Effective Poly (Cyclotriphosphazene-Co-4,4′-Sulfonyldiphenol)@rGO Sheets for Tetracycline Adsorption: Fabrication, Characterization, Adsorption Kinetics and Thermodynamics
Abstract
1. Introduction
2. Experimental Details
2.1. Materials
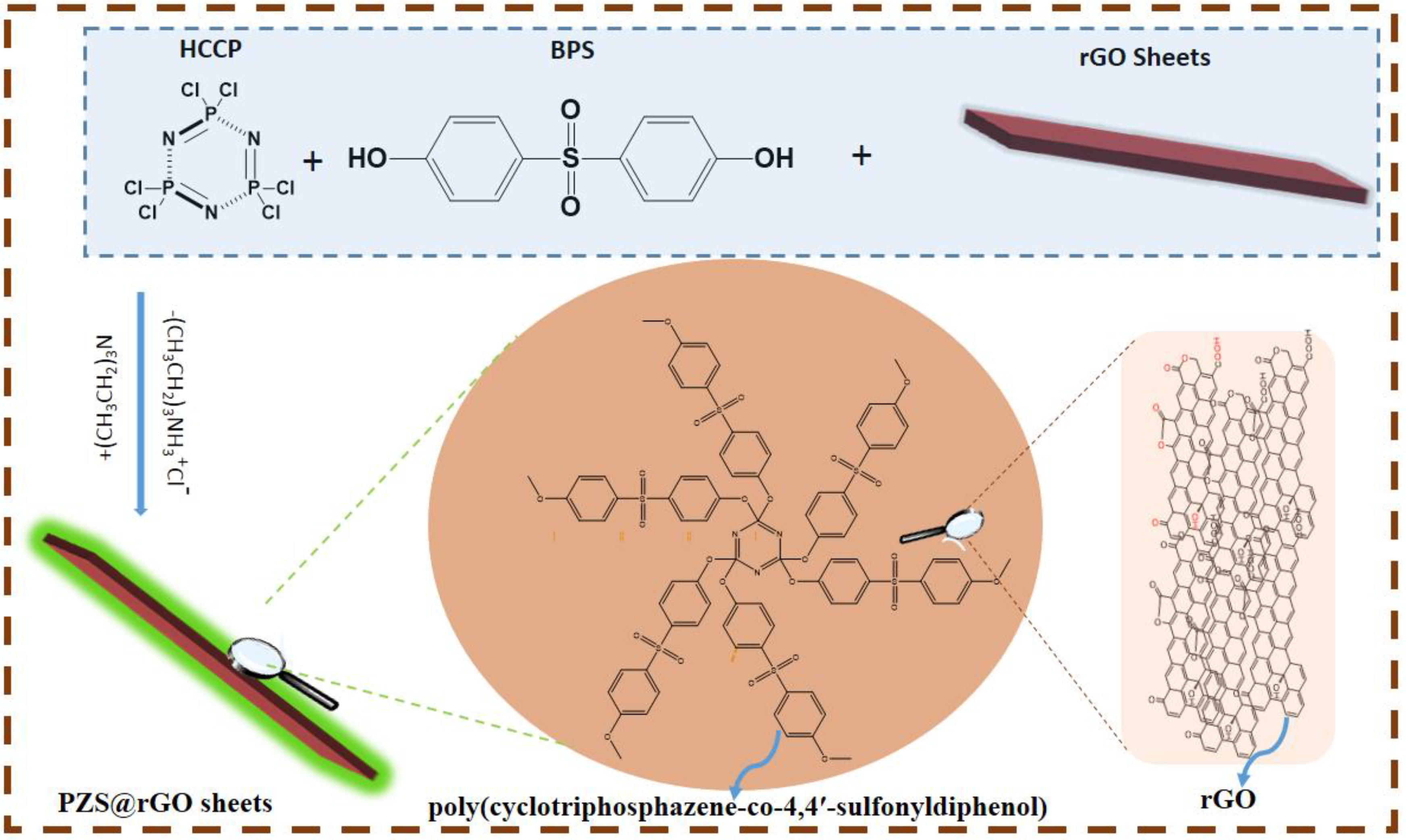
2.2. Synthesis of PZS@rGO
2.3. Adsorption of PZS@rGO Materials for Tetracycline Hydrochloride
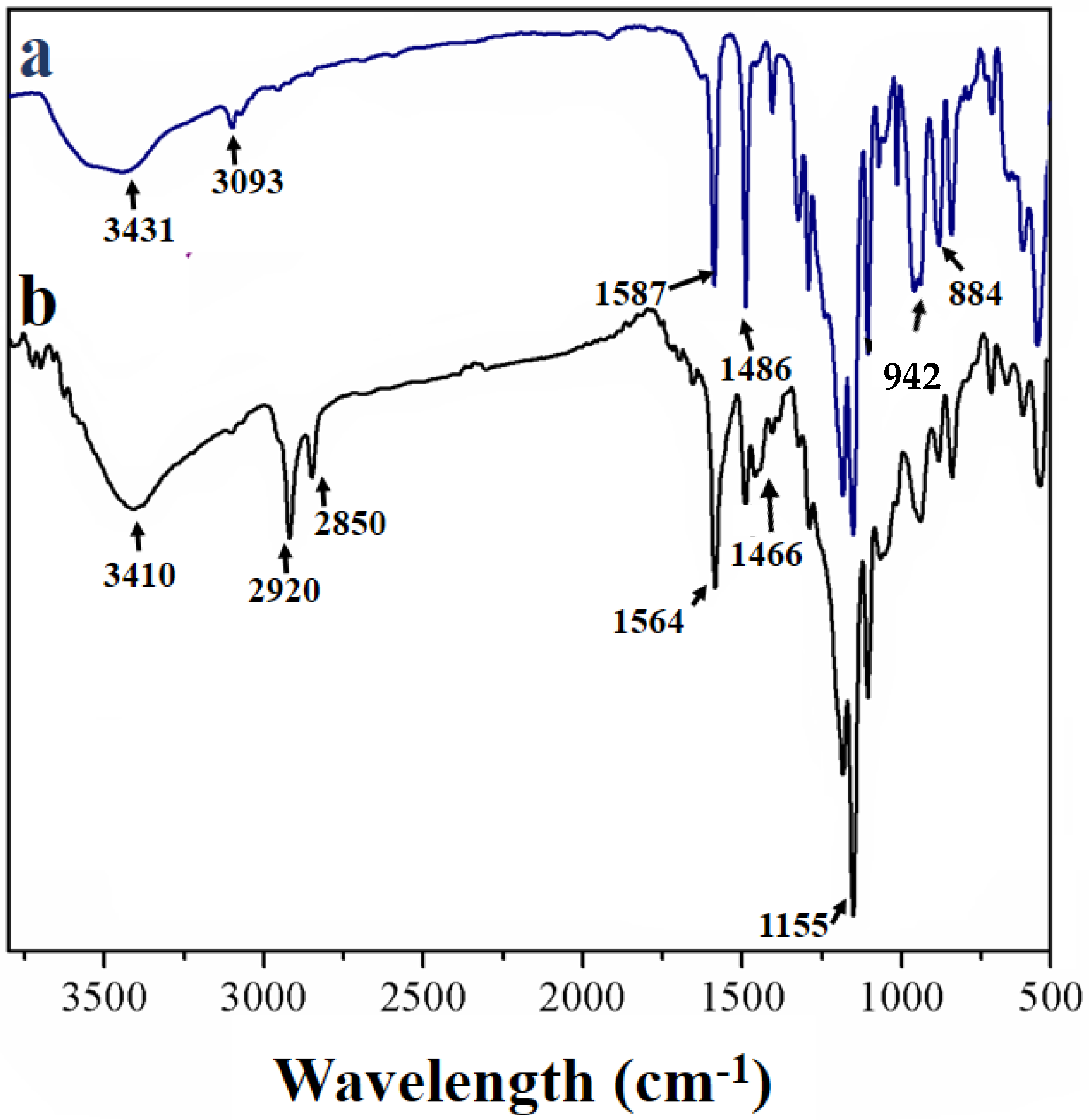
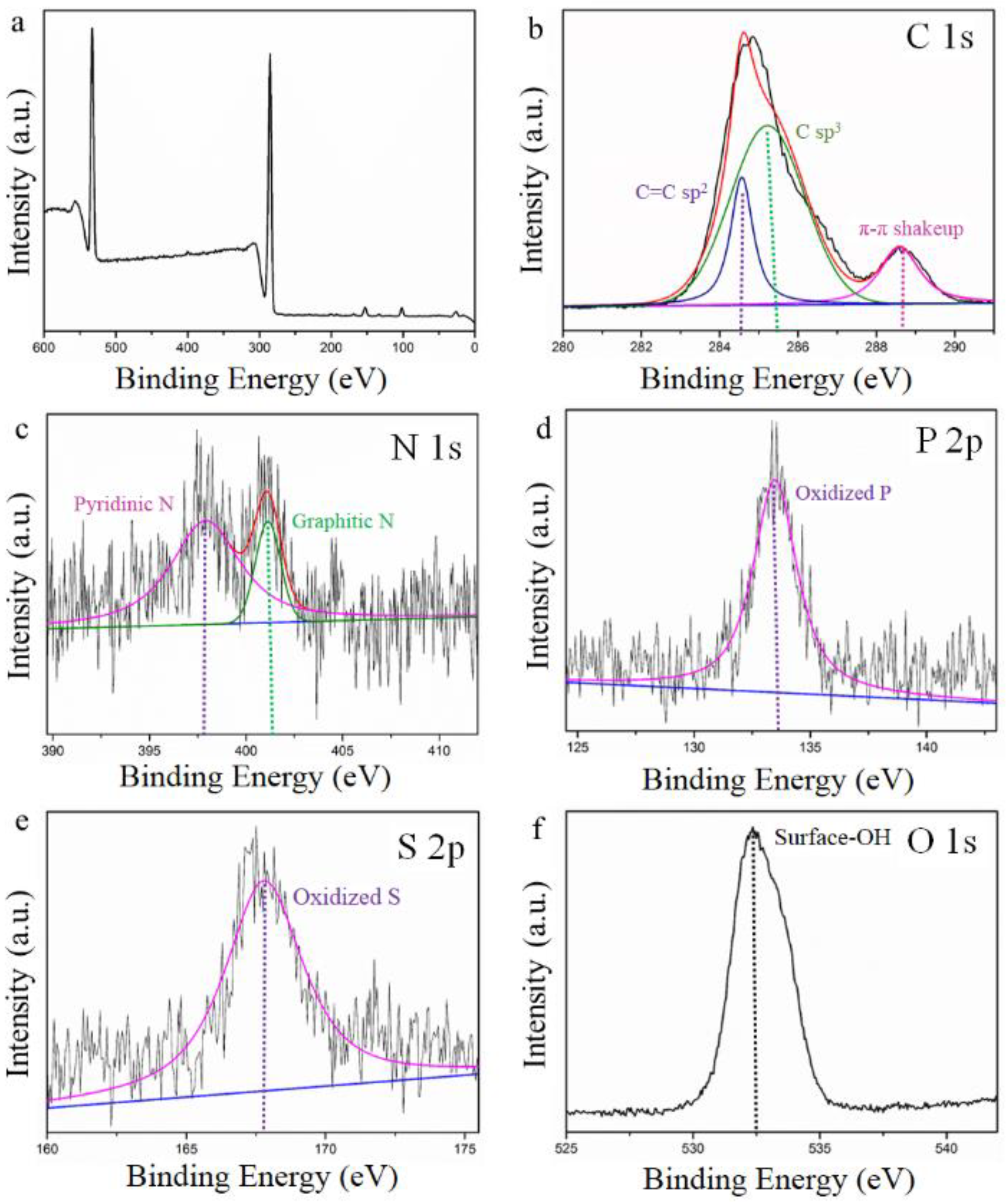
2.4. Characterization
3. Results and Discussion
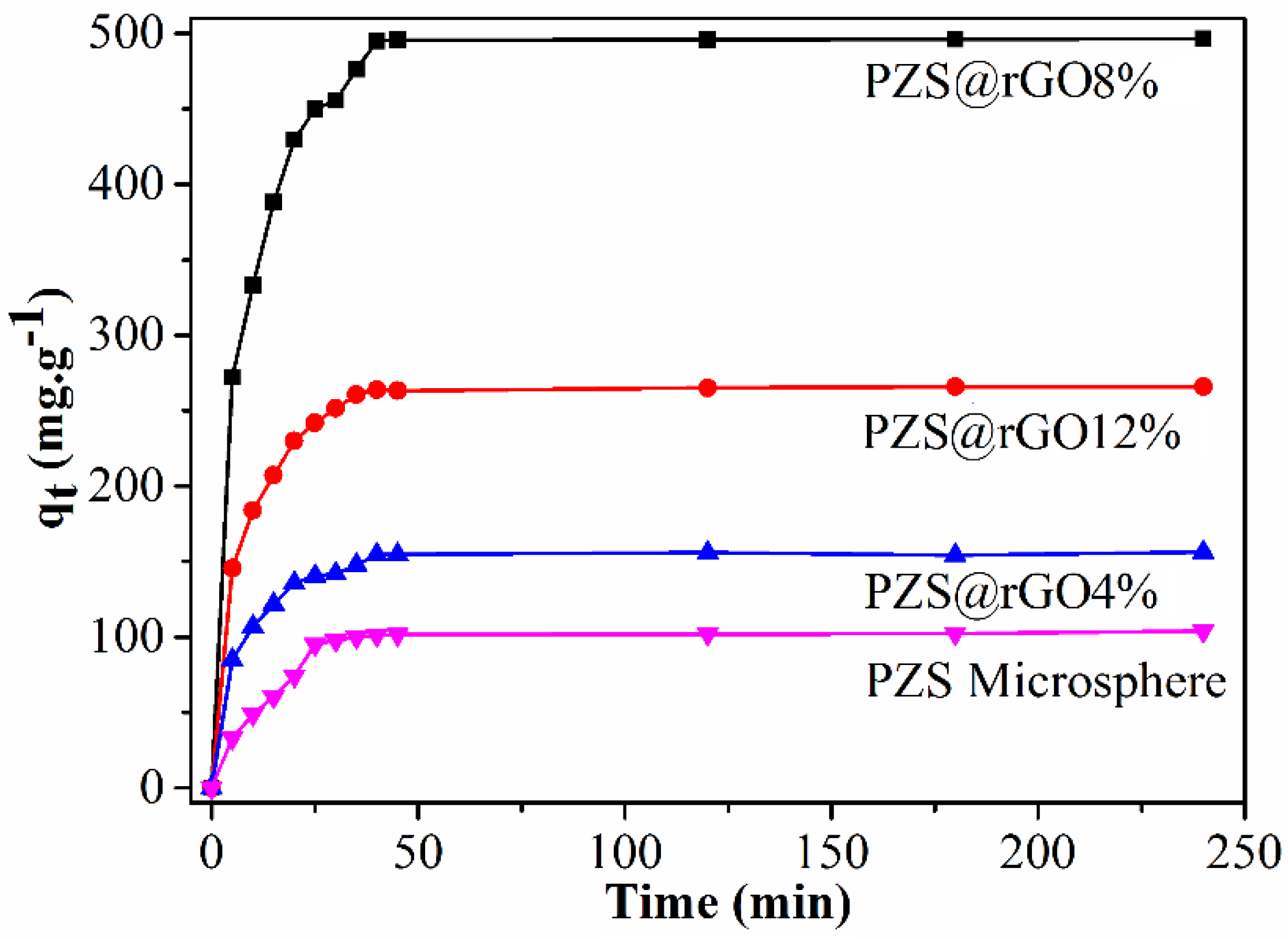
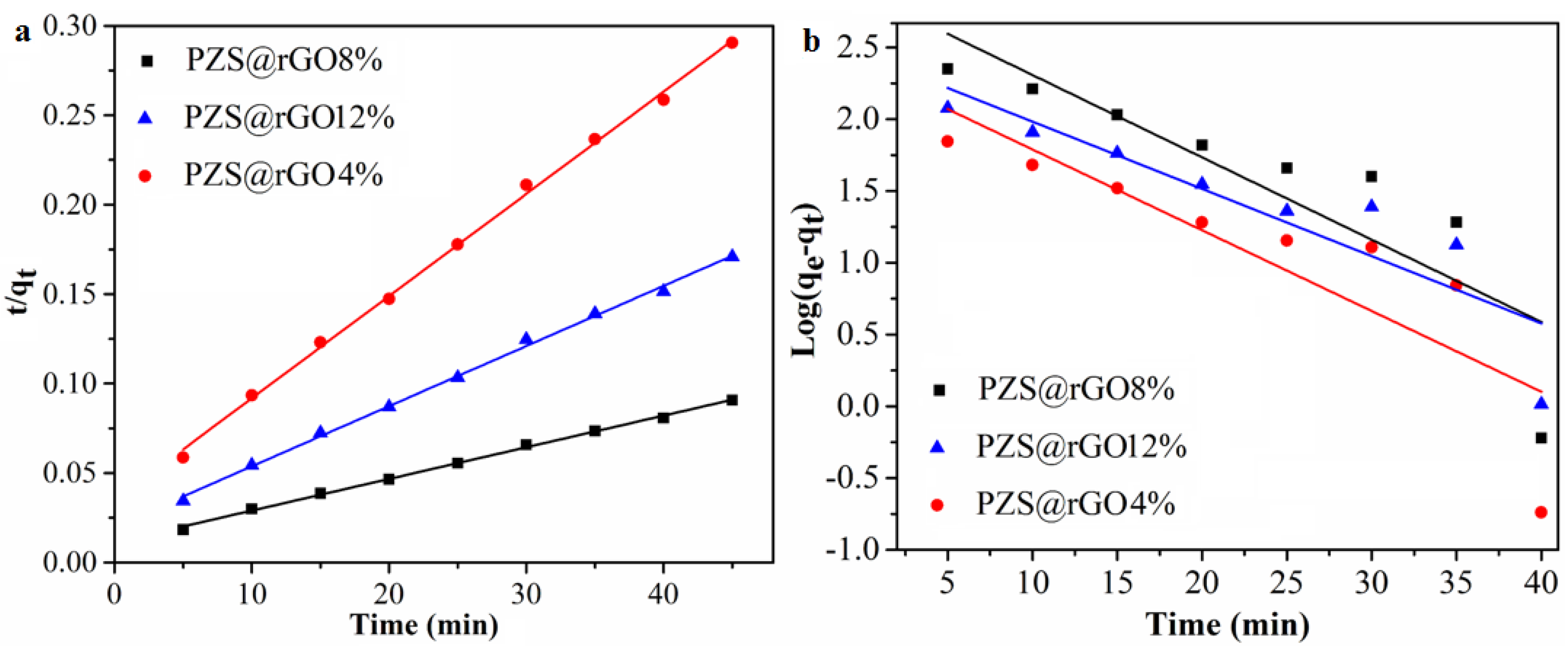
3.1. Adsorption Kinetics and Adsorption Capacity
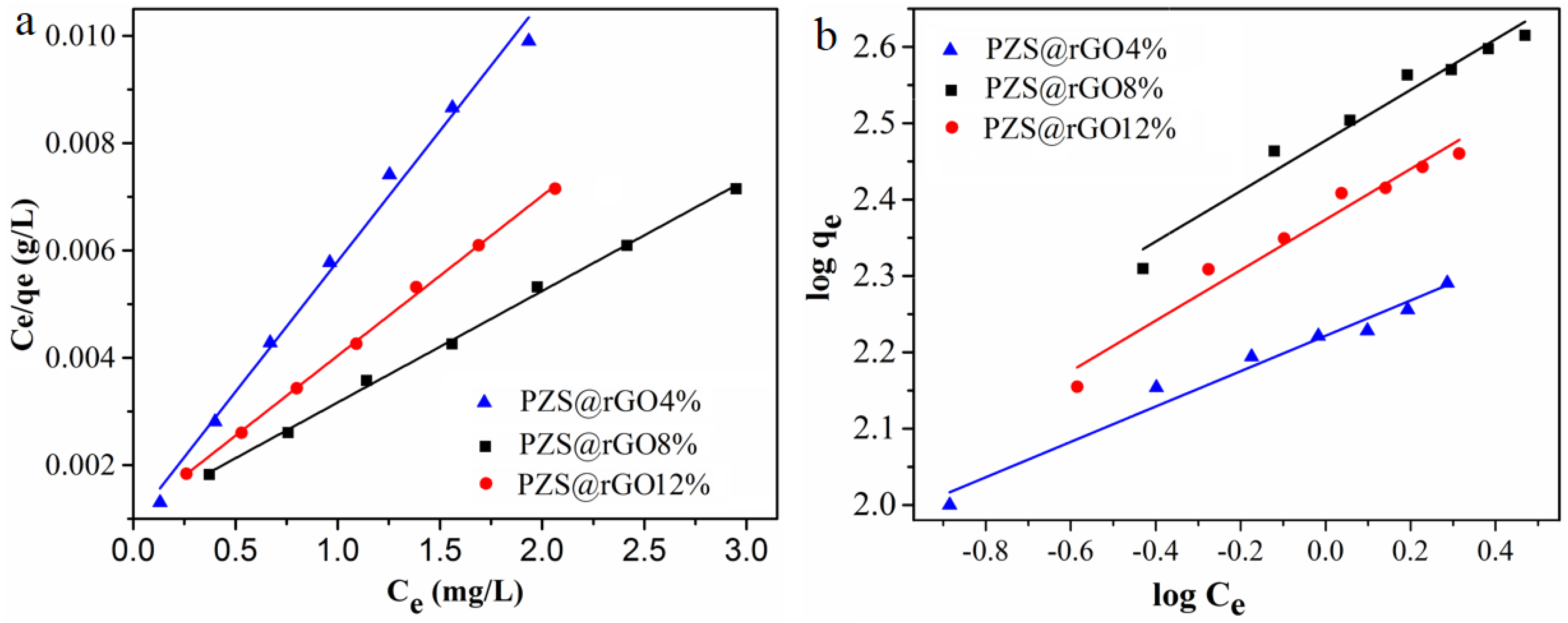
3.2. Adsorption Isotherms
3.3. Batch Experiments
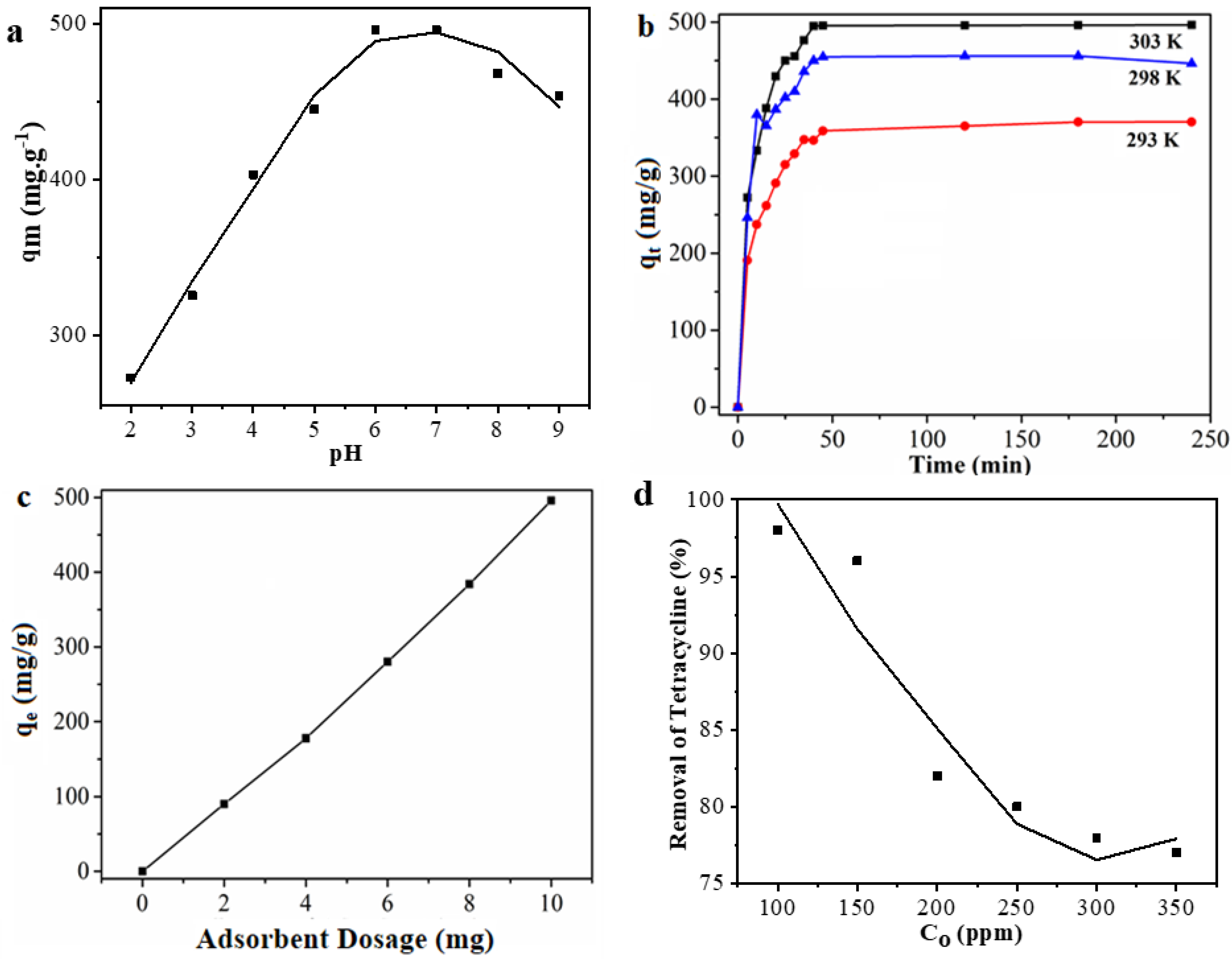
3.3.1. Effect of pH
3.3.2. Effect of Temperature
3.3.3. Effect of Dosage
3.3.4. Effect of Initial Concentrations
3.4. Adsorption Thermodynamics
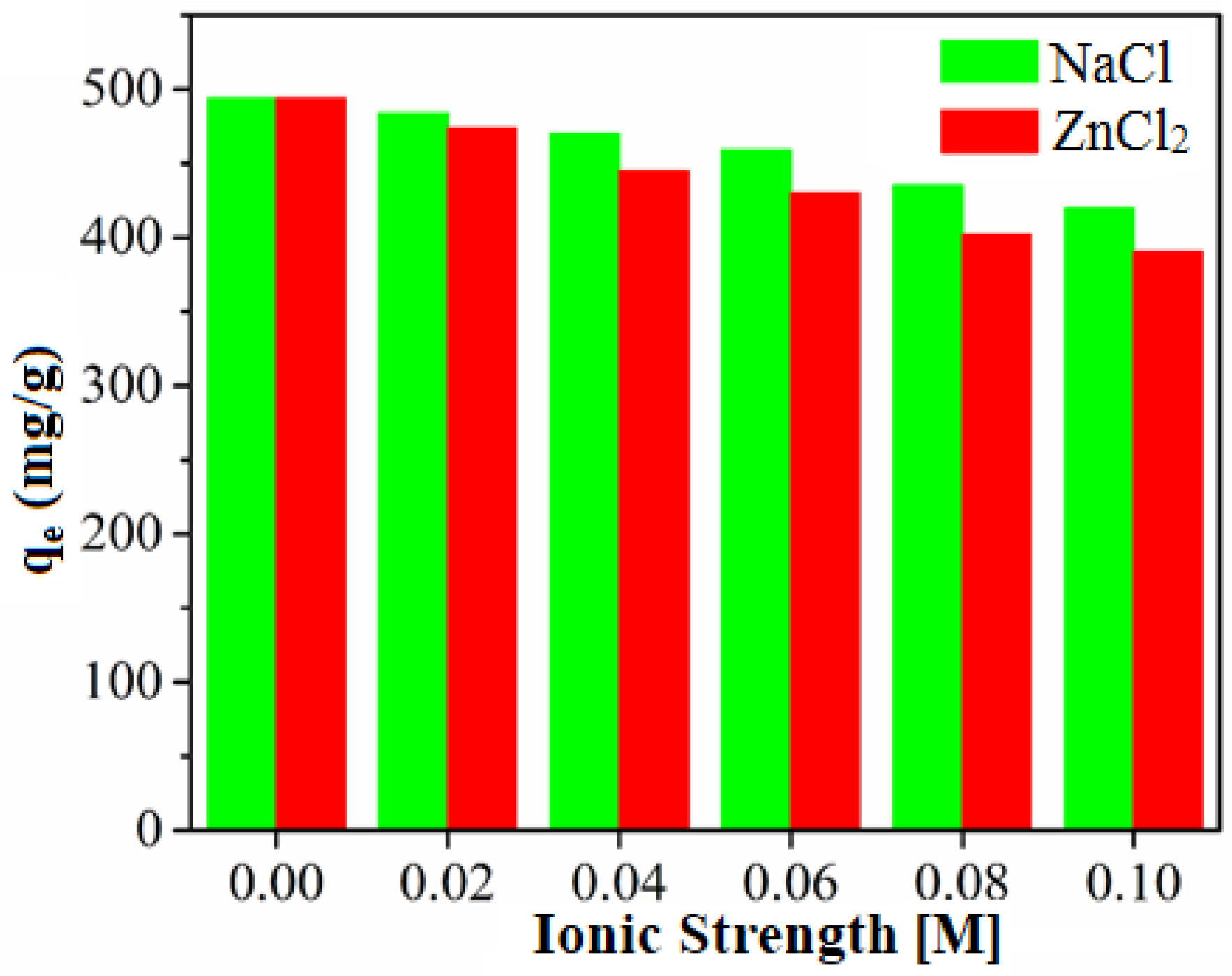
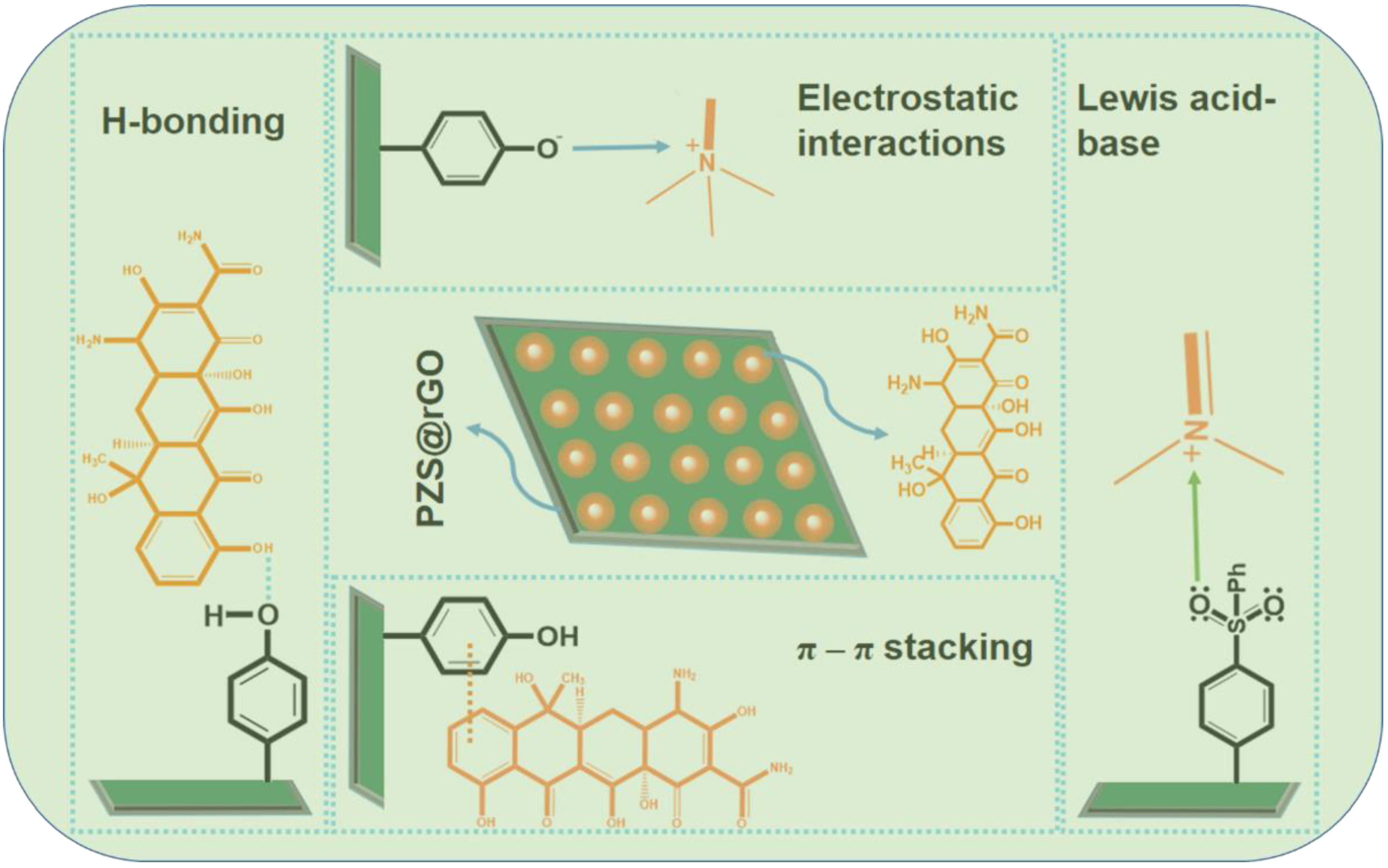
3.5. Mechanism
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ahmed, M.B.; Zhou, J.L.; Ngo, H.H.; Guo, W. Adsorptive removal of antibiotics from water and wastewater: Progress and challenges. Sci. Total. Environ. 2015, 532, 112–126. [Google Scholar] [CrossRef]
- Sassman, S.A.; Lee, L.S. Sorption of three tetracyclines by several soils: Assessing the role of pH and cation exchange. Environ. Sci. Technol. 2005, 39, 7452–7459. [Google Scholar] [CrossRef]
- Westerhoff, P.; Yoon, Y.; Snyder, S.; Wert, E. Fate of endocrine-disruptor, pharmaceutical, and personal care product chemicals during simulated drinking water treatment processes. Environ. Sci. Technol. 2005, 39, 6649–6663. [Google Scholar] [CrossRef] [PubMed]
- Tanis, E.; Hanna, K.; Emmanuel, E. Experimental and modeling studies of sorption of tetracycline onto iron oxides-coated quartz. Colloids Surf. A Physicochem. Eng. Asp. 2008, 327, 57–63. [Google Scholar] [CrossRef]
- Sun, H.; Shi, X.; Mao, J.; Zhu, D. Tetracycline sorption to coal and soil humic acids: An examination of humic structural heterogeneity. Environ. Toxicol. Chem. 2010, 29, 1934–1942. [Google Scholar] [CrossRef] [PubMed]
