Early Recognition of the PCL/Fibrous Carbon Nanocomposites Interaction with Osteoblast-like Cells by Raman Spectroscopy
Abstract
:1. Introduction
2. Materials and Methods
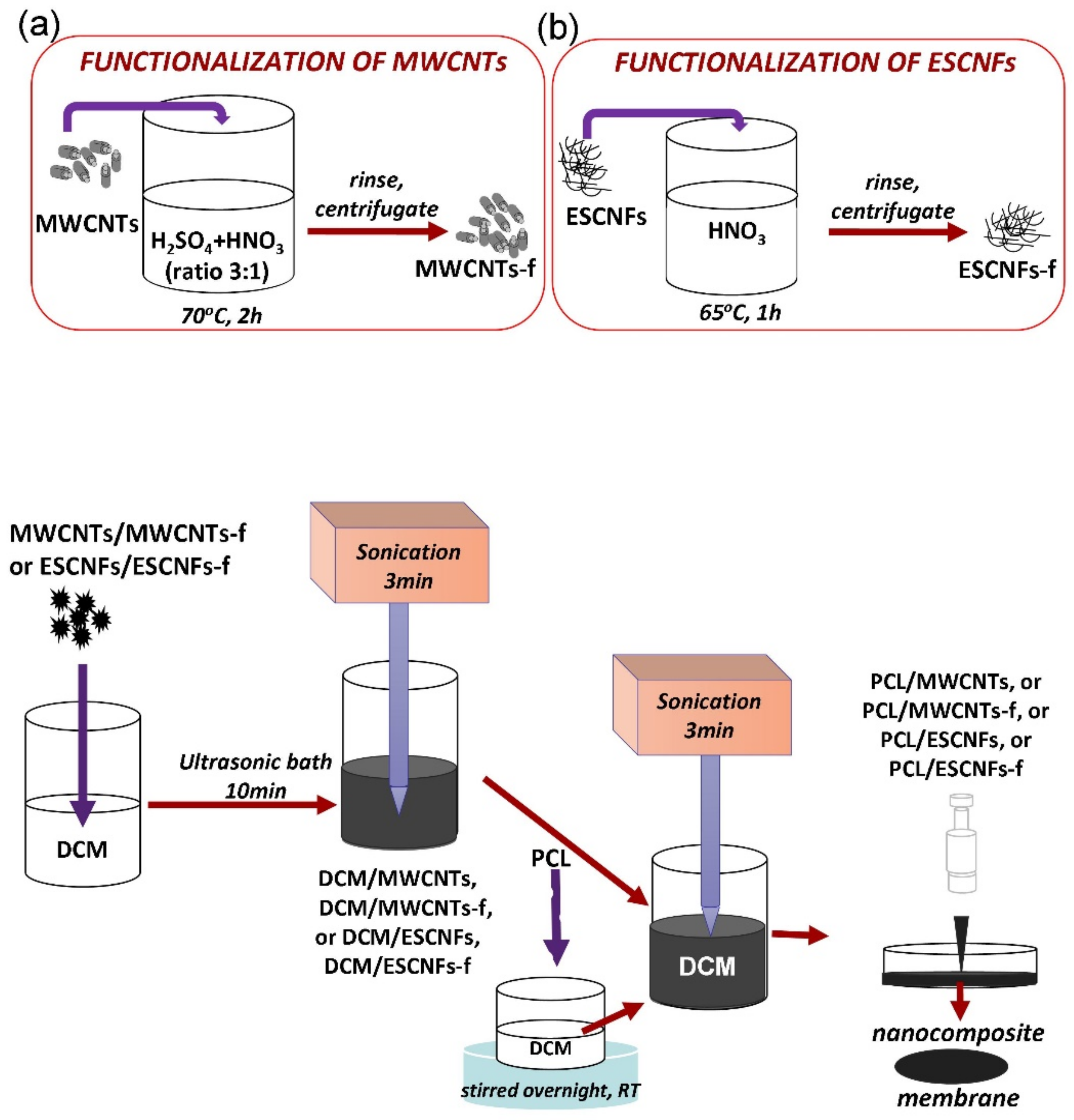
2.1. Fabrication of Nanocomposite Membranes
2.2. Contact Angle Measurements and Surface Free Energy Evaluation
2.3. Cell Culture
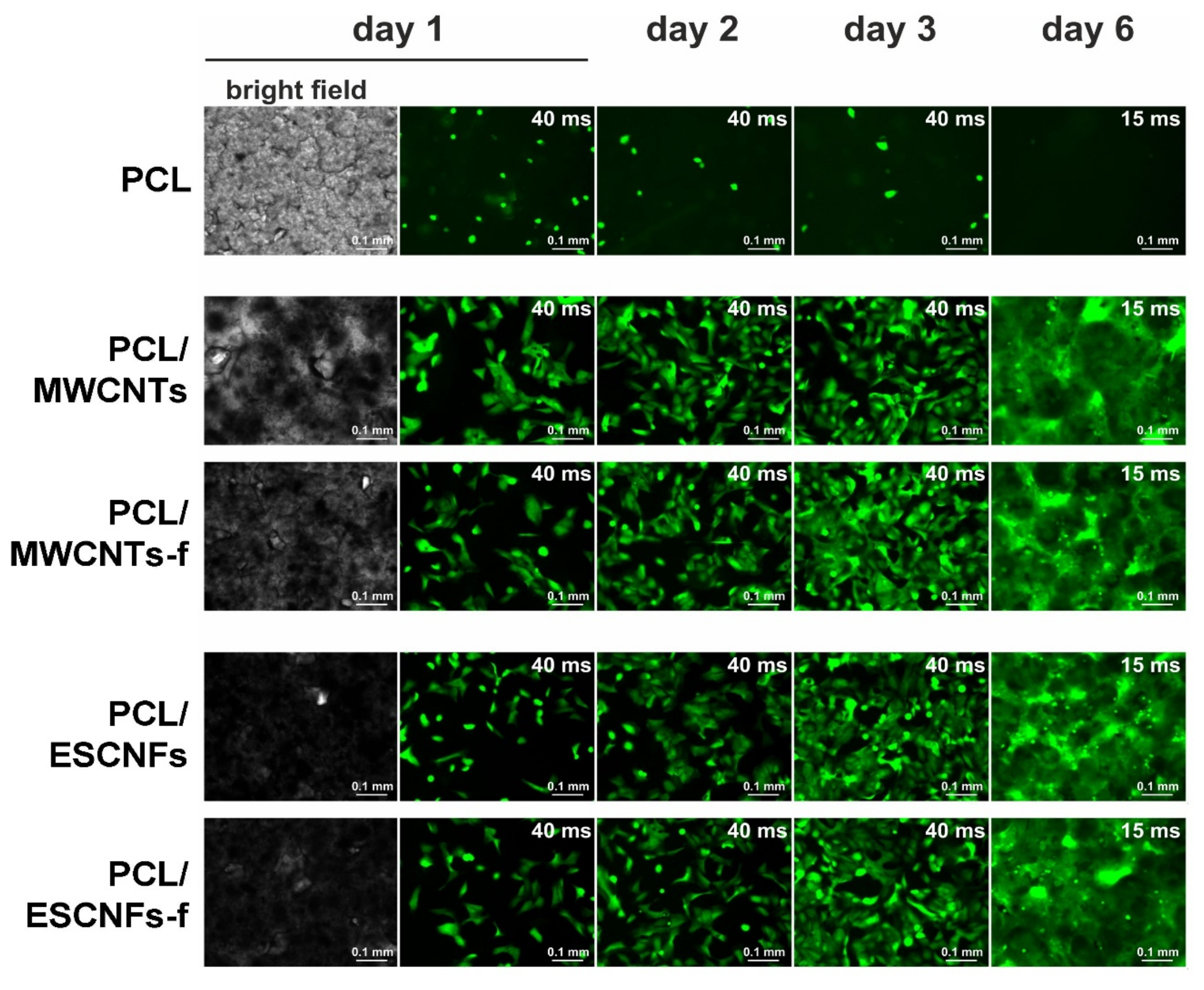
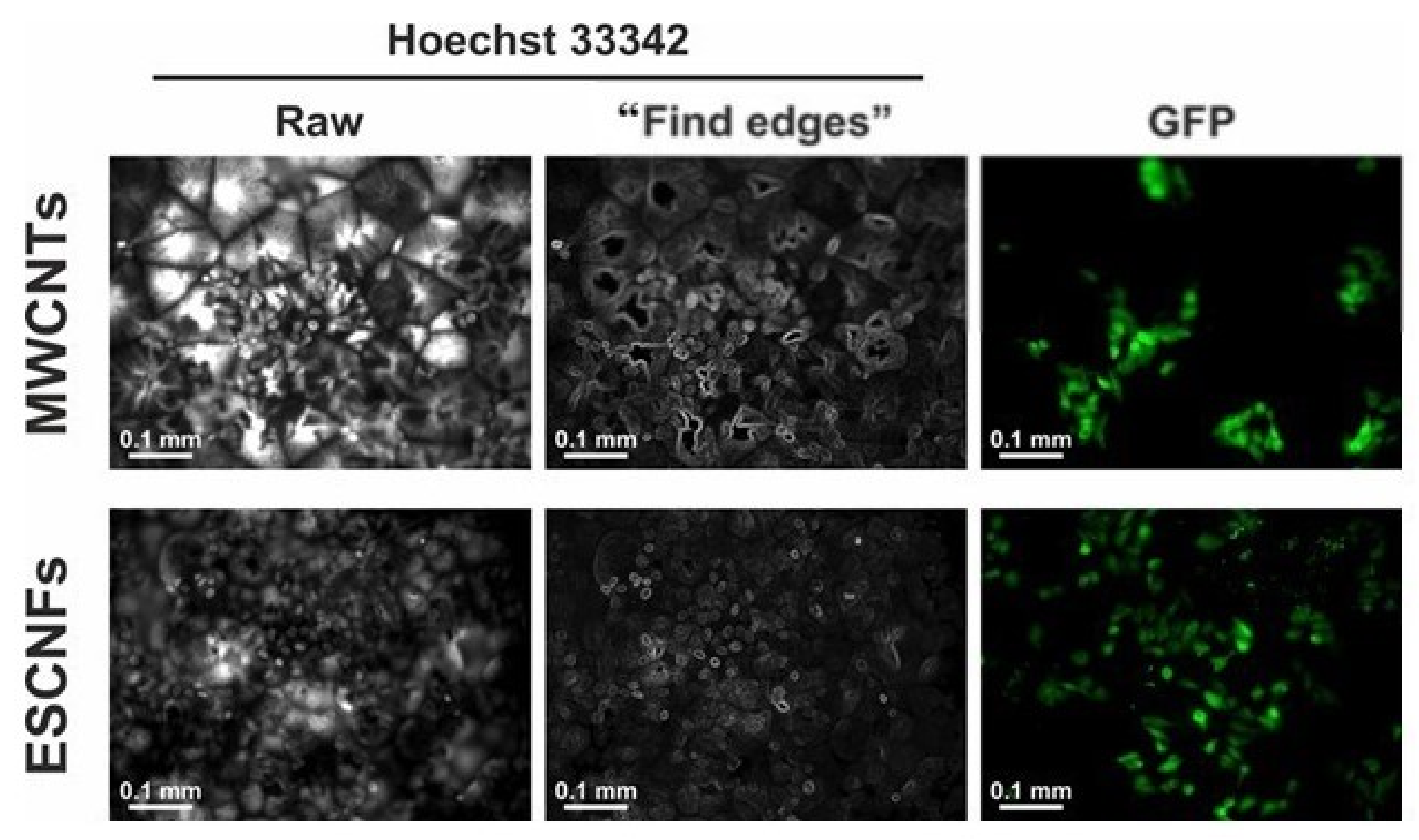
2.4. Fluorescence Microscopy
2.5. Raman Microspectroscopy
3. Results and Discussion
3.1. The Morphology of Membranes of PCL with Fibrous Carbon Nanoparticles
3.2. Contact Angle Measurements and Surface Free Energy (SFE)
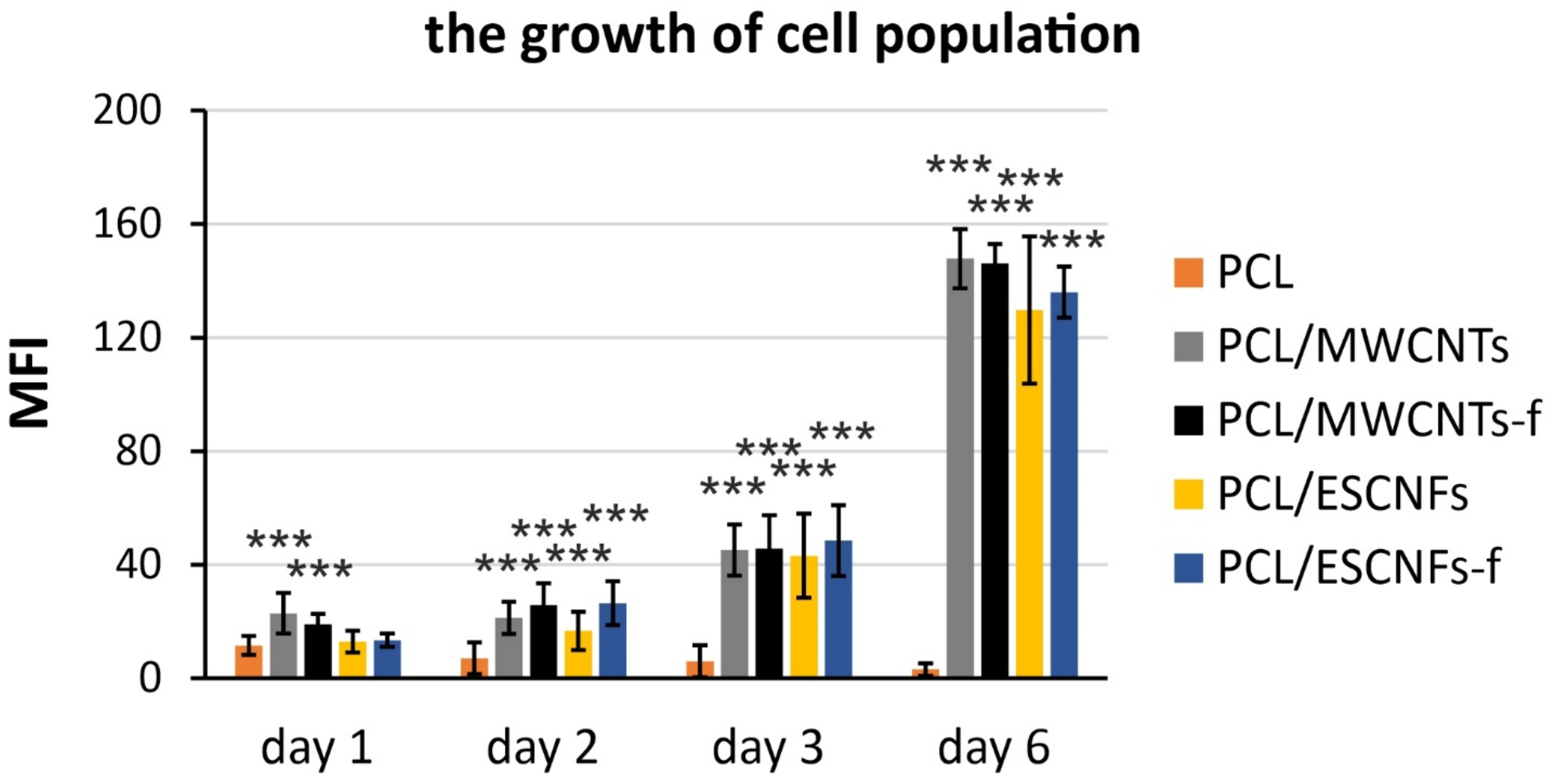
3.3. The Comparison of Growth of U-2 OS Cells on the Membranes of PCL with Fibrous Carbon Nanoparticles
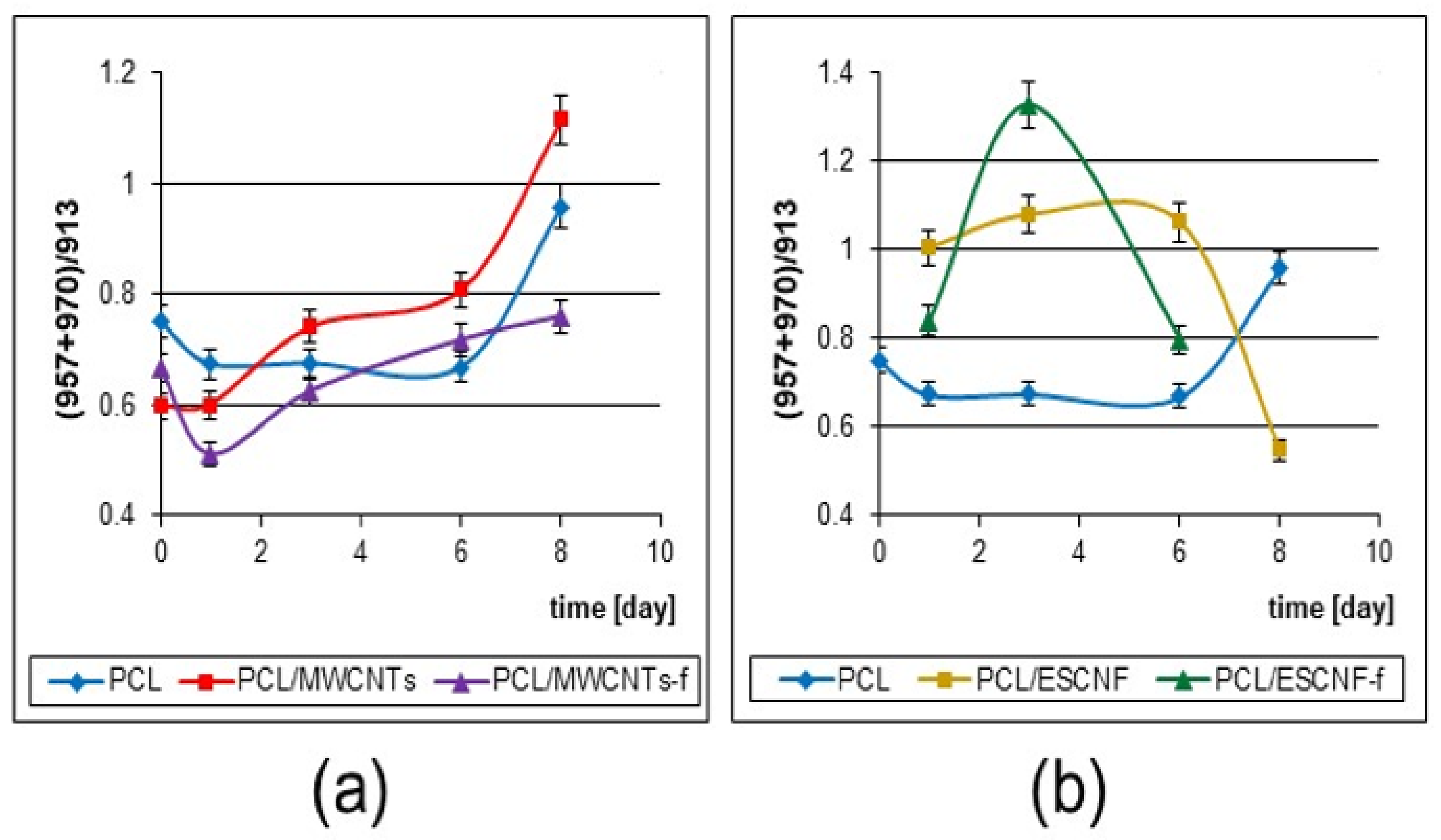
3.4. Raman Microspectroscopic Analysis of the Membranes of PCL with Fibrous Carbon Nanoparticles/Cells Interactions
3.4.1. PCL Matrix Crystallinity
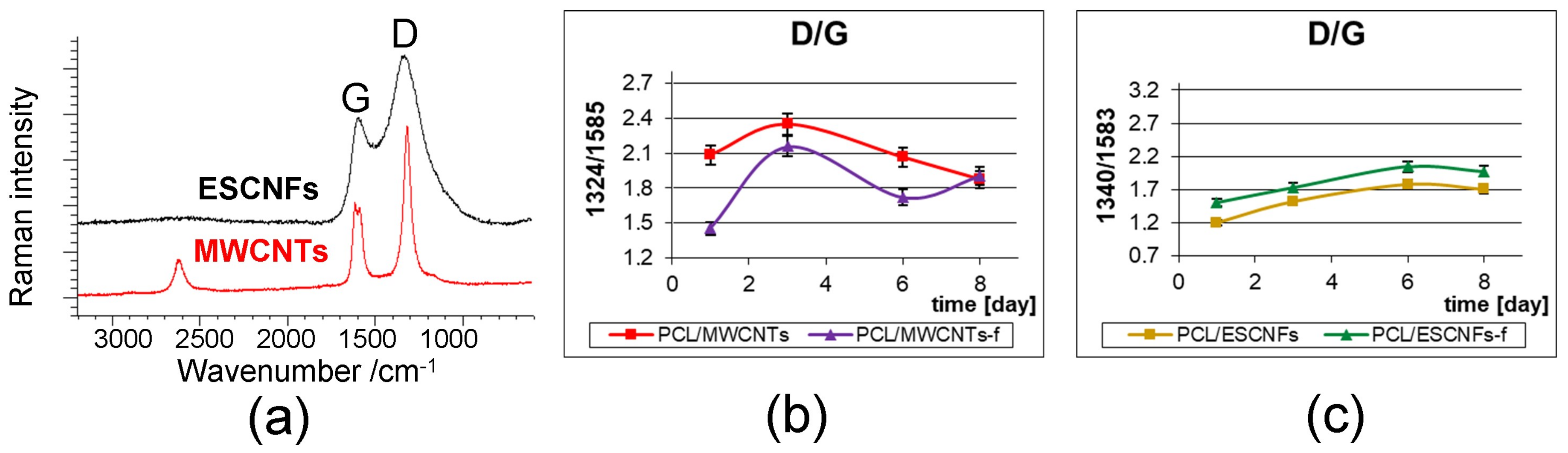
3.4.2. The Arrangement of Carbon Nanostructures
3.4.3. Raman Spectroscopy of U-2 OS Cell Development on PCL Membranes with Fibrous Carbon Nanoparticles
3.5. Morphology of U-2 OS Cells Growing on PCL Membranes with Fibrous Carbon Nanoparticles
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Daar, A.S.; Greenwood, H.L. A proposed definition of regenerative medicine. J. Tissue Eng. Regen. Med. 2007, 1, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Mason, C.; Dunnill, P. A brief definition of regenerative medicine. Regen. Med. 2008, 3, 1–5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Atala, A. Regenerative medicine strategies. J. Pediatr. Surg. 2012, 47, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Webster, T.J. Nanotechnology and nanomaterials: Promises for improved tissue regeneration. Nano Today 2009, 4, 66–80. [Google Scholar] [CrossRef]
- O’Brien, F.J. Biomaterials & scaffolds for tissue engineering. Mater. Today 2011, 14, 88–95. [Google Scholar] [CrossRef]
- Khang, D.; Carpenter, J.; Chun, Y.W.; Pareta, R.; Webster, T.J. Nanotechnology for regenerative medicine. Biomed. Microdevices 2010, 12, 575–587. [Google Scholar] [CrossRef]
