Ligand-Length Modification in CsPbBr3 Perovskite Nanocrystals and Bilayers with PbS Quantum Dots for Improved Photodetection Performance
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis and Purification of CsPbBr3 Nanocrystals
2.3. Characterization of PNCs
2.4. Device Fabrication
2.5. Characterization of Films and Devices
3. Results and Discussion
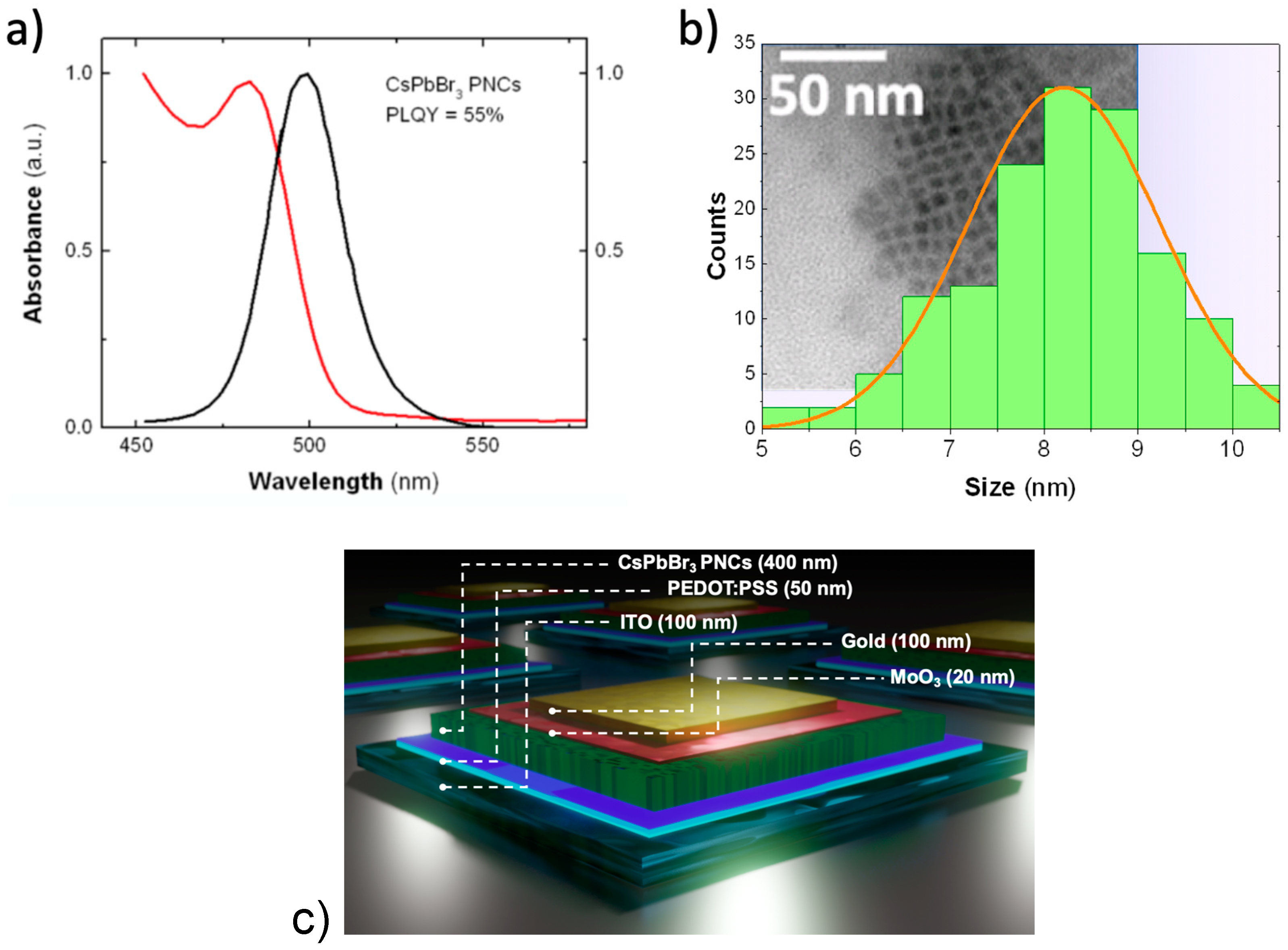
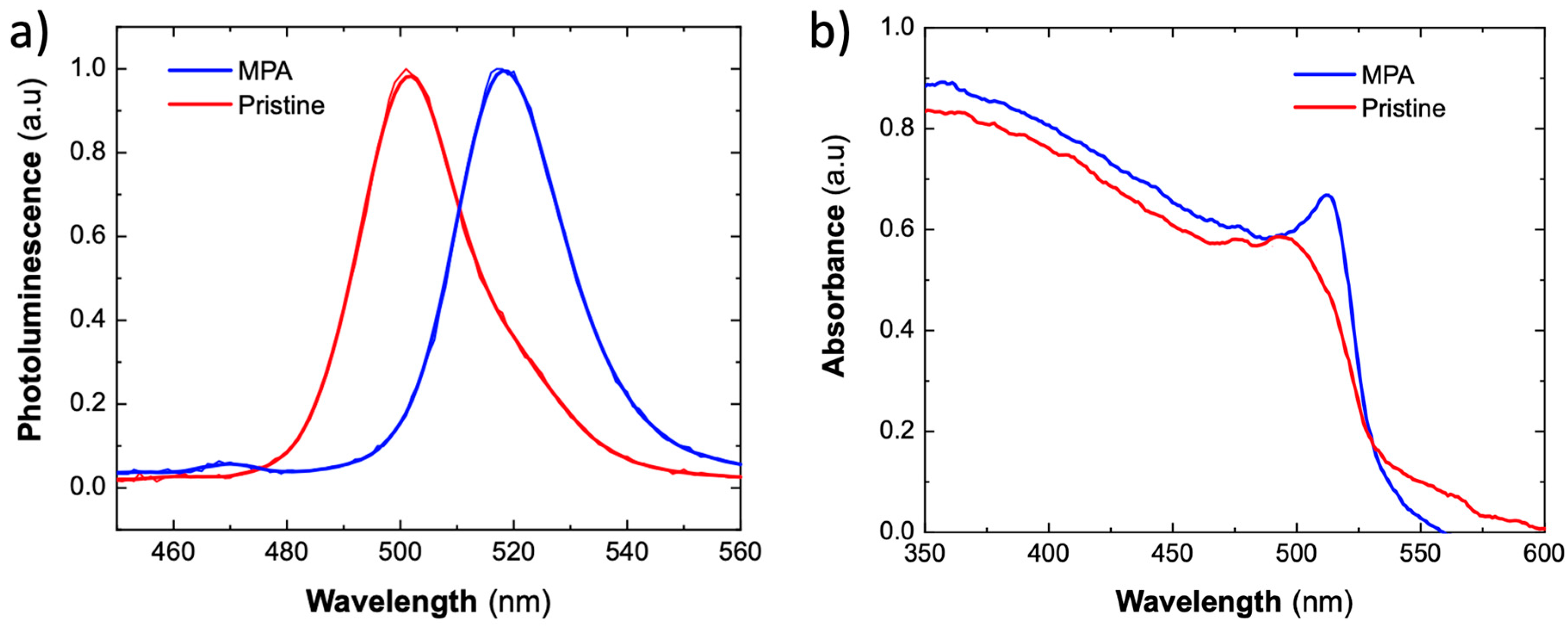
3.1. Thin Films of CsPbBr3 PNCs
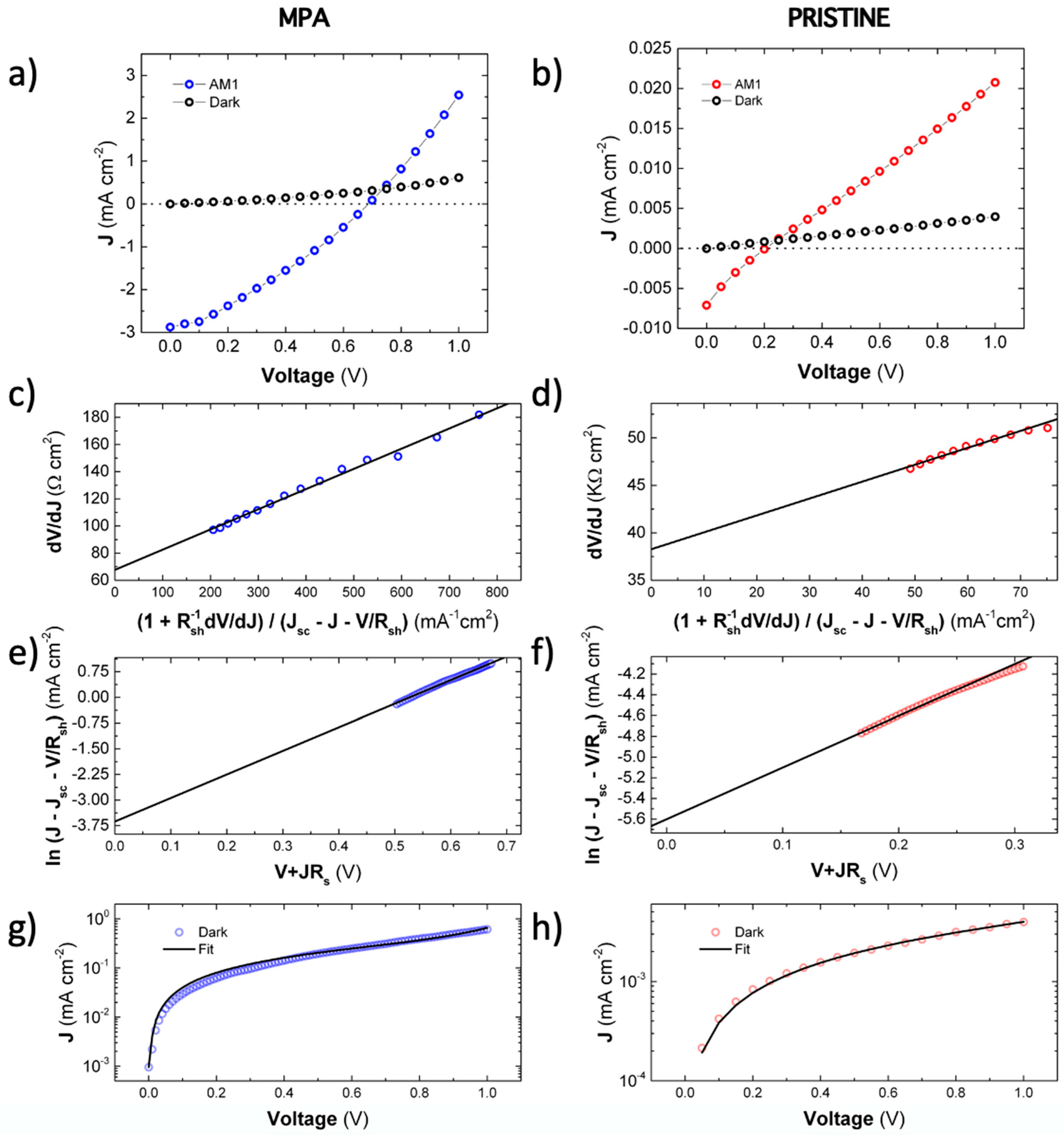
3.2. Electro-Optical Characterization of Schottky Heterostructures
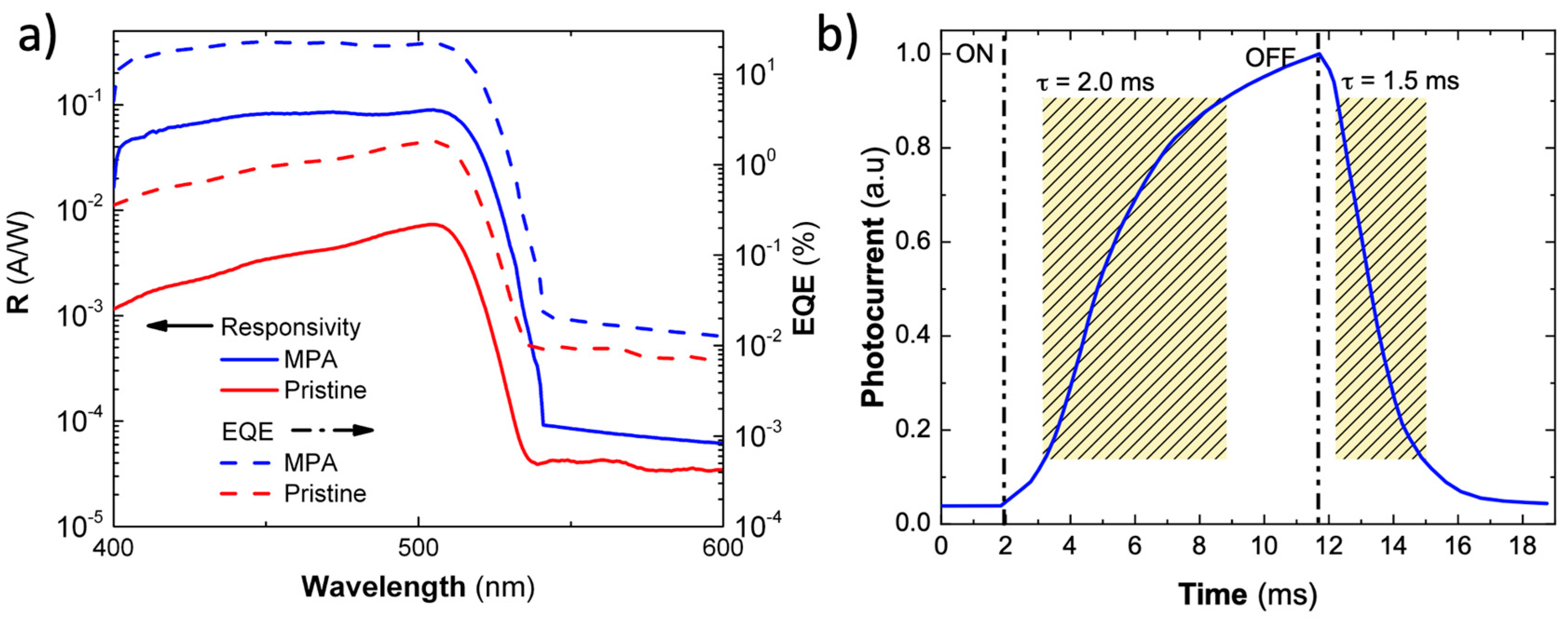
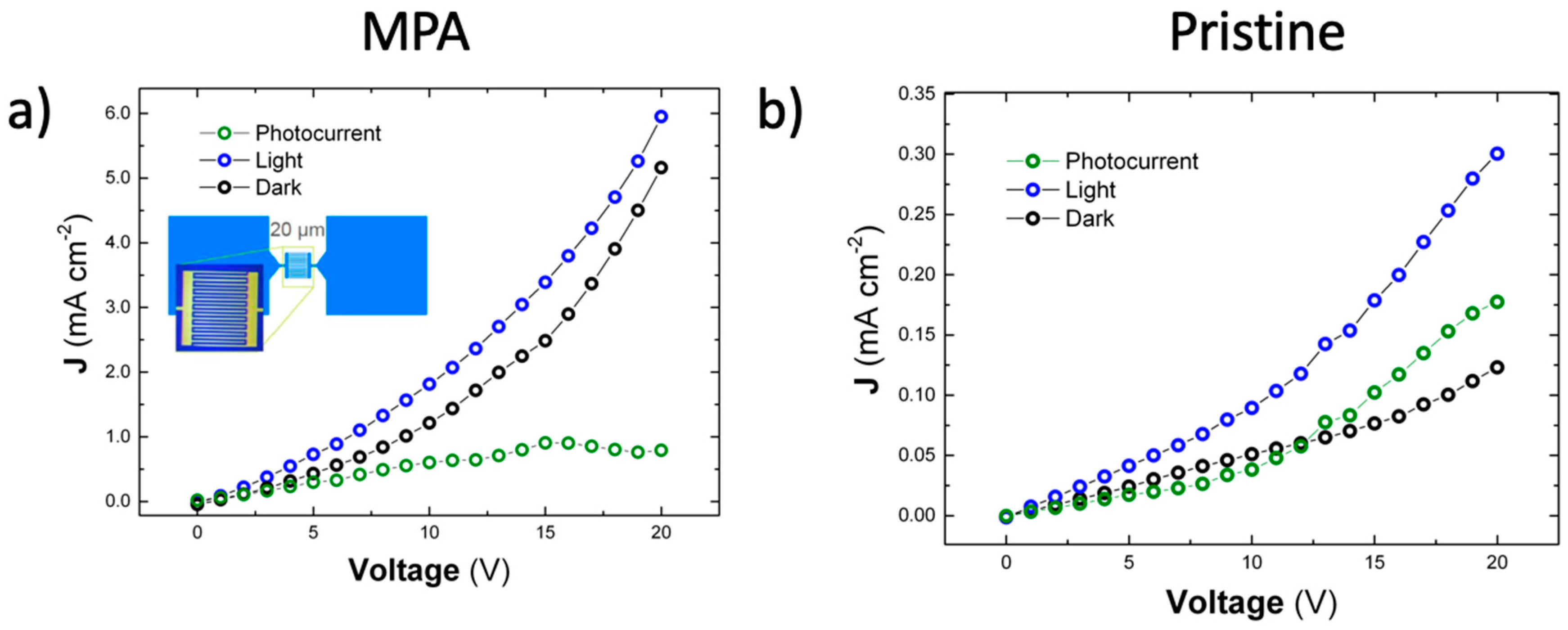
3.3. Electro-Optical Characterization of Photoconductors
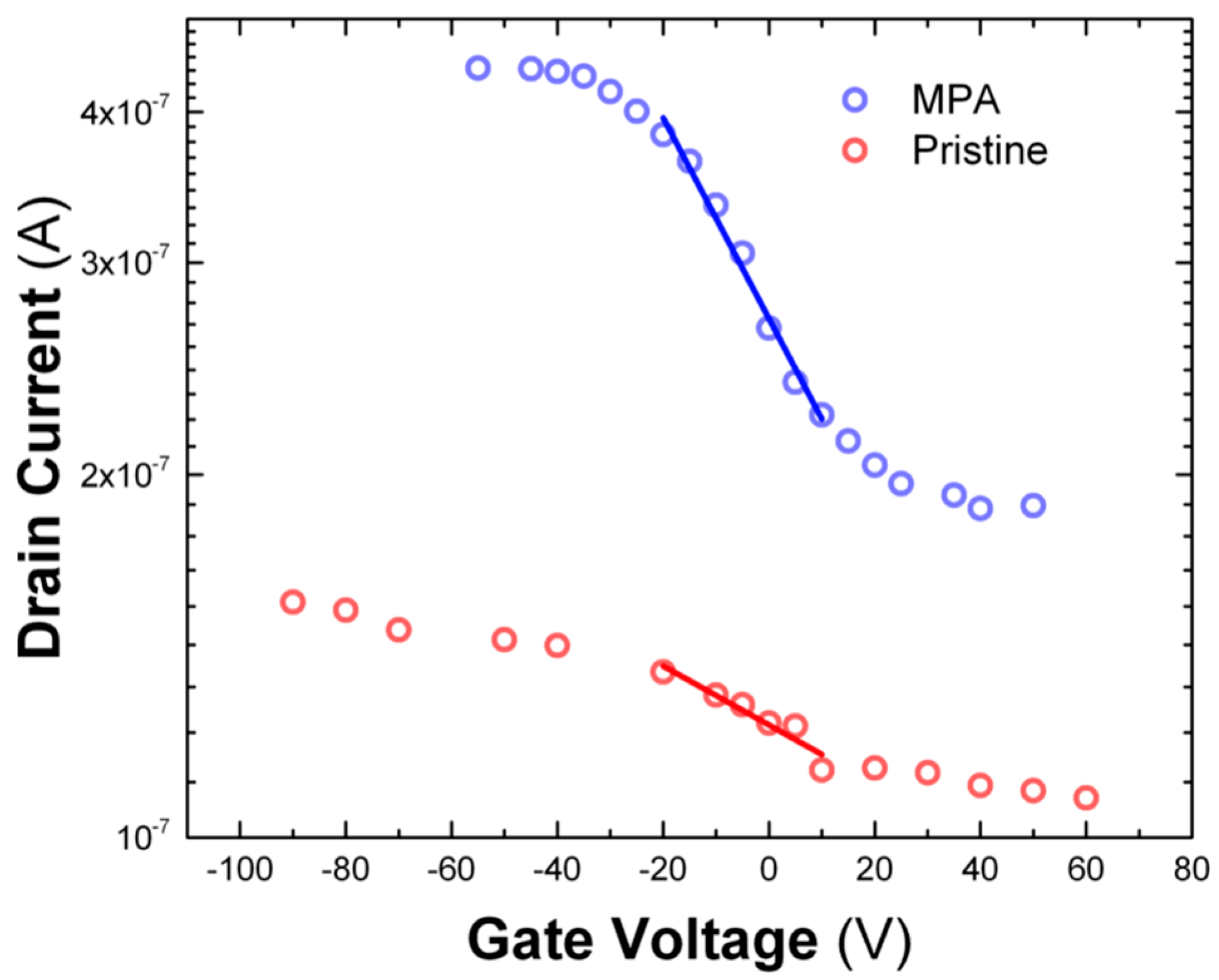
3.4. Characterization of the FET Devices
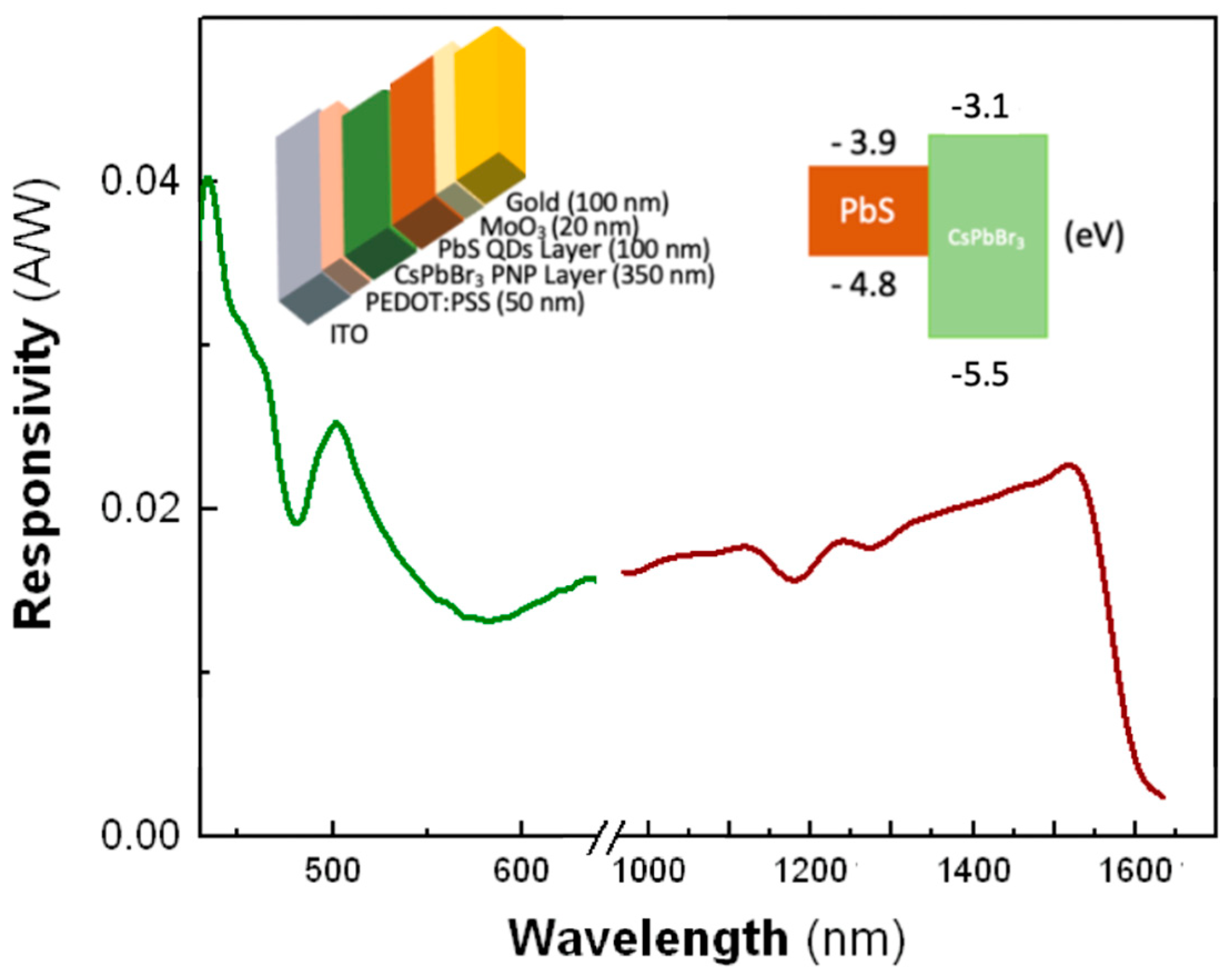
3.5. Tandem PbS-CsPbBr3 Photodiodes
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kovalenko, M.V.; Protesescu, L.; Bodnarchuk, M.I. Properties and potential optoelectronic applications of lead halide perovskite nanocrystals. Science 2017, 358, 745–750. [Google Scholar] [CrossRef] [PubMed]
- Protesescu, L.; Yakunin, S.; Bodnarchuk, M.I.; Krieg, F.; Caputo, R.; Hendon, C.H.; Yang, R.X.; Walsh, A.; Kovalenko, M.V. Nanocrystals of Cesium Lead Halide Perovskites (CsPbX3, X = Cl, Br, and I): Novel Optoelectronic Materials Showing Bright Emission with Wide Color Gamut. Nano Lett. 2015, 15, 3692–3696. [Google Scholar] [CrossRef] [PubMed]
- Wu, K.; Liang, G.; Shang, Q.; Ren, Y.; Kong, D.; Lian, T. Ultrafast Interfacial Electron and Hole Transfer from CsPbBr3 Perovskite Quantum Dots. J. Am. Chem. Soc. 2015, 137, 12792–12795. [Google Scholar] [CrossRef] [PubMed]
- Colbert, A.E.; Wu, W.; Janke, E.M.; Ma, F.; Ginger, D.S. Effects of Ligands on Charge Generation and Recombination in Hybrid Polymer/Quantum Dot Solar Cells. J. Phys. Chem. C 2015, 119, 24733–24739. [Google Scholar] [CrossRef]
- Huang, H.; Polavarapu, L.; Sichert, J.A.; Susha, A.S.; Urban, A.S.; Rogach, A.L. Colloidal lead halide perovskite nanocrystals: Synthesis, optical properties and applications. NPG Asia Mater. 2016, 8, e328. [Google Scholar] [CrossRef]
