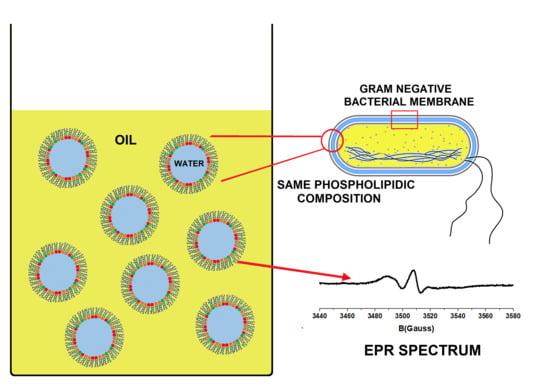
Bioinspired Nanoemulsions Stabilized by Phosphoethanolamine and Phosphoglycerol Lipids
Abstract
1. Introduction
2. Materials and Methods
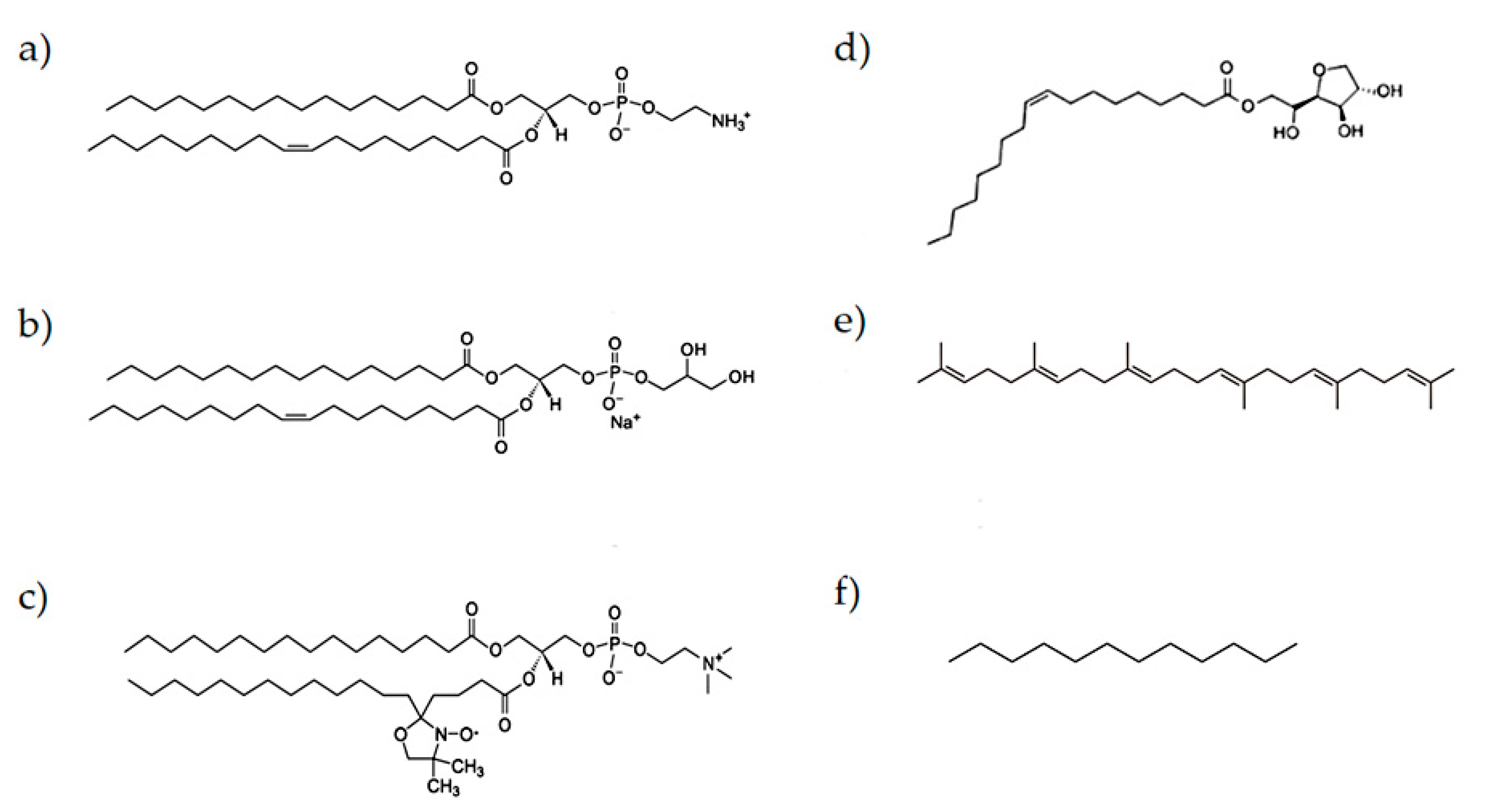
2.1. Materials
2.2. Samples Preparation
2.3. DLS Measurements
2.4. EPR Characterization
2.5. Interfacial Tension Measurements
2.6. Statistical Analysis
3. Results and Discussion
3.1. Emulsion Design
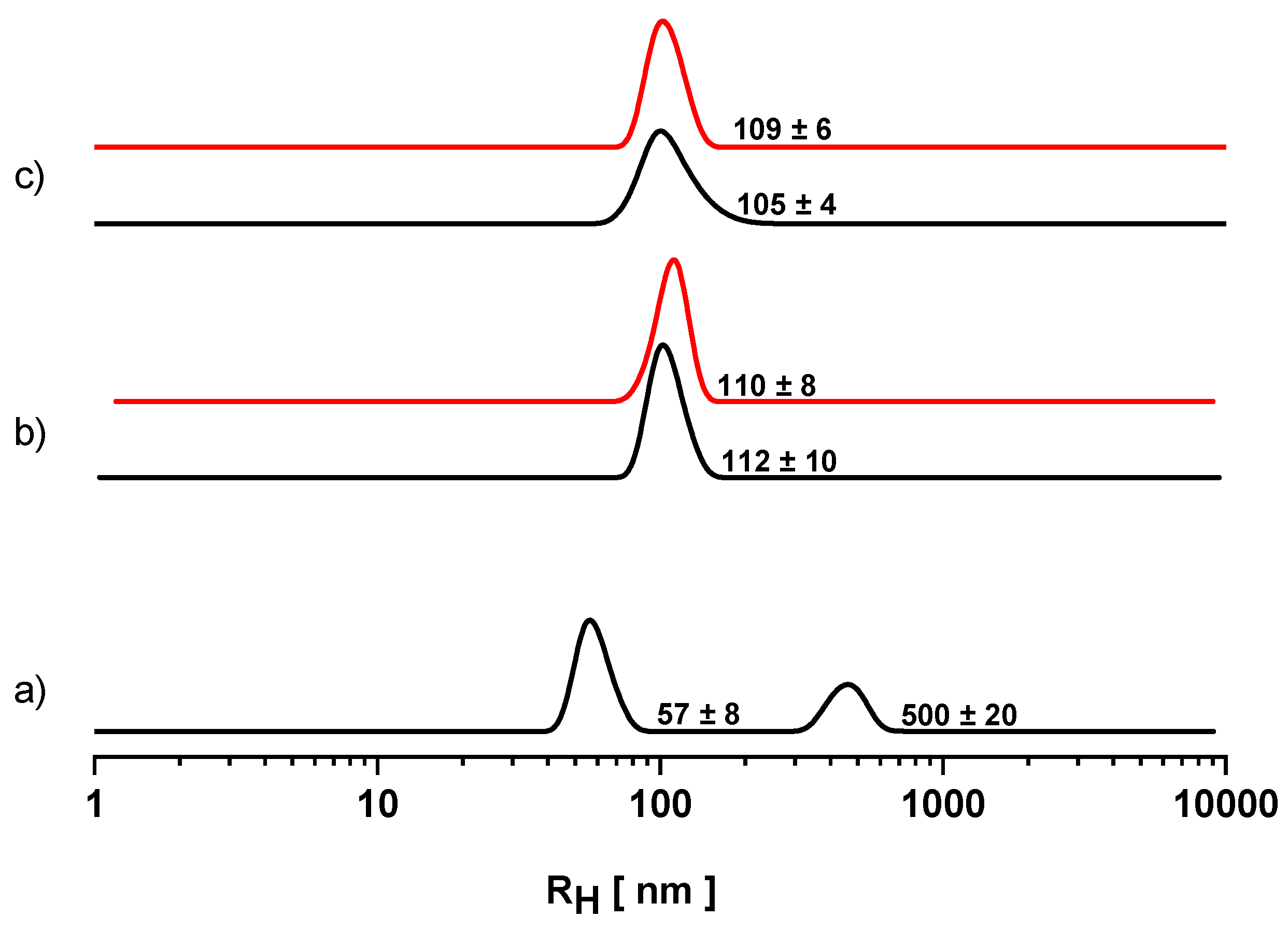
3.2. Dynamic Light Scattering
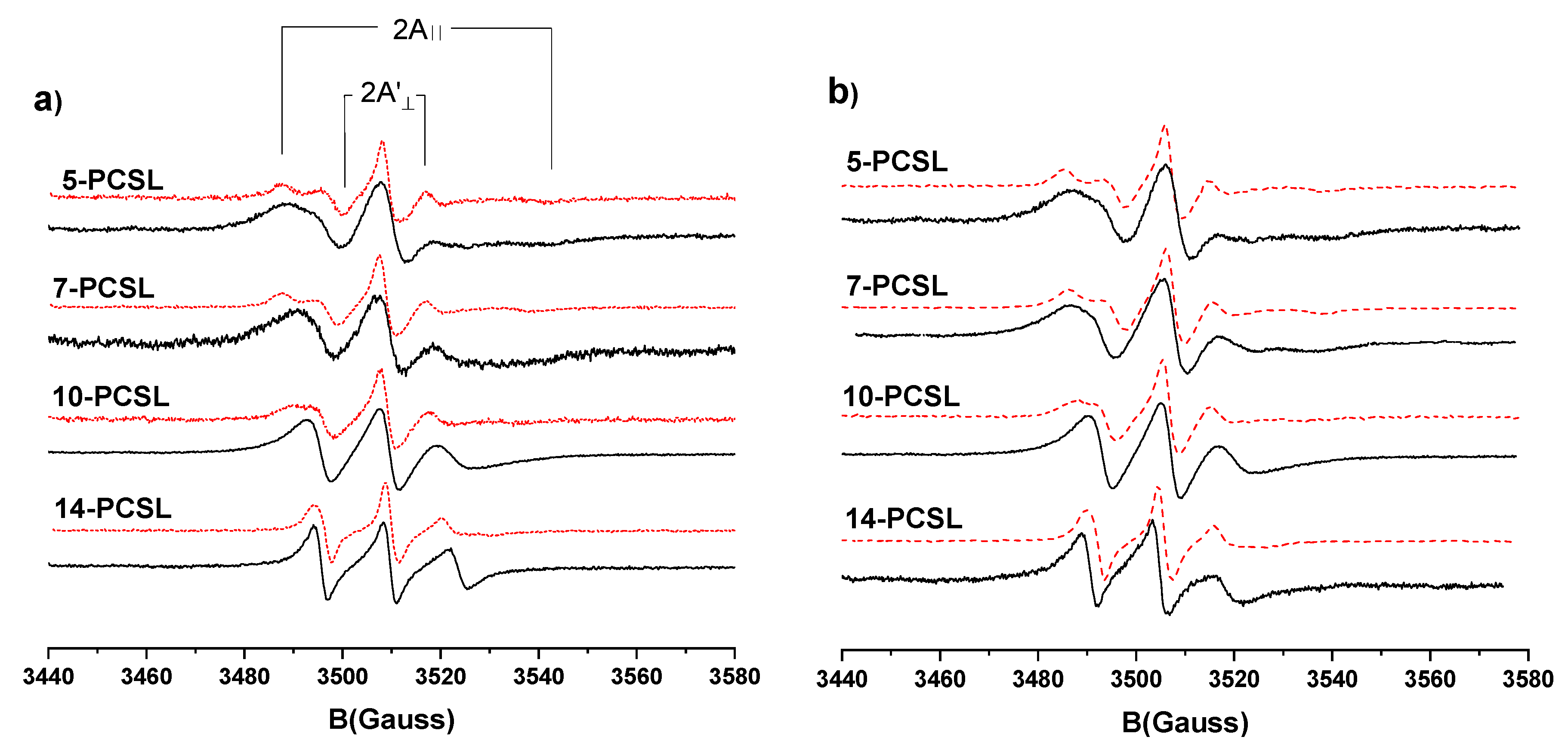
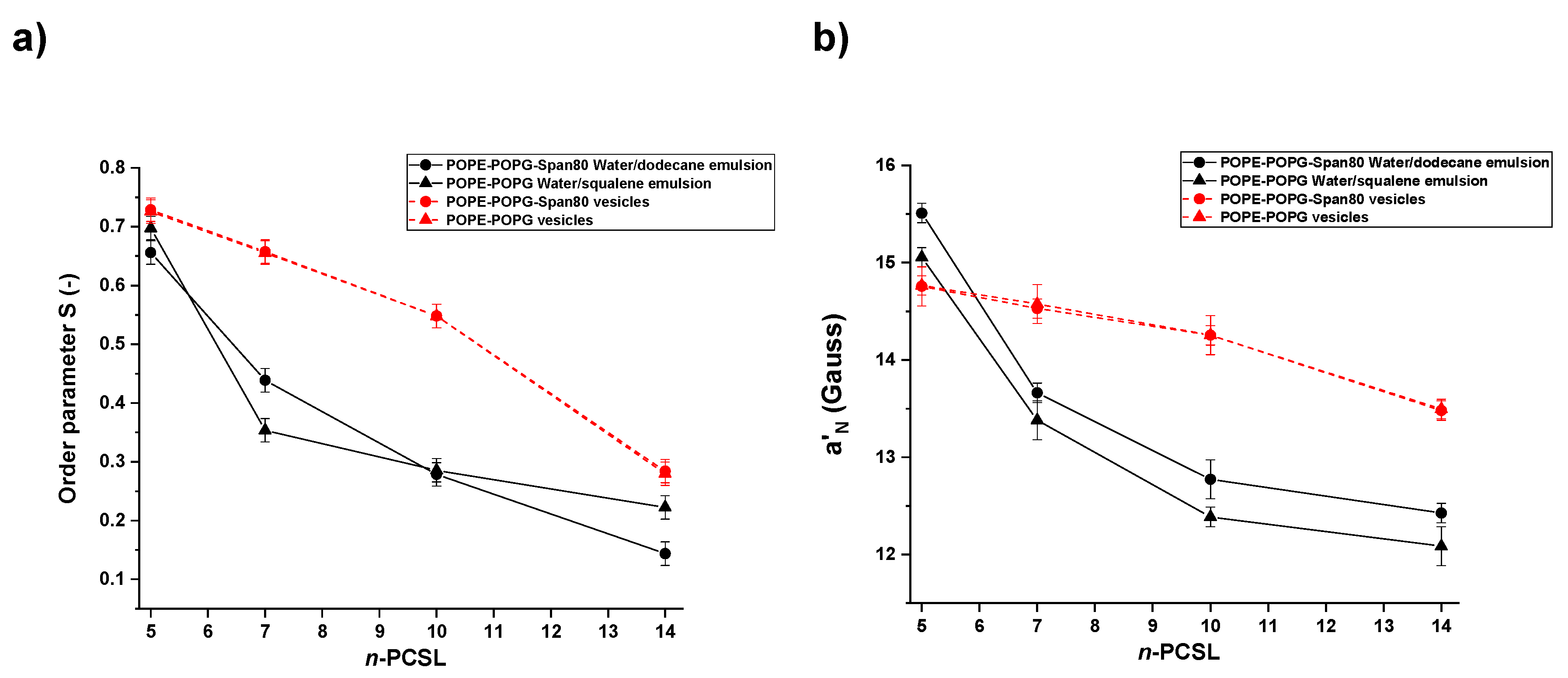
3.3. Electron Paramagnetic Resonance
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kabalnov, A.; Wennerström, H. Macroemulsion Stability: The Oriented Wedge Theory Revisited. Langmuir 1996, 12, 276–292. [Google Scholar] [CrossRef]
- Bueschelberger, H.G. Lecithins. In Emulsifiers in Food Technology; Whitehurst, R.J., Ed.; Blackwell Publishing: Oxford, UK, 2004. [Google Scholar]
- Lucks, J.S.; Müller, B.W.; Klütsch, K. Parenteral Fat Emulsions: Structure, Stability, and Applications. In Pharmaceutical Emulsions and Suspensions, 2nd ed.; Nielloud, F., Marti-Mestres, G., Eds.; CRC Press: New York, NY, USA, 2000; pp. 229–245. [Google Scholar]
- Friberg, S. Liquid crystalline phases in emulsions. J. Colloid Interface Sci. 1971, 37, 291–295. [Google Scholar] [CrossRef]
- Handa, T.; Saito, H.; Miyajima, K. Phospholipid monolayers at the triolein-saline interface: production of microemulsion particles and conversion of monolayers to bilayers. Biochemistry 1990, 29, 2884–2890. [Google Scholar] [CrossRef] [PubMed]
