Sustainable Biomass-Derived Carbon Electrodes for Potassium and Aluminum Batteries: Conceptualizing the Key Parameters for Improved Performance
Abstract
1. Introduction
2. Biomass-Based Anodes for Battery Application
3. Effect of Preparation Conditions on Biochar Physicochemical Properties
4. Potassium-Ion Battery (KIBs)
| Biomass Source | Synthesis Method zand Morphology | Specific Surface Area (SBET, m2 g−1) | Potential (V vs. K/K+) | Current Rate (mA g−1) | Initial Discharge Capacity (mA h g−1) | Capacity Retention (mA h g−1)/(cycles) | Ref. |
|---|---|---|---|---|---|---|---|
| Ganoderma lucidum spore | Anode prepared by one-step carbonization with no activation. Hollow-cage structure with no high porosity and a high degree of graphitization with a typical carbon amorphous structure. | 104.4 | 0.02–3.0 | 1000 | ~450 | ~125 (700) | [52] |
| Rice husk | Carbonization temperatures from 900 to 1500 °C. The highly amorphous microstructure is rich in mesopores. | 365 | 0.01–3.0 | 30 | ~250 | 104 (500) | [54] |
| Bamboo | Carbonization and KOH-treated activation. Medium porosity carbon anodes with crystalline structures with a high presence of defects and low graphitization degree. | 339 | 0.01–2.8 | 50 | ~450 | 204 (300) | [57] |
| Sugarcane bagasse | Biomass activated with NiCl2. Highly amorphous and microporous materials doped with nitrogen. | ~467 | 0.1–3.0 | 100 | 142 | 100.4 (400) | [58] |
| Chitin | NaOH/urea activated carbon anodes. Meso-macro porous chitin microspheres-like materials | 563 | 0.01–3.0 | 0.12 to 36 Coulombic | ~320 | 180 (4000) | [59] |
| Corn silk | Hydrothermal treatment and one-step carbonization. The carbon materials possess larger lattice spacing, amorphous structures, and a very low degree of graphitization with many defects. | - | 0.01–3.0 | 100 | ~886 | ~121 (2600) | [60] |
| Hemp core | One-step carbonization with fluorine doping process. Material wrapped by fluorine-containing nanotubes rich in defects with wide pore sizes and a low degree of graphitization. | 780 | 0.001–3.0 | 200 | ~822 | 369.6 (500) | [61] |
| Maple leaves | Carbonization and HNO3-treated activation. Low porosity carbon anodes containing O/N functional groups with higher graphitization degree. | 62.6 | 0.01–3.0 | 50 | ~934 | ~142 (1000) | [62] |
| Cyanobacteria powder | Two-step carbonization with NaCl and KCl activation. Nitrogen/oxygen co-doped hierarchically mesoporous carbon. Amorphous structures with a high degree of graphitization. | 473 | 0.01–3.0 | 50 | 912 | ~104 (1000) | [63] |
5. Aluminum Battery
| Source | Synthesis Method and Morphology | Specific Surface Area (SSA, m2 g−1) | Operating Potential (V) | Current Rate (mA g−1) | Initial Discharge Capacity (mA h g−1) | Capacity Retention (mA h g−1)/(Cycles) | Ref. |
|---|---|---|---|---|---|---|---|
| Sucrose | Sucrose was carbonized over customized silica template particles and washed with NaOH etching. The carbonization was carried out at 900 °C under H2/Ar atmosphere. | 1185 | 0.01–2.25 | 500 | 82 | 70 (1000) | [71] |
| Tar pitch | The reaction mixture of tar pitch and KOH (1:4 wt.%) was heated at 850 °C to produce activated carbon (AC). The porosity of AC is further modified by heat treatment at 750 °C under N2 flow. | 1980 | 0.01–2.25 | 500 | 51 | 95 (1000) | [71] |
| Commercial CMK-3 | - | 1000–2000 | 0.5–2.3 | 980 | ~27 | 33 (36,000) | [88] |
| Birch sawdust | Carbonized birch sawdust (400 °C) activated with 4 wt.% of NaOH under Ar flow at 600–850 °C. | 3300 | 0.0–2.5 | 283 | 82 | 69 (40) | [87] |
| Coconut shell | coconut shell chars were activated with 5 wt.% of KOH at 850 °C under an argon atmosphere. | 2686 | 0.01–2.2 | 1000 | 90 | 81 (1500) | [76] |
| Human Hair | Hair samples brunt at 300 °C were activated with 2 wt.% of NaOH at 750 °C in Ar flow. | - | 0.2–2.45 | 50 | 103 | 100 (50) | [91] |
6. Conclusions, Challenges and Future Works
- Difficulties in selecting proper and suitable biomass and carbon preparation methods to obtain desirable and tailored shapes/pores despite thermochemical routes employed in the conversion of biomass-precursor into carbon electrode materials.
- Issues on controlling and tailoring the pore geometry and size (pore structure), and interlayer carbon spacing by using chemical activators to obtain high and stable SSA suitable for any kind of battery application.
- a better understanding of the mechanism(s) and effect(s) of SSA, pore structure, interlayer spacing, and surface chemistry on electrochemical performance in the preparation of high-efficient biomass-carbon electrodes in a battery application.
- Weak development of more sustainable (e.g., cheaper and environmentally friendly) large-scale synthesis capabilities for biomass-carbon electrodes with suitable structures and properties for commercial battery application.
- A deeper understanding of the composition of biomass precursors and the biomass-related effects on the properties of the final bio-based carbons.
- Deepening in the charge storage mechanisms (intercalation versus pseudocapacitance) in connection with textural properties, pore size, distribution, etc.
- A deeper understanding of the carbon electrode properties on the large volumetric variation induced by large ions (K+, AlCl4−, and Al2Cl7−) size during charge and discharge processes and their sluggish reactions kinetic, so carbon electrodes with outstanding physicochemical features must be developed to overcome these issues.
