Impact of Iron on the Fe–Co–Ni Ternary Nanocomposites Structural and Magnetic Features Obtained via Chemical Precipitation Followed by Reduction Process for Various Magnetically Coupled Devices Applications
Abstract
1. Introduction
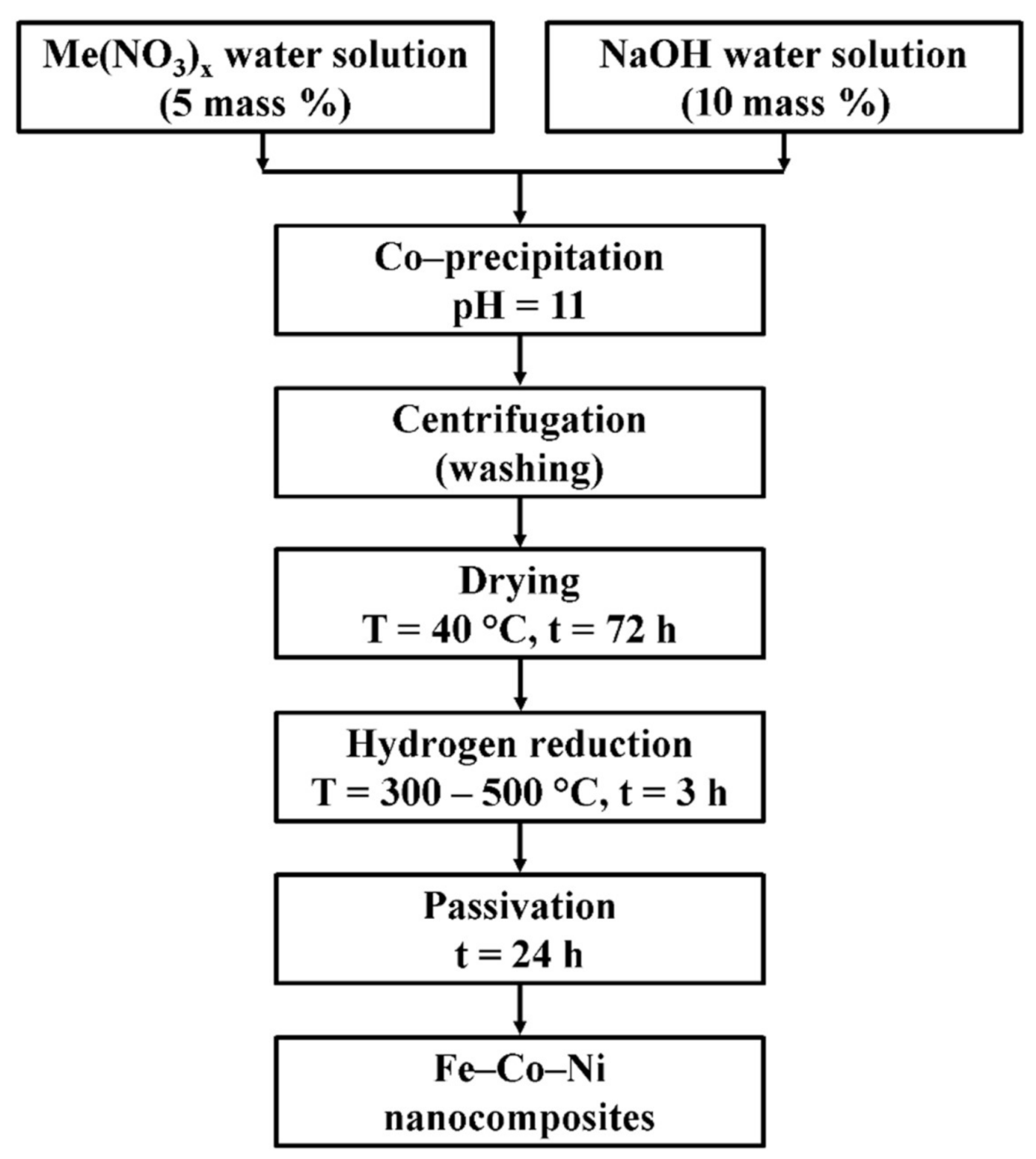
2. Materials and Methods
3. Results and Discussion
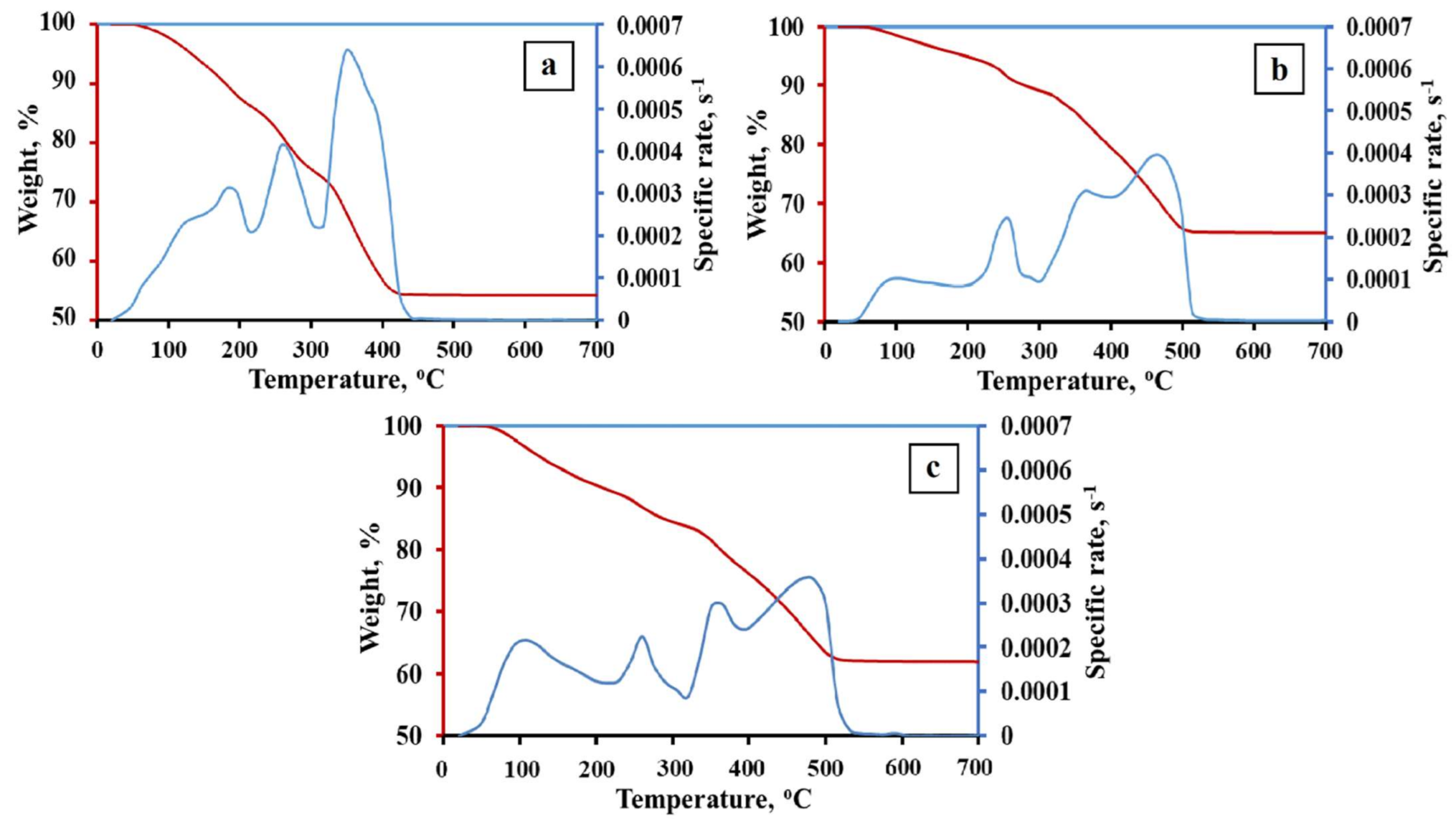
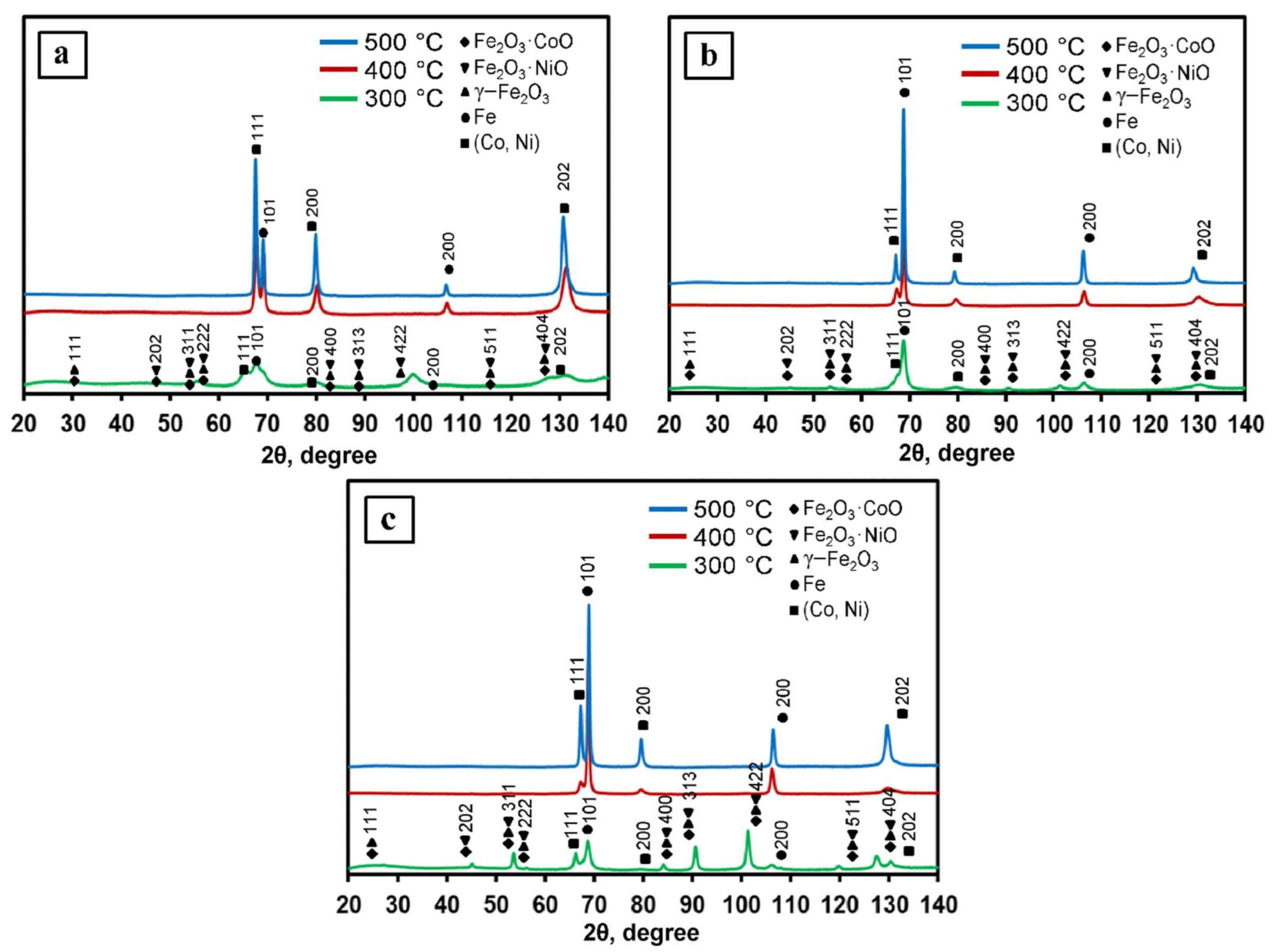
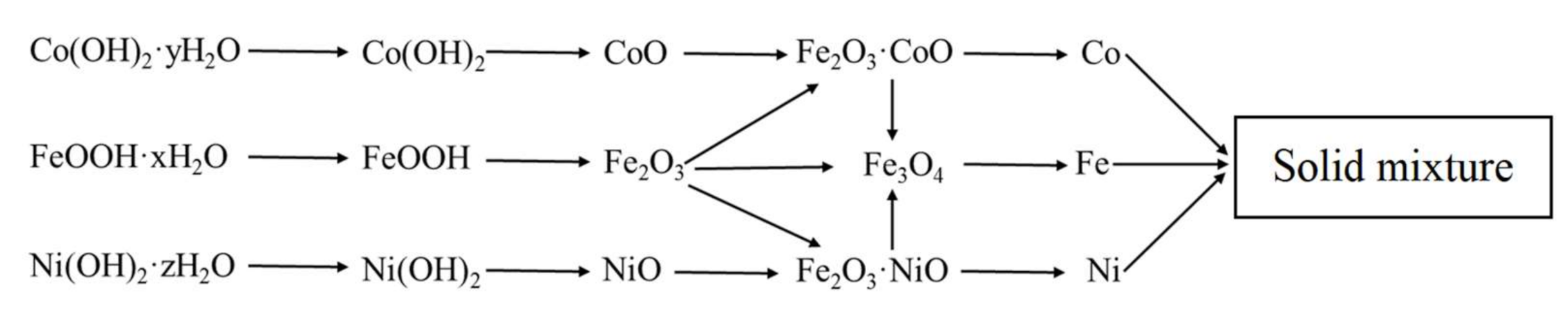
3.1. Phase Formation of Fe–Co–Ni Nanocomposites
3.2. Dispersion and Morphological Characteristics
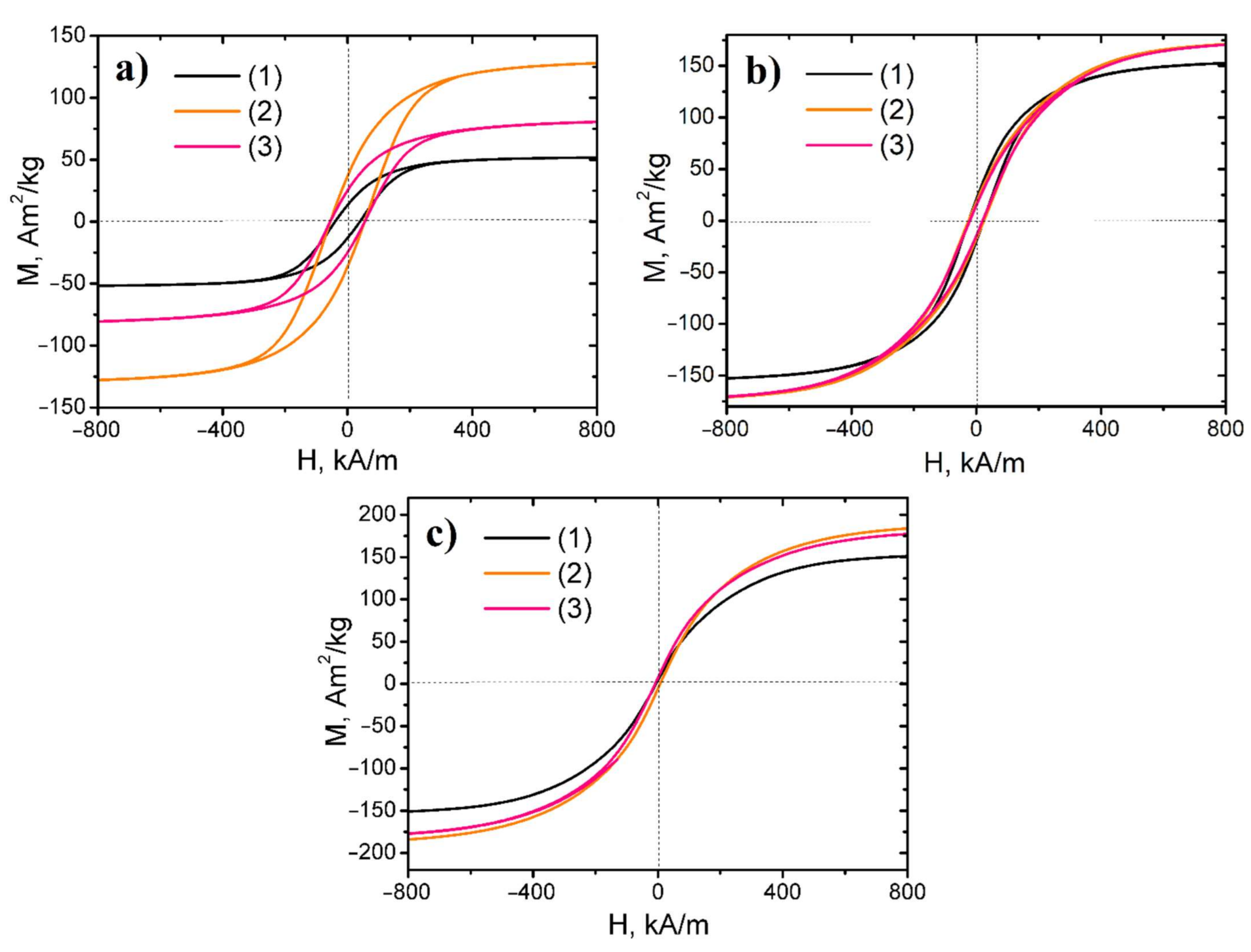
3.3. Magnetic Properties
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Mohapatra, J.; Xing, M.; Elkins, J.; Liu, J.P. Hard and semi-hard magnetic materials based on cobalt and cobalt alloys. J. Alloys Compd. 2020, 824, 153874. [Google Scholar] [CrossRef]
- Zhu, K.; Ju, Y.; Xu, J.; Yang, Z.; Gao, S.; Hou, Y. Magnetic Nanomaterials: Chemical Design, Synthesis, and Potential Applications. Acc. Chem. Res. 2018, 51, 404–413. [Google Scholar] [CrossRef]
- Kalska-Szostko, B.; Klekotka, U.; Olszewskib, W.; Satuł, D. Multilayered and alloyed Fe–Co and Fe–Ni nanowires physicochemical studies. J. Magn. Magn. Mater. 2019, 484, 67–73. [Google Scholar] [CrossRef]
- Solanki, V.; Lebedev, O.I.; Seikh, M.M.; Mahato, N.K.; Kundu, A.K. Synthesis and characterization of Co-Ni and Fe–Ni alloy nanoparticles. J. Magn. Magn. Mater. 2016, 42015, 39–44. [Google Scholar] [CrossRef]
- Raanaei, H.; Eskandari, H.; Mohammad-Hosseini, V. Structural and magnetic properties of nanocrystalline Fe–Co–Ni alloy processed by mechanical alloying. J. Magn. Magn. Mater. 2016, 39815, 190–195. [Google Scholar] [CrossRef]