- Burg, K.J.L.; Porter, S.; Kellam, J.F. Biomaterial developments for bone tissue engineering. Biomaterials 2000, 21, 2347–2359. [Google Scholar] [CrossRef]
- Roseti, L.; Parisi, V.; Petretta, M.; Cavallo, C.; Desando, G.; Bartolotti, I.; Grigolo, B. Scaffolds for Bone Tissue Engineering: State of the art and new perspectives. Mater. Sci. Eng. C 2017, 78, 1246–1262. [Google Scholar] [CrossRef]
- Farokhi, M.; Mottaghitalab, F.; Samani, S.; Shokrgozar, M.A.; Kundu, S.C.; Reis, R.L.; Fatahi, Y.; Kaplan, D.L. Silk fibroin/hydroxyapatite composites for bone tissue engineering. Biotechnol. Adv. 2018, 36, 68–91. [Google Scholar] [CrossRef]
- Karageorgiou, V.; Kaplan, D. Porosity of 3D biomaterial scaffolds and osteogenesis. Biomaterials 2005, 26, 5474–5491. [Google Scholar] [CrossRef]
- Ge, Z.; Jin, Z.; Cao, T. Manufacture of degradable polymeric scaffolds for bone regeneration. Biomed. Mater. 2008, 3, 22001. [Google Scholar] [CrossRef]
- Salinas, A.J.; Esbrit, P.; Vallet-Regí, M. A tissue engineering approach based on the use of bioceramics for bone repair. Biomater. Sci. 2013, 1, 40–51. [Google Scholar] [CrossRef]
- Rogel, M.R.; Qiu, H.; Ameer, G.A. The role of nanocomposites in bone regeneration. J. Mater. Chem. 2008, 18, 4233–4241. [Google Scholar] [CrossRef]
- Sheikh, Z.; Najeeb, S.; Khurshid, Z.; Verma, V.; Rashid, H.; Glogauer, M. Biodegradable materials for bone repair and tissue engineering applications. Materials 2015, 8, 5744–5794. [Google Scholar] [CrossRef]
- Taddei, P.; Di Foggia, M.; Causa, F.; Ambrosio, L.; Fagnano, C. In vitro bioactivity of poly(ε-caprolactone)-apatite (PCL-AP) scaffolds for bone tissue engineering: The influence of the PCL/AP ratio. Int. J. Artif. Organs 2006, 29, 719–725. [Google Scholar] [CrossRef]
- White, A.A.; Best, S.M.; Kinloch, I.A. Hydroxyapatite-carbon nanotube composites for biomedical applications: A review. Int. J. Appl. Ceram. Technol. 2007, 4, 1–13. [Google Scholar] [CrossRef]
- Paluszkiewicz, C.; Wesełucha-Birczyńska, A.; Stodolak-Zych, E.; Hasik, M. 2D IR correlation analysis of chitosan-MMT nanocomposite system. Vib. Spectrosc. 2012, 60, 185–188. [Google Scholar] [CrossRef]
- Abedalwafa, M.; Wang, F.; Wang, L.; Li, C. Biodegradable poly-epsilon-caprolactone (PCL) for tissue engineering applications: A review. Rev. Adv. Mater. Sci. 2013, 34, 123–140. [Google Scholar]
