Nanostructured Polyelectrolyte Complexes Based on Water-Soluble Thiacalix[4]Arene and Pillar[5]Arene: Self-Assembly in Micelleplexes and Polyplexes at Packaging DNA
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. Scanning Electron Microscopy (SEM)
2.3. Dynamic Light Scattering (DLS)
2.3.1. Particles’ Size
2.3.2. Zeta Potentials
2.4. UV-Visible Spectroscopy
2.5. Fluorescence Spectroscopy
2.6. Circular Dichroism (CD) Studies
3. Results
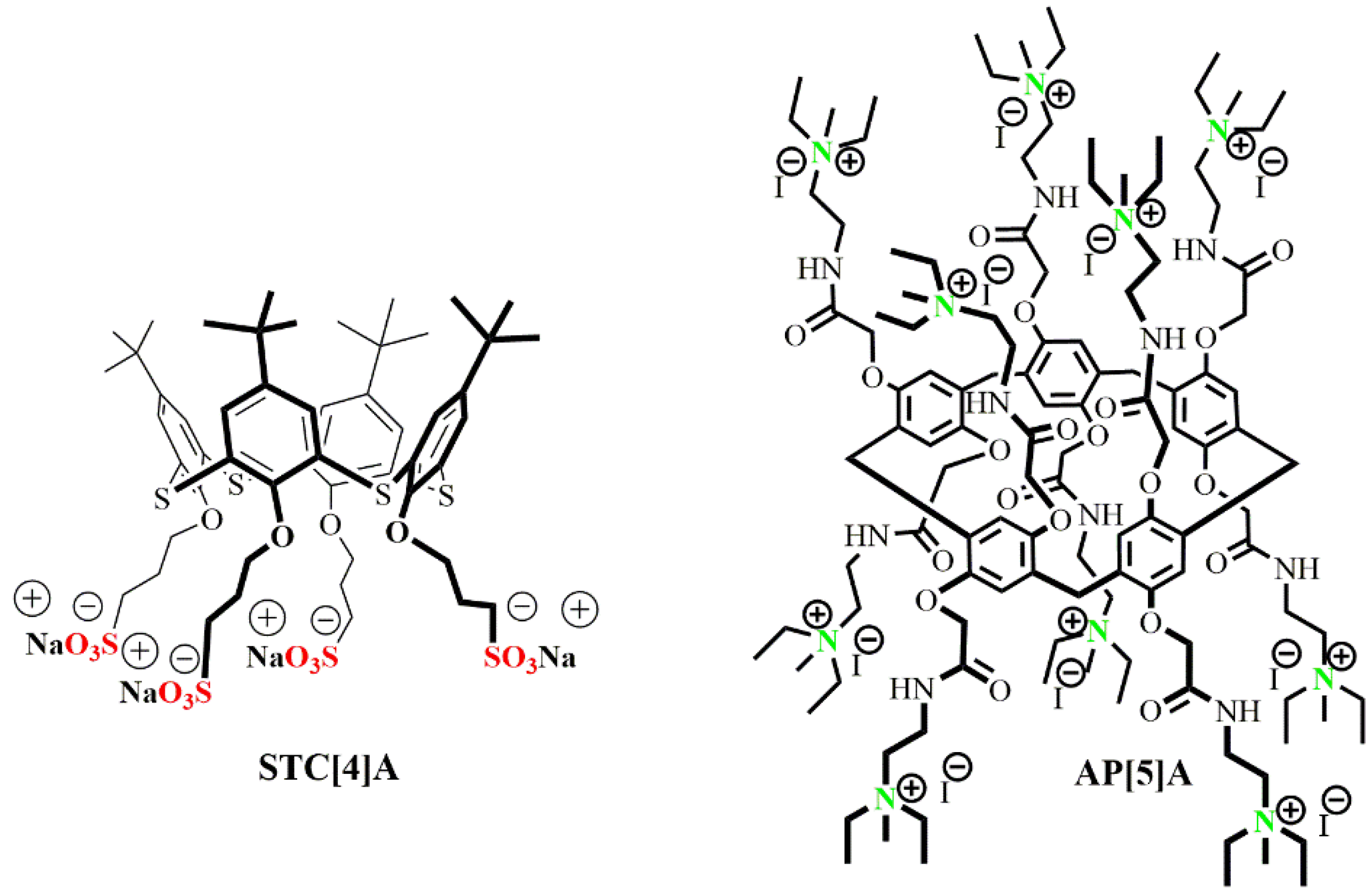
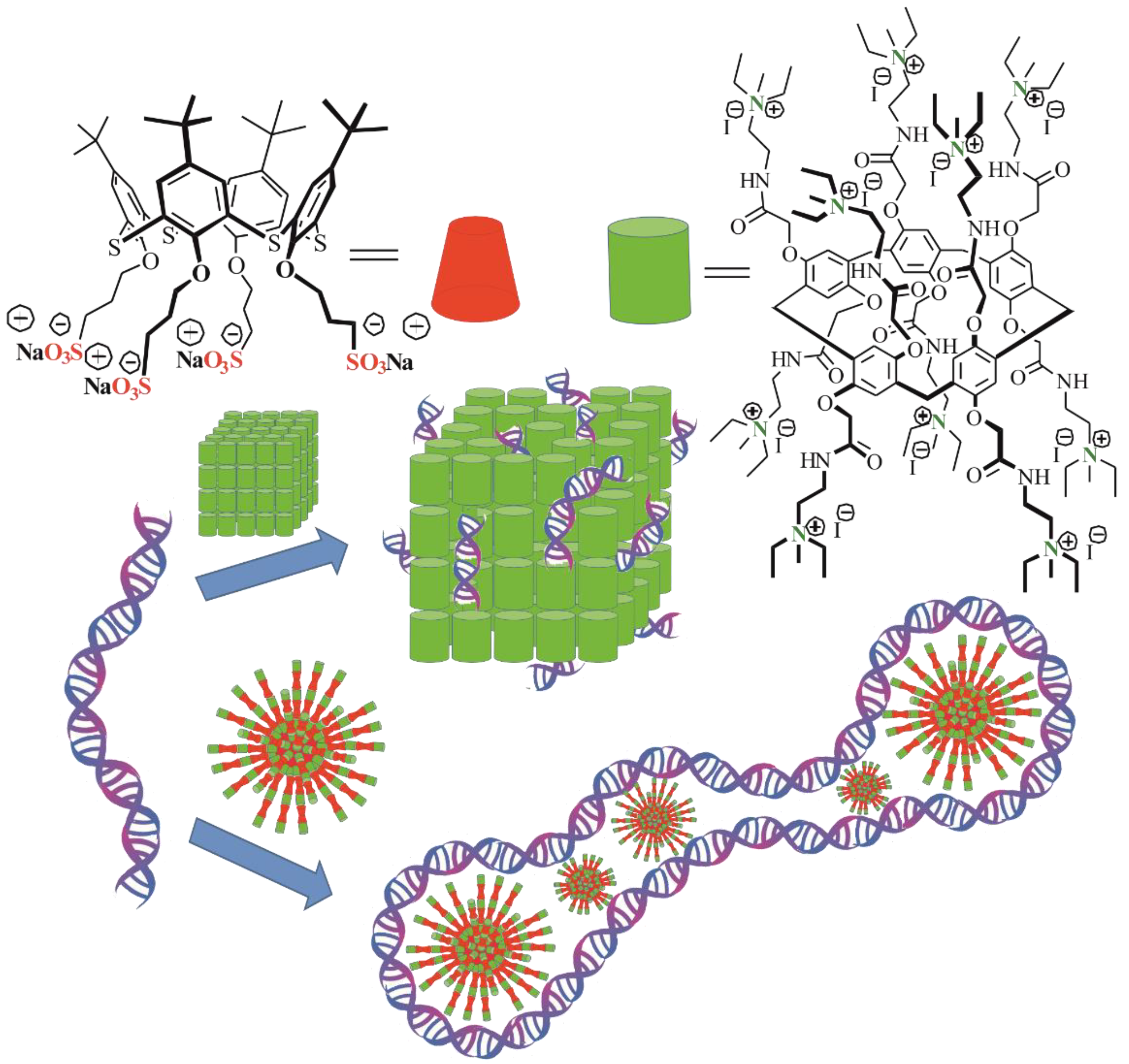
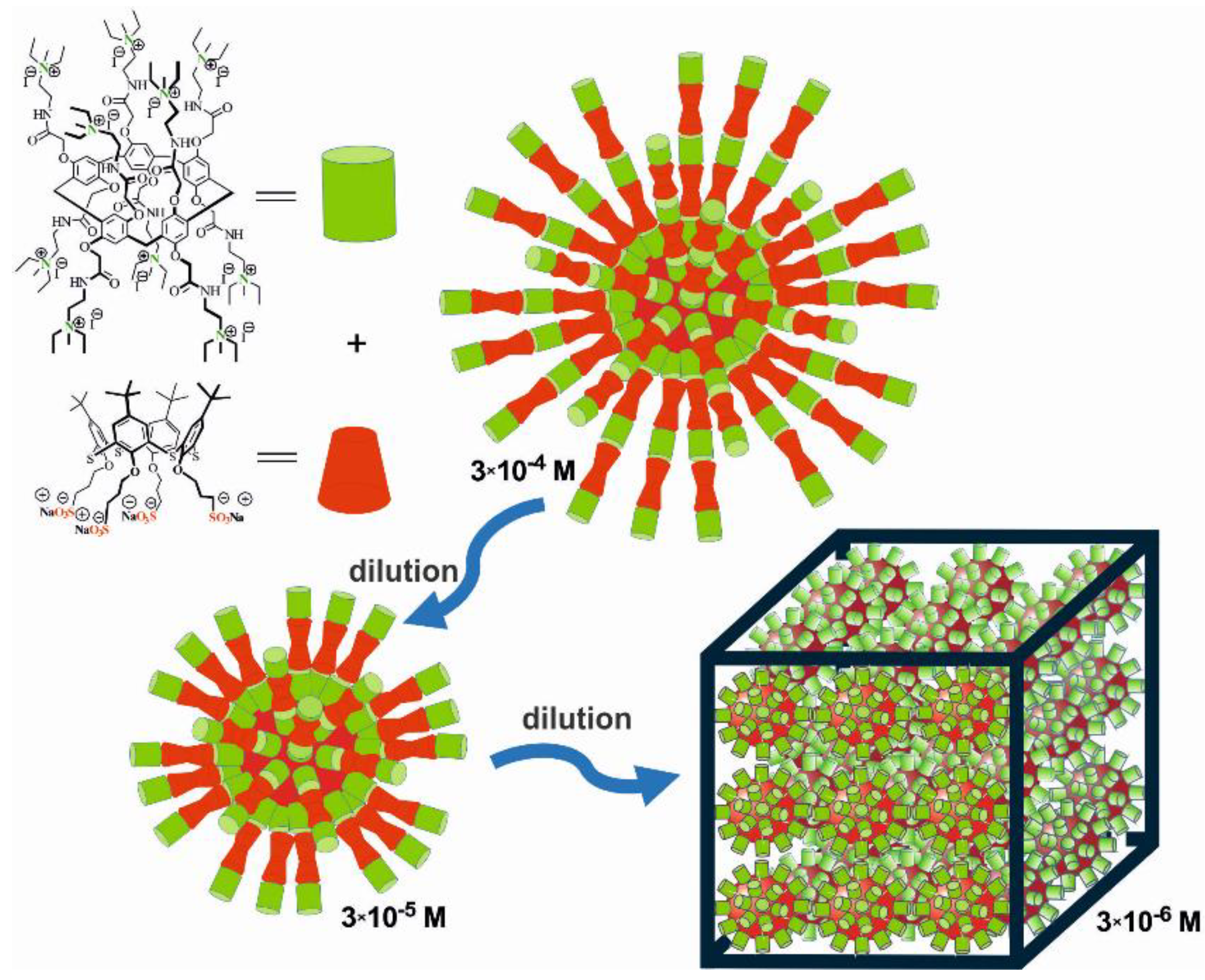
3.1. Construction of the AP[5]A/STC[4]A Co-Assemblies
3.2. Co-Assembly with DNA
3.2.1. UV Spectroscopy
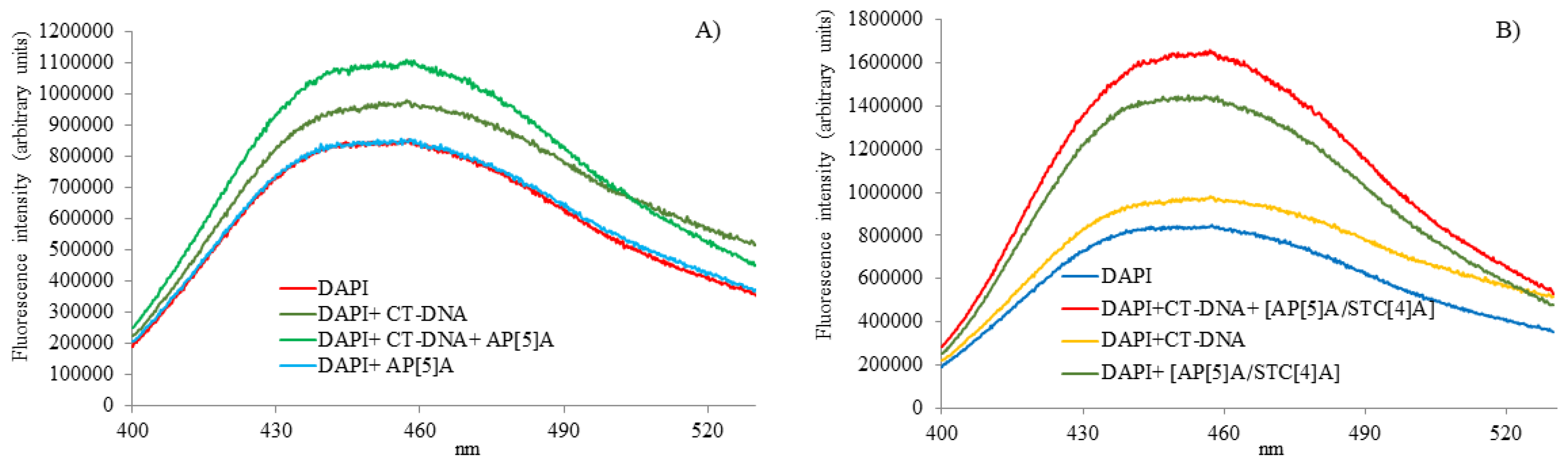
3.2.2. Fluorescence Spectroscopy
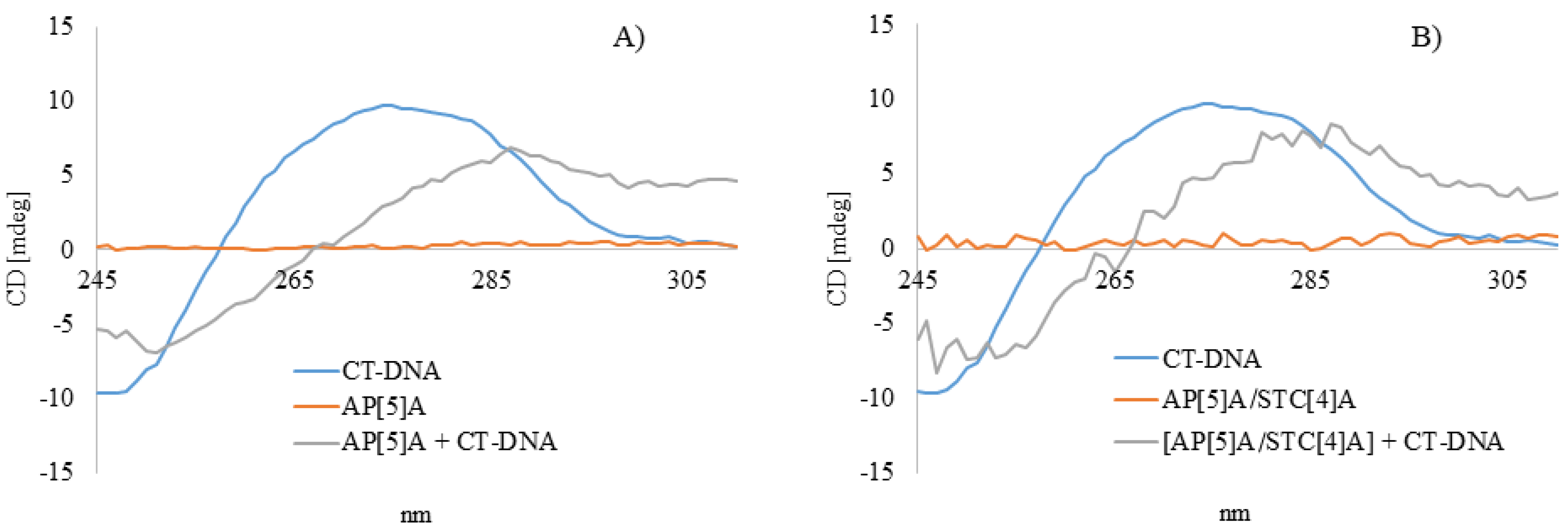
3.2.3. Circular Dichroism Spectroscopy
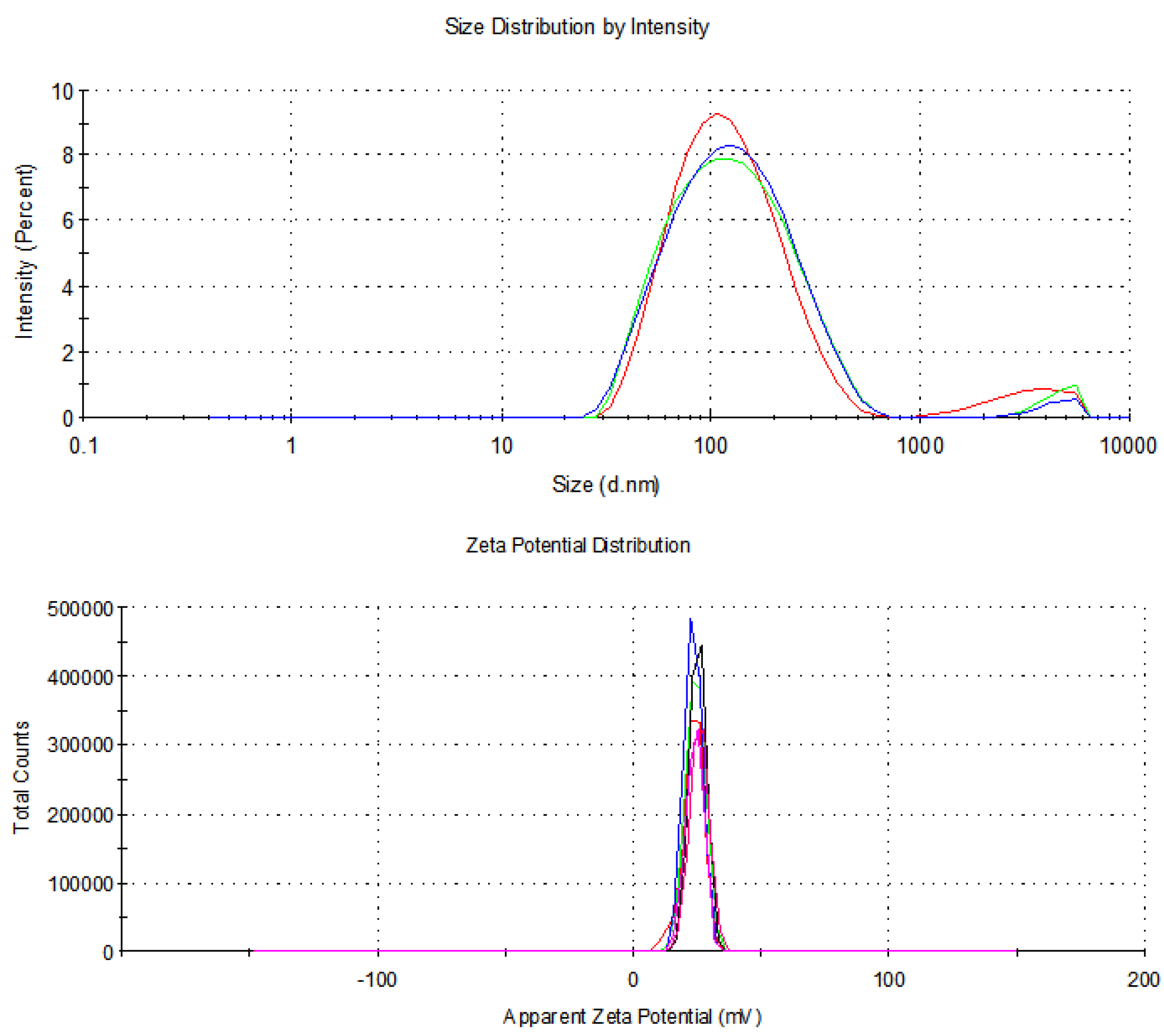
3.2.4. Dynamic Light Scattering
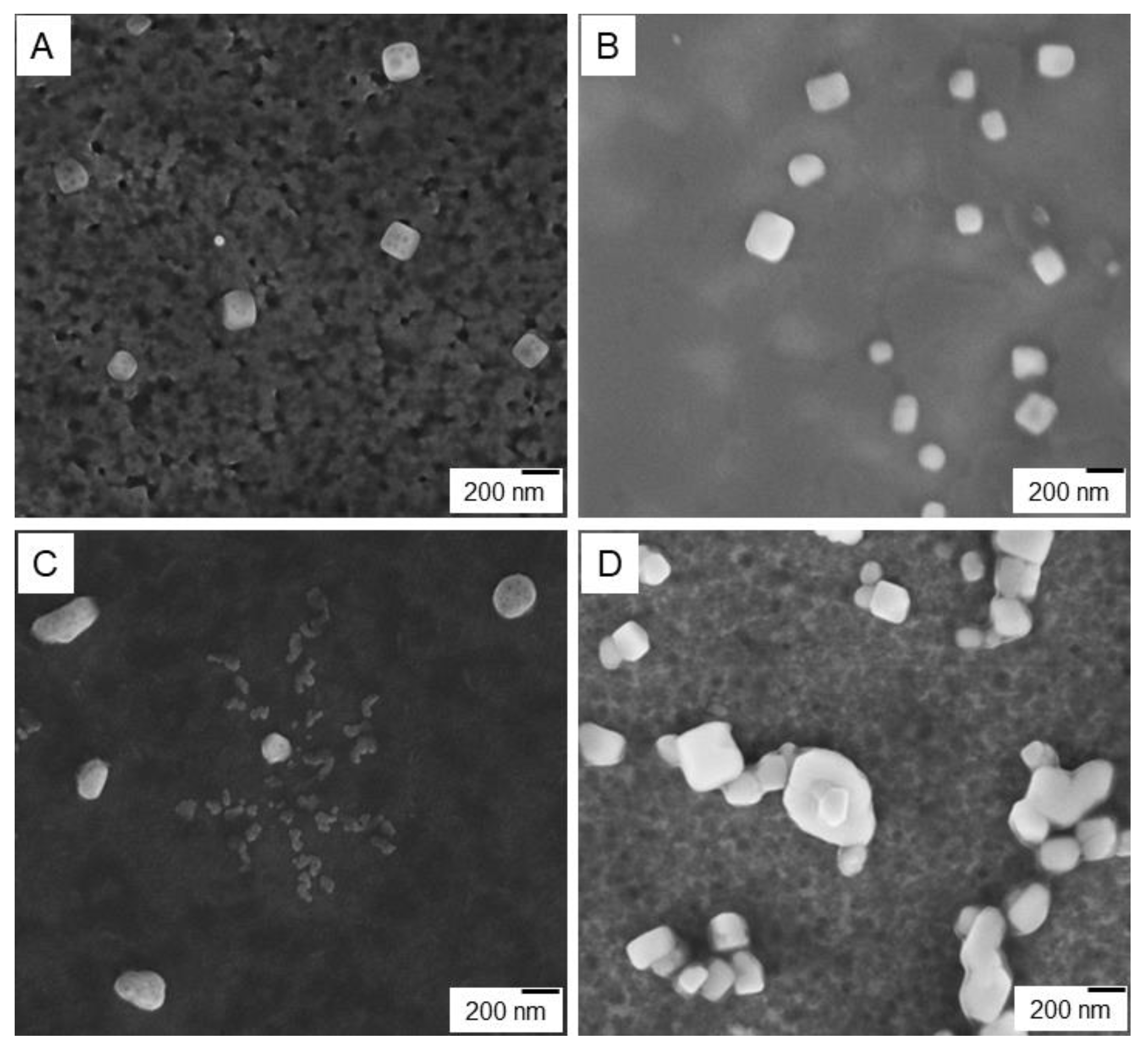
3.2.5. Scanning Electron Microscopy
3.3. Determination of the Proposed Structure of Co-Interpolyelectrolyte Aggregates AP[5]A/STC[4]A
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Li, L.-L.; An, H.-W.; Peng, B.; Zheng, R.; Wang, H. Self-assembled nanomaterials: Design principles, the nanostructural effect, and their functional mechanisms as antimicrobial or detection agents. Mater. Hor. 2019, 6, 1794–1811. [Google Scholar] [CrossRef]
- Ariga, K.; Nishikawa, M.; Mori, T.; Takeya, J.; Shrestha, L.K.; Hill, J.P. Self-assembly as a key player for materials nanoarchitectonics. Sci. Technol. Adv. Mat. 2019, 20, 51–95. [Google Scholar] [CrossRef]
- Sevim, S.; Sorrenti, A.; Franco, C.; Furukawa, S.; Pané, S.; deMello, A.J.; Puigmartí-Luis, J. Self-assembled materials and supramolecular chemistry within microfluidic environments: From common thermodynamic states to non-equilibrium structures. Chem. Soc. Rev. 2018, 47, 3788–3803. [Google Scholar] [CrossRef]
- Amit, M.; Yuran, S.; Gazit, E.; Reches, M.; Ashkenasy, N. Tailor-Made Functional Peptide Self-Assembling Nanostructures. Adv. Mater. 2018, 30, 1707083. [Google Scholar] [CrossRef]