- Chen, Y.; Wu, X.; Chu, Y.; Zhou, J.; Zhou, B.; Huang, J. Hybrid Field-Effect Transistors and Photodetectors Based on Organic Semiconductor and CsPbI3 Perovskite Nanorods Bilayer Structure. Nano-Micro Lett. 2018, 10, 1–9. [Google Scholar] [CrossRef]
- Yang, T.; Zheng, Y.; Du, Z.; Liu, W.; Yang, Z.; Gao, F.; Wang, L.; Chou, K.-C.; Hou, X.; Yang, W. Superior Photodetectors Based on All-Inorganic Perovskite CsPbI 3 Nanorods with Ultrafast Response and High Stability. ACS Nano 2018, 12, 1611–1617. [Google Scholar] [CrossRef]
- Zhang, D.; Yang, Y.; Bekenstein, Y.; Yu, Y.; Gibson, N.A.; Wong, A.B.; Eaton, S.W.; Kornienko, N.; Kong, Q.; Lai, M.; et al. Synthesis of Composition Tunable and Highly Luminescent Cesium Lead Halide Nanowires through Anion-Exchange Reactions. J. Am. Chem. Soc. 2016, 138, 7236–7239. [Google Scholar] [CrossRef]
- Yang, D.; Fang, H.; Dong, A.; Liu, L.; Hu, W.; Xu, Y.; Zhang, X.; Luo, W.; Lv, L.; Xu, F.; et al. Generalized colloidal synthesis of high-quality, two-dimensional cesium lead halide perovskite nanosheets and their applications in photodetectors. Nanoscale 2016, 8, 13589–13596. [Google Scholar]
- Song, J.; Xu, L.; Li, J.; Xue, J.; Dong, Y.; Li, X.; Zeng, H. Monolayer and Few-Layer All-Inorganic Perovskites as a New Family of Two-Dimensional Semiconductors for Printable Optoelectronic Devices. Adv. Mater. 2016, 4861–4869. [Google Scholar] [CrossRef]
- Ramasamy, P.; Lim, D.H.; Kim, B.; Lee, S.H.; Lee, M.S.; Lee, J.S. All-inorganic cesium lead halide perovskite nanocrystals for photodetector applications. Chem. Commun. 2016, 52, 2067–2070. [Google Scholar] [CrossRef] [PubMed]
- Jang, D.M.; Kim, D.H.; Park, K.; Park, J.; Lee, J.W.; Song, J.K. Ultrasound synthesis of lead halide perovskite nanocrystals. J. Mater. Chem. C 2016, 4, 10625–10629. [Google Scholar] [CrossRef]
- Dong, Y.; Gu, Y.; Zou, Y.; Song, J.; Xu, L.; Li, J.; Xue, J.; Li, X.; Zeng, H. Improving All-Inorganic Perovskite Photodetectors by Preferred Orientation and Plasmonic Effect. Small 2016, 12, 5622–5632. [Google Scholar] [CrossRef]
- Tong, Y.; Yao, E.-P.; Manzi, A.; Bladt, E.; Wang, K.; Döblinger, M.; Bals, S.; Müller-Buschbaum, P.; Urban, A.S.; Polavarapu, L.; et al. Spontaneous Self-Assembly of Perovskite Nanocrystals into Electronically Coupled Supercrystals: Toward Filling the Green Gap. Adv. Mater. 2018, 30, 1801117. [Google Scholar] [CrossRef] [PubMed]
