Interfacial Modification of Mesoporous TiO2 Films with PbI2-Ethanolamine-Dimethyl Sulfoxide Solution for CsPbIBr2 Perovskite Solar Cells
Abstract
1. Introduction
2. Experiment
2.1. Device Fabrication
2.2. Device Characterizations
3. Results and Discussion
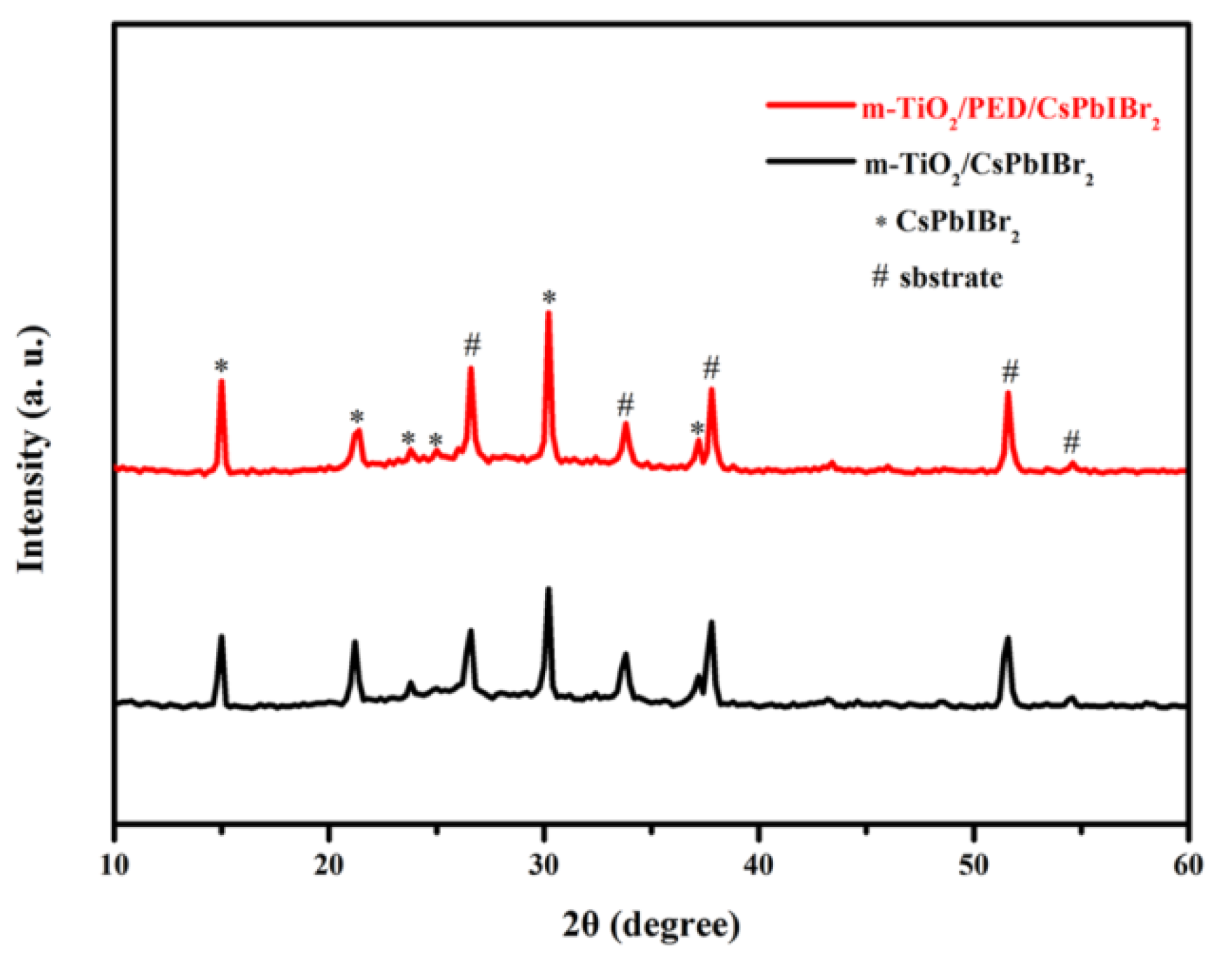
3.1. X-ray Diffraction Studies and Compositional Analysis
3.2. Morphological Characterization
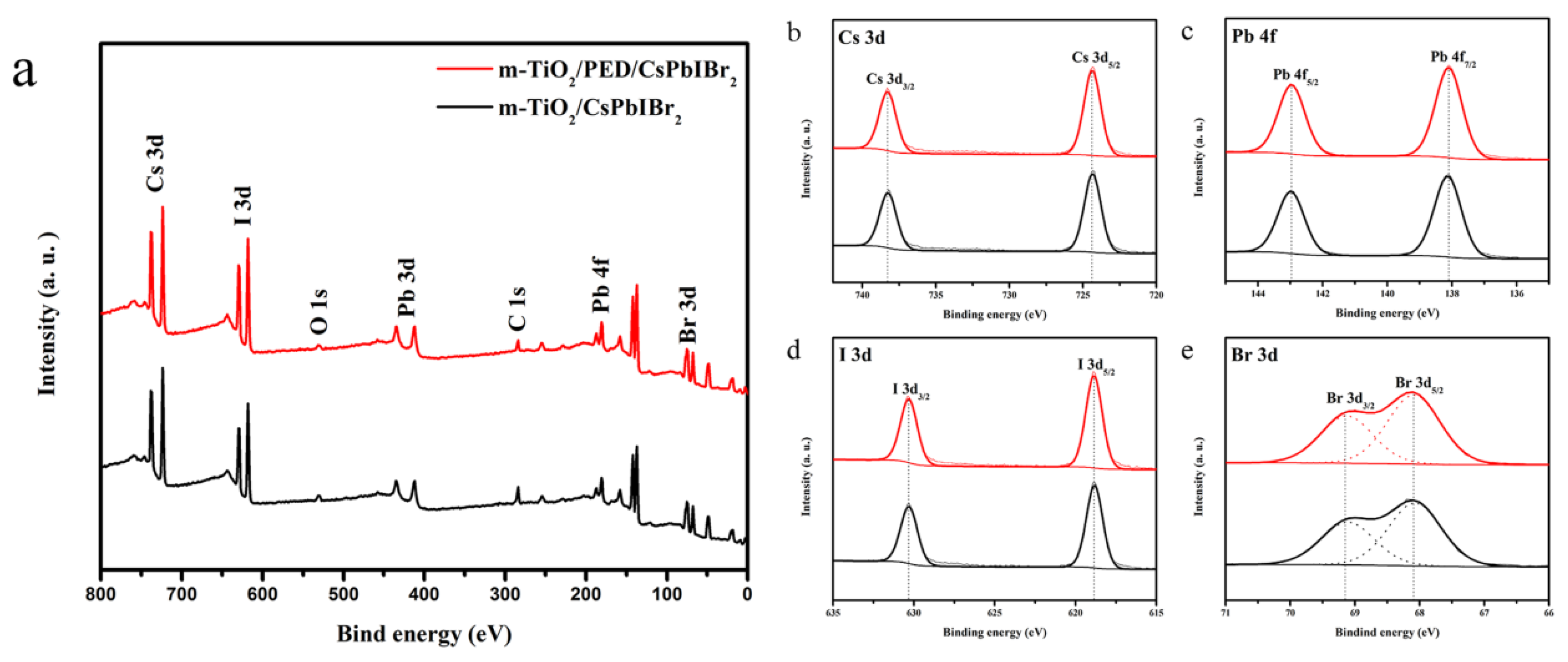
3.3. XPS Analysis
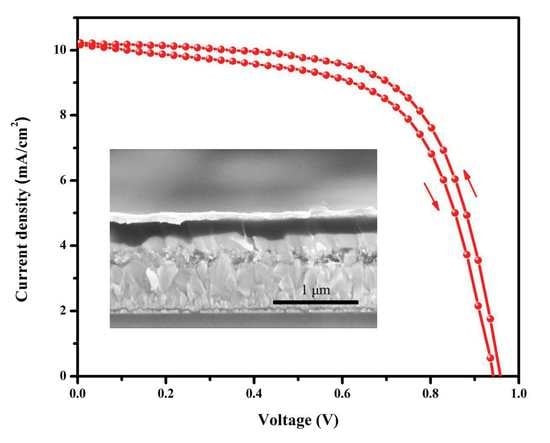
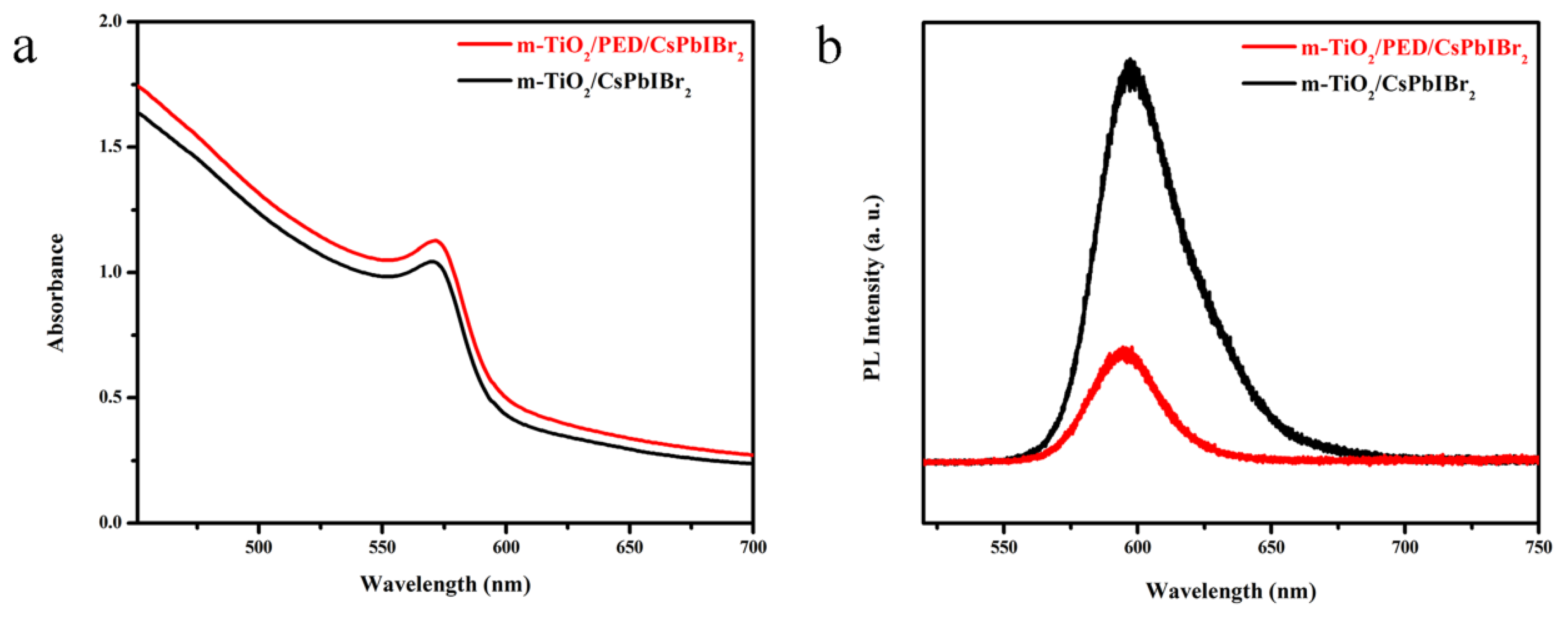
3.4. Optical Properties and Photovoltaic Performances
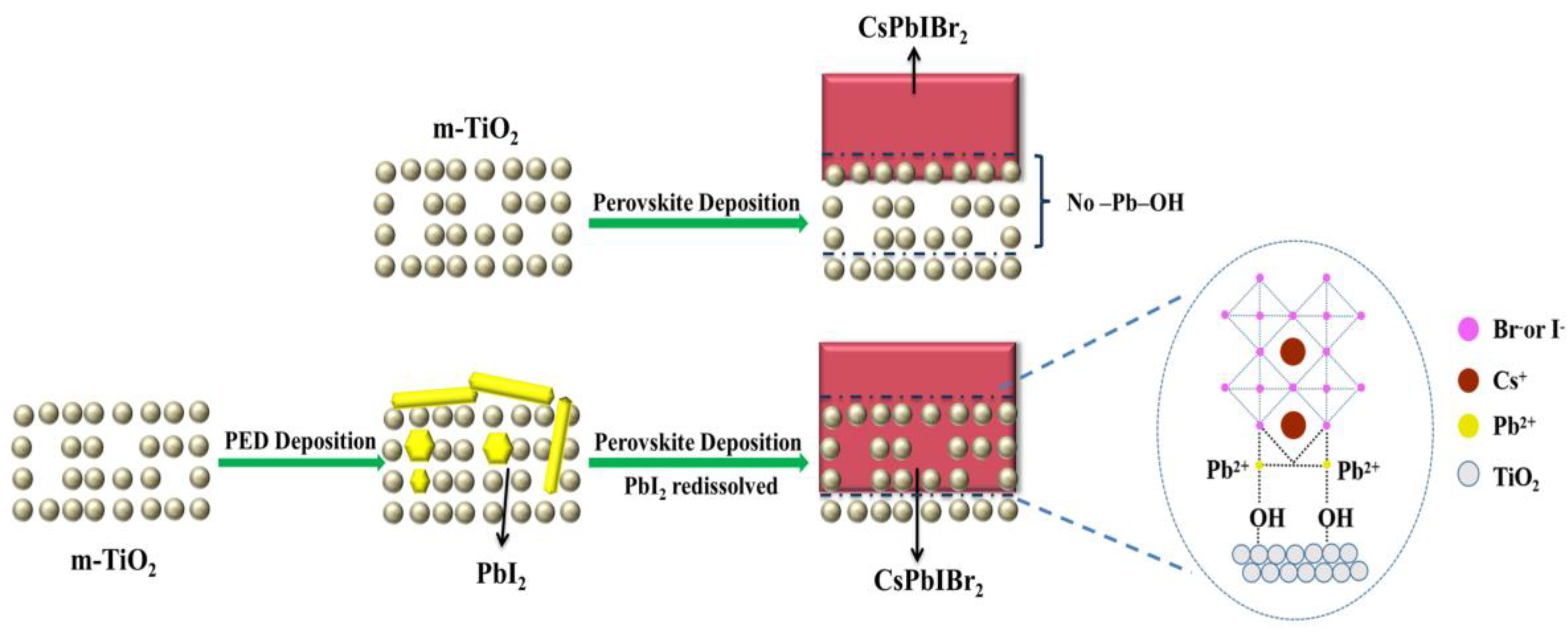
3.5. Possible Mechanism
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Conings, B.; Drijkoningen, J.; Gauquelin, N.; Babayigit, A.; D’Haen, J.; D’Olieslaeger, L.; Ethirajan, A.; Verbeeck, J.; Manca, J.; Mosconi, E.; et al. Intrinsic thermal instability of methylammonium lead trihalide perovskite. Adv. Energy Mater. 2015, 5, 1500477. [Google Scholar] [CrossRef]
- Han, Y.; Meyer, S.; Dkhissi, Y.; Weber, K.; Pringle, J.M.; Bach, U.; Spiccia, L.; Cheng, Y.B. Degradation observations of encapsulated planar CH3NH3PbI3 perovskite solar cells at high temperatures and humidity. J. Mater. Chem. A. 2015, 3, 8139–8147. [Google Scholar] [CrossRef]
- Park, Y.H.; Jeong, I.; Bae, S.; Son, H.J.; Lee, P.; Lee, J.; Lee, C.H.; Ko, M.J. Inorganic rubidium cation as an enhancer for photovoltaic performance and moisture stability of HC(NH2)2PbI3 perovskite solar cells. Adv. Funct. Mater. 2017, 27, 1605988. [Google Scholar] [CrossRef]
- Akbulatov, A.F.; Luchkin, S.Y.; Frolova, L.A.; Dremova, N.N.; Gerasimov, K.L.; Zhidkov, I.S.; Anokhin, D.V.; Kurmaev, E.Z.; Stevenson, K.J.; Troshin, P.A. Probing the intrinsic thermal and photochemical stability of hybrid and inorganic lead halide perovskites. J. Phys. Chem. Lett. 2017, 8, 1211–1218. [Google Scholar] [CrossRef]
