Synthesis of D-Limonene Loaded Polymeric Nanoparticles with Enhanced Antimicrobial Properties for Potential Application in Food Packaging
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
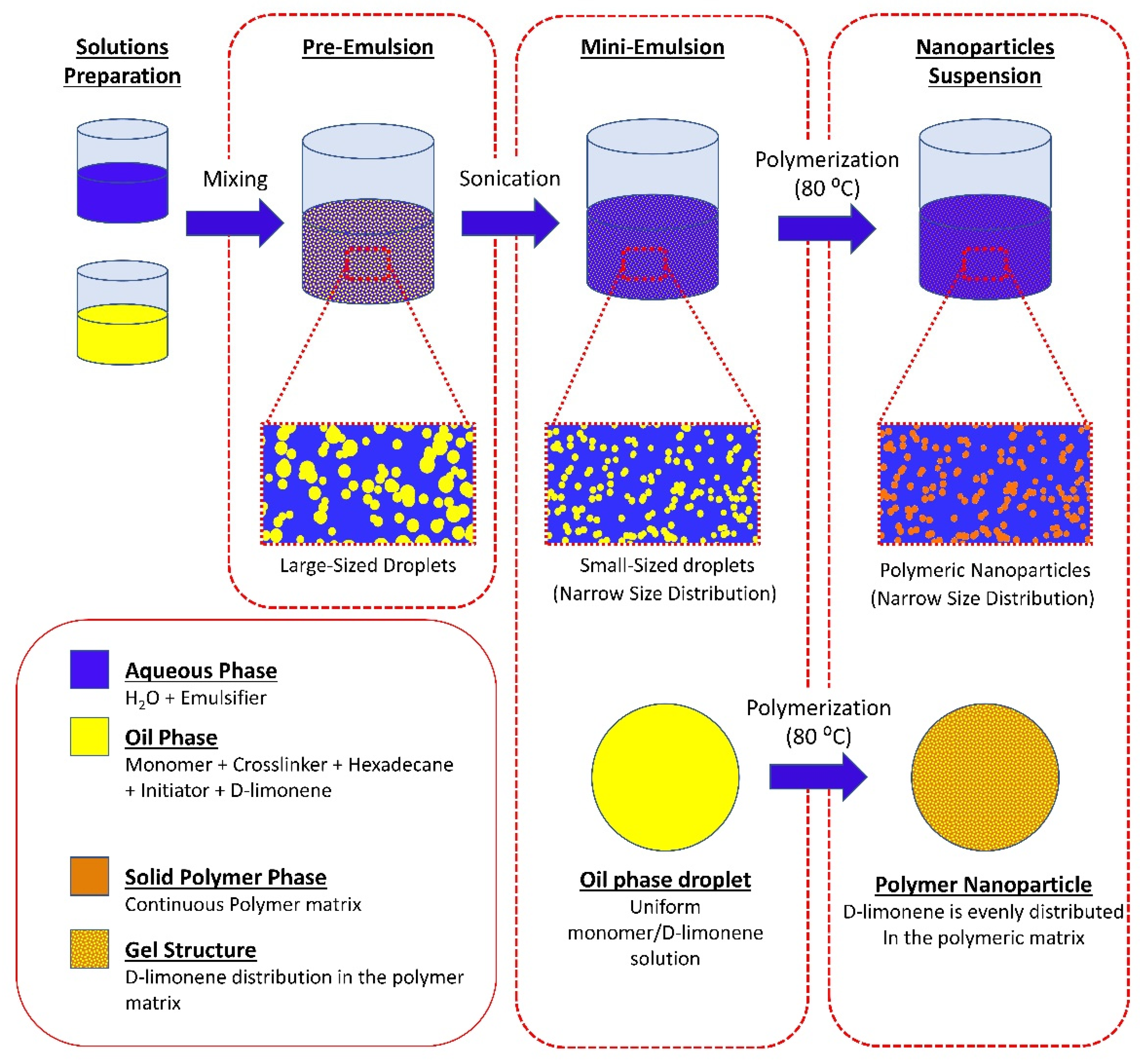
2.2. Synthesis of Polymeric Nanoparticles
2.3. Design of Experiments (DOE)
2.4. Thermogravimetric Analysis (TGA)