- Aristilde, L.; Marichal, C.; Miéhé-Brendlé, J.; Lanson, B.; Charlet, L. Interactions of oxytetracycline with a smectite clay: A spectroscopic study with molecular simulations. Environ. Sci. Technol. 2010, 44, 7839–7845. [Google Scholar] [CrossRef]
- Figueroa, R.A.; Leonard, A.; Mackay, A.A. Modeling tetracycline antibiotic sorption to clays. Environ. Sci. Technol. 2004, 38, 476–483. [Google Scholar] [CrossRef]
- Chao, Y.; Zhu, W.; Chen, F.; Wang, P.; Da, Z.; Wu, X.; Ji, H.; Yan, S.; Li, H. Commercial diatomite for adsorption of tetracycline antibiotic from aqueous solution. Sep. Sci. Technol. 2014, 49, 2221–2227. [Google Scholar] [CrossRef]
- Gao, S.; Chen, Y.; Su, J.; Wang, M.; Wei, X.; Jiang, T.; Wang, Z.L. Triboelectric nanogenerator powered electrochemical degradation of organic pollutant using pt-free carbon materials. ACS Nano 2017, 11, 3965–3972. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, M.; Tian, M.; Zhu, Y.; Wei, X.; Jiang, T.; Gao, S. An innovative electro-fenton degradation system self-powered by triboelectric nanogenerator using biomass-derived carbon materials as cathode catalyst. Nano Energy 2017, 42, 314–321. [Google Scholar] [CrossRef]
- Wang, P.; He, Y.-L.; Huang, C.-H. Reactions of tetracycline antibiotics with chlorine dioxide and free chlorine. Water Res. 2011, 45, 1838–1846. [Google Scholar] [CrossRef]
- Zhu, X.-D.; Wang, Y.; Sun, R.-J.; Zhou, D.-M. Photocatalytic degradation of tetracycline in aqueous solution by nanosized TiO2. Chemosphere 2013, 92, 925–932. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Pacheco, C.; Sánchez-Polo, M.; Rivera-Utrilla, J.; López-Peñalver, J. Tetracycline removal from waters by integrated technologies based on ozonation and biodegradation. Chem. Eng. J. 2011, 178, 115–121. [Google Scholar] [CrossRef]
- Okoli, C.P.; Ofomaja, A.E. Development of sustainable magnetic polyurethane polymer nanocomposite for abatement of tetracycline antibiotics aqueous pollution: Response surface methodology and adsorption dynamics. J. Clean. Prod. 2019, 217, 42–55. [Google Scholar] [CrossRef]