- Pergushov, D.V.; Müller, A.H.E.; Schacher, F.H. Micellar Interpolyelectrolyte Complexes. Chem. Soc. Rev. 2012, 41, 6888–6901. [Google Scholar] [CrossRef] [PubMed]
- van der Gucht, J.; Spruijt, E.; Lemmers, M.; Cohen Stuart, M.A. Polyelectrolyte Complexes: Bulk Phases and Colloidal Systems. J. Colloid Interface Sci. 2011, 361, 407–422. [Google Scholar] [CrossRef]
- Zhao, Q.; An, Q.F.; Ji, Y.; Qian, J.; Gao, C. Polyelectrolyte Complex Membranes for Pervaporation, Nanofiltration and Fuel Cell Applications. J. Membr. Sci. 2011, 379, 19–45. [Google Scholar] [CrossRef]
- Ahmadiannamini, P.; Li, X.; Goyens, W.; Joseph, N.; Meesschaert, B.; Vankelecom, I.F.J. Multilayered Polyelectrolyte Complex Based Solvent Resistant Nanofiltration Membranes Prepared from Weak Polyacids. J. Membr. Sci. 2012, 394−395, 98–106. [Google Scholar] [CrossRef]
- Delcea, M.; Möhwald, H.; Skirtach, A.G. Stimuli-Responsive LbL Capsules and Nanoshells for Drug Delivery. Adv. Drug Deliv. Rev. 2011, 63, 730–747. [Google Scholar] [CrossRef]
- Kim, S.H.; Jeong, J.H.; Lee, S.H.; Kim, S.W.; Park, T.G. Local and Systemic Delivery of VEGF siRNA Using Polyelectrolyte Complex Micelles for Effective Treatment of Cancer. J. Control. Release 2008, 129, 107–116. [Google Scholar] [CrossRef]
- Wang, Q.; Schlenoff, J.B. The Polyelectrolyte Complex/Coacervate Continuum. Macromolecules 2014, 47, 3108–3116. [Google Scholar] [CrossRef]
- Lounis, F.M.; Chamieh, J.; Gonzalez, P.; Cottet, H.; Leclercq, L. Prediction of Polyelectrolyte Complex Stoichiometry for Highly Hydrophilic Polyelectrolytes. Macromolecules 2016, 49, 3881–3888. [Google Scholar] [CrossRef]
- Fu, J.; Fares, H.M.; Schlenoff, J.B. Ion-Pairing Strength in Polyelectrolyte Complexes. Macromolecules 2017, 50, 1066–1074. [Google Scholar] [CrossRef]
- Love, J.C.; Estroff, L.A.; Kriebel, J.K.; Nuzzo, R.G.; Whitesides, G.M. Self-assembled monolayers of thiolates on metals as a form of nanotechnology. Chem. Rev. 2005, 105, 1103–1169. [Google Scholar] [CrossRef]
