Environmentally Friendly Improvement of Plasmonic Nanostructure Functionality towards Magnetic Resonance Applications
Abstract
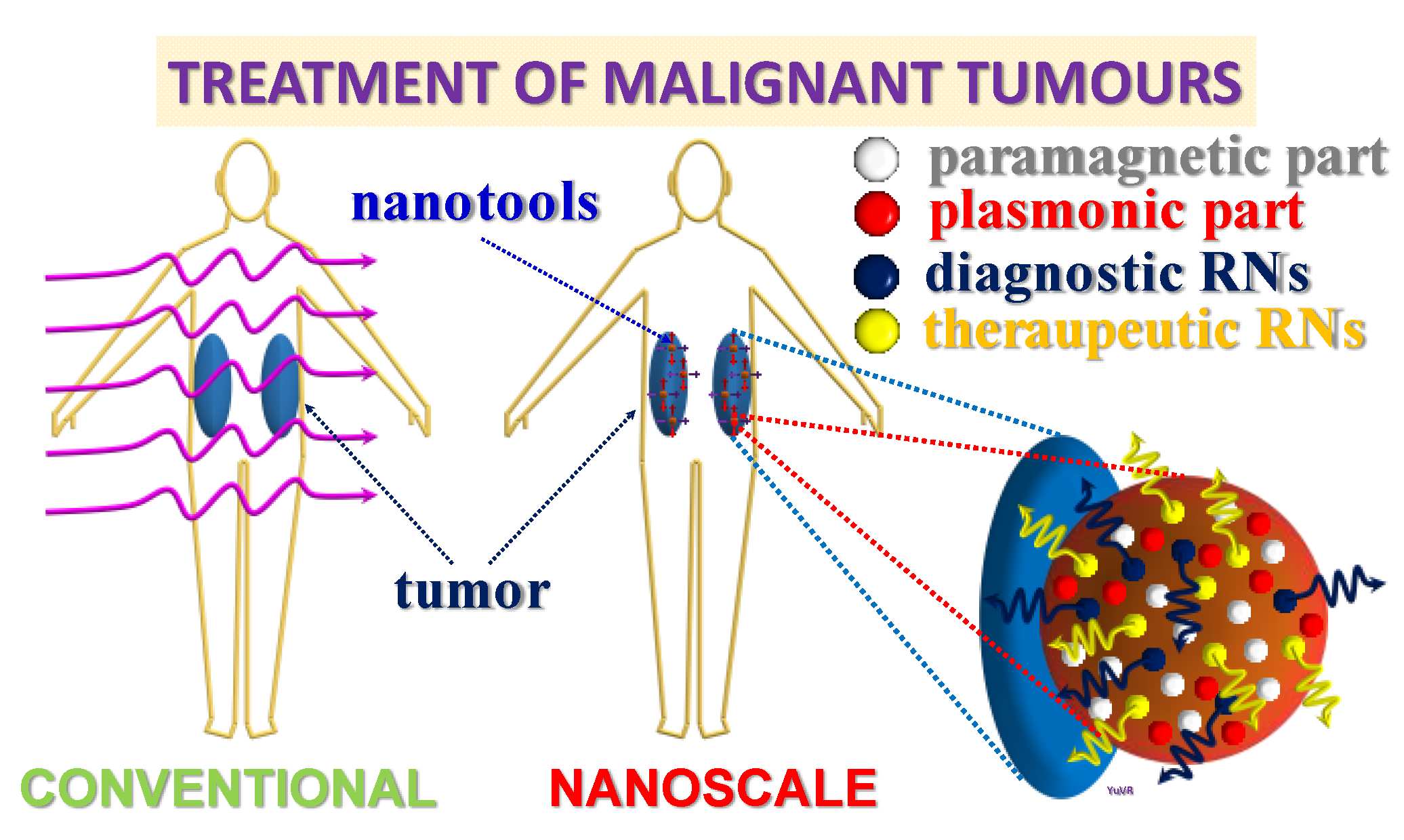
1. Introduction
2. Materials and Methods
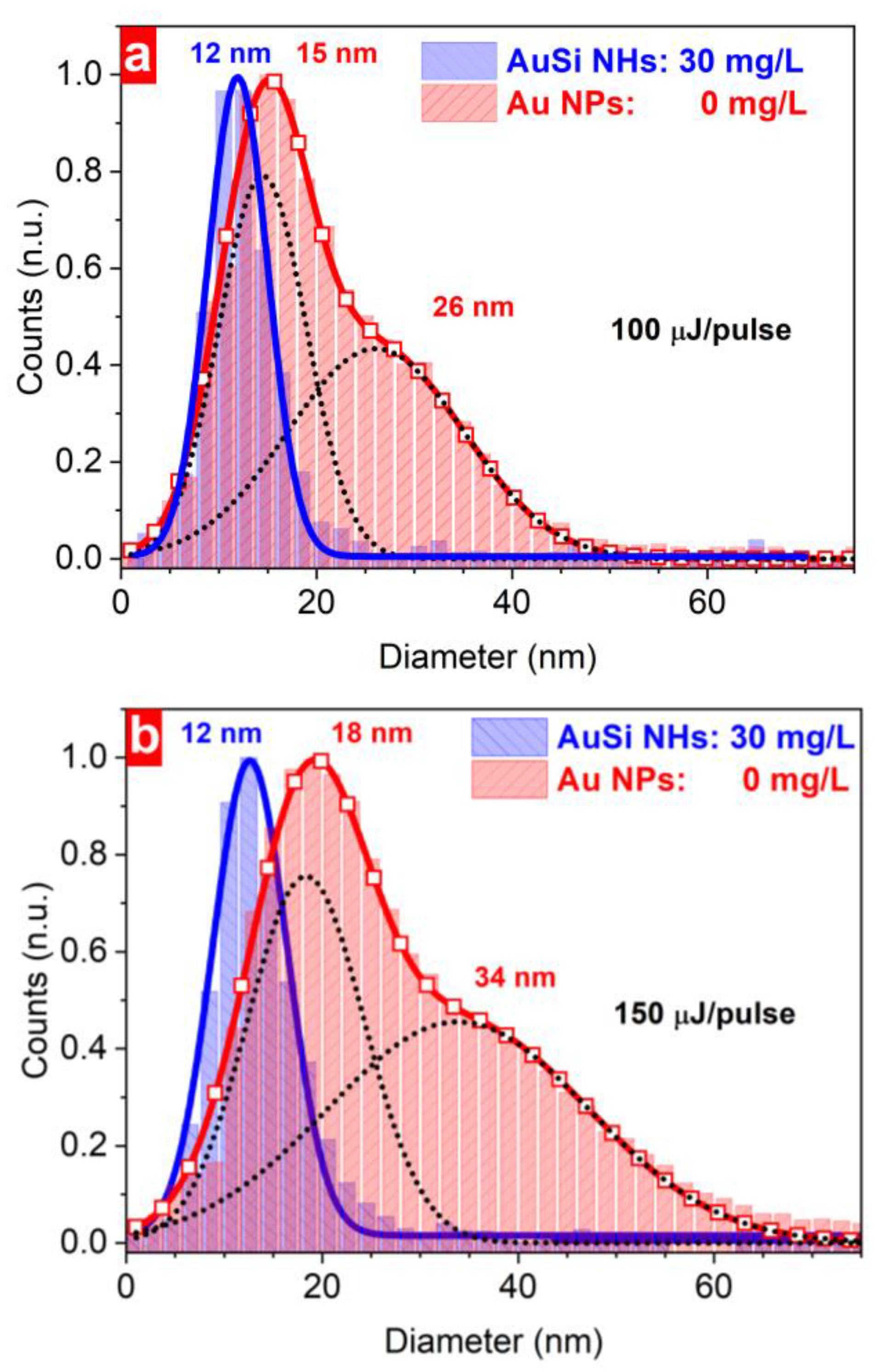
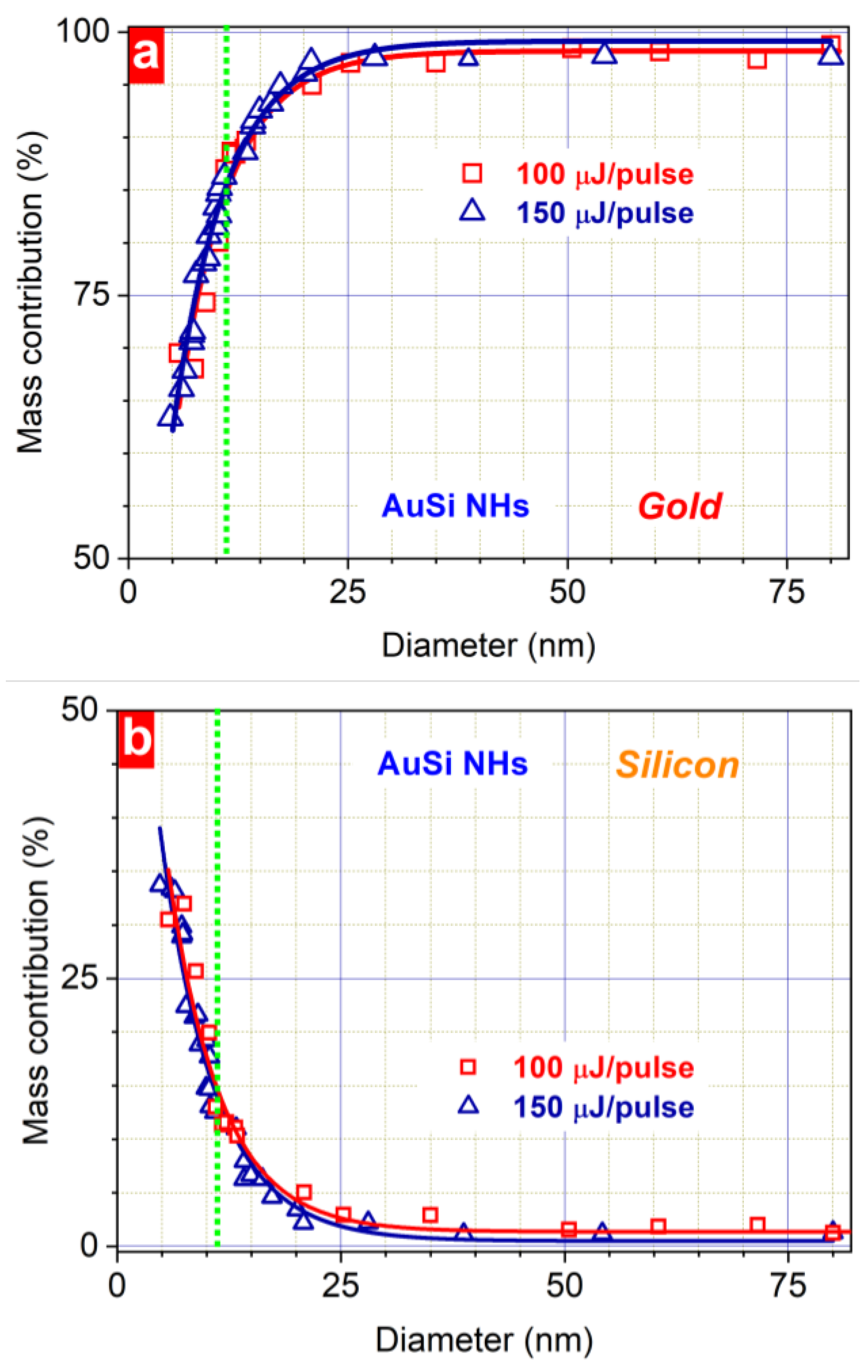
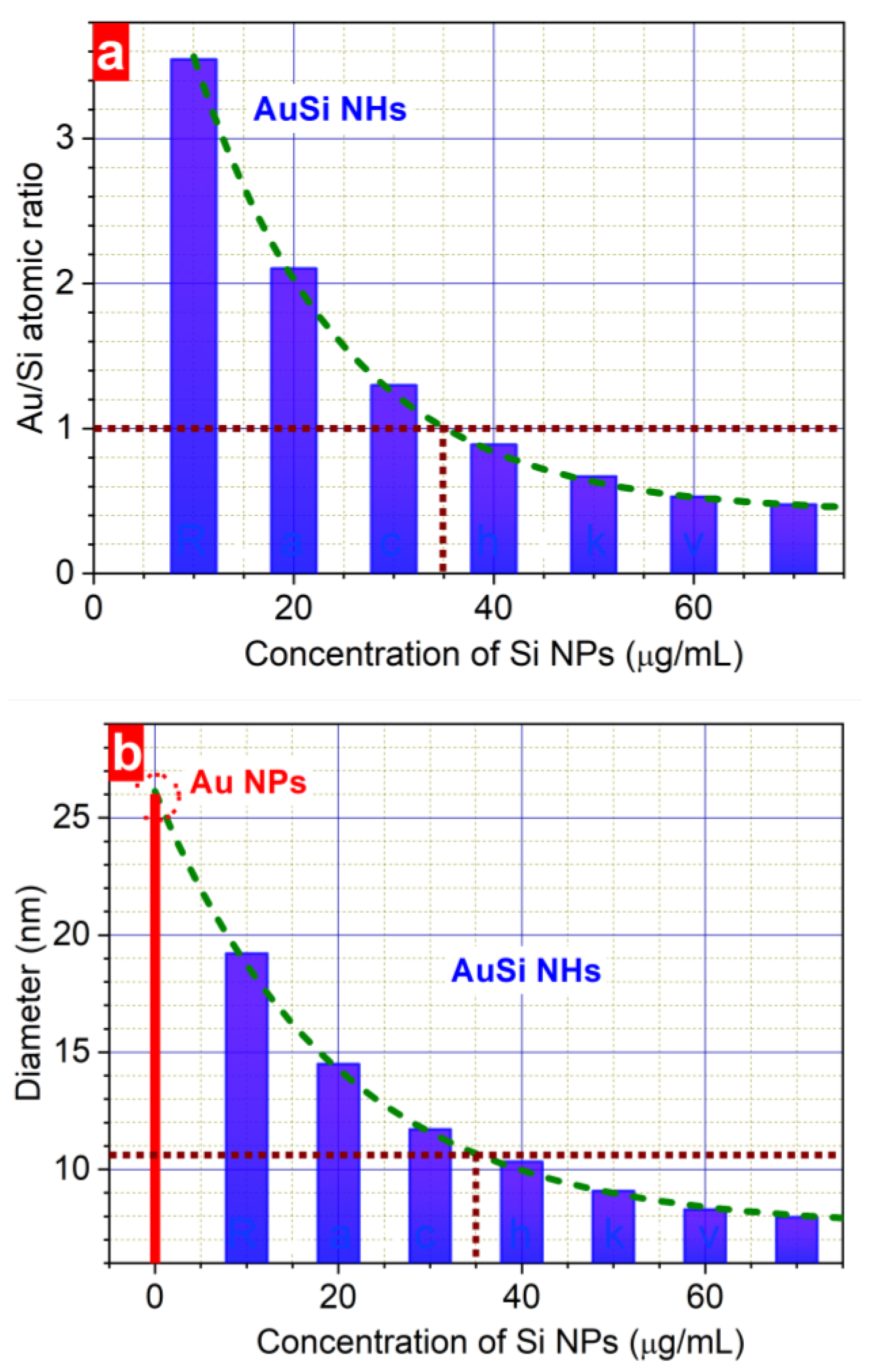
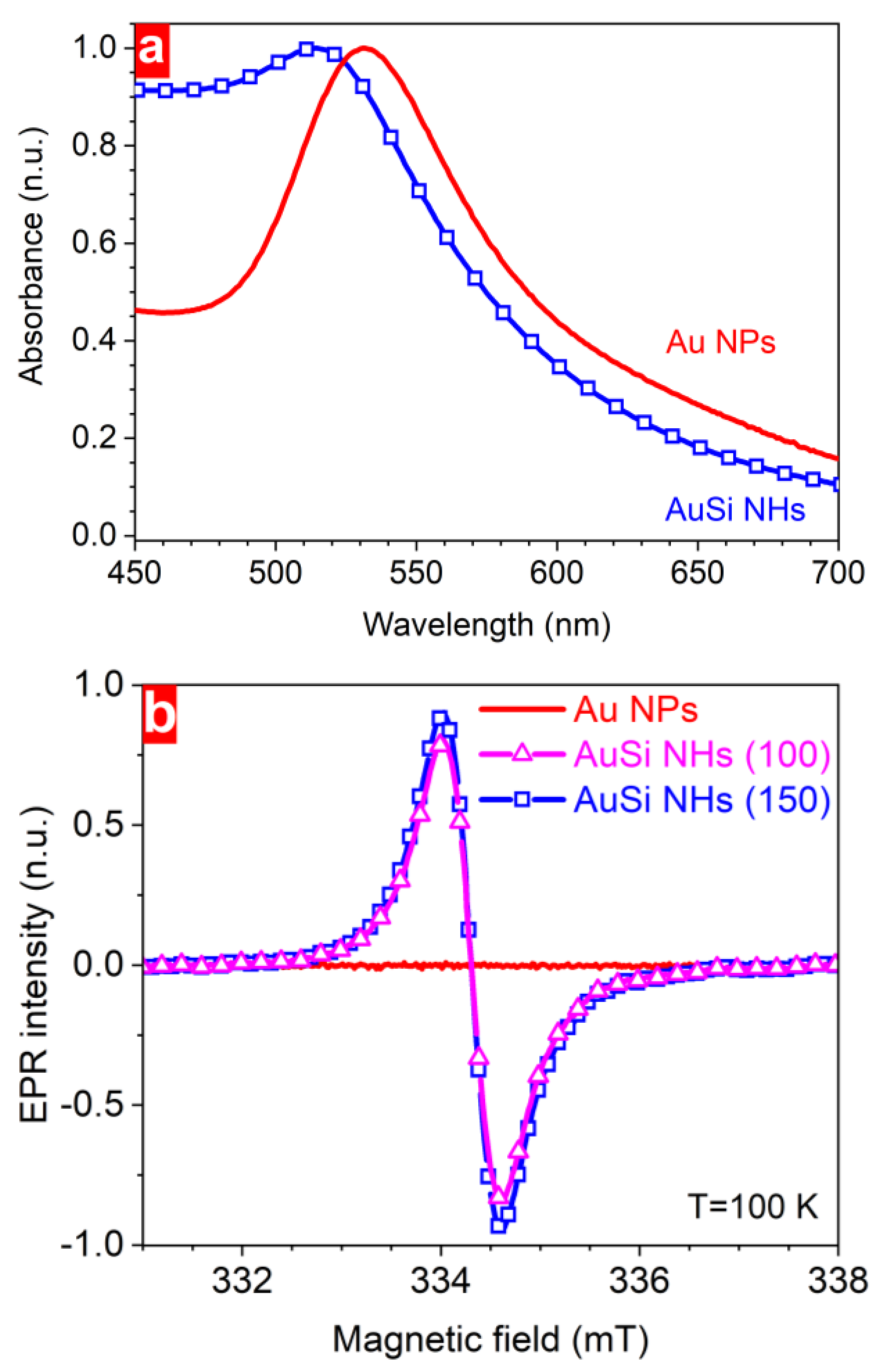
3. Results
4. Discussion
| Interplanar Distance (Å) | Planes | XRD Peaks (°) | |||
|---|---|---|---|---|---|
| Au NPs | AuSi NHs | Literature | Experiments | Literature | |
| 2.38 | 2.39 | 2.35 | (111) | 38.4 | 38.2 |
| 2.12 | 2.08 | 2.04 | (200) | 44.8 | 44.4 |
| 1.47 | 1.48 | 1.44 | (220) | 64.8 | 64.8 |
| 1.26 | 1.26 | 1.23 | (311) | 77.5 | 77.6 |
| 1.19 | 1.19 | 1.18 | (222) | 82.3 | 82.0 |
| 0.95 | - | 0.94 | (331) | - | - |
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Xie, F.; Pang, J.S.; Centeno, A.; Ryan, M.P.; Riley, D.J.; Alford, N.M. Nanoscale control of Ag nanostructures for plasmonic fluorescence enhancement of near-infrared dyes. Nano Res. 2013, 6, 496–510. [Google Scholar] [CrossRef]
- Uusitalo, S.; Popov, A.; Ryabchikov, Y.V.; Bibikova, O.; Alakomi, H.-L.; Juvonen, R.; Kontturi, V.; Siitonen, S.; Kabashin, A.; Meglinski, I.; et al. Surface enhanced Raman spectroscopy for identification and discrimination of beverage spoilage yeasts using patterned substrates and gold nanoparticles. J. Food Eng. 2017, 212, 47–54. [Google Scholar] [CrossRef]
- Yakimchuk, D.V.; Kaniukov, E.Y.; Lepeshov, S.; Bundyukova, V.D.; Demyanov, S.E.; Arzumanyanm, G.M.; Doroshkevich, N.V.; Mamatkulov, K.Z.; Bochmann, A.; Presselt, M.; et al. Self-organized spatially separated silver 3D dendrites as efficient plasmonic nanostructures for surface-enhanced Raman spectroscopy applications. J. Appl. Phys. 2019, 126, 233105. [Google Scholar] [CrossRef]
- Kornilova, A.V.; Gorbachevskii, M.V.; Kuralbayeva, G.A.; Jana, S.; Novikov, A.A.; Eliseev, A.A.; Vasiliev, A.N.; Timoshenko, V.Y. Plasmonic properties of halloysite nanotubes with immobilized silver nanoparticles for applications in surface-enhanced Raman scattering. Phys. Status Solidi A 2019, 216, 1800886. [Google Scholar] [CrossRef]
- Ryabchikov, Y.V.; Lysenko, V.; Nychyporuk, T. Enhanced Thermal sensitivity of silicon nanoparticles embedded in (nano–Ag)/SiNx for luminescent thermometry. J. Phys. Chem. C 2014, 118, 12515–12519. [Google Scholar] [CrossRef]
- Mantri, Y.; Jokerst, J.V. Engineering plasmonic nanoparticles for enhanced photoacoustic imaging. ACS Nano 2020, 14, 9408–9422. [Google Scholar] [CrossRef]