- Nautiyal, P.; Seikh, M.M.; Lebedev, O.I.; Kundu, A.K. Sol-gel synthesis of Fe–Co nanoparticles and magnetization study. J. Magn. Magn. Mater. 2015, 3771, 402–405. [Google Scholar] [CrossRef]
- Suman, G. Chemical, Physical and Mechanical Properties of Nanomaterials and its Applications. Ph.D. Thesis, University of Iowa, Iowa City, IA, USA, 2013. [Google Scholar] [CrossRef]
- Jeevanandam, J.; Barhoum, A.; Chan, Y.S.; Dufresne, A.; Beilstein, M.K.D. Review on nanoparticles and nanostructured materials: History, sources, toxicity and regulations. J. Nanotechnol. 2018, 9, 1050–1074. [Google Scholar] [CrossRef]
- Leslie-Pelecky, D.L.; Rieke, R.D. Magnetic properties of nanostructured materials. Chem. Mater. 1996, 8, 1770–1783. [Google Scholar] [CrossRef]
- Li, Q.; Kartikowati, C.W.; Horie, S.; Ogi, T.; Iwaki, T.; Okuyama, K. Correlation between particle size/domain structure and magnetic properties of highly crystalline Fe3O4 nanoparticles. Sci. Rep. 2017, 7, 9894. [Google Scholar] [CrossRef]
- Li, X.; Takahashi, S. Synthesis and magnetic properties of Fe–Co–Ni nanoparticles by hydrogen plasma-metal reaction. J. Magn. Magn. Mater. 2000, 214, 195–203. [Google Scholar] [CrossRef]
- Jartych, E. On the magnetic properties of mechanosynthesized Co–Fe–Ni ternary alloys. J. Magn. Magn. Mater. 2011, 323, 209–216. [Google Scholar] [CrossRef]
- Rodrigues, A.P.G.; Gomes, D.K.S.; Araújo, J.H.; Melo, D.M.A.; Oliveira, N.A.S.; Braga, R.M. Nanoferrites of nickel doped with cobalt: Influence of Co2+ on the structural and magnetic properties. J. Magn. Magn. Mater. 2015, 374, 748–754. [Google Scholar] [CrossRef]
- Pugachev, V.M.; Zaharov, Y.A.; Datiy, K.A.; Popova, A.N.; Bogomyakov, A.S. The Temperature Effect on Properties of Fe–Co–Ni Nanostructured System. Eurasian Chem. Technol. J. 2015, 17, 193–200. [Google Scholar] [CrossRef]
