Nanoparticles for Cerenkov and Radioluminescent Light Enhancement for Imaging and Radiotherapy
Abstract
1. Introduction
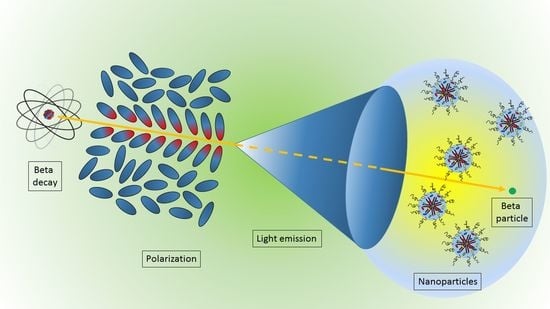
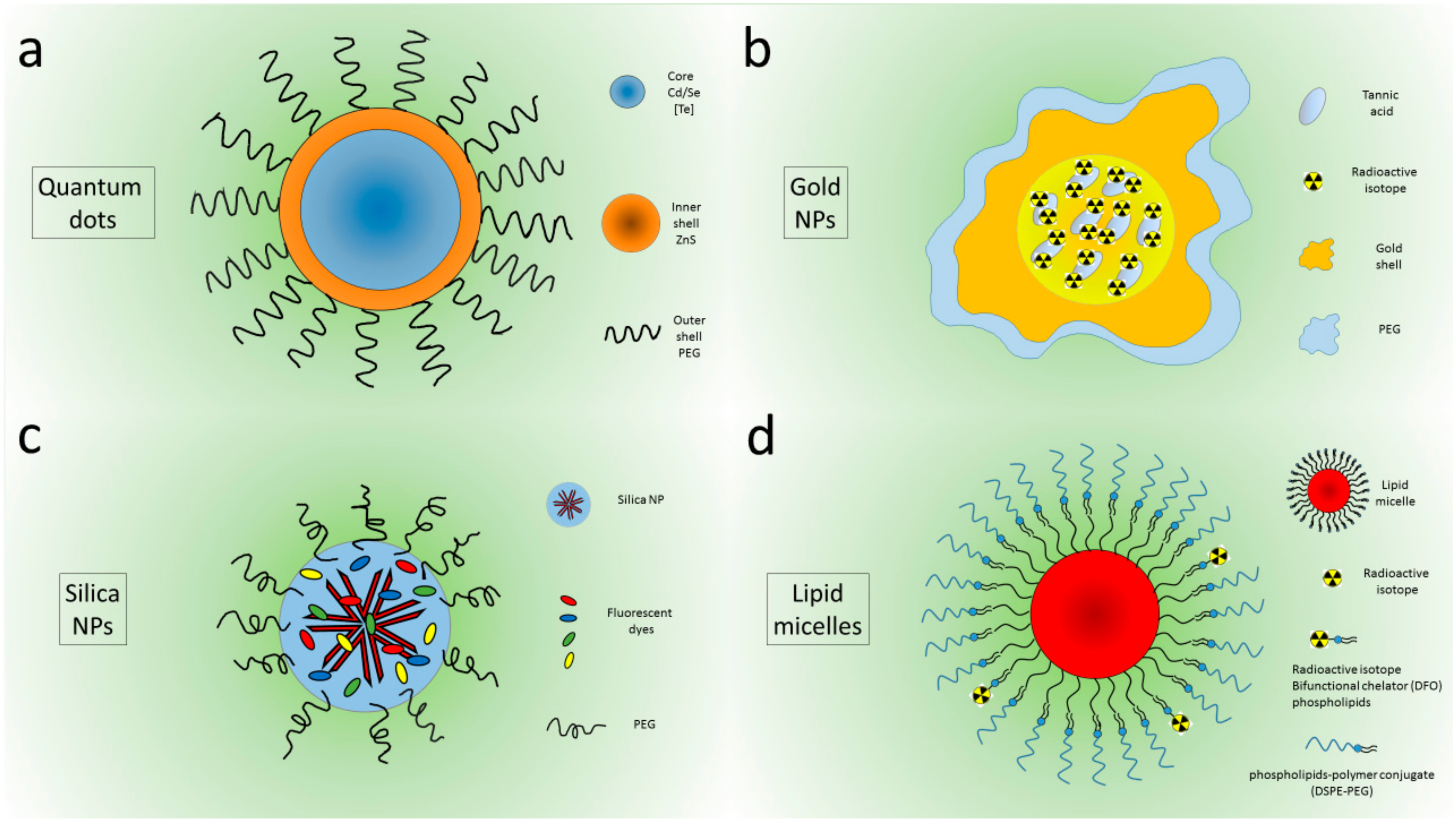
2. Physics of Cerenkov Light Production
3. Nanoparticles for Imaging and Therapy Using Cerenkov Sources
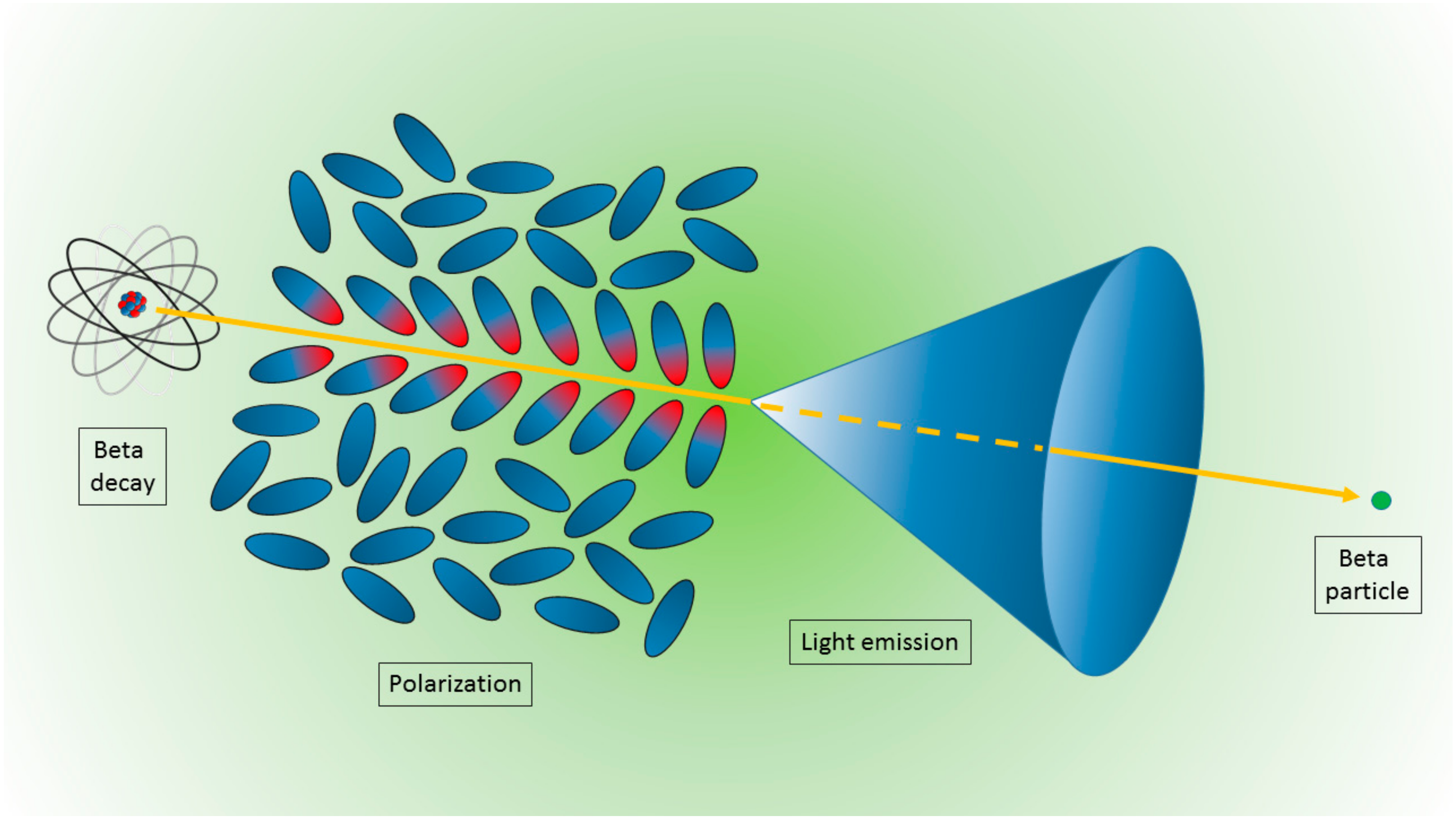
3.1. Nanoparticles
3.2. Toxicity
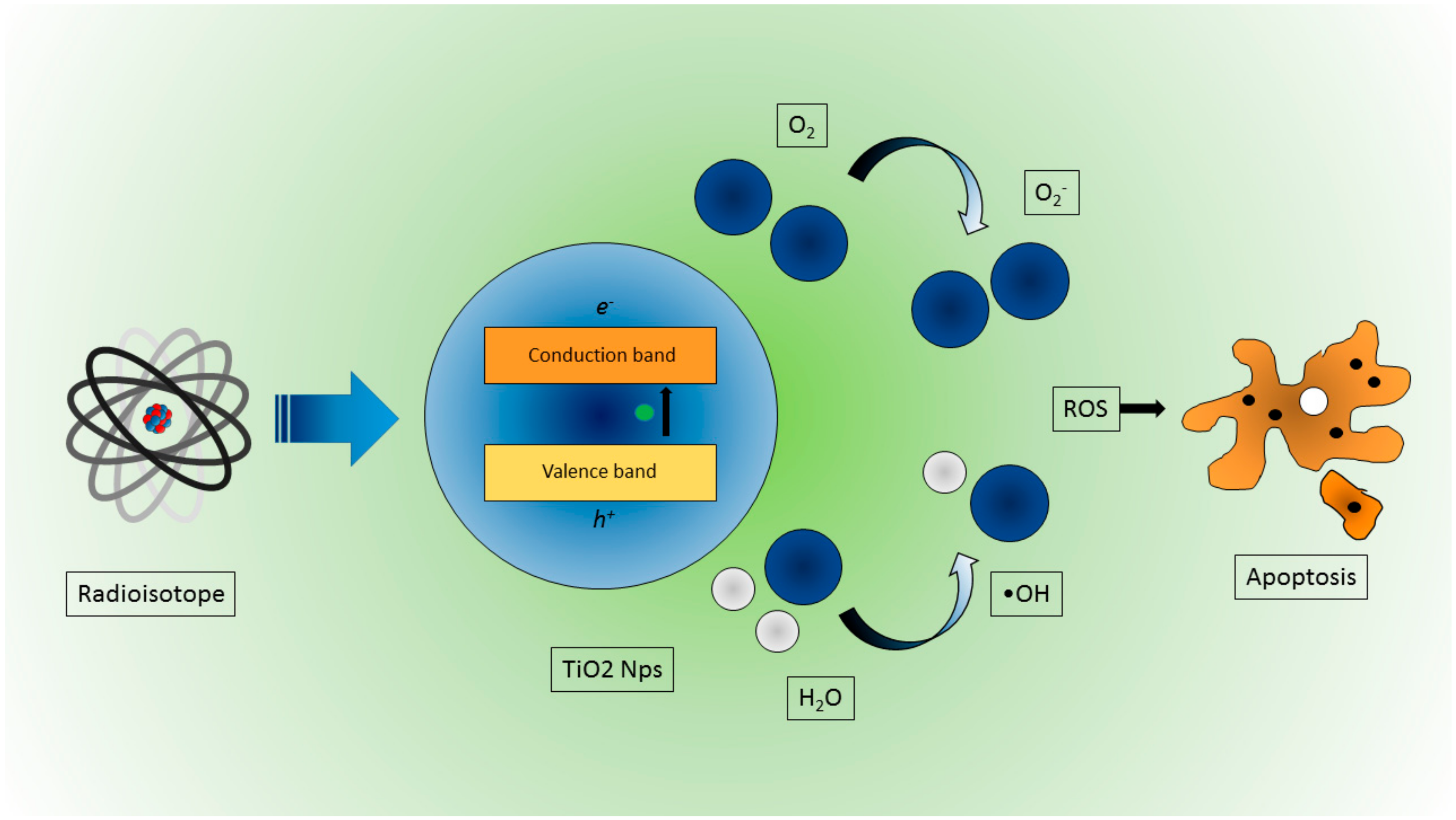
3.3. Radioisotope/Nanoparticle Interaction
3.4. Optical and Chemical Properties
3.5. Applications to Cancer Models
3.6. CR Sources
3.7. Imaging Techniques
4. Photodynamic Therapy Using Cerenkov Sources
5. Quick Overview
6. Discussion and Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Xu, Y.; Liu, H.; Cheng, Z. Harnessing the power of radionuclides for optical imaging: Cerenkov luminescence imaging. J. Nucl. Med. 2011, 52, 2009–2018. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Wang, J.; Cheng, Z. Cerenkov radiation: A multi-functional approach for biological sciences. Front. Phys. 2014, 2, 1–14. [Google Scholar] [CrossRef]
- Spinelli, A.E.; Boschi, F. Novel biomedical applications of Cerenkov radiation and radioluminescence imaging. Phys. Med. 2015, 31, 120–129. [Google Scholar] [CrossRef] [PubMed]
- Tanha, K.; Pashazadeh, A.M.; Pogue, B.W. Review of biomedical Čerenkov luminescence imaging applications. Biomed. Opt. Express 2015, 6, 3053–3065. [Google Scholar] [CrossRef]
- Shaffer, T.M.; Pratt, E.C.; Grimm, J. Utilizing the power of Cerenkov light with nanotechnology. Nat. Nanotechnol. 2017, 12, 106–117. [Google Scholar] [CrossRef]
- Jacques, S.L. Optical properties of biological tissues: A review. Phys. Med. Biol. 2013, 58, R37. [Google Scholar] [CrossRef]
- Jelley, J.V. Cerenkov radiation and its applications. Br. J. Appl. Phys. 1955, 6, 227–232. [Google Scholar] [CrossRef]
- Spinelli, A.E.; Marengo, M.; Calandrino, R.; Sbarbati, A.; Boschi, F. Optical imaging of radioisotopes: A novel multimodal approach to molecular imaging. Q. J. Nucl. Med. Mol. Imaging 2012, 56, 280–290. [Google Scholar]
- Alivisatos, A.P. Semiconductor clusters, nanocrystals, and quantum dots. Science 1996, 271, 933–937. [Google Scholar] [CrossRef]
- Boschi, F.; De Sanctis, F. Overview of the optical properties of fluorescent nanoparticles for optical imaging. Eur. J. Histochem. 2017, 61, 2830. [Google Scholar] [CrossRef]
- Liu, H.; Ren, G.; Miao, Z.; Zhang, X.; Tang, X.; Han, P.; Gambhir, S.S.; Sanjiv, S.; Cheng, Z. Molecular Optical Imaging with Radioactive Probes. PLoS ONE 2010, 5, e9470. [Google Scholar] [CrossRef] [PubMed]
- Dothager, R.S.; Goiffon, R.J.; Jackson, E.; Harpstrite, S.; Piwnica-Worms, D. Cerenkov radiation energy transfer (CRET) imaging: A novel method for optical imaging of PET isotopes in biological systems. PLoS ONE 2010, 5, e13300. [Google Scholar] [CrossRef] [PubMed]
- Boschi, F.; Spinelli, A.E. Quantum dots excitation using pure beta minus radioisotopes emitting Cerenkov radiation. RSC Adv. 2012, 2, 11049–11052. [Google Scholar] [CrossRef]
- Thorek, D.L.J.; Ogirala, A.; Beattie, B.J.; Grimm, J. Quantitative imaging of disease signatures through radioactive decay signal conversion. Nat. Med. 2013, 19, 1345–1350. [Google Scholar] [CrossRef]
- Sun, X.; Huang, X.; Guo, J.; Zhu, W.; Ding, Y.; Niu, G.; Wang, A.; Kiesewetter, D.O.; Wang, Z.L.; Sun, S.; et al. Self-illuminating 64Cu-Doped CdSe/ZnS nanocrystals for in vivo tumor imaging. J. Am. Chem. Soc. 2014, 136, 1706–1709. [Google Scholar] [CrossRef]
- Zhao, Y.; Shaffer, T.M.; Das, S.; Pérez-Medina, C.; Mulder, W.J.M.; Grimm, J. Near-Infrared Quantum Dot and 89Zr Dual-Labeled Nanoparticles for in Vivo Cerenkov Imaging. Bioconjugate Chem. 2017, 28, 600–608. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, Y.; Luehmann, H.; Xia, X.; Wan, D.; Cutler, C.; Xia, Y. Radioluminescent gold nanocages with controlled radioactivity for real-time in vivo imaging. Nano Lett. 2013, 13, 581–585. [Google Scholar] [CrossRef]
- Hu, H.; Huang, P.; Weiss, O.J.; Yan, X.; Yue, X.; Zhang, M.G.; Tang, Y.; Nie, L.; Ma, Y.; Niu, G.; et al. PET and NIR optical imaging using self-illuminating 64Cu-doped chelator-free gold nanoclusters. Biomaterials 2014, 35, 9868–9876. [Google Scholar] [CrossRef]
- Black, K.C.L.; Wang, Y.; Luehmann, H.P.; Cai, X.; Xing, W.; Pang, B.; Zhao, Y.; Cutler, C.S.; Wang, L.V.; Liu, Y.; et al. Radioactive 198Au-doped nanostructures with different shapes for in vivo analyses of their biodistribution, tumor uptake, and intratumoral distribution. ACS Nano 2014, 8, 4385–4394. [Google Scholar] [CrossRef]
- Volotskova, O.; Sun, C.; Stafford, J.H.; Koh, A.L.; Ma, X.; Cheng, Z.; Cui, B.; Pratx, G.; Xing, L. Efficient Radioisotope Energy Transfer by Gold Nanoclusters for Molecular Imaging. Small 2015, 11, 4002–4008. [Google Scholar] [CrossRef]
- Lee, S.B.; Yoon, G.; Lee, S.W.; Jeong, S.Y.; Ahn, B.C.; Lim, D.K.; Lee, J.; Jeon, Y.H. Combined Positron Emission Tomography and Cerenkov Luminescence Imaging of Sentinel Lymph Nodes Using PEGylated Radionuclide-Embedded Gold Nanoparticles. Small 2016, 12, 4894–4901. [Google Scholar] [CrossRef]
- Lee, S.B.; Li, Y.; Lee, I.K.; Cho, S.J.; Kim, S.K.; Lee, S.W.; Lee, J.; Jeon, Y.H. In vivo detection of sentinel lymph nodes with PEGylated crushed gold shell @ radioactive core nanoballs. J. Ind. Eng. Chem. 2019, 70, 196–203. [Google Scholar] [CrossRef]