- Gerosa, C.; Crisponi, G.; Nurchi, V.M.; Saba, L.; Cappai, R.; Cau, F.; Faa, G.; Eyken, P.V.; Scartozzi, M.; Floris, G.; et al. Gold nanoparticles: A new golden era in oncology. Pharmaceuticals 2020, 13, 192. [Google Scholar] [CrossRef]
- Shakeri-Zadedh, A.; Zareyi, H.; Sheervalilou, R.; Laurent, S.; Ghaznavi, H.; Samadin, H. Gold nanoparticles-mediated bubbles in cancer nanotechnology. J. Control Release 2021, 330, 49–60. [Google Scholar] [CrossRef]
- Konstantinova, E.A.; Demin, V.A.; Vorontzov, A.S.; Ryabchikov, Y.V.; Belogorokhov, I.A.; Osminkina, L.A.; Forsh, P.A.; Kashkarov, P.K.; Timoshenko, V.Y. Electron paramagnetic resonance and photoluminescence study of Si nanocrystals—Photosensitizers of singlet oxygen molecules. J. Non-Cryst. Solids 2006, 352, 1156–1159. [Google Scholar] [CrossRef]
- Ryabchikov, Y.V.; Alekseev, S.A.; Lysenko, V.; Bremond, G.; Bluet, J.-M. Photoluminescence thermometry with alkyl–terminated silicon nanoparticles dispersed in low-polar liquids. Phys. Status Solidi RRL 2013, 7, 414–417. [Google Scholar] [CrossRef]
- Ryabchikov, Y.V.; Alekseev, S.A.; Lysenko, V.V.; Bremond, G.; Bluet, J.-M. Luminescence behavior of silicon and carbon nanoparticles dispersed in low-polar liquids. Nanoscale Res. Lett. 2012, 7, 365. [Google Scholar] [CrossRef] [PubMed]
- Ryabchikov, Y.V.; Belogorokhov, I.A.; Vorontsov, A.S.; Osminkina, L.A.; Timoshenko, V.Y.; Kashkarov, P.K. Dependence of the singlet oxygen photosensitization efficiency on morphology of porous silicon. Phys. Status Solidi A 2007, 204, 1271–1275. [Google Scholar] [CrossRef]
- Ryabchikov, Y.V.; Belogorokhov, I.A.; Gongalskiy, M.B.; Osminkina, L.A.; Timoshenko, V.Y. Photosensitized generation of singlet oxygen in powders and aqueous suspensions of silicon nanocrystals. Semiconductors 2011, 45, 1059–1063. [Google Scholar] [CrossRef]
- Tieu, T.; Alba, M.; Elnathan, R.; Cifuentes-Rius, A.; Voelcekr, N.H. Advances in porous silicon–based nanomaterials for diagnostic and therapeutic applications. Adv. Ther. 2019, 2, 1800095. [Google Scholar] [CrossRef]
- Xu, Z.; Ma, X.; Gao, Y.-E.; Hou, M.; Xue, P.; Li, C.M.; Kang, Y. Multifunctional silica nanoparticles as a promising theranostic platform for biomedical applications. Mater. Chem. Front. 2017, 1, 1257–1272. [Google Scholar] [CrossRef]
- Peng, F.; Su, Y.; Zhong, Y.; Fan, C.; Lee, F.-S.; He, Y. Silicon nanomaterials platform for bioimaging, biosensing, and cancer therapy. Acc. Chem. Res. 2014, 47, 612–623. [Google Scholar] [CrossRef] [PubMed]
- Zabotnov, S.V.; Kashaev, F.V.; Shuleiko, D.V.; Gongalesky, M.B.; Golovan, L.A.; Kashkarov, P.K.; Kurakina, D.A.; Agrba, P.D.; Sergeeva, E.A.; Kirilin, M.Y. Silicon nanoparticles as contrast agents in the methods of optical biomedical diagnostics. Quantum Electron. 2017, 47, 638–646. [Google Scholar] [CrossRef]