- Hazarika, A.; Zhao, Q.; Gaulding, E.A.; Christians, J.A.; Dou, B.; Marshall, A.R.; Moot, T.; Berry, J.J.; Johnson, J.C.; Luther, J.M. Perovskite Quantum Dot Photovoltaic Materials beyond the Reach of Thin Films: Full-Range Tuning of A-Site Cation Composition. ACS Nano 2018, 12, 10327–10337. [Google Scholar] [CrossRef]
- Maulu, A.; Rodríguez-Cantó, P.J.; Navarro-Arenas, J.; Abargues, R.; Sánchez-Royo, J.F.; García-Calzada, R.; Martínez Pastor, J.P. Strongly-coupled PbS QD solids by doctor blading for IR photodetection. RSC Adv. 2016, 6, 80201–80212. [Google Scholar] [CrossRef]
- Abargues, R.; Navarro, J.; Rodríguez-Cantó, P.J.; Maulu, A.; Sánchez-Royo, J.F.; Martínez-Pastor, J.P. Enhancing the photocatalytic properties of PbS QD solids: The ligand exchange approach. Nanoscale 2019, 11, 1978–1987. [Google Scholar] [CrossRef]
- Ji, G.; Zhao, W.; Wei, J.; Yan, L.; Han, Y.; Luo, Q.; Yang, S.; Hou, J.; Ma, C.Q. 12.88% efficiency in doctor-blade coated organic solar cells through optimizing the surface morphology of a ZnO cathode buffer layer. J. Mater. Chem. A 2019, 7, 212–220. [Google Scholar] [CrossRef]
- Huang, H.; Lin, H.; Kershaw, S.V.; Susha, A.S.; Choy, W.C.H.; Rogach, A.L. Polyhedral Oligomeric Silsesquioxane Enhances the Brightness of Perovskite Nanocrystal-Based Green Light-Emitting Devices. J. Phys. Chem. Lett. 2016, 7, 4398–4404. [Google Scholar] [CrossRef]
- Cai, Y.; Wang, L.; Zhou, T.; Zheng, P.; Li, Y.; Xie, R.-J. Improved stability of CsPbBr3 perovskite quantum dots achieved by suppressing interligand proton transfer and applying a polystyrene coating. Nanoscale 2018, 10, 21441–21450. [Google Scholar] [CrossRef]
- Boles, M.A.; Engel, M.; Talapin, D. V Self-Assembly of Colloidal Nanocrystals: From Intricate Structures to Functional Materials. Chem. Rev. 2016, 116, 11220–11289. [Google Scholar] [CrossRef]
- Fu, W.; Shi, Y.; Qiu, W.; Wang, L.; Nan, Y.; Shi, M.; Li, H.; Chen, H. High efficiency hybrid solar cells using post-deposition ligand exchange by monothiols. Phys. Chem. Chem. Phys. 2012, 14, 12094–12098. [Google Scholar] [CrossRef] [PubMed]