- Kamkaew, A.; Cheng, L.; Goel, S.; Valdovinos, H.F.; Barnhart, T.E.; Liu, Z.; Cai, W. Cerenkov Radiation Induced Photodynamic Therapy Using Chlorin e6-Loaded Hollow Mesoporous Silica Nanoparticles. ACS Appl. Mater. Interfaces 2016, 8, 26630–26637. [Google Scholar] [CrossRef] [PubMed]
- Goel, S.; Ferreira, C.A.; Chen, F.; Ellison, P.A.; Siamof, C.M.; Barnhart, T.E.; Cai, W. Activatable Hybrid Nanotheranostics for Tetramodal Imaging and Synergistic Photothermal/Photodynamic Therapy. Adv. Mater. 2017, 30, 1704367. [Google Scholar] [CrossRef]
- Pratt, E.C.; Shaffer, T.M.; Zhang, Q.; Drain, C.M.; Grimm, J. Nanoparticles as multimodal photon transducers of ionizing radiation. Nat. Nanotechnol. 2018, 13, 418–426. [Google Scholar] [CrossRef] [PubMed]
- Genovese, D.; Petrizza, L.; Prodi, L.; Rampazzo, E.; De Sanctis, F.; Spinelli, A.E.; Boschi, F.; Zaccheroni, N. Tandem Dye-Doped Nanoparticles for NIR Imaging via Cerenkov Resonance Energy Transfer. Front Chem. 2020, 8, 71. [Google Scholar] [CrossRef] [PubMed]
- Satterlee, A.B.; Yuan, H.; Huang, L. A radio-theranostic nanoparticle with high specific drug loading for cancer therapy and imaging. J. Control. Release 2015, 217, 170–182. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.Y.; Chen, C.C.; Lin, L.T.; Chang, C.H.; Chen, L.C.; Wang, H.E.; Lee, T.W.; Lee, Y.J. PEGylated liposome-encapsulated rhenium-188 radiopharmaceutical inhibits proliferation and epithelial–mesenchymal transition of human head and neck cancer cells in vivo with repeated therapy. Cell Death Discov. 2018, 4, 100. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.; Pratx, G.; Carpenter, C.M.; Liu, H.; Cheng, Z.; Gambhir, S.S.; Xing, L. Synthesis and Radioluminescence of PEGylated Eu3+-doped Nanophosphors as Bioimaging Probes. Adv. Mater. 2011, 23, H195–H199. [Google Scholar] [CrossRef]
- Carpenter, C.M.; Sun, C.; Pratx, G.; Liu, H.; Cheng, Z.; Xing, L. Radioluminescent nanophosphors enable multiplexed small-animal imaging. Opt. Express 2012, 20, 11598–11604. [Google Scholar] [CrossRef]
- Cao, X.; Chen, X.; Kang, F.; Cao, X.; Zhan, Y.; Wang, J.; Wu, K.; Liang, J. Sensitivity improvement of Cerenkov luminescence endoscope with terbium doped Gd2O2S nanoparticles. Appl. Phys. Lett. 2015, 106, 213702. [Google Scholar] [CrossRef]
- Hu, Z.; Qu, Y.; Wang, K.; Zhang, X.; Zha, J.; Song, T.; Bao, C.; Liu, H.; Wang, Z.; Wang, J.; et al. In vivo nanoparticle-mediated radiopharmaceutical-excited fluorescence molecular imaging. Nat. Commun. 2015, 6, 7560. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Zhao, M.; Qu, Y.; Zhang, X.; Zhang, M.; Liu, M.; Guo, H.; Zhang, Z.; Wang, J.; Yang, W.; et al. In Vivo 3-dimensional radiopharmaceutical-excited fluorescence tomography. J. Nucl. Med. 2017, 58, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Chi, C.; Liu, M.; Guo, H.; Zhang, Z.; Zeng, C.; Ye, J.; Wang, J.; Tian, J.; Yang, W.; et al. Nanoparticle-mediated radiopharmaceutical-excited fluorescence molecular imaging allows precise image-guided tumor-removal surgery. Nanomed. Nanotechnol. Biol. Med. 2017, 13, 1323–1331. [Google Scholar] [CrossRef]
- Liu, N.; Shi, J.; Wang, Q.; Guo, J.; Hou, Z.; Su, X.; Zhang, H.; Sun, X. In Vivo Repeatedly Activated Persistent Luminescence Nanoparticles by Radiopharmaceuticals for Long-Lasting Tumor Optical Imaging. Small 2020, 16, 2001494. [Google Scholar] [CrossRef]
- Black, K.C.L.; Ibricevic, A.; Gunsten, S.P.; Flores, J.A.; Gustafson, T.P.; Raymond, J.E.; Samarajeewa, S.; Shrestha, R.; Felder, S.E.; Cai, T.; et al. In vivo fate tracking of degradable nanoparticles for lung gene transfer using PET and Ĉerenkov imaging. Biomaterials 2016, 98, 53–63. [Google Scholar] [CrossRef] [PubMed]
- Duan, D.; Liu, H.; Xu, Y.; Han, Y.; Xu, M.; Zhang, Z.; Liu, Z. Activating TiO2 Nanoparticles: Gallium-68 Serves as a High-Yield Photon Emitter for Cerenkov-Induced Photodynamic Therapy. ACS Appl. Mater. Interfaces 2018, 10, 5278–5286. [Google Scholar] [CrossRef]
- Lee, S.B.; Kumar, D.; Li, Y.; Lee, I.K.; Cho, S.J.; Kim, S.K.; Lee, S.W.; Jeong, S.Y.; Lee, J.; Jeon, Y.H. PEGylated crushed gold shell-radiolabeled core nanoballs for in vivo tumor imaging with dual positron emission tomography and Cerenkov luminescent imaging. J. Nanobiotechnol. 2018, 16, 41. [Google Scholar] [CrossRef]
- Ferreira, C.A.; Ni, D.; Rosenkrans, Z.T.; Cai, W. Radionuclide-Activated Nanomaterials and Their Biomedical Applications. Angew. Chem. Int. Ed. 2019, 58, 13232–13252. [Google Scholar] [CrossRef]
- Park, J.C.; Yu, M.K.; An, G.I.; Park, S.-I.; Oh, J.; Kim, H.J.; Kim, J.H.; Wang, E.K.; Hong, I.H.; Ha, Y.S.; et al. Facile preparation of a hybrid nanoprobe for triple-modality optical/PET/MR imaging. Small 2010, 6, 2863–2868. [Google Scholar] [CrossRef]
- Ni, D.; Ferreira, C.A.; Barnhart, T.E.; Quach, V.; Yu, B.; Jiang, D.; Wei, W.; Liu, H.; Engle, J.W.; Hu, P.; et al. Magnetic Targeting of Nanotheranostics Enhances Cerenkov Radiation-Induced Photodynamic Therapy. J. Am. Chem. Soc. 2018, 140, 14971–14979. [Google Scholar] [CrossRef] [PubMed]