- Kirillin, M.Y.; Sergeeva, E.A.; Agrba, P.D.; Krainov, A.D.; Ezhov, A.A.; Shuleiko, D.V.; Kashkarov, P.K.; Zabotnov, S.V. Laser-ablated silicon nanoparticles: Optical properties and perspectives in optical coherence tomography. Laser Phys. 2015, 25, 075604–075610. [Google Scholar] [CrossRef]
- Joo, J.; Liu, X.; Kotamraju, R.V.; Ruoslahti, E.; Nam, Y.; Sailor, M.J. Gated luminescence imaging of silicon nanoparticles. ACS Nano 2015, 9, 6233–6241. [Google Scholar] [CrossRef] [PubMed]
- Nekovic, E.; Storey, C.J.; Kaplan, A.; Theis, W.; Canham, L.T. A gentle sedimentation process for size-selecting porous silicon microparticles to be used for drug delivery via fine gauge needle administration. Silicon 2022, 14, 589–596. [Google Scholar] [CrossRef]
- Schevchenko, S.N.; Burhardt, M.; Sheval, E.V.; Natashina, U.A.; Grosse, C.; Nikolaev, A.L.; Gopin, A.V.; Neugebauer, U.; Kudryavtsev, A.A.; Sivakov, V.; et al. Antimicrobial effect of biocompatible silicon nanoparticles activated using therapeutic ultrasound. Langmuir 2017, 33, 2603–2609. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Su, L.; Ge, X.; Zhang, W.; Li, Q.; Zhang, X.; Ye, J.; Lin, L.; Song, J.; Yang, H. Dual activated NIR-II fluorescence and photoacoustic imaging-guided cancer chemo-radiotherapy using hybrid plasmonic-fluorescent assemblies. Nano Res. 2020, 13, 3268–3277. [Google Scholar] [CrossRef]
- Yang, Z.; Ding, X.; Jiang, J. Facile synthesis of magnetic–plasmonic nanocomposites as T1 MRI contrast enhancing and photothermal therapeutic agents. Nano Res. 2016, 9, 787–799. [Google Scholar] [CrossRef]
- Chatterjee, K.; Sarkar, S.; Rao, K.J. Core/shell nanoparticles in biomedical applications. Adv. Colloid Interface Sci. 2014, 209, 8–39. [Google Scholar] [CrossRef] [PubMed]
- Moreira, A.F.; Rodrigues, C.F.; Reis, C.A.; Costa, E.C.; Correia, I.J. Gold-core silica shell nanoparticles application in imaging and therapy: A review. Microporous Mesoporous Mater. 2018, 270, 168–179. [Google Scholar] [CrossRef]
- Li, J.; Zhang, W.; Gao, Y.; Tong, H.; Chen, Z.; Shi, J.; Santos, H.E.; Xia, B. Near-infrared light and magnetic field dual-responsive porous silicon-based nanocarriers to overcome multidrug resistance in breast cancer cells with enhanced efficiency. J. Mater. Chem. B 2020, 8, 546–557. [Google Scholar] [CrossRef]
- Jin, R.; Liu, Z.; Bai, Y.; Zhou, Y.; Gooding, Y.J.; Chen, X. Core–Satellite Mesoporous Silica–Gold Nanotheranostics for Biological Stimuli Triggered Multimodal Cancer Therapy. Adv. Funct. Mater. 2018, 28, 1801961. [Google Scholar] [CrossRef]