- Crisp, R.W.; Kroupa, D.M.; Marshall, A.R.; Miller, E.M.; Zhang, J.; Beard, M.C.; Luther, J.M. Metal Halide Solid-State Surface Treatment for High Efficiency PbS and PbSe QD Solar Cells. Sci. Rep. 2015, 5, 9945. [Google Scholar] [CrossRef] [PubMed]
- Baker, D.R.; Kamat, P.V. Tuning the emission of CdSe quantum dots by controlled trap enhancement. Langmuir 2010, 26, 11272–11276. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Huang, J.; Zhu, H.; Li, Y.; Tang, J.-X.; Jiang, Y. Surface Ligand Engineering for Near-Unity Quantum Yield Inorganic Halide Perovskite QDs and High-Performance QLEDs. Chem. Mater. 2018, 30, 6099–6107. [Google Scholar] [CrossRef]
- Zhou, L.; Yu, K.; Yang, F.; Cong, H.; Wang, N.; Zheng, J.; Zuo, Y.; Li, C.; Cheng, B.; Wang, Q. Insight into the effect of ligand-exchange on colloidal CsPbBr3 perovskite quantum dot/mesoporous-TiO2 composite-based photodetectors: Much faster electron injection. J. Mater. Chem. C 2017, 5, 6224–6233. [Google Scholar] [CrossRef]
- Gong, M.; Sakidja, R.; Goul, R.; Ewing, D.; Casper, M.; Stramel, A.; Elliot, A.; Wu, J.Z. High-Performance All-Inorganic CsPbCl3 Perovskite Nanocrystal Photodetectors with Superior Stability. ACS Nano 2019, 13, 1772–1783. [Google Scholar] [CrossRef]
- Balazs, D.M.; Rizkia, N.; Fang, H.H.; Dirin, D.N.; Momand, J.; Kooi, B.J.; Kovalenko, M.V.; Loi, M.A. Colloidal Quantum Dot Inks for Single-Step-Fabricated Field-Effect Transistors: The Importance of Postdeposition Ligand Removal. ACS Appl. Mater. Interfaces 2018, 10, 5626–5632. [Google Scholar] [CrossRef]
- Maulu, A.; Navarro-Arenas, J.; Rodríguez-Cantó, P.; Sánchez-Royo, J.; Abargues, R.; Suárez, I.; Martínez-Pastor, J. Charge Transport in Trap-Sensitized Infrared PbS Quantum-Dot-Based Photoconductors: Pros and Cons. Nanomaterials 2018, 8, 677. [Google Scholar] [CrossRef]
- Li, Y.; Shi, Z.-F.; Li, X.-J.; Shan, C.-X. Photodetectors based on inorganic halide perovskites: Materials and devices. Chinese Phys. B 2019, 28, 017803. [Google Scholar] [CrossRef]
- Li, Y.; Shi, Z.F.; Li, S.; Lei, L.Z.; Ji, H.F.; Wu, D.; Xu, T.T.; Tian, Y.T.; Li, X.J. High-performance perovskite photodetectors based on solution-processed all-inorganic CsPbBr3 thin films. J. Mater. Chem. C 2017, 5, 8355–8360. [Google Scholar] [CrossRef]