- Daniel, M.-C.; Astruc, D. Gold Nanoparticles: Assembly, Supramolecular Chemistry, Quantum-Size-Related Properties, and Applications toward Biology, Catalysis, and Nanotechnology. Chem. Rev. 2004, 104, 293–346. [Google Scholar] [CrossRef]
- Chen, M.; Diao, G.W.; Li, C.H.; Zhou, X.M. Phase transition of silver nanoparticles from aqueous solution to chloroform with the help of inclusion complexes of p-sulfonated calix[4]arene and alkanethiol molecules and its application in the size sorting of nanoparticles. Nanotechnology 2007, 18, 175706. [Google Scholar] [CrossRef]
- Chen, M.; Ding, W.H.; Kong, Y.; Diao, G.W. Conversion of the Surface Property of Oleic Acid Stabilized Silver Nanoparticles from Hydrophobic to Hydrophilic Based on Host−Guest Binding Interaction. Lanmuir 2008, 24, 3471–3478. [Google Scholar] [CrossRef]
- Baldini, L.; Sansone, F.; Massera, C.; Casnati, A.; Ugozzoli, F.; Ungaro, R. Designing nanoporous crystalline materials by self-assembly: 2D hydrogen-bonded networks from upper rim calix[4]arene diamide derivatives. Inorg. Chim. Acta 2007, 360, 970–980. [Google Scholar] [CrossRef]
- Seganish, J.L.; Santacroce, P.V.; Salimian, K.J.; Fettinger, J.C.; Zavalij, P.; Davis, J.T. Regulating supramolecular function in membranes: Calixarenes that enable or inhibit transmembrane Cl- transport. Angew. Chem. Int. Ed. 2006, 45, 3334–3338. [Google Scholar] [CrossRef]
- Schafer, C.; Eckel, R.; Ros, R.; Mattay, J.; Anselmetti, D. Photochemical Single-Molecule Affinity Switch. J. Am. Chem. Soc. 2007, 129, 1488–1489. [Google Scholar] [CrossRef]
- Organo, V.G.; Leontiev, A.V.; Sgarlata, V.; Dias, H.V.R.; Rudkevich, D.M. Supramolecular Features of Calixarene-Based Synthetic Nanotubes. Angew. Chem. Int. Ed. 2005, 44, 3043–3047. [Google Scholar] [CrossRef]
- Atwood, J.L.; Steed, J.W. Encyclopedia of Supramolecular Chemistry; CRC Press: Boca Raton, FL, USA, 2004; pp. 1–1744. [Google Scholar]
- Gorbatchuk, V.V.; Savelyeva, L.S.; Ziganshin, M.A.; Antipin, I.S.; Sidorov, V.A. Molecular recognition of organic guest vapor by solid adamantylcalix[4]arene. Russ. Chem. Bull. 2004, 53, 60–64. [Google Scholar] [CrossRef]
