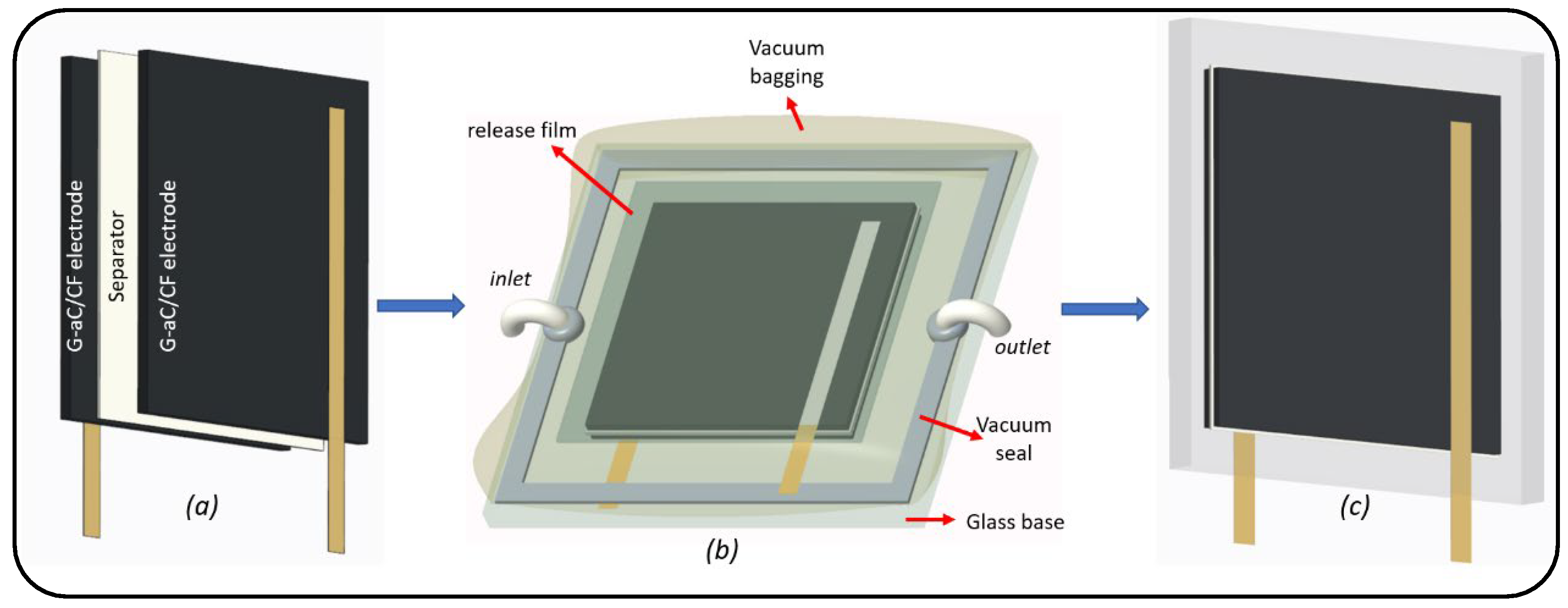
2.5. Fabrication of Symmetrical Structural Supercapacitor (SSSC)
2.6. Material Characterizations
2.7. Electrochemical Measurements
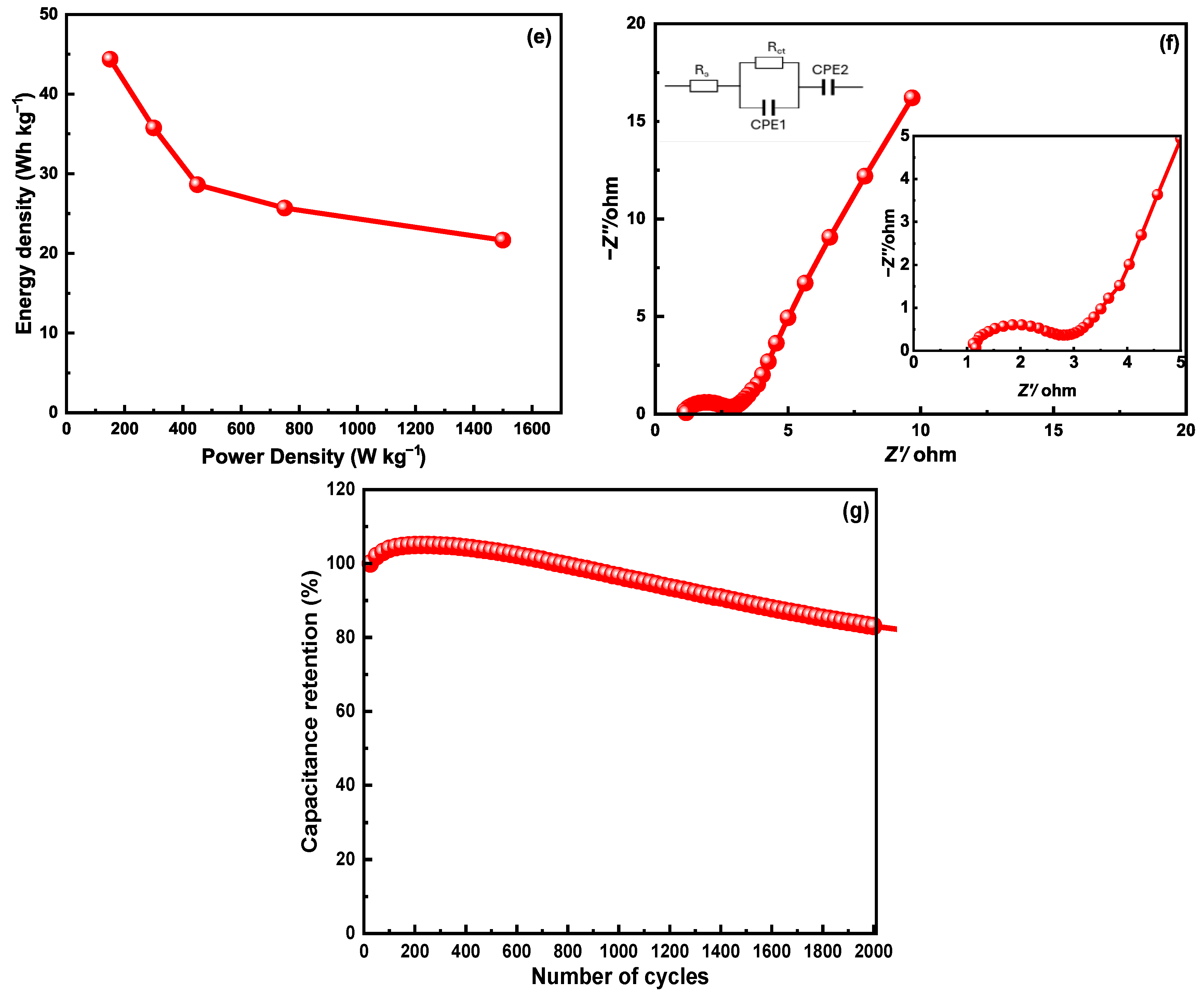
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Data Availability Statement
Conflicts of Interest
Correction Statement
Abbreviations
| G | Graphene |
| GO | Graphene oxide |
| rGO | Reduced graphene oxide |
| LED | Light-emitting diode |
| SSC | Structural supercapacitor |
| SC | Supercapacitor |
| ESD | Energy storage device |
| ESC | Energy storage composites |
| EDLC | Electric double layer capacitor |
| CF | Carbon fiber |
| GA | Graphene aerogel |
| CFRP | Carbon fiber-reinforced polymer |
| CNT | Carbon nanotubes |
| PAN | Polyacrylonitrile |
| PPy | Polypyrrole |
| CAG | Carbon Aerogel |
| GNP | Graphene nanoplatelets |
| aC | Activated carbon |
| HCL | Hydrochloric acid |
| KOH | Potassium hydroxide |
| EG | Ethylene glycol |
| PEGDGE | Polyethylene glycol diglycidyl ether |
| TETA | Triethylenetetramine |
| EMIMTFSI | 1-Ethyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide |
| LiFSI | lithium bis(fluorosulfonyl)imide |
| VRTM | vacuum-assisted resin transfer molding |
| SEM | Scanning Electron Microscope |
| CV | Cyclic voltammetry |
| BET | Brunauer–Emmett–Teller |
| XPS | X-ray photoelectron spectroscopy |
References
- Carlstedt, D.; Asp, L.E. Performance analysis framework for structural battery composites in electric vehicles. Compos. Part B Eng. 2020, 186, 107822. [Google Scholar] [CrossRef]
- Akbar, F. Weight Optimisation of Electric Vehicle through Hybrid Structural Batteries. Int. J. Automot. Mech. Eng. 2020, 17, 8310–8325. [Google Scholar] [CrossRef]
- Zhou, H.; Su, Y.; Zhang, J.; Li, H.; Zhou, L.; Huang, H. A novel embedded all-solid-state composite structural supercapacitor based on activated carbon fiber electrode and carbon fiber reinforced polymer matrix. Chem. Eng. J. 2023, 454, 140222. [Google Scholar] [CrossRef]
- Lamba, P.; Singh, P.; Singh, P.; Singh, P.; Bharti; Kumar, A.; Gupta, M.; Kumar, Y. Recent advancements in supercapacitors based on different electrode materials: Classifications, synthesis methods and comparative performance. J. Energy Storage 2022, 48, 103871. [Google Scholar] [CrossRef]
- Ahmad, F.; Zahid, M.; Jamil, H.; Khan, M.A.; Atiq, S.; Bibi, M.; Shahbaz, K.; Adnan, M.; Danish, M.; Rasheed, F. Advances in graphene-based electrode materials for high-performance supercapacitors: A review. J. Energy Storage 2023, 72, 108731. [Google Scholar] [CrossRef]
- Wang, K.; Wang, Z.; Wang, C.; Zhang, X.; Wu, L. Carbon fiber electrodes for composite structural supercapacitor: Preparation and modification methods. J. Energy Storage 2024, 98, 113129. [Google Scholar] [CrossRef]
- Wu, K.-J.; Young, W.-B.; Young, C. Structural supercapacitors: A mini-review of their fabrication, mechanical & electrochemical properties. J. Energy Storage 2023, 72, 108358. [Google Scholar] [CrossRef]
- Asp, L.E.; Johansson, M.; Lindbergh, G.; Xu, J.; Zenkert, D. Structural battery composites: A review. Funct. Compos. Struct. 2019, 1, 042001. [Google Scholar] [CrossRef]
- Cao, Y.; Sun, S.; Li, W.; Chang, Y.; Gu, N.; Yao, J.; Sun, Y. Challenge and technological trends of flexible solid-state supercapacitors. J. Energy Storage 2024, 97, 112837. [Google Scholar] [CrossRef]
- Galos, J.; Pattarakunnan, K.; Best, A.S.; Kyratzis, I.L.; Wang, C.H.; Mouritz, A.P. Energy Storage Structural Composites with Integrated Lithium-Ion Batteries: A Review. Adv. Mater. Technol. 2021, 6, 2001059. [Google Scholar] [CrossRef]
- Subhani, K.; Hameed, N.; Al-Qatatsheh, A.; Ince, J.; Mahon, P.J.; Lau, A.; Salim, N.V. Multifunctional structural composite supercapacitors based on MnO2-nanowhiskers decorated carbon fibers. J. Energy Storage 2022, 56, 105936. [Google Scholar] [CrossRef]