- Thorek, D.L.; Das, S.; Grimm, J. Molecular imaging using nanoparticle quenchers of Cerenkov luminescence. Small 2014, 10, 3729–3734. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kotagiri, N.; Sudlow, G.P.; Akers, W.J.; Achilefu, S. Breaking the depth dependency of phototherapy with Cerenkov radiation and low-radiance-responsive nanophotosensitizers. Nat. Nanotechnol. 2015, 10, 370–379. [Google Scholar] [CrossRef] [PubMed]
- Reed, N.A.; Raliya, R.; Tang, R.; Xu, B.; Mixdorf, M.; Achilefu, S.; Biswas, P. Electrospray Functionalization of Titanium Dioxide Nanoparticles with Transferrin for Cerenkov Radiation Induced Cancer Therapy. ACS Appl. Bio. Mater. 2019, 2, 1141–1147. [Google Scholar] [CrossRef]
- Brown, A. The impact of metastable lutetium 177 on a Nuclear Medicine Department. J. Nucl. Med. 2020, 61, 3021. [Google Scholar]
- Bieke, L.; de Klerk, J.M. Clinical applications of Re-188-labelled radiopharmaceuticals for radionuclide therapy. Nucl. Med. Commun. 2006, 27, 223–229. [Google Scholar]
- Lee, S.B.; Ahn, S.B.; Lee, S.W.; Jeong, S.Y.; Ghilsuk, Y.; Ahn, B.C.; Kim, E.M.; Jeong, H.J.; Lee, J.; Lim, D.K.; et al. Radionuclide-embedded gold nanoparticles for enhanced dendritic cell-based cancer immunotherapy, sensitive and quantitative tracking of dendritic cells with PET and Cerenkov luminescence. NPG Asia Mater. 2016, 8, e281. [Google Scholar] [CrossRef]
- Paik, T.; Chacko, A.M.; Mikitsh, J.L.; Friedberg, J.S.; Pryma, D.A.; Murray, C.B. Shape-Controlled Synthesis of Isotopic Yttrium-90-Labeled Rare Earth Fluoride Nanocrystals for Multimodal Imaging. ACS Nano 2015, 9, 8718–8728. [Google Scholar] [CrossRef]
- Daniell, M.D.; Hill, J.S. A history of photodynamic therapy. ANZ J. Surg. 1991, 61, 340–348. [Google Scholar] [CrossRef]
- Ackroyd, R.; Kelty, C.; Brown, N.; Reed, M. The History of Photodetection and Photodynamic Therapy. Photochem. Photobiol. 2001, 74, 656–669. [Google Scholar] [CrossRef]
- Spinelli, A.E.; D’Ambrosio, D.; Calderan, L.; Marengo, M.; Sbarbati, A.; Boschi, F. Cerenkov radiation allows in vivo optical imaging of positron emitting radiotracers. Phys. Med. Biol. 2010, 55, 483–495. [Google Scholar] [CrossRef] [PubMed]
- Hartl, B.A.; Hirschberg, H.; Marcu, L.; Cherry, S.R. Activating Photodynamic Therapy in vitro with Cerenkov Radiation Generated from Yttrium-90. J. Environ. Pathol. Toxicol. Oncol. 2016, 35, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Pratx, G.; Kapp, D.S. Is Cherenkov luminescence bright enough for photodynamic therapy? Nat. Nanotechnol. 2018, 13, 354. [Google Scholar] [CrossRef] [PubMed]
- Magill, S.; Nayfeh, M.; Fizari, M.; Malloy, J.; Maximenko, Y.; Xie, J.; Yu, H. Enhanced UV light detection using wavelength-shifting properties of Silicon nanoparticles. J. Instrum. 2015, 10, P05008. [Google Scholar] [CrossRef]
- Cao, X.; Chen, X.; Kang, F.; Zhan, Y.; Cao, X.; Wang, J.; Liang, J.; Tian, J. Intensity Enhanced Cerenkov Luminescence Imaging Using Terbium-Doped Gd2O2S Microparticles. ACS Appl. Mater. Interfaces 2015, 7, 11775–11782. [Google Scholar] [CrossRef]
- Kaittanis, C.; Shaffer, T.M.; Bolaender, A.; Appelbaum, Z.; Appelbaum, J.; Chiosis, G.; Grimm, J. Multifunctional MRI/PET Nanobeacons Derived from the in Situ Self-Assembly of Translational Polymers and Clinical Cargo through Coalescent Intermolecular Forces. Nano Lett. 2015, 15, 8032–8043. [Google Scholar] [CrossRef]
- Ai, X.; Mu, J.; Xing, B. Recent advances of light-mediated theranostics. Theranostics 2016, 6, 2439–2457. [Google Scholar] [CrossRef]
- Ouyang, Z.; Liu, B.; Yasmin-Karim, S.; Sajo, E.; Ngwa, W. Nanoparticle-aided external beam radiotherapy leveraging the Čerenkov effect. Phys. Medica 2016, 32, 944–947. [Google Scholar] [CrossRef][Green Version]
- Gao, Y.; Ma, X.; Kang, F.; Yang, W.; Liu, Y.; Wang, Z.; Ma, W.; Wang, Z.; Li, G.; Cao, X.; et al. Enhanced Cerenkov luminescence tomography analysis based on Y2O3:Eu3+ rare earth oxide nanoparticles. Biomed. Opt. Express 2018, 9, 6091–6102. [Google Scholar] [CrossRef]
- Ghandi, K.; Wang, F.; Landry, C.; Mostafavi, M. Naked Gold Nanoparticles and hot Electrons in Water. Sci. Rep. 2018, 8, 7258. [Google Scholar] [CrossRef]
- Sheng, J.; Wang, X.; Yan, J.; Pan, D.; Yang, R.; Wang, L.; Xub, Y.; Yang, M. Theranostic radioiodine-labelled melanin nanoparticles inspired by clinical brachytherapy seeds. J. Mater. Chem. B 2018, 6, 8163. [Google Scholar] [CrossRef] [PubMed]
- Kavadiya, S.; Biswas, P. Design of Cerenkov radiation–assisted photoactivation of TiO2 nanoparticles and reactive oxygen species generation for cancer treatment. J. Nucl. Med. 2019, 60, 702–709. [Google Scholar] [CrossRef] [PubMed]
- Jia, M.J.; Bruza, P.; Andreozzi, J.M.; Jarvis, L.A.; Gladstone, D.J.; Pogue, B.W. Cherenkov-excited luminescence scanned imaging using scanned beam differencing and iterative deconvolution in dynamic plan radiation delivery in a human breast phantom geometry. Med. Phys. 2019, 46, 3067–3077. [Google Scholar] [CrossRef] [PubMed]
- Tang, R.; Zheleznyak, A.; Mixdorf, M.; Ghai, A.; Prior, J.; Black, K.C.L.; Shokeen, M.; Reed, N.; Biswas, P.; Achilefu, S. Osteotropic Radiolabeled Nanophotosensitizer for Imaging and Treating Multiple Myeloma. ACS Nano 2020, 14, 4255–4264. [Google Scholar] [CrossRef]