2.5. Pyrolysis/Gas Chromatography-Mass Spectrometry (Py/GC-MS)
2.6. Particles Size Distribution
2.7. Scanning Electron Microscopy (SEM)
2.8. Determination of Antimicrobial Properties
2.9. Mathematical Modeling
2.10. Statistical Analysis
3. Results
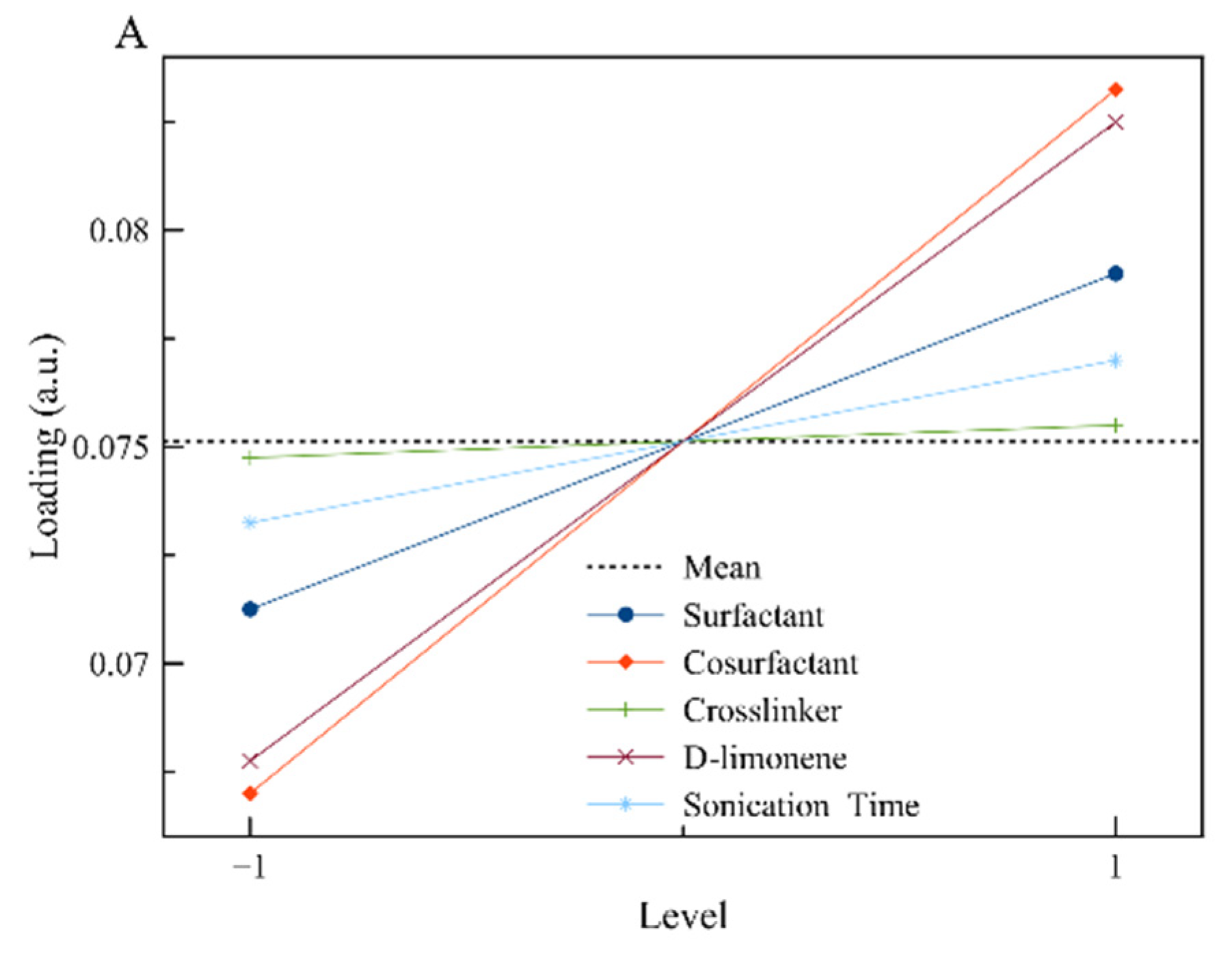
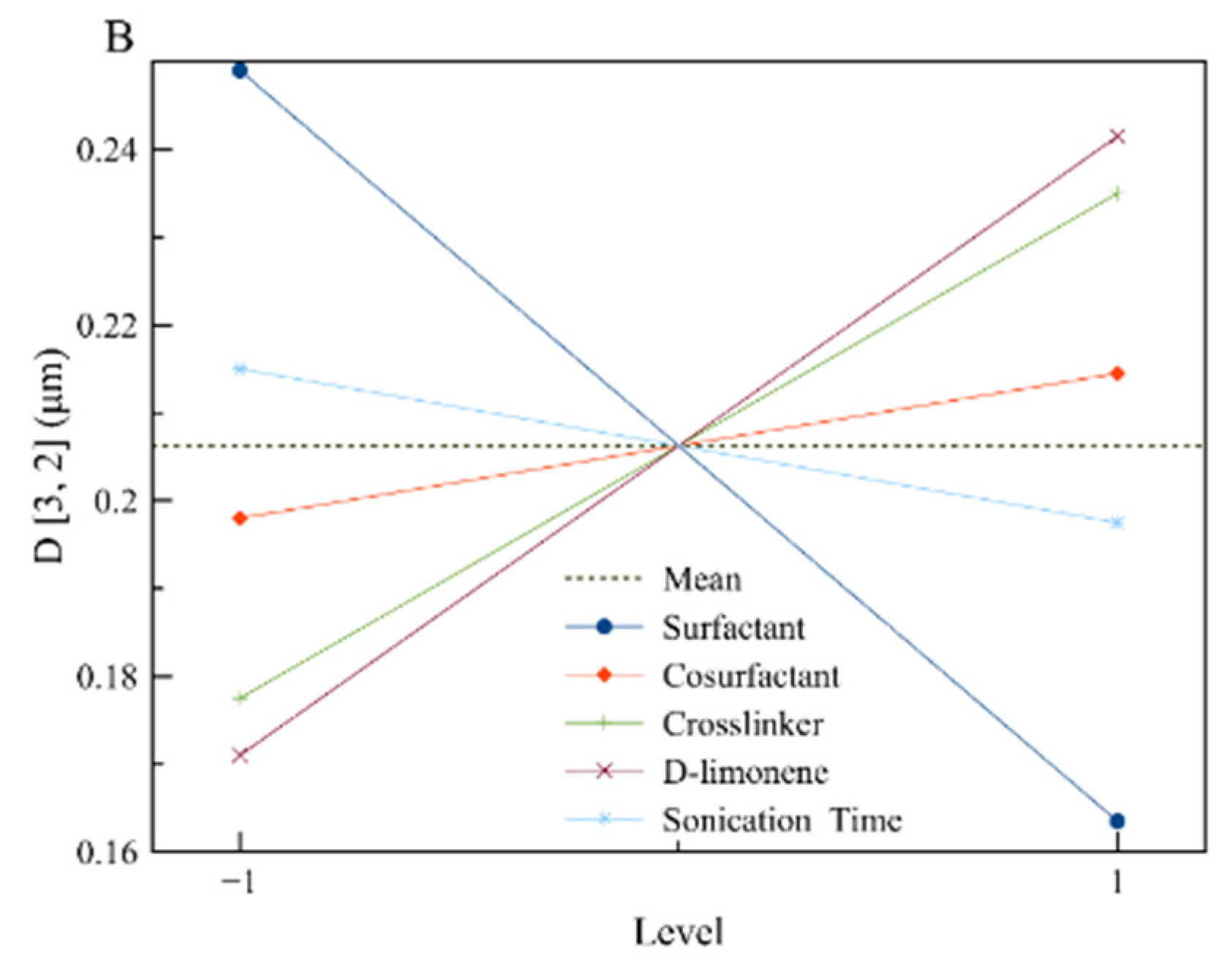
3.1. Design of Experiments