- Zhang, J.; Claverie, J.; Chaker, M.; Ma, D. Colloidal metal nanoparticles prepared by laser ablation and their applications. ChemPhysChem 2017, 18, 986–1006. [Google Scholar] [CrossRef]
- Zhang, D.; Gökce, B.; Barcikowski, S. Laser synthesis and processing of colloids: Fundamentals and applications. Chem. Rev. 2017, 117, 3990–4103. [Google Scholar] [CrossRef]
- Scaramuzza, S.; Zerbetto, M.; Amendola, V. Synthesis of gold nanoparticles in liquid environment by laser ablation with geometrically confined configurations: Insights to improve size control and productivity. J. Phys. Chem. C 2016, 17, 9453–9463. [Google Scholar] [CrossRef]
- Taheri, M.; Mansour, N. Silicon nanoparticles produced by two-step nanosecond pulsed laser ablation in ethanol for enhanced blue emission properties. Silicon 2020, 12, 789–797. [Google Scholar] [CrossRef]
- Shuaib, E.P.; Yogesh, G.K.; Sastikumar, D. Amorphous and photoluminescent crystalline silicon carbide nanoparticles synthesized by laser ablation in liquids. Mater. Today Proc. 2022, 50, 2745–2750. [Google Scholar] [CrossRef]
- Ryabchikov, Y.V. Size modification of optically active contamination-free silicon nanoparticles with paramagnetic defects by their fast synthesis and dissolution. Phys. Status Solidi A 2019, 216, 1800685. [Google Scholar] [CrossRef]
- Patrone, L.; Nelson, D.; Safarov, V.I.; Sentis, M.; Marine, W. Photoluminescence of silicon nanoclusters with reduced size dispersion produced by laser ablation. J. Appl. Phys. 2000, 87, 3829. [Google Scholar] [CrossRef]
- Amoruso, S.; Bruzzese, R.; Spinello, N.; Velotta, R.; Vitiello, M.; Wang, X. Generation of silicon nanoparticles via femtosecond laser ablation in vacuum. Appl. Phys. Lett. 2004, 84, 4502. [Google Scholar] [CrossRef]
- Guadagnini, A.; Agnoli, S.; Badocco, D.; Pastore, P.; Coral, D.; Fernandéz van Raap, M.B.; Forrer, D.; Amendola, V. Facile synthesis by laser ablation in liquid of nonequilibrium cobalt-silver nanoparticles with magnetic and plasmonic properties. J. Colloid Interface Sci. 2021, 585, 267–275. [Google Scholar] [CrossRef]
- Amendola, V.; Scaramuzza, S.; Agnoli, S.; Granozzi, G.; Meneghetti, M.; Campo, G.; Bonanni, V.; Pineider, F.; Sangregorio, C.; Ghigna, P.; et al. Laser generation of iron-doped silver nanotruffles with magnetic and plasmonic properties. Nano Res. 2015, 8, 4007–4023. [Google Scholar] [CrossRef]
- Torresan, V.; Forrer, D.; Guadagnini, A.; Badocco, D.; Pastore, P.; Cesarin, M.; Selloni, A.; Coral, D.; Ceolin, M.; Fernandéz van Raap, M.B.; et al. 4D Multimodal nanomedicines made of nonequilibrium Au–Fe alloy nanoparticles. ACS Nano 2020, 14, 12840–12853. [Google Scholar] [CrossRef]
