Dry Generation of CeO2 Nanoparticles and Deposition onto a Co-Culture of A549 and THP-1 Cells in Air-Liquid Interface—Dosimetry Considerations and Comparison to Submerged Exposure
Abstract
1. Introduction
2. Materials and Methods
2.1. CeO2 NPs Information and Physico-Chemical Properties
2.2. Cell Culture and Reagents
2.3. Preparation of CeO2 Dispersions
2.4. Size Characterization in Cell Medium
2.5. Preparations of Co-Cultures of A549 and THP-1 Cells
2.6. Submerged Exposures in Inserts and Plates
2.7. Aerosol Generation and Deposition in ALI
2.8. Scanning Electron Microscopy (SEM) and Energy Dispersive Spectroscopy (EDS)
2.9. Quantification of Deposition/Cellular Dose Using Inductively Coupled Plasma Mass Spectrometry (ICP-MS)
2.10. Analysis of Cytotoxicity and Inflammatory Potential in Co-Cultures
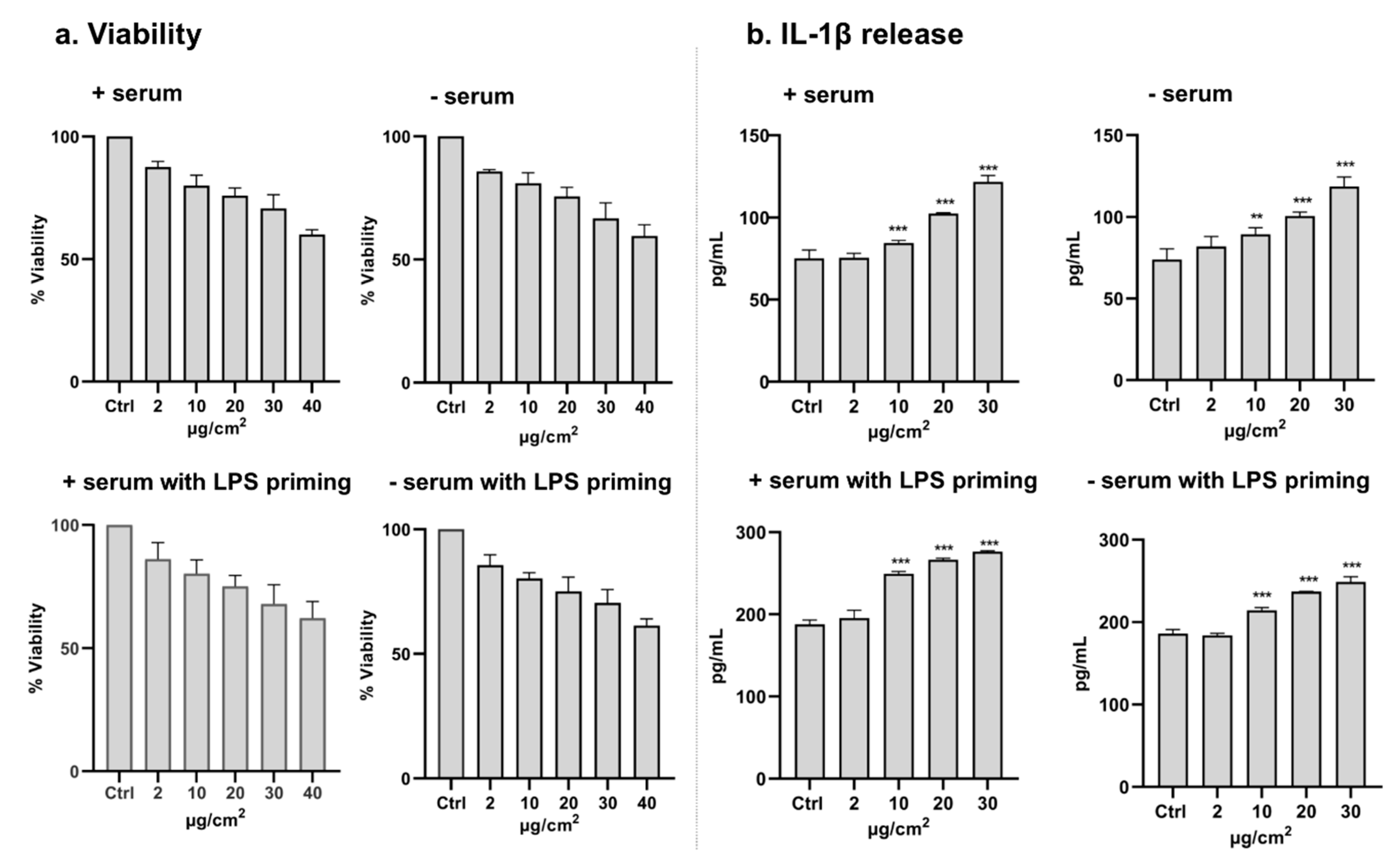
2.11. Analysis of Cytotoxicity and IL-1β Release in THP-1 Cells (in Plates)
2.12. Statistical Analysis
3. Results
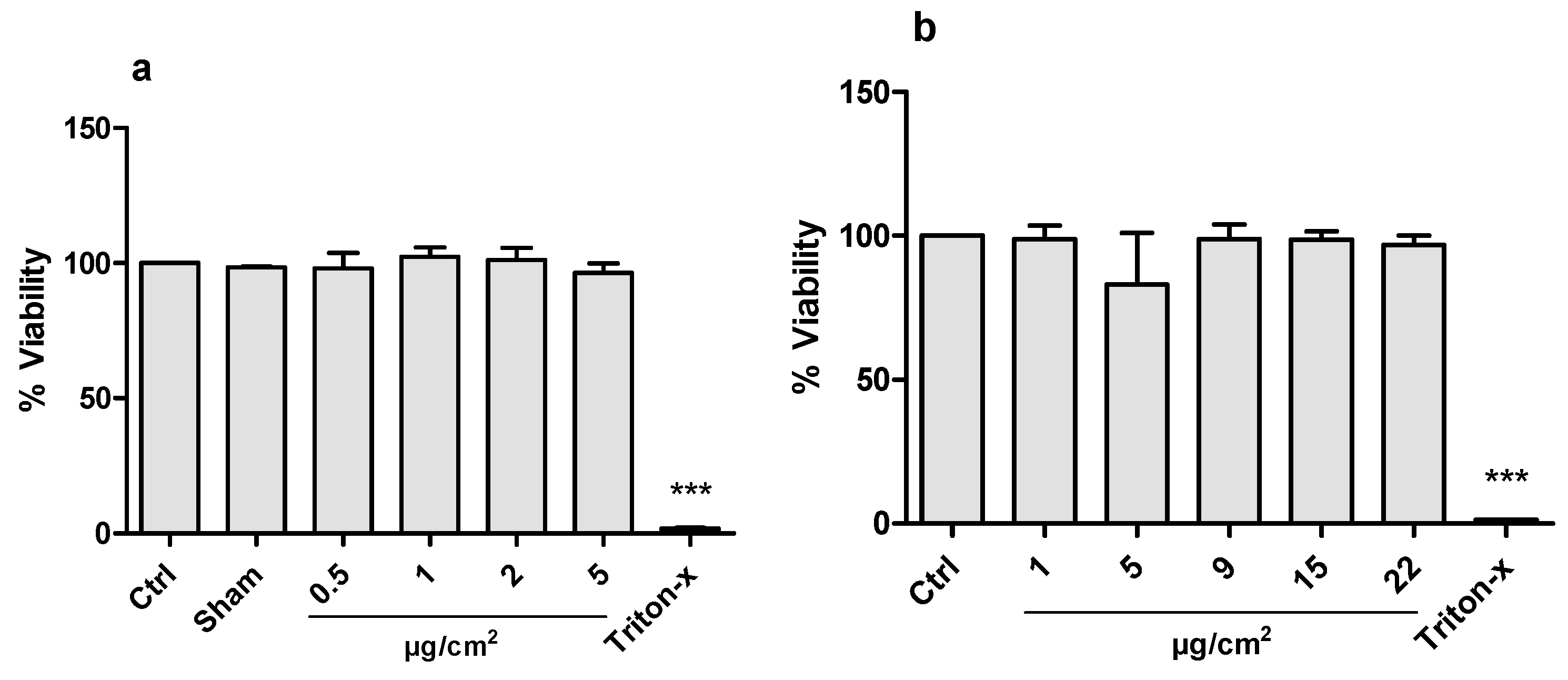
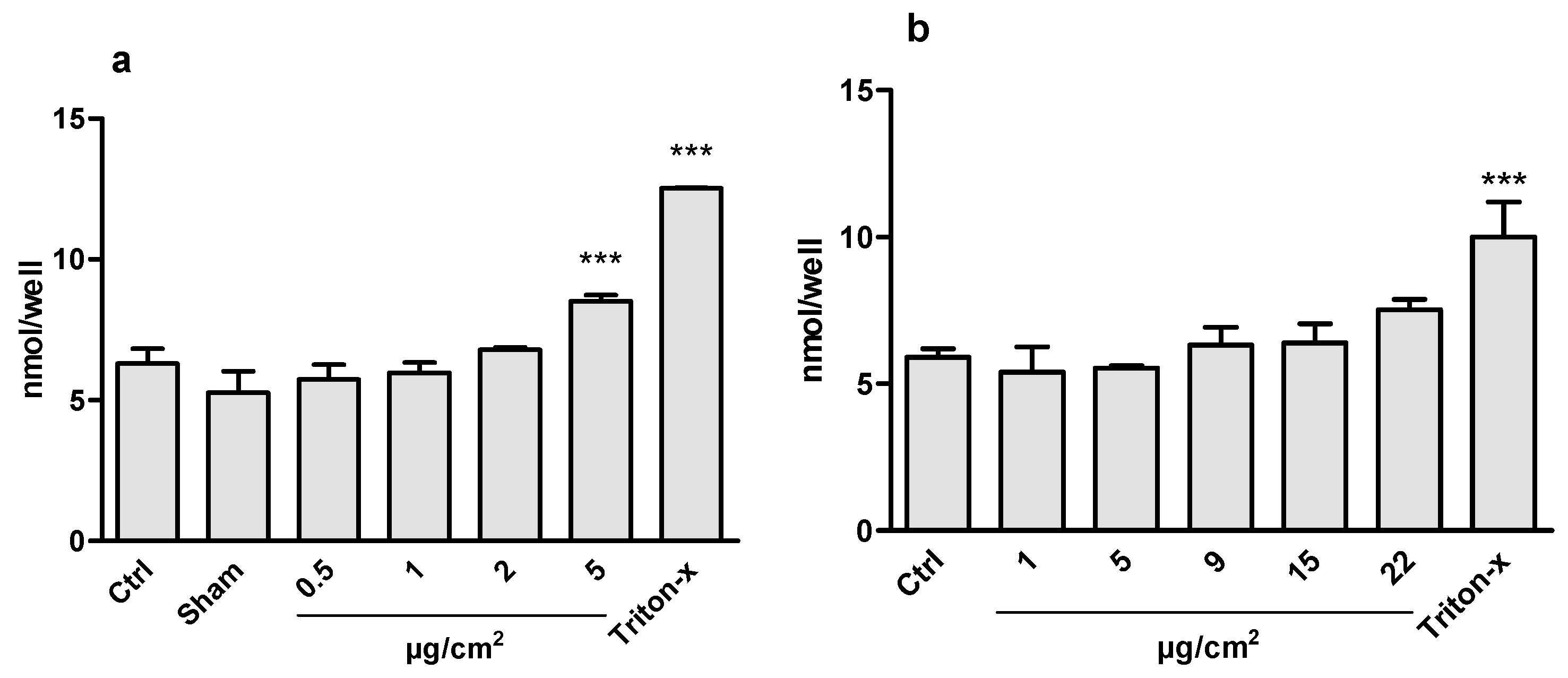
3.1. Aerosol Generation and CeO2 Deposition in ALI and Submerge
3.2. CeO2 Characterization in Cell Medium
3.3. Cytotoxicity
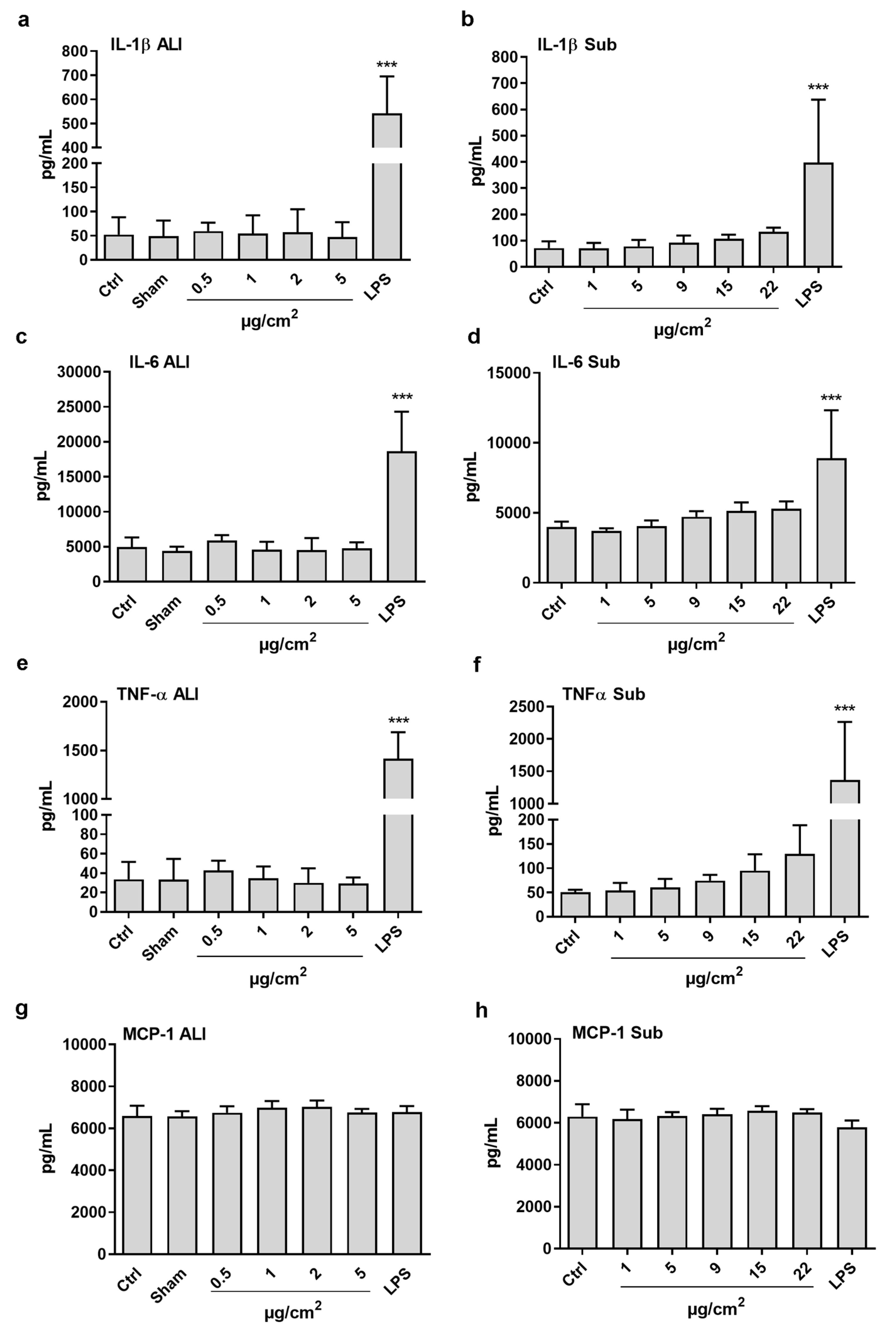
3.4. Inflammatory Potential in Co-Cultures
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Paur, H.-R.; Cassee, F.R.; Teeguarden, J.; Fissan, H.; Diabate, S.; Aufderheide, M.; Kreyling, W.G.; Hänninen, O.; Kasper, G.; Riediker, M.; et al. In-vitro cell exposure studies for the assessment of nanoparticle toxicity in the lung—A dialog between aerosol science and biology. J. Aerosol Sci. 2011, 42, 668–692. [Google Scholar] [CrossRef]