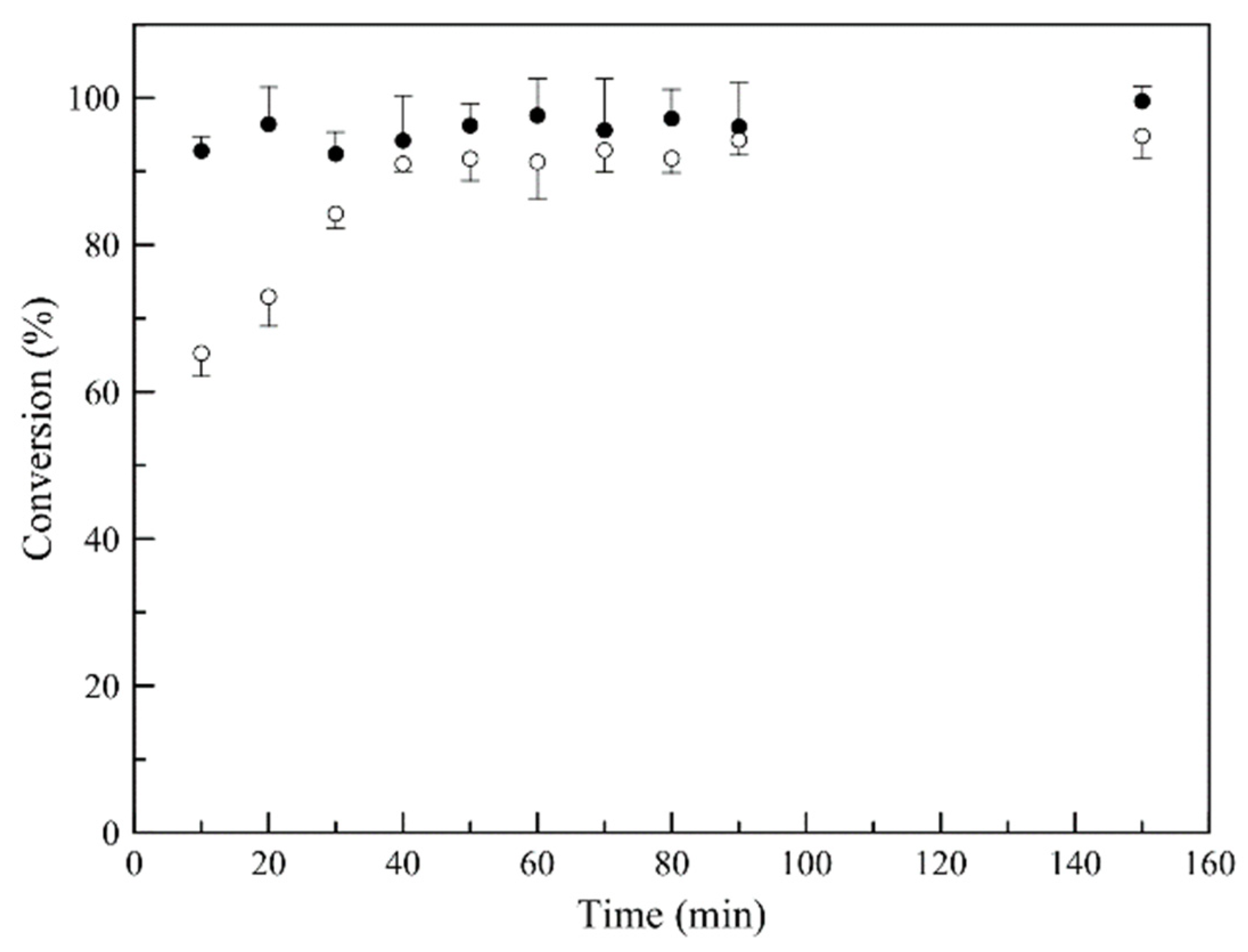
3.2. Synthesis of Latex Particles
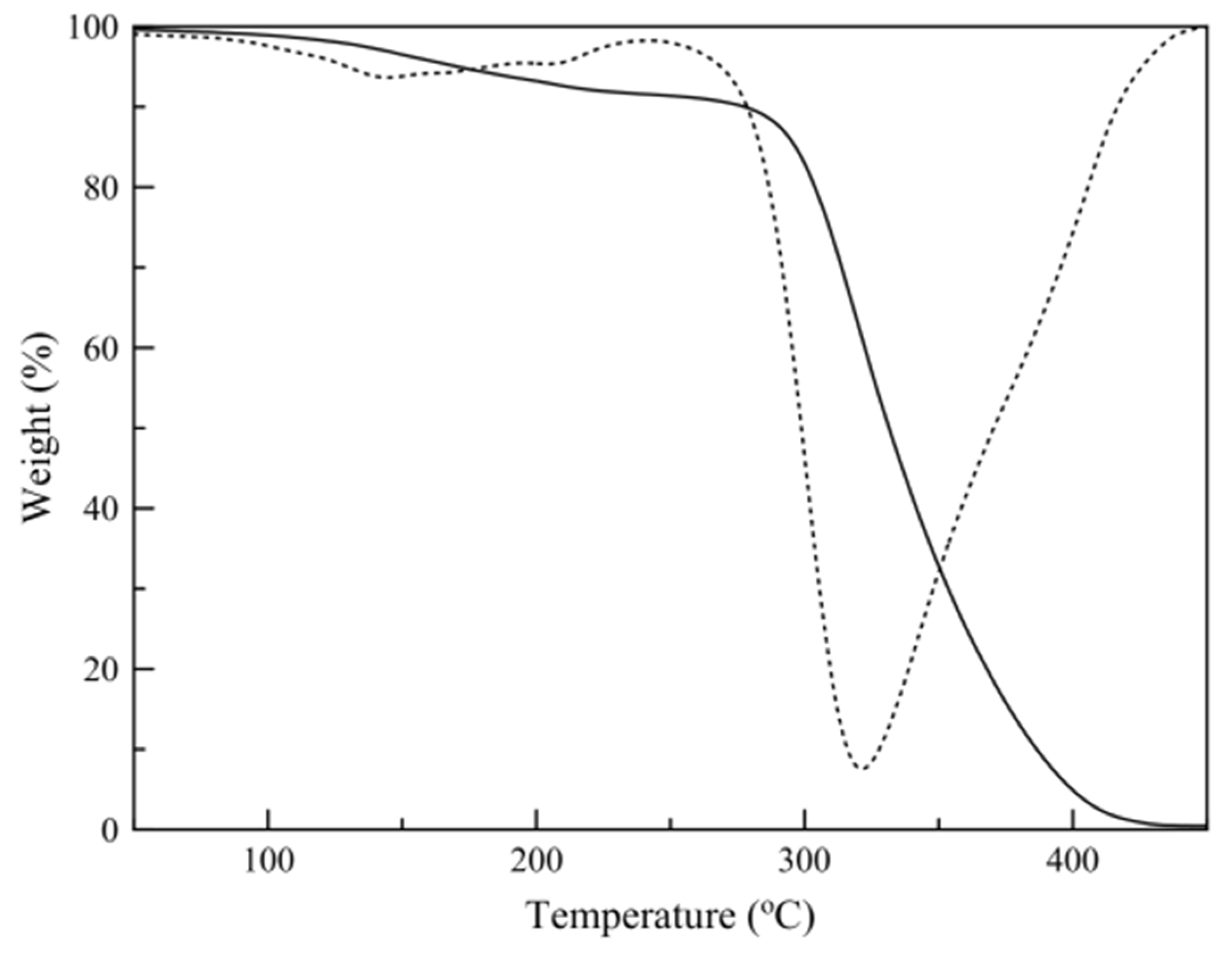
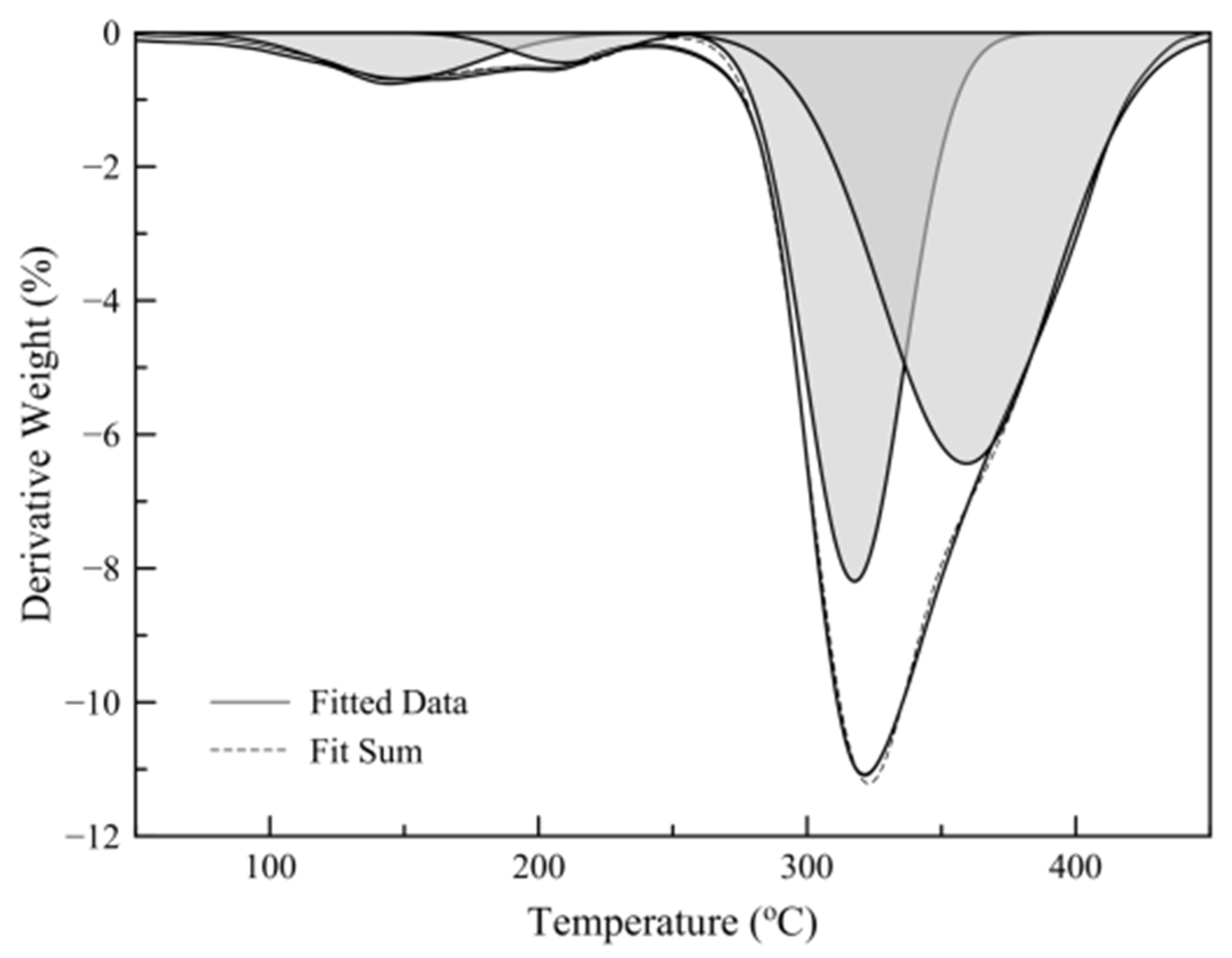
3.3. Thermogravimetric Analysis (TGA)
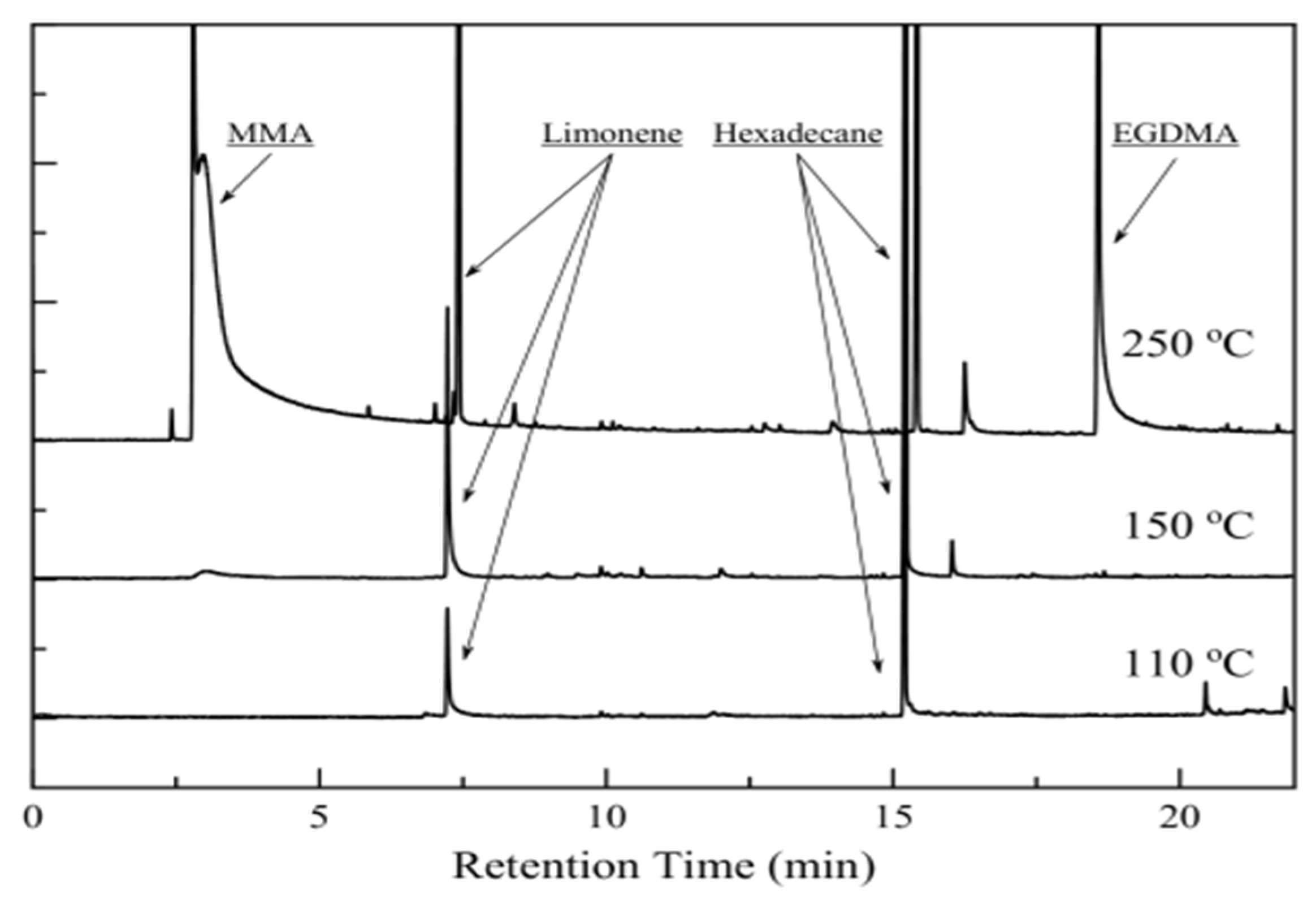
3.4. Py/GC-MS Analysis
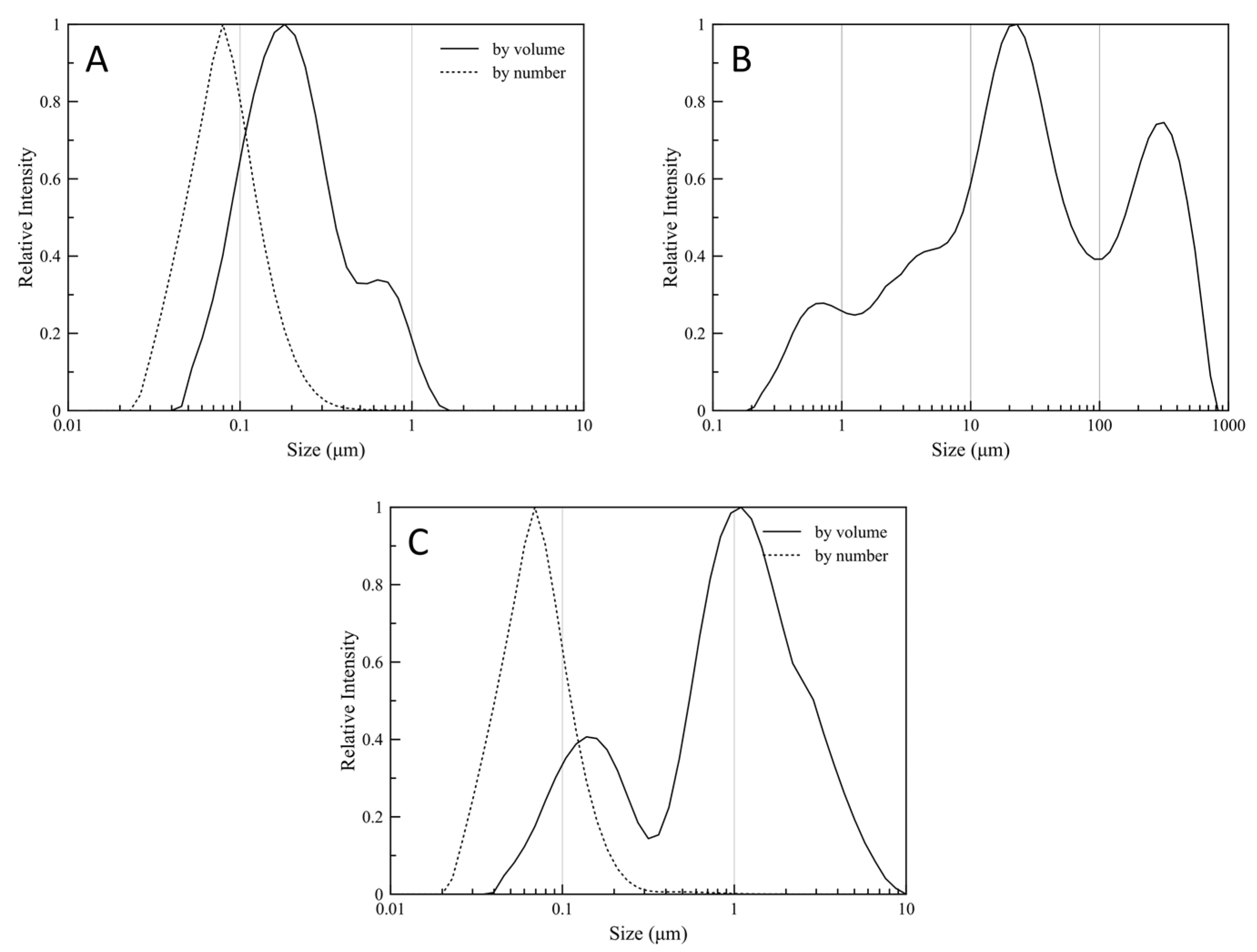
3.5. Particles Size Distribution Analysis
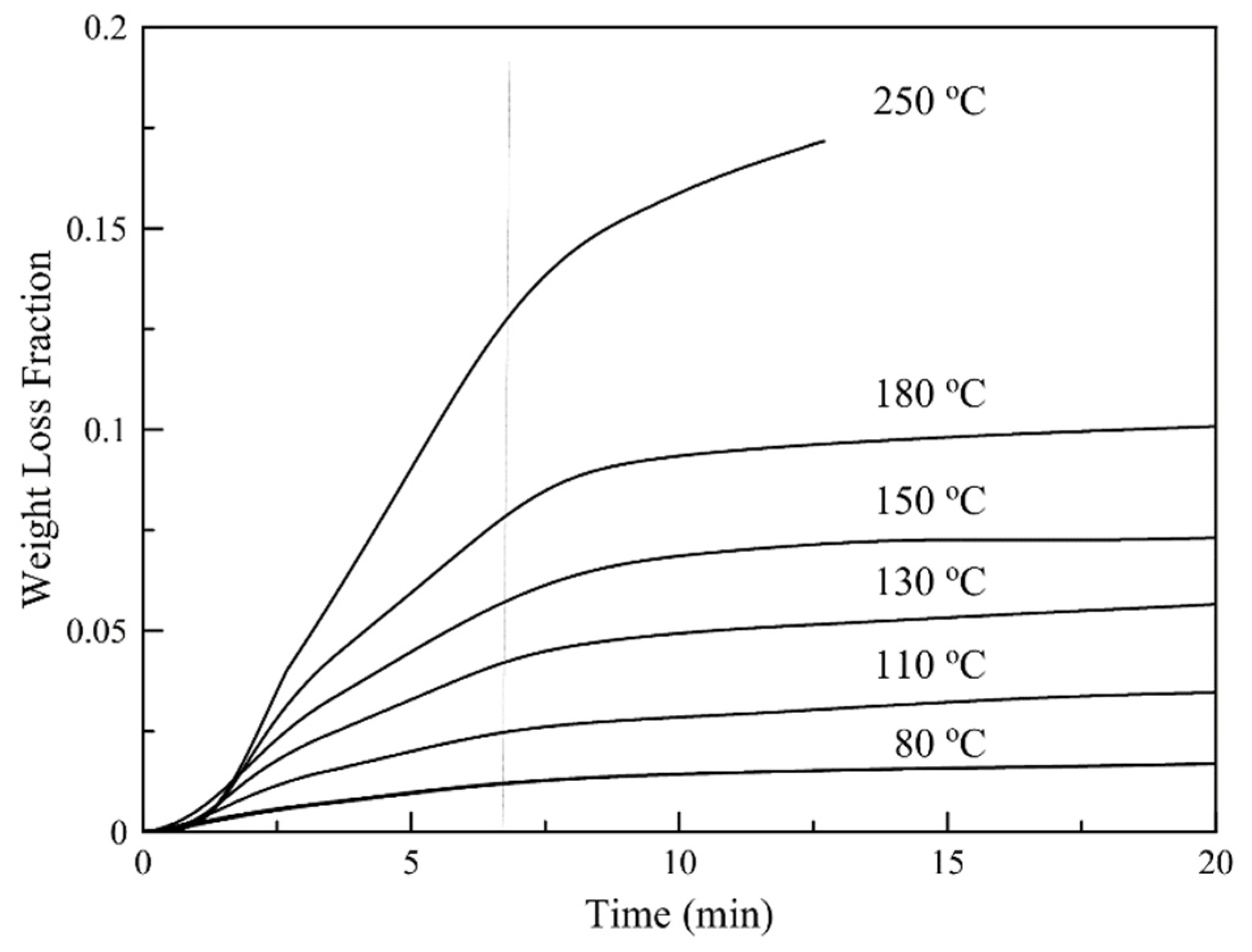