- Ziganshin, M.A.; Yakimova, L.S.; Khayarov, K.R.; Gorbatchuk, V.V.; Vysotsky, M.O.; Böhmer, V. Guest exchange in dimeric capsules of a tetraurea calix[4]arene in the solid state. Chem. Commun. 2006, 37, 3897–3899. [Google Scholar] [CrossRef]
- Yakimova, L.S.; Ziganshin, M.A.; Sidorov, V.A.; Kovalev, V.V.; Shokova, E.A.; Tafeenko, V.A.; Gorbatchuk, V.V. Molecular Recognition of Organic Vapors by Adamantylcalix[4]arene in QCM Sensor Using Partial Binding Reversibility. J. Phys. Chem. B 2008, 112, 15569–15575. [Google Scholar] [CrossRef]
- Stoikov, I.I.; Mostovaya, O.A.; Yakimova, L.S.; Yantemirova, A.A.; Antipin, I.S.; Konovalov, A.I. Phosphorus-bridged calixarene phosphites: Dramatic influence of a tert-butyl group at the upper rim of the macrocycle upon anion binding. Mendeleev Commun. 2010, 20, 359–360. [Google Scholar] [CrossRef]
- Ogoshi, T.; Hasegawa, Y.; Aoki, T.; Ishimori, Y.; Inagi, S.; Yamagishi, T.-A. Reduction of Emeraldine Base Form of Polyaniline by Pillar[5]arene Based on Formation of Poly(pseudorotaxane) Structure. Macromolecules 2011, 44, 7639–7644. [Google Scholar] [CrossRef]
- Ogoshi, T.; Kitajima, K.; Aoki, T.; Yamagishi, T.A.; Nakamoto, Y. Effect of an Intramolecular Hydrogen Bond Belt and Complexation with the Guest on the Rotation Behavior of Phenolic Units in Pillar[5]arenes. J. Phys. Chem. Lett. 2010, 1, 817–821. [Google Scholar] [CrossRef]
- Li, C.; Xu, Q.; Li, J.; Yao, F.; Jia, X. Complex interactions of pillar[5]arene with paraquats and bis(pyridinium) derivatives. Org. Biomol. Chem. 2010, 8, 1568–1576. [Google Scholar] [CrossRef]
- Shurpik, D.N.; Yakimova, L.S.; Rizvanov, I.K.; Plemenkov, V.V.; Stoikov, I.I. Water-soluble pillar[5]arenes: Synthesis and characterization of the inclusion complexes with p-toluenesulfonic acid. Macroheterocycles 2015, 8, 128–134. [Google Scholar] [CrossRef]
- Xu, Z.; Jia, S.; Wang, W.; Yuan, Z.; Ravoo, B.J.; Guo, D.-S. Heteromultivalent peptide recognition by co-assembly of cyclodextrin and calixarene amphiphiles enables inhibition of amyloid fibrillation. Nat. Chem. 2019, 86, 86–93. [Google Scholar] [CrossRef]