- Kötz, R.; Carlen, M. Principles and applications of electrochemical capacitors. Electrochim. Acta 2000, 45, 2483–2498. [Google Scholar] [CrossRef]
- Xu, Y.; Lu, W.; Xu, G.; Chou, T.-W. Structural supercapacitor composites: A review. Compos. Sci. Technol. 2021, 204, 108636. [Google Scholar] [CrossRef]
- Asfaw, H.D.; Kucernak, A.; Greenhalgh, E.S.; Shaffer, M.S. Electrochemical performance of supercapacitor electrodes based on carbon aerogel-reinforced spread tow carbon fiber fabrics. Compos. Sci. Technol. 2023, 238, 110042. [Google Scholar] [CrossRef]
- Raza, M.A.; Yousaf, M.I.; Akram, S.; Siddique, A.; Ashraf, M.; Fu, D. High-performance supercapacitor based on NiCo2S4/g-C3N4/polyaniline deposited on carbon cloth for structural stability. Synth. Met. 2024, 301, 117534. [Google Scholar] [CrossRef]
- Greenhalgh, E.S.; Nguyen, S.; Valkova, M.; Shirshova, N.; Shaffer, M.S.; Kucernak, A. A critical review of structural supercapacitors and outlook on future research challenges. Compos. Sci. Technol. 2023, 235, 109968. [Google Scholar] [CrossRef]
- Nayan, R.; Sinha, S.; Dixit, V.; Satnami, M.L.; Ghosh, K.K.; Pervez, S.; Deb, M.K.; Shrivas, K.; Rai, M.K.; Yenchalwar, S.G. PANI-grafted boron, nitrogen co-doped carbon fiber: An outstanding, high-performance supercapacitor electrode. J. Energy Storage 2024, 96, 112668. [Google Scholar] [CrossRef]
- Chen, Q.; Zhang, L.; Rahman, A.; Zhou, Z.; Wu, X.-F.; Fong, H. Hybrid multi-scale epoxy composite made of conventional carbon fiber fabrics with interlaminar regions containing electrospun carbon nanofiber mats. Compos. Part A Appl. Sci. Manuf. 2011, 42, 2036–2042. [Google Scholar] [CrossRef]
- Zeng, L.; Sun, J.; Zhao, T.; Ren, Y.; Wei, L. Balancing the specific surface area and mass diffusion property of electrospun carbon fibers to enhance the cell performance of vanadium redox flow battery. Int. J. Hydrogen Energy 2020, 45, 12565–12576. [Google Scholar] [CrossRef]
- Birch, M.E.; Ruda-Eberenz, T.A.; Chai, M.; Andrews, R.; Hatfield, R.L. Properties that influence the specific surface areas of carbon nanotubes and nanofibers. Ann. Occup. Hyg. 2013, 57, 1148–1166. [Google Scholar]
- Ding, Y.; Qi, G.; Cui, Q.; Yang, J.; Zhang, B.; Du, S. High-Performance Multifunctional Structural Supercapacitors Based on In Situ and Ex Situ Activated-Carbon-Coated Carbon Fiber Electrodes. Energy Fuels 2022, 36, 2171–2178. [Google Scholar] [CrossRef]
- Lingappan, N.; Lim, S.; Lee, G.-H.; Tung, H.T.; Lee, W. Recent advances on fiber-reinforced multifunctional composites for structural supercapacitors. Funct. Compos. Struct. 2022, 4, 012001. [Google Scholar] [CrossRef]
- Senokos, E.; Anthony, D.B.; Rubio, N.; Ribadeneyra, M.C.; Greenhalgh, E.S.; Shaffer, M.S. Robust Single-Walled Carbon Nanotube-Infiltrated Carbon Fiber Electrodes for Structural Supercapacitors: From Reductive Dissolution to High Performance Devices. Adv. Funct. Mater. 2023, 33, 2212697. [Google Scholar] [CrossRef]
- Qian, H.; Kucernak, A.R.; Greenhalgh, E.S.; Bismarck, A.; Shaffer, M.S. Multifunctional structural supercapacitor composites based on carbon aerogel modified high performance carbon fiber fabric. ACS Appl. Mater. Interfaces 2013, 5, 6113–6122. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Romate, X.F.; Del Bosque, A.; Artigas-Arnaudas, J.; Muñoz, B.K.; Sánchez, M.; Ureña, A. A proof of concept of a structural supercapacitor made of graphene coated woven carbon fibers: EIS study and mechanical performance. Electrochim. Acta 2021, 370, 137746. [Google Scholar] [CrossRef]
- Almeida, D.A.L.; Couto, A.; Ferreira, N.G. Flexible polyaniline/reduced graphene oxide/carbon fiber composites applied as electrodes for supercapacitors. J. Alloys Compd. 2019, 788, 453–460. [Google Scholar] [CrossRef]