- Biomass-derived ACs are potential anode candidates for future battery technology such as ABs. Studies regarding the Al3+ storage mechanism and its consequences on the physical, chemical, and physicochemical properties of ACs have not progressed much. Hence we recommend pursuing research in this direction as the next immediate step.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Wang, M.; Ma, J.; Yang, H.; Lu, G.; Yang, S.; Chang, Z. Nitrogen and Cobalt Co-Coped Carbon Materials Derived from Biomass Chitin as High-Performance Electrocatalyst for Aluminum-Air Batteries. Catalysts 2019, 9, 954. [Google Scholar] [CrossRef]
- Sultana, I.; Rahman, M.M.; Chen, Y.; Glushenkov, A.M. Potassium-Ion Battery Anode Materials Operating through the Alloying–Dealloying Reaction Mechanism. Adv. Funct. Mater. 2018, 28, 1703857. [Google Scholar] [CrossRef]
- Li, F.; Wei, Z.; Manthiram, A.; Feng, Y.; Ma, J.; Mai, L. Sodium-based batteries: From critical materials to battery systems. J. Mater. Chem. A 2019, 7, 9406–9431. [Google Scholar] [CrossRef]
- Mukherjee, S.; Banwait, A.; Grixti, S.; Koratkar, N.; Singh, C.V. Adsorption and Diffusion of Lithium and Sodium on Defective Rhenium Disulfide: A First Principles Study. ACS Appl. Mater. Interfaces 2018, 10, 5373–5384. [Google Scholar] [CrossRef]
- Fang, G.; Wu, Z.; Zhou, J.; Zhu, C.; Cao, X.; Lin, T.; Chen, Y.; Wang, C.; Pan, A.; Liang, S. Observation of Pseudocapacitive Effect and Fast Ion Diffusion in Bimetallic Sulfides as an Advanced Sodium-Ion Battery Anode. Adv. Energy Mater. 2018, 8, 1703155. [Google Scholar] [CrossRef]
- Xiong, P.; Zhao, X.; Xu, Y. Nitrogen-Doped Carbon Nanotubes Derived from Metal–Organic Frameworks for Potassium-Ion Battery Anodes. ChemSusChem 2018, 11, 202–208. [Google Scholar] [CrossRef]
- Komaba, S.; Hasegawa, T.; Dahbi, M.; Kubota, K. Potassium intercalation into graphite to realize high-voltage/high-power potassium-ion batteries and potassium-ion capacitors. Electrochem. Commun. 2015, 60, 172–175. [Google Scholar] [CrossRef]
- Zhou, Z.; Li, N.; Wang, P.; Song, W.L.; Jiao, S.; Chen, H.; Fang, D. All-carbon positive electrodes for stable aluminium batteries. J. Energy Chem. 2020, 42, 17–26. [Google Scholar] [CrossRef]
- Zhang, K.; Kirlikovali, K.O.; Suh, J.M.; Choi, J.-W.; Jang, H.W.; Varma, R.S.; Farha, O.K.; Shokouhimehr, M. Recent Advances in Rechargeable Aluminum-Ion Batteries and Considerations for Their Future Progress. ACS Appl. Energy Mater. 2020, 3, 6019–6035. [Google Scholar] [CrossRef]
- Uemura, Y.; Chen, C.Y.; Hashimoto, Y.; Tsuda, T.; Matsumoto, H.; Kuwabata, S. Graphene Nanoplatelet Composite Cathode for a Chloroaluminate Ionic Liquid-Based Aluminum Secondary Battery. ACS Appl. Energy Mater. 2018, 1, 2269–2274. [Google Scholar] [CrossRef]
- Kim, D.J.; Yoo, D.-J.; Otley, M.T.; Prokofjevs, A.; Pezzato, C.; Owczarek, M.; Lee, S.J.; Choi, J.W.; Stoddart, J.F. Rechargeable aluminium organic batteries. Nat. Energy 2019, 4, 51–59. [Google Scholar] [CrossRef]
- Chen, J.; Zhu, Q.; Jiang, L.; Liu, R.; Yang, Y.; Tang, M.; Wang, J.; Wang, H.; Guo, L. Rechargeable Aqueous Aluminum Organic Batteries. Angew. Chemie Int. Ed. 2021, 60, 5794–5799. [Google Scholar] [CrossRef]
- Zhou, L.; Zhang, Z.; Cui, L.; Xiong, F.; An, Q.; Zhou, Z.; Yu, X.-F.; Chu, P.K.; Zhang, K. High-capacity and small-polarization aluminum organic batteries based on sustainable quinone-based cathodes with Al3+ insertion. Cell Reports Phys. Sci. 2021, 2, 100354. [Google Scholar] [CrossRef]
- Han, X.; Li, S.; Song, W.-L.; Chen, N.; Chen, H.; Huang, S.; Jiao, S. Stable High-Capacity Organic Aluminum–Porphyrin Batteries. Adv. Energy Mater. 2021, 11, 2101446. [Google Scholar] [CrossRef]
- Zhang, W.; Huang, W.; Zhang, Q. Organic Materials as Electrodes in Potassium-Ion Batteries. Chem.–A Eur. J. 2021, 27, 6131–6144. [Google Scholar] [CrossRef]
- Li, C.; Wang, K.; Li, J.; Zhang, Q. Nanostructured potassium–organic framework as an effective anode for potassium-ion batteries with a long cycle life. Nanoscale 2020, 12, 7870–7874. [Google Scholar] [CrossRef]
- Ding, C.; Li, C.; Tian, H.; Tong, Y.; Huang, W.; Zhang, Q. Recent Progress on Organic Electrode Materials for Multivalent (Zn, Al, Mg, Ca) Secondary Batteries. Batter. Supercaps 2022, 5, e202200160. [Google Scholar] [CrossRef]
- Zhou, W.; Zhang, M.; Kong, X.; Huang, W.; Zhang, Q. Recent Advance in Ionic-Liquid-Based Electrolytes for Rechargeable Metal-Ion Batteries. Adv. Sci. 2021, 8, 2004490. [Google Scholar] [CrossRef]
