Portable Real-Time Detection of Pb(II) Using a CMOS MEMS-Based Nanomechanical Sensing Array Modified with PEDOT:PSS
Abstract
1. Introduction
2. Materials and Methods
2.1. Design and Fabrication of Microcantilever Sensing Array Device
2.2. Experimental Chemicals and Instruments
2.3. Sensor Surface Modification
3. Results and Discussion
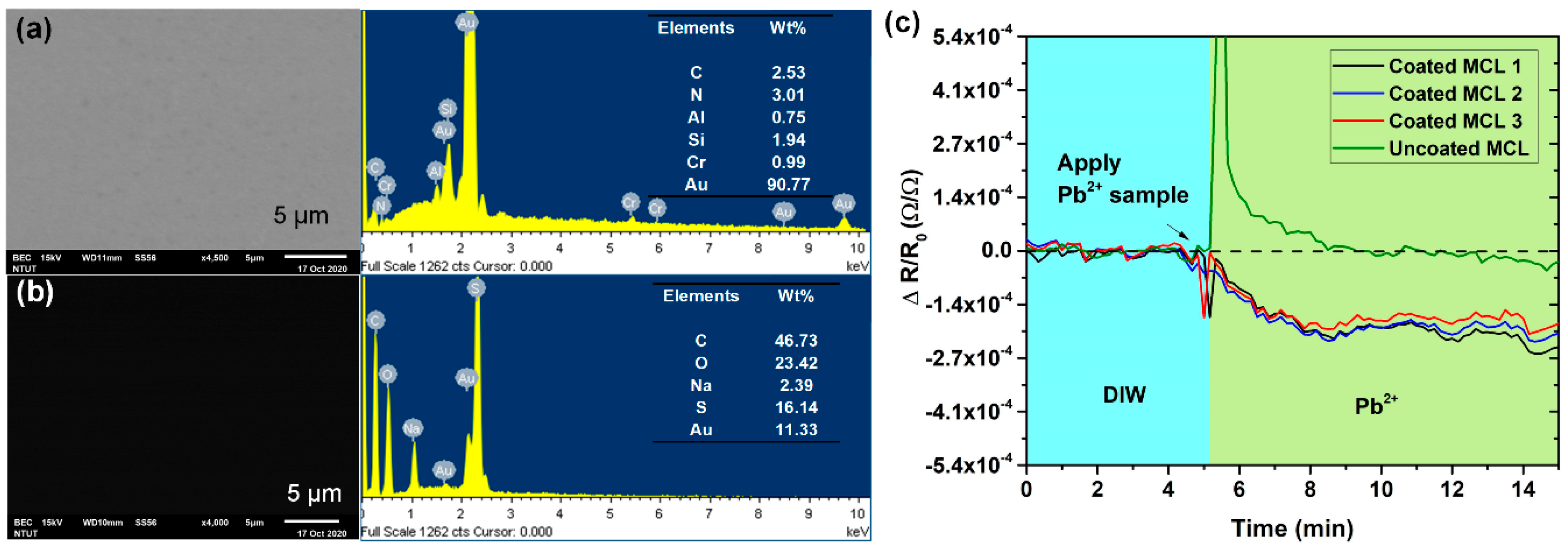
3.1. Sensor Characterizations
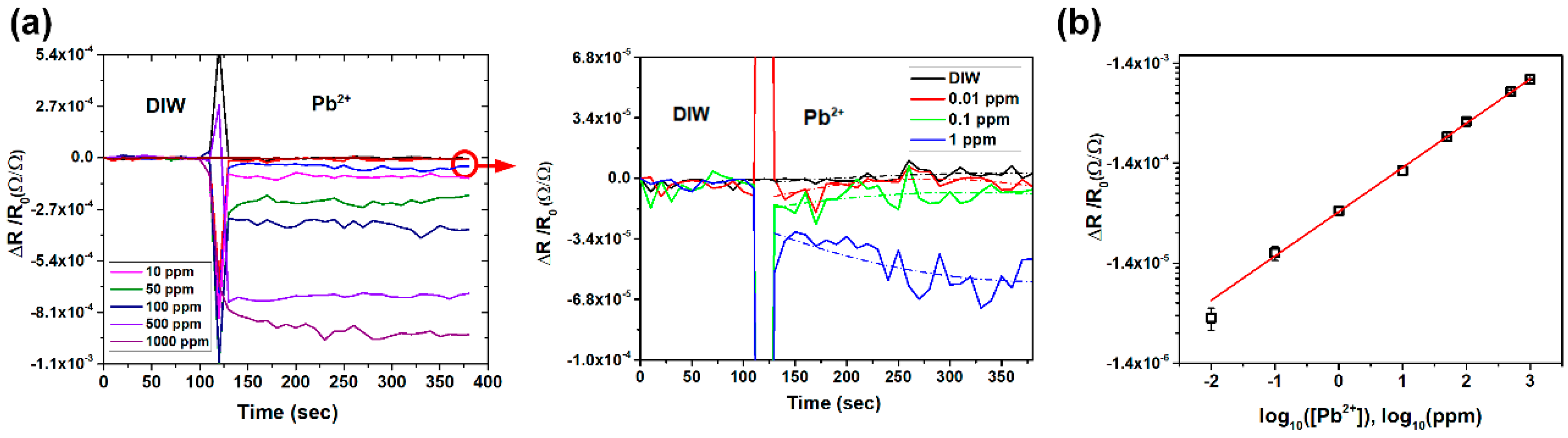
3.2. Detection of Lead Ions
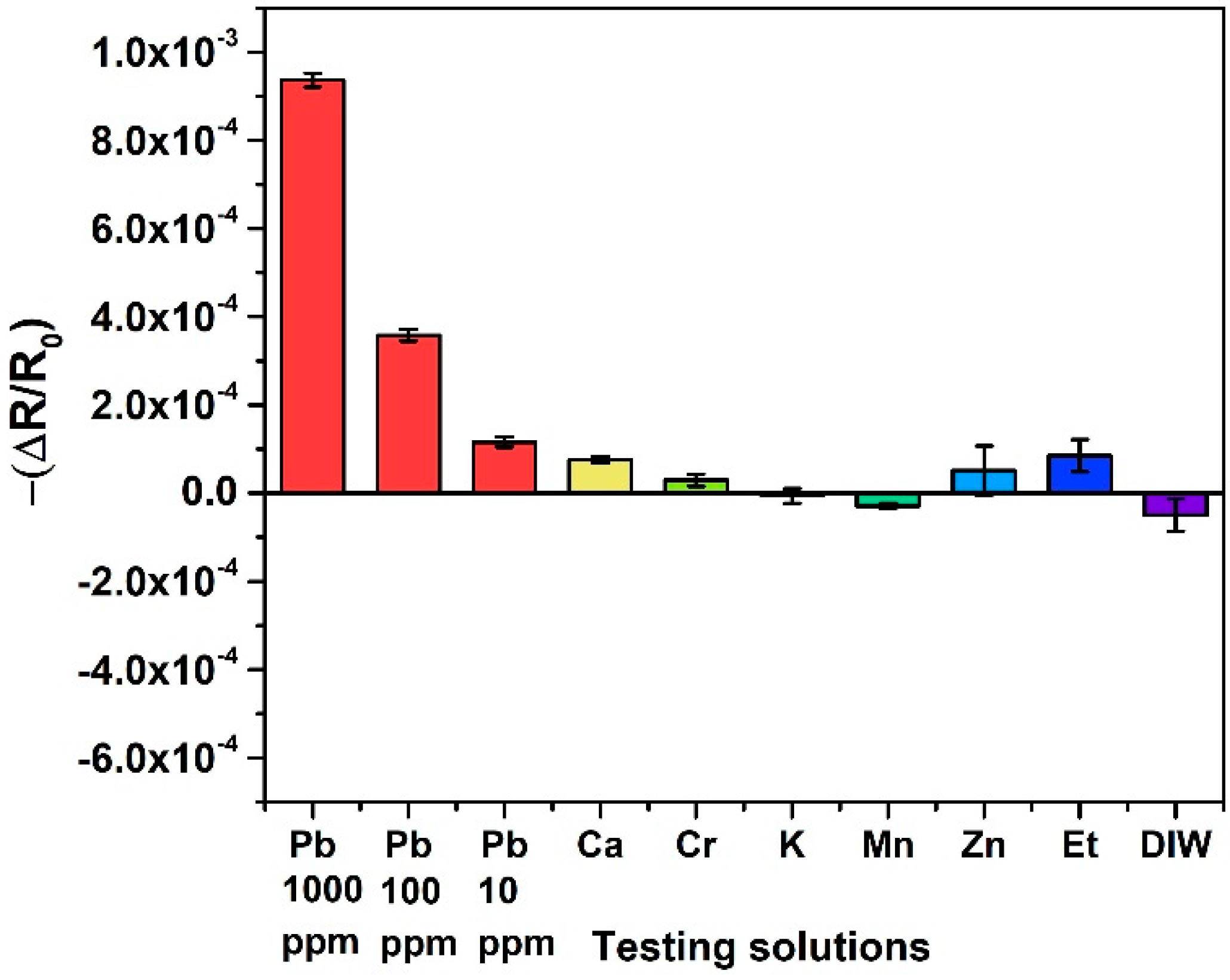
3.3. Selectivity
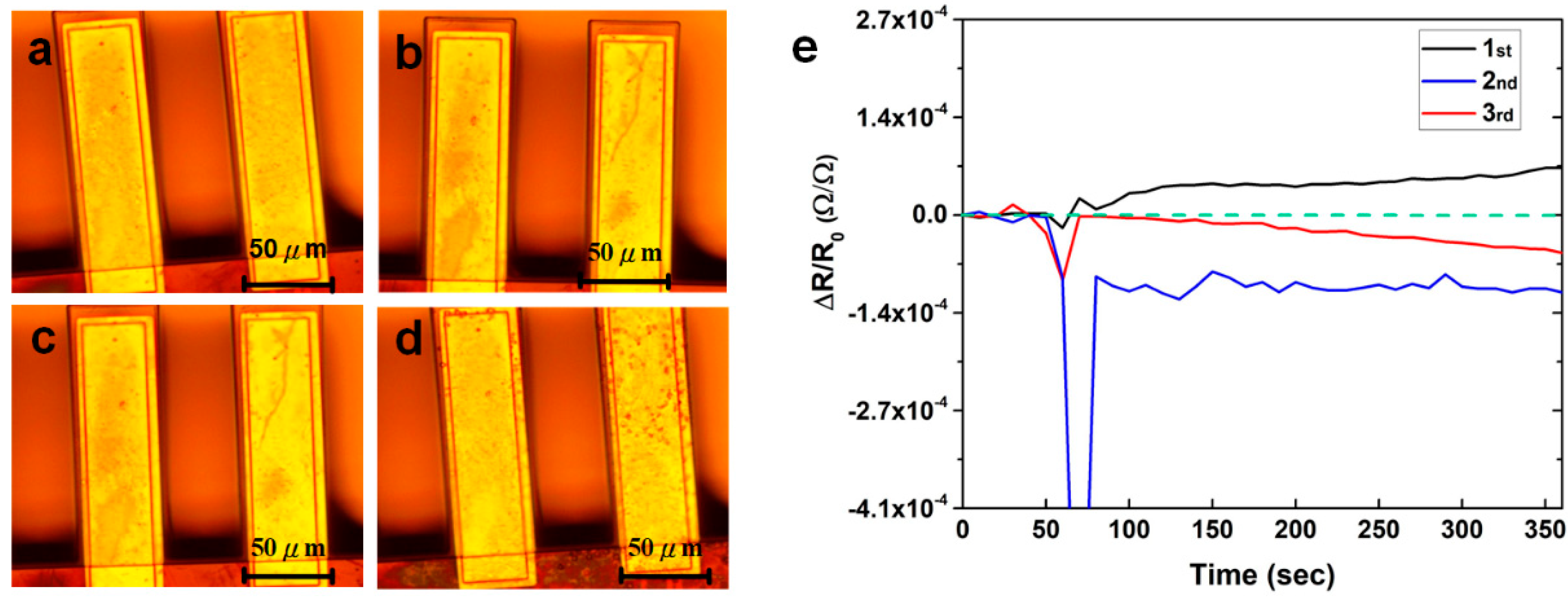
3.4. Reusability of the Sensor
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fernandez-Luqueno, F.; López-Valdez, F.; Gamero-Melo, P.; Luna-Suárez, S.; Aguilera-González, E.N.; Martínez, A.I.; García-Guillermo, M.D.S.; Hernández-Martínez, G.; Herrera-Mendoza, R.; Álvarez-Garza, M.A. Heavy metal pollution in drinking water-a global risk for human health: A review. Afr. J. Environ. Sci. Technol. 2013, 7, 567–584. [Google Scholar]
- Gumpu, M.B.; Sethuraman, S.; Krishnan, U.M.; Rayappan, J.B.B. A review on detection of heavy metal ions in water–an electrochemical approach. Sens. Actuators B Chem. 2015, 213, 515–533. [Google Scholar] [CrossRef]
- Raymond, J. Blood lead levels in children aged <5 years—United States, 2007–2013. MMWR. Morb. Mortal. Wkly. Rep. 2016, 63, 66–72. [Google Scholar] [PubMed]
- Sanders, T.; Liu, Y.; Buchner, V.; Tchounwou, P.B. Neurotoxic effects and biomarkers of lead exposure: A review. Rev. Environ. Health 2009, 24, 15–46. [Google Scholar] [CrossRef] [PubMed]
- Patrick, L. Lead toxicity part II: The role of free radical damage and the use of antioxidants in the pathology and treatment of lead toxicity. Altern. Med. Rev. 2006, 11, 11. [Google Scholar]