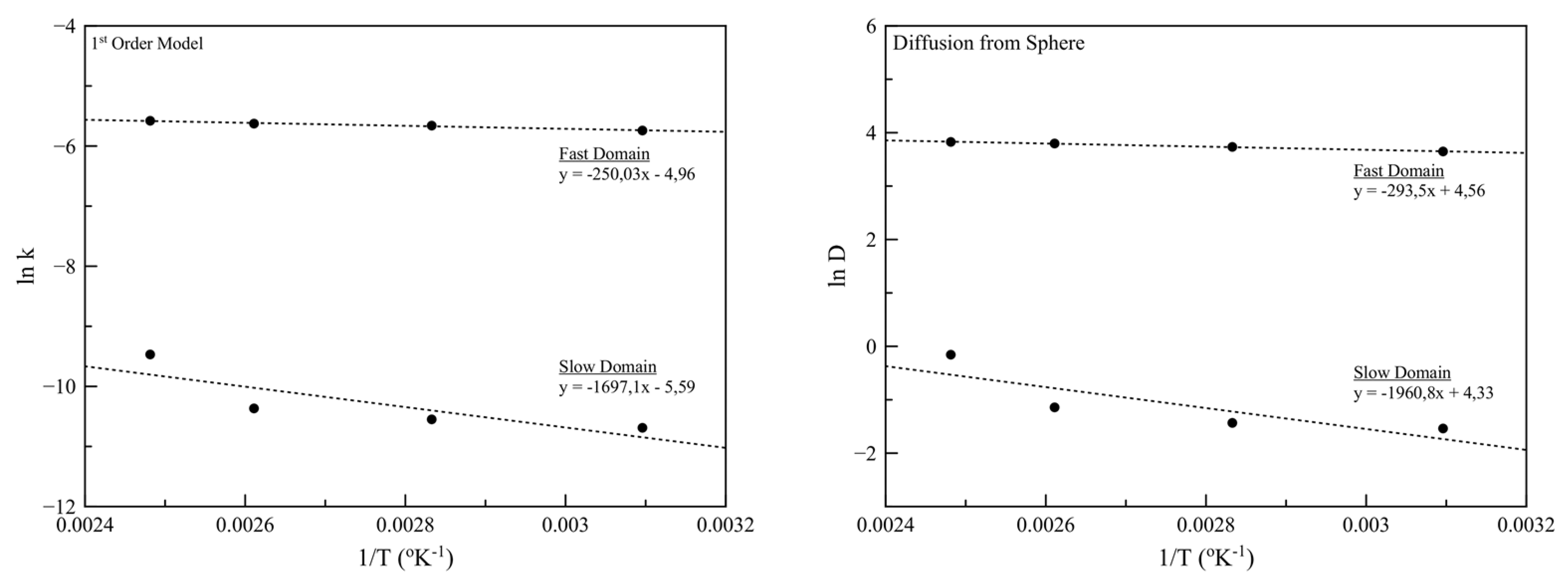
3.6. Isothermal TGA Analysis
- The porous bed of the sample is considered as a single batch and is stable, with no form disposition or changes during the process.
- Nitrogen flow is plug-flow, with a constant rate.
- Particles are isotropic of equal shape and equal initial oil concentration.
- The effective coefficient of diffusion through the particles is constant.
- There is no resistance to the mass transfer of the oil from the external surfaces of the particles.
- The oil is uniformly distributed in the particles.
- The oil is considered as a single component.
- Particle clusters are considered as individual spherical particles, consisted of uniform material.
