Effect of Na- and Organo-Modified Montmorillonite/Essential Oil Nanohybrids on the Kinetics of the In Situ Radical Polymerization of Styrene
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of NaMMT/EO and orgMMT/EO Hybrids
2.3. Preparation of the Initial Monomer/MMT Mixtures
2.4. Synthesis of PS/MMTEO Nanocomposites by the In-Situ Bulk Radical Polymerization Technique
2.5. Synthesis of Composites Based on PS with Model Thymol, Carvacrol and Estragol
2.6. Measurements
3. Results
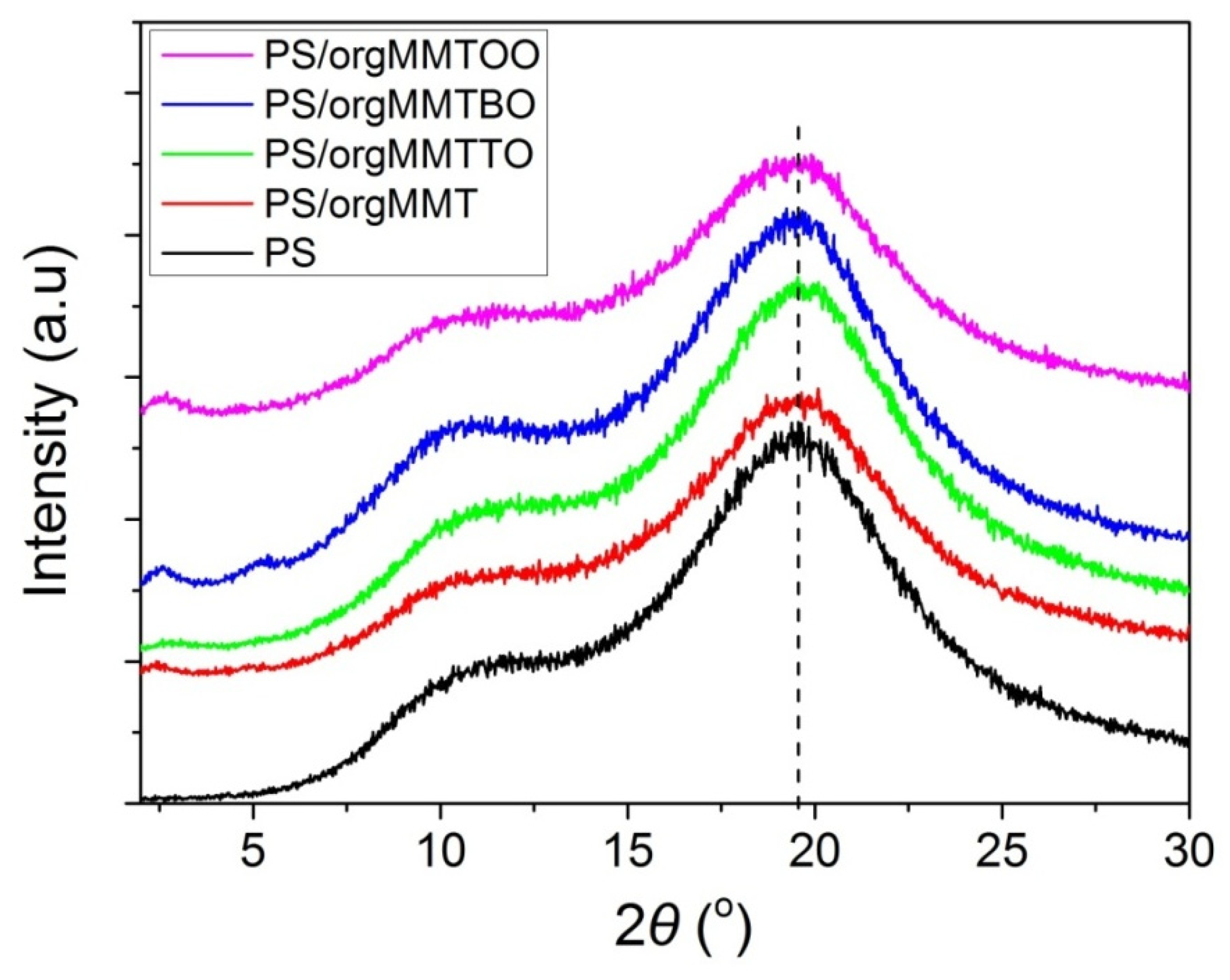
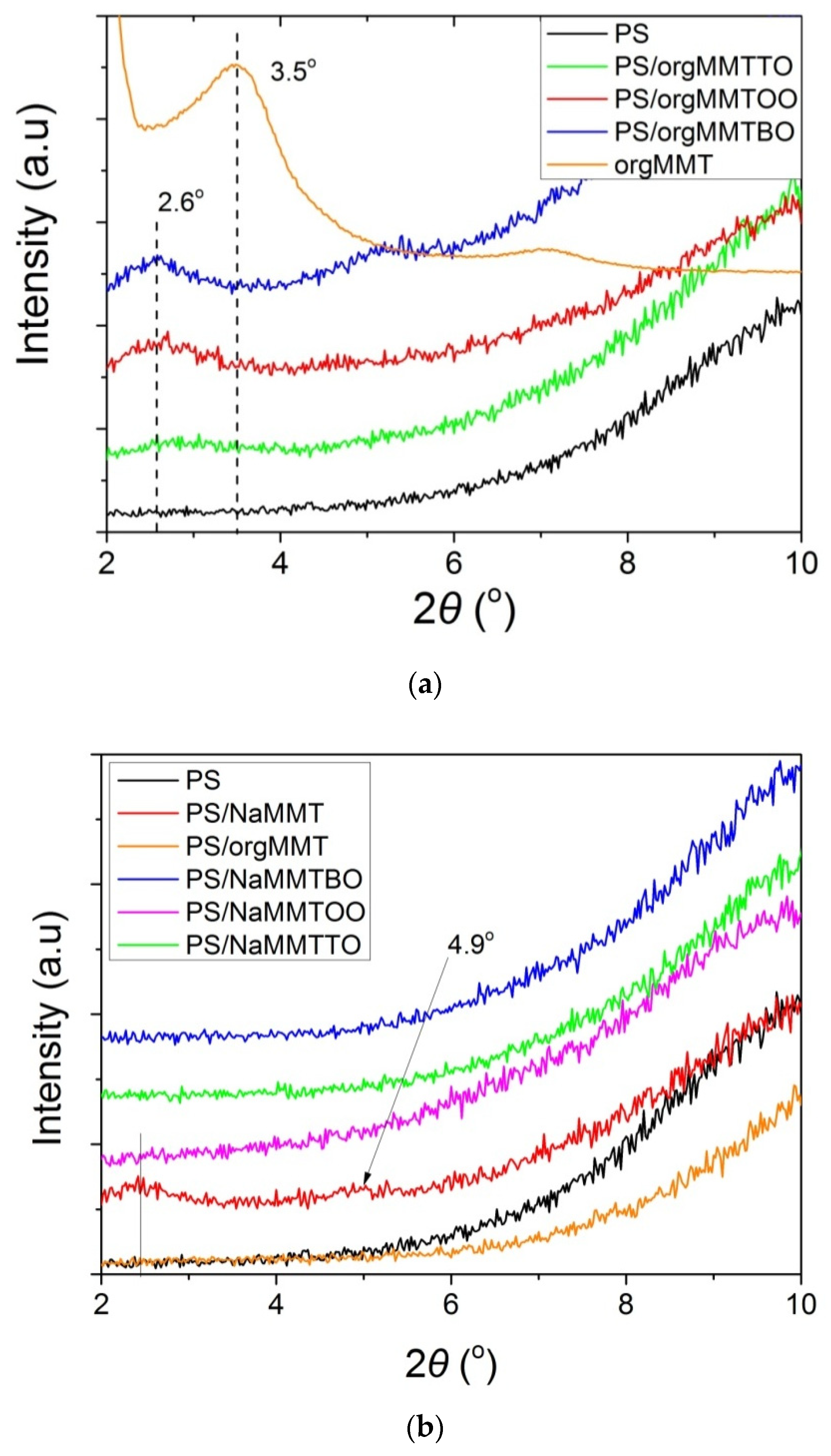
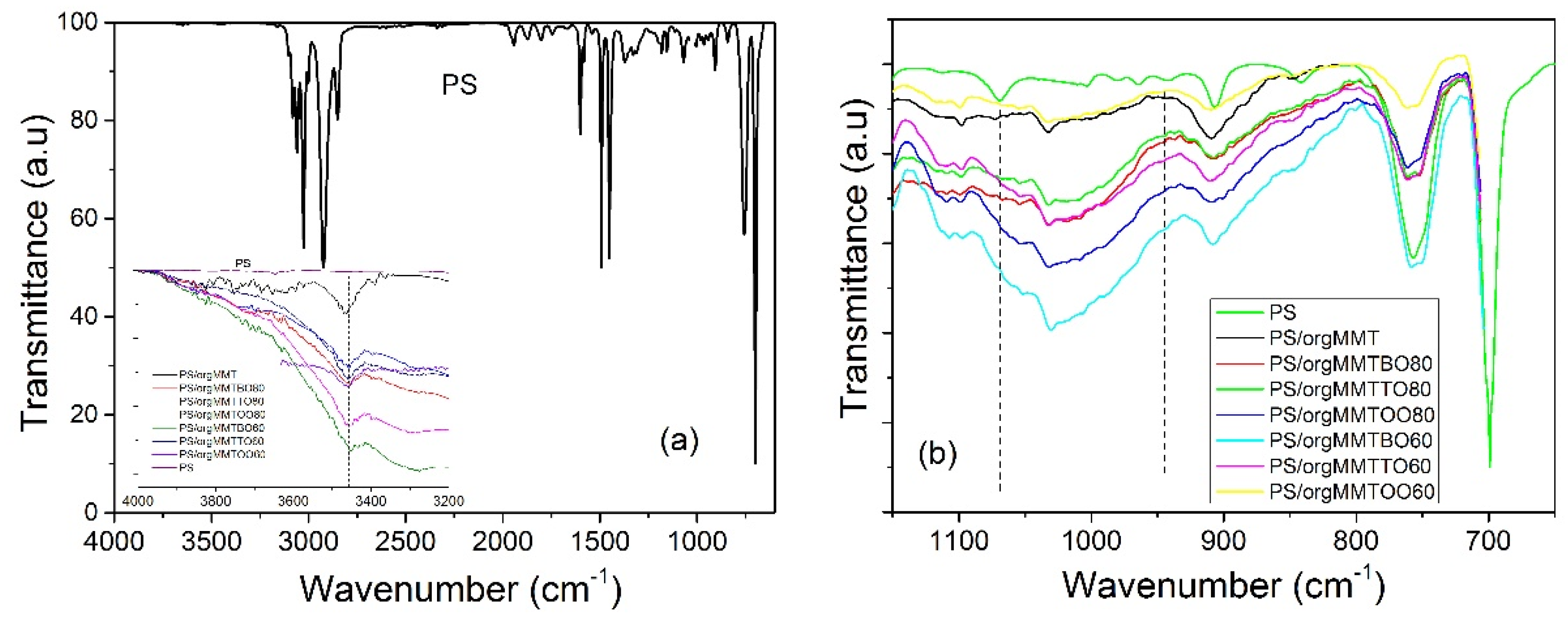
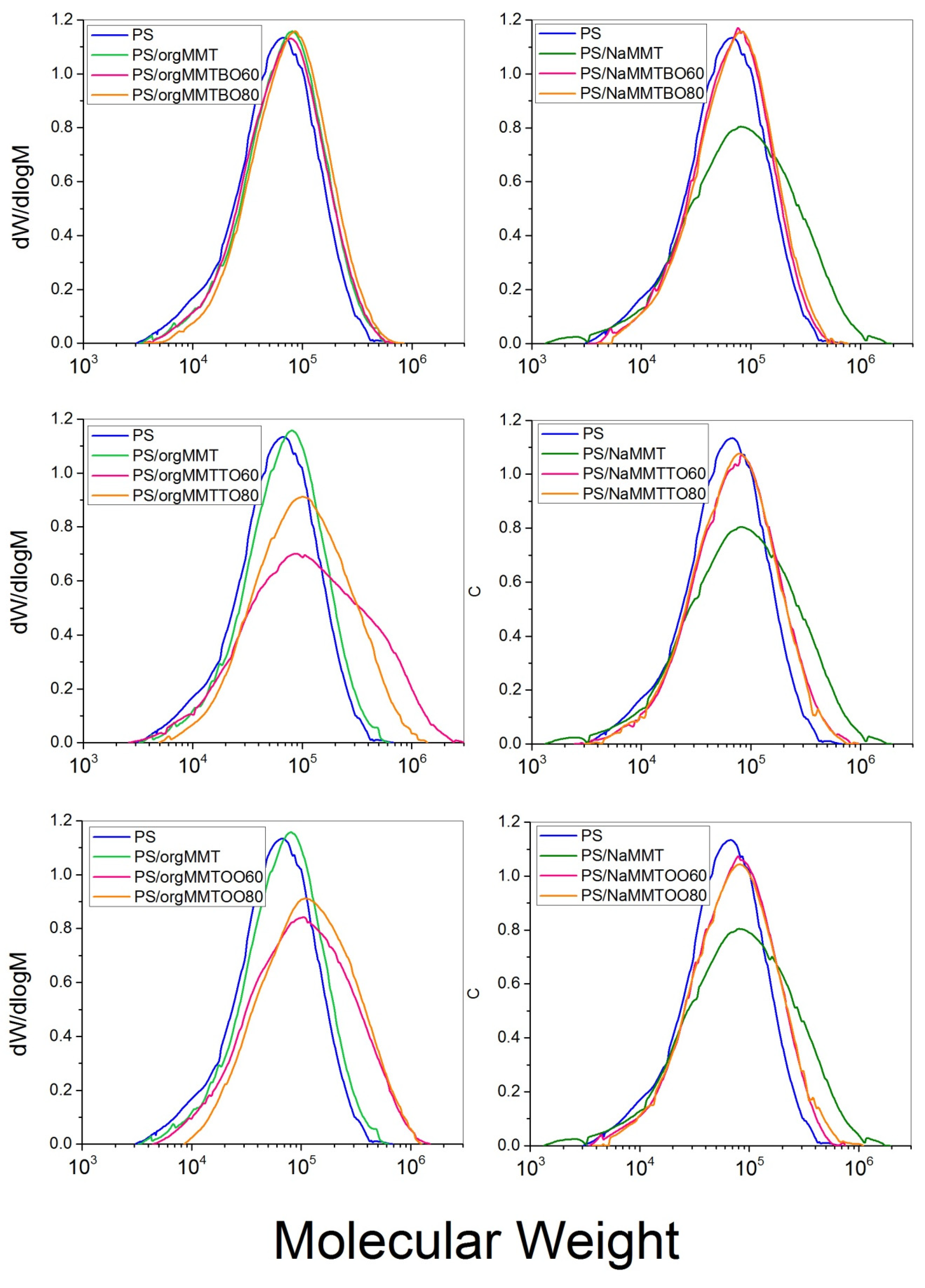
3.1. Characterization of the PS/MMTEO Nanocomposites
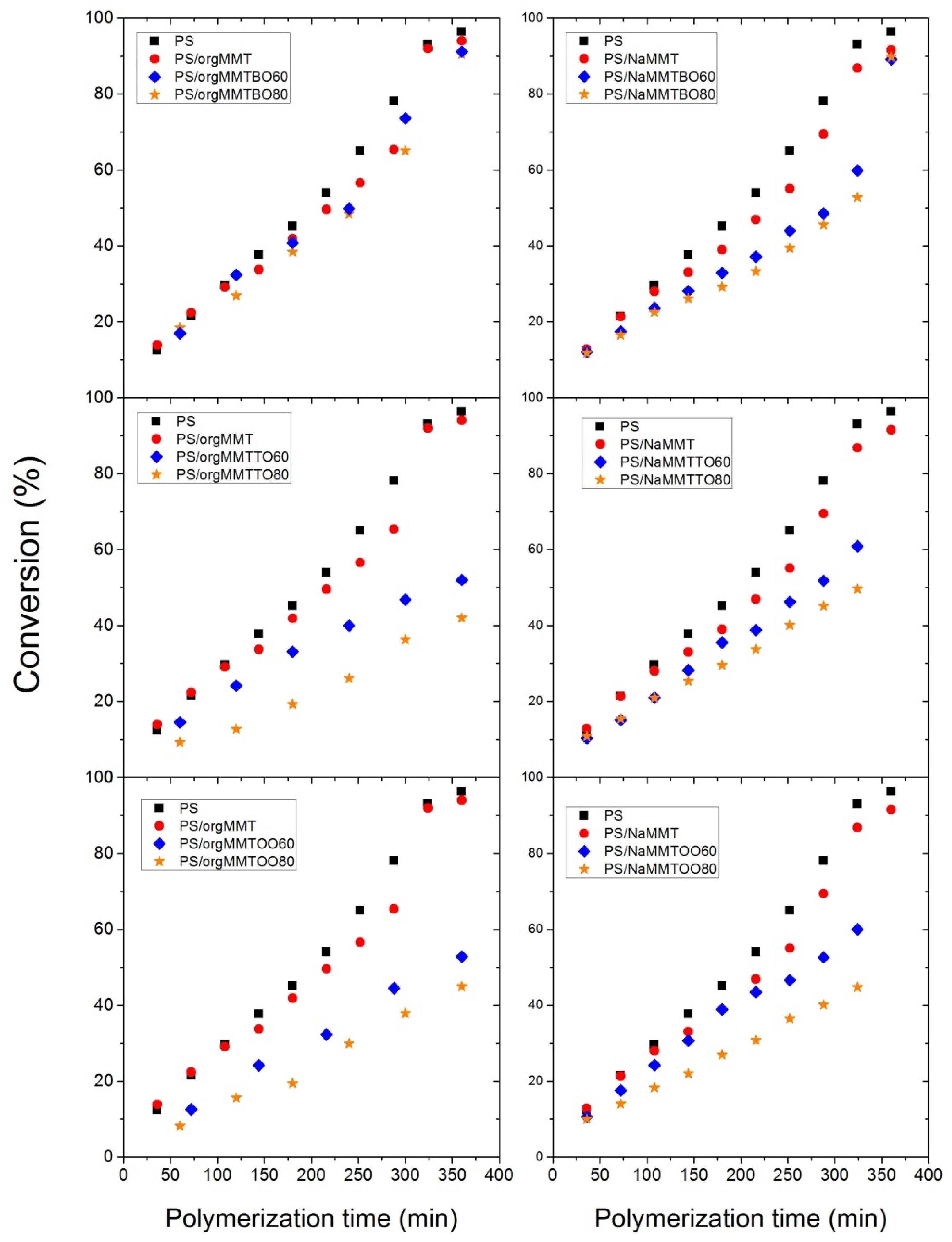
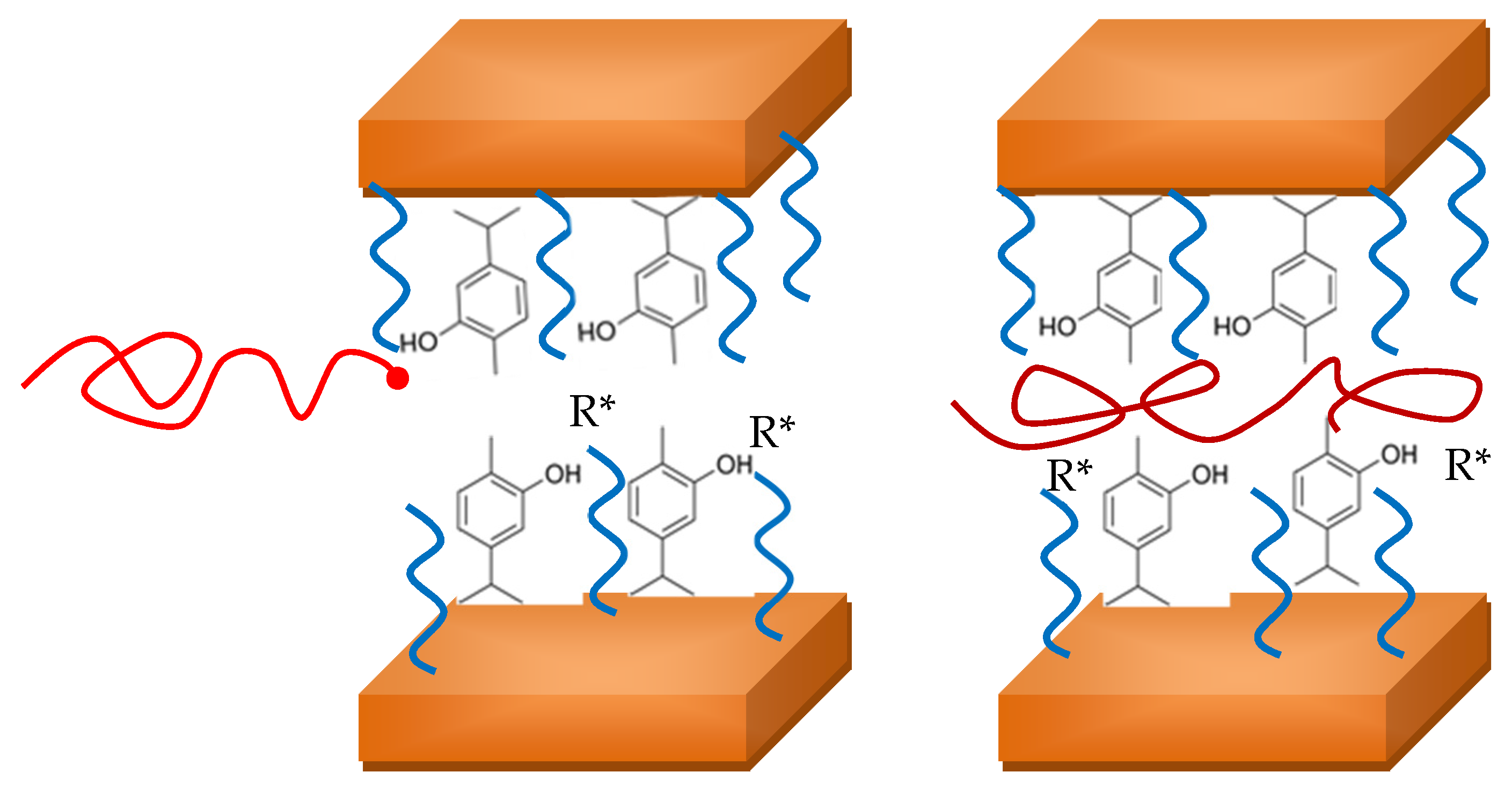
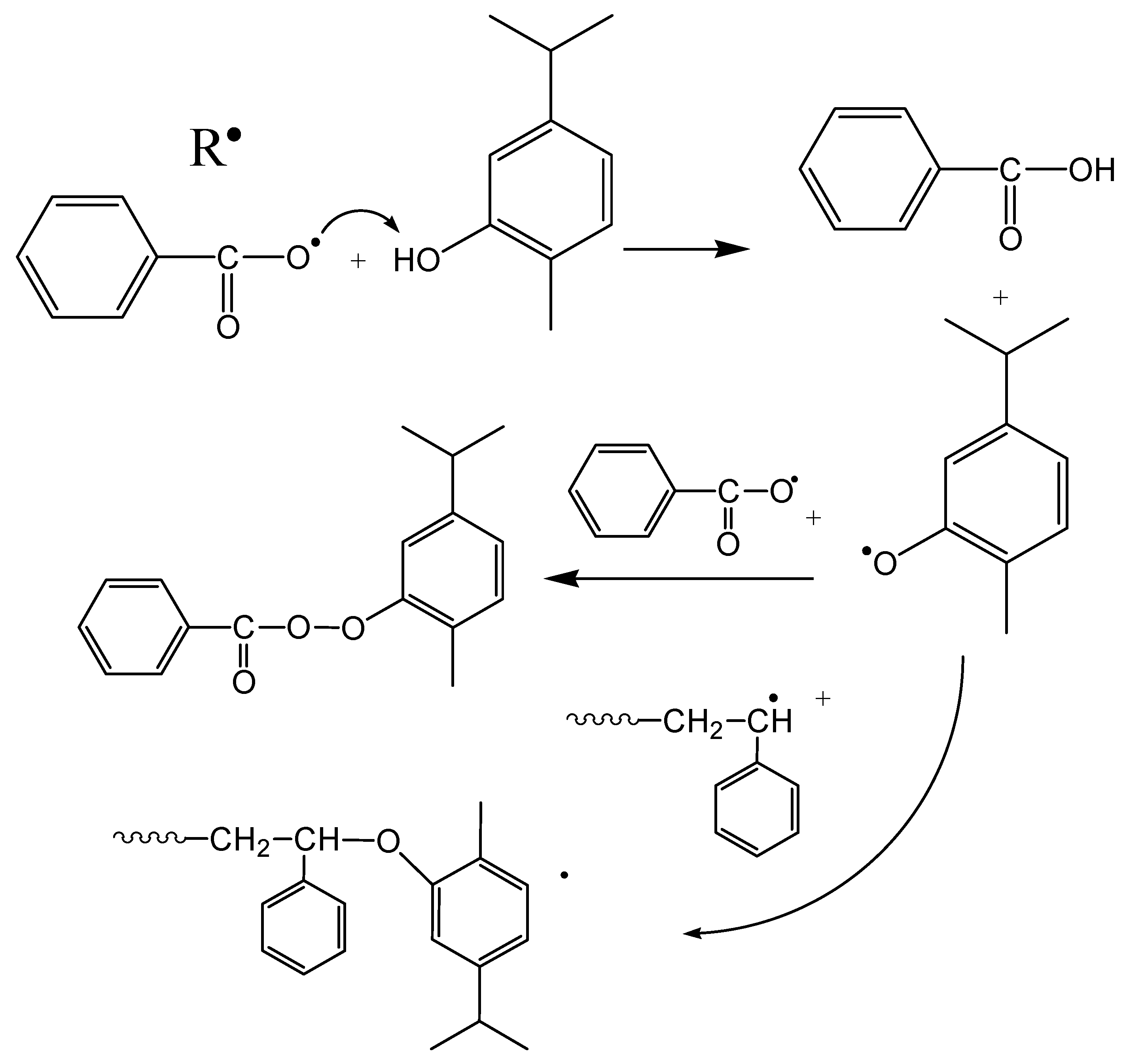
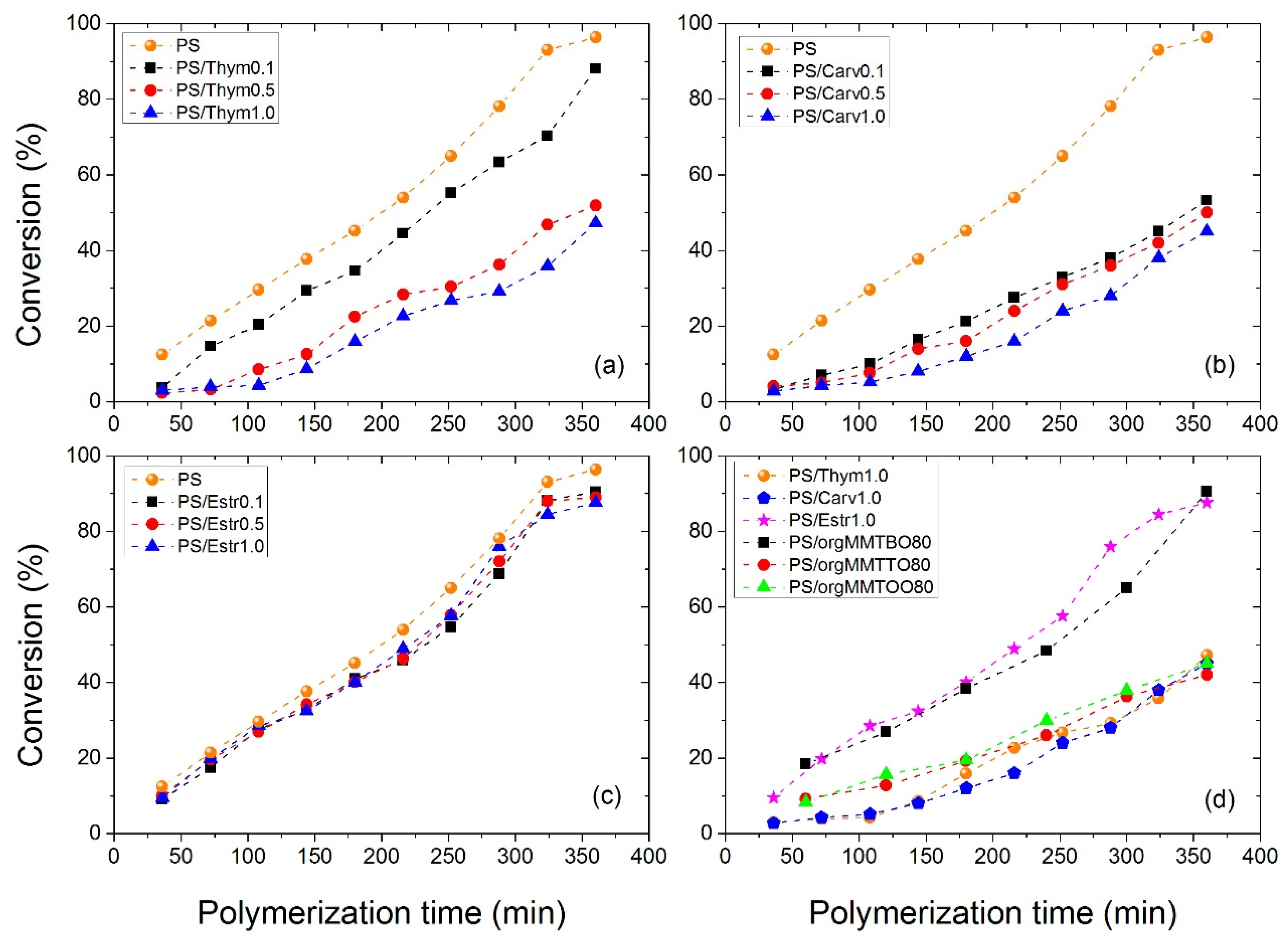
3.2. Polymerization Kinetics
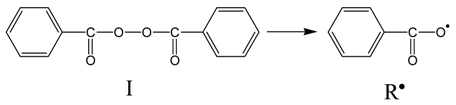
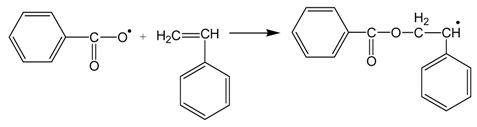
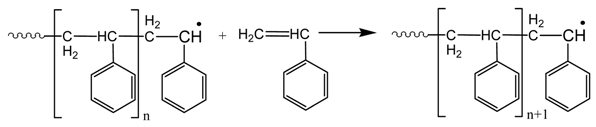
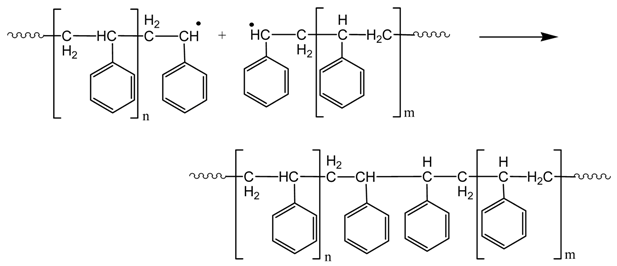
3.3. Thermal Properties of the Materials Produced
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sánchez-González, L.; Vargas, M.; González-Martínez, C.; Chiralt, A.; Cháfer, M. Use of Essential Oils in Bioactive Edible Coatings: A Review. Food Eng. Rev. 2011, 3, 1–16. [Google Scholar] [CrossRef]
- Vilarinho, F.; Vaz, M.F.; Silva, A.S. The use of montmorillonite (MMT) in food nanocomposites: Methods of incorporation, characterization of MMT/polymer nanocomposites and main consequences in the properties. Recent Pat. Food Nutr. Agric. 2020, 11, 13–26. [Google Scholar] [CrossRef] [PubMed]
- Souza, V.G.L.; Pires, J.R.A.; Rodrigues, C.; Rodrigues, P.F.; Lopes, A.; Silva, R.J.; Caldeira, J.; Duarte, M.P.; Fernandes, F.B.; Coelhoso, I.M.; et al. Physical and morphological characterization of chitosan/montmorillonite films incorporated with ginger essential oil. Coatings 2019, 9, 700. [Google Scholar] [CrossRef]
- Souza, V.G.L.; Pires, J.R.A.; Vieira, É.T.; Coelhoso, I.M.; Duarte, M.P.; Fernando, A.L. Activity of chitosan-montmorillonite bionanocomposites incorporated with rosemary essential oil: From in vitro assays to application in fresh poultry meat. Food Hydrocoll. 2019, 89, 241–252. [Google Scholar] [CrossRef]