- Song, X.; Liu, X.; Yu, D.; Huo, C.; Ji, J.; Li, X.; Zhang, S.; Zou, Y.; Zhu, G.; Wang, Y.; et al. Boosting Two-Dimensional MoS 2 /CsPbBr 3 Photodetectors via Enhanced Light Absorbance and Interfacial Carrier Separation. ACS Appl. Mater. Interfaces 2018, 10, 2801–2809. [Google Scholar] [CrossRef]
- Yao, Y.; Zhang, B.; Green, M.A.; Conibeer, G.; Shrestha, S.K. Photovoltaic effect in Ge nanocrystals/c-silicon heterojunctions devices. In Proceedings of the 2010 35th IEEE Photovoltaic Specialists Conference, Honolulu, HI, USA, 20–25 June 2010; pp. 1889–1893. [Google Scholar]
- Hu, L.; Wang, W.; Liu, H.; Peng, J.; Cao, H.; Shao, G.; Xia, Z.; Ma, W.; Tang, J. PbS colloidal quantum dots as an effective hole transporter for planar heterojunction perovskite solar cells. J. Mater. Chem. A 2015, 3, 515–518. [Google Scholar] [CrossRef]
- Huang, H.; Bodnarchuk, M.I.; Kershaw, S.V.; Kovalenko, M.V.; Rogach, A.L. Lead Halide Perovskite Nanocrystals in the Research Spotlight: Stability and Defect Tolerance. ACS Energy Lett. 2017, 2, 2071–2083. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, J.B.; Zaiats, G.; Wappes, I.; Kamat, P.V. CsPbBr3Solar Cells: Controlled Film Growth through Layer-by-Layer Quantum Dot Deposition. Chem. Mater. 2017, 29, 9767–9774. [Google Scholar] [CrossRef]
- Akkerman, Q.A.; Gandini, M.; Di Stasio, F.; Rastogi, P.; Palazon, F.; Bertoni, G.; Ball, J.M.; Prato, M.; Petrozza, A.; Manna, L. Strongly emissive perovskite nanocrystal inks for high-voltage solar cells. Nat. Energy 2017, 2, 16194. [Google Scholar] [CrossRef]
- Yang, Z.; Wang, M.; Li, J.; Dou, J.; Qiu, H.; Shao, J. Spray-Coated CsPbBr3 Quantum Dot Films for Perovskite Photodiodes. ACS Appl. Mater. Interfaces 2018, 10, 26387–26395. [Google Scholar] [CrossRef]
- Zhang, Y.; Wu, G.; Liu, F.; Ding, C.; Zou, Z.; Shen, Q. Photoexcited carrier dynamics in colloidal quantum dot solar cells: Insights into individual quantum dots, quantum dot solid films and devices. Chem. Soc. Rev. 2020, 49, 49–84. [Google Scholar] [CrossRef]
- Kirchartz, T.; Deledalle, F.; Tuladhar, P.S.; Durrant, J.R.; Nelson, J. On the Differences between Dark and Light Ideality Factor in Polymer:Fullerene Solar Cells. J. Phys. Chem. Lett. 2013, 4, 2371–2376. [Google Scholar] [CrossRef]