- Li, Z.; Chang, P.-H.; Jean, J.-S.; Jiang, W.-T.; Wang, C.-J. Interaction between tetracycline and smectite in aqueous solution. J. Colloid Interface Sci. 2010, 341, 311–319. [Google Scholar] [CrossRef]
- Chen, W.-R.; Huang, C.-H. Adsorption and transformation of tetracycline antibiotics with aluminum oxide. Chemosphere 2010, 79, 779–785. [Google Scholar] [CrossRef]
- Wu, Z.-S.; Ren, W.; Gao, L.; Liu, B.; Jiang, C.; Cheng, H.-M. Synthesis of high-quality graphene with a pre-determined number of layers. Carbon 2009, 47, 493–499. [Google Scholar] [CrossRef]
- Ghadim, E.E.; Manouchehri, F.; Soleimani, G.; Hosseini, H.; Kimiagar, S.; Nafisi, S. Adsorption properties of tetracycline onto graphene oxide: Equilibrium, kinetic and thermodynamic studies. PLoS ONE 2013, 8, e79254. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Huang, X.; Li, W.; Fu, J.; Tang, X. Preparation of novel hybrid inorganic–organic microspheres with active hydroxyl groups using ultrasonic irradiation via one-step precipitation polymerization. Mater. Lett. 2008, 62, 1389–1392. [Google Scholar] [CrossRef]
- Seong, J.-Y.; Jun, Y.J.; Kim, B.M.; Park, Y.M.; Sohn, Y.S. Synthesis and characterization of biocompatible poly(organophosphazenes) aiming for local delivery of protein drugs. Int. J. Pharm. 2006, 314, 90–96. [Google Scholar] [CrossRef]
- Deng, M.; Kumbar, S.G.; Nair, L.S.; Weikel, A.L.; Allcock, H.R.; Laurencin, C.T. Biomimetic structures: Biological implications of dipeptide-substituted polyphosphazene-polyester blend nanofiber matrices for load-bearing bone regeneration. Adv. Funct. Mater. 2011, 21, 2641–2651. [Google Scholar] [CrossRef]
- Allcock, H.R. Recent developments in polyphosphazene materials science. Curr. Opin. Solid State Mater. Sci. 2006, 10, 231–240. [Google Scholar] [CrossRef]
- Ali, S.; Zuhra, Z.; Butler, I.S.; Dar, S.U.; Hameed, M.U.; Wu, D.; Zhang, L.; Wu, Z. High-throughput synthesis of cross-linked poly(cyclotriphosphazene-co-bis(aminomethyl)ferrocene) microspheres and their performance as a superparamagnetic, electrochemical, fluorescent and adsorbent material. Chem. Eng. J. 2017, 315, 448–458. [Google Scholar] [CrossRef]