- Tymoczko, A.; Kamp, M.; Rehbock, C.; Kienle, L.; Cattaruzza, E.; Barcikowski, S.; Amendola, V. One-step synthesis of Fe–Au core–shell magnetic-plasmonic nanoparticles driven by interface energy minimization. Nanoscale Horiz. 2019, 4, 1326–1332. [Google Scholar] [CrossRef]
- Amendola, V.; Scaramuzza, S.; Carraro, F.; Cattaruzza, E. Formation of alloy nanoparticles by laser ablation of Au/Fe multilayer films in liquid environment. J. Colloid Interface Sci. 2017, 489, 18–27. [Google Scholar] [CrossRef]
- Nasiri, P.; Doranian, D.; Sari, A.H. Synthesis of Au/Si nanocomposite using laser ablation method. Opt. Laser Technol. 2019, 113, 217–224. [Google Scholar] [CrossRef]
- Saraeva, I.N.; Luong, N.V.; Kudryashov, S.I.; Rudenko, A.A.; Khmelnitskiy, R.A.; Shakhmin, A.L.; Kharin, A.Y.; Ionin, A.A.; Zayarny, D.A.; Tung, D.H.; et al. Laser synthesis of colloidal Si@Au and Si@Ag nanoparticles in water via plasma-assisted reduction. J. Photochem. Photobiol. 2018, 360, 125–131. [Google Scholar] [CrossRef]
- Ryabchikov, Y.V. Facile laser synthesis of multimodal composite silicon/gold nanoparticles with variable chemical composition. J. Nanoparticle Res. 2019, 21, 85. [Google Scholar] [CrossRef]
- Kögler, M.; Ryabchikov, Y.V.; Uusitalo, S.; Popov, A.; Tselikov, G.; Välimaa, A.-L.; Al-Kattan, A.; Hiltunen, J.; Laitinen, R.; Neubauer, P.; et al. Bare laser–synthesized Au–based nanoparticles as non–disturbing SERS probes for bacteria identification. J. Biophotonics 2018, 11, e201700225. [Google Scholar] [CrossRef] [PubMed]
- Kutrovskaya, S.; Arekelian, S.; Kucherik, A.; Osipov, A.; Evlyukhin, A.; Kavokin, A.V. The synthesis of hybrid gold-silicon nano particles in a liquid. Sci. Rep. 2017, 7, 10284. [Google Scholar] [CrossRef]
- Taheri, M.; Hajiesmaeilbaigi, F.; Motamedi, A.; Golian, Y. Nonlinear optical response of gold/silicon nanocomposite prepared by consecutive laser ablation. Laser Phys. 2015, 25, 065901. [Google Scholar] [CrossRef]
- Saraeva, I.N.; Kudryashov, S.I.; Lednev, V.N.; Makarov, S.V.; Pershin, S.M.; Rudenko, A.A.; Zayarny, D.A.; Ionin, A.A. Single- and multishot femtosecond laser ablation of silicon and silver in air and liquid environments: Plume dynamics and surface modification. Appl. Surf. Sci. 2019, 476, 576–586. [Google Scholar] [CrossRef]
- Kohsakowski, S.; Seiser, F.; Wiederrecht, J.-P.; Reichenberger, S.; Vinnay, T.; Barcikowski, S.; Marzun, G. Effective size separation of laser-generated, surfactant-free nanoparticles by continuous centrifugation. Nanotechnology 2020, 31, 095603. [Google Scholar] [CrossRef]