- Zhou, W.; Zhao, Y.C.; Zhou, X.; Fu, R.; Li, Q.; Zhao, Y.; Liu, K.H.; Yu, D.P.; Zhao, Q. Light-independent ionic transport in inorganic perovskite and ultrastable Cs-based perovskite solar cells. J. Phys. Chem. Lett. 2017, 8, 4124–4128. [Google Scholar] [CrossRef]
- Sutter-Fella, C.M.; Ngo, Q.P.; Cefarin, N.; Gardner, K.L.; Tamura, N.; Stan, C.V.; Drisdell, W.S.; Javey, A.; Toma, F.M.; Sharp, I.D. Cation-dependent light-induced halide demixing in hybrid organic-inorganic perovskites. Nano Lett. 2018, 18, 3473–3480. [Google Scholar] [CrossRef]
- Kojima, A.; Teshima, K.; Shirai, Y.; Miyasaka, T. Organometal halide perovskites as visible-light sensitizers for photovoltaic cells. J. Am. Chem. Soc. 2009, 131, 6050–6051. [Google Scholar] [CrossRef]
- Jeon, N.J.; Na, H.; Jung, E.H.; Yang, T.-Y.; Lee, Y.G.; Kim, G.; Shin, H.-W.; Seok, S.I.; Lee, J.; Seo, J. A fluorene-terminated hole-transporting material for highly efficient and stable perovskite solar cells. Nat. Energy 2018, 3, 682–688. [Google Scholar] [CrossRef]
- Christians, J.A.; Schulz, P.; Tinkham, J.S.; Schloemer, T.H.; Harvey, S.P.; Tremolet de Villers, B.J.; Sellinger, A.; Berry, J.J.; Luther, J.M. Tailored interfaces of unencapsulated perovskite solar cells for >1,000 hour operational stability. Nat. Energy 2018, 3, 68–74. [Google Scholar] [CrossRef]
- Chen, H.; Xiang, S.; Li, W.; Liu, H.; Zhu, L.; Yang, S. Inorganic perovskite solar cells: A rapidly growing field. Solar RRL 2018, 2, 1700188. [Google Scholar] [CrossRef]
- Bian, H.; Bai, D.L.; Jin, Z.W.; Wang, K.; Liang, L.; Wang, H.R.; Zhang, J.R.; Wang, Q.; (Frank)Liu, S.Z. Graded bandgap CsPbI2+xBr1-x perovskite solar cells with a stabilized efficiency of 14.4%. Joule 2018, 2, 1500–1510. [Google Scholar] [CrossRef]