- Abbas, Y.; Basharat, M.; Liu, W.; Khan, M.S.; Zhang, S.; Ali, S.; Wu, Z.; Wu, D. Substantial role of nitrogen and sulfur in quaternary-atom-doped multishelled carbon nanospheres for the oxygen evolution reaction. ACS Sustain. Chem. Eng. 2020, 8, 4284–4291. [Google Scholar] [CrossRef]
- Abbas, Y.; Zuhra, Z.; Basharat, M.; Qiu, M.; Wu, Z.; Wu, D.; Ali, S. Morphology control of novel cross-linked ferrocenedimethanol derivative cyclophosphazenes: From microspheres to nanotubes and their enhanced physicochemical performances. J. Phys. Chem. B 2019, 123, 4148–4156. [Google Scholar] [CrossRef]
- Abbas, Y.; Ali, S.; Basharat, M.; Zou, W.; Yang, F.; Liu, W.; Zhang, S.; Wu, Z.; Akhtar, N.; Wu, D. Heteroatom-doped carbon nanoparticle–ionic liquid composites as electrochemical sensors for uric acid. ACS Appl. Nano Mater. 2020, 3, 11383–11390. [Google Scholar] [CrossRef]
- Abbas, Y.; Zuhra, Z.; Wu, Z.; Wu, D.; Ali, S. Poly(ferrocenedimethano)cyclotriphosphazene to homogenously Fe, N, P, O doped carbon nanotubes: An efficient and tremendous electrocatalyst for oxygen reduction reaction. J. Electrochem. Soc. 2019, 166, H297–H303. [Google Scholar] [CrossRef]
- Zhu, L.; Xu, Y.; Yuan, W.; Xi, J.; Huang, X.; Tang, X.; Zheng, S. One-pot synthesis of poly(cyclotriphosphazene-co-4,4′-sulfonyldiphenol) nanotubes via an in situ template approach. Adv. Mater. 2006, 18, 2997–3000. [Google Scholar] [CrossRef]
- Fu, J.; Huang, X.; Huang, Y.; Zhang, J.; Tang, X. One-pot noncovalent method to functionalize multi-walled carbon nanotubes using cyclomatrix-type polyphosphazenes. Chem. Commun. 2008, 1049–1051. [Google Scholar] [CrossRef]
- Wang, M.; Fu, J.; Huang, D.; Zhang, C.; Xu, Q. Silver nanoparticles-decorated polyphosphazene nanotubes: Synthesis and applications. Nanoscale 2013, 5, 7913–7919. [Google Scholar] [CrossRef]
- Fu, J.; Chen, Z.; Xu, Q.; Chen, J.; Huang, X.; Tang, X. The production of porous carbon nanofibers from cross-linked polyphosphazene nanofibers. Carbon 2011, 49, 1037–1039. [Google Scholar] [CrossRef]
- Wei, W.; Huang, X.; Tao, Y.; Chen, K.; Tang, X. Enhancement of the electrocapacitive performance of manganese dioxide by introducing a microporous carbon spheres network. Phys. Chem. Chem. Phys. 2012, 14, 5966–5972. [Google Scholar] [CrossRef]