- Javaid, A.; Irfan, M. Multifunctional structural supercapacitors based on graphene nanoplatelets/carbon aerogel composite coated carbon fiber electrodes. Mater. Res. Express 2018, 6, 016310. [Google Scholar] [CrossRef]
- Subhani, K.; Jin, X.; Mahon, P.J.; Lau, A.K.T.; Salim, N.V. Graphene aerogel modified carbon fiber reinforced composite structural supercapacitors. Compos. Commun. 2021, 24, 100663. [Google Scholar] [CrossRef]
- Islam, M.S.; Faisal, S.N.; Tong, L.; Roy, A.K.; Zhang, J.; Haque, E.; Minett, A.I.; Wang, C.H. N-doped reduced graphene oxide (rGO) wrapped carbon microfibers as binder-free electrodes for flexible fibre supercapacitors and sodium-ion batteries. J. Energy Storage 2021, 37, 102453. [Google Scholar] [CrossRef]
- Qian, H.; Diao, H.; Shirshova, N.; Greenhalgh, E.S.; Steinke, J.G.; Shaffer, M.S.; Bismarck, A. Activation of structural carbon fibres for potential applications in multifunctional structural supercapacitors. J. Colloid Interface Sci. 2013, 395, 241–248. [Google Scholar] [CrossRef]
- Radhakrishnan, S.; Kim, D.-Y.; Yu, S.; Kim, J.-W.; Lee, K.-y.; Jeong, I.Y.; Kim, B.-S. A flexible, binder-free electrodes using nickel-cobalt sulfur modified carbon nanofibers for asymmetric supercapacitor applications. Electrochim. Acta 2024, 498, 144683. [Google Scholar] [CrossRef]
- Javaid, A.; Khalid, O.; Shakeel, A.; Noreen, S. Multifunctional structural supercapacitors based on polyaniline deposited carbon fiber reinforced epoxy composites. J. Energy Storage 2021, 33, 102168. [Google Scholar] [CrossRef]
- Zhang, J.; Seyedin, S.; Gu, Z.; Salim, N.; Wang, X.; Razal, J.M. Liquid Crystals of Graphene Oxide: A Route Towards Solution-Based Processing and Applications. Part. Part. Syst. Charact. 2017, 34, 16000396. [Google Scholar] [CrossRef]
- Reenu; Sonia; Phor, L.; Kumar, A.; Chahal, S. Electrode materials for supercapacitors: A comprehensive review of advancements and performance. J. Energy Storage 2024, 84, 110698. [Google Scholar] [CrossRef]
- Farjadian, F.; Abbaspour, S.; Sadatlu, M.A.A.; Mirkiani, S.; Ghasemi, A.; Hoseini-Ghahfarokhi, M.; Mozaffari, N.; Karimi, M.; Hamblin, M.R. Recent developments in graphene and graphene oxide: Properties, synthesis, and modifications: A review. ChemistrySelect 2020, 5, 10200–10219. [Google Scholar] [CrossRef]
- Johra, F.T.; Jung, W.-G. Hydrothermally reduced graphene oxide as a supercapacitor. Appl. Surf. Sci. 2015, 357, 1911–1914. [Google Scholar] [CrossRef]
- Yuan, S.; Fan, W.; Jin, Y.; Wang, D.; Liu, T. Free-standing flexible graphene-based aerogel film with high energy density as an electrode for supercapacitors. Nano Mater. Sci. 2021, 3, 68–74. [Google Scholar] [CrossRef]
- Ahmad, N.; Rinaldi, A.; Sidoli, M.; Magnani, G.; Morenghi, A.; Scaravonati, S.; Vezzoni, V.; Pasetti, L.; Fornasini, L.; Ridi, F.; et al. High performance quasi-solid-state supercapacitor based on activated carbon derived from asparagus waste. J. Energy Storage 2024, 99, 113267. [Google Scholar] [CrossRef]
- Sagadevan, S.; Balakrishnan, T.; Rahman, M.Z.; Soga, T.; Randriamahazaka, H.; Kakavandi, B.; Johan, M.R. Agricultural biomass-based activated carbons for efficient and sustainable supercapacitors. J. Energy Storage 2024, 97, 112878. [Google Scholar] [CrossRef]
- Atchudan, R.; Perumal, S.; Sundramoorthy, A.; Manoj, D.; Kumar, R.; Almansour, A.; Lee, Y. Facile Synthesis of Functionalized Porous Carbon by Direct Pyrolysis of Anacardium occidentale Nut-Skin Waste and Its Utilization towards Supercapacitors. Nanomaterials 2023, 13, 1654. [Google Scholar] [CrossRef]