- Nordén, T.P.; Siekmann, B.; Lundquist, S.; Malmsten, M. Physicochemical characterisation of a drug-containing phospholipid-stabilised o/w emulsion for intravenous administration. Eur. J. Pharm. Sci. 2001, 13, 393–401. [Google Scholar] [CrossRef]
- Klang, V.; Valenta, C. Lecithin-based nanoemulsions. J. Drug Deliv. Sci. Technol. 2011, 21, 55–76. [Google Scholar] [CrossRef]
- Ushikubo, F.; Cunha, R. Stability mechanisms of liquid water-in-oil emulsions. Food Hydrocoll. 2014, 34, 145–153. [Google Scholar] [CrossRef]
- Pautot, S.; Frisken, B.; Weitz, D.A. Production of unilamellar vesicles using an inverted emulsion. Langmuir 2003, 19, 2870–2879. [Google Scholar] [CrossRef]
- Sommerling, J.-H.; de Matos, M.B.C.; Hildebrandt, E.; Dessy, A.; Kok, R.J.; Nirschl, H.; Leneweit, G. Instability mechanisms of water-in-oil nanoemulsions with phospholipids: Temporal and morphological structures. Langmuir 2018, 34, 572–584. [Google Scholar] [CrossRef]
- Vrignaud, S.; Anton, N.; Gayet, P.; Benoit, J.-P.; Saulnier, P. Reverse micelle-loaded lipid nanocarriers: A novel drug delivery system for the sustained release of doxorubicin hydrochloride. Eur. J. Pharm. Biopharm. 2011, 79, 197–204. [Google Scholar] [CrossRef]
- Zhu, Q.; Pan, Y.; Jia, X.; Li, J.; Zhang, M.; Yin, L. Review on the stability mechanism and application of water-in-oil emulsions encapsulating various additives. Compr. Rev. Food Sci. F 2019, 18, 1660–1675. [Google Scholar] [CrossRef]