- Dastidar, S.; Li, S.; Smolin, S.Y.; Baxter, J.B.; Fafarman, A.T. Slow electron–hole recombination in lead iodide perovskites does not require a molecular dipole. ACS Energy Lett. 2017, 2, 2239–2244. [Google Scholar] [CrossRef]
- Liang, J.; Liu, J.; Jin, Z. All-inorganic halide perovskites for optoelectronics: Progress and prospects. Solar RRL 2017, 1, 1700086. [Google Scholar] [CrossRef]
- Wang, Q.; Zheng, X.; Deng, Y.; Zhao, J.; Chen, Z.; Huang, J. Stabilizing the α-Phase of CsPbI3 perovskite by sulfobetaine zwitterions in one-step spin-coating films. Joule 2017, 1, 371–382. [Google Scholar] [CrossRef]
- Eperon, G.E.; Paterno, G.M.; Sutton, R.J.; Zampetti, A.; Haghighirad, A.A.; Cacialli, F.; Snaith, H.J. Inorganic caesium lead iodide perovskite solar cells. J. Mater. Chem. A. 2015, 3, 19688–19695. [Google Scholar] [CrossRef]
- Hu, Y.; Bai, F.; Liu, X.; Ji, Q.; Miao, X.; Qiu, T.; Zhang, S. Bismuth incorporation stabilized α-CsPbI3 for fully inorganic perovskite solar cells. ACS Energy Lett. 2017, 2, 2219–2227. [Google Scholar] [CrossRef]
- Chen, C.Y.; Lin, H.Y.; Chiang, K.M.; Tsai, W.L.; Huang, Y.C.; Tsao, C.S.; Lin, H.-W. All-vacuum-deposited stoichiometrically balanced inorganic cesium lead halide perovskite solar cells with stabilized efficiency exceeding 11%. Adv. Mater. 2017, 29, 1605290. [Google Scholar] [CrossRef]
- Xiang, W.; Wang, Z.; Kubicki, D.J.; Tress, W.; Luo, J.; Prochowicz, D.; Akin, S.; Emsley, L.; Zhou, J.; Dietler, G.; et al. Hagfeldt, Europium-doped CsPbI2Br for stable and highly efficient inorganic perovskite solar cells. Joule 2019, 3, 205–214. [Google Scholar] [CrossRef]
- Shen, E.; Chen, J.; Tian, Y.; Luo, Y.; Shen, Y.; Sun, Q.; Jin, T.; Shi, G.; Li, Y.; Tang, J. Interfacial energy level tuning for efficient and thermostable CsPbI2Br perovskite solar cells. Adv. Sci. 2020, 7, 1901952. [Google Scholar] [CrossRef]