- Sun, Z.; Zhang, M.; Yin, H.; Hu, Q.; Krishnan, S.; Huang, Z.; Qi, H.; Wang, X. Tailoring hierarchically porous structure of biomass-derived carbon for high-performance supercapacitors. Renew. Energy 2023, 219, 119375. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, Y.; Zhao, H.; Li, L.; Ju, D.; Wang, C.; An, B. Biomass-derived porous carbon with a good balance between high specific surface area and mesopore volume for supercapacitors. Nanomaterials 2022, 12, 3804. [Google Scholar] [CrossRef] [PubMed]
- Rajivgandhi, P.; Thirumal, V.; Sekar, A.; Kim, J. Biomass-Derived Activated Porous Carbon from Foxtail Millet Husk to Utilizing High-Performance Symmetric Supercapacitor Applications. Nanomaterials 2025, 15, 575. [Google Scholar] [CrossRef] [PubMed]
- Nazhipkyzy, M.; Yeleuov, M.; Sultakhan, S.T.; Maltay, A.B.; Zhaparova, A.A.; Assylkhanova, D.D.; Nemkayeva, R.R. Electrochemical performance of chemically activated carbons from sawdust as supercapacitor electrodes. Nanomaterials 2022, 12, 3391. [Google Scholar] [CrossRef]
- Li, X.; Tang, Y.; Song, J.; Yang, W.; Wang, M.; Zhu, C.; Zhao, W.; Zheng, J.; Lin, Y. Self-supporting activated carbon/carbon nanotube/reduced graphene oxide flexible electrode for high performance supercapacitor. Carbon 2018, 129, 236–244. [Google Scholar] [CrossRef]
- Singh, S.; Sahoo, R.K.; Shinde, N.M.; Yun, J.M.; Mane, R.S.; Kim, K.H. Synthesis of Bi2O3-MnO2 nanocomposite electrode for wide-potential window high performance supercapacitor. Energies 2019, 12, 3320. [Google Scholar] [CrossRef]
- Afroze, J.D.; Tong, L.; Abden, M.J.; Chen, Y. Multifunctional hierarchical graphene-carbon fiber hybrid aerogels for strain sensing and energy storage. Adv. Compos. Hybrid Mater. 2023, 6, 18. [Google Scholar] [CrossRef]
- Ishfaq, A.; Nguyen, S.N.; Greenhalgh, E.S.; Shaffer, M.S.; Kucernak, A.R.; Asp, L.E.; Zenkert, D.; Linde, P. Multifunctional design, feasibility and requirements for structural power composites in future electric air taxis. J. Compos. Mater. 2023, 57, 817–828. [Google Scholar] [CrossRef]
- Yu, S.; Li, Y.; Pan, N. KOH activated carbon/graphene nanosheets composites as high performance electrode materials in supercapacitors. RSC Adv. 2014, 4, 48758–48764. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, S.; Wen, X.; Chen, X.; Wen, Y.; Shi, X.; Mijowska, E. High yield conversion of biowaste coffee grounds into hierarchical porous carbon for superior capacitive energy storage. Sci. Rep. 2020, 10, 3518. [Google Scholar] [CrossRef]
- Pandey, D.; Sambath Kumar, K.; Henderson, L.N.; Suarez, G.; Vega, P.; Salvador, H.R.; Roberson, L.; Thomas, J. Energized Composites for Electric Vehicles: A Dual Function Energy-Storing Supercapacitor-Based Carbon Fiber Composite for the Body Panels. Small 2022, 18, 2107053. [Google Scholar] [CrossRef]
- Wu, S.; Chen, G.; Kim, N.Y.; Ni, K.; Zeng, W.; Zhao, Y.; Tao, Z.; Ji, H.; Lee, Z.; Zhu, Y. Creating Pores on Graphene Platelets by Low-Temperature KOH Activation for Enhanced Electrochemical Performance. Small 2016, 12, 2376–2384. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, X.; Zhang, H.; Sun, X.; Zhang, D.; Ma, Y. High-performance supercapacitors based on a graphene–activated carbon composite prepared by chemical activation. RSC Adv. 2012, 2, 7747–7753. [Google Scholar] [CrossRef]
- Ma, C.; Fan, Q.; Dirican, M.; Subjalearndee, N.; Cheng, H.; Li, J.; Song, Y.; Shi, J.; Zhang, X. Rational design of meso-/micro-pores for enhancing ion transportation in highly-porous carbon nanofibers used as electrode for supercapacitors. Appl. Surf. Sci. 2021, 545, 148933. [Google Scholar] [CrossRef]
- Mendoza, R.; Oliva, J.; Rodriguez-Gonzalez, V. Effect of the micro-, meso-and macropores on the electrochemical performance of supercapacitors: A review. Int. J. Energy Res. 2022, 46, 6989–7020. [Google Scholar] [CrossRef]
- Suriyakumar, S.; Bhardwaj, P.; Grace, A.N.; Stephan, A.M. Role of polymers in enhancing the performance of electrochemical supercapacitors: A review. Batter. Supercaps 2021, 4, 571–584. [Google Scholar] [CrossRef]