- Kulikov, I.; Panjwani, N.A.; Vereshchagin, A.A.; Spallek, D.; Lukianov, D.A.; Alekseeva, E.V.; Levin, O.V.; Behrends, J. Spins at work: Probing charging and discharging of organic radical batteries by electron paramagnetic resonance spectroscopy. Energy Environ. Sci. 2022, 15, 3275–3290. [Google Scholar] [CrossRef]
- Kabashin, A.V.; Meunier, M. Synthesis of colloidal nanoparticles during femtosecond laser ablation of gold in water. J. Appl. Phys. 2003, 94, 7941. [Google Scholar] [CrossRef]
- Shih, C.-Y.; Shugaev, M.V.; Wu, C.; Zhigilei, L.V. The effect of pulse duration on nanoparticle generation in pulsed laser ablation in liquids: Insights from large-scale atomistic simulations. Phys. Chem. Chem. Phys. 2020, 22, 7077–7099. [Google Scholar] [CrossRef] [PubMed]
- Starinskiy, S.V.; Shukhov, Y.G.; Bulgakov, A.V. Effect of nanoparticle sizes on the extinction spectrum of colloidal solutions produced by laser ablation of gold in water. Quantum Electron. 2017, 47, 343. [Google Scholar] [CrossRef]
- Kabashin, A.V.; Meunier, M.; Kingston, C.; Luong, J.H.T. Fabrication and characterization of gold nanoparticles by femtosecond laser ablation in an aqueous solution of cyclodextrins. J. Phys. Chem. B 2003, 107, 4527–4531. [Google Scholar] [CrossRef]
- Spellauge, M.; Doñate-Buendía, C.; Barcikowski, S.; Gökce, B. Comparison of ultrashort pulse ablation of gold in air and water by time-resolved experiments. Light Sci. Appl. 2022, 11, 68. [Google Scholar] [CrossRef]
- Taccogna, F. Nucleation and growth of nanoparticles in a plasma by laser ablation in liquid. J. Plasma Phys. 2015, 81, 495810509. [Google Scholar] [CrossRef]
- Maximova, K.; Aristov, A.; Sentis, M.; Kabashin, A.V. Size-controllable synthesis of bare gold nanoparticles by femtosecond laser fragmentation in water. Nanotechnology 2015, 26, 065601. [Google Scholar] [CrossRef]
- Huang, H.; Zhigilei, L.V. Computational study of laser fragmentation in liquid: Phase explosion, inverse Leidenfrost effect at the nanoscale, and evaporation in a nanobubble. Sci. China Phys. Mech. Astron. 2022, 65, 274206. [Google Scholar] [CrossRef]
- Hwang, S.-W.; Tao, H.; Kim, D.-A.; Cheng, H.; Song, J.-K.; Rill, E.; Brenckle, M.A.; Panilaitis, B.; Won, S.M.; Kim, Y.-S.; et al. A physically transient form of silicon electronics. Science 2012, 337, 1640–1644. [Google Scholar] [CrossRef]
- Sylvestre, J.-P.; Poulin, S.; Kabashin, A.V.; Sacher, E.; Meunier, M.; Luong, J.H.T. Surface chemistry of gold nanoparticles produced by laser ablation in aqueous media. J. Phys. Chem. B 2004, 108, 16864–16869. [Google Scholar] [CrossRef]