- Sun, T.; Xie, J.; Guo, W.; Li, D.-S.; Zhang, Q. Covalent–Organic Frameworks: Advanced Organic Electrode Materials for Rechargeable Batteries. Adv. Energy Mater. 2020, 10, 1904199. [Google Scholar] [CrossRef]
- Xie, J.; Zhang, Q. Recent progress in aqueous monovalent-ion batteries with organic materials as promising electrodes. Mater. Today Energy 2020, 18, 100547. [Google Scholar] [CrossRef]
- Kumar, A.; Bhattacharya, T.; Mozammil Hasnain, S.M.; Kumar Nayak, A.; Hasnain, M.S. Applications of biomass-derived materials for energy production, conversion, and storage. Mater. Sci. Energy Technol. 2020, 3, 905–920. [Google Scholar] [CrossRef]
- Dos Reis, G.S.; Larsson, S.H.; de Oliveira, H.P.; Thyrel, M.; Lima, E.C. Sustainable biomass activated carbons as electrodes for battery and Supercapacitors–A mini-review. Nanomaterials 2020, 10, 1398. [Google Scholar] [CrossRef] [PubMed]
- Dos Reis, G.S.; de Oliveira, H.P.; Larsson, S.H.; Thyrel, M.; Lima, E.C. A short review on the electrochemical performance of hierarchical and nitrogen-doped activated biocarbon-based electrodes for supercapacitors. Nanomaterials 2021, 11, 424. [Google Scholar] [CrossRef] [PubMed]
- dos Reis, G.S.; Pinheiro Lima, R.M.A.; Larsson, S.H.; Subramaniyam, C.M.; Dinh, V.M.; Thyrel, M.; de Oliveira, H.P. Flexible supercapacitors of biomass-based activated carbon-polypyrrole on eggshell membranes. J. Environ. Chem. Eng. 2021, 9, 106155. [Google Scholar] [CrossRef]
- Vassilev, S.V.; Baxter, D.; Andersen, L.K.; Vassileva, C.G. An overview of the chemical composition of biomass. Fuel 2010, 89, 913–933. [Google Scholar] [CrossRef]
- Jache, B.; Binder, J.O.; Abe, T.; Adelhelm, P. A comparative study on the impact of different glymes and their derivatives as electrolyte solvents for graphite co-intercalation electrodes in lithium-ion and sodium-ion batteries. Phys. Chem. Chem. Phys. 2016, 18, 14299–14316. [Google Scholar] [CrossRef]
- Guan, Z.; Guan, Z.; Li, Z.; Liu, J.; Yu, K. Characterization and Preparation of Nano-porous Carbon Derived from Hemp Stems as Anode for Lithium-Ion Batteries. Nanoscale Res. Lett. 2019, 14, 338. [Google Scholar] [CrossRef]
- Yu, K.; Wang, J.; Song, K.; Wang, X.; Liang, C.; Dou, Y. Hydrothermal Synthesis of Cellulose-Derived Carbon Nanospheres from Corn Straw as Anode Materials for Lithium ion Batteries. Nanomaterials 2019, 9, 93. [Google Scholar] [CrossRef]
- Wang, G.; Yu, M.; Feng, X. Carbon materials for ion-intercalation involved rechargeable battery technologies. Chem. Soc. Rev. 2021, 50, 2388–2443. [Google Scholar] [CrossRef]
- dos Reis, G.S.; Larsson, S.H.; Mathieu, M.; Thyrel, M.; Pham, T.N. Application of design of experiments (DoE) for optimised production of micro- and mesoporous Norway spruce bark activated carbons. Biomass Convers. Biorefinery 2021, 1, 1–19. [Google Scholar] [CrossRef]
- Rodriguez Correa, C.; Otto, T.; Kruse, A. Influence of the biomass components on the pore formation of activated carbon. Biomass Bioenergy 2017, 97, 53–64. [Google Scholar] [CrossRef]
- Duan, X.H.; Srinivasakannan, C.; Yang, K.B.; Peng, J.H.; Zhang, L.B. Effects of heating method and activating agent on the porous structure of activated carbons from coconut shells. Waste Biomass Valorization 2012, 3, 131–139. [Google Scholar] [CrossRef]
- dos Reis, G.S.; Larsson, S.H.; Thyrel, M.; Pham, T.N.; Lima, E.C.; de Oliveira, H.P.; Dotto, G.L. Preparation and Application of Efficient Biobased Carbon Adsorbents Prepared from Spruce Bark Residues for Efficient Removal of Reactive Dyes and Colors from Synthetic Effluents. Coatings 2021, 11, 772. [Google Scholar] [CrossRef]
- Guy, M.; Mathieu, M.; Anastopoulos, I.P.; Martínez, M.G.; Rousseau, F.; Dotto, G.L.; de Oliveira, H.P.; Lima, E.C.; Thyrel, M.; Larsson, S.H.; et al. Process Parameters Optimization, Characterization, and Application of KOH-Activated Norway Spruce Bark Graphitic Biochars for Efficient Azo Dye Adsorption. Molecules 2022, 27, 456. [Google Scholar] [CrossRef]
- Xiao, X.; Chen, B.; Chen, Z.; Zhu, L.; Schnoor, J.L. Insight into Multiple and Multilevel Structures of Biochars and Their Potential Environmental Applications: A Critical Review. Environ. Sci. Technol. 2018, 52, 5027–5047. [Google Scholar] [CrossRef]
- dos Reis, G.S.; Guy, M.; Mathieu, M.; Jebrane, M.; Lima, E.C.; Thyrel, M.; Dotto, G.L.; Larsson, S.H. A comparative study of chemical treatment by MgCl2, ZnSO4, ZnCl2, and KOH on physicochemical properties and acetaminophen adsorption performance of biobased porous materials from tree bark residues. Colloids Surf. A Physicochem. Eng. Asp. 2022, 642, 128626. [Google Scholar] [CrossRef]