- Pautot, S.; Frisken, B.; Weitz, D.A. Engineering asymmetric vesicles. Proc. Natl. Acad. Sci. USA 2003, 100, 10718–10721. [Google Scholar] [CrossRef]
- Shanthilal, J.a.B.; Bhattacharya, S. Nanoparticles and nanotechnology in food. In Conventional and Advanced Food Processing Technologies; Bhattacharya, S., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 2014; pp. 567–594. [Google Scholar]
- Gnanadesigan, M.; Nandagopalan, V.; Kapildev, G.; Gundappa, M. Nano drugs for curing malaria: The plausibility. In Applications of Targeted Nano Drugs and Delivery Systems; Mohapatra, S.S., Ranjan, S., Dasgupta, N., Mishra, R.K., Thomas, S., Eds.; Elsevier: Amsterdam, NL, USA, 2019; pp. 451–467. [Google Scholar]
- Whitesides, G.M. Bioinspiration: something for everyone. Interface Focus 2015, 5, 20150031. [Google Scholar] [CrossRef]
- Delcour, A.H. Outer membrane permeability and antibiotic resistance. Biochim. Biophys. Acta 2009, 1794, 808–816. [Google Scholar] [CrossRef]
- Zimmerberg, J.; Kozlov, M.M. How proteins produce cellular membrane curvature. Nat. Rev. Mol. Cell Biol. 2006, 7, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Annunziata, O. Effect of macromolecular polydispersity on diffusion coefficients measured by rayleigh interferometry. J. Phys. Chem. B 2008, 112, 3633–3643. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lomakin, A.; Teplow, D.; Benedek, G. Quasielastic light scattering for protein assembly studies. Methods Mol. Biol. 2005, 299, 153–174. [Google Scholar] [CrossRef]