- Hu, Y.; Meng, L.; Niu, L.; Lu, Q. Facile synthesis of superparamagnetic Fe3O4@polyphosphazene@Au shells for magnetic resonance imaging and photothermal therapy. ACS Appl. Mater. Interfaces 2013, 5, 4586–4591. [Google Scholar] [CrossRef]
- Chen, Z.; Fu, J.; Wang, M.; Wang, X.; Zhang, J.; Xu, Q. Adsorption of cationic dye (methylene blue) from aqueous solution using poly(cyclotriphosphazene-co-4,4′-sulfonyldiphenol) nanospheres. Appl. Surf. Sci. 2014, 289, 495–501. [Google Scholar] [CrossRef]
- Wei, W.; Lu, R.; Xie, H.; Zhang, Y.; Bai, X.; Gu, L.; Da, R.; Liu, X. Selective adsorption and separation of dyes from an aqueous solution on organic–inorganic hybrid cyclomatrix polyphosphazene submicro-spheres. J. Mater. Chem. A 2015, 3, 4314–4322. [Google Scholar] [CrossRef]
- Yang, S.; Zhu, Y.; Cao, C.; Peng, L.; Li, S.; Zhai, D.; Song, W. A general route to coat poly (cyclotriphosphazene-co-4, 4′-sulfonyldiphenol) on various substrates and the derived N, P, S-doped hollow carbon shells for catalysis. Nanoscale 2017, 9, 13538–13545. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Chen, X.; Xu, H.; Zou, Y.; Gu, R.; Xu, M.; Jen, A.K.-Y.; Chen, H. Highly-efficient fabrication of nanoscrolls from functionalized graphene oxide by Langmuir–Blodgett method. Carbon 2010, 48, 4475–4482. [Google Scholar] [CrossRef]
- Wang, W.; Wang, X.; Xing, J.; Gong, Q.; Wang, H.; Wang, J.; Chen, Z.; Ai, Y.; Wang, X. Multi-heteroatom doped graphene-like carbon nanospheres with 3D inverse opal structure: A promising bisphenol-A remediation material. Environ. Sci. Nano 2019, 6, 809–819. [Google Scholar] [CrossRef]
- Zhong, H.-X.; Wang, J.; Zhang, Y.-W.; Xu, W.-L.; Xing, W.; Xu, D.; Zhang, Y.-F.; Zhang, X.-B. ZIF-8 derived graphene-based nitrogen-doped porous carbon sheets as highly efficient and durable oxygen reduction electrocatalysts. Angew. Chem. Int. Ed. 2014, 53, 14235–14239. [Google Scholar] [CrossRef]