- González-Hourcade, M.; Simões dos Reis, G.; Grimm, A.; Dinh, V.M.; Lima, E.C.; Larsson, S.H.; Gentili, F.G. Microalgae biomass as a sustainable precursor to produce nitrogen-doped biochar for efficient removal of emerging pollutants from aqueous media. J. Clean. Prod. 2022, 348, 131280. [Google Scholar] [CrossRef]
- Tang, J.; Hou, L.; Hu, T.; Fan, S.; Zhou, X.; Yang, J. Influence of oxygen content on the electrochemical behavior of SiOx@C anodes for Li-ion battery. Compos. Commun. 2021, 23, 100544. [Google Scholar] [CrossRef]
- Peng, Y.; Chen, Z.; Zhang, R.; Zhou, W.; Gao, P.; Wu, J.; Liu, H.; Liu, J.; Hu, A.; Chen, X. Oxygen-Containing Functional Groups Regulating the Carbon/Electrolyte Interfacial Properties Toward Enhanced K+ Storage. Nano-Micro Lett. 2021, 13, 192. [Google Scholar] [CrossRef] [PubMed]
- Jerigová, M.; Odziomek, M.; López-Salas, N. “We Are Here!” Oxygen Functional Groups in Carbons for Electrochemical Applications. ACS Omega 2022, 7, 11544–11554. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Zhu, W.; Yuan, F.; Zhang, D.; Li, Z.; Wang, Q.; Li, W.; Wang, H.; Wang, B. Hierarchical porous carbon nanofibers with enhanced capacitive behavior as a flexible self-supporting anode for boosting potassium storage. J. Power Sources 2022, 523, 231043. [Google Scholar] [CrossRef]
- Xu, Y.; Zhang, C.; Zhou, M.; Fu, Q.; Zhao, C.; Wu, M.; Lei, Y. Highly nitrogen doped carbon nanofibers with superior rate capability and cyclability for potassium ion batteries. Nat. Commun. 2018, 9, 1720. [Google Scholar] [CrossRef]
- Molaiyan, P.; Dos Reis, G.S.; Karuppiah, D.; Subramaniyam, C.M.; García-Alvarado, F.; Lassi, U. Recent Progress in Biomass-Derived Carbon Materials for Li-Ion and Na-Ion Batteries—A Review. Batteries 2023, 9, 116. [Google Scholar] [CrossRef]
- Chen, W.; Wan, M.; Liu, Q.; Xiong, X.; Yu, F.; Huang, Y. Heteroatom-Doped Carbon Materials: Synthesis, Mechanism, and Application for Sodium-Ion Batteries. Small Methods 2019, 3, 1800323. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, L.; Meng, Y.; Li, X.; Guo, Y.; Xiao, D. Sodium storage in fluorine-rich mesoporous carbon fabricated by low-temperature carbonization of polyvinylidene fluoride with a silica template. RSC Adv. 2016, 6, 110850–110857. [Google Scholar] [CrossRef]
- Lu, Z.; Gao, D.; Yi, D.; Yang, Y.; Wang, X.; Yao, J. sp2/sp3 Hybridized Carbon as an Anode with Extra Li-Ion Storage Capacity: Construction and Origin. ACS Cent. Sci. 2020, 6, 1451–1459. [Google Scholar] [CrossRef]
- Kabir, H.; Zhu, H.; May, J.; Hamal, K.; Kan, Y.; Williams, T.; Echeverria, E.; McIlroy, D.N.; Estrada, D.; Davis, P.H.; et al. The sp2-sp3 carbon hybridization content of nanocrystalline graphite from pyrolyzed vegetable oil, comparison of electrochemistry and physical properties with other carbon forms and allotropes. Carbon N. Y. 2019, 144, 831–840. [Google Scholar] [CrossRef]
- Vejpravová, J. Mixed sp2–sp3 Nanocarbon Materials: A Status Quo Review. Nanomaterials 2021, 11, 2469. [Google Scholar] [CrossRef]
- Liu, X.; Ji, T.; Guo, H.; Wang, H.; Li, J.; Liu, H.; Shen, Z. Effects of Crystallinity and Defects of Layered Carbon Materials on Potassium Storage: A Review and Prediction. Electrochem. Energy Rev. 2022, 5, 401–433. [Google Scholar] [CrossRef]
- Wang, A.; Kadam, S.; Li, H.; Shi, S.; Qi, Y. Review on modeling of the anode solid electrolyte interphase (SEI) for lithium-ion batteries. npj Comput. Mater. 2018, 4, 15. [Google Scholar] [CrossRef]
- Yuan, X.; Zhu, B.; Feng, J.; Wang, C.; Cai, X.; Qin, R. Recent advance of biomass-derived carbon as anode for sustainable potassium ion battery. Chem. Eng. J. 2021, 405, 126897. [Google Scholar] [CrossRef]
- Yang, M.; Dai, J.; He, M.; Duan, T.; Yao, W. Biomass-derived carbon from Ganoderma lucidum spore as a promising anode material for rapid potassium-ion storage. J. Colloid Interface Sci. 2020, 567, 256–263. [Google Scholar] [CrossRef]
- Wu, Z.; Zou, J.; Zhang, Y.; Lin, X.; Fry, D.; Wang, L.; Liu, J. Lignin-derived hard carbon anode for potassium-ion batteries: Interplay among lignin molecular weight, material structures, and storage mechanisms. Chem. Eng. J. 2022, 427, 131547. [Google Scholar] [CrossRef]
- Li, W.; Li, Z.; Zhang, C.; Liu, W.; Han, C.; Yan, B.; An, S.; Qiu, X. Hard carbon derived from rice husk as anode material for high performance potassium-ion batteries. Solid State Ionics 2020, 351, 115319. [Google Scholar] [CrossRef]