- Tyan, H.L.; Wu, C.Y.; Wei, K.H. Effect of montmorillonite on thermal and moisture absorption properties of polyimide of different chemical structures. J. Appl. Polym. Sci. 2001, 81, 1742–1747. [Google Scholar] [CrossRef]
- Wesełucha-Birczyńska, A.; Świętek, M.; Sołtysiak, E.; Galiński, P.; Płachta, Ł.; Piekara, K.; Błazewicz, M. Raman spectroscopy and the material study of nanocomposite membranes from poly(ε-caprolactone) with biocompatibility testing in osteoblast-like cells. Analyst 2015, 140, 2311–2320. [Google Scholar] [CrossRef]
- Wesełucha-Birczyńska, A.; Morajka, K.; Stodolak-Zych, E.; Długoń, E.; Dużyja, M.; Lis, T.; Gubernat, M.; Ziąbka, M.; Błażewicz, M. Raman studies of the interactions of fibrous carbon nanomaterials with albumin. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2018, 196, 262–267. [Google Scholar] [CrossRef]
- Wesełucha-Birczyńska, A.; Stodolak-Zych, E.; Piś, W.; Długoń, E.; Benko, A.; Błażewicz, M. A model of adsorption of albumin on the implant surface titanium and titanium modified carbon coatings (MWCNT-EPD). 2D correlation analysis. J. Mol. Struct. 2016, 1124, 61–70. [Google Scholar] [CrossRef]
- Tran, P.A.; Zhang, L.; Webster, T.J. Carbon nanofibers and carbon nanotubes in regenerative medicine. Adv. Drug Deliv. Rev. 2009, 61, 1097–1114. [Google Scholar] [CrossRef]
- Fraczek-Szczypta, A.; Dlugon, E.; Weselucha-Birczynska, A.; Nocun, M.; Blazewicz, M. Multi walled carbon nanotubes deposited on metal substrate using EPD technique. A spectroscopic study. J. Mol. Struct. 2013, 1040, 238–245. [Google Scholar] [CrossRef]
- Wesełucha-Birczyńska, A.; Frączek-Szczypta, A.; Długoń, E.; Paciorek, K.; Bajowska, A.; Kościelna, A.; Błażewicz, M. Application of Raman spectroscopy to study of the polymer foams modified in the volume and on the surface by carbon nanotubes. Vib. Spectrosc. 2014, 72, 50–56. [Google Scholar] [CrossRef]
- Kołodziej, A.; Długoń, E.; Świętek, M.; Ziąbka, M.; Dawiec, E.; Gubernat, M.; Michalec, M.; Wesełucha-Birczyńska, A. A Raman Spectroscopic Analysis of Polymer Membranes with Graphene Oxide and Reduced Graphene Oxide. J. Compos. Sci. 2021, 5, 20. [Google Scholar] [CrossRef]