- Pradhan, S.; Hedberg, J.; Blomberg, E.; Wold, S.; Odnevall Wallinder, I. Effect of sonication on particle dispersion, administered dose and metal release of non-functionalized, non-inert metal nanoparticles. J. nanopart. Res. Interdiscip. Forum Nanoscale Sci. Technol. 2016, 18, 285. [Google Scholar] [CrossRef]
- Cronholm, P.; Midander, K.; Karlsson, H.L.; Elihn, K.; Odnevall Wallinder, I.; Moller, L. Effect of sonication and serum proteins on copper release from copper nanoparticles and the toxicity towards lung epithelial cells. Nanotoxicology 2011, 5, 269–281. [Google Scholar] [CrossRef]
- Lesniak, A.; Fenaroli, F.; Monopoli, M.P.; Aberg, C.; Dawson, K.A.; Salvati, A. Effects of the presence or absence of a protein corona on silica nanoparticle uptake and impact on cells. ACS Nano 2012, 6, 5845–5857. [Google Scholar] [CrossRef]
- Limbach, L.K.; Li, Y.; Grass, R.N.; Brunner, T.J.; Hintermann, M.A.; Muller, M.; Gunther, D.; Stark, W.J. Oxide nanoparticle uptake in human lung fibroblasts: Effects of particle size, agglomeration, and diffusion at low concentrations. Environ. Sci. Technol. 2005, 39, 9370–9376. [Google Scholar] [CrossRef] [PubMed]
- Teeguarden, J.G.; Hinderliter, P.M.; Orr, G.; Thrall, B.D.; Pounds, J.G. Particokinetics in vitro: Dosimetry considerations for in vitro nanoparticle toxicity assessments. Toxicol. Sci. Off. J. Soc. Toxicol. 2007, 95, 300–312. [Google Scholar] [CrossRef] [PubMed]
- Lenz, A.G.; Stoeger, T.; Cei, D.; Schmidmeir, M.; Semren, N.; Burgstaller, G.; Lentner, B.; Eickelberg, O.; Meiners, S.; Schmid, O. Efficient bioactive delivery of aerosolized drugs to human pulmonary epithelial cells cultured in air-liquid interface conditions. Am. J. Respir. Cell Mol. Biol. 2014, 51, 526–535. [Google Scholar] [CrossRef] [PubMed]