- Wei, J.; Hu, Y.; Liang, Y.; Kong, B.; Zhang, J.; Song, J.; Bao, Q.; Simon, G.; Jiang, S.P.; Wang, H. Nitrogen-doped nanoporous carbon/graphene nano-sandwiches: Synthesis and application for efficient oxygen reduction. Adv. Funct. Mater. 2015, 25, 5768–5777. [Google Scholar] [CrossRef]
- You, B.; Jiang, N.; Sheng, M.; Drisdell, W.S.; Yano, J.; Sun, Y. Bimetal–organic framework self-adjusted synthesis of support-free nonprecious electrocatalysts for efficient oxygen reduction. ACS Catal. 2015, 5, 7068–7076. [Google Scholar] [CrossRef]
- Razmjooei, F.; Singh, K.P.; Song, M.Y.; Yu, J.-S. Enhanced electrocatalytic activity due to additional phosphorous doping in nitrogen and sulfur-doped graphene: A comprehensive study. Carbon 2014, 78, 257–267. [Google Scholar] [CrossRef]
- Wu, J.; Zheng, X.; Jin, C.; Tian, J.; Yang, R. Ternary doping of phosphorus, nitrogen, and sulfur into porous carbon for enhancing electrocatalytic oxygen reduction. Carbon 2015, 92, 327–338. [Google Scholar] [CrossRef]
- Haul, R.; Gregg, S.J.; Sing, K.S.W. Adsorption, Surface Area and Porosity, 2nd ed.; Academic press: London, UK, 1982; pp. 1–303. [Google Scholar]
- Yan, Q.; Zhang, Z.; Zhang, Y.; Umar, A.; Guo, Z.; O’Hare, D.; Wang, Q. Hierarchical Fe3O4 core-shell layered double hydroxide composites as magnetic adsorbents for anionic dye removal from wastewater. Eur. J. Inorg. Chem. 2015, 2015, 4182–4191. [Google Scholar] [CrossRef]
- Chen, R.; Yu, J.; Xiao, W. Hierarchically porous MnO2 microspheres with enhanced adsorption performance. J. Mater. Chem. A 2013, 1, 11682–11690. [Google Scholar] [CrossRef]
- Acosta, R.; Fierro, V.; de Yuso, A.M.; Nabarlatz, D.A.; Celzard, A. Tetracycline adsorption onto activated carbons produced by KOH activation of tyre pyrolysis char. Chemosphere 2016, 149, 168–176. [Google Scholar] [CrossRef]
- Alam, S.; Khan, M.; Bibi, W.; Zekker, I.; Burlakovs, J.; Ghangrekar, M.; Bhowmick, G.; Kallistova, A.; Pimenov, N.; Zahoor, M. Preparation of activated carbon from the wood of Paulownia tomentosa as an efficient adsorbent for the removal of acid red 4 and methylene blue present in wastewater. Water 2021, 13, 1453. [Google Scholar] [CrossRef]