- Hopley, E.L.; Salmasi, S.; Kalaskar, D.M.; Seifalian, A.M. Carbon nanotubes leading the way forward in new generation 3D tissue engineering. Biotechnol. Adv. 2014, 32, 1000–1014. [Google Scholar] [CrossRef]
- Khatri, Z.; Nakashima, R.; Mayakrishnan, G.; Lee, K.H.; Park, Y.H.; Wei, K.; Kim, I.S. Preparation and characterization of electrospun poly(ε-caprolactone)-poly(l-lactic acid) nanofiber tubes. J. Mater. Sci. 2013, 48, 3659–3664. [Google Scholar] [CrossRef]
- Kriparamanan, R.; Aswath, P.; Zhou, A.; Tang, L.; Nguyen, K.T. Nanotopography: Cellular responses to nanostructured materials. J. Nanosci. Nanotechnol. 2006, 6, 1905–1919. [Google Scholar] [CrossRef]
- Wesełucha-Birczyńska, A.; Moskal, P.; Dużyja, M.; Stodolak-Zych, E.; Długoń, E.; Kluska, S.; Sacharz, J.; Błażewicz, M. 2D correlation Raman spectroscopy of model micro- and nano-carbon layers in interactions with albumin, human and animal. J. Mol. Struct. 2018, 1171, 587–593. [Google Scholar] [CrossRef]
- Wesełucha-Birczyńska, A.; Stodolak-Zych, E.; Turrell, S.; Cios, F.; Krzuś, M.; Długoń, E.; Benko, A.; Niemiec, W.; Błazewicz, M. Vibrational spectroscopic analysis of a metal/carbon nanotube coating interface and the effect of its interaction with albumin. Vib. Spectrosc. 2016, 85, 185–195. [Google Scholar] [CrossRef]
- Kołodziej, A.; Wesełucha-Birczyńska, A.; Moskal, P.; Stodolak-zych, E.; Dużyja, M.; Długoń, E.; Sacharz, J.; Błażewicz, M. 2D-Raman Correlation Spectroscopy as a Method to Recognize of the Interaction at the Interface of Carbon Layer and Albumin. J. Autom. Mob. Robot. Intell. Syst. 2019, 13. [Google Scholar] [CrossRef]
- Wesełucha-Birczyńska, A.; Kołodziej, A.; Świętek, M.; Moskal, P.; Skalniak, Ł.; Długoń, E.; Błażewicz, M. Does 2D correlation Raman spectroscopy distinguish polymer nanomaterials due to the nanoaddition? J. Mol. Struct. 2020, 1217, 128342. [Google Scholar] [CrossRef]
- Kołodziej, A.; Wesełucha-Birczyńska, A.; Świętek, M.; Skalniak, Ł.; Błażewicz, M. A 2D-Raman correlation spectroscopy study of the interaction of the polymer nanocomposites with carbon nanotubes and human osteoblast-like cells interface. J. Mol. Struct. 2020, 1212. [Google Scholar] [CrossRef]