- Micó-Vicent, B.; Viqueira, V.; Ramos, M.; Luzi, F.; Dominici, F.; Torre, L.; Jiménez, A.; Puglia, D.; Garrigós, M.C. Effect of lemon waste natural dye and essential oil loaded into laminar nanoclays on thermomechanical and color properties of polyester based bionanocomposites. Polymers 2020, 12, 1451. [Google Scholar] [CrossRef]
- Tornuk, F.; Sagdic, O.; Hancer, M.; Yetim, H. Development of LLDPE based active nanocomposite films with nanoclays impregnated with volatile compounds. Food Res. Int. 2018, 107, 337–345. [Google Scholar] [CrossRef]
- Giannakas, A.; Tsagkalias, I.; Achilias, D.S.; Ladavos, A. A novel method for the preparation of inorganic and organo-modified montmorillonite essential oil hybrids. Appl. Clay Sci. 2017, 146, 362–370. [Google Scholar] [CrossRef]
- Giannakas, A. Na-Montmorillonite vs. Organically Modified Montmorillonite as Essential Oil Nanocarriers for Melt-Extruded Low-Density Poly-Ethylene Nanocomposite Active Packaging Films with a Controllable and Long-Life Antioxidant Activity. Nanomaterials 2020, 10, 1027. [Google Scholar] [CrossRef]
- Giannakas, A.; Ladavos, A. Preparation and Characterization of Polystyrene/Organolaponite Nanocomposites. Polym. Plast. Technol. Eng. 2012, 51, 1411–1415. [Google Scholar] [CrossRef]
- Elshafie, H.S.; Camele, I. An Overview of the Biological Effects of Some Mediterranean Essential Oils on Human Health. Biomed Res. Int. 2017, 2017, 1–14. [Google Scholar] [CrossRef]
- Wińska, K.; Mączka, W.; Łyczko, J.; Grabarczyk, M.; Czubaszek, A.; Szumny, A. Essential Oils as Antimicrobial Agents—Myth or Real Alternative? Molecules 2019, 24, 2130. [Google Scholar] [CrossRef] [PubMed]
- Méndez-Vilas, A. Microbial Pathogens and Strategies for Combating Them: Science, Technology and Education; Formatex Research Center: Norristown, PA, USA, 2013; ISBN 9788493984397. [Google Scholar]
- Vokou, D.; Kokkini, S.; Bessiere, J.-M. Geographic variation of Greek oregano (Origanum vulgare ssp. hirtum) essential oils. Biochem. Syst. Ecol. 1993, 21, 287–295. [Google Scholar] [CrossRef]