- Pogue, B.W.; Feng, J.; LaRochelle, E.P.; Bruza, P.; Lin, H.; Zhang, R.; Shell, J.R.; Dehghani, H.; Davis, S.C.; Vinogradov, S.A.; et al. Maps of in vivo oxygen pressure with submillimetre resolution and nanomolar sensitivity enabled by Cherenkov-excited luminescence scanned imaging. Nat. Biomed. Eng. 2018, 2, 254–264. [Google Scholar] [CrossRef]
- Romano, F.; Angeloni, S.; Morselli, G.; Mazzaro, R.; Morandi, V.; Shell, J.R.; Cao, X.; Pogue, B.W.; Ceroni, P. Water-soluble silicon nanocrystals as NIR luminescent probes for time-gated biomedical imaging. Nanoscale 2020, 12, 7921–7926. [Google Scholar] [CrossRef]
- Darafsheh, A.; Najmr, S.; Paik, T.; Tenuto, M.E.; Murray, C.B.; Finlay, J.C.; Friedberg, J.S. Characterization of rare-earth-doped nanophosphors for photodynamic therapy excited by clinical ionizing radiation beams. In Optical Methods for Tumor Treatment and Detection: Mechanisms and Techniques in Photodynamic Therapy XXIV; Photonics West-Biomedical Optics: San Francisco, CA, USA, 2015; Volume 930812. [Google Scholar]
- Kim, M.M.; Darafsheh, A. Light Sources and Dosimetry Techniques for Photodynamic Therapy. Photochem. Photobiol. 2020, 96, 280–294. [Google Scholar] [CrossRef]
- Sun, W.; Zhou, Z.; Pratx, G.; Chen, X.; Chen, H. Nanoscintillator-Mediated X-Ray Induced Photodynamic Therapy for Deep-Seated Tumors: From Concept to Biomedical Applications. Theranostics 2020, 10, 1296–1318. [Google Scholar] [CrossRef]
| N | NPs | Source | NP–CR Source Link | Applications | Main Results | Note | Instrument | Reference |
|---|---|---|---|---|---|---|---|---|
| 01 | CdSe/ZnS Quantum dots QD655, QD705, QD800 | 131 I | Separated | In vitro and in vivo. | Feasibility of using radiation luminescence as an internal source to illuminate QDs. | IVIS 200 and IVIS Spectrum | Liu et al. (2010) [11] | |
| 02 | Quantum dots (Qtracker705) | 64Cu, 18F and 99mTc | Separated | In vitro and in vivo. | Qtracker705 and 18F-FDG showed CRET ratios in vitro as high as 8.8 ± 1.1. In vivo in pseudo tumor impregnated with Qtracker705 following intravenous injection of 18F-FDG showed CRET ratios as high as 3.5 ± 0.3. No efficient energy transfer detected with 99mTc. | Definition of Cerenkov radiation energy transfer (CRET) ratio. | IVIS 100 | Dothager et al. (2010) [12] |
| 03 | Tyramine conjugated superparamagnetic iron oxide nanoparticle (TCL-SPION) C = 4–11 nm H ~40 nm | 124I | Bound | In vitro and in vivo. Sentinel lymph nodes (SLNs) detection of mouse bearing breast 4T1 tumor. | Facilitated noninvasive differentiation between tumor-metastasized sentinel lymph nodes (SLNs) and tumor-free SLNs | Triple-modality optical/PET/MRI. | IVIS 200 | Park et al. (2010) [40] |
| 04 | Radioluminescent nanophosphors (RLNPs) Barium yttrium fluoride (Ba0.55Y0.3F2) doped with EuropiumC = 14 nm | 18F | Separated | In vitro and in vivo. | Presentation of facile synthesis and surface modification process to produce water-soluble radioluminescent lanthanide-doped nanophosphors. FDG-stimulated optical imaging of the mice bearing inclusions clearly displayed enhanced emission at 700 nm on the flank containing RLNPs | PET and X-ray validation | IVIS Spectrum | Sun et al. (2011) [29] |
| 05 | Radioluminescent nanophosphors (RLNPs) Barium yttrium fluoride (Ba0.55Y0.3F2) nanocrystals doped with terbium (0.5%) or europium (0.5%) C = 14 nm H ~27 nm | 18F and X-ray tube | Separated | In vitro and in vivo. | RLNPs doped with terbium or europium can be distinguished in optical images in gelatin phantoms. RLNPs aid in the down-conversion of Cerenkov light emitted from the radiopharmaceutical. | PET validation. | IVIS Spectrum | Carpenter et al. (2012) [30] |
| 06 | Quantum dots (QD800) | 32P | Separated | In vitro. | Primary CR and beta particles contribute almost equally to the excitation of the QDs. Good agreement of the light intensity emission with the inverse squared law of the NP–CR source distance. | P32 source covered alternatively with plexiglass slabs or black paper to obtain pure Cerenkov source or pure beta emitter source | IVIS Spectrum | Boschi et al. (2012) [13] |
| 07 | Au nanocages C = 33 nm | 198Au | Incorporated | In vitro, in vivo, ex vivo. murine mammary carcinoma (EMT-6) | Au nanocages show emission with wavelengths in the visible and near-infrared regions, enabling luminescence imaging of the whole mouse in vivo, as well as the organs ex vivo. | First incorporation of CR source into the nanostructure for imaging purpose | IVIS Lumina II XR | Wang et al. (2013) [17] |
| 08 | Fluorescenin (FAM/FITC). Cyanine (Cy5.5, Cy7 ICG). Quantum dots (QD565, QD605, QD800). Au NPs | 68Ga, 18F, 89Zr | Separated | In vitro, in vivo. Mouse squamous cell carcinoma (SCC-7) and human breast cancer (BT-20). | Reduced background signal compared to conventional fluorescence imaging. Approach useful to quantitatively determine prognostically relevant enzymatic activity. | PET and CT validation | IVIS200 | Thorek et al. (2013) [14] |
| 09 | Gold nanocluster C = 2.56 ± 0.50 nm | 64Cu | Bound | In vitro, in vivo, ex vivo. Human primary glioblastoma (U87). | Portion of the energy of Cerenkov radiation serves to excite AuNCs in 64Cu-doped AuNCs. 64Cu-doped AuNCs can be applied as an alternative indicator of PET signal. | Self-illuminating gold nanocluster for dual-modality PET and near-infrared (NIR) fluorescence imaging | IVIS Lumina II | Hu et al. (2014) [18] |