- Mills, W.B.; Porcella, D.B.; Ungs, M.J.; Gherini, S.A.; Summers, K.V. Water-Quality Assessment: A Screening Procedure for Toxic and Conventional Pollutants in Surface and Ground Water. Part 2, (Revised 1985); JACA Corp.: Fort Washington, MD, USA, 1985. [Google Scholar]
- Porento, M.; Sutinen, V.; Julku, T.; Oikari, R. Detection of copper in water using on-line plasma-excited atomic absorption spectroscopy (AAS). Appl. Spectrosc. 2011, 65, 678–683. [Google Scholar] [CrossRef]
- Karimi, M.; Sereshti, H.; Khojeh, V.; Samadi, S. Ligandless-ultrasound-assisted emulsification microextraction followed by inductively coupled plasma-optical emission spectrometry for simultaneous determination of heavy metals in water samples. Int. J. Environ. Anal. Chem. 2013, 93, 401–415. [Google Scholar] [CrossRef]
- Trujillo, I.S.; Alonso, E.V.; de Torres, A.G.; Pavón, J.M.C. Development of a solid phase extraction method for the multielement determination of trace metals in natural waters including sea-water by FI-ICP-MS. Microchem. J. 2012, 101, 87–94. [Google Scholar] [CrossRef]
- Zhou, Q.; Xing, A.; Zhao, K. Simultaneous determination of nickel, cobalt and mercury ions in water samples by solid phase extraction using multiwalled carbon nanotubes as adsorbent after chelating with sodium diethyldithiocarbamate prior to high performance liquid chromatography. J. Chromatogr. A 2014, 1360, 76–81. [Google Scholar] [CrossRef]
- Bansod, B.; Kumar, T.; Thakur, R.; Rana, S.; Singh, I. A review on various electrochemical techniques for heavy metal ions detection with different sensing platforms. Biosens. Bioelectron. 2017, 94, 443–455. [Google Scholar] [CrossRef]