- Yin, C.; Tao, C.-a.; Cai, F.; Song, C.; Gong, H.; Wang, J. Effects of activation temperature on the deoxygenation, specific surface area and supercapacitor performance of graphene. Carbon 2016, 109, 558–565. [Google Scholar] [CrossRef]
- Raj, C.J.; Rajesh, M.; Manikandan, R.; Lee, W.-g.; Yu, K.H.; Kim, B.C. Direct fabrication of two-dimensional copper sulfide nanoplates on transparent conducting glass for planar supercapacitor. J. Alloys Compd. 2018, 735, 2378–2383. [Google Scholar] [CrossRef]
- Okhay, O.; Tkach, A. Graphene/reduced graphene oxide-carbon nanotubes composite electrodes: From capacitive to battery-type behaviour. Nanomaterials 2021, 11, 1240. [Google Scholar] [CrossRef] [PubMed]
- Zhu, D.; Hou, J.; Zhang, L.; Gao, Y.; Dai, B.; Lian, Y.; Yan, H.; Zhang, H. Microbial porous carbon by low-alkali activation for flexible supercapacitors. J. Electron. Mater. 2021, 50, 6733–6740. [Google Scholar] [CrossRef]
- Wang, C.; Yuan, X.; Guo, G.; Liang, H.; Ma, Z.; Li, P. Salt template tuning morphology and porosity of biomass-derived N-doped porous carbon with high redox-activation for efficient energy storage. Colloids Surf. A Physicochem. Eng. Asp. 2022, 650, 129552. [Google Scholar] [CrossRef]
- Babaahmadi, V.; Pourhosseini, S.; Norouzi, O.; Naderi, H.R. Designing 3D ternary hybrid composites composed of graphene, biochar and manganese dioxide as high-performance supercapacitor electrodes. Nanomaterials 2023, 13, 1866. [Google Scholar] [CrossRef]
- Hubert, O.; Todorovic, N.; Bismarck, A. Towards separator-free structural composite supercapacitors. Compos. Sci. Technol. 2022, 217, 109126. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, G.; Huang, Z.; Li, Y.; Yang, J.; Long, Z. A high potential (2.5 V) carbon fiber paper-based asymmetric supercapacitor with oxygen vacancies and hierarchical porous hollow structure. Chem. Eng. J. 2024, 496, 154165. [Google Scholar] [CrossRef]
- Li, X.; Wang, J.; Zhao, Y.; Ge, F.; Komarneni, S.; Cai, Z. Wearable solid-state supercapacitors operating at high working voltage with a flexible nanocomposite electrode. ACS Appl. Mater. Interfaces 2016, 8, 25905–25914. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Xuan, H.; Lin, G.; Wang, F.; Chen, Z.; Dong, X. A melamine-assisted chemical blowing synthesis of N-doped activated carbon sheets for supercapacitor application. J. Power Sources 2016, 319, 262–270. [Google Scholar] [CrossRef]
- Nunes, W.G.; Freitas, B.G.; Beraldo, R.M.; Filho, R.M.; Da Silva, L.M.; Zanin, H. A rational experimental approach to identify correctly the working voltage window of aqueous-based supercapacitors. Sci. Rep. 2020, 10, 19195. [Google Scholar] [CrossRef]
- Ganguly, A.; Karakassides, A.; Benson, J.; Hussain, S.; Papakonstantinou, P. Multifunctional structural supercapacitor based on urea-activated graphene nanoflakes directly grown on carbon fiber electrodes. ACS Appl. Energy Mater. 2020, 3, 4245–4254. [Google Scholar] [CrossRef]
- Pernice, M.F.; Qi, G.; Senokos, E.; Anthony, D.B.; Nguyen, S.; Valkova, M.; Greenhalgh, E.S.; Shaffer, M.S.; Kucernak, A.R. Mechanical, electrochemical and multifunctional performance of a CFRP/carbon aerogel structural supercapacitor and its corresponding monofunctional equivalents. Multifunct. Mater. 2022, 5, 025002. [Google Scholar] [CrossRef]
- Zhao, Y.; Xu, H.; Cai, G.; Yan, C.; Liu, D.; Chen, G.; Zhu, Y. Multi-functional structural supercapacitor based on manganese oxide-hydroxide nanowires modified carbon fiber fabric electrodes. Polym. Compos. 2022, 43, 8458–8470. [Google Scholar] [CrossRef]
- Deka, B.K.; Hazarika, A.; Kim, J.; Park, Y.-B.; Park, H.W. Multifunctional CuO nanowire embodied structural supercapacitor based on woven carbon fiber/ionic liquid–polyester resin. Compos. Part A Appl. Sci. Manuf. 2016, 87, 256–262. [Google Scholar] [CrossRef]