- Rao, H.; Ye, S.; Gu, F.; Zhao, Z.; Liu, Z.; Bian, Z.; Huang, C. Morphology controlling of all-inorganic perovskite at low temperature for efficient rigid and flexible solar cells. Adv. Energy Mater. 2018, 8, 1800758. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, T.; Kan, M.; Zhao, Y. Bifunctional stabilization of all-inorganic α-CsPbI3 perovskite for 17% efficiency photovoltaics. J. Am. Chem. Soc. 2018, 140, 12345–12348. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Zhao, S.; Liu, A.; Kamata, Y.; Teo, S.; Yang, S.; Xu, Z.; Hayase, S.; Ma, T. Niobium, Incorporation into CsPbI2Br for stable and efficient all-inorganic perovskite solar cells. ACS Appl. Mater. Interfaces 2019, 11, 19994–20003. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.S.; Jin, Z.W.; Zhang, J.R.; Bai, D.L.; Bian, H.; Wang, K.; Sun, J.; Wang, Q.; Frank Liu, S.Z. All-ambient processed binary CsPbBr3-CsPb2Br5 perovskites with synergistic enhancement for high-efficiency Cs-Pb-Br-based solar cells. ACS Appl. Mater. Interfaces. 2018, 10, 7145–7154. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.; Wang, C.; Wang, Y.; Xu, Z.; Lu, Z.; Ma, Y.; Zhu, H.; Hu, Y.; Xiao, C.; Yi, X.; et al. All-inorganic perovskite solar cells. J. Am. Chem. Soc. 2016, 138, 15829–15832. [Google Scholar] [CrossRef]
- Duan, J.; Zhao, Y.; He, B.; Tang, Q. High-purity inorganic perovskite films for solar cells with 9.72% efficiency. Angew. Chem. 2018, 57, 3787–3791. [Google Scholar] [CrossRef]
- Lau, C.F.J.; Deng, X.F.; Ma, Q.S.; Zheng, J.H.; Yun, J.S.; Green, M.A.; Huang, S.J.; Ho-Baillie, A.W.Y. CsPbIBr2 perovskite solar cell by spray-assisted deposition. ACS Energy Lett. 2016, 1, 573–577. [Google Scholar] [CrossRef]