- Stefanaki, A.; Cook, C.M.; Lanaras, T.; Kokkini, S. The Oregano plants of Chios Island (Greece): Essential oils of Origanum onites L. growing wild in different habitats. Ind. Crop. Prod. 2016, 82, 107–113. [Google Scholar] [CrossRef]
- Wei, H.-K.; Chen, G.; Wang, R.-J.; Peng, J. Oregano essential oil decreased susceptibility to oxidative stress-induced dysfunction of intestinal epithelial barrier in rats. J. Funct. Foods 2015, 18, 1191–1199. [Google Scholar] [CrossRef]
- Lambert, R.J.W.; Skandamis, P.N.; Coote, P.J.; Nychas, G.J.E. A study of the minimum inhibitory concentration and mode of action of oregano essential oil, thymol and carvacrol. J. Appl. Microbiol. 2001, 91, 453–462. [Google Scholar] [CrossRef]
- Teixeira, B.; Marques, A.; Ramos, C.; Serrano, C.; Matos, O.; Neng, N.R.; Nogueira, J.M.; Saraiva, J.A.; Nunes, M.L. Chemical composition and bioactivity of different oregano (Origanum vulgare) extracts and essential oil. J. Sci. Food Agric. 2013, 93, 2707–2714. [Google Scholar] [CrossRef]
- Perdones, A.; Chiralt, A.; Vargas, M. Properties of film-forming dispersions and films based on chitosan containing basil or thyme essential oil. Food Hydrocoll. 2016, 57, 271–279. [Google Scholar] [CrossRef]
- Borugă, O.; Jianu, C.; Mişcă, C.; Goleţ, I.; Gruia, A.T.; Horhat, F.G. Thymus vulgaris essential oil: Chemical composition and antimicrobial activity. J. Med. Life 2014, 7, 56–60. [Google Scholar]
- Bozin, B.; Mimica-Dukic, N.; Simin, N.; Anackov, G. Characterization of the volatile composition of essential oils of some lamiaceae spices and the antimicrobial and antioxidant activities of the entire oils. J. Agric. Food Chem. 2006, 54, 1822–1828. [Google Scholar] [CrossRef]
- Politeo, O.; Jukic, M.; Milos, M. Chemical composition and antioxidant capacity of free volatile aglycones from basil (Ocimum basilicum L.) compared with its essential oil. Food Chem. 2007, 101, 379–385. [Google Scholar] [CrossRef]
- Opalchenova, G.; Obreshkova, D. Comparative studies on the activity of basil—An essential oil from Ocimum basilicum L.-against multidrug resistant clinical isolates of the genera Staphylococcus, Enterococcus and Pseudomonas by using different test methods. J. Microbiol. Methods 2003, 54, 105–110. [Google Scholar] [CrossRef]