- Elihn, K.; Cronholm, P.; Karlsson, H.L.; Midander, K.; Odnevall Wallinder, I.; Moller, L. Cellular dose of partly soluble Cu particle aerosols at the air-liquid interface using an in vitro lung cell exposure system. J. Aerosol Med. Pulm. Drug Deliv. 2013, 26, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Geiser, M.; Jeannet, N.; Fierz, M.; Burtscher, H. Evaluating Adverse Effects of Inhaled Nanoparticles by Realistic In Vitro Technology. Nanomaterials 2017, 7, 49. [Google Scholar] [CrossRef]
- Cargnello, M.; Doan-Nguyen, V.V.; Gordon, T.R.; Diaz, R.E.; Stach, E.A.; Gorte, R.J.; Fornasiero, P.; Murray, C.B. Control of metal nanocrystal size reveals metal-support interface role for ceria catalysts. Science 2013, 341, 771–773. [Google Scholar] [CrossRef]
- Zhang, J.; Nazarenko, Y.; Zhang, L.; Calderon, L.; Lee, K.B.; Garfunkel, E.; Schwander, S.; Tetley, T.D.; Chung, K.F.; Porter, A.E.; et al. Impacts of a nanosized ceria additive on diesel engine emissions of particulate and gaseous pollutants. Environ. Sci. Technol. 2013, 47, 13077–13085. [Google Scholar] [CrossRef] [PubMed]
- Pagliari, F.; Mandoli, C.; Forte, G.; Magnani, E.; Pagliari, S.; Nardone, G.; Licoccia, S.; Minieri, M.; Di Nardo, P.; Traversa, E. Cerium oxide nanoparticles protect cardiac progenitor cells from oxidative stress. ACS Nano 2012, 6, 3767–3775. [Google Scholar] [CrossRef] [PubMed]