- Huang, H.; Chen, T.; Liu, X.; Ma, H. Ultrasensitive and simultaneous detection of heavy metal ions based on three-dimensional graphene-carbon nanotubes hybrid electrode materials. Anal. Chim. Acta 2014, 852, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Parra, E.J.; Blondeau, P.; Crespo, G.A.; Rius, F.X. An effective nanostructured assembly for ion-selective electrodes. An ionophore covalently linked to carbon nanotubes for Pb 2+ determination. Chem. Commun. 2011, 47, 2438–2440. [Google Scholar] [CrossRef] [PubMed]
- Ruecha, N.; Rodthongkum, N.; Cate, D.M.; Volckens, J.; Chailapakul, O.; Henry, C.S. Sensitive electrochemical sensor using a graphene–polyaniline nanocomposite for simultaneous detection of Zn (II), Cd (II), and Pb (II). Anal. Chim. Acta 2015, 874, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Zhai, N.; Snyder, J.H.; Chen, Q.; Liu, P.; Jin, L.; Zheng, Q.; Lin, F.; Hu, J.; Zhou, H. Colorimetric detection of Hg 2+ and Pb 2+ based on peroxidase-like activity of graphene oxide–gold nanohybrids. Anal. Methods 2015, 7, 1951–1957. [Google Scholar] [CrossRef]
- Li, T.; Wang, E.; Dong, S. Lead (II)-induced allosteric G-quadruplex DNAzyme as a colorimetric and chemiluminescence sensor for highly sensitive and selective Pb2+ detection. Anal. Chem. 2010, 82, 1515–1520. [Google Scholar] [CrossRef] [PubMed]
- Verma, R.; Gupta, B.D. Detection of heavy metal ions in contaminated water by surface plasmon resonance based optical fibre sensor using conducting polymer and chitosan. Food Chem. 2015, 166, 568–575. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Zhou, X.; Guo, S.; Wu, N. Detection of lead (II) with a “turn-on” fluorescent biosensor based on energy transfer from CdSe/ZnS quantum dots to graphene oxide. Biosens. Bioelectron. 2013, 43, 69–74. [Google Scholar] [CrossRef]