- Hussain, A.I.; Anwar, F.; Sherazi, S.T.H.; Przybylski, R. Chemical composition, antioxidant and antimicrobial activities of basil (Ocimum basilicum) essential oils depends on seasonal variations. Food Chem. 2008, 108, 986–995. [Google Scholar] [CrossRef] [PubMed]
- Su, H.J.; Chao, C.J.; Chang, H.Y.; Wu, P.C. The effects of evaporating essential oils on indoor air quality. Atmos. Environ. 2007, 41, 1230–1236. [Google Scholar] [CrossRef]
- Campos-Requena, V.H.; Rivas, B.L.; Pérez, M.A.; Garrido-Miranda, K.A.; Pereira, E.D. Polymer/clay nanocomposite films as active packaging material: Modeling of antimicrobial release. Eur. Polym. J. 2015, 71, 461–475. [Google Scholar] [CrossRef]
- Giannakas, A.E.; Leontiou, A.A. Montmorillonite Composite Materials and Food Packaging. In Composites Materials for Food Packaging; John Wiley & Sons: Hoboken, NJ, USA, 2018; pp. 1–71. ISBN 978-1-119-16024-3. [Google Scholar]
- Kinninmonth, M.A.; Liauw, C.M.; Verran, J.; Taylor, R.; Edwards-Jones, V.; Shaw, D.; Webb, M. Investigation into the suitability of layered silicates as adsorption media for essential oils using FTIR and GC-MS. Appl. Clay Sci. 2013, 83–84, 415–425. [Google Scholar] [CrossRef]
- El Miz, M.; Salhi, S.; El Bachiri, A.; Wathelet, J.; Tahani, A. Adsorption of essential oil components of Lavandula angustifolia on sodium modified bentonite from Nador (North-East Morocco). Afr. J. Biotechnol. 2015, 13, 3413–3425. [Google Scholar]
- El Miz, M.; Salhi, S.; Chraibi, I.; El Bachiri, A.; Fauconnier, M.-L.; Tahani, A. Characterization and adsorption study of thymol on pillared bentonite. Open J. Phys. Chem. 2014, 4, 98–116. [Google Scholar] [CrossRef]
- Nguemtchouin, M.G.; Ngassoum, M.B.; Chalier, P.; Kamga, R.; Ngamo, L.S.; Cretin, M. Ocimum gratissimum essential oil and modified montmorillonite clay, a means of controlling insect pests in stored products. J. Stored Prod. Res. 2013, 52, 57–62. [Google Scholar] [CrossRef]
- Alexandre, M.; Dubois, P. Polymer-layered silicate nanocomposites: Preparation, properties and used of a new class of materials. Mater. Sci. Eng. R Rep. 2000, 28, 1–63. [Google Scholar] [CrossRef]