- Russo Krauss, I.; Imperatore, R.; De Santis, A.; Luchini, A.; Paduano, L.; D’Errico, G. Structure and dynamics of cetyltrimethylammonium chloride-sodium dodecylsulfate (CTAC-SDS) catanionic vesicles: High-value nano-vehicles from low-cost surfactants. J. Colloid Interface Sci. 2017, 501, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Caudwell, D.R.; Trusler, J.P.M.; Vesovic, V.; Wakeham, W.A. The viscosity and density of n-dodecane and n-octadecane at pressures up to 200 MPa and temperatures up to 473 K. Int. J. Thermophys. 2004, 25, 1339. [Google Scholar] [CrossRef]
- Popa, O.; Băbeanu, N.E.; Popa, I.; Niță, S.; Dinu-Pârvu, C.E. Methods for obtaining and determination of squalene from natural sources. Biomed. Res. Int. 2015, 367202. [Google Scholar] [CrossRef]
- Dvolaitzky, C.T.M. Spin labels. In Surfactant Solutions: New Methods of Investigations; Zana, R., Ed.; Marcel Dekker Inc: New York, NY, USA, 1987; pp. 359–397. [Google Scholar]
- Kim, H.; Kim, K.; Lee, H.-R.; Jo, H.; Jeong, D.-w.; Ryu, J.; Gweon, D.-G.; Choi, S.Q. Formation of stable adhesive water-in-oil emulsions using a phospholipid and cosurfactants. J. Ind. Eng. Chem. 2017, 55, 198–203. [Google Scholar] [CrossRef]
- Fukuhira, Y.; Yabu, H.; Ijiro, K.; Shimomura, M. Interfacial tension governs the formation of self-organized honeycomb-patterned polymer films. Soft Matter 2009, 5, 2037–2041. [Google Scholar] [CrossRef]