- Zaera, F. Probing liquid/solid interfaces at the molecular level. Chem. Rev. 2012, 112, 2920–2986. [Google Scholar] [CrossRef]
- Benko, A.; Przekora, A.; Wesełucha-Birczyńska, A.; Nocuń, M.; Ginalska, G.; Błażewicz, M. Fabrication of multi-walled carbon nanotube layers with selected properties via electrophoretic deposition: Physicochemical and biological characterization. Appl. Phys. A Mater. Sci. Process. 2016, 122. [Google Scholar] [CrossRef] [Green Version]
- Musiol, P.; Szatkowski, P.; Gubernat, M.; Weselucha-Birczynska, A.; Blazewicz, S. Comparative study of the structure and microstructure of PAN-based nano- and micro-carbon fibers. Ceram. Int. 2016, 42, 11603–11610. [Google Scholar] [CrossRef]
- Panek, A.; Frączek-Szczypta, A.; Długoń, E.; Nocuń, M.; Paluszkiewicz, C.; Błażewicz, M. Genotoxicity study of carbon nanoforms using a comet assay. Acta Phys. Pol. A 2018, 133, 306–308. [Google Scholar] [CrossRef]
- Niforou, K.N.; Anagnostopoulos, A.K.; Vougas, K.; Kittas, C.; Gorgoulis, V.G.; Tsangaris, G.T. The proteome profile of the human osteosarcoma U2OS cell line. Cancer Genom. Proteom. 2008, 5, 63–77. [Google Scholar]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Kołodziej, A.; Wesełucha-Birczyńska, A.; Świętek, M.; Skalniak, Ł.; Błażewicz, M. Raman microspectroscopic investigations of polymer nanocomposites: Evaluation of physical and biophysical properties. Int. J. Polym. Mater. Polym. Biomater. 2019, 68, 44–52. [Google Scholar] [CrossRef]
- Waters, J.C.; Wittmann, T. Concepts in Quantitative Fluorescence Microscopy. In Methods in Cell Biology; Wilson, L., Tran, P., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2014; ISBN 0091679X. [Google Scholar]
- Kister, G.; Cassanas, G.; Bergounhon, M.; Hoarau, D.; Vert, M. Structural characterization and hydrolytic degradation of solid copolymers of D, L-lactide-co-ε-caprolactone by Raman spectroscopy. Polymer 2000, 41, 925–932. [Google Scholar] [CrossRef]
- Kumar, S.; Rai, A.K.; Singh, V.B.; Rai, S.B. Vibrational spectrum of glycine molecule. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2005, 61, 2741–2746. [Google Scholar] [CrossRef]
- Rippon, W.B.; Koenig, J.L.; Walton, A.G. Raman Spectroscopy of Proline Oligomers and Poly-L-proline. J. Am. Chem. Soc. 1970, 92, 7455–7459. [Google Scholar] [CrossRef]