- Alam, S.; Khan, M.; Umar, A.; Khattak, R.; Rahman, N.; Zekker, I.; Burlakovs, J.; Rubin, S.; Ghangrekar, M.; Bhowmick, G.; et al. Preparation of Pd–Ni nanoparticles supported on activated carbon for efficient removal of basic blue 3 from water. Water 2021, 13, 1211. [Google Scholar] [CrossRef]
- Umar, A.; Khan, M.; Alam, S.; Zekker, I.; Burlakovs, J.; Rubin, S.D.; Bhowmick, G.; Kallistova, A.; Pimenov, N.; Zahoor, M. Synthesis and characterization of Pd-Ni bimetallic nanoparticles as efficient adsorbent for the removal of acid orange 8 present in wastewater. Water 2021, 13, 1095. [Google Scholar] [CrossRef]
- Li, N.; Zhou, L.; Jin, X.; Owens, G.; Chen, Z. Simultaneous removal of tetracycline and oxytetracycline antibiotics from wastewater using a ZIF-8 metal organic-framework. J. Hazard. Mater. 2019, 366, 563–572. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Chen, T.; Liu, J.; Li, D. Adsorption of tetracycline antibiotics from an aqueous solution onto graphene oxide/calcium alginate composite fibers. RSC Adv. 2018, 8, 2616–2621. [Google Scholar] [CrossRef]
- Rafatullah, M.; Ismail, S.; Ahmad, A. Optimization study for the desorption of methylene blue dye from clay based adsorbent coating. Water 2019, 11, 1304. [Google Scholar] [CrossRef]
- Lei, C.; Zhu, X.; Zhu, B.; Jiang, C.; Le, Y.; Yu, J. Superb adsorption capacity of hierarchical calcined Ni/Mg/Al layered double hydroxides for Congo red and Cr(VI) ions. J. Hazard. Mater. 2017, 321, 801–811. [Google Scholar] [CrossRef]
- dos Santos Lins, P.V.; Henrique, D.C.; Ide, A.H.; e Silva, C.L.D.P.; Meili, L. Evaluation of caffeine adsorption by MgAl-LDH/biochar composite. Environ. Sci. Pollut. Res. 2019, 26, 31804–31811. [Google Scholar] [CrossRef]
- Extremera, R.; Pavlovic, I.; Perez, M.R.; Barriga, C. Removal of acid orange 10 by calcined Mg/Al layered double hydroxides from water and recovery of the adsorbed dye. Chem. Eng. J. 2012, 213, 392–400. [Google Scholar] [CrossRef]


| Adsorbents | C0 (ppm) | qe,exp | Pseudo-First-Order Model | Pseudo-Second-Order Model | ||||
|---|---|---|---|---|---|---|---|---|
| (mg g−1) | qe,cal | k1 | R2 | qe,cal | k2 | R2 | ||
| (mg g−1) | (min−1) | (mg g−1) | (g mg−1 min−1) | |||||