- Hirst, S.M.; Karakoti, A.S.; Tyler, R.D.; Sriranganathan, N.; Seal, S.; Reilly, C.M. Anti-inflammatory properties of cerium oxide nanoparticles. Small 2009, 5, 2848–2856. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.Y.; Zhao, H.; Mercer, R.R.; Barger, M.; Rao, M.; Meighan, T.; Schwegler-Berry, D.; Castranova, V.; Ma, J.K. Cerium oxide nanoparticle-induced pulmonary inflammation and alveolar macrophage functional change in rats. Nanotoxicology 2011, 5, 312–325. [Google Scholar] [CrossRef]
- Keller, J.; Wohlleben, W.; Ma-Hock, L.; Strauss, V.; Groters, S.; Kuttler, K.; Wiench, K.; Herden, C.; Oberdorster, G.; van Ravenzwaay, B.; et al. Time course of lung retention and toxicity of inhaled particles: Short-term exposure to nano-Ceria. Arch. Toxicol. 2014, 88, 2033–2059. [Google Scholar] [CrossRef]
- Dekkers, S.; Ma-Hock, L.; Lynch, I.; Russ, M.; Miller, M.R.; Schins, R.P.F.; Keller, J.; Romer, I.; Kuttler, K.; Strauss, V.; et al. Differences in the toxicity of cerium dioxide nanomaterials after inhalation can be explained by lung deposition, animal species and nanoforms. Inhal. Toxicol. 2018, 30, 273–286. [Google Scholar] [CrossRef]
- Loret, T.; Peyret, E.; Dubreuil, M.; Aguerre-Chariol, O.; Bressot, C.; le Bihan, O.; Amodeo, T.; Trouiller, B.; Braun, A.; Egles, C.; et al. Air-liquid interface exposure to aerosols of poorly soluble nanomaterials induces different biological activation levels compared to exposure to suspensions. Part. Fibre Toxicol. 2016, 13, 58. [Google Scholar] [CrossRef]
- Panas, A.; Comouth, A.; Saathoff, H.; Leisner, T.; Al-Rawi, M.; Simon, M.; Seemann, G.; Dossel, O.; Mulhopt, S.; Paur, H.R.; et al. Silica nanoparticles are less toxic to human lung cells when deposited at the air-liquid interface compared to conventional submerged exposure. Beilstein J. Nanotechnol. 2014, 5, 1590–1602. [Google Scholar] [CrossRef]