- Qi, G.; Nguyen, S.; Anthony, D.B.; Kucernak, A.R.; Shaffer, M.S.; Greenhalgh, E.S. The influence of fabrication parameters on the electrochemical performance of multifunctional structural supercapacitors. Multifunct. Mater. 2021, 4, 034001. [Google Scholar] [CrossRef]
- Gao, B.; Zhang, R.; He, M.; Sun, L.; Wang, C.; Liu, L.; Zhao, L.; Cui, H.; Cao, A. Effect of a multiscale reinforcement by carbon fiber surface treatment with graphene oxide/carbon nanotubes on the mechanical properties of reinforced carbon/carbon composites. Compos. Part A Appl. Sci. Manuf. 2016, 90, 433–440. [Google Scholar] [CrossRef]
- Sanchez, J.S.; Xu, J.; Xia, Z.; Sun, J.; Asp, L.E.; Palermo, V. Electrophoretic coating of LiFePO4/Graphene oxide on carbon fibers as cathode electrodes for structural lithium ion batteries. Compos. Sci. Technol. 2021, 208, 108768. [Google Scholar] [CrossRef]
- Xiong, C.; Li, T.; Zhao, T.; Dang, A.; Li, H.; Ji, X.; Jin, W.; Jiao, S.; Shang, Y.; Zhang, Y. Reduced graphene oxide-carbon nanotube grown on carbon fiber as binder-free electrode for flexible high-performance fiber supercapacitors. Compos. Part B Eng. 2017, 116, 7–15. [Google Scholar] [CrossRef]
- Kaniyoor, A.; Ramaprabhu, S. A Raman spectroscopic investigation of graphite oxide derived graphene. AIP Adv. 2012, 2, 032183. [Google Scholar] [CrossRef]
- Chen, J.; Wu, J.; Ge, H.; Zhao, D.; Liu, C.; Hong, X. Reduced graphene oxide deposited carbon fiber reinforced polymer composites for electromagnetic interference shielding. Compos. Part A Appl. Sci. Manuf. 2016, 82, 141–150. [Google Scholar] [CrossRef]
- Jiang, G.; Pickering, S.J. Structure–property relationship of recycled carbon fibres revealed by pyrolysis recycling process. J. Mater. Sci. 2016, 51, 1949–1958. [Google Scholar] [CrossRef]
- Dobiášová, L.; Starý, V.; Glogar, P.; Valvoda, V. Analysis of carbon fibers and carbon composites by asymmetric X-ray diffraction technique. Carbon 1999, 37, 421–425. [Google Scholar] [CrossRef]
- Yao, Z.; Wang, C.; Lu, R.; Su, S.; Qin, J.; Wang, Y.; Ma, Z.; Wei, H.; Wang, Q. Fracture investigation of functionalized carbon nanotubes-grown carbon fiber fabrics/epoxy composites. Compos. Sci. Technol. 2020, 195, 108161. [Google Scholar] [CrossRef]
- Emiru, T.F.; Ayele, D.W. Controlled synthesis, characterization and reduction of graphene oxide: A convenient method for large scale production. Egypt. J. Basic Appl. Sci. 2017, 4, 74–79. [Google Scholar] [CrossRef]
- Cen, T.; Chen, L.; Zhang, X.; Tian, Y.; Fan, X. A novel fiber-shaped asymmetric supercapacitor prepared by twisting carbon fiber/carbon nanotube/MnO2 and carbon fiber/carbon nanotube/polypyrrole electrodes+. Electrochim. Acta 2021, 367, 137488. [Google Scholar] [CrossRef]
- Folaranmi, G.; Bechelany, M.; Sistat, P.; Cretin, M.; Zaviska, F. Comparative investigation of activated carbon electrode and a novel activated carbon/graphene oxide composite electrode for an enhanced capacitive deionization. Materials 2020, 13, 5185. [Google Scholar] [CrossRef]
- Folaranmi, G.; Bechelany, M.; Sistat, P.; Cretin, M.; Zaviska, F. Activated carbon blended with reduced graphene oxide nanoflakes for capacitive deionization. Nanomaterials 2021, 11, 1090. [Google Scholar] [CrossRef]
- Allahbakhsh, A.; Sharif, F.; Mazinani, S.; Kalaee, M. Synthesis and characterization of graphene oxide in suspension and powder forms by chemical exfoliation method. Int. J. Nano Dimens. 2014, 5, 11–20. [Google Scholar]
- Baishya, P.; Maji, T.K. A comparative study on the properties of graphene oxide and activated carbon based sustainable wood starch composites. Int. J. Biol. Macromol. 2018, 115, 970–977. [Google Scholar] [CrossRef]