- Shyuan, L.K.; Heng, L.Y.; Ahmad, M.; Aziz, S.A.; Ishak, Z. Evaluation of pesticide and heavy metal toxicity using immobilized enzyme alkaline phosphatase with an electrochemical biosensor. Asian J. Biochem. 2008, 3, 359–365. [Google Scholar] [CrossRef]
- Yin, Y.; Li, S.; Wang, S.; Jia, S.; Ren, J.; Farrell, G.; Lewis, E.; Wang, P. Ultra-high-resolution detection of Pb 2+ ions using a black phosphorus functionalized microfiber coil resonator. Photonics Res. 2019, 7, 622–629. [Google Scholar] [CrossRef]
- Megarajan, S.; Kanth, K.R.; Anbazhagan, V. Highly selective rapid colorimetric sensing of Pb2+ ion in water samples and paint based on metal induced aggregation of N-decanoyltromethamine capped gold nanoparticles. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2020, 239, 118485. [Google Scholar]
- Minas, G.; Ribeiro, J.C.; Wolffenbuttel, R.F.; Correia, J.H. On-chip integrated CMOS optical detection microsystem for spectrophotometric analyses in biological microfluidic systems. In Proceedings of the IEEE International Symposium on Industrial Electronics, Dubrovnik, Croatia, 20–23 June 2005; Volume 3, pp. 1133–1138. [Google Scholar]
- Boas, L.V.; Faustino, V.; Lima, R.; Miranda, J.M.; Minas, G.; Fernandes, C.S.V.; Catarino, S.O. Assessment of the deformability and velocity of healthy and artificially impaired red blood cells in narrow polydimethylsiloxane (PDMS) microchannels. Micromachines 2018, 9, 384. [Google Scholar] [CrossRef] [PubMed]
- Teh, H.B.; Li, H.; Li, S.F.Y. Highly sensitive and selective detection of Pb 2+ ions using a novel and simple DNAzyme-based quartz crystal microbalance with dissipation biosensor. Analyst 2014, 139, 5170–5175. [Google Scholar] [CrossRef] [PubMed]