- Ma, Q.; Huang, S.; Wen, X.; Green, M.A.; Ho-Baillie, A.W.Y. Hole transport layer Free inorganic CsPbIBr2 perovskite solar cell by dual source thermal evaporation. Adv. Energy Mater. 2016, 6, 1502202. [Google Scholar] [CrossRef]
- Lin, J.; Lai, M.; Dou, L.; Kley, C.S.; Chen, H.; Peng, F.; Sun, J.; Lu, D.; Hawks, S.A.; Xie, C.; et al. Thermochromic halide perovskite solar cells. Nat. Mater. 2018, 17, 261–267. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhu, W.; Chen, D.; Zhang, Z.; Lin, Z.; Chang, J.; Zhang, J.; Zhang, C.; Hao, Y. Light processing enables efficient carbon-based, all-inorganic planar CsPbIBr2 solar cells with high photovoltages. ACS Appl. Mater. Interfaces 2019, 11, 2997–3005. [Google Scholar] [CrossRef]
- Sutton, R.J.; Eperon, G.E.; Miranda, L.; Parrott, E.S.; Kamino, B.A.; Patel, J.B.; Horantner, M.T.; Johnston, M.B.; Haghighirad, A.A.; Moore, D.T. Bandgap-tunable cesium lead halide perovskites with high thermal stability for efficient solar cells. Adv. Energy Mater. 2016, 6, 1502458. [Google Scholar] [CrossRef]
- Guo, Y.; Yin, X.; Liu, J.; Wen, S.; Wu, Y.; Que, W. Inorganic CsPbIBr2-based perovskite solar cells: Fabrication technique modification and efficiency improvement. Sol. RRL 2019, 3, 1900135. [Google Scholar] [CrossRef]
- Liu, C.; Li, W.; Chen, J.; Fan, J.; Mai, Y.; Schropp, R.E. Ultra-thin MoOx as cathode buffer layer for the improvement of all-inorganic CsPbIBr2 perovskite solar cells. Nano Energy 2017, 41, 75–83. [Google Scholar] [CrossRef]
- Liang, J.; Liu, Z.H.; Qiu, L.B.; Hawash, Z.; Meng, L.Q.; Wu, Z.F.; Jiang, Y.; Ono, L.K.; Qi, Y.B. Enhancing optical, electronic, crystalline, and morphological properties of cesium lead halide by Mn substitution for high-stability all-inorganic perovskite solar cells with carbon electrodes. Adv. Energy Mater. 2018, 8, 1800504. [Google Scholar] [CrossRef]