- Breitenstein, O.; Bauer, J.; Lotnyk, A.; Wagner, J.-M. Defect induced non-ideal dark I–V characteristics of solar cells. Superlattices Microstruct. 2009, 45, 182–189. [Google Scholar] [CrossRef]
- Zolfaghari, Z.; Hassanabadi, E.; Pitarch-Tena, D.; Yoon, S.J.; Shariatinia, Z.; van de Lagemaat, J.; Luther, J.M.; Mora-Seró, I. Operation Mechanism of Perovskite Quantum Dot Solar Cells Probed by Impedance Spectroscopy. ACS Energy Lett. 2019, 4, 251–258. [Google Scholar] [CrossRef]
- Tress, W.; Yavari, M.; Domanski, K.; Yadav, P.; Niesen, B.; Correa Baena, J.P.; Hagfeldt, A.; Graetzel, M. Interpretation and evolution of open-circuit voltage, recombination, ideality factor and subgap defect states during reversible light-soaking and irreversible degradation of perovskite solar cells. Energy Environ. Sci. 2018, 11, 151–165. [Google Scholar] [CrossRef]
- Zhang, Z.-Y.; Wang, H.-Y.; Zhang, Y.-X.; Hao, Y.-W.; Sun, C.; Zhang, Y.; Gao, B.-R.; Chen, Q.-D.; Sun, H.-B. The Role of Trap-assisted Recombination in Luminescent Properties of Organometal Halide CH3NH3PbBr3 Perovskite Films and Quantum Dots. Sci. Rep. 2016, 6, 27286. [Google Scholar] [CrossRef] [PubMed]
- Liao, P.; Zhao, X.; Li, G.; Shen, Y.; Wang, M. A New Method for Fitting Current-Voltage Curves of Planar Heterojunction Perovskite Solar Cells. Nano-micro Lett. 2018, 10, 5. [Google Scholar] [CrossRef]
- Cappelletti, M.A.; Casas, G.A.; Cédola, A.P.; Peltzer y Blancá, E.L.; Marí Soucase, B. Study of the reverse saturation current and series resistance of p-p-n perovskite solar cells using the single and double-diode models. Superlattices Microstruct. 2018, 123, 338–348. [Google Scholar] [CrossRef]
- Yang, Z.; Surrente, A.; Galkowski, K.; Miyata, A.; Portugall, O.; Sutton, R.J.; Haghighirad, A.A.; Snaith, H.J.; Maude, D.K.; Plochocka, P.; et al. Impact of the Halide Cage on the Electronic Properties of Fully Inorganic Cesium Lead Halide Perovskites. ACS Energy Lett. 2017, 2, 1621–1627. [Google Scholar] [CrossRef]
- Dong, R.; Fang, Y.; Chae, J.; Dai, J.; Xiao, Z.; Dong, Q.; Yuan, Y.; Centrone, A.; Zeng, X.C.; Huang, J. High-Gain and Low-Driving-Voltage Photodetectors Based on Organolead Triiodide Perovskites. Adv. Mater. 2015, 27, 1912–1918. [Google Scholar] [CrossRef] [PubMed]