- Ishii, F. Phospholipids in emulsion and dispersion systems. J. Jpn. Oil Chem. Soc. 1992, 41, 787–792. [Google Scholar] [CrossRef][Green Version]
- Whitaker, J.R.; Voragen, A.G.J.; Wong, D.W.S. Handbook of Food Enzymology; CRC Press: New York, NY, USA, 2002. [Google Scholar]
- Ciobanasu, C.; Rzeszutek, A.; Kubitscheck, U.; Willumeit, R. NKCS, a mutant of the NK-2 peptide, causes severe distortions and perforations in bacterial, but not human model lipid membranes. Molecules 2015, 20, 6941–6958. [Google Scholar] [CrossRef]
- Alam, M.; Aramaki, K. Liquid crystal-based emulsions: Progress and prospects. J. Oleo Sci. 2014, 63, 97–108. [Google Scholar] [CrossRef]
- Fox, C.B. Squalene emulsions for parenteral vaccine and drug delivery. Molecules 2009, 14, 3286–3312. [Google Scholar] [CrossRef]
- Marsh, D. Handbook of Lipid Bilayers, 2nd ed.; CRC Press: New York, NY, USA, 2013. [Google Scholar]
- Knoth, A.; Scherze, I.; Muschiolik, G. Stability of water-in-oil-emulsions containing phosphatidylcholine-depleted lecithin. Food Hydrocoll. 2005, 19, 635–640. [Google Scholar] [CrossRef]
- Eastoe, J. Microemulsions. In Colloid Science, 2nd ed.; Cosgrove, T., Ed.; John Wiley & Sons Ltd.: Hoboken, NJ, USA, 2010; pp. 91–116. [Google Scholar]
- Kassem, M.G.A.; Ahmed, A.-M.M.; Abdel-Rahman, H.H.; Moustafa, A.H.E. Use of Span 80 and Tween 80 for blending gasoline and alcohol in spark ignition engines. Energy Rep. 2019, 5, 221–230. [Google Scholar] [CrossRef]