- Ohshima, H.; Healy, T.W.; White, L.R. Accurate analytic expressions for the surface charge density/surface potential relationship and double-layer potential distribution for a spherical colloidal particle. J. Colloid Interface Sci. 1982, 90, 17–26. [Google Scholar] [CrossRef]
- Heuer-Jungemann, A.; Feliu, N.; Bakaimi, I.; Hamaly, M.; Alkilany, A.; Chakraborty, I.; Masood, A.; Casula, M.F.; Kostopoulou, A.; Oh, E.; et al. The role of ligands in the chemical synthesis and applications of inorganic nanoparticles. Chem. Rev. 2019, 119, 4819–4880. [Google Scholar] [CrossRef] [PubMed]
- Bulgakov, A.V.; Bulgakova, N.M. Recent advances in nanoparticle generation in liquids by lasers: Revealing formation mechanisms and tailoring properties. Sci. China Phys. Mech. 2022, 65, 274207. [Google Scholar] [CrossRef]
- Sivakumar, M.; Venkatakrishnan, K.; Tan, B. Characterization of MHz pulse repetition rate femtosecond laser-irradiated gold-coated silicon surfaces. Nanoscale Res. Lett. 2011, 6, 78. [Google Scholar] [CrossRef] [PubMed]
- Chromik, R.; Zavalij, L.; Johnson, M.D.; Cotts, E.J. Calorimetric investigation of the formation of metastable silicides in Au/a-Si thin film multilayers. J. Appl. Phys. 2002, 91, 8992–8998. [Google Scholar] [CrossRef]
- Chang, J.F.; Young, T.F.; Yang, Y.L.; Ueng, H.Y.; Chang, T.C. Silicide formation of Au thin films on (100) Si during annealing. Mater. Chem. Phys. 2004, 83, 199–203. [Google Scholar] [CrossRef]
- Ji, H.Y.; Parida, B.; Park, S.; Kim, M.; Peck, J.H.; Kim, K. Eutectic temperature effect on Au thin film for the formation of Si nanostructures by hot wire chemical vapor deposition. Curr. Photovolt. Res. 2013, 1, 63–68. [Google Scholar]
- Yan, W.; Petkov, V.; Mahurin, S.M.; Overbury, S.H.; Dai, S. Powder XRD analysis and catalysis characterization of ultra-small gold nanoparticles deposited on titania-modified SBA-15. Catal. Commun. 2005, 6, 404–408. [Google Scholar] [CrossRef]
- Pashley, D.W.; Stowell, M.J. Electron microscopy and diffraction of twinned structures in evaporated films of gold. Philos. Mag. 1963, 8, 1605–1632. [Google Scholar] [CrossRef]
- Dahal, N.; Wright, J.T.; Willey, T.M.; Meulenberg, R.W.; Chikan, V. Preparation of iron and gold silicide nanodomains on silicon (111) by the reaction of gold, iron-gold core-shell, and alloy nanoparticles with triethylsilane. ACS Appl. Mater. Interfaces 2010, 2, 2238–2247. [Google Scholar] [CrossRef]
- Hasegawa, S.; Watanabe, S.; Inokuma, T.; Kurata, Y. Structure and grain boundary defects of recrystallized silicon films prepared from amorphus silicon deposited using disilane. J. Appl. Phys. 1995, 77, 1938. [Google Scholar] [CrossRef]