| 10 | Quantum dots (CdSe/ZnS QDs) QD526 QD580 QD636 H = from 14.1 to 28.4 nm PEG-coated | 64Cu | Bound | In vitro, in vivo Human primary glioblastoma (U87). | Favorable imaging without the issue of dissociation of 64Cu from the particles and controllable and enhanced long-wavelength luminescence emission detectable by in vivo imaging. | First time for direct doping of 64Cu PET isotope into QDs via a cation-exchange reaction and endowing them with luminescence properties. PET validation | IVIS Lumina II | Sun et al. (2014) [15] |
| 11 | Au nanostructures: nanospheres, nanodisks, nanorods, nanocages | 198Au | Incorporated | In vitro, in vivo, ex vivo. Murine mammary carcinoma (EMT-6) | Nanospheres showed the best blood circulation, the lowest clearance by the reticuloendothelial system, and the highest overall tumor uptake relative to nanodisks, nanorods, and nanocages. Nanorods and nanocages could reach the cores of the tumors, whereas nanospheres and nanodisks were only observed on the surfaces | PET and autoradiography validation | IVIS 100 | Black et al. (2014) [19] |
| 12 | Superparamagnetic iron oxide nanoparticles (SPIO) | 18F | Separated | In vitro, in vivo. Human fibrosarcoma (HT1080) | Demonstration of quenching of Cerenkov emissions using nanoparticle platforms to provide disease-relevant information including tumor vascularity and specific antigen expression | Several proof of principle models using nanoparticles and clinically approved agents PET validation | IVIS 200 | Thorek et al. (2014) [42] |
| 13 | (PEG)-coated TiO2 nanoparticles, transferrin-coated TiO2 nanoparticles titanocene-transferrin- TiO2 nanoparticles | 18F, 64Cu | Separated | In vitro, in vivo, ex-vivo Human fibrosarcoma (HT1080) | Observed a remarkable shrinkage of the tumor volume (40 ± 5%) within three days of CR-induced therapy initiation. Complete tumor regression was achieved by 30 days and translated into complete remission without a significant loss in body weight up to four months posttreatment | Phototherapy Fluorescence imaging and PET validation | Kotagiri et al. (2015) [43] | |
| 14 | Si nanoparticles | Deuterium lamp | Separated | Not for bioimaging applications. New sensor materials field. | Placing a film of nanoparticles in front of a standard visible-wavelength detecting photosensor, the response of the sensor was significantly enhanced at wavelengths < 320 nm. | To enhance Cerenkov emission and for all experiments requiring sensitivity to UV photons | Hamamatsu MPPC | Magill et al. (2015) [54] |
| 15 | Terbium doped Gd2O2S (Gd2O2S:Tb)nanoparticles | 68Ga | Separated | In vitro, in vivo Gastro intestinal tumor | 50-fold improvement in detection sensitivity, which guaranteed meeting the demands of the clinical diagnosis of gastrointestinal tract tumors. | Endoscopy | EMCCD camera iXon3 888, Andor | Cao et al. (2015) [31] |
| 16 | Terbium doped Gd2O2S Microparticles C = few µm | 18F | Separated | In vitro, in vivo Gastro intestinal tumor | RLMPs significantly improve the intensity and the penetration capacity of CLI, which has been extended to as deep as 15 mm. Microparticles can be excited by gamma rays, but can barely be excited by Cerenkov luminescence. | IVIS system | Cao et al. (2015) [55] | |
| 17 | Europium oxide nanoparticles (EO) C = 85 ± 22 nm. | 18F, 99mTc, 131I | Separated | In vitro, in vivo, ex vivo human breast tumor (Bcap-37) mouse breast tumor (4T1) human glioma tumor (U87MG) human liver tumor (HepG2) | Gamma radiation is the major cause of EO excitation. Strong optical signals with high signal-to-background ratios, an ideal tissue penetration spectrum and activatable imaging ability. More effective detection of tumor lesions with low radioactive tracer uptake or small tumor lesions. | Comparison with Quantum dots (QD620) PET validation | IVIS system | Hu et al. (2015) [32] |
| 18 | Gold nanoclusters conjugated with blood serum proteins (AuNCs) | 18F, 90Y, 99mTc | Separated | In vitro, in vivo, ex-vivo Breast carcinoma (MDA-MB-231) | AuNCs convert beta-decaying radioisotope energy into tissue-penetrating optical signals between 620 and 800 nm with 18F and 90 Y but not with 99mTc. Optical emission from AuNCs is not proportional to Cerenkov radiation, indicating that the energy transfer between the radionuclide and AuNC is only partially mediated by Cerenkov photons. Excitation by high-energy photons is highly inefficient. | Definition of luminescence output (LO) of AuNC as a result of interactions with radioisotope | IVIS Spectrum | Volotskova et al. (2015) [20] |
| 19 | GdF3:90Y/Y nanoplates C = from 8.1 ± 1.2 nm to 15.5 ± 1.3 nm | 90Y | Incorporated | In vitro | Synthesis of a plethora of nanocrystals with different shapes doped with 90Y. Linear relationship between total radiance and radioactivity, suggesting that 90Y-doped nanocrystals are applicable for quantitative optical imaging studies. | Evaluation of MRI capabilities of 90Y-doped GdF3 nanocrystals | IVIS Lumina II | Paik et al. (2015) [48] |