- Tiwari, R.R.; Natarajan, U. Thermal and mechanical properties of melt processed intercalated poly(methyl methacrylate)–organoclay nanocomposites over a wide range of filler loading. Polym. Int. 2008, 57, 738–743. [Google Scholar] [CrossRef]
- Kumar, S.; Jog, J.P.; Natarajan, U. Preparation and characterization of poly(methyl methacrylate)–clay nanocomposites via melt intercalation: The effect of organoclay on the structure and thermal properties. J. Appl. Polym. Sci. 2003, 89, 1186–1194. [Google Scholar] [CrossRef]
- Dhibar, A.K.; Mallick, S.; Rath, T.; Khatua, B.B. Effect of clay platelet dispersion as affected by the manufacturing techniques on thermal and mechanical properties of PMMA-clay nanocomposites. J. Appl. Polym. Sci. 2009, 113, 3012–3018. [Google Scholar] [CrossRef]
- Nikolaidis, A.K.; Achilias, D.S.; Karayannidis, G.P. Synthesis and characterization of PMMA/organomodified montmorillonite nanocomposites prepared by in situ bulk polymerization. Ind. Eng. Chem. Res. 2011, 50, 571–579. [Google Scholar] [CrossRef]
- Achilias, D.S.; Siafaka, P.; Nikolaidis, A.K. Polymerization Kinetics and Thermal Properties of Poly(alkyl methacrylate)/Organomodified Montmorillonite Nanocomposites. Polym. Int. 2012, 61, 1510–1518. [Google Scholar] [CrossRef]
- Verros, G.D.; Achilias, D.S. Towards the development of a mathematical model for the bulk in situ radical polymerization of methyl methacrylate in the presence of nano-additives. Can. J. Chem. Eng. 2016, 94, 1783–1791. [Google Scholar] [CrossRef]
- Siddiqui, M.N.; Redhwi, H.H.; Vakalopoulou, E.; Tsagkalias, I.; Ioannidou, M.D.; Achilias, D.S. Synthesis, characterization and reaction kinetics of PMMA/silver nanocomposites prepared via in situ radical polymerization. Eur. Polym. J. 2015, 72, 256–269. [Google Scholar] [CrossRef]
- Tsagkalias, I.S.; Vlachou, A.; Verros, G.D.; Achilias, D.S. Effect of Graphene oxide or Functionalized Graphene Oxide on the Copolymerization Kinetics of Styrene/n-butyl Methacrylate. Polymers 2019, 11, 999. [Google Scholar] [CrossRef] [PubMed]
- Tsagkalias, I.; Proskynitopoulou, V.; Verros, G.D.; Achilias, D.S. Effect of graphene oxide on the kinetics of the radical polymerization of styrene. Materials today Proc. 2018, 5, 27517–27525. [Google Scholar] [CrossRef]