- Xue, X.; Lou, B.; Pang, W.; Han, S.; Zhang, J.; Wu, J.; Tian, L.; Shi, N.; Liu, D. The Impact of Defective Carbon Interface Engineering on the Lithiation Behavior of Silicon-Graphite Composite Electrodes. Carbon 2025, 238, 120255. [Google Scholar] [CrossRef]
- Dharmasiri, B.; Randall, J.; Yin, Y.; Andersson, G.G.; Doeven, E.H.; Francis, P.S.; Henderson, L.C. Carbon reinforced carbon fibers: Using surface modification as a route to enhanced physical performance. Compos. Sci. Technol. 2022, 218, 109217. [Google Scholar] [CrossRef]
- Zhang, H.; Li, A.; Wang, J.; Zhang, Y.; Zhao, Z.; Zhao, H.; Cheng, M.; Wang, C.; Wang, J.; Zhang, S.; et al. Graphene integrating carbon fiber and hierarchical porous carbon formed robust flexible “carbon-concrete” supercapacitor film. Carbon 2018, 126, 500–506. [Google Scholar] [CrossRef]
- Lee, W.; Lee, J.; Reucroft, P. XPS study of carbon fiber surfaces treated by thermal oxidation in a gas mixture of O2/(O2+ N2). Appl. Surf. Sci. 2001, 171, 136–142. [Google Scholar] [CrossRef]
- Woodhead, A.L.; de Souza, M.L.; Church, J.S. An investigation into the surface heterogeneity of nitric acid oxidized carbon fiber. Appl. Surf. Sci. 2017, 401, 79–88. [Google Scholar] [CrossRef]
- Al-Gaashani, R.; Najjar, A.; Zakaria, Y.; Mansour, S.; Atieh, M. XPS and structural studies of high quality graphene oxide and reduced graphene oxide prepared by different chemical oxidation methods. Ceram. Int. 2019, 45, 14439–14448. [Google Scholar] [CrossRef]
- Guo, Y.; Liu, Q.; Li, Z.; Zhang, Z.; Fang, X. Enhanced photocatalytic hydrogen evolution performance of mesoporous graphitic carbon nitride co-doped with potassium and iodine. Appl. Catal. B Environ. 2018, 221, 362–370. [Google Scholar] [CrossRef]
- Hao, Y.; Zhang, S.; Tao, P.; Shen, T.; Huang, Z.; Yan, J.; Chen, Y. Pillaring Effect of K Ion Anchoring for Stable V2O5-Based Zinc-Ion Battery Cathodes. ChemNanoMat 2020, 6, 797–805. [Google Scholar] [CrossRef]
- Barr, T.L. An XPS study of Si as it occurs in adsorbents, catalysts, and thin films. Appl. Surf. Sci. 1983, 15, 1–35. [Google Scholar] [CrossRef]
- Jensen, D.S.; Kanyal, S.S.; Madaan, N.; Vail, M.A.; Dadson, A.E.; Engelhard, M.H.; Linford, M.R. Silicon (100)/sio2 by xps. Surf. Sci. Spectra 2013, 20, 36–42. [Google Scholar] [CrossRef]





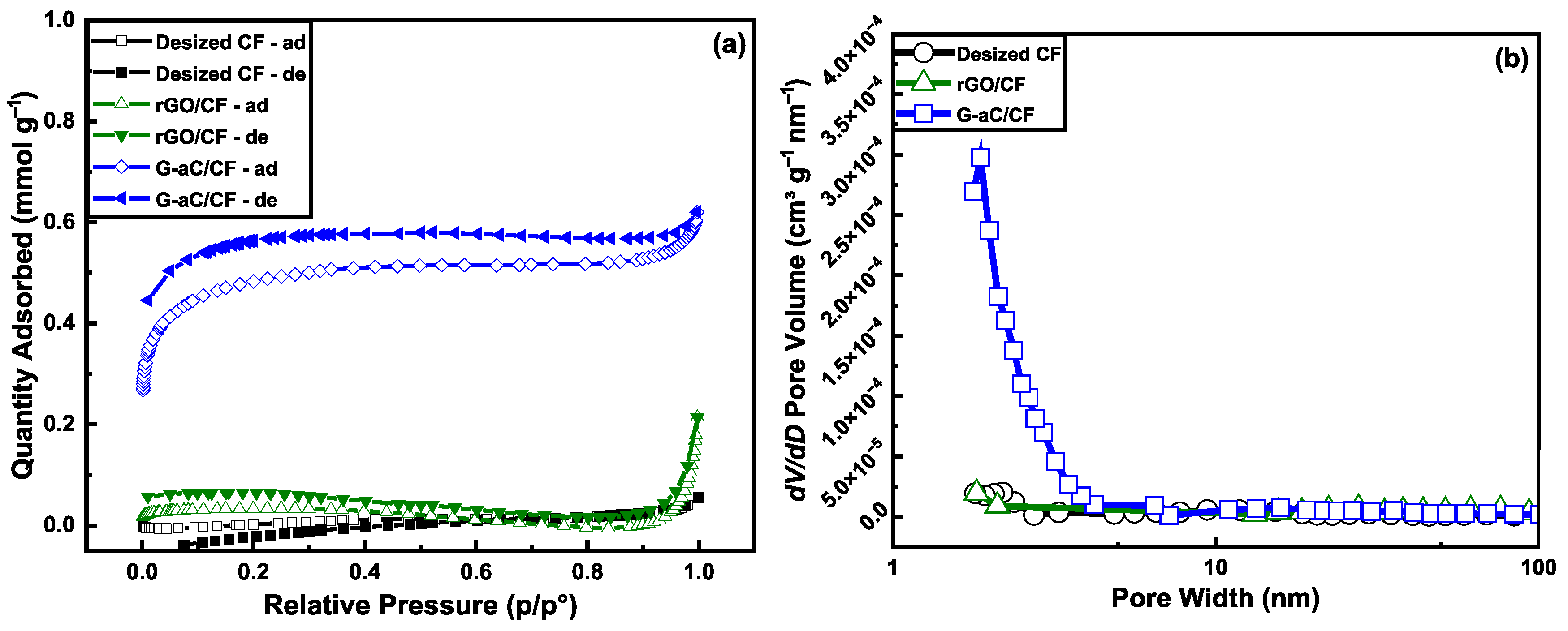





| Sample Material | Loading of Active Material on CF (wt.%) | BET Surface Area (m2 g−1) | Pore Volume (cm3 g−1) | Pore Width (nm) | Specific Capacitance (F g−1) |
|---|---|---|---|---|---|
| Desized—CF | - | 0.16 | 0.0015 | 8.5 | 9.38 |
| rGO/CF | 3.2 | 2.36 | 0.0020 | 2.46 | 22.54 |
| G-aC/CF2 | 13.8 | 34.21 | 0.0190 | 2.25 | 172 |
| G-aC powder | - | 64.27 | 0.0650 | 4.15 ± 0.25 | - |
| Electrode | Material Loading | Device Type | Electrolyte | Capacitance (C) | Energy Density (E) | Power Density (P) | References |
|---|---|---|---|---|---|---|---|
| G-aC/CF | 13.8 wt.% | Three-electrode cell | 6 M KCl | 172 F g−1 | 44.36 Wh kg−1 | 150 W kg−1 | Current work |
| SSC | PEGDGE (83 wt.%), EMIMTFSI (10 wt.%), TETA (7 wt.%), 2 g of LiFSI | 0.227 F g−1 | 0.9897 Wh kg−1 | 3.5 W kg−1 | |||
| CF Fabric—CAG | 22 wt.% | Three-electrode cell | 3 M KCl | 14.3 ± 0.2 F g−1 | - | - | [24] |
| 15.9 wt.% | Three-electrode cell | 3 M KCl | 8.7 ± 0.3 F g−1 | - | - | ||
| 9.5 wt.% | SSC | EMITFSI | 0.885 F g−1 | 1230 µWh kg−1 | 420 µW kg−1 | ||
| SSC | 82.6 wt.% PEGDGE,10 wt.% EMITFSI, 7.4 wt.% TETA | 0.602 F g−1 | 840 µWh kg−1 | 32.8 µW kg−1 | |||
| GNP-WCF | - | SSC | 33.75 wt.% of LY, 33.75 wt.% of a PEGDGE, 30 wt.% of EMITSFI + TiO2, DDS hardener | 0.0096 F g−1 | 0.00286 Wh kg−1 | 1.139 W kg−1 | [25] |
| CF-CA/GNP | 5 wt.% | SSC | DGEBA (43.9 wt.%), 1 M lithium perchlorate in propylene carbonate (50 wt.%), triethylene tetramine (6.1 wt.%) | 0.4 F g−1 | 0.8 Wh kg−1 | 108 W kg−1 | [27] |