- Zhou, G.; Chang, J.; Cui, S.; Pu, H.; Wen, Z.; Chen, J. Real-time, selective detection of Pb2+ in water using a reduced graphene oxide/gold nanoparticle field-effect transistor device. ACS Appl. Mater. Interfaces 2014, 6, 19235–19241. [Google Scholar] [CrossRef] [PubMed]
- Peng, R.-P.; Xing, L.-B.; Wang, X.-J.; Wu, C.; Chen, B.; Ji, H.-F.; Wu, L.-Z.; Tung, C.-H. Detection of Pb2+ in Aqueous Solution by Using a DNA-modified Microcantilever. Anal. Sci. 2016, 32, 1065–1069. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Ji, H.-F. Detection of Pb2+ using a hydrogel swelling microcantilever sensor. Anal. Sci. 2004, 20, 9–11. [Google Scholar] [CrossRef]
- Serrano-Santos, M.B.; Llobet, E.; Schäfer, T. Quartz crystal microbalance with dissipation measurement for proving the potential of ionic liquids as selective sensing Materials. Procedia Eng. 2011, 25, 1169–1172. [Google Scholar] [CrossRef]
- Ku, Y.-F.; Huang, L.-S.; Yen, Y.-K. A real-time thermal self-elimination method for static mode operated freestanding piezoresistive microcantilever-based biosensors. Biosensors 2018, 8, 18. [Google Scholar] [CrossRef]
- Láng, G.G.; Ujvári, M.; Bazsó, F.; Vesztergom, S.; Ujhelyi, F. In situ monitoring of the electrochemical degradation of polymer films on metals using the bending beam method and impedance spectroscopy. Electrochim. Acta 2012, 73, 59–69. [Google Scholar] [CrossRef]
- Xiong, C.; Qu, H.; Chen, W.; Zhang, L.; Qiu, L.; Zheng, L.; Xia, F. Real-time detection of Cu (II) with PEDOT: PSS based organic electrochemical transistors. Sci. China Chem. 2017, 60, 1205–1211. [Google Scholar] [CrossRef]
- Yasri, N.G.; Halabi, A.J.; Istamboulie, G.; Noguer, T. Chronoamperometric determination of lead ions using PEDOT:PSS modified carbon electrodes. Talanta 2011, 85, 2528–2533. [Google Scholar] [CrossRef]