| 20 | Lipid-calcium-phosphate nanoparticle (177Lu-LCP) H = 36 ± 9 nm | 177Lu | Bound | In vitro, in vivo, ex vivo Human nonsmall cell lung cancer cells (H460) Human bladder cancer cells (UMUC3) | 177Lu-LCP functioned as in vivo anticancer therapy in addition to radiographic imaging via the dual decay modes of 177Lu. Treatment with just one dose of 177Lu-LCP showed significant in vivo tumor inhibition in two subcutaneous xenograft tumor models. | Tumor accumulation of 177Lu-LCP was measured using both SPECT and Cerenkov imaging modalities in live mice. | IVIS Kinetic | Satterlee et al. (2015) [27] |
| 21 | Glucose-based polymer dextran (89Zr-PNP) C = from 34 to 82 nm | 89Zr | Bound | In vitro, in vivo | 89Zr-PNP guided the surgical resection of sentinel lymph nodes, utilizing their Cerenkov luminescence. PNP also made it possible to monitor drug release via MRI, through the quenching of the gadolinium signal by the coloaded drug, making them a new multifunctional theranostic nanotechnology platform. | PET/CT validation | IVIS Spectrum | Kaittanis et al. (2015) [56] |
| 22 | Gd2O2S: Eu3+ nanophosphors | 89Zr | Bound | In vitro, in vivo, ex vivo | Excitation of Gd2O2S:Eu nanoparticles by 89Zr was successfully observed. Increasing the nanoparticle concentration or radioactivity increased the intensity of the emission signals. The distance between the donor and the receptor significantly influenced the RL intensity. | PET validation | IVIS Spectrum | Ai et al. (2016) [57] |
| 23 | Titanium dioxide (titania) nanoparticles (NPs) C = 5 nm | X-ray external beam | Separated | In vitro Human lung carcinoma cells (A549) | 6 MV radiation produced the most CR per unit dose deposition, i.e., about 10 times higher than that of 18F. Synergistic effect for the combination of ionizing radiation and titania NPs was observed in the 6MV experiments, where 20% more cancer cells were killed in the group with both radiation and NPs compared to with radiation alone. | External beam radiotherapy Monte Carlo simulations with 18F, 192Ir and 60Co as internal sources | Ouyang et al. (2016) [58] | |
| 24 | Poly(acrylamidoethylamine)-b-poly (DL-lactide) block copolymer-based degradable, cationic, shell-cross-linked knedel-like NPs (Dg-cSCKs)H = 135 ± 40 nm | 123I, 124I, 131I, 76Br | Bound | In vitro, in vivo, ex vivo. | In vivo characterization of pharmacokinetics and fate of the NPs by radiolabeling Dg-cSCKs using a multimodal, noninvasive imaging approach that incorporated positron emission tomography (PET) and Cerenkov luminescence imaging. | Intratracheal injection for lung gene transfer. PET validation | IVIS 100 | Black et al. (2016) [36] |
| 25 | Radioiodine embedded gold (Au) nanoparticles (Rie AuNPs) C = 5, 20 and 42 nm | 124I, 125I | Bound | In vitro, in vivo, ex vivo. | Simple and straightforward synthetic scheme for producing gold-based imaging agents that are applicable as a dual bio-imaging modality by combining nuclear imaging and CLI. | PET/SPECT validation | IVIS Lumina III | Lee et al. (2016) [47] |
| 26 | Peghilated radioiodine embedded gold (Au) nanoparticles (PEG-RIe-AuNPs) C = 20 nm | 124I | Bound | In vitro, in vivo, ex vivo. | In vivo imaging reveals sentinel lymph nodes as early as 1 h post PEG-RIe-AuNP-injection, with peak signals achieved at 6 h postinjection. The data provide strong evidence that PEG-RIe-AuNPs are promising as potential lymphatic tracers in biomedical imaging for pre- and intra- operative surgical guidance | NPs useful for sentinel lymph node detection via PET and CLI PET/CT validation | IVIS Lumina III | Lee et al. (2016) [21] |
| 27 | Hollow mesoporous silica nanoparticles ([89Zr]HMSN-Ce6) H ∼130 ± 2.1 nm | 89Zr | Bound | In vitro, in vivo, ex vivo. Murine breast cancer (4T1) | In vitro cell viability experiments demonstrated dose-dependent cell deconstruction as a function of the concentration of Ce6 and 89Zr. In vivo studies showed inhibition of tumor growth when mice were subcutaneously injected with [89Zr]HMSN-Ce6, and histological analysis of the tumor section showed damage to tumor tissues, implying that reactive oxygen species mediated the destruction. | Photodynamic Therapy Activation of chlorin e6 (Ce6) to generate reactive oxygen species (ROS) PET imaging | IVIS Spectrum | Kamkaew et al. (2016) [23] |
| 28 | Europium oxide (EO) nanoparticles C = 85 ± 22 nm. | 131I, 18F, 68Ga, 99mTc | Separated | In vitro, in vivo, ex vivo. Human hepatocellular carcinomas (HCC). | A mixture of 68GaCl3 and EO nanoparticles yielded the strongest optical signals compared with the other mixtures. Radiopharmaceutical-excited fluorescence tomography (REFT) can detect more tumors than small-Animal PET in hepatocellular carcinoma-bearing mice, and achieved more accurate 3D distribution information than Cerenkov luminescence tomography. | PET/CT imaging. | IVIS Kinetic | Hu et al. (2017) [33] |
| 29 | Dual-labeled nanoparticles based on lipid micelles (89Zr-QD-MC) nanoemulsions (89Zr-QD-NE), and polymeric biocompatible nanoplatforms (89Zr-QD-BP). H = from 45 to 75 nm | 89Zr | Bound | In vitro, in vivo, ex vivo. Human prostate cancer (DU145) | The intensity of converted light is linearly related to the concentration of the spectral converter, and the slope is related to the quantum yield of the fluorophore. Pharmacokinetics, biodistribution, and whole-body imaging of QD and 89Zr dual-labeled nanoparticles. | PET/CT imaging | IVIS Spectrum | Zhao et al. (2017) [16] |