| CF fabric - GA | 28 wt.% | Three-electrode cell | 6 M KCl | 92.7 F g−1 | 1 Wh kg−1 | 677 W kg−1 | [28] |
| SSC | PEDGE (75 wt.%), TETA (11.4 wt.%) and EMITFSI (13.6 wt.%) | 0.0056 F g−1 | - | 0.0225 W kg−1 | |||
| AC/WCF | 15 wt.% | SSC | DGEBA:AG80 (7:3), EMIM-TFSI, D400 polyether amine | 1.56 F g−1 | 1.53 Wh kg−1 | 42.66 W kg−1 | [21] |
| CAG-Spread tow CF | 20–30 wt.% | Symmetrical SC | EMI-TFSI | 4.5 F g−1 | 2.6 Wh kg−1 | 440 W kg−1 | [14] |
| 1:1 ACC-CSS | - | SSC | PVA-KOH gel electrolyte | 0.0088 F g−1 | 0.0099 Wh kg−1 | 0.4455 W kg−1 | [3] |
| Three-electrode cell | 1 M KOH | 0.467 F g−1 | - | - | |||
| MnOOH-NWs@WCF | - | SSC | EMIMTFSI (69 wt.%), LiTFSI (30 wt.%), PC (1 wt.%), Epoxy resins (E-51 and PEGDGE), D-230 | 0.0771 F cm2 | 0.0812 Wh kg−1 | 0.04737 W kg−1 | [70] |
| PANI-CF | 0.05 mg cm−2 | SSC | DGEBA (43%), LiClO4 (50%), PC, TETA (7%) | 0.02005 F cm2 | 0.0446 Wh kg−1 | 1.59 W kg−1 | [32] |
| PANI-ACF | 0.05 mg cm−2 | SSC | 0.02224 F cm2 | 0.0494 Wh kg−1 | 21.63 W kg−1 | ||
| CF-Urea-GNF | - | SSC | PEGDGE/EMIMTFSI | 0.048 F g−1 | 7 × 10−5 Wh kg−1 | 0.788 W kg−1 | [68] |
| CAG-CF | 18.4 wt.% | SSC | BADGE, IPDA, EMIM-TFSI | 2.4 F g−1 | 2.15 Wh kg−1 | 98.8 W kg−1 | [72] |
| Symmetrical SC | EMIM-TFSI | 2.17 F g−1 | 1.55 Wh kg−1 | 61.7 W kg−1 | |||
| CNT-CF | 17 wt.% | SSC | PEGDGE, EMIMTFSI, TETA | 0.173 F g−1 | 0.055 Wh kg−1 | 17 W kg−1 | [23] |
| PEGDGE, EMIMTFSI + additional 6 wt.% IL, TETA | 0.967 F g−1 | 0.3 Wh kg−1 | 84.5 W kg−1 | ||||
| Symmetrical SC | EMIMTFSI | 13.26 F g−1 | 3.8 Wh kg−1 | 57,789 W kg−1 | |||
| CAG-CF | - | SSC | DGEBA (47.2 wt.%), IPDA 11.8 wt.%, EMIM-TFSI (41 wt.%) | 0.212 F g−1 | 0.093 Wh kg−1 | 5.2 W kg−1 | [69] |
| Symmetrical SC | EMIMTFSI | 1.731 F g−1 | 1.405 Wh kg−1 | 301 W kg−1 | |||
| CF-GNP | - | SSC | PEGDGE (82.6 wt.%), TETA (7.4 wt.%) and EMIBF4 (10 wt.%) | 0.623 F g−1 | 0.0169 Wh kg−1 | 5.2 W kg−1 | [63] |
| 1.44 F g−1 | - | - |
| Sample | Element | Atomic Concentrations (at%) |
|---|---|---|
| Desized CF | C 1s | 71.5 |
| O 1s | 27.12 | |
| N 1s | 1.73 | |
| rGO/CF | C 1s | 88.83 |
| O 1s | 8.23 | |
| N 1s | 2.88 | |
| G-aC/CF | C 1s | 85.77 |
| O 1s | 14.09 | |
| N 1s | 0.14 |
| Samples | Surface Functional Group | Binding Energy | Atomic Concentrations |
|---|---|---|---|
| Desized CF | C-C | 284.85 | 69.28 |
| C-O | 286.26 | 16.3 | |
| C=O | 287.54 | 6.62 | |
| O-C=O | 288.91 | 7.8 | |
| rGO/CF | C-C | 284.77 | 40.41 |
| C-O | 286.67 | 29.82 | |
| C=O | 287.72 | 16.22 | |
| O-C=O | 289.81 | 6.41 | |
| π-π* | 291.89 | 7.41 | |
| G-aC/CF | C-C | 284.77 | 34.99 |
| C-O | 286.08 | 7.61 | |
| C=O | 288.08 | 4.97 | |
| O-C=O | 289.28 | 5.07 | |
| π-π* | 291.4 | 1.01 | |
| K2P 3/2 | 292.83 | 29.11 | |
| K2P 1/2 | 295.59 | 17.24 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gangipamula, V.; Subhani, K.; Mahon, P.J.; Salim, N. Nanomaterial Functionalized Carbon Fiber-Reinforced Composites with Energy Storage Capabilities. Nanomaterials 2025, 15, 1325. https://doi.org/10.3390/nano15171325
Gangipamula V, Subhani K, Mahon PJ, Salim N. Nanomaterial Functionalized Carbon Fiber-Reinforced Composites with Energy Storage Capabilities. Nanomaterials. 2025; 15(17):1325. https://doi.org/10.3390/nano15171325
Chicago/Turabian StyleGangipamula, Venkatesh, Karamat Subhani, Peter J. Mahon, and Nisa Salim. 2025. "Nanomaterial Functionalized Carbon Fiber-Reinforced Composites with Energy Storage Capabilities" Nanomaterials 15, no. 17: 1325. https://doi.org/10.3390/nano15171325
APA StyleGangipamula, V., Subhani, K., Mahon, P. J., & Salim, N. (2025). Nanomaterial Functionalized Carbon Fiber-Reinforced Composites with Energy Storage Capabilities. Nanomaterials, 15(17), 1325. https://doi.org/10.3390/nano15171325