- Chokprasombat, K.; Pinitsoontorn, S.; Maensiri, S. Effects of Ni content on nanocrystalline Fe–Co–Ni ternaryalloys synthesized by a chemical reduction method. J. Magn. Magn. Mater. 2016, 405, 174–180. [Google Scholar] [CrossRef]
- Sun, Y.; Zhou, X.; Liu, Y.; Zhao, G.; Jiang, Y. Effect of magnetic nanoparticles on the properties of magnetic rubber. Mater. Res. Bull. 2010, 45, 878–881. [Google Scholar] [CrossRef]
- Newnham, R.E.; Giniewicz, J.R. Fiber Reinforcements and General Theory of Composites. Compr. Compos. Mater. 2000. [Google Scholar] [CrossRef]
- Kalia, S.; Kango, S.; Kumar, A.; Haldorai, Y.; Kumari, B.; Kumar, R. Magnetic polymer nanocomposites for environmental and biomedical applications. Colloid Polym. Sci. 2014, 292, 2025–2052. [Google Scholar] [CrossRef]
- Jänis, A.; Olsson, R.T.; Savage, S.J.; Gedde, U.W.; Klement, U. Microwave absorbing properties of ferrite-based nanocomposites. Behav. Mech. Multifunct. Compos. Mater. 2007, 6526, 1–10. [Google Scholar] [CrossRef]
- Ahmad, S.H.; Abdullah, M.H.; Hui, D.; Yusoff, A.N.; Puryanti, D. Magnetic and microwave absorbing properties of magnetite-thermoplastic natural rubber nanocomposites. J. Magn. Magn. Mater. 2010, 322, 3401–3409. [Google Scholar] [CrossRef]
- Li, N.; Hu, C.; Cao, M. Enhanced microwave absorbing performance of CoNi alloy nanoparticles anchored on a spherical carbon monolith. Phys Chem. Chem. Phys. 2013, 15, 7685–7689. [Google Scholar] [CrossRef]
- Zelensky, V.A.; Ankudinov, A.B.; Alymov, M.I.; Rubtsov, N.M.; Tregubova, I.V. Application of nickel nanopowders of different morphology for the synthesis of highly porous materials by powder metallurgy. IOP Conf. Ser. Mater. Sci. Eng. 2019, 558, 012055. [Google Scholar] [CrossRef]