| 30 | Europium oxide (EO) nanoparticles C = 85 ± 22 nm. | 18F, 11C | Separated | In vitro, in vivo, ex vivo. Mouse breast cancer (4T1) Human hepatocellular carcinoma (HCC). | By mixing the 18F–FDG and EO nanoparticles, strong near-infrared fluorescent light is emitted, and its peak is 620 nm. The total flux is almost 70 times higher than the sum of the optical signals of EO nanoparticles and 18F–FDG alone. EO at a very low dose can be excited by radiopharmaceuticals of very low dose to produce an optical signal. Mediated radiopharmaceutical-excited fluorescent (REF) image-guided cancer surgery strategy technique exhibited excellent performance in detecting invisible ultrasmall tumors (even less than 1 mm) and residual tumor tissue. | For precise image-guided tumor-removal surgery. Employs the internal dual excitation of EO nanoparticles by both gamma rays and Cerenkov luminescence of radiopharmaceuticals. | IVIS Kinetic | Hu et al. (2017) [34] |
| 31 | Citrate-capped (Cit) copper sulfide (CuS) nanoparticles on the surface of [89Zr]-labeled hollow mesoporous silica nanoshells (HMSN) filled with porphyrin molecules H~150 nm (HMSN), ~10 nm (CuS-Cit) | 89Zr | Bound | In vitro, in vivo, ex vivo. Murine breast cancer (4T1). | Development of a novel, biocompatible, hybrid nanoplatform to seek and treat cancer in vivo. [89Zr]-labeled HMSN shell, CuS nanosatellites and photosensitizer porphyrin, self-assemble for Tetramodal Imaging and Synergistic Photothermal/Photodynamic Therapy Localized and synergistic phototherapy shows complete tumor eradication with no recurrence or long-term toxicity. | Photodynamic therapy PET, fluorescence, Cerenkov Luminescence and Cerenkov Radiation Energy Transfer-based imaging, and, photothermal/photodynamic therapy Activation of TCPP and doxorubicin | IVIS Spectrum | Goel et al. (2018) [24] |
| 32 | 68Ga-labeled bovine serum albumin (68Ga-BSA) and dextran-modified TiO2 nanoparticles (D-TiO2 NPs) H = from 73.2 to 83.2 nm | 68Ga, 18F | Separated | In vitro, in vivo, ex vivo. Murine breast cancer (4T1) | 68Ga is a more potent radionuclide than 18F for CR-induced PDT. The tumor volumes in mice treated by 68Ga-BSA and D-TiO2 NPs were significantly inhibited, whereas no significant difference in tumor volumes was found between the control group and other treatment groups. | Photodynamic therapy PET/CT validation | Duan et al. (2018) [37] | |
| 33 | PEGylated crushed gold shell-radioactive iodide-124-labeled gold core nanoballs (PEG-124I-Au@AuCBs) C = 20 nm | 124I | Bound | In vivo, in vitro, ex vivo Murine breast cancer (4T1) | PEG-124I-Au@AuCBs showed high stability and sensitivity in various pH solutions, serum, and in vivo conditions. Combined PET/CLI clearly revealed tumor lesions at 1 h after injection of particles, and both signals remained visible in tumor lesions at 24 h. | PET validation | IVIS Lumina III | Lee et al. (2018) [38] |
| 34 | Amorphous silica NP (SNP) H = 163.7 ± 4.8 nm TiO2 H = 12.8 ± 1.5 nm, HfO2 H = 34.9 ± 0.7 nm, Eu2O3 H = 134 ± 18 nm Gd2O3 H = 75.7 ± 6.8 nm YAG:Ce H = 36.6 ± 4.0 nm Bi2O3 H = 201.2 ± 4.4 nm AuNP H = 85.8 ± 0.009 nm | 3H, 35S, 177Lu, 32P, 18F, 89Zr, 68Ga, 90Y 99mTc | Separated | In vitro, in vivo | β-scintillation contributes appreciably to excitation and reactivity in certain nanoparticle systems. The excitation by radionuclides of nanoparticles composed of large atomic number atoms generates X-rays, enabling multiplexed imaging through single photon emission computed tomography. Optical imaging and therapy using radionuclides with emission energies below the Cerenkov threshold are feasible, thereby expanding the list of applicable radionuclides. | SPECT/CT validation | IVIS Spectrum | Pratt et al. (2018) [25] |
| 35 | Magnetic nanoparticle(Zn0.4Mn0.6) Fe2O4 s (MNPs) with 89Zr radiolabeling and porphyrin molecules (TCPP) surface modification (89Zr-MNP/TCPP) C = 20 nm | 89Zr | Bound | In vivo, in vitro, ex-vivo Murine breast cancer (4T1) | In vivo biodistribution of the 89Zr-MNP/TCPP imaging (FL), Cerenkov luminescence (CL) and CRET Imaging. High NPs tumor accumulation in the presence of an external magnetic field. The intensity spectrum of 89Zr-MNP/TCPP was completely different from that of free 89Zr and exhibited much stronger emission at a long wavelength, i.e., from 600 to 800 nm, reaching the maximum at around 660 nm. | Photodynamic therapy PET confirmation | IVIS system | Ni et al. (2018) [41] |
| 36 | Y2O3:Eu3+ rare earth nanoparticles (RENPS) C = 51.5 ± 7.5 nm | 68Ga | Separated | In vitro. | Enhanced CL penetration and intensity (over 3 times better) by using Y2O3:Eu3+ RENPs. 3D reconstruction method which is able to acquire more accurate spatial information in vivo, as well as some quantitative information. | PET/CT validation | IVIS Kinetic | Gao et al. (2018) [59] |
| 37 | Gold nanoparticles C = 15 nm | Electron pulse | Separated | In vitro. No imaging | Cerenkov absorption spectrum in water with different gold nanoparticle concentrations | Ghandi et al. (2018) [60] | ||