- Gelse, K.; Pöschl, E.; Aigner, T. Collagens-Structure, function, and biosynthesis. Adv. Drug Deliv. Rev. 2003, 55, 1531–1546. [Google Scholar] [CrossRef] [Green Version]
- Khoshnoodi, J.; Pedchenko, V.; Hudson, B.G. Mammalian collagen IV. Microsc. Res. Tech. 2008, 71, 357–370. [Google Scholar] [CrossRef] [Green Version]
- Mizuno, K.; Adachi, E.; Imamura, Y.; Katsumata, O.; Hayashi, T. The fibril structure of type V collagen triple-helical domain. Micron 2001, 32, 317–323. [Google Scholar] [CrossRef]
- Bächinger, H.P.; Mizuno, K.; Vranka, J.A.; Boudko, S.P. Collagen Formation and Structure. In Comprehensive Natural Products II: Chemistry and Biology; Elsevier Ltd.: Amsterdam, The Netherlands, 2010; Volume 5, ISBN 9780080453828. [Google Scholar]
- Bogin, O.; Kvansakul, M.; Rom, E.; Singer, J.; Yayon, A.; Hohenester, E. Insight into Schmid metaphyseal chondrodysplasia from the crystal structure of the collagen X NC1 domain trimer. Structure 2002, 10, 165–173. [Google Scholar] [CrossRef] [Green Version]
- Tu Raman Spectroscopy in Biology: Principles and Applications; John Wiley & Sons, Ltd: New York, NY, USA, 1982.
- Zhu, G.; Zhu, X.; Fan, Q.; Wan, X. Raman spectra of amino acids and their aqueous solutions. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2011, 78, 1187–1195. [Google Scholar] [CrossRef]
- Dresselhaus, M.S.; Dresselhaus, G.; Saito, R.; Jorio, A. Raman spectroscopy of carbon nanotubes. Phys. Rep. 2005, 409, 47–99. [Google Scholar] [CrossRef]
- Wesełucha-Birczyńska, A.; Babeł, K.; Jurewicz, K. Carbonaceous materials for hydrogen storage investigated by 2D Raman correlation spectroscopy. Vib. Spectrosc. 2012, 60, 206–211. [Google Scholar] [CrossRef]
- Rehman, I.U.; Movasaghi, Z.; Rehman, S. Vibrational Spectroscopy for Tissue Analysis, 1st ed.; CRC Press: Boca Raton, FL, USA, 2012; ISBN 9780429106118. [Google Scholar] [CrossRef]
- Tuinstra, F.; Koenig, J.L. Raman Spectrum of Graphite. J. Chem. Phys. 1970, 53, 1126–1130. [Google Scholar] [CrossRef] [Green Version]
- Wesełucha-Birczyńska, A.; Długoń, E.; Kołodziej, A.; Bilska, A.; Sacharz, J.; Błażewicz, M. Multi-wavelength Raman microspectroscopic studies of modified monwoven carbon scaffolds for tissue engineering applications. J. Mol. Struct. 2020, 1220, 128665. [Google Scholar] [CrossRef]