- Sing, C.; Friedrichs, S.; Ceccone, G.; Gibson, N.; Alstrup Jensen, K.; Levin, M.; Goenaga Infante, H.; Carlander, D.; Rasmussen, K. Cerium Dioxide, NM-211, NM-212, NM-213. Characterization and Test Item Preparation, 2014, JRC Repository: NM-Series of Representative Manufactured Nanomaterials; EUR 26649; The European Commission’s (EC): Brussels, Belgium, 2014. [Google Scholar]
- Ji, J.; Hedelin, A.; Malmlof, M.; Kessler, V.; Seisenbaeva, G.; Gerde, P.; Palmberg, L. Development of Combining of Human Bronchial Mucosa Models with XposeALI(R) for Exposure of Air Pollution Nanoparticles. PLoS ONE 2017, 12, e0170428. [Google Scholar] [CrossRef]
- Ji, J.; Upadhyay, S.; Xiong, X.; Malmlof, M.; Sandstrom, T.; Gerde, P.; Palmberg, L. Multi-cellular human bronchial models exposed to diesel exhaust particles: Assessment of inflammation, oxidative stress and macrophage polarization. Part. Fibre Toxicol. 2018, 15, 19. [Google Scholar] [CrossRef]
- Ji, J.; Ganguly, K.; Mihai, X.; Sun, J.; Malmlof, M.; Gerde, P.; Upadhyay, S.; Palmberg, L. Exposure of normal and chronic bronchitis-like mucosa models to aerosolized carbon nanoparticles: Comparison of pro-inflammatory oxidative stress and tissue injury/repair responses. Nanotoxicology 2019, 13, 1362–1379. [Google Scholar] [CrossRef] [PubMed]
- Stoehr, L.C.; Endes, C.; Radauer-Preiml, I.; Boyles, M.S.; Casals, E.; Balog, S.; Pesch, M.; Petri-Fink, A.; Rothen-Rutishauser, B.; Himly, M.; et al. Assessment of a panel of interleukin-8 reporter lung epithelial cell lines to monitor the pro-inflammatory response following zinc oxide nanoparticle exposure under different cell culture conditions. Part. Fibre Toxicol. 2015, 12, 29. [Google Scholar] [CrossRef] [PubMed]