- Wu, F.; Liu, L.; Yuan, Y.; Li, Y.; Bai, Y.; Li, T.; Lu, J.; Wu, C. Expanding Interlayer Spacing of Hard Carbon by Natural K + Doping to Boost Na-Ion Storage. ACS Appl. Mater. Interfaces 2018, 10, 27030–27038. [Google Scholar] [CrossRef]
- Wang, Q.; Gao, C.; Zhang, W.; Luo, S.; Zhou, M.; Liu, Y.; Liu, R.; Zhang, Y.; Wang, Z.; Hao, A. Biomorphic carbon derived from corn husk as a promising anode materials for potassium ion battery. Electrochim. Acta 2019, 324, 134902. [Google Scholar] [CrossRef]
- Tian, S.; Guan, D.; Lu, J.; Zhang, Y.; Liu, T.; Zhao, X.; Yang, C.; Nan, J. Synthesis of the electrochemically stable sulfur-doped bamboo charcoal as the anode material of potassium-ion batteries. J. Power Sources 2020, 448, 227572. [Google Scholar] [CrossRef]
- Deng, Q.; Liu, H.; Zhou, Y.; Luo, Z.; Wang, Y.; Zhao, Z.; Yang, R. N-doped three-dimensional porous carbon materials derived from bagasse biomass as an anode material for K-ion batteries. J. Electroanal. Chem. 2021, 899, 115668. [Google Scholar] [CrossRef]
- Chen, C.; Wang, Z.; Zhang, B.; Miao, L.; Cai, J.; Peng, L.; Huang, Y.; Jiang, J.; Huang, Y.; Zhang, L.; et al. Nitrogen-rich hard carbon as a highly durable anode for high-power potassium-ion batteries. Energy Storage Mater. 2017, 8, 161–168. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhao, R.; Li, Y.; Zhu, X.; Zhang, B.; Lang, X.; Zhao, L.; Jin, B.; Zhu, Y.; Jiang, Q. Potassium-ion batteries with novel N, O enriched corn silk-derived carbon as anode exhibiting excellent rate performance. J. Power Sources 2021, 481, 228644. [Google Scholar] [CrossRef]
- Wang, P.; Gong, Z.; Wang, D.; Hu, R.; Ye, K.; Gao, Y.; Zhu, K.; Yan, J.; Wang, G.; Cao, D. Facile fabrication of F-doped biomass carbon as high-performance anode material for potassium-ion batteries. Electrochim. Acta 2021, 389, 138799. [Google Scholar] [CrossRef]
- Liu, M.; Jing, D.; Shi, Y.; Zhuang, Q. Superior potassium storage in natural O/N–doped hard carbon derived from maple leaves. J. Mater. Sci. Mater. Electron. 2019, 30, 8911–8919. [Google Scholar] [CrossRef]
- Sun, Y.; Xiao, H.; Li, H.; He, Y.; Zhang, Y.; Hu, Y.; Ju, Z.; Zhuang, Q.; Cui, Y. Nitrogen/Oxygen Co-Doped Hierarchically Porous Carbon for High-Performance Potassium Storage. Chemistry 2019, 25, 7359–7365. [Google Scholar] [CrossRef]
- Petnikota, S.; Chua, R.; Boopathi, K.M.; Satish, R.; Bonaccorso, F.; Pellegrini, V.; Srinivasan, M. An Insight into the Electrochemical Activity of Al-doped V2O3. J. Electrochem. Soc. 2020, 167, 100514. [Google Scholar] [CrossRef]
- Petnikota, S.; Koch, D.; Imran, M.; Buha, J.; Panda, J.K.; Garakani, M.A.; Marasco, L.; Gamucci, A.; Bonaccorso, F.; Pellegrini, V. Spray Coated Few-Layer Graphene as Aluminium Battery Cathode. Sustain. Energy Fuels 2022, 6, 4311–4321. [Google Scholar] [CrossRef]
- Lin, M.C.; Gong, M.; Lu, B.; Wu, Y.; Wang, D.Y.; Guan, M.; Angell, M.; Chen, C.; Yang, J.; Hwang, B.J.; et al. An ultrafast rechargeable aluminium-ion battery. Nature 2015, 520, 324–328. [Google Scholar] [CrossRef]
- Muñoz-Torrero, D.; Palma, J.; Marcilla, R.; Ventosa, E. A critical perspective on rechargeable Al-ion battery technology. Dalt. Trans. 2019, 48, 9906–9911. [Google Scholar] [CrossRef]
- Efimova, A.; Pfützner, L.; Schmidt, P. Thermal stability and decomposition mechanism of 1-ethyl-3-methylimidazolium halides. Thermochim. Acta 2015, 604, 129–136. [Google Scholar] [CrossRef]
- Zhang, L.; Chen, L.; Luo, H.; Zhou, X.; Liu, Z. Large-Sized Few-Layer Graphene Enables an Ultrafast and Long-Life Aluminum-Ion Battery. Adv. Energy Mater. 2017, 7, 1700034. [Google Scholar] [CrossRef]
- Chen, H.; Xu, H.; Wang, S.; Huang, T.; Xi, J.; Cai, S.; Guo, F.; Xu, Z.; Gao, W.; Gao, C. Ultrafast all-climate aluminum-graphene battery with quarter-million cycle life. Sci. Adv. 2017, 3, eaao7233. [Google Scholar] [CrossRef]
- Li, C.; Rath, P.C.; Lu, S.X.; Patra, J.; Su, C.Y.; Bresser, D.; Passerini, S.; Chang, J.K. Ordered nano-structured mesoporous CMK-8 and other carbonaceous positive electrodes for rechargeable aluminum batteries. Chem. Eng. J. 2021, 417, 129131. [Google Scholar] [CrossRef]
- Yang, S.; Knickle, H. Design and analysis of aluminum/air battery system for electric vehicles. J. Power Sources 2002, 112, 162–173. [Google Scholar] [CrossRef]
- About Lithium|London Metal Exchange. Available online: https://www.lme.com/en/metals/ev/about-lithium (accessed on 27 August 2022).
- LME Aluminium|London Metal Exchange. Available online: https://www.lme.com/en/metals/non-ferrous/lme-aluminium#Trading+day+summary (accessed on 27 August 2022).