- Tehrani, P.; Kanciurzewska, A.; Crispin, X.; Robinson, N.D.; Fahlman, M.; Berggren, M. The effect of pH on the electrochemical over-oxidation in PEDOT: PSS films. Solid State Ion. 2007, 177, 3521–3527. [Google Scholar] [CrossRef]
- Hu, Z. Thin Film Stress Studies Using Microcantilevers and Microcantilever Sensors. Ph.D. Thesis, The University of Tennessee, Knoxville, Tennessee, May 2000. [Google Scholar]
- Seel, S.C.; Thompson, C.V. Piezoresistive microcantilevers for in situ stress measurements during thin film deposition. Rev. Sci. Instrum. 2005, 76, 75103. [Google Scholar] [CrossRef]
- Jiang, R.; Liu, N.; Gao, S.; Mamat, X.; Su, Y.; Wagberg, T.; Li, Y.; Hu, X.; Hu, G. A facile electrochemical sensor based on PyTS–CNTs for simultaneous determination of cadmium and lead ions. Sensors 2018, 18, 1567. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yen, Y.-K.; Lai, C.-Y. Portable Real-Time Detection of Pb(II) Using a CMOS MEMS-Based Nanomechanical Sensing Array Modified with PEDOT:PSS. Nanomaterials 2020, 10, 2454. https://doi.org/10.3390/nano10122454
Yen Y-K, Lai C-Y. Portable Real-Time Detection of Pb(II) Using a CMOS MEMS-Based Nanomechanical Sensing Array Modified with PEDOT:PSS. Nanomaterials. 2020; 10(12):2454. https://doi.org/10.3390/nano10122454
Chicago/Turabian StyleYen, Yi-Kuang, and Chao-Yu Lai. 2020. "Portable Real-Time Detection of Pb(II) Using a CMOS MEMS-Based Nanomechanical Sensing Array Modified with PEDOT:PSS" Nanomaterials 10, no. 12: 2454. https://doi.org/10.3390/nano10122454
APA StyleYen, Y.-K., & Lai, C.-Y. (2020). Portable Real-Time Detection of Pb(II) Using a CMOS MEMS-Based Nanomechanical Sensing Array Modified with PEDOT:PSS. Nanomaterials, 10(12), 2454. https://doi.org/10.3390/nano10122454