- Chen, X.-M.; Chen, Y.; Yu, Q.; Gu, B.-H.; Liu, Y. Supramolecular assemblies with near-infrared emission mediated in two stages by cucurbituril and amphiphilic calixarene for lysosome- targeted cell imaging. Angew. Chem. Int. Ed. 2018, 57, 12519–12523. [Google Scholar] [CrossRef]
- Yakimova, L.; Padnya, P.; Tereshina, D.; Kunafina, A.; Nugmanova, A.; Osin, Y.; Evtugyn, V.; Stoikov, I. Interpolyelectrolyte mixed nanoparticles from anionic and cationic thiacalix[4]arenes for selective recognition of model biopolymers. J. Mol. Liq. 2019, 279, 9–17. [Google Scholar] [CrossRef]
- Yakimova, L.S.; Gilmanova, L.H.; Evtugyn, V.G.; Osin, Y.N.; Stoikov, I.I. Self-assembled fractal hybrid dendrites from water-soluble anionic (Thia)calix[4] arenes and Ag+. J. Nanopart. Res. 2017, 19, 173–183. [Google Scholar] [CrossRef]
- Khan, G.S.; Shah, A.; Zia-ur-Rehman; Barker, D. Chemistry of DNA minor groove binding agents. J. Phtoch. Photobio. B 2012, 115, 105–118. [Google Scholar] [CrossRef]
- Kapuscinski, J.; Skoczylas, B. Fluorescent complexes of DNA with DAPI 4′-6-diamidine-2-phenyl indole 2HCl or DC14′-6-dicarboxyamide-2-phenyl indole. Nucleic Acids Res. 1978, 5, 3775–3799. [Google Scholar] [CrossRef][Green Version]
- Biancardi, A.; Biver, T.; Secco, F.; Mennucci, B. An investigation of the photophysical properties of minor groove bound and intercalated DAPI through quantum-mechanical and spectroscopic tools. Phys. Chem. Chem. Phys. 2013, 15, 4596–4603. [Google Scholar] [CrossRef]
- Kasyanenko, N.A.; Tikhomirov, R.A.; Bakulev, V.M.; Demidov, V.N.; Chikhirzhina, E.V.; Moroshkina, E.B. DNA Complexes with Cobalt(II) Phthalocyanine Disodium Disulfonate. ACS Omega 2019, 4, 16935–16942. [Google Scholar] [CrossRef]
- Zadmard, R.; Schrader, T. DNA recognition with large calixarene dimers. Angew. Chem. Int. Ed. 2006, 45, 2703–2706. [Google Scholar] [CrossRef]
- Pabbathi, A.; Samanta, A. Spectroscopic and Molecular Docking Study of the Interaction of DNA with a Morpholinium Ionic Liquid. J. Phys. Chem. B 2015, 119, 11099–11105. [Google Scholar] [CrossRef]