- Majoni, S. Thermal and flammability study of polystyrene composites containing magnesium–aluminum layered double hydroxide (MgAl–C16 LDH), and an organophosphate. J. Therm Anal. Calorim 2015, 120, 1435–1443. [Google Scholar] [CrossRef]
- Bera, O.; Pilic, B.; Pavlicevic, J.; Jovicic, M.; Holló, B.; Mészáros Szécsényi, K.; Spirkova, M. Preparation and thermal properties of polystyrene/silica nanocomposites. Thermochim. Acta 2011, 515, 1–5. [Google Scholar] [CrossRef]
- Achilias, D.S. A review of modeling of diffusion controlled polymerization reactions. Macromol. Theory Simul. 2007, 16, 319–347. [Google Scholar] [CrossRef]
- Siddiqui, M.N.; Redhwi, H.H.; Verros, G.D.; Achilias, D.S. Evaluating the role of nanomontmorillonite in bulk in situ radical polymerization kinetics of butyl methacrylate through a simulation model. Ind. Eng. Chem. Res. 2014, 53, 11303–11311. [Google Scholar] [CrossRef]
- Michailidis, M.; Verros, G.D.; Deliyanni, E.; Andriotis, E.G.; Achilias, D.S. An experimental and theoretical study of butyl methacrylate in situ radical polymerization kinetics in the presence of graphene oxide nanoadditive. J. Polym. Sci. Part A Polym. Chem. 2017, 55, 1433–1441. [Google Scholar] [CrossRef]
- Blanco, I.; Cicala, G.; Tosto, C.; Bottino, F.A. Kinetic Study of the Thermal and Thermo-Oxidative Degradations of Polystyrene Reinforced with Multiple-Cages POSS. Polymers 2020, 12, 2742. [Google Scholar] [CrossRef] [PubMed]
- Peterson, J.D.; Vyazovkin, S.; Wight, C.A. Kinetics of the Thermal and Thermo-Oxidative Degradation of Polystyrene, Polyethylene and Poly(propylene). Macromol. Chem. Phys. 2001, 202, 775–784. [Google Scholar] [CrossRef]
- Suresh, K.; Kumar, R.V.; Kumar, M.; Jeyapriya, M.; Anbarasan, R.; Pugazhenthi, G. Sonication-assisted synthesis of polystyrene (PS)/organoclay nanocomposites: Influence of clay content. Appl. Nanosci. 2017, 7, 215–223. [Google Scholar] [CrossRef]
- Chen, K.; Wilkie, C.A.; Vyazovkin, S. Nanoconfinement revealed in degradation and relaxation studies of two structurally different polystyrene–clay systems. J. Phys. Chem. B 2007, 111, 12685–12692. [Google Scholar] [CrossRef]
- Vyazovkin, S.; Dranca, I.; Fan, X.; Advincula, R. Degradation and relaxation kinetics of polystyrene-clay nanocomposites prepared by surface-initiated polymerization. J. Phys. Chem. B 2004, 108, 11672–11679. [Google Scholar] [CrossRef]