- Liang, J.; Zhao, P.Y.; Wang, C.X.; Wang, Y.R.; Hu, Y.; Zhu, G.Y.; Ma, L.B.; Liu, J.; Jin, Z. CsPb0.9Sn0.1IBr2 based all-Inorganic perovskite solar cells with exceptional efficiency and stability. J. Am. Chem. Soc. 2017, 139, 14009–14012. [Google Scholar] [CrossRef]
- Kim, S.; Chung, T.; Bae, S.; Lee, S.-W.; Lee, K.D.; Kim, H.; Lee, S.; Kang, Y.; Lee, H.S.; Kim, D. Improved performance and thermal stability of perovskite solar cells prepared via a modified sequential deposition process. Org. Electron. 2017, 41, 266–273. [Google Scholar] [CrossRef]
- Zhu, W.D.; Zhang, Q.N.; Chen, D.Z.; Zhang, Z.Y.; Lin, Z.H.; Chang, J.J.; Zhang, J.C.; Zhang, C.F.; Hao, Y. Intermolecular exchange boosts efficiency of air-stable, carbon-based all-Inorganic planar CsPbIBr2 perovskite solar cells to Over 9%. Adv. Energy Mater. 2018, 8, 1802080. [Google Scholar] [CrossRef]
- Lu, J.J.; Chen, S.C.; Zheng, Q.D. Defect Passivation of CsPbIBr2 perovskites for high-performance solar cells with large open-circuit voltage of 1.28 V. ACS Appl. Energy Mater. 2018, 1, 5872–5878. [Google Scholar] [CrossRef]
- Hwang, T.; Lee, S.; Kim, J.; Kim, J.; Kim, C.; Shin, B.; Park, B. Tailoring the mesoscopic TiO2 layer: Concomitant parameters for enabling high-performance perovskite solar cells. Nanoscale Res. Lett. 2017, 12, 57. [Google Scholar] [CrossRef]
- Divitini, G.; Cacovich, S.; Matteocci, F.; Cinà, L.; Carlo, A.D.; Ducati, C. In situ observation of heat-induced degradation of perovskite solar cells. Nat. Energy 2016, 1, 15012. [Google Scholar] [CrossRef]
- Yang, B.; Wang, M.; Hu, X.F.; Zhou, T.W.; Zang, Z.G. Highly efficient semitransparent CsPbIBr2 perovskite solar cells via low temperature processed In2S3 as electron-transport-layer. Nano Energy 2019, 57, 718–727. [Google Scholar] [CrossRef]