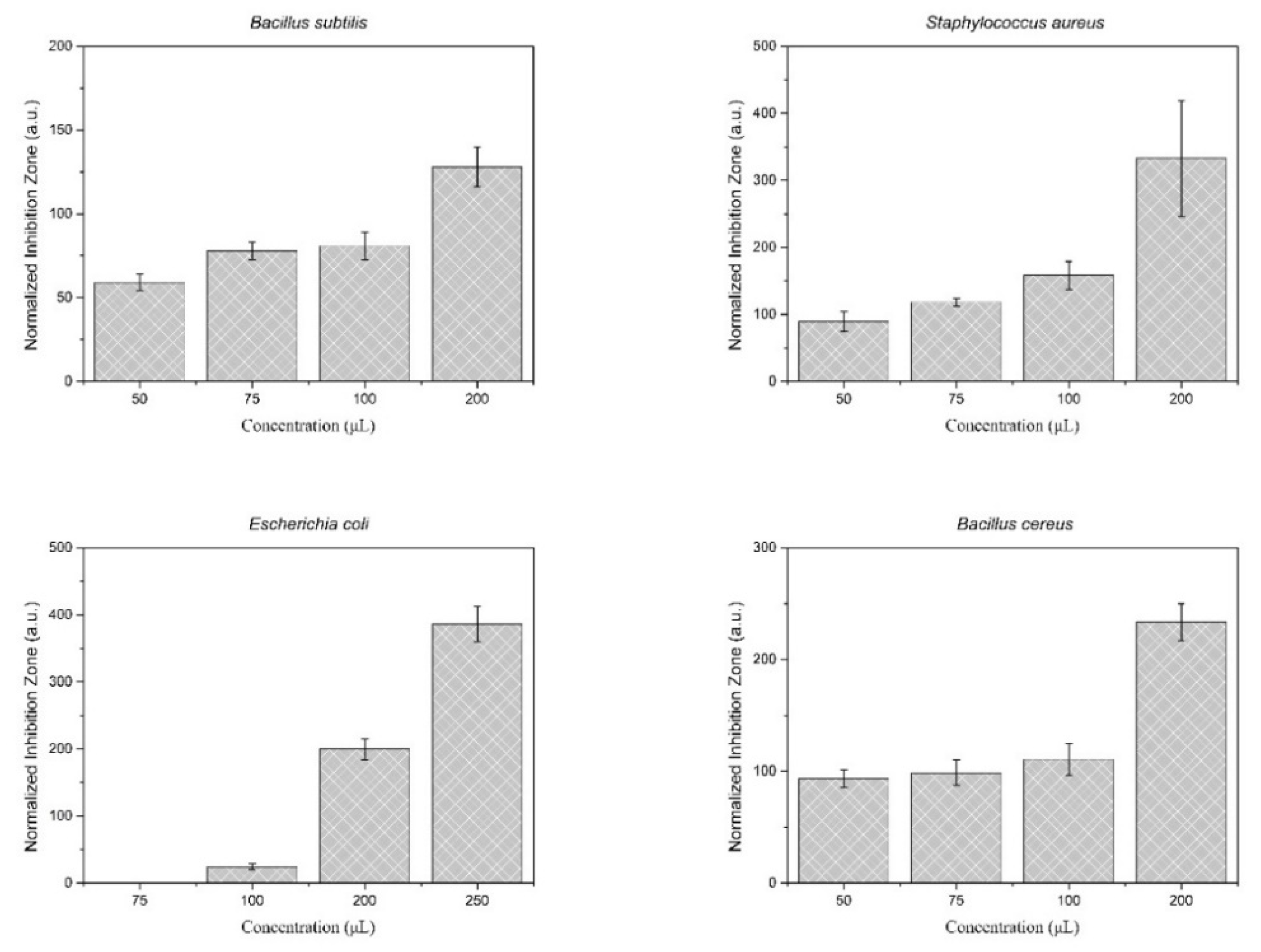
3.7. Antimicrobial Activity Study
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Elshafie, H.S.; Camele, I. An Overview of the Biological Effects of Some Mediterranean Essential Oils on Human Health. BioMed Res. Int. 2017, 2017, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Wińska, K.; Wińska, K.; Łyczko, J.; Mączka, W.; Czubaszek, A.; Szumny, A. Essential Oils as Antimicrobial Agents—Myth or Real Alternative? Molecules 2019, 24, 2130. [Google Scholar] [CrossRef] [PubMed]
- Méndez-Vilas, A. Microbial Pathogens and Strategies for Combating Them: Science, Technology and Education; Formatex Research Center: Norristown, PA, USA, 2013; ISBN 9788493984397. [Google Scholar]
- González-Mas, M.C.; Rambla, J.L.; López-Gresa, M.P.; Blázquez, M.A.; Granell, A. Volatile Compounds in Citrus Essential Oils: A Comprehensive Review. Front. Plant Sci. 2019, 10, 12. [Google Scholar] [CrossRef] [PubMed]
- Dagli, N.; Dagli, R.J.; Mahmoud, R.S.; Baroudi, K. Essential oils, their therapeutic properties, and implication in dentistry: A review. J. Int. Soc. Prev. Community Dent. 2015, 5, 335–340. [Google Scholar] [CrossRef]
- Alahi, E.; Mukhopadhyay, S.C. Detection Methodologies for Pathogen and Toxins: A Review. Sensors 2017, 17, 1885. [Google Scholar] [CrossRef]
- Logan, N. Bacillus and relatives in foodborne illness. J. Appl. Microbiol. 2011, 112, 417–429. [Google Scholar] [CrossRef]
- Guimarães, A.C.; Meireles, L.M.; Lemos, M.F.; Guimarães, M.C.C.; Endringer, D.C.; Fronza, M.; Scherer, R. Antibacterial Activity of Terpenes and Terpenoids Present in Essential Oils. Molecules 2019, 24, 2471. [Google Scholar] [CrossRef]
- Han, Y.; Sun, Z.; Chen, W. Antimicrobial Susceptibility and Antibacterial Mechanism of Limonene against Listeria monocytogenes. Molecules 2019, 25, 33. [Google Scholar] [CrossRef]
- Da Silva, J.K.R.; Figueiredo, P.L.; Byler, K.G.; Setzer, W.N. Essential Oils as Antiviral Agents, Potential of Essential Oils to Treat SARS-CoV-2 Infection: An In-Silico Investigation. Int. J. Mol. Sci. 2020, 21, 3426. [Google Scholar] [CrossRef]
- Kumar, K.J.S.; Vani, M.G.; Wang, C.-S.; Chen, C.-C.; Chen, Y.-C.; Lu, L.-P.; Huang, C.-H.; Lai, C.-S.; Wang, S.-Y. Geranium and Lemon Essential Oils and Their Active Compounds Downregulate Angiotensin-Converting Enzyme 2 (ACE2), a SARS-CoV-2 Spike Receptor-Binding Domain, in Epithelial Cells. Plants 2020, 9, 770. [Google Scholar] [CrossRef]
- Boukhatem, M.N.; Setzer, W.N. Aromatic Herbs, Medicinal Plant-Derived Essential Oils, and Phytochemical Extracts as Potential Therapies for Coronaviruses: Future Perspectives. Plants 2020, 9, 800. [Google Scholar] [CrossRef] [PubMed]
- Thuy, B.T.P.; My, T.T.A.; Hai, N.T.T.; Hieu, L.T.; Hoa, T.T.; Loan, H.T.P.; Triet, N.T.; Van Anh, T.T.; Quy, P.T.; Van Tat, P.; et al. Investigation into SARS-CoV-2 Resistance of Compounds in Garlic Essential Oil. ACS Omega 2020, 5, 8312–8320. [Google Scholar] [CrossRef] [PubMed]
- Vimalanathan, S.; Hudson, J. Anti-Influenza Virus Activity of Essential Oils and Vapors. Am. J. Essent. Oils Nat. Prod. 2014, 2, 47–53. [Google Scholar]
- Theisinger, S.; Schoeller, K.; Osborn, B.; Sarkar, M.; Landfester, K. Encapsulation of a Fragrance via Miniemulsion Polymerization for Temperature-Controlled Release. Macromol. Chem. Phys. 2009, 210, 411–420. [Google Scholar] [CrossRef]
- Mahato, N.; Sharma, K.; Koteswararao, R.; Sinha, M.; Baral, E.; Cho, M.H. Citrus essential oils: Extraction, authentication and application in food preservation. Crit. Rev. Food Sci. Nutr. 2017, 59, 611–625. [Google Scholar] [CrossRef]
- Bora, H.; Kamle, M.; Mahato, D.K.; Tiwari, P.; Kumar, P. Citrus Essential Oils (CEOs) and Their Applications in Food: An Overview. Plants 2020, 9, 357. [Google Scholar] [CrossRef]
- Chaiwarit, T.; Ruksiriwanich, W.; Jantanasakulwong, K.; Jantrawut, P. Use of Orange Oil Loaded Pectin Films as Antibacterial Material for Food Packaging. Polymers 2018, 10, 1144. [Google Scholar] [CrossRef]
- Figueroa-Lopez, K.J.; Cabedo, L.; Lagaron, J.M.; Torres-Giner, S. Development of Electrospun Poly(3-hydroxybutyrate-co-3-hydroxyvalerate) Monolayers Containing Eugenol and Their Application in Multilayer Antimicrobial Food Packaging. Front. Nutr. 2020, 7, 140. [Google Scholar] [CrossRef]
- Lan, W.; Liang, X.; Lan, W.; Ahmed, S.; Liu, Y.; Qin, W. Electrospun Polyvinyl Alcohol/d-Limonene Fibers Prepared by Ultrasonic Processing for Antibacterial Active Packaging Material. Molecules 2019, 24, 767. [Google Scholar] [CrossRef]
- Lan, W.; Wang, S.; Chen, M.; Sameen, D.E.; Lee, K.; Liu, Y. Developing poly(vinyl alcohol)/chitosan films incorporate with d-limonene: Study of structural, antibacterial, and fruit preservation properties. Int. J. Biol. Macromol. 2020, 145, 722–732. [Google Scholar] [CrossRef]
- McKay, S.; Sawant, P.; Fehlberg, J.; Almenar, E. Antimicrobial activity of orange juice processing waste in powder form and its suitability to produce antimicrobial packaging. Waste Manag. 2021, 120, 230–239. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Rhim, J.-W. Fabrication of Copper Sulfide Nanoparticles and Limonene Incorporated Pullulan/Carrageenan-Based Film with Improved Mechanical and Antibacterial Properties. Polymers 2020, 12, 2665. [Google Scholar] [CrossRef]