| 38 | 188Re-liposome H = 74.2 ± 9.1 nm | 188Re | Bound | In vitro, in vivo, ex vivo Human hypopharyngeal carcinoma cells (FaDu) Human tongue carcinoma (SAS) Human oral squamous carcinoma (OECM-1) | CLI demonstrate an increased accumulation of 188Re-liposome in the tumor lesion of nude mice with repeated doses compared to a single dose. | IVIS 50 | Chang et al. (2018) [28] | |
| 39 | Radioiodine-labelled melanin nanoparticles (MNP–Ag–131I) C = 6 nm H = 12 nm | 131I | Bound | In vitro, in vivo, ex vivo Human prostate carcinoma (PC3) | Synthesis of MNP–Ag–131I for therapeutic purposes which can be used for both single-photon emission computed tomography and Cherenkov radiation imaging. The beta rays of 131I make it a good candidate as a cancer cell killer. | MNP used as a platform for SPECT and CLI for accurate localization in brachytherapy. | IVIS Spectrum | Sheng et al. (2020) [61] |
| 40 | PEGylated crushed gold shell @ radioiodine-labeled core nanoparticles (PEG-124I-Au@AuCBs) C = 20 nm | 124I | Bound | In vitro, in vivo, ex vivo. | PEG-124I-Au@AuCBs are promising lymphatic tracers for dual imaging PET/CLI. NPs allowed for high-sensitivity detection of SLNs within 1 h postinjection, and their accumulation persisted until 24 h in a clinical application under intraoperative conditions. | PET/CT validation | IVIS Lumina III | Lee et al. (2019) [22] |
| 41 | TiO2 nanoparticles coated with the glycoprotein transferrin (Tf) (Tf/TiO2) C = 25 ± 3.2 nm | 18F | Separated | In vitro. No imaging Human multiple myeloma (MM1.S) and human fibrosarcoma (HT1080) | The use of an electrospray system is efficient for coating the protein transferrin (Tf) on the surface of titanium dioxide (TiO2) nanoparticles in a single step. Tf/TiO2 nanoparticles improved cell killing for MM1.S multiple myeloma cells from 23% to 57%, compared to Tf/TiO2 nanoparticles prepared using conventional functionalization methods. | Cerenkov Radiation Induced Cancer Therapy | Reed et al. (2019) [44] | |
| 42 | TiO2 nanoparticles | 18F | Separated | No imaging | Mathematic model that integrates Cerenkov physics, light interaction with matter, and photocatalytic reaction engineering. The model investigates the concentration of TiO2 nanoparticles and the activity of the radionuclide 18F-FDG on the number of photons and ROS generation. The model can be used for other radionuclides and nanoparticles, and can provide guidance on the concentration and size of TiO2 nanoparticles and the radionuclide activity needed for efficient cancer therapy. | Theoretical investigation Validation with comparison to experimental reports in the literature | Kavadiya et al. (2019) [62] | |
| 43 | Europium nanoparticles Microspheres | X ray from LINAC | Separated | New methodological approach to reconstruct Cherenkov excited luminescence intensity distributions starting from a three-dimensional dataset. The method makes it possible to visualize and localize luminescence/fluorescence tagged vasculature, lymph nodes, or superficial tagged regions with most dynamic treatment plans. | Cherenkov excited luminescence scanning imaging (CELSI) | ICCD Princeton Instruments | Jia et al. (2019) [63] | |
| 44 | Silica nanoparticles (Plus-NPs) H = 25 nm | 32P | Separated | In vitro | Synthesized pluronic–silica nanoparticles doped with five different dyes that were chosen to efficiently absorb CR in all the visible ranges and to efficiently funnel the excitation energy toward the lowest energy dye, a Cy7 derivative, presenting a fluorescence emission in the near-infrared region (NIR). | IVIS Spectrum | Genovese et al. (2020) [26] | |
| 45 | Chelate-Free Radiolabeling and Transferrin coating of TiO2 NPs (89Zr -TiO2- Tf NPs) | 89Zr | Bound | In vitro, in vivo, ex vivo. Multiple myeloma (MM1.S) | Design of a theranostic nanoplatform (89Zr -TiO2-Tf NPs) for targeting bone marrow, imaging the distribution of NPs, and stimulating ROS generation for cell killing. Single dose of NPs inhibited cancer growth | Photodynamic therapy PET/CT imaging | IVIS Lumina | Tang et al. (2020) [64] |
| 46 | Persistent Luminescent ZnGa2O4:Cr3+, NPs (ZGCs) C = 60–80 nm | 18F | Separated | In vitro, in vivo, ex vivo. Murine breast cancer (4T1) | 18F can efficiently excite ZGCs Nanoparticles for both fluorescence and afterglow luminescence via Cerenkov resonance energy transfer, as well as ionizing radiation. | PET imaging | IVIS Lumina | Liu et al. (2020) 35] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boschi, F.; Spinelli, A.E. Nanoparticles for Cerenkov and Radioluminescent Light Enhancement for Imaging and Radiotherapy. Nanomaterials 2020, 10, 1771. https://doi.org/10.3390/nano10091771
Boschi F, Spinelli AE. Nanoparticles for Cerenkov and Radioluminescent Light Enhancement for Imaging and Radiotherapy. Nanomaterials. 2020; 10(9):1771. https://doi.org/10.3390/nano10091771
Chicago/Turabian StyleBoschi, Federico, and Antonello Enrico Spinelli. 2020. "Nanoparticles for Cerenkov and Radioluminescent Light Enhancement for Imaging and Radiotherapy" Nanomaterials 10, no. 9: 1771. https://doi.org/10.3390/nano10091771
APA StyleBoschi, F., & Spinelli, A. E. (2020). Nanoparticles for Cerenkov and Radioluminescent Light Enhancement for Imaging and Radiotherapy. Nanomaterials, 10(9), 1771. https://doi.org/10.3390/nano10091771