- Pautke, C.; Schieker, M.; Tischer, T.; Kolk, A.; Neth, P.; Mutschler, W.; Milz, S. Characterization of Osteosarcoma Cell Lines MG-63, Saos-2 and U-2 OS in Comparison to Human Osteoblasts. Anticancer Res. 2004, 24, 3743–3748. [Google Scholar]
- Shoulders, M.D.; Raines, R.T. Collagen structure and stability. Annu. Rev. Biochem. 2009, 78, 929–958. [Google Scholar] [CrossRef] [Green Version]
- Woodruff, M.A.; Hutmacher, D.W. The return of a forgotten polymer—Polycaprolactone in the 21st century. Prog. Polym. Sci. 2010, 35, 1217–1256. [Google Scholar] [CrossRef] [Green Version]



| Material | Contact Angle for Diiodomethane [°] | Surface Free Energy [mN/m] | Disperse Part [mN/m] | Polar Part [mN/m] | ||||
|---|---|---|---|---|---|---|---|---|
| Value | StDev | Value | StDev | Value | StDev | Value | StDev | |
| PCL | 29.31 | 2.76 | 45.83 | 1.42 | 44.51 | 1.12 | 1.32 | 0.29 |
| PCL/MWCNTs | 26.58 | 4.80 | 47.66 | 2.36 | 45.57 | 1.80 | 2.09 | 0.56 |
| PCL/MWCNTs-f | 25.98 | 3.07 | 46.70 | 1.60 | 45.83 | 1.13 | 0.88 | 0.48 |
| PCL/ESCNFs | 29.31 | 3.20 | 45.93 | 1.62 | 44.51 | 1.30 | 1.43 | 0.32 |
| PCL/ESCNFs-f | 35.71 | 3.02 | 42.88 | 2.02 | 41.70 | 1.42 | 1.19 | 0.60 |
| Raman Bands [cm−1] | Assignment | ||||
|---|---|---|---|---|---|
| PCL | PCL/ MWCNTs | PCL/ MWCNTs-f | PCL/ ESCNFs | PCL/ ESCNFs-f | |
| 712 ± 1 | 712 ± 1 | 713 ± 2 | 713 ± 1 | 713 ± 2 | δ(CH2), δ(NH2), Gly; CS, Cys [44,45,46,47,48,49,50,51] |
| 865 ± 1 | 862 ± 1 | 862 ± 2 | 861 ± 1 | 861 ± 2 | ν(C-COO) PCL (amorph); o.o.p. δ(CH2), Pro; collagen [20,43,45,52] |
| 913 ± 1 | 913 ± 1 | 912 ± 1 | 912 ± 1 | 912 ± 1 | ν(C-COO), PCL (cryst); τ(CH2)&τ(NH2), Gly; collagen [20,43,44,52] |
| 958 ± 1 | 958 ± 1 | 957 ± 1 | 958 ± 1 | 958 ± 1 | ν(C-COO), PCL; ring str., Pro [20,43,45] |
| 1038 ± 1 | 1038 ± 1 | 1037 ± 1 | 1038 ± 1 | 1037 ± 1 | ν(COC), PCL; ω(CH2), Pro; ν(CN)&ν(CC), Gly [20,43,44,45] |
| 1064 ± 1 | 1064 ± 1 | 1064 ± 1 | 1064 ± 1 | 1064 ± 1 | ν(COC), PCL (amorph) [20,43] |
| 1109 ± 1 | 1109 ± 1 | 1108 ± 1 | 1109 ± 1 | 1108 ± 1 | ν(COC), PCL (cryst); collagen [20,43,52] |
| 1284 ± 1 | 1284 ± 1 | 1284 ± 1 | 1283 ± 1 | 1283 ± 1 | ω(CH2), PCL (cyst); δ(CH2), Pro [20,43,45] |