- Guo, Y.X.; Yin, X.T.; Liu, J.; Que, W.X. Highly efficient CsPbIBr2 perovskite solar cells with efficiency over 9.8% fabricated using a preheating-assisted spin-coating method. J. Mater. Chem. A 2019, 7, 19008–19016. [Google Scholar] [CrossRef]
- Ahn, N.; Jeon, I.; Yoon, J.; Kauppinen, E.; Matsuo, Y.; Maruyama, S.; Choi, M. Carbon-sandwiched perovskite solar cell. J. Mater. Chem. A 2018, 6, 1382–1389. [Google Scholar] [CrossRef]
- Bai, Y.; Chen, H.; Xiao, S.; Xue, Q.; Zhang, T.; Zhu, Z.; Li, Q.; Hu, C.; Yang, Y.; Hu, Z.C.; et al. Effects of a molecular monolayer modification of NiO nanocrystal layer surfaces on perovskite crystallization and interface contact toward faster hole extraction and higher photovoltaic performance. Adv. Funct. Mater. 2016, 26, 2950–2958. [Google Scholar] [CrossRef]
- Peng, Q.M.; Guo, J.X.; Zhang, Q.R.; Xiang, J.Y.; Liu, B.Z.; Zhou, A.G.; Liu, R.P.; Tian, Y.J. Unique lead adsorption behavior of activated hydroxyl group in two-dimensional titanium carbide. J. Am. Chem. Soc. 2014, 136, 4113–4116. [Google Scholar] [CrossRef]
- Shi, B.; Yao, X.; Hou, F.H.; Guo, S.; Li, Y.C.; Wei, C.C.; Ding, Y.; Li, Y.L.; Zhao, Y.; Zhang, X.D. Unraveling the passivation process of PbI2 to enhance the efficiency of planar perovskite solar cells. J. Phys. Chem. C. 2018, 122, 21269–21276. [Google Scholar] [CrossRef]
- Yu, H.; Lu, H.P.; Xie, F.Y.; Zhou, S.; Zhao, N. Native defect-induced hysteresis behavior in organolead iodide perovskite solar cells. Adv. Funct. Mater. 2016, 26, 1411–1419. [Google Scholar] [CrossRef]
- Ahn, N.; Kwak, K.; Jang, M.S.; Yoon, H.; Lee, B.Y.; Lee, J.K.; Pikhitsa, P.V.; Byun, J.; Choi, M. Trapped charge-driven degradation of perovskite solar cells. Nat. Commun. 2016, 7, 13422. [Google Scholar] [CrossRef]