- Banerjee, D.; Pal, S.K. Dynamics in the DNA Recognition by DAPI: Exploration of the Various Binding Modes. J. Phys. Chem. B 2008, 112, 1016–1021. [Google Scholar] [CrossRef]
- Ankerfors, C.; Bertin, A.; Bouhallab, S.; Croguennec, T.; Müller, M.; Petzold, G.; Schwarz, S.; Wagberg, L. Polyelectrolyte Complexes in the Dispersed and Solid State II; Springer: Berlin, Germany, 2014; pp. 1–256. [Google Scholar] [CrossRef]
- Bhattacharjee, S. DLS and zeta potential What they are and what they are not? J. Control. Release 2016, 235, 337–351. [Google Scholar] [CrossRef]
- Lohani, N.; Rajeswari, M.R. Preferential binding of anticancer drugs to triplex DNA compared to duplex DNA: A spectroscopic and calorimetric study. RSC Adv. 2016, 6, 39903–39917. [Google Scholar] [CrossRef]
- Shahabadi, N.; Mohammadi, S. Synthesis characterization and DNA interaction studies of a new Zn (II) complex containing different dinitrogen aromatic ligands. Bioinorg. Chem. Appl. 2012, 571913–571921. [Google Scholar] [CrossRef]
- Gomathi, R.; Ramu, A.; Murugan, A. Evaluation of DNA Binding, Cleavage, and Cytotoxic Activity of Cu (II), Co (II), and Ni (II) Schiff Base Complexes of 1-Phenylindoline-2, 3-dione with Isonicotinohydrazide. Bioinorg. Chem. Appl. 2014, 215392. [Google Scholar] [CrossRef]
- Norden, B.; Kurucsev, T. Analysing DNA complexes by circular and linear dichroism. J. Mol. Recognit. 1994, 7, 141–156. [Google Scholar] [CrossRef]
- Kaushik, M.; Kaushik, S.; Roy, K.; Singh, A.; Mahendru, S.; Kumar, M.; Chaudhary, S.; Ahmed, S.; Kukreti, S. A bouquet of DNA structures: Emerging diversity. Biochem. Biophys. Rep. 2016, 5, 388–395. [Google Scholar] [CrossRef][Green Version]
- Banavali, N.K.; Roux, B. Free Energy Landscape of A-DNA to B-DNA Conversion in Aqueous Solution. J. Am. Chem. Soc. 2005, 127, 6866–6876. [Google Scholar] [CrossRef]