- Hinderliter, P.M.; Minard, K.R.; Orr, G.; Chrisler, W.B.; Thrall, B.D.; Pounds, J.G.; Teeguarden, J.G. ISDD: A computational model of particle sedimentation, diffusion and target cell dosimetry for in vitro toxicity studies. Part. Fibre Toxicol. 2010, 7, 36. [Google Scholar] [CrossRef]
- Herzog, F.; Loza, K.; Balog, S.; Clift, M.J.; Epple, M.; Gehr, P.; Petri-Fink, A.; Rothen-Rutishauser, B. Mimicking exposures to acute and lifetime concentrations of inhaled silver nanoparticles by two different in vitro approaches. Beilstein J. Nanotechnol. 2014, 5, 1357–1370. [Google Scholar] [CrossRef]
- Latvala, S.; Vare, D.; Karlsson, H.L.; Elihn, K. In vitro genotoxicity of airborne Ni-NP in air-liquid interface. J. Appl. Toxicol. JAT 2017, 37, 1420–1427. [Google Scholar] [CrossRef]
- Loret, T.; Rogerieux, F.; Trouiller, B.; Braun, A.; Egles, C.; Lacroix, G. Predicting the in vivo pulmonary toxicity induced by acute exposure to poorly soluble nanomaterials by using advanced in vitro methods. Part. Fibre Toxicol. 2018, 15, 25. [Google Scholar] [CrossRef]
- Bhattacharya, K.; Kilic, G.; Costa, P.M.; Fadeel, B. Cytotoxicity screening and cytokine profiling of nineteen nanomaterials enables hazard ranking and grouping based on inflammogenic potential. Nanotoxicology 2017, 11, 809–826. [Google Scholar] [CrossRef]
- Cho, W.S.; Duffin, R.; Bradley, M.; Megson, I.L.; MacNee, W.; Lee, J.K.; Jeong, J.; Donaldson, K. Predictive value of in vitro assays depends on the mechanism of toxicity of metal oxide nanoparticles. Part. Fibre Toxicol. 2013, 10, 55. [Google Scholar] [CrossRef]