- Sarkar, D.K.; Bera, S.; Dhara, S.; Nair, K.G.M.; Narasimhan, S.V.; Chowdhury, S. XPS studies on silicide formation in ion beam irradiated Au/Si system. Appl. Surf. Sci. 1997, 120, 159–164. [Google Scholar] [CrossRef]
- Swain, B.P.; Swain, B.S. Analysis of chemical bonding and structural network of gold silicide in core–shell silicon nanowire. J. Mater. Eng. Perform. 2018, 27, 2687–2692. [Google Scholar] [CrossRef]
- Sarkar, D.K.; Bera, S.; Dhara, S.; Narasimhan, S.V.; Chowdhury, S.; Nair, K.G.M. GIXRD and XPS study of the ion beam mixed Au/Si(111) system. Solid State Commun. 1998, 105, 351–356. [Google Scholar] [CrossRef]
- Petriev, V.M.; Tischenko, V.K.; Mikhailovskaya, A.A.; Popov, A.A.; Tselikov, G.; Zelepukin, I.; Deyev, S.M.; Kaprin, A.D.; Ivanov, S.; Timoshenko, Y.V.; et al. Nuclear nanomedicine using Si nanoparticles as safe and effective carriers of 188Re radionuclide for cancer therapy. Sci. Rep. 2019, 9, 2017. [Google Scholar] [CrossRef] [PubMed]
- Skrabkova, H.S.; Bubenschikov, V.B.; Kodina, G.E.; Lunev, A.S.; Larenkov, A.A.; Epshtein, N.B.; Kabashin, A.V. 68Ga-adsorption on the Si-nanoparticles. IOP Conf. Ser. Mater. Sci. Eng. 2019, 487, 012026. [Google Scholar] [CrossRef]
- Bhaumik, J.; Mittal, A.K.; Banerjee, A.; Chisti, Y.; Banerjee, U.C. Applications of phototheranostic nanoagents in photodynamic therapy. Nano Res. 2015, 8, 1373–1394. [Google Scholar] [CrossRef]
- Pallares, R.M.; Abergel, R.J. Nanoparticles for targeted cancer radiotherapy. Nano Res. 2020, 13, 2887–2897. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Flimelová, M.; Ryabchikov, Y.V.; Behrends, J.; Bulgakova, N.M. Environmentally Friendly Improvement of Plasmonic Nanostructure Functionality towards Magnetic Resonance Applications. Nanomaterials 2023, 13, 764. https://doi.org/10.3390/nano13040764
Flimelová M, Ryabchikov YV, Behrends J, Bulgakova NM. Environmentally Friendly Improvement of Plasmonic Nanostructure Functionality towards Magnetic Resonance Applications. Nanomaterials. 2023; 13(4):764. https://doi.org/10.3390/nano13040764
Chicago/Turabian StyleFlimelová, Miroslava, Yury V. Ryabchikov, Jan Behrends, and Nadezhda M. Bulgakova. 2023. "Environmentally Friendly Improvement of Plasmonic Nanostructure Functionality towards Magnetic Resonance Applications" Nanomaterials 13, no. 4: 764. https://doi.org/10.3390/nano13040764
APA StyleFlimelová, M., Ryabchikov, Y. V., Behrends, J., & Bulgakova, N. M. (2023). Environmentally Friendly Improvement of Plasmonic Nanostructure Functionality towards Magnetic Resonance Applications. Nanomaterials, 13(4), 764. https://doi.org/10.3390/nano13040764