- Wang, Y.; Liao, X.; Li, S.; Luo, Y.; Yang, Q.; Li, G. Poly(methyl methacrylate nanocomposites based on graphene oxide: A comparative investigation of the effects of surface chemistry on properties and foaming behavior. Polym. Int. 2016, 65, 1195–1203. [Google Scholar] [CrossRef]


| Sample | NaMMT/orgMMT (% Relative to Monomer) | Nominal Composition of EO to Clays (%) | ||
|---|---|---|---|---|
| Basil | Thyme | Oregano | ||
| PS | - | - | - | - |
| PS/orgMMT | 3 | - | - | - |
| PS/orgMMTBO60 | 3 | 60 | - | - |
| PS/orgMMTTO60 | 3 | - | 60 | - |
| PS/orgMMTOO60 | 3 | - | - | 60 |
| PS/orgMMTBO80 | 3 | 80 | - | - |
| PS/orgMMTTO80 | 3 | - | 80 | - |
| PS/orgMMTOO80 | 3 | - | - | 80 |
| PS/NaMMT | 3 | - | - | - |
| PS/NaMMTBO60 | 3 | 60 | - | - |
| PS/NaMMTTO60 | 3 | - | 60 | - |
| PS/NaMMTOO60 | 3 | - | - | 60 |
| PS/NaMMTBO80 | 3 | 80 | - | - |
| PS/NaMMTTO80 | 3 | - | 80 | - |
| PS/NaMMTOO80 | 3 | - | - | 80 |
| Sample | PD | Tg (°C) | ||
|---|---|---|---|---|
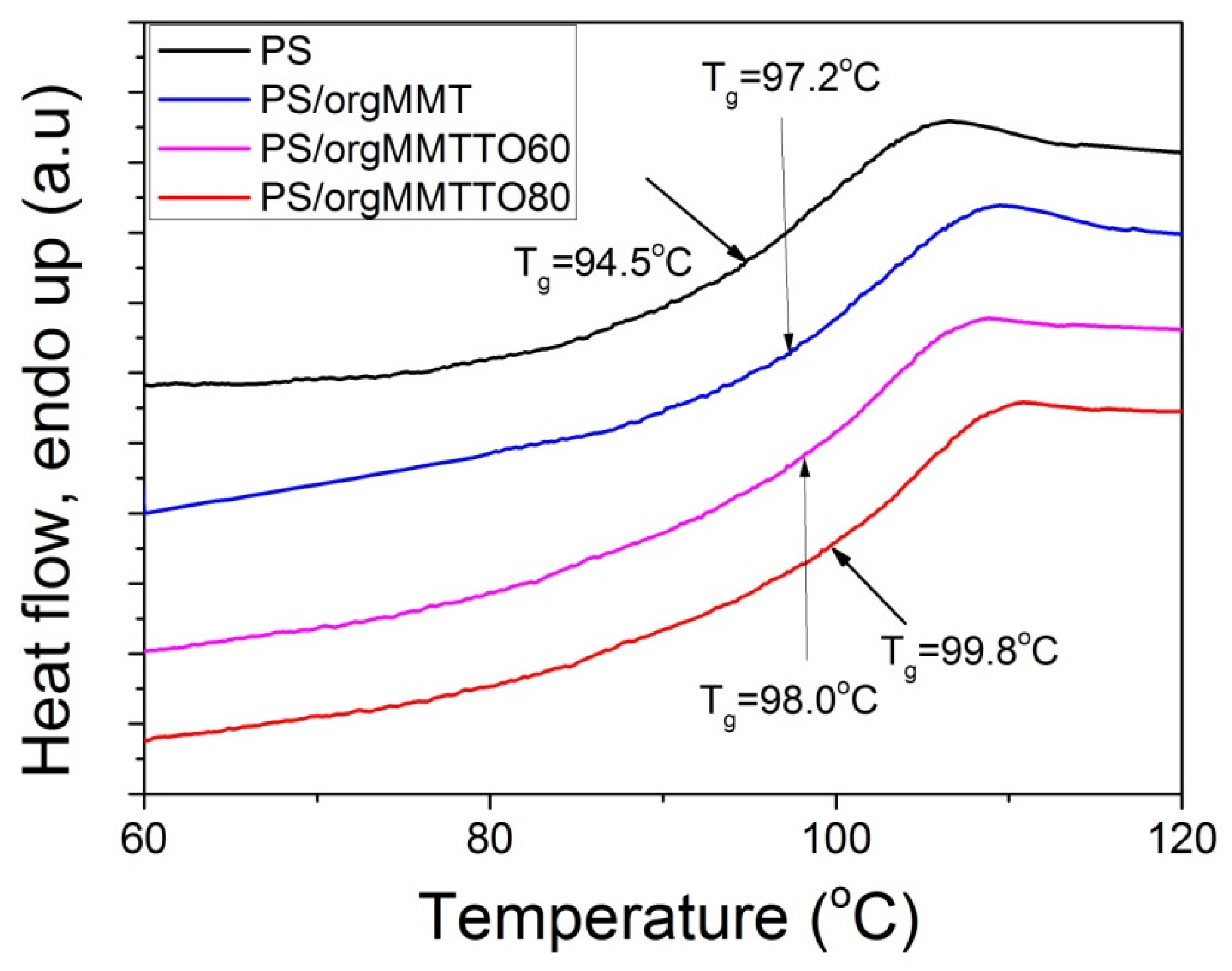
| PS | 37,120 | 77,590 | 2.09 | 94.5 |
| PS/orgMMT | 44,680 | 90,300 | 2.02 | 97.2 |
| PS/orgMMTBO60 | 45,310 | 90,460 | 2.00 | 97.6 |
| PS/orgMMTTO60 | 52,200 | 222,610 | 4.26 | 98.0 |
| PS/orgMMTOO60 | 56,870 | 159,830 | 2.81 | 97.2 |
| PS/orgMMTBO80 | 50,010 | 100,520 | 2.01 | 99.7 |
| PS/orgMMTTO80 | 61,380 | 152,420 | 2.48 | 99.8 |
| PS/orgMMTOO80 | 73,860 | 173,160 | 2.34 | 98.5 |
| PS/NaMMT | 38,610 | 140,120 | 3.63 | 96.0 |
| PS/NaMMTBO60 | 44,070 | 86,450 | 1.96 | 97.1 |
| PS/NaMMTTO60 | 45,750 | 100,440 | 2.20 | 97.4 |
| PS/NaMMTOO60 | 44,960 | 96,390 | 2.14 | 96.8 |
| PS/NaMMTBO80 | 47,510 | 91,830 | 1.93 | 99.3 |
| PS/NaMMTTO80 | 45,900 | 97,260 | 2.12 | 98.9 |
| PS/NaMMTOO80 | 47,810 | 102,900 | 2.15 | 98.2 |
| Sample | PD | ||
|---|---|---|---|
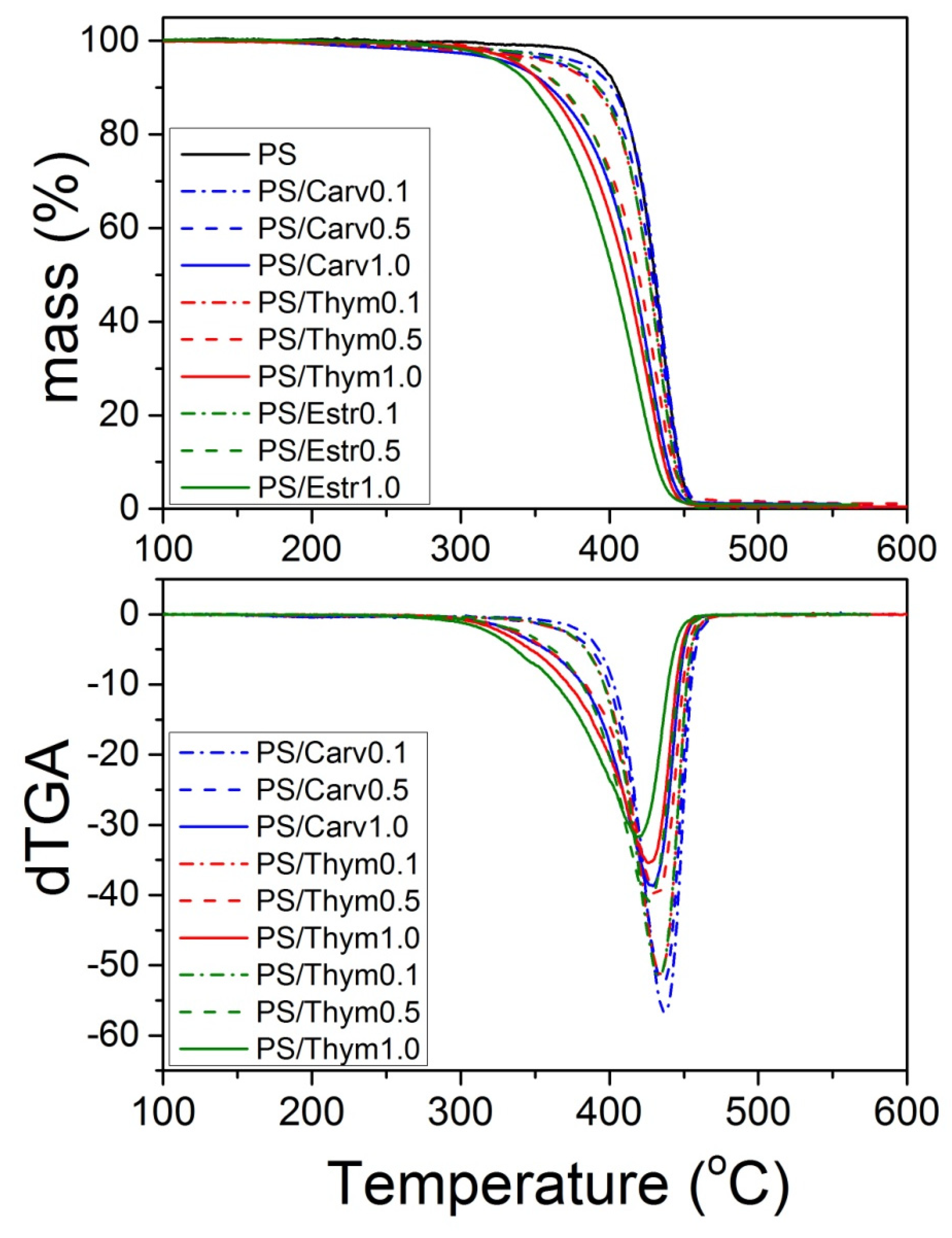
| PS | 37,120 | 77,590 | 2.09 |
| PS/Thym0.1 | 38,240 | 81,070 | 2.12 |
| PS/Thym0.5 | 44,880 | 101,430 | 2.26 |
| PS/Thym1.0 | 54,770 | 154,990 | 2.83 |
| PS/Carv0.1 | 38,270 | 83,420 | 2.18 |
| PS/Carv0.5 | 46,830 | 101,610 | 2.17 |
| PS/Carv1.0 | 51,930 | 144,360 | 2.78 |