- USA Gasoline Prices, 22-Aug-2022, GlobalPetrolPrices. Available online: https://www.globalpetrolprices.com/USA/gasoline_prices/ (accessed on 27 August 2022).
- Thanwisai, P.; Chaiyapo, N.; Phuenhinlad, P.; Kanaphan, Y.; Nash, J.; Chotsuwan, C.; Klamchuen, A.; Wang, Y.; Nann, T.; Meethong, N. Mesoporous and defective activated carbon cathode for AlCl4− anion storage in non-aqueous aluminium-ion batteries. Carbon N. Y. 2022, 191, 195–204. [Google Scholar] [CrossRef]
- Gifford, P.R.; Palmisano, J.B. An Aluminum/Chlorine Rechargeable Cell Employing a Room Temperature Molten Salt Electrolyte. J. Electrochem. Soc. 1988, 135, 650–654. [Google Scholar] [CrossRef]
- Yang, Y.; Han, C.; Jiang, B.; Iocozzia, J.; He, C.; Shi, D.; Jiang, T.; Lin, Z. Graphene-based materials with tailored nanostructures for energy conversion and storage. Mater. Sci. Eng. R Rep. 2016, 102, 1–72. [Google Scholar] [CrossRef]
- Wang, D.Y.; Wei, C.Y.; Lin, M.C.; Pan, C.J.; Chou, H.L.; Chen, H.A.; Gong, M.; Wu, Y.; Yuan, C.; Angell, M.; et al. Advanced rechargeable aluminium ion battery with a high-quality natural graphite cathode. Nat. Commun. 2017, 8, 1–7. [Google Scholar] [CrossRef]
- Wang, S.; Jiao, S.; Song, W.L.; Chen, H.S.; Tu, J.; Tian, D.; Jiao, H.; Fu, C.; Fang, D.N. A novel dual-graphite aluminum-ion battery. Energy Storage Mater. 2018, 12, 119–127. [Google Scholar] [CrossRef]
- Zhang, E.; Cao, W.; Wang, B.; Yu, X.; Wang, L.; Xu, Z.; Lu, B. A novel aluminum dual-ion battery. Energy Storage Mater. 2018, 11, 91–99. [Google Scholar] [CrossRef]
- Pan, W.; Wang, Y.; Zhang, Y.; Kwok, H.Y.H.; Wu, M.; Zhao, X.; Leung, D.Y.C. A low-cost and dendrite-free rechargeable aluminium-ion battery with superior performance. J. Mater. Chem. A 2019, 7, 17420–17425. [Google Scholar] [CrossRef]