- Gubendran, A.; Palsamy Kesavan, M.; Ayyanaar, S.; Mitu, L.; Athappan, P.; Rajesh, J. Non-enolisable Knoevenagel condensate appended Schiff bases-metal (II) complexes: Spectral characteristics, DNA-binding and nuclease activities. Spectrochim. Acta A 2017, 181, 39–46. [Google Scholar] [CrossRef]
- Luo, J.; Xie, Z.; Lam, J.W.Y.; Cheng, L.; Chen, H.; Qiu, C.; Kwok, H.S.; Zhan, X.; Liu, Y.; Zhu, D.; et al. Aggregation-induced emission of 1-methyl-1,2,3,4,5-pentaphenylsilole. Chem. Commun. 2001, 18, 1740–1741. [Google Scholar] [CrossRef]
- Yakimova, L.S.; Puplampu, J.B.; Evtugin, G.A.; Stoikov, I.I. Polyfunctional branched nitrogen-containing p-tert-butylthiacalix[4]arene derivatives as efficient agents for packaging calf thymus DNA. Russ. Chem. Bull. 2017, 66, 1515–1523. [Google Scholar] [CrossRef]
- Kabanov, A.V.; Astafieva, I.V.; Chikindas, M.L.; Rosenblat, G.F.; Kiselev, V.I.; Severin, E.S.; Kabanov, V.A. DNA interpolyelectrolyte complexes as a tool for efficient cell transformation. Biopolymers 1991, 31, 1437–1443. [Google Scholar] [CrossRef] [PubMed]
- Kabanov, A.V.; Kabanov, V.A. DNA Complexes with Polycations for the Delivery of Genetic Material into Cells. Bioconjugate Chem. 1995, 6, 7–20. [Google Scholar] [CrossRef] [PubMed]
- Tan, Z.; Jiang, Y.; Zhang, W.; Karls, L.; Lodge, T.P.; Reineke, T.M. Polycation Architecture and Assembly Direct Successful Gene Delivery: Micelleplexes Outperform Polyplexes via Optimal DNA Packaging. J. Am. Chem. Soc. 2019, 141, 15804–15817. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Reineke, T.M.; Lodge, T.P. Complexation of DNA with Cationic Copolymer Micelles: Effects of DNA Length and Topology. Macromolecules 2018, 51, 1150–1160. [Google Scholar] [CrossRef]
- Jiang, Y.; Lodge, T.P.; Reineke, T.M. Packaging pDNA by Polymeric ABC Micelles Simultaneously Achieves Colloidal Stability and Structural Control. J. Am. Chem. Soc. 2018, 140, 11101–11111. [Google Scholar] [CrossRef]
- Stein, H.N. The Preparation of Dispersions in Liquids; CRC Press: Boca Raton, FL, USA, 1995; pp. 1–264. [Google Scholar]
| Molar Ratio | [AP[5]A/STC[4]A]+CT-DNA | ||
|---|---|---|---|
| PDI | d, nm | ζ, mV | |
| 1:10 | 0.32 ± 0.04 | 199 ± 2 | −44 ± 3 |