| PS/Estr0.1 | 37,170 | 80,290 | 2.16 |
| PS/Estr0.5 | 36,800 | 79,500 | 2.16 |
| PS/Estr1.0 | 38,390 | 81,400 | 2.12 |
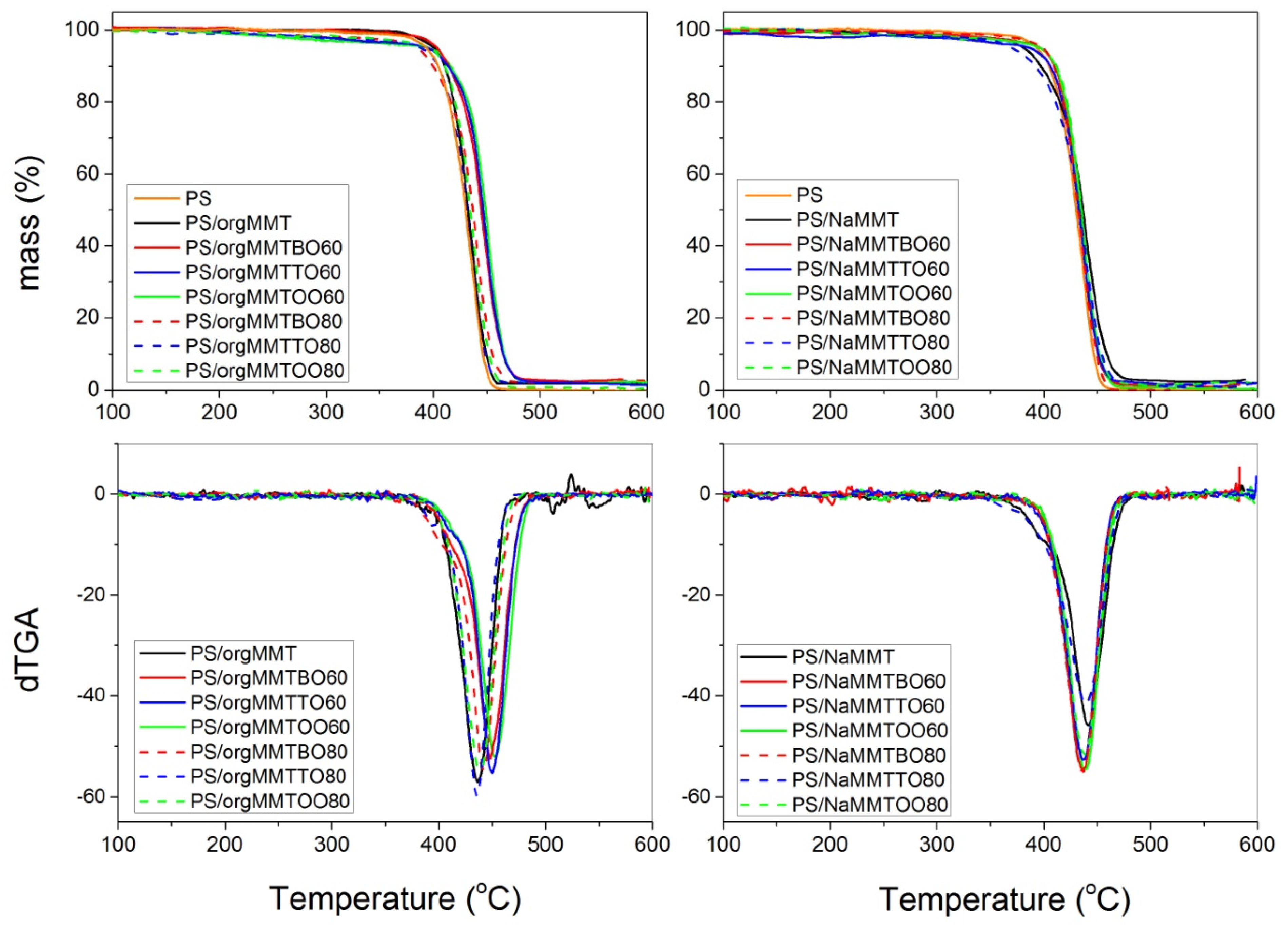
| Sample | T4% (°C) | T20% (°C) | Tmax (°C) | Residual at 600 °C |
|---|---|---|---|---|
| PS | 390 | 415 | 435 | 0.0 |
| PS/orgMMT | 397 | 419 | 437 | 1.8 |
| PS/orgMMTBO60 | 400 | 428 | 448 | 2.3 |
| PS/orgMMTTO60 | 383 | 430 | 450 | 2.0 |
| PS/orgMMTOO60 | 387 | 432 | 452 | 2.2 |
| PS/orgMMTBO80 | 379 | 417 | 442 | 2.1 |
| PS/orgMMTTO80 | 380 | 416 | 437 | 2.0 |
| PS/orgMMTOO80 | 370 | 420 | 438 | 1.8 |
| PS/NaMMT | 390 | 415 | 441 | 2.3 |
| PS/NaMMTBO60 | 393 | 419 | 437 | 1.8 |
| PS/NaMMTTO60 | 375 | 417 | 437 | 2.0 |
| PS/NaMMTOO60 | 388 | 420 | 438 | 2.0 |
| PS/NaMMTBO80 | 390 | 418 | 437 | 1.5 |
| PS/NaMMTTO80 | 365 | 412 | 439 | 1.6 |
| PS/NaMMTOO80 | 388 | 421 | 437 | 1.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsagkalias, I.S.; Loukidi, A.; Chatzimichailidou, S.; Salmas, C.E.; Giannakas, A.E.; Achilias, D.S. Effect of Na- and Organo-Modified Montmorillonite/Essential Oil Nanohybrids on the Kinetics of the In Situ Radical Polymerization of Styrene. Nanomaterials 2021, 11, 474. https://doi.org/10.3390/nano11020474
Tsagkalias IS, Loukidi A, Chatzimichailidou S, Salmas CE, Giannakas AE, Achilias DS. Effect of Na- and Organo-Modified Montmorillonite/Essential Oil Nanohybrids on the Kinetics of the In Situ Radical Polymerization of Styrene. Nanomaterials. 2021; 11(2):474. https://doi.org/10.3390/nano11020474
Chicago/Turabian StyleTsagkalias, Ioannis S., Alexandra Loukidi, Stella Chatzimichailidou, Constantinos E. Salmas, Aris E. Giannakas, and Dimitris S. Achilias. 2021. "Effect of Na- and Organo-Modified Montmorillonite/Essential Oil Nanohybrids on the Kinetics of the In Situ Radical Polymerization of Styrene" Nanomaterials 11, no. 2: 474. https://doi.org/10.3390/nano11020474
APA StyleTsagkalias, I. S., Loukidi, A., Chatzimichailidou, S., Salmas, C. E., Giannakas, A. E., & Achilias, D. S. (2021). Effect of Na- and Organo-Modified Montmorillonite/Essential Oil Nanohybrids on the Kinetics of the In Situ Radical Polymerization of Styrene. Nanomaterials, 11(2), 474. https://doi.org/10.3390/nano11020474