| 1305 ± 1 | 1305 ± 1 | 1305 ± 1 | 1305 ± 1 | 1304 ± 1 | ω(CH2), PCL (cryst and amorph) [20,43] |
| - | 1323 ± 1 | 1323 ± 1 | 1340 ± 2 | 1341 ± 2 | D1-disorder-induced A1g mode in graphite plane; δ(CH2), Pro [45,53,54] |
| 1418 ± 1 | 1418 ± 1 | 1418 ± 1 | 1418 ± 1 | 1418 ± 2 | δ(CH2), PCL; γ(CH2)), Gly [20,43,44,52] |
| 1441 ± 1 | 1441 ± 1 | 1441 ± 1 | 1441 ± 1 | 1441 ± 1 | δ(CH2), PCL (cryst.); δ(CH2), Pro [20,43,45] |
| 1469 ± 1 | 1468 ± 1 | 1467 ± 1 | 1466 ± 1 | 1470 ± 1 | δ(CH2)), PCL; collagen [20,43,52] |
| - | 1587 ± 1 | 1585 ± 1 | 1584 ± 1 | 1585 ± 1 | corresponding to G-graphite tangential mode [30,31,53] |
| - | 1615 ± 1 | 1614 ± 1 | 1615 ± 1 | 1614 ± 1 | D2-band due to due to in-plane defects and heteroatoms [54] |
| 1723 ± 1 | 1723 ± 1 | 1723 ± 1 | 1724 ± 1 | 1723 ± 1 | ν(C=O), PCL (cryst) [20,43] |
| 1732 ± 1 | 1733 ± 1 | 1733 ± 1 | 1732 ± 1 | 1723 ± 1 | ν(C=O), PCL (amorph) [20,43] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wesełucha-Birczyńska, A.; Kołodziej, A.; Świętek, M.; Skalniak, Ł.; Długoń, E.; Pajda, M.; Błażewicz, M. Early Recognition of the PCL/Fibrous Carbon Nanocomposites Interaction with Osteoblast-like Cells by Raman Spectroscopy. Nanomaterials 2021, 11, 2890. https://doi.org/10.3390/nano11112890
Wesełucha-Birczyńska A, Kołodziej A, Świętek M, Skalniak Ł, Długoń E, Pajda M, Błażewicz M. Early Recognition of the PCL/Fibrous Carbon Nanocomposites Interaction with Osteoblast-like Cells by Raman Spectroscopy. Nanomaterials. 2021; 11(11):2890. https://doi.org/10.3390/nano11112890
Chicago/Turabian StyleWesełucha-Birczyńska, Aleksandra, Anna Kołodziej, Małgorzata Świętek, Łukasz Skalniak, Elżbieta Długoń, Maria Pajda, and Marta Błażewicz. 2021. "Early Recognition of the PCL/Fibrous Carbon Nanocomposites Interaction with Osteoblast-like Cells by Raman Spectroscopy" Nanomaterials 11, no. 11: 2890. https://doi.org/10.3390/nano11112890
APA StyleWesełucha-Birczyńska, A., Kołodziej, A., Świętek, M., Skalniak, Ł., Długoń, E., Pajda, M., & Błażewicz, M. (2021). Early Recognition of the PCL/Fibrous Carbon Nanocomposites Interaction with Osteoblast-like Cells by Raman Spectroscopy. Nanomaterials, 11(11), 2890. https://doi.org/10.3390/nano11112890