- Mollet, H.; Grubermann, D. Formulation Technology; Wiley-VCH: Weinheim, Germany, 2001; pp. 59–104. [Google Scholar]
- Marsh, D. Electron spin resonance in membrane research: protein-lipid interactions from challenging beginnings to state of the art. Eur. Biophys. J. 2010, 39, 513–525. [Google Scholar] [CrossRef]
- Fedotov, V.D.; Vylegzhanina, N.N.; Altshuler, A.E.; Shlenkin, V.I.; Zuev, Y.F.; Garti, N. An electron spin resonance study of the soybean phosphatidylcholine-based reversed micelles. Appl. Magn. Reson. 1998, 14, 497–512. [Google Scholar] [CrossRef]
- Vitiello, G.; Falanga, A.; Petruk, A.A.; Merlino, A.; Fragneto, G.; Paduano, L.; Galdiero, S.; D’Errico, G. Fusion of raft-like lipid bilayers operated by a membranotropic domain of the HSV-type I glycoprotein gH occurs through a cholesterol-dependent mechanism. Soft Matter 2015, 11, 3003–3016. [Google Scholar] [CrossRef]
- Vitiello, G.; Fragneto, G.; Petruk, A.A.; Falanga, A.; Galdiero, S.; D’Ursi, A.M.; Merlino, A.; D’Errico, G. Cholesterol modulates the fusogenic activity of a membranotropic domain of the FIV glycoprotein gp36. Soft Matter 2013, 9, 6442–6456. [Google Scholar] [CrossRef]
- Hildebrandt, E.; Dessy, A.; Sommerling, J.-H.; Guthausen, G.; Nirschl, H.; Leneweit, G. Interactions between phospholipids and organic phases: Insights into lipoproteins and nanoemulsions. Langmuir 2016, 32, 5821–5829. [Google Scholar] [CrossRef] [PubMed]