| PZS@rGO4% | 100 | 155 | 223.87 | 0.12 | 0.685 | 175.13 | 9.4 × 10−4 | 0.998 |
| PZS@rGO8% | 100 | 496 | 761.18 | 0.13 | 0.711 | 565.97 | 2.77 × 10−4 | 0.998 |
| PZS@rGO12% | 100 | 265 | 282.87 | 0.107 | 0.769 | 297.61 | 5.161 × 10−4 | 0.998 |
| Adsorbents | Langmuir | Freundlich | ||||
|---|---|---|---|---|---|---|
| b (L mg−1) | qm (mg g−1) | R2 | Kf | n | R2 | |
| PZS@rGO8% | 2.08 | 480 | 0.998 | 295 | 3.1 | 0.956 |
| PZS@rGO12% | 2.81 | 336 | 0.998 | 234 | 2.9 | 0.951 |
| PZS@rGO4% | 5.20 | 205.7 | 0.997 | 165.95 | 4.34 | 0.942 |
| Samples | T/(K) | ΔG0/(kJ mol−1) | ΔS0/(kJ K−1 mol−1) | ΔH0s/(kJ mol−1) |
|---|---|---|---|---|
| PZS@rGO8% | 293 | −2.07 | 15.2 | 2.64 |
| 298 | −1.95 | |||
| 303 | −1.84 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmad, M.; Nawaz, T.; Alam, M.M.; Abbas, Y.; Ali, S.; Imran, M.; Zhang, S.; Wu, Z. Effective Poly (Cyclotriphosphazene-Co-4,4′-Sulfonyldiphenol)@rGO Sheets for Tetracycline Adsorption: Fabrication, Characterization, Adsorption Kinetics and Thermodynamics. Nanomaterials 2021, 11, 1540. https://doi.org/10.3390/nano11061540
Ahmad M, Nawaz T, Alam MM, Abbas Y, Ali S, Imran M, Zhang S, Wu Z. Effective Poly (Cyclotriphosphazene-Co-4,4′-Sulfonyldiphenol)@rGO Sheets for Tetracycline Adsorption: Fabrication, Characterization, Adsorption Kinetics and Thermodynamics. Nanomaterials. 2021; 11(6):1540. https://doi.org/10.3390/nano11061540
Chicago/Turabian StyleAhmad, Muhammad, Tehseen Nawaz, Mohammad Mujahid Alam, Yasir Abbas, Shafqat Ali, Muhammad Imran, Shuangkun Zhang, and Zhanpeng Wu. 2021. "Effective Poly (Cyclotriphosphazene-Co-4,4′-Sulfonyldiphenol)@rGO Sheets for Tetracycline Adsorption: Fabrication, Characterization, Adsorption Kinetics and Thermodynamics" Nanomaterials 11, no. 6: 1540. https://doi.org/10.3390/nano11061540
APA StyleAhmad, M., Nawaz, T., Alam, M. M., Abbas, Y., Ali, S., Imran, M., Zhang, S., & Wu, Z. (2021). Effective Poly (Cyclotriphosphazene-Co-4,4′-Sulfonyldiphenol)@rGO Sheets for Tetracycline Adsorption: Fabrication, Characterization, Adsorption Kinetics and Thermodynamics. Nanomaterials, 11(6), 1540. https://doi.org/10.3390/nano11061540