- Arrieta, M.P.; Lopez, J.M.; Ferrándiz, S.; Peltzer, M.A. Characterization of PLA-limonene blends for food packaging applications. Polym. Test. 2013, 32, 760–768. [Google Scholar] [CrossRef]
- Escamilla-García, M.; Calderón-Domínguez, G.; Chanona-Pérez, J.; Mendoza-Madrigal, A.G.; Di Pierro, P.; García-Almendárez, B.; Amaro-Reyes, A.; Regalado-González, C. Physical, Structural, Barrier, and Antifungal Characterization of Chitosan–Zein Edible Films with Added Essential Oils. Int. J. Mol. Sci. 2017, 18, 2370. [Google Scholar] [CrossRef] [PubMed]
- Blanco-Padilla, A.; Soto, K.M.; Iturriaga, M.H.; Mendoza, S. Food Antimicrobials Nanocarriers. Sci. World J. 2014, 2014, 1–11. [Google Scholar] [CrossRef]
- Thickett, S.C.; Teo, G.H. Recent advances in colloidal nanocomposite designviaheterogeneous polymerization techniques. Polym. Chem. 2019, 10, 2906–2924. [Google Scholar] [CrossRef]
- Landfester, K. Miniemulsions for Nanoparticle Synthesis. In Colloid Chemistry II; Topics in Current Chemistry; Antonietti, M., Ed.; Springer: Berlin/Heidelberg, Germany, 2003; Volume 227, pp. 75–123. ISBN 978-3-540-00418-9. [Google Scholar]
- Kermabon-Avon, G.; Bressy, C.; Margaillan, A. Statistical design strategies to optimize properties in miniemulsion polymerization of methyl methacrylate. Eur. Polym. J. 2009, 45, 1208–1216. [Google Scholar] [CrossRef]
- Andriotis, E.G.; Achilias, D.S. Optimizing the synthesis of bio-based polymers using naturally occurring monomers by response surface methodology. Fresenius Environ. Bull. 2013, 22, 3808–3814. [Google Scholar]
- Box, G.E.P.; Hunter, J.S.; Hunter, W.G. Statistics for Experimenters: Design, Innovation, and Discovery, Wiley Series in Probability and Statistics, 2nd ed.; Wiley-Interscience: Hoboken, NJ, USA, 2005; ISBN 978-0-471-71813-0. [Google Scholar]
- Bauer, A.W.; Kirby, W.M.; Sherris, J.C.; Turck, M. Antibiotic susceptibility testing by a standardized single disk method. Am. J. Clin. Pathol. 1966, 45, 493–496. [Google Scholar] [CrossRef]
- Trikas, E.D.; Melidou, M.; Papi, R.; Zachariadis, G.A.; Kyriakidis, D.A. Extraction, separation and identification of anthocyanins from red wine by-product and their biological activities. J. Funct. Foods 2016, 25, 548–558. [Google Scholar] [CrossRef]
- Karavasili, C.; Tsonga, C.; Andreadis, I.; Andriotis, E.G.; Papachristou, E.T.; Papi, R.M.; Tzetzis, D.; Fatouros, D.G. Physico-Mechanical and Finite Element Analysis Evaluation of 3D Printable Alginate-Methylcellulose Inks for Wound Healing Applications. Carbohydr. Polym. 2020, 247, 116666. [Google Scholar] [CrossRef] [PubMed]
- Andriotis, E.G.; Achilias, D.S. Role of Polylimonene as a Bio-Based Additive in Thermal Oxidation of High Impact Polystyrene. Macromol. Symp. 2013, 331, 173–180. [Google Scholar] [CrossRef]
- Andriotis, E.G.; Koumbis, A.E.; Achilias, D. Nitroxide-mediated polymerization of styrene and limonene in the framework of synthesis of potentially functional polymers using naturally occurring terpenes. Polym. Bull. 2020, 1–20. [Google Scholar] [CrossRef]
- Coulson, C.A.; Crank, J. The Mathematics of Diffusion. Math. Gaz. 1958, 42, 165. [Google Scholar] [CrossRef]
- Hosh, U.G.; Alley, J.W.T.; Uthy, R.G.L. Particle-Scale Investigation of PAH Desorption Kinetics and Thermodynamics from Sediment. Environ. Sci. Technol. 2001, 35, 3468–3475. [Google Scholar] [CrossRef]
- Shor, L.M.; Rockne, K.J.; Taghon, G.; Young, L.Y.; Kosson, D.S. Desorption Kinetics for Field-Aged Polycyclic Aromatic Hydrocarbons from Sediments. Environ. Sci. Technol. 2003, 37, 1535–1544. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, F.; de Moura, M.R.; Capparelli Mattoso, L.H. Biodegradable Hydrogel as Delivery Vehicle for the Controlled Release of Pesticide. In Pesticides-Formulations, Effects, Fate; Stoytcheva, M., Ed.; InTech: London, UK, 2011; ISBN 978-953-307-532-7. [Google Scholar]
- Ibáñez, M.D.; Sanchez, N.; Blázquez, M.A. Encapsulated Limonene: A Pleasant Lemon-Like Aroma with Promising Application in the Agri-Food Industry. A Review. Molecules 2020, 25, 2598. [Google Scholar] [CrossRef] [PubMed]
- Vishwakarma, G.S.; Gautam, N.; Babu, J.N.; Mittal, S.; Jaitak, V. Polymeric Encapsulates of Essential Oils and Their Constituents: A Review of Preparation Techniques, Characterization, and Sustainable Release Mechanisms. Polym. Rev. 2016, 56, 668–701. [Google Scholar] [CrossRef]
- Sansukcharearnpon, A.; Wanichwecharungruang, S.; Leepipatpaiboon, N.; Kerdcharoen, T.; Arayachukeat, S. High loading fragrance encapsulation based on a polymer-blend: Preparation and release behavior. Int. J. Pharm. 2010, 391, 267–273. [Google Scholar] [CrossRef]
- Paramita, V.; Furuta, T.; Yoshii, H. High-Oil-Load Encapsulation of Medium-Chain Triglycerides and d-Limonene Mixture in Modified Starch by Spray Drying. J. Food Sci. 2012, 77, E38–E44. [Google Scholar] [CrossRef]