- Kong, Y.; Tang, C.; Huang, X.; Nanjundan, A.K.; Zou, J.; Du, A.; Yu, C. Thermal Reductive Perforation of Graphene Cathode for High-Performance Aluminum-Ion Batteries. Adv. Funct. Mater. 2021, 31, 2010569. [Google Scholar] [CrossRef]
- Das, S.K.; Mahapatra, S.; Lahan, H. Aluminium-ion batteries: Developments and challenges. J. Mater. Chem. A 2017, 5, 6347–6367. [Google Scholar] [CrossRef]
- Teobaldi, G.; Ohnishi, H.; Tanimura, K.; Shluger, A.L. The effect of van der Waals interactions on the properties of intrinsic defects in graphite. Carbon N. Y. 2010, 48, 4145–4161. [Google Scholar] [CrossRef]
- Lu, N.; Guo, H.; Wang, L.; Wu, X.; Zeng, X.C. van der Waals trilayers and superlattices: Modification of electronic structures of MoS2 by intercalation. Nanoscale 2014, 6, 4566–4571. [Google Scholar] [CrossRef]
- Shkolnikov, E.I.; Novaev, E.M.; Kochanova, S.A.; Ilyukhin, A.S.; Vervikishko, D.E.; Yudina, T.F.; Bratkov, I.V.; Glukhov, L.M. Carbon materials as a cathode for aluminum-ion battery. Mater. Today Proc. 2018, 5, 26073–26077. [Google Scholar] [CrossRef]
- Zafar, Z.A.; Imtiaz, S.; Li, R.; Zhang, J.; Razaq, R.; Xin, Y.; Li, Q.; Zhang, Z.; Huang, Y. A super-long life rechargeable aluminum battery. Solid State Ionics 2018, 320, 70–75. [Google Scholar] [CrossRef]
- Volperts, A.; Dobele, G.; Zhurinsh, A.; Vervikishko, D.; Shkolnikov, E.; Ozolinsh, J. Wood-based activated carbons for supercapacitor electrodes with a sulfuric acid electrolyte. New Carbon Mater. 2017, 32, 319–326. [Google Scholar] [CrossRef]
- Das, S.K. Graphene: A Cathode Material of Choice for Aluminum-Ion Batteries. Angew. Chemie Int. Ed. 2018, 57, 16606–16617. [Google Scholar] [CrossRef]
- Divya, S.; Nann, T. High Voltage Carbon-Based Cathodes for Non-Aqueous Aluminium-Ion Batteries. ChemElectroChem 2021, 8, 492–499. [Google Scholar] [CrossRef]

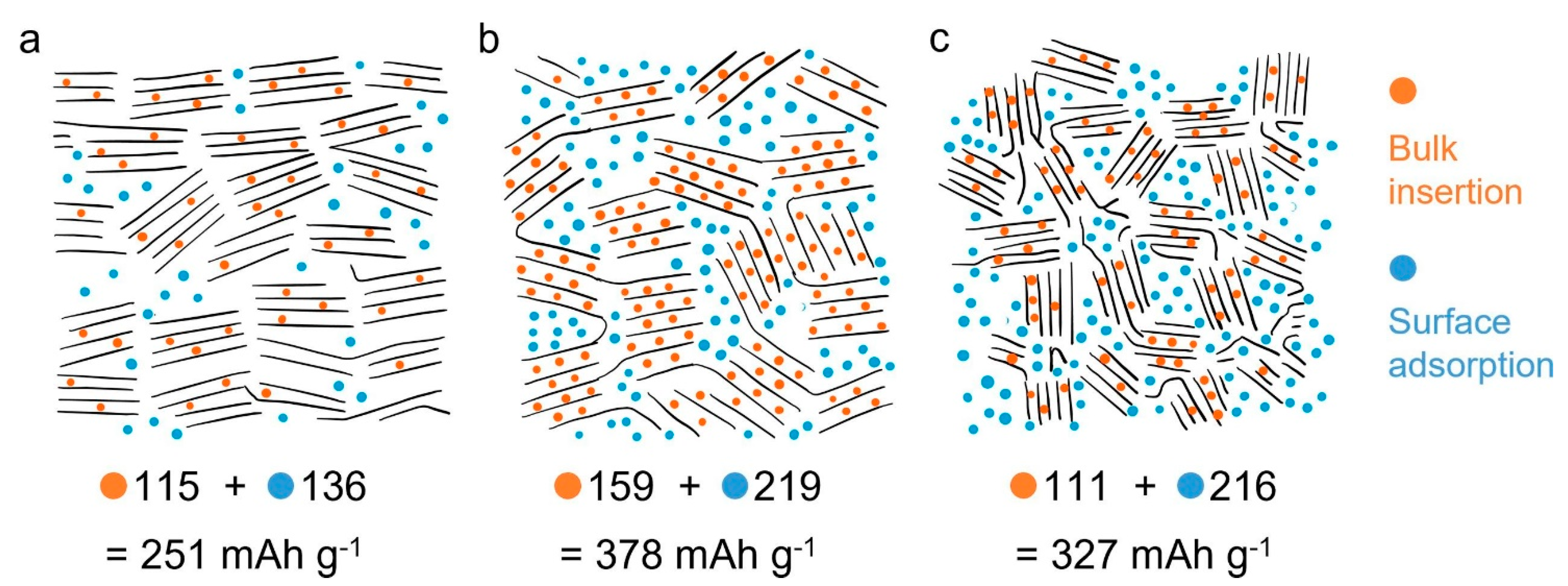


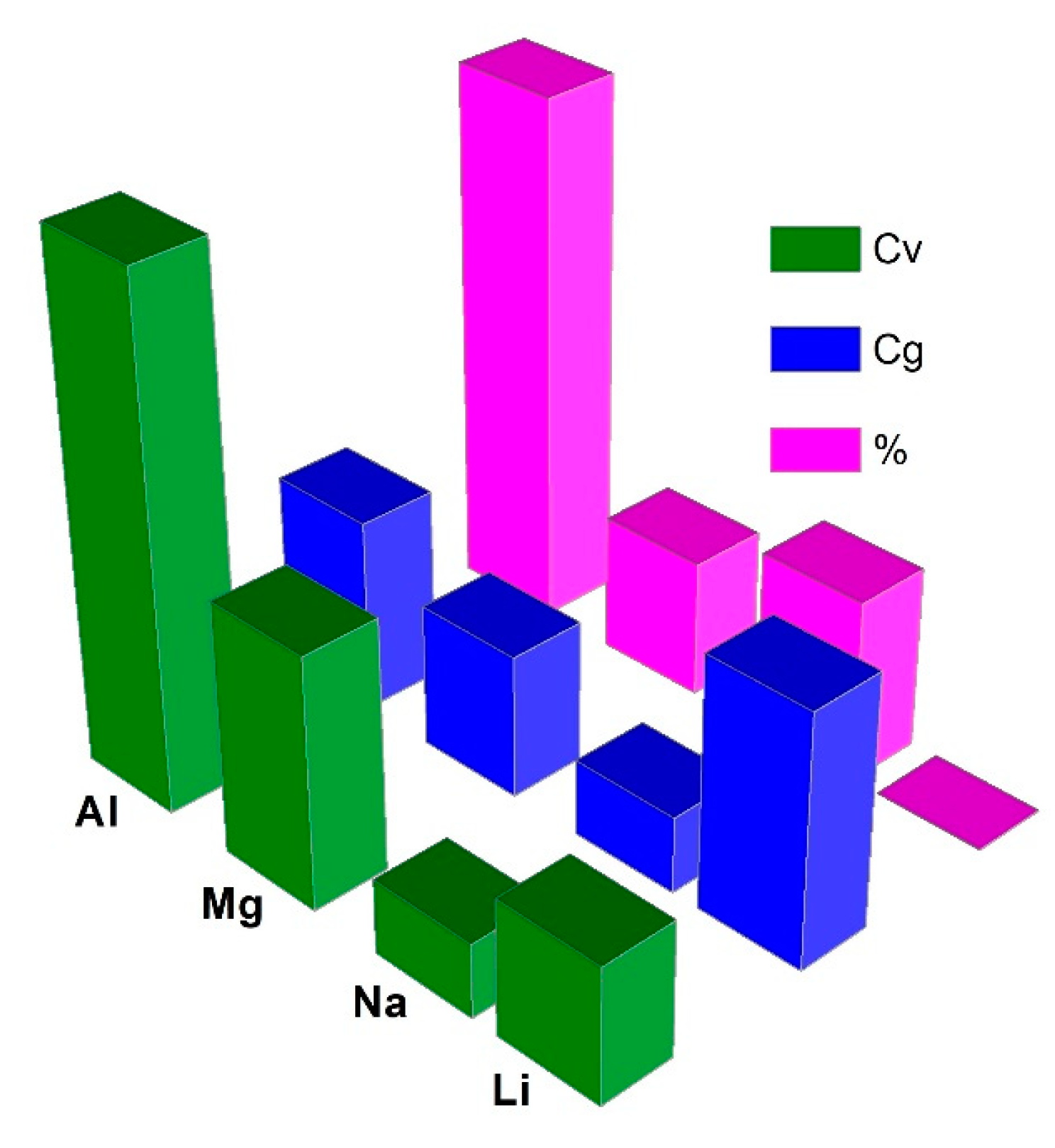

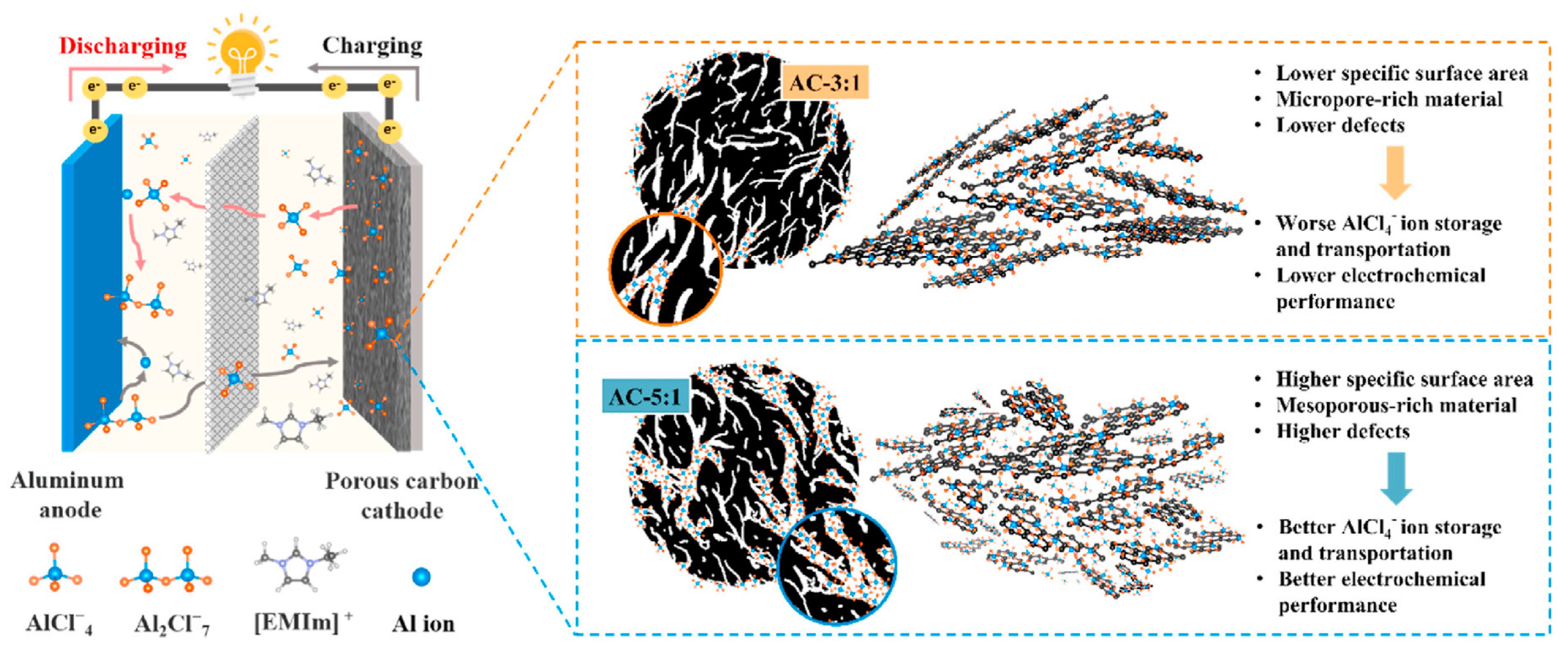