- Algadi, H.; Mahata, C.; Woo, J.; Lee, M.; Kim, M.; Lee, T. Enhanced Photoresponsivity of All-Inorganic (CsPbBr3) Perovskite Nanosheets Photodetector with Carbon Nanodots (CDs). Electronics 2019, 8, 678. [Google Scholar] [CrossRef]
- Clifford, J.P.; Konstantatos, G.; Johnston, K.W.; Hoogland, S.; Levina, L.; Sargent, E.H. Fast, sensitive and spectrally tuneable colloidal-quantum-dot photodetectors. Nat. Nanotechnol. 2009, 4, 40–44. [Google Scholar] [CrossRef]
- Li, Y.; Shi, Z.; Lei, L.; Ma, Z.; Zhang, F.; Li, S.; Wu, D.; Xu, T.; Li, X.; Shan, C.; et al. Controllable Vapor-Phase Growth of Inorganic Perovskite Microwire Networks for High-Efficiency and Temperature-Stable Photodetectors. ACS Photonics 2018, 5, 2524–2532. [Google Scholar] [CrossRef]
- Haruta, Y.; Ikenoue, T.; Miyake, M.; Hirato, T. Fabrication of (101)-oriented CsPbBr 3 thick films with high carrier mobility using a mist deposition method. Appl. Phys. Express 2019, 12, 085505. [Google Scholar] [CrossRef]
- Zhang, P.; Zhang, G.; Liu, L.; Ju, D.; Zhang, L.; Cheng, K.; Tao, X. Anisotropic Optoelectronic Properties of Melt-Grown Bulk CsPbBr 3 Single Crystal. J. Phys. Chem. Lett. 2018, 9, 5040–5046. [Google Scholar] [CrossRef] [PubMed]
- Herz, L.M. Charge-Carrier Mobilities in Metal Halide Perovskites: Fundamental Mechanisms and Limits. ACS Energy Lett. 2017, 2, 1539–1548. [Google Scholar] [CrossRef]
- Karani, A.; Yang, L.; Bai, S.; Futscher, M.H.; Snaith, H.J.; Ehrler, B.; Greenham, N.C.; Di, D. Perovskite/Colloidal Quantum Dot Tandem Solar Cells: Theoretical Modeling and Monolithic Structure. ACS Energy Lett. 2018, 3, 869–874. [Google Scholar] [CrossRef]
- Han, J.; Luo, S.; Yin, X.; Zhou, Y.; Nan, H.; Li, J.; Li, X.; Oron, D.; Shen, H.; Lin, H. Hybrid PbS Quantum-Dot-in-Perovskite for High-Efficiency Perovskite Solar Cell. Small 2018, 14, 1801016. [Google Scholar] [CrossRef]
- Sanchez, R.S.; de la Fuente, M.S.; Suarez, I.; Muñoz-Matutano, G.; Martinez-Pastor, J.P.; Mora-Sero, I. Tunable light emission by exciplex state formation between hybrid halide perovskite and core/shell quantum dots: Implications in advanced LEDs and photovoltaics. Sci. Adv. 2016, 2, e1501104. [Google Scholar] [CrossRef]
- Yu, Y.; Zhang, Y.; Jin, L.; Chen, Z.; Li, Y.; Cao, M.; Che, Y.; Yao, J.; Li, Q. Highly photosensitive vertical photodetectors based on CsPbBr3 and PbS quantum dot layered heterojunction. In Proceedings of the Optoelectronic Devices and Integration VII, Beijing, China, 5 November 2018; Volume 10814, p. 38. [Google Scholar]
| Sample | |||||||
|---|---|---|---|---|---|---|---|
| Pristine | 4 ± 2 | (3 ± 1) × 104 | 6 ± 2 | (5 ± 2) × 10−8 | (5 ± 3) × 10−8 | (2 ± 1) × 10−3 | (5 ± 1) × 10−1 |
| MPA | 8 ± 3 | 50 ± 20 | 5 ± 1 | (4 ± 1) × 10−5 | 0 | (1.5 ± 1.0) × 10−1 | (5 ± 1) × 10−1 |
| Sample | ||||
|---|---|---|---|---|
| MPA | 0.8 | 0.33 | 0.35 | 10−3 |
| Pristine | 0.03 | 0.03 | 0.40 | 10−4 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Navarro Arenas, J.; Soosaimanickam, A.; Pashaei Adl, H.; Abargues, R.; P. Boix, P.; Rodríguez-Cantó, P.J.; Martínez-Pastor, J.P. Ligand-Length Modification in CsPbBr3 Perovskite Nanocrystals and Bilayers with PbS Quantum Dots for Improved Photodetection Performance. Nanomaterials 2020, 10, 1297. https://doi.org/10.3390/nano10071297
Navarro Arenas J, Soosaimanickam A, Pashaei Adl H, Abargues R, P. Boix P, Rodríguez-Cantó PJ, Martínez-Pastor JP. Ligand-Length Modification in CsPbBr3 Perovskite Nanocrystals and Bilayers with PbS Quantum Dots for Improved Photodetection Performance. Nanomaterials. 2020; 10(7):1297. https://doi.org/10.3390/nano10071297
Chicago/Turabian StyleNavarro Arenas, Juan, Ananthakumar Soosaimanickam, Hamid Pashaei Adl, Rafael Abargues, Pablo P. Boix, Pedro J. Rodríguez-Cantó, and Juan P. Martínez-Pastor. 2020. "Ligand-Length Modification in CsPbBr3 Perovskite Nanocrystals and Bilayers with PbS Quantum Dots for Improved Photodetection Performance" Nanomaterials 10, no. 7: 1297. https://doi.org/10.3390/nano10071297
APA StyleNavarro Arenas, J., Soosaimanickam, A., Pashaei Adl, H., Abargues, R., P. Boix, P., Rodríguez-Cantó, P. J., & Martínez-Pastor, J. P. (2020). Ligand-Length Modification in CsPbBr3 Perovskite Nanocrystals and Bilayers with PbS Quantum Dots for Improved Photodetection Performance. Nanomaterials, 10(7), 1297. https://doi.org/10.3390/nano10071297