- Chen, J.; Jiang, Q.-D.; Chai, Y.-P.; Zhang, H.; Peng, P.; Yang, X.-X. Natural Terpenes as Penetration Enhancers for Transdermal Drug Delivery. Molecules 2016, 21, 1709. [Google Scholar] [CrossRef] [PubMed]
- Chikhoune, A.; Hazzit, M.; Kerbouche, L.; Baaliouamer, A.; Aissat, K. Tetraclinis articulata(Vahl) Masters essential oils: Chemical composition and biological activities. J. Essent. Oil Res. 2013, 25, 300–307. [Google Scholar] [CrossRef]
- Lv, F.; Liang, H.; Yuan, Q.; Li, C. In vitro antimicrobial effects and mechanism of action of selected plant essential oil combinations against four food-related microorganisms. Food Res. Int. 2011, 44, 3057–3064. [Google Scholar] [CrossRef]
- Zahi, M.R.; El Hattab, M.; Liang, H.; Yuan, Q. Enhancing the antimicrobial activity of d-limonene nanoemulsion with the inclusion of ε-polylysine. Food Chem. 2017, 221, 18–23. [Google Scholar] [CrossRef]
- The Potential Risks Arising from Nanoscience and Nanotechnologies on Food and Feed Safety. EFSA J. 2009, 7, 7. [CrossRef]
- European Food Safety Authority (EFSA). Table of Public Comments on the EFSA Draft Nanotechnology Opinion. EFSA Support. Publ. 2009, 6, 6. [CrossRef][Green Version]


| Factor | Name | Low Value (−1) | High Value (+1) |
|---|---|---|---|
| A | Surfactant (% w/w) | 3 | 5 |
| B | Cosurfactant (% w/w) | 2 | 5 |
| C | Crosslinker (% w/w) | 5 | 10 |
| D | D-limonene (% w/w) | 5 | 15 |
| E | Sonication Time (s) | 180 | 300 |
| Run | A | B | C | D | E | Loading (a.u.) | Span (a.u.) | D[4,3] (μm) | D[3,2] (μm) | d (0.1) (μm) | d (0.5) (μm) | d (0.9) (μm) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | −1 | 1 | 1 | 1 | −1 | 0.097 | 1.527 | 0.754 | 0.371 | 0.039 | 0.072 | 0.149 |
| 2 | 1 | 1 | -1 | −1 | 1 | 0.093 | 1.341 | 0.219 | 0.140 | 0.035 | 0.064 | 0.121 |
| 3 | 1 | −1 | 1 | −1 | −1 | 0.061 | 1.398 | 0.263 | 0.159 | 0.035 | 0.066 | 0.127 |
| 4 | 1 | 1 | 1 | 1 | 1 | 0.085 | 1.467 | 0.318 | 0.186 | 0.036 | 0.068 | 0.136 |
| 5 | −1 | 1 | -1 | −1 | −1 | 0.058 | 1.302 | 0.351 | 0.161 | 0.034 | 0.063 | 0.117 |
| 6 | −1 | −1 | 1 | −1 | 1 | 0.059 | 1.429 | 0.407 | 0.224 | 0.041 | 0.076 | 0.150 |
| 7 | −1 | −1 | −1 | 1 | 1 | 0.071 | 1.393 | 0.487 | 0.240 | 0.040 | 0.074 | 0.142 |
| 8 | 1 | −1 | −1 | 1 | −1 | 0.077 | 1.393 | 0.297 | 0.169 | 0.035 | 0.066 | 0.127 |
| Estimate | |||
|---|---|---|---|
| Effect | Loading | Span (a.u.) | D[3,2] (μm) |
| A | 0.00783 | 0.0130 | −0.0855 |
| B | 0.01578 | 0.0060 | 0.0165 |
| C | 0.00091 | 0.0980 | 0.0575 |
| D | 0.01489 | 0.0775 | 0.0705 |
| E | 0.00367 | 0.0025 | −0.0175 |
| Run | A | B | C | D | E | Loading (a.u.) | Span (a.u.) | D[4,3] (μm) | D[3,2] (μm) |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 5 (% w/w) | 3 (% w/w) | 5 (% w/w) | 10 (% w/w) | 180 (s) | 0.125 | 1.349 | 0.314 | 0.162 |
| 2 | 0.096 | 1.341 | 0.330 | 0.169 | |||||
| 3 | 0.108 | 1.306 | 0.294 | 0.159 | |||||
| Average | 0.109 | 1.332 | 0.313 | 0.163 |
| By Volume | Span | D[4,3] (μm) | D[3,2] (μm) | d (0,1) (μm) | d (0,5) (μm) | d (0,9) (μm) |
|---|---|---|---|---|---|---|
| Latex | 2.746 | 0.227 | 0.135 | 0.073 | 0.16 | 0.513 |
| Re-dispersion | 2.737 | 1.118 | 0.331 | 0.113 | 0.861 | 2.47 |
| Dry Powder | 12.930 | 80.690 | 3.700 | 1.310 | 20.820 | 270.540 |
| 1st Order | Diffusion from Sphere | ||||||
|---|---|---|---|---|---|---|---|
| Temperature (°C) | f (Fast Fraction) | kf × 10−3 (s−1) | ks × 10−3 (s−1) | r2 | Df (μ2/s) | Ds (μ2/s) | r2 |
| 50 | 0.120 | 3.21 | 0.023 | 0.997 | 38.41 | 0.215 | 0.968 |
| 80 | 0.115 | 3.48 | 0.026 | 0.998 | 41.82 | 0.239 | 0.968 |
| 110 | 0.256 | 3.60 | 0.032 | 0.999 | 44.60 | 0.319 | 0.962 |
| 130 | 0.421 | 3.77 | 0.077 | 0.998 | 45.93 | 0.855 | 0.966 |
| 150 | 0.629 | 3.28 | 0 | 0.992 | 39.05 | 0.490 | 0.939 |
| 180 | 0.864 | 2.86 | 0 | 0.991 | 35.28 | 3.181 | 0.946 |
| Ea (kJ/mol), Fast Domain | Ea (kJ/mol), Slow Domain | |
|---|---|---|
| 1st Order kinetic Model | 30.1 | 204.1 |
| Diffusion from Sphere Model | 35.3 | 235.8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Andriotis, E.G.; Papi, R.M.; Paraskevopoulou, A.; Achilias, D.S. Synthesis of D-Limonene Loaded Polymeric Nanoparticles with Enhanced Antimicrobial Properties for Potential Application in Food Packaging. Nanomaterials 2021, 11, 191. https://doi.org/10.3390/nano11010191
Andriotis EG, Papi RM, Paraskevopoulou A, Achilias DS. Synthesis of D-Limonene Loaded Polymeric Nanoparticles with Enhanced Antimicrobial Properties for Potential Application in Food Packaging. Nanomaterials. 2021; 11(1):191. https://doi.org/10.3390/nano11010191
Chicago/Turabian StyleAndriotis, Eleftherios G., Rigini M. Papi, Adamantini Paraskevopoulou, and Dimitris S. Achilias. 2021. "Synthesis of D-Limonene Loaded Polymeric Nanoparticles with Enhanced Antimicrobial Properties for Potential Application in Food Packaging" Nanomaterials 11, no. 1: 191. https://doi.org/10.3390/nano11010191
APA StyleAndriotis, E. G., Papi, R. M., Paraskevopoulou, A., & Achilias, D. S. (2021). Synthesis of D-Limonene Loaded Polymeric Nanoparticles with Enhanced Antimicrobial Properties for Potential Application in Food Packaging. Nanomaterials, 11(1), 191. https://doi.org/10.3390/nano11010191