- Alymov, M.; Rubtsov, N.M.; Seplyarskii, B.S. Preparation and characterization of iron nanoparticles protected by an oxide film. Inorg. Mater. 2017, 53, 911–915. [Google Scholar] [CrossRef]
- Taylor, A.; Sinclair, H. On the determination of lattice parameters by the debye-scherrer method. Proc. Phys. Soc. 1945, 57, 126–135. [Google Scholar] [CrossRef]
- Brunauer, S.; Emmett, P.H.; Teller, E. Adsorption of Gases in Multimolecular Layers. J. Am. Chem. Soc. 1938, 60, 309–319. [Google Scholar] [CrossRef]
- Michel, F.; Courard, L. Particle Size Distribution of Limestone Fillers: Granulometry and Specific Surface Area Investigations. Part. Sci. Technol. 2014, 32, 334–340. [Google Scholar] [CrossRef]
- Chaly, V.P. Metal hydroxide. The regularities of formation, composition, structure and properties. Kiev: Nauk. Dumka 1972. (In Russian) [Google Scholar]
- Tikhomirov, S.A.; Alymov, M.I.; Tregubova, I.V.; Shustov, V.S. Influence that cobalt hydroxide reduction regimes have on dispersion and reduction degree of cobalt nanopowders. Nanotechnol. Russ. 2011, 6, 268. [Google Scholar] [CrossRef]
- Manukyan, K.V.; Avetisyan, A.G.; Shuck, C.E.; Chatilyan, H.A.; Rouvimov, S.; Kharatyan, S.L.; Mukasyan, A.S. Nickel Oxide Reduction by Hydrogen: Kinetics and Structural Transformations. J. Phys. Chem. C 2015, 119, 16131–16138. [Google Scholar] [CrossRef]
- Bannykh, O.A.; Budberg, P.B.; Alisova, S.P. Phase Diagrams of Binary and Multicomponent Systems Based on Iron. Mosc. Metallurgiya 1986. (In Russian) [Google Scholar]
- Zaharov, Y.A.; Pugachev, V.M.; Ovcharenko, V.I.; Datiy, K.A.; Popova, A.N.; Bogomyakov, A.S. Phase Composition and Magnetic Properties of Nanostructured Fe–Co–Ni Powders. Phys. Status. Solidi. 2018, 255, 1700175. [Google Scholar] [CrossRef]
- Muratov, D.G.; Kozhitov, L.V.; Karpenkov, D.Y.; Yakushko, E.V.; Korovin, E.Y.; Vasil’ev, A.V.; Popkova, A.V.; Kazaryan, T.M.; Shadrinov, A.V. Synthesis and Magnetic Properties of Fe–Co–Ni/C Nanocomposites. Russ. Phys. J. 2018, 60, 1924–1930. [Google Scholar] [CrossRef]
- Muratov, D.G.; Kozhitov, L.V.; Emelyanov, S.G.; Vasilyev, A.V.; Popkova, A.V.; Pavlova, A.A. Synthesis of Nanoparticles of Fe–Co–Ni Three-component Alloy Capsulated Into Carbon Matrix of Fe–Co–Ni/C Nanocomposites. J. Nano-Electron. Phys. 2016, 6, 03037. [Google Scholar] [CrossRef]
- Barabash, S.M.; Koval, Y.N. Structure and properties of metals and alloys. Kiev Nauk. Dumka 1986. (In Russian) [Google Scholar]
- Respaud, M.; Broto, J.M.; Rakoto, H.; Fert, A.R.; Thomas, L.; Barbara, B.; Verelst, M.; Snoeck, E.; Lecante, P.; Mosset, A.; et al. Surface effects on the magnetic properties of ultrafine cobalt particles. Phys. Rev. B 1998, 57, 2925. [Google Scholar] [CrossRef]
- Coey, J.M.D. Magnetism and Magnetic Materials; Cambridge University Press: Cambridge, UK, 2010. [Google Scholar]
- Kurian, M.; Thankachan, S.; Nair, D.S.; Aswathy, E.K.; Babu, A.; Thomas, A.; KT, B.K. Structural, magnetic, and acidic properties of cobalt ferrite nanoparticles synthesised by wet chemical methods. J. Adv. Ceram. 2015, 4, 199–205. [Google Scholar] [CrossRef]
- Beddow, J.K. Particulate Science and Technology; Chemical Publishing: New York, NY, USA, 1980. [Google Scholar]
- Bottoni, G.; Candolfo, D.; Cecchetti, M.; Corradi, A.R.; Masoli, F. Influence of the packing density on the coercivity of iron particles for magnetic recording. J. Magn. Magn. Mater. 1993, 120, 167–171. [Google Scholar] [CrossRef]
- Bottoni, G.; Candolfo, D.; Cecchetti, A.; Masoli, F. Interparticle interactions and magnetic parameters of iron powders for magnetic recording. J. Magn. Magn. Mater. 1992, 116, 285–290. [Google Scholar] [CrossRef]
- Ramesh, S.R.; Ramanand, A.; Ponnusamy, S.; Muthamizhchelvan, C. Synthesis and characterization of NiFe2O4 nanoparticles and nanorods. J. Alloys Compd. 2013, 563, 6–11. [Google Scholar] [CrossRef]
- Sivakumar, P.; Ramesh, R.; Ramanand, A.; Ponnusamy, S. Synthesis and characterization of NiFe2O4 nanosheet via polymer assisted co-precipitation method. Mater. Lett. 2011, 65, 483–485. [Google Scholar] [CrossRef]
- Nairan, A.; Khan, M.; Khan, U.; Iqbal, M.; Riaz, S.; Naseem, S. Temperature-dependent magnetic response of antiferromagnetic doping in cobalt ferrite nanostructures. Nanomaterials 2016, 6, 73. [Google Scholar] [CrossRef]
- Maaz, K.; Karim, S.; Mumtaz, A.; Hasanain, S.K.; Liu, J.; Duan, J.L. Synthesis and magnetic characterization of nickel ferrite nanoparticles prepared by co-precipitation route. J. Magn. Magn. Mater. 2009, 321, 1838. [Google Scholar] [CrossRef]
- Ouaissa, M.; Benyoussef, A.; Abo, G.S.; Ouaissa, S.; Hafid, M.; Belaiche, M. Effects of Crystal Fields and Exchange Interactions on Magnetic Properties of Nickel Spinel Ferrite. J. Supercond. Nov. Magn. 2015, 28, 1371–1377. [Google Scholar] [CrossRef]
- Perron, H.; Mellier, T.; Domain, C.; Roques, J.; Simoni, E.; Drot, R.; Catalette, H. Structural investigation and electronic properties of the nickel ferrite NiFe2O4: A periodic density functional theory approach. J. Phys. Condens. Mater. 2007, 19, 346219. [Google Scholar] [CrossRef]

| № | Sample | Reduction Temperature, °C | Element Composition by EDX, mass.%. | Lattice Period a, nm | Content of Co in bcc Phase, at. % | Content of Ni in fcc Phase, at.%. | |||
|---|---|---|---|---|---|---|---|---|---|
| Fe | Co | Ni | bcc | fcc | |||||
| 1 | 1Fe–Co–Ni | 300 | 32.3 | 34.9 | 32.8 | – | 0.3555 | – | 74.5 |
| 2 | 3Fe–Co–Ni | 0.2864 | 0.3568 | 30 | 62 | ||||
| 3 | 5Fe–Co–Ni | 0.2870 | – | ~20 | – | ||||
| 4 | 1Fe–Co–Ni | 400 | 57.9 | 21.2 | 20.9 | 0.2854 | 0.3554 | 52 | 74.5 |
| 5 | 3Fe–Co–Ni | 0.2864 | 0.3569 | 30 | 62 | ||||
| 6 | 5Fe–Co–Ni | 0.2868 | 0.3577 | 15 | 60 | ||||
| 7 | 1Fe–Co–Ni | 500 | 70.5 | 15.1 | 14.4 | 0.2856 | 0.3563 | 48 | 63 |
| 8 | 3Fe–Co–Ni | 0.2865 | 0.3586 | 25 | 31 | ||||
| 9 | 5Fe–Co–Ni | 0.2860 | 0.3580 | 40 | 59 | ||||
| No. | Sample | Reduction Temperature, °C | Specific Surface Area Sa, m2/g | Average Size of Particles D, nm |
|---|---|---|---|---|
| 1 | 1Fe–Co–Ni | 300 | 20.3 | 54 |
| 2 | 3Fe–Co–Ni | 24.2 | 49 | |
| 3 | 5Fe–Co–Ni | 53.3 | 36 | |
| 4 | 1Fe–Co–Ni | 400 | 5.9 | 77 |
| 5 | 3Fe–Co–Ni | 7.0 | 75 | |
| 6 | 5Fe–Co–Ni | 8.5 | 72 | |
| 7 | 1Fe–Co–Ni | 500 | 0.5 | 151 |
| 8 | 3Fe–Co–Ni | 1.2 | 135 | |
| 9 | 5Fe–Co–Ni | 1.9 | 115 |
| Sample | Reduction Temperature, °C | Magnetic State, Ferro/para | Ms, A m2 kg−1 (emu/g) | Mr, A m2 kg−1 (emu/g) | Mr/Ms | HcM, kA/m |
|---|---|---|---|---|---|---|
| 1Fe–Co–Ni | 300 | Ferromagnetic | 52.3 | 14.8 | 0.28 | 41.6 |
| 3Fe–Co–Ni | Ferromagnetic | 130.9 | 39.7 | 0.30 | 60.16 | |
| 5Fe–Co–Ni | Ferromagnetic | 84.1 | 27.0 | 0.32 | 60.64 | |
| 1Fe–Co–Ni | 400 | Ferromagnetic | 155.8 | 25.1 | 0.16 | 24.32 |
| 3Fe–Co–Ni | Ferromagnetic | 176.6 | 21.5 | 0.12 | 26.32 | |
| 5Fe–Co–Ni | Ferromagnetic | 176.6 | 17.6 | 0.1 | 21.84 | |
| 1Fe–Co–Ni | 500 | Ferromagnetic | 156.3 | 4.8 | 0.03 | 4.72 |
| 3Fe–Co–Ni | Ferromagnetic | 192.5 | 6.2 | 0.03 | 5.04 | |
| 5Fe–Co–Ni | Ferromagnetic | 185.2 | 3.7 | 0.02 | 5.92 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen, T.H.; Karunakaran, G.; Konyukhov, Y.V.; Minh, N.V.; Karpenkov, D.Y.; Burmistrov, I.N. Impact of Iron on the Fe–Co–Ni Ternary Nanocomposites Structural and Magnetic Features Obtained via Chemical Precipitation Followed by Reduction Process for Various Magnetically Coupled Devices Applications. Nanomaterials 2021, 11, 341. https://doi.org/10.3390/nano11020341
Nguyen TH, Karunakaran G, Konyukhov YV, Minh NV, Karpenkov DY, Burmistrov IN. Impact of Iron on the Fe–Co–Ni Ternary Nanocomposites Structural and Magnetic Features Obtained via Chemical Precipitation Followed by Reduction Process for Various Magnetically Coupled Devices Applications. Nanomaterials. 2021; 11(2):341. https://doi.org/10.3390/nano11020341
Chicago/Turabian StyleNguyen, Tien Hiep, Gopalu Karunakaran, Yu.V. Konyukhov, Nguyen Van Minh, D.Yu. Karpenkov, and I.N. Burmistrov. 2021. "Impact of Iron on the Fe–Co–Ni Ternary Nanocomposites Structural and Magnetic Features Obtained via Chemical Precipitation Followed by Reduction Process for Various Magnetically Coupled Devices Applications" Nanomaterials 11, no. 2: 341. https://doi.org/10.3390/nano11020341
APA StyleNguyen, T. H., Karunakaran, G., Konyukhov, Y. V., Minh, N. V., Karpenkov, D. Y., & Burmistrov, I. N. (2021). Impact of Iron on the Fe–Co–Ni Ternary Nanocomposites Structural and Magnetic Features Obtained via Chemical Precipitation Followed by Reduction Process for Various Magnetically Coupled Devices Applications. Nanomaterials, 11(2), 341. https://doi.org/10.3390/nano11020341