| Metal Anode (Counter Electrode) | Cg (A h g−1) | Cv (A h cm−3) | Eo (V) | rion (Å) | Abundance (wt.%) |
|---|---|---|---|---|---|
| Li+/Li | 3.86 | 2.06 | −3.0 | 0.76 | 0.006 |
| Na+/Na | 1.17 | 1.13 | −2.7 | 1.02 | 2.8 |
| Mg2+/Mg | 2.20 | 3.84 | −2.4 | 0.72 | 2.1 |
| Al3+/Al | 2.98 | 8.04 | −1.7 | 0.53 | 8.1 |
| K+/K | 0.685 | 0.61 | −2.9 | 1.38 | 2.09 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reis, G.S.D.; Petnikota, S.; Subramaniyam, C.M.; de Oliveira, H.P.; Larsson, S.; Thyrel, M.; Lassi, U.; García Alvarado, F. Sustainable Biomass-Derived Carbon Electrodes for Potassium and Aluminum Batteries: Conceptualizing the Key Parameters for Improved Performance. Nanomaterials 2023, 13, 765. https://doi.org/10.3390/nano13040765
Reis GSD, Petnikota S, Subramaniyam CM, de Oliveira HP, Larsson S, Thyrel M, Lassi U, García Alvarado F. Sustainable Biomass-Derived Carbon Electrodes for Potassium and Aluminum Batteries: Conceptualizing the Key Parameters for Improved Performance. Nanomaterials. 2023; 13(4):765. https://doi.org/10.3390/nano13040765
Chicago/Turabian StyleReis, Glaydson Simões Dos, Shaikshavali Petnikota, Chandrasekar M. Subramaniyam, Helinando Pequeno de Oliveira, Sylvia Larsson, Mikael Thyrel, Ulla Lassi, and Flaviano García Alvarado. 2023. "Sustainable Biomass-Derived Carbon Electrodes for Potassium and Aluminum Batteries: Conceptualizing the Key Parameters for Improved Performance" Nanomaterials 13, no. 4: 765. https://doi.org/10.3390/nano13040765
APA StyleReis, G. S. D., Petnikota, S., Subramaniyam, C. M., de Oliveira, H. P., Larsson, S., Thyrel, M., Lassi, U., & García Alvarado, F. (2023). Sustainable Biomass-Derived Carbon Electrodes for Potassium and Aluminum Batteries: Conceptualizing the Key Parameters for Improved Performance. Nanomaterials, 13(4), 765. https://doi.org/10.3390/nano13040765