- Dunnick, K.M.; Morris, A.M.; Badding, M.A.; Barger, M.; Stefaniak, A.B.; Sabolsky, E.M.; Leonard, S.S. Evaluation of the effect of valence state on cerium oxide nanoparticle toxicity following intratracheal instillation in rats. Nanotoxicology 2016, 10, 992–1000. [Google Scholar] [CrossRef]
- Wiemann, M.; Vennemann, A.; Sauer, U.G.; Wiench, K.; Ma-Hock, L.; Landsiedel, R. An in vitro alveolar macrophage assay for predicting the short-term inhalation toxicity of nanomaterials. J. Nanobiotechnol. 2016, 14, 16. [Google Scholar] [CrossRef]
- Teeguarden, J.G.; Mikheev, V.B.; Minard, K.R.; Forsythe, W.C.; Wang, W.; Sharma, G.; Karin, N.; Tilton, S.C.; Waters, K.M.; Asgharian, B.; et al. Comparative iron oxide nanoparticle cellular dosimetry and response in mice by the inhalation and liquid cell culture exposure routes. Part. Fibre Toxicol. 2014, 11, 46. [Google Scholar] [CrossRef] [PubMed]


| Deposited Mass ALI (μg/cm2) | Deposited Mass sub. (μg/cm2) | Added Mass sub. (μg/cm2) |
|---|---|---|
| 0 | 0 | 0 |
| 0.5 ± 0.05 | 1 ± 0.4 | 2 |
| 1 ± 0.2 | 5 ± 0.6 | 10 |
| 2 ± 0.6 | 9 ± 1.5 | 20 |
| 5 ± 0.7 | 15 ± 1.1 | 30 |
| - | 22 ± 1.9 | 40 |
| Size (nm) | Scattered Light Intensity (Kcounts/s) | |||
|---|---|---|---|---|
| 0 h | 24 h | 0 h | 24 h | |
| Probe sonication (MilliQ + albumin) | 236 ± 11 | 235 ± 17 | 2150 ± 56 | 868 ± 241 |
| Bath sonication (+ serum) | 865 ± 238 | 396 ± 47 | 1396 ± 186 | 265 ± 122 |
| Bath sonication (- serum) | 3279 ± 177 | - | 1392 ± 35 | 8 ± 8 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cappellini, F.; Di Bucchianico, S.; Karri, V.; Latvala, S.; Malmlöf, M.; Kippler, M.; Elihn, K.; Hedberg, J.; Odnevall Wallinder, I.; Gerde, P.; et al. Dry Generation of CeO2 Nanoparticles and Deposition onto a Co-Culture of A549 and THP-1 Cells in Air-Liquid Interface—Dosimetry Considerations and Comparison to Submerged Exposure. Nanomaterials 2020, 10, 618. https://doi.org/10.3390/nano10040618
Cappellini F, Di Bucchianico S, Karri V, Latvala S, Malmlöf M, Kippler M, Elihn K, Hedberg J, Odnevall Wallinder I, Gerde P, et al. Dry Generation of CeO2 Nanoparticles and Deposition onto a Co-Culture of A549 and THP-1 Cells in Air-Liquid Interface—Dosimetry Considerations and Comparison to Submerged Exposure. Nanomaterials. 2020; 10(4):618. https://doi.org/10.3390/nano10040618
Chicago/Turabian StyleCappellini, Francesca, Sebastiano Di Bucchianico, Venkatanaidu Karri, Siiri Latvala, Maria Malmlöf, Maria Kippler, Karine Elihn, Jonas Hedberg, Inger Odnevall Wallinder, Per Gerde, and et al. 2020. "Dry Generation of CeO2 Nanoparticles and Deposition onto a Co-Culture of A549 and THP-1 Cells in Air-Liquid Interface—Dosimetry Considerations and Comparison to Submerged Exposure" Nanomaterials 10, no. 4: 618. https://doi.org/10.3390/nano10040618
APA StyleCappellini, F., Di Bucchianico, S., Karri, V., Latvala, S., Malmlöf, M., Kippler, M., Elihn, K., Hedberg, J., Odnevall Wallinder, I., Gerde, P., & Karlsson, H. L. (2020). Dry Generation of CeO2 Nanoparticles and Deposition onto a Co-Culture of A549 and THP-1 Cells in Air-Liquid Interface—Dosimetry Considerations and Comparison to Submerged Exposure. Nanomaterials, 10(4), 618. https://doi.org/10.3390/nano10040618