- Lin, H.S.; Jeno, I.I.; Chen, Y.Q.; Yang, X.Y.; Nakagawa, T.; Maruyama, S.; Manzhos, S.; Matsuo, Y. Highly selective and scalable fullerene-cation-mediated synthesis accessing cyclo [60] fullerenes with five-membered carbon ring and their application to perovskite solar cells. Chem. Mater. 2019, 31, 8432–8439. [Google Scholar] [CrossRef]

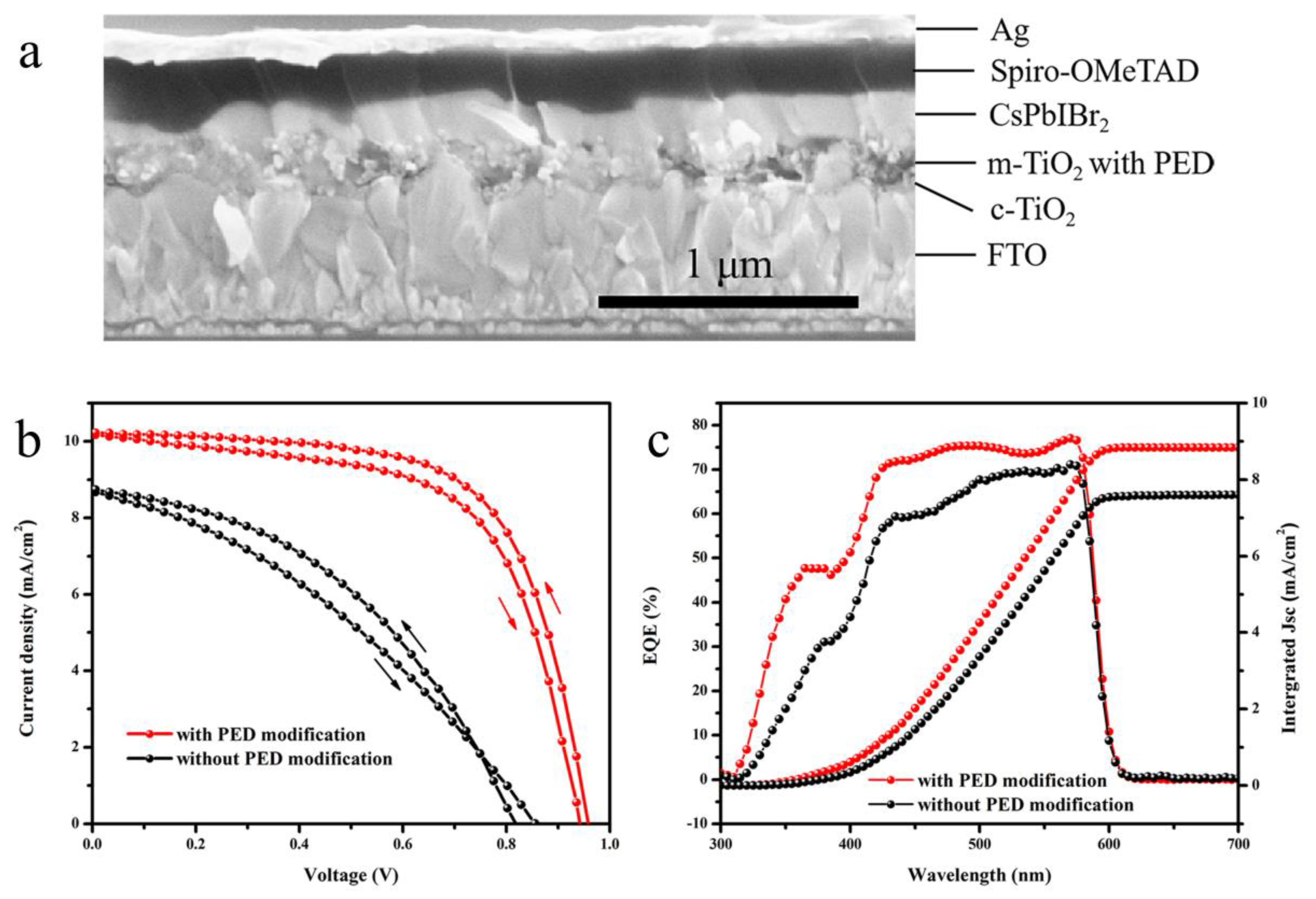
| Substrate | Scan | Jsc (mA∙cm−2) | Voc (V) | FF | PCE (%) |
|---|---|---|---|---|---|
| Without PED | forward | 8.69 | 0.86 | 0.35 | 2.62 |
| reverse | 8.74 | 0.82 | 0.43 | 3.04 | |
| With PED | forward | 10.28 | 0.94 | 0.62 | 5.93 |
| reverse | 10.22 | 0.96 | 0.65 | 6.39 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meng, X.; Chi, K.; Li, Q.; Cao, Y.; Song, G.; Liu, B.; Yang, H.; Fu, W. Interfacial Modification of Mesoporous TiO2 Films with PbI2-Ethanolamine-Dimethyl Sulfoxide Solution for CsPbIBr2 Perovskite Solar Cells. Nanomaterials 2020, 10, 962. https://doi.org/10.3390/nano10050962
Meng X, Chi K, Li Q, Cao Y, Song G, Liu B, Yang H, Fu W. Interfacial Modification of Mesoporous TiO2 Films with PbI2-Ethanolamine-Dimethyl Sulfoxide Solution for CsPbIBr2 Perovskite Solar Cells. Nanomaterials. 2020; 10(5):962. https://doi.org/10.3390/nano10050962
Chicago/Turabian StyleMeng, Xianwei, Kailin Chi, Qian Li, Yu Cao, Gengxin Song, Bao Liu, Haibin Yang, and Wuyou Fu. 2020. "Interfacial Modification of Mesoporous TiO2 Films with PbI2-Ethanolamine-Dimethyl Sulfoxide Solution for CsPbIBr2 Perovskite Solar Cells" Nanomaterials 10, no. 5: 962. https://doi.org/10.3390/nano10050962
APA StyleMeng, X., Chi, K., Li, Q., Cao, Y., Song, G., Liu, B., Yang, H., & Fu, W. (2020). Interfacial Modification of Mesoporous TiO2 Films with PbI2-Ethanolamine-Dimethyl Sulfoxide Solution for CsPbIBr2 Perovskite Solar Cells. Nanomaterials, 10(5), 962. https://doi.org/10.3390/nano10050962