- Pradilla, D.; Barrera, A.; Saetran, M.G.; Sorland, G.; Alvarez, O. Mechanisms of physical stabilization of concentrated water-in-oil emulsions probed by pulse field gradient nuclear magnetic resonance and rheology through a multiscale approach. Langmuir 2018, 34, 9489–9499. [Google Scholar] [CrossRef] [PubMed]
| System | Interfacial Tension γ (mN m−1) |
|---|---|
| dodecane/water | 52 ± 1 |
| squalene/water | 49 ± 1 |
| dodecane/water POPE-POPG | 20 ± 1 |
| squalene/water POPE-POPG | 12 ± 1 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caianiello, C.; D'Avino, M.; Cavasso, D.; Paduano, L.; D'Errico, G. Bioinspired Nanoemulsions Stabilized by Phosphoethanolamine and Phosphoglycerol Lipids. Nanomaterials 2020, 10, 1185. https://doi.org/10.3390/nano10061185
Caianiello C, D'Avino M, Cavasso D, Paduano L, D'Errico G. Bioinspired Nanoemulsions Stabilized by Phosphoethanolamine and Phosphoglycerol Lipids. Nanomaterials. 2020; 10(6):1185. https://doi.org/10.3390/nano10061185
Chicago/Turabian StyleCaianiello, Carlo, Marcellino D'Avino, Domenico Cavasso, Luigi Paduano, and Gerardino D'Errico. 2020. "Bioinspired Nanoemulsions Stabilized by Phosphoethanolamine and Phosphoglycerol Lipids" Nanomaterials 10, no. 6: 1185. https://doi.org/10.3390/nano10061185
APA StyleCaianiello, C., D'Avino, M., Cavasso, D., Paduano, L., & D'Errico, G. (2020). Bioinspired Nanoemulsions Stabilized by Phosphoethanolamine and Phosphoglycerol Lipids. Nanomaterials, 10(6), 1185. https://doi.org/10.3390/nano10061185