Genotoxicity of Aluminum and Aluminum Oxide Nanomaterials in Rats Following Oral Exposure
Abstract
1. Introduction
2. Material and Methods
2.1. Chemicals, NMs and Dispersion
2.2. Animals and Experimental Design
2.3. Tissue Collection and Sample Preparation
2.4. Alkaline Comet Assay and FpG-Modified Comet Assay
2.5. Bone Marrow Micronucleus Assay (BMMN)
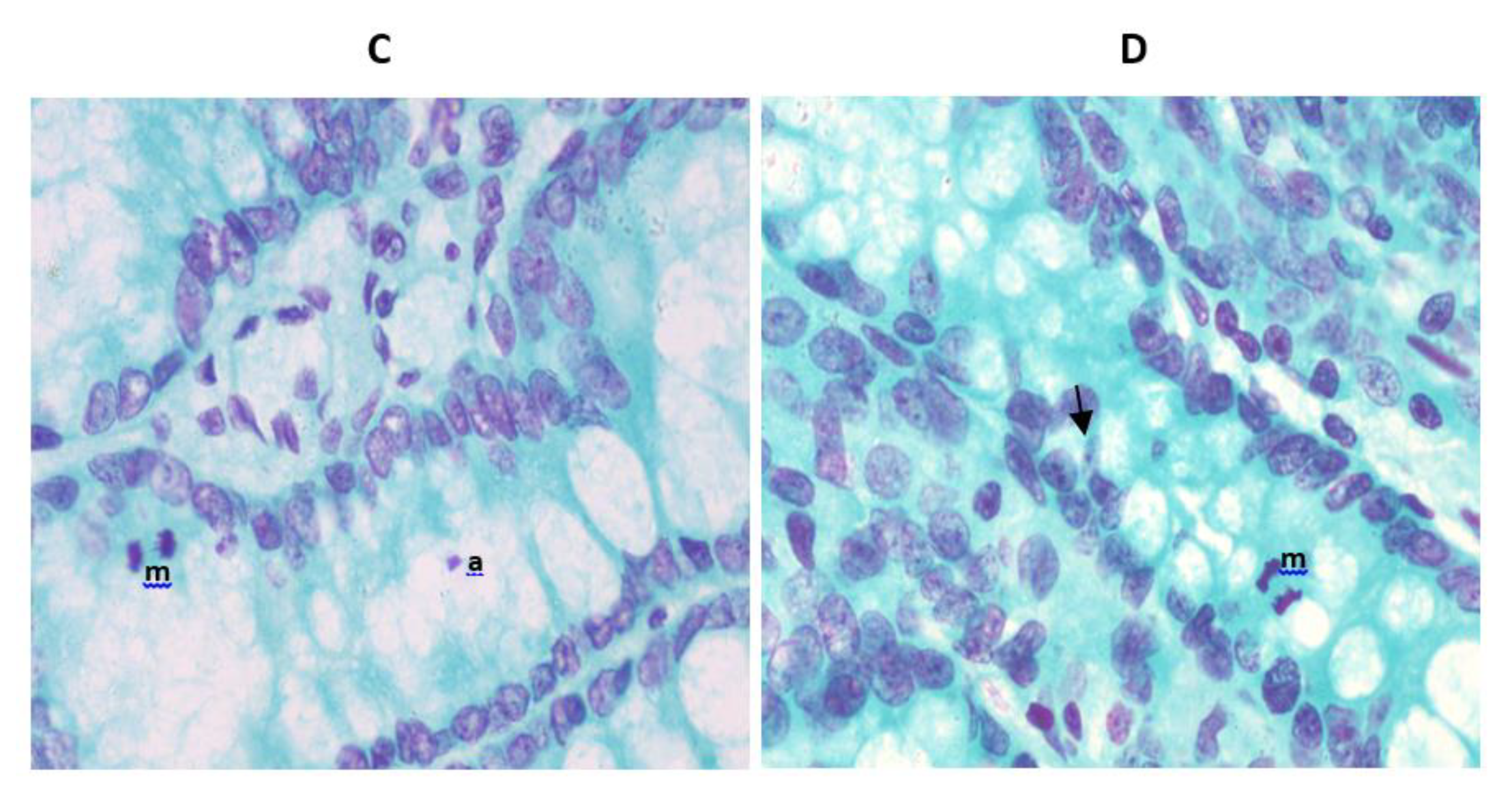
2.6. Colon Micronucleus Assay
2.7. Statistical Analysis
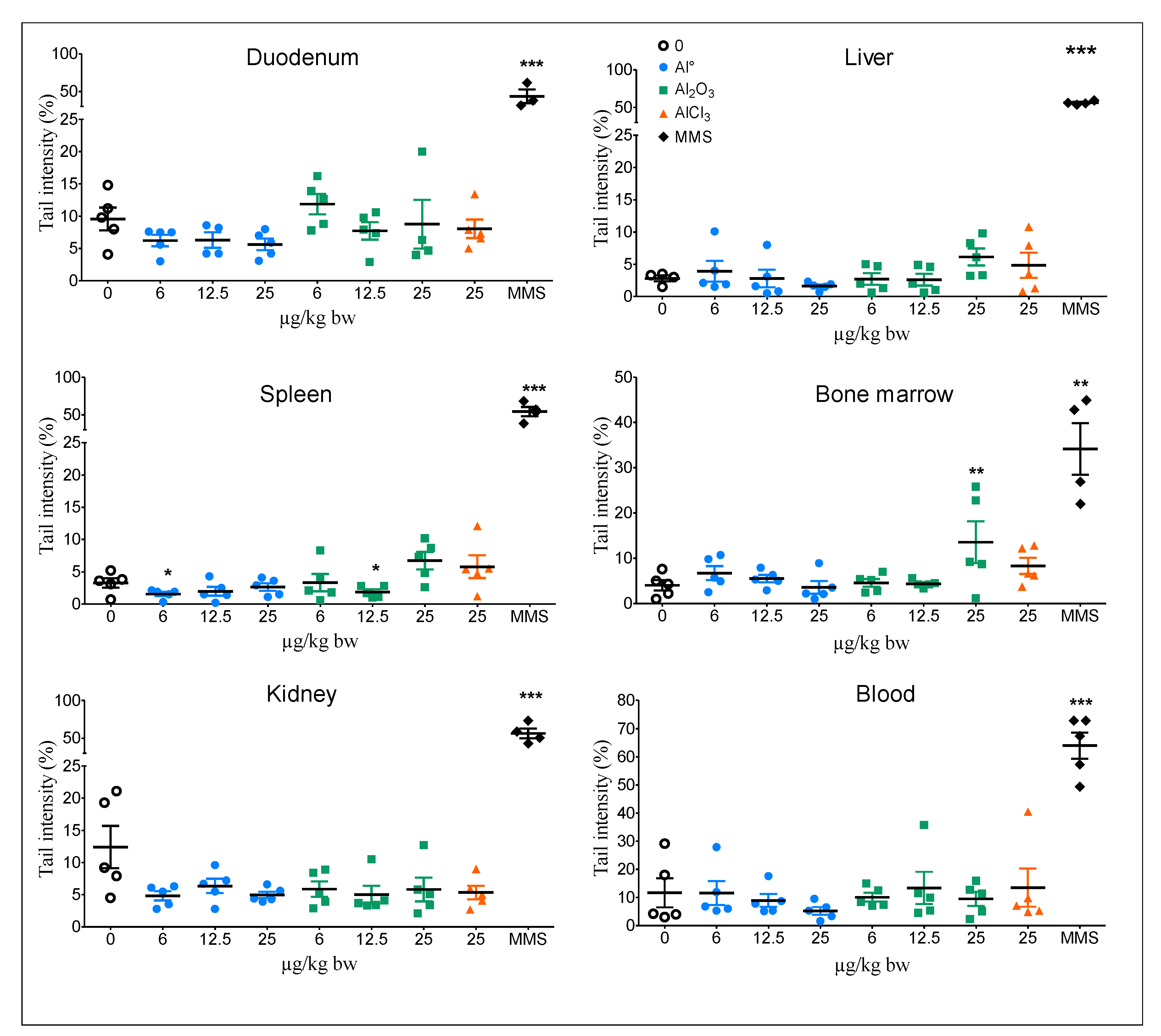
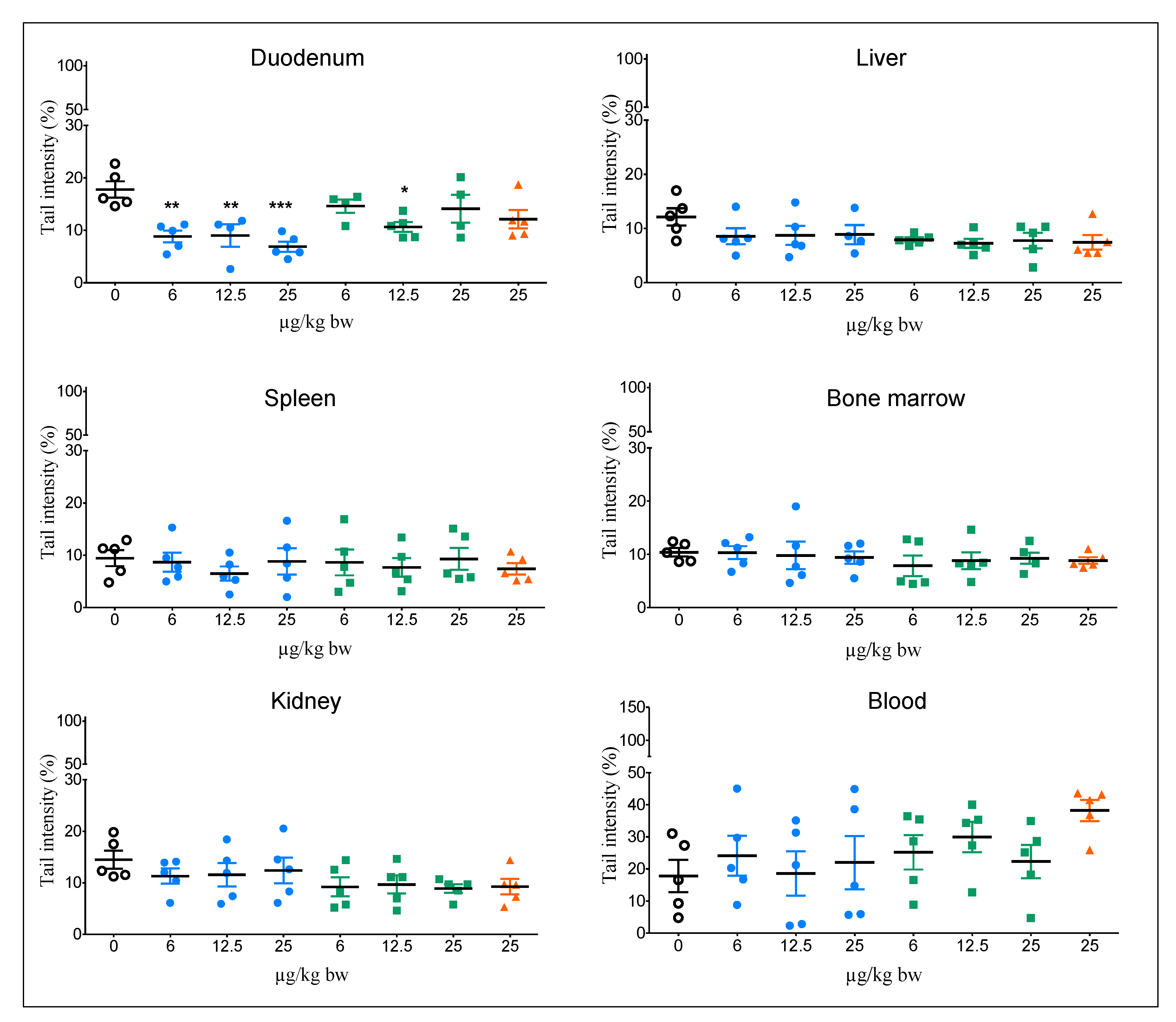
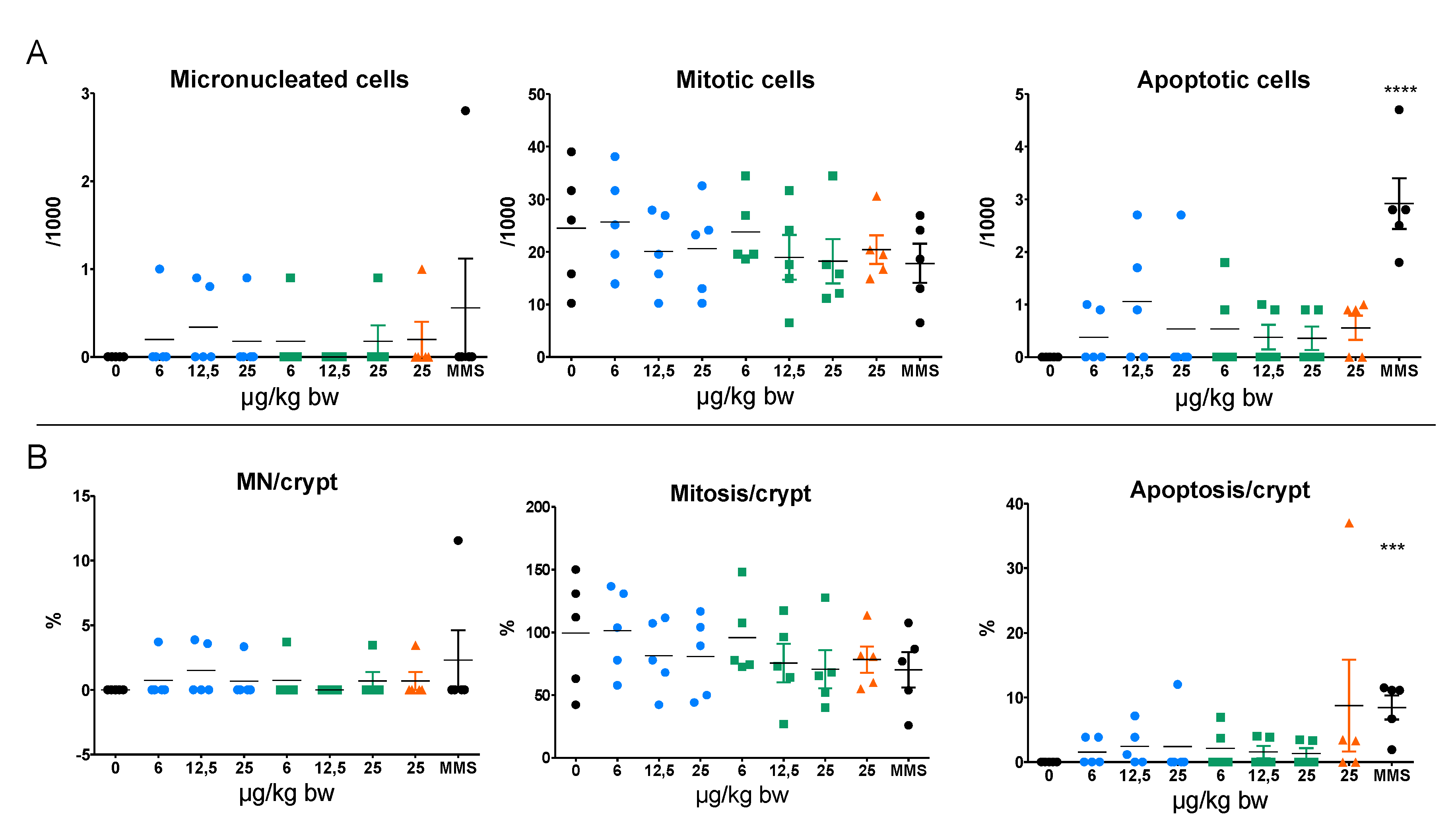
3. Results
3.1. Comet Assay
3.2. Bone Marrow Micronucleus Assay (BMMN)
3.3. Micronucleus Assay in Colon
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- European Food Safety Authority. Safety of aluminium from dietary intake—Scientific Opinion of the Panel on Food Additives, Flavourings, Processing Aids and Food Contact Materials (AFC). EFSA J. 2008, 6, 754. [Google Scholar]
- Willhite, C.C.; Karyakina, N.A.; Yokel, R.A.; Yenugadhati, N.; Wisniewski, T.M.; Arnold, I.M.; Momoli, F.; Krewski, D. Systematic review of potential health risks posed by pharmaceutical, occupational and consumer exposures to metallic and nanoscale aluminum, aluminum oxides, aluminum hydroxide and its soluble salts. Crit. Rev. Toxicol. 2014, 44, 1–80. [Google Scholar] [CrossRef] [PubMed]
- Vignal, C.; Desreumaux, P.; Body-Malapel, M. Gut: An underestimated target organ for Aluminum. Morphologie 2016, 100, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Saiyed, S.M.; Yokel, R.A. Aluminium content of some foods and food products in the USA, with aluminium food additives. Food Addit. Contam. 2005, 22, 234–244. [Google Scholar] [CrossRef] [PubMed]
- Krewski, D.; Yokel, R.A.; Nieboer, E.; Borchelt, D.; Cohen, J.; Harry, J.; Kacew, S.; Lindsay, J.; Mahfouz, A.M.; Rondeau, V. Human health risk assessment for aluminium, aluminium oxide, and aluminium hydroxide. J. Toxicol. Environ. Health. B Crit. Rev. 2007, 10, 1–269. [Google Scholar] [CrossRef] [PubMed]
- Gehrke, I.; Geiser, A.; Somborn-Schulz, A. Innovations in nanotechnology for water treatment. Nanotechnol. Sci. Appl. 2015, 8, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Castranova, V. Toxicology of nanomaterials used in nanomedicine. J. Toxicol. Environ. Health B Crit. Rev. 2011, 14, 593–632. [Google Scholar] [CrossRef]
- Frey, A.; Neutra, M.R.; Robey, F.A. Peptomer aluminum oxide nanoparticle conjugates as systemic and mucosal vaccine candidates: Synthesis and characterization of a conjugate derived from the C4 domain of HIV-1MN gp120. Bioconjug. Chem. 1997, 8, 424–433. [Google Scholar] [CrossRef]
- Narayan, R.J.; Adiga, S.P.; Pellin, M.J.; Curtiss, L.A.; Hryn, A.J.; Stafslien, S.; Chisholm, B.; Shih, C.-C.; Shih, C.-M.; Lin, S.-J.; et al. Atomic layer deposition-based functionalization of materials for medical and environmental health applications. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2010, 368, 2033–2064. [Google Scholar] [CrossRef]
- JECFA Summary and Conclusions of the Sixty-Seventh Meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA). Available online: http://www.fao.org/3/a-at874e.pdf (accessed on 1 February 2020).
- Balasubramanyam, A.; Sailaja, N.; Mahboob, M.; Rahman, M.F.; Misra, S.; Hussain, S.M.; Grover, P. Evaluation of genotoxic effects of oral exposure to aluminum oxide nanomaterials in rat bone marrow. Mutat. Res. 2009, 676, 41–47. [Google Scholar] [CrossRef]
- Park, E.J.; Sim, J.; Kim, Y.; Han, B.S.; Yoon, C.; Lee, S.; Cho, M.H.; Lee, B.S.; Kim, J.H. A 13-week repeated-dose oral toxicity and bioaccumulation of aluminum oxide nanoparticles in mice. Arch. Toxicol. 2015, 89, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Shrivastava, R.; Raza, S.; Yadav, A.; Kushwaha, P.; Flora, S.J. Effects of sub-acute exposure to TiO2, ZnO and Al2O3 nanoparticles on oxidative stress and histological changes in mouse liver and brain. Drug Chem. Toxicol. 2014, 37, 336–347. [Google Scholar] [CrossRef] [PubMed]
- Prabhakar, P.V.; Reddy, U.A.; Singh, S.P.; Balasubramanyam, A.; Rahman, M.F.; Indu Kumari, S.; Agawane, S.B.; Murty, U.S.; Grover, P.; Mahboob, M. Oxidative stress induced by aluminum oxide nanomaterials after acute oral treatment in Wistar rats. J. Appl. Toxicol. 2012, 32, 436–445. [Google Scholar] [CrossRef] [PubMed]
- ECHA. Registration dossier Aluminum oxide. Available online: https://echa.europa.eu/fr/registration-dossier/-/registered-dossier/16039/7/7/1 (accessed on 5 January 2020).
- Tsaousi, A.; Jones, E.; Case, C.P. The in vitro genotoxicity of orthopaedic ceramic (Al2O3) and metal (CoCr alloy) particles. Mutat. Res. 2010, 697, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Alarifi, S.; Ali, D.; Alkahtani, S. Nanoalumina induces apoptosis by impairing antioxidant enzyme systems in human hepatocarcinoma cells. Int. J. Nanomed. 2015, 10, 3751–3760. [Google Scholar]
- Rajiv, S.; Jerobin, J.; Saranya, V.; Nainawat, M.; Sharma, A.; Makwana, P.; Gayathri, C.; Bharath, L.; Singh, M.; Kumar, M.; et al. Comparative cytotoxicity and genotoxicity of cobalt (II, III) oxide, iron (III) oxide, silicon dioxide, and aluminum oxide nanoparticles on human lymphocytes in vitro. Hum. Exp. Toxicol. 2016, 35, 170–183. [Google Scholar] [CrossRef]
- Di Virgilio, A.L.; Reigosa, M.; Arnal, P.M.; Fernandez Lorenzo de Mele, M. Comparative study of the cytotoxic and genotoxic effects of titanium oxide and aluminium oxide nanoparticles in Chinese hamster ovary (CHO-K1) cells. J. Hazard. Mater. 2010, 177, 711–718. [Google Scholar] [CrossRef]
- Lima, P.D.; Leite, D.S.; Vasconcellos, M.C.; Cavalcanti, B.C.; Santos, R.A.; Costa-Lotufo, L.V.; Pessoa, C.; Moraes, M.O.; Burbano, R.R. Genotoxic effects of aluminum chloride in cultured human lymphocytes treated in different phases of cell cycle. Food Chem. Toxicol. 2007, 45, 1154–1159. [Google Scholar] [CrossRef]
- Lankoff, A.; Banasik, A.; Duma, A.; Ochniak, E.; Lisowska, H.; Kuszewski, T.; Gozdz, S.; Wojcik, A. A comet assay study reveals that aluminium induces DNA damage and inhibits the repair of radiation-induced lesions in human peripheral blood lymphocytes. Toxicol. Lett. 2006, 161, 27–36. [Google Scholar] [CrossRef]
- Paz, L.N.; Moura, L.M.; Feio, D.C.; Cardoso, M.S.; Ximenes, W.L.; Montenegro, R.C.; Alves, A.P.; Burbano, R.R.; Lima, P.D. Evaluation of in vivo and in vitro toxicological and genotoxic potential of aluminum chloride. Chemosphere 2017, 175, 130–137. [Google Scholar] [CrossRef]
- Hartmann, N.B.; Jensen, K.A.; Baun, A.; Rasmussen, K.; Rauscher, H.; Tantra, R.; Cupi, D.; Gilliland, D.; Pianella, F.; Riego Sintes, J.M. Techniques and Protocols for Dispersing Nanoparticle Powders in Aqueous Media-Is there a Rationale for Harmonization? J. Toxicol. Environ. Health B Crit. Rev. 2015, 18, 299–326. [Google Scholar] [CrossRef] [PubMed]
- Jensen, K.A.; Kembouche, Y.; Christiansen, E.; Jacobsen, N.R.; Wallin, H.; Guiot, C.; Spalla, O.; Witschger, O. Final Protocol for Producing Suitable Manufactured Nanomaterial Exposure Media. Web-Report. The Generic NANOGENOTOX Dispersion Protocol—Standard Operation Procedure (SOP). Available online: https://www.anses.fr/en/system/files/nanogenotox_deliverable_5.pdf (accessed on 1 February 2020).
- Sieg, H.; Kastner, C.; Krause, B.; Meyer, T.; Burel, A.; Bohmert, L.; Lichtenstein, D.; Jungnickel, H.; Tentschert, J.; Laux, P.; et al. Impact of an Artificial Digestion Procedure on Aluminum-Containing Nanomaterials. Langmuir 2017, 33, 10726–10735. [Google Scholar] [CrossRef] [PubMed]
- Krause, B.; Meyer, T.; Sieg, H.; Kästner, C.; Reichardt, P.; Tentschert, J.; Jungnickel, H.; Estrela-Lopis, I.; Burel, A.; Chevance, S.; et al. Characterization of aluminum, aluminum oxide and titanium dioxide nanomaterials using a combination of methods for particle surface and size analysis. RSC Adv. 2018, 8, 14377–14388. [Google Scholar] [CrossRef]
- OCDE. Test No. 489: In Vivo Mammalian Alkaline Comet Assay; OECD Publishing: Paris, France, 2016. [Google Scholar]
- Tarantini, A.; Huet, S.; Jarry, G.; Lanceleur, R.; Poul, M.; Tavares, A.; Vital, N.; Louro, H.; Joao Silva, M.; Fessard, V. Genotoxicity of synthetic amorphous silica nanoparticles in rats following short-term exposure. Part 1: Oral route. Environ. Mol. Mutagen. 2015, 56, 218–227. [Google Scholar] [CrossRef]
- McKelvey-Martin, V.J.; Green, M.H.; Schmezer, P.; Pool-Zobel, B.L.; De Meo, M.P.; Collins, A. The single cell gel electrophoresis assay (comet assay): A European review. Mutat. Res. 1993, 288, 47–63. [Google Scholar] [CrossRef]
- Zeller, A.; Duran-Pacheco, G.; Guerard, M. An appraisal of critical effect sizes for the benchmark dose approach to assess dose-response relationships in genetic toxicology. Arch. Toxicol. 2017, 91, 3799–3807. [Google Scholar] [CrossRef]
- Tice, R.R.; Agurell, E.; Anderson, D.; Burlinson, B.; Hartmann, A.; Kobayashi, H.; Miyamae, Y.; Rojas, E.; Ryu, J.C.; Sasaki, Y.F. Single cell gel/comet assay: Guidelines for in vitro and in vivo genetic toxicology testing. Environ. Mol. Mutagen. 2000, 35, 206–221. [Google Scholar] [CrossRef]
- OCDE. Test No. 474: Mammalian Erythrocyte Micronucleus Test; OECD Publishing: Paris, France, 2016. [Google Scholar]
- Zhang, Q.; Wang, H.; Ge, C.; Duncan, J.; He, K.; Adeosun, S.O.; Xi, H.; Peng, H.; Niu, Q. Alumina at 50 and 13 nm nanoparticle sizes have potential genotoxicity. J. Appl. Toxicol. 2017, 37, 1053–1064. [Google Scholar] [CrossRef]
- Akbaba, G.B.; Turkez, H. Investigation of the Genotoxicity of Aluminum Oxide, beta-Tricalcium Phosphate, and Zinc Oxide Nanoparticles In Vitro. Int. J. Toxicol. 2018, 37, 216–222. [Google Scholar] [CrossRef]
- Hashimoto, M.; Imazato, S. Cytotoxic and genotoxic characterization of aluminum and silicon oxide nanoparticles in macrophages. Dent. Mater. 2015, 31, 556–564. [Google Scholar] [CrossRef]
- Jalili, P.; Huet, S.; Burel, A.; Krause, B.-C.; Fontana, C.; Gauffre, F.; Guichard, Y.; Laux, P.; Luch, A.; Hogeveen, K.; et al. Genotoxic impact of aluminum-containing nanomaterials in human intestinal and hepatic cells. Manuscript in preparation.
- McKenna, D.J.; Gallus, M.; McKeown, S.R.; Downes, C.S.; McKelvey-Martin, V.J. Modification of the alkaline Comet assay to allow simultaneous evaluation of mitomycin C-induced DNA cross-link damage and repair of specific DNA sequences in RT4 cells. DNA Repair 2003, 2, 879–890. [Google Scholar] [CrossRef]
- Chen, P.; Sjogren, C.A.; Larsen, P.B.; Schnittger, A. A multi-level response to DNA damage induced by aluminium. Plant J. 2019, 98, 479–491. [Google Scholar] [CrossRef] [PubMed]
- Yousef, M.I.; Mutar, T.F.; Kamel, M.A.E. Hepato-renal toxicity of oral sub-chronic exposure to aluminum oxide and/or zinc oxide nanoparticles in rats. Toxicol. Rep. 2019, 6, 336–346. [Google Scholar] [CrossRef] [PubMed]
- Minigalieva, I.A.; Katsnelson, B.A.; Privalova, L.I.; Sutunkova, M.P.; Gurvich, V.B.; Shur, V.Y.; Shishkina, E.V.; Valamina, I.E.; Makeyev, O.H.; Panov, V.G.; et al. Combined Subchronic Toxicity of Aluminum (III), Titanium (IV) and Silicon (IV) Oxide Nanoparticles and Its Alleviation with a Complex of Bioprotectors. Int. J. Mol. Sci. 2018, 19, 837. [Google Scholar] [CrossRef]
- Morsy, G.M.; El-Ala, K.S.; Ali, A.A. Studies on fate and toxicity of nanoalumina in male albino rats: Lethality, bioaccumulation and genotoxicity. Toxicol. Ind. Health 2016, 32, 344–359. [Google Scholar] [CrossRef]
- Demir, E.; Akca, H.; Turna, F.; Aksakal, S.; Burgucu, D.; Kaya, B.; Tokgun, O.; Vales, G.; Creus, A.; Marcos, R. Genotoxic and cell-transforming effects of titanium dioxide nanoparticles. Environ. Res. 2015, 136, 300–308. [Google Scholar] [CrossRef]
- Gao, C.-H.; Mortimer, M.; Zhang, M.; Holden, P.A.; Cai, P.; Wu, S.; Xin, Y.; Wu, Y.; Huang, Q. Impact of metal oxide nanoparticles on in vitro DNA amplification. Peer J. 2019, 7, e7228. [Google Scholar] [CrossRef]
- Shah, S.A.; Yoon, G.H.; Ahmad, A.; Ullah, F.; Amin, F.U.; Kim, M.O. Nanoscale-alumina induces oxidative stress and accelerates amyloid beta (Aβ) production in ICR female mice. Nanoscale 2015, 7, 15225–15237. [Google Scholar] [CrossRef]
- Canli, E.G.; Atli, G.; Canli, M. Response of the antioxidant enzymes of the erythrocyte and alterations in the serum biomarkers in rats following oral administration of nanoparticles. Environ. Toxicol. Pharmacol. 2017, 50, 145–150. [Google Scholar] [CrossRef]
- Li, X.; Zhang, C.; Zhang, X.; Wang, S.; Meng, Q.; Wu, S.; Yang, H.; Xia, Y.; Chen, R. An acetyl-L-carnitine switch on mitochondrial dysfunction and rescue in the metabolomics study on aluminum oxide nanoparticles. Part Fibre Toxicol. 2016, 13, 4. [Google Scholar] [CrossRef]
- Sliwinska, A.; Kwiatkowski, D.; Czarny, P.; Milczarek, J.; Toma, M.; Korycinska, A.; Szemraj, J.; Sliwinski, T. Genotoxicity and cytotoxicity of ZnO and Al2O3 nanoparticles. Toxicol. Mech. Methods. 2015, 25, 176–183. [Google Scholar] [CrossRef] [PubMed]
- Morsy, G.M.; El-Ala, K.S.; Ali, A.A. Studies on fate and toxicity of nanoalumina in male albino rats: Some haematological, biochemical and histological aspects. Toxicol. Ind. Health. 2016, 32, 634–655. [Google Scholar] [CrossRef] [PubMed]
- Canli, E.G.; Atli, G.; Canli, M. Responses of biomarkers belonging to different metabolic systems of rats following oral administration of aluminium nanoparticle. Environ. Toxicol. Pharm. 2019, 69, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Demir, E.; Burgucu, D.; Turna, F.; Aksakal, S.; Kaya, B. Determination of TiO2, ZrO2, and Al2O3 nanoparticles on genotoxic responses in human peripheral blood lymphocytes and cultured embyronic kidney cells. J. Toxicol. Environ. Health A 2013, 76, 990–1002. [Google Scholar] [CrossRef]
- M’Rad, I.; Jeljeli, M.; Rihane, N.; Hilber, P.; Sakly, M.; Amara, S. Aluminium oxide nanoparticles compromise spatial learning and memory performance in rats. Excli. J. 2018, 17, 200–210. [Google Scholar]
- Turkez, H.; Yousef, M.I.; Geyikoglu, F. Propolis prevents aluminium-induced genetic and hepatic damages in rat liver. Food Chem. Toxicol. 2010, 48, 2741–2746. [Google Scholar] [CrossRef]
- Geyikoglu, F.; Turkez, H.; Bakir, T.O.; Cicek, M. The genotoxic, hepatotoxic, nephrotoxic, haematotoxic and histopathological effects in rats after aluminium chronic intoxication. Toxicol. Ind. Health 2013, 29, 780–791. [Google Scholar] [CrossRef]
- Turkez, H.; Geyikoglu, F.; Tatar, A. Borax counteracts genotoxicity of aluminum in rat liver. Toxicol. Ind. Health 2013, 29, 775–779. [Google Scholar] [CrossRef]
- Cao, Z.; Geng, X.; Jiang, X.; Gao, X.; Liu, K.; Li, Y. Melatonin Attenuates AlCl3-Induced Apoptosis and Osteoblastic Differentiation Suppression by Inhibiting Oxidative Stress in MC3T3-E1 Cells. Biol. Trace Elem. Res. 2019. [Google Scholar] [CrossRef]
- Martinez, C.S.; Vera, G.; Ocio, J.A.U.; Pecanha, F.M.; Vassallo, D.V.; Miguel, M.; Wiggers, G.A. Aluminum exposure for 60days at an equivalent human dietary level promotes peripheral dysfunction in rats. J. Inorg. Biochem. 2018, 181, 169–176. [Google Scholar] [CrossRef]
- Yang, X.; Yu, K.; Wang, H.; Zhang, H.; Bai, C.; Song, M.; Han, Y.; Shao, B.; Li, Y.; Li, X. Bone impairment caused by AlCl3 is associated with activation of the JNK apoptotic pathway mediated by oxidative stress. Food Chem. Toxicol. 2018, 116, 307–314. [Google Scholar] [CrossRef]
- Cheng, D.; Zhang, X.; Xu, L.; Li, X.; Hou, L.; Wang, C. Protective and prophylactic effects of chlorogenic acid on aluminum-induced acute hepatotoxicity and hematotoxicity in mice. Chem. Interact. 2017, 273, 125–132. [Google Scholar] [CrossRef] [PubMed]
- D’Souza, S.P.; Vijayalaxmi, K.K.; Naik, P. Assessment of genotoxicity of aluminium acetate in bone marrow, male germ cells and fetal liver cells of Swiss albino mice. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2014, 766, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Banasik, A.; Lankoff, A.; Piskulak, A.; Adamowska, K.; Lisowska, H.; Wojcik, A. Aluminum-induced micronuclei and apoptosis in human peripheral-blood lymphocytes treated during different phases of the cell cycle. Environ. Toxicol. 2005, 20, 402–406. [Google Scholar] [CrossRef] [PubMed]
- Villarini, M.; Gambelunghe, A.; Giustarini, D.; Ambrosini, M.V.; Fatigoni, C.; Rossi, R.; Dominici, L.; Levorato, S.; Muzi, G.; Piobbico, D.; et al. No evidence of DNA damage by co-exposure to extremely low frequency magnetic fields and aluminum on neuroblastoma cell lines. Mutat. Res. 2017, 823, 11–21. [Google Scholar] [CrossRef]
| NM | NM-Code | Average Particle Size a (nm) | SSA b (m2/g) | Purity c | Bulk Density, True Density d (g/cm3) | Morphology | Pdi e | Z-Average Size in the Stock Solution Dispersion at 0 h e (nm) | Solubility f (24h) (%) |
|---|---|---|---|---|---|---|---|---|---|
| Al0 | NM-0015-HP | 18 | 40–60 | >99% | 2.7 | Spherical | 0.17 ± 0.004 | 254 ± 4 | 0.48 ± 0.02 |
| 0.008–0.2 | |||||||||
| γ-Al2o3 | NM-0036-HP | 20 | <200 | 99% | - | Spherical | 0.23 ± 0.015 | 168 ± 3 | 0.15 ± 0.01 |
| 0.9 |
| Genotoxicity | Myelotoxicity | ||
|---|---|---|---|
| Micronucleated PCE/2 000 PCE | %PCEs | ||
| Doses (mg/kg bw/day) | Mean ± SD | Mean ± SD | |
| Control | 1.3 ± 0.91 | 72.5 ± 26 | |
| Al0 | 6 | 2.0 ± 2.0 | 66.3 ± 13 |
| 12.5 | 1.5 ± 0.8 | 71.5 ± 8 | |
| 25 | 2.1 ± 1.0 | 74.7 ± 16 | |
| Al2O3 | 6 | 1.1 ± 0.7 | 68.4 ± 18 |
| 12.5 | 1.6 ± 1.3 | 65.8 ± 17 | |
| 25 | 0.9 ± 0.8 | 56.7 ± 17 | |
| AlCl3 | 25 | 1.5 ± 0.7 | 65.9 ± 19 |
| MMS | 100, 100, 80 | 16.7 ± 3.7 * | 44.5 ± 12 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jalili, P.; Huet, S.; Lanceleur, R.; Jarry, G.; Hegarat, L.L.; Nesslany, F.; Hogeveen, K.; Fessard, V. Genotoxicity of Aluminum and Aluminum Oxide Nanomaterials in Rats Following Oral Exposure. Nanomaterials 2020, 10, 305. https://doi.org/10.3390/nano10020305
Jalili P, Huet S, Lanceleur R, Jarry G, Hegarat LL, Nesslany F, Hogeveen K, Fessard V. Genotoxicity of Aluminum and Aluminum Oxide Nanomaterials in Rats Following Oral Exposure. Nanomaterials. 2020; 10(2):305. https://doi.org/10.3390/nano10020305
Chicago/Turabian StyleJalili, Pégah, Sylvie Huet, Rachelle Lanceleur, Gérard Jarry, Ludovic Le Hegarat, Fabrice Nesslany, Kevin Hogeveen, and Valérie Fessard. 2020. "Genotoxicity of Aluminum and Aluminum Oxide Nanomaterials in Rats Following Oral Exposure" Nanomaterials 10, no. 2: 305. https://doi.org/10.3390/nano10020305
APA StyleJalili, P., Huet, S., Lanceleur, R., Jarry, G., Hegarat, L. L., Nesslany, F., Hogeveen, K., & Fessard, V. (2020). Genotoxicity of Aluminum and Aluminum Oxide Nanomaterials in Rats Following Oral Exposure. Nanomaterials, 10(2), 305. https://doi.org/10.3390/nano10020305