| 1:7 | 0.30 ± 0.01 | 192 ± 2 | −41 ± 2 |
| 1:3 | 0.23 ± 0.01 | 186 ± 1 | −36 ± 1 |
| c,* mol/L | STC[4]A [34] | AP[5]A | AP[5]A/STC[4]A | ||||
|---|---|---|---|---|---|---|---|
| PDI | d, nm | PDI | d, nm | PDI | d, nm | ζ, mV | |
| 3 × 10−4 | 0.41 ± 0.07 | 480 ± 73 | 0.40 ± 0.22 | 183 ± 12 | 0.18 ± 0.01 | 129 ± 5 | +37 ± 1 |
| 3 × 10−5 | 0.32 ± 0.02 | 168 ± 8 | 0.45 ± 0.19 | 167 ± 54 | 0.27 ± 0.02 | 124 ± 4 | +16 ± 1 |
| 3 × 10−6 | 0.38 ± 0.04 | 262 ± 25 | 0.35 ± 0.07 | 200 ± 38 | 0.40 ± 0.06 | 154 ± 19 | +10 ± 1 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yakimova, L.S.; Nugmanova, A.R.; Mostovaya, O.A.; Vavilova, A.A.; Shurpik, D.N.; Mukhametzyanov, T.A.; Stoikov, I.I. Nanostructured Polyelectrolyte Complexes Based on Water-Soluble Thiacalix[4]Arene and Pillar[5]Arene: Self-Assembly in Micelleplexes and Polyplexes at Packaging DNA. Nanomaterials 2020, 10, 777. https://doi.org/10.3390/nano10040777
Yakimova LS, Nugmanova AR, Mostovaya OA, Vavilova AA, Shurpik DN, Mukhametzyanov TA, Stoikov II. Nanostructured Polyelectrolyte Complexes Based on Water-Soluble Thiacalix[4]Arene and Pillar[5]Arene: Self-Assembly in Micelleplexes and Polyplexes at Packaging DNA. Nanomaterials. 2020; 10(4):777. https://doi.org/10.3390/nano10040777
Chicago/Turabian StyleYakimova, Luidmila S., Aigul R. Nugmanova, Olga A. Mostovaya, Alena A. Vavilova, Dmitriy N. Shurpik, Timur A. Mukhametzyanov, and Ivan I. Stoikov. 2020. "Nanostructured Polyelectrolyte Complexes Based on Water-Soluble Thiacalix[4]Arene and Pillar[5]Arene: Self-Assembly in Micelleplexes and Polyplexes at Packaging DNA" Nanomaterials 10, no. 4: 777. https://doi.org/10.3390/nano10040777
APA StyleYakimova, L. S., Nugmanova, A. R., Mostovaya, O. A., Vavilova, A. A., Shurpik, D. N., Mukhametzyanov, T. A., & Stoikov, I. I. (2020). Nanostructured Polyelectrolyte Complexes Based on Water-Soluble Thiacalix[4]Arene and Pillar[5]Arene: Self-Assembly in Micelleplexes and Polyplexes at Packaging DNA. Nanomaterials, 10(4), 777. https://doi.org/10.3390/nano10